| Research Article | ||
Open Vet. J.. 2025; 15(10): 5069-5077 Open Veterinary Journal, (2025), Vol. 15(10): 5069-5077 Research Article Role of pollen beetles (Carpophilus aterrimus: Nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae)Marwan Keshlaf1*, Robert Spooner-Hart2, and Hamida Mirwan11Department of Plant Protection, Faculty of Agriculture, University of Tripoli, Tripoli, Libya 2Schools of Science and Health, University of Western Sydney, Sydney, Australia *Corresponding Author: Marwan M. Keshlaf, Department of Plant Protection, Faculty of Agriculture, University of Tripoli, Tripoli, Libya. Email: m.keshlaf [at] UOT.edu.ly Submitted: 23/04/2025 Revised: 18/08/2025 Accepted: 10/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: Cotton (Gossypium hirsutum) is an important global crop, and insect-mediated pollination significantly influences its productivity. Although honeybees (Apis mellifera) are widely recognized as effective cotton pollinators, other insect visitors, such as pollen beetles (Carpophilus aterrimus), frequently occur in cotton fields, but their role in pollination remains poorly understood. Aim: This study investigated the role of C. aterrimus in cotton pollination and examined how its presence affects honeybee foraging behavior. Understanding these interactions under Australian field conditions would provide valuable insights for optimizing integrated pest and pollinator management strategies in cotton agroecosystems. Methods: Experiments were conducted in Narrabri, Australia, using a combination of caging and bagging trials. Periodic observations were carried out throughout the cotton flowering period to assess the impact of beetle presence on honeybee visitation. Results: Carpophilus aterrimus did not enhance cotton yield, and high beetle densities negatively affected boll set and deterred honeybee visitation. Conclusion: Carpophilus aterrimus is not an effective pollinator and may reduce cotton productivity. Keywords: Carpophilus aterrimus, Apis mellifera, pollination efficiency, productivity, honeybee-beetle interaction. IntroductionCotton (Gossypium hirsutum L.) is a partially cross-pollinated crop, with insects serving as primary pollen transfer agents (McGregor, 1976). Numerous studies from various countries have documented a wide array of insect visitors to cotton, including pest species, pollinators, and biological control agents (Moffett et al., 1981; Tanda, 1983; El-Sarrag et al., 1993). Hymenoptera are typically dominant among beneficial insects, whereas Lepidoptera and Coleoptera are less commonly observed. Several studies have shown that honeybees (Apis mellifera) can enhance both the quantity and quality of cotton through cross-pollination, leading to improved yields (McGregor, 1976; Free, 1993). For instance, studies conducted in Australia (Rhodes, 2002; Keshlaf, 2009) found that cotton fields with high honeybee visitation produced heavier, more productive bolls with increased seed and lint content. Beetles are among the earliest known pollinators in the evolutionary history of flowering plants and play a crucial role in the pollination of many crops and wild plants (Crowson, 1981; George et al., 1989; Nadel and Peña, 1994; García-Robledo et al., 2004; Blanche and Cunningham, 2005; Kono and Tobe, 2007; Gottsberger and G, 2016). Families such as Cetoniinae, Nitidulidae, and Cantharidae are notable contributors. Cetoniinae beetles are primary pollinators of crops such as date palm (Phoenix dactylifera) and large flowering plants such as Magnolia grandiflora (Henderson, 1986; Proctor et al., 1996). Nitidulidae beetles, including Carpophilus hemipterus, contribute to the pollination of fruit crops such as fig (Ficus carica) and apple (Malus domestica) (Jordano, 1987; Compton et al., 1994). Cantharidae beetles are also important pollinators of wild plants such as Lilium species and various medicinal plants (Faegri and van der Pijl, 1979). In cotton fields, Collops vittatus (Say) (Melyridae) and Conotelus mexicanus Murray (Nitidulidae) have been reported in cotton flowers in Arizona, suggesting a potential role in pollination (Moffett et al., 1983). Interestingly, while the pollen beetle Meligethes aeneus (F.) (Nitidulidae) is known to damage oilseed rape flowers, it has also been shown to enhance pollination in the absence of bumblebees (Åhman et al., 2009). However, not all beetles contribute positively; their impact varies depending on their behavior and interactions with the crop. Other floral visitors, including pest species such as beetles and aphids, can influence the foraging behavior of honeybees. For example, honeybee foragers avoid flowers occupied by M. aeneus (Kirk et al., 1995), and aphid infestations in G. hirsutum have been shown to reduce honeybee visitation and pollination, ultimately affecting seed set (Levan and Holway, 2015). These interactions can decrease pollinator activity and crop yield, often without obvious damage. Such findings highlight the dual roles that floral visitors may play as both pollinators and floral resource competitors. In Australia, the pollen beetle Carpophilus aterrimus Macleay (Nitidulidae) is frequently observed in cotton flowers (Llewellyn and Fitt, 1996; Rhodes, 2002; Llewellyn et al., 2007). However, its role in cotton pollination remains unclear. Understanding the interactions between pest and pollinator species is essential, as some beetles may act as secondary pollinators, whereas others may disrupt pollination dynamics and reduce productivity by deterring primary pollinators or damaging floral structures (Gagic et al., 2019). This study aimed to investigate the role of the pollen beetle C. aterrimus in cotton pollination and assess its interaction with the foraging behavior of honeybees. Specifically, we examined whether the presence of C. aterrimus enhances the pollination outcomes of cotton plants under field conditions. We also evaluated whether the presence of pollen beetles on flowers influences honeybee visitation rates. Based on these aims, we hypothesized that (1) C. aterrimus contributes positively to cotton pollination by increasing seed and lint production and (2) pollen beetle presence on flowers may alter honeybee foraging patterns by either deterrence or attraction. This study seeks to clarify the dual role of C. aterrimus as both a potential pollinator and pest by addressing these questions, contributing to a more nuanced understanding of its ecological impact on cotton production. Materials and MethodsTwo experiments were conducted in a commercially irrigated transgenic cotton field (Bollgard ІІ® Sicot 71BR) at the ACRI, Narrabri, New South Wales (30.30˚S, 149.8˚E), during the flowering period from early January to mid-February 2007. The 18-hectare field was divided into two plots: Area 1 (16 ha) and Area 2 (2 ha), with Area 2 sown one month later than Area 1. Abundance of pollen beetlesThe abundance of C. aterrimus pollen beetles was assessed weekly by randomly collecting more than 100 cotton flowers from the field between 12:30 and 13:00. The flowers were placed into sealable plastic bags, and the mean number of beetles per flower was calculated. Observations of pollen beetles, including their diurnal activity, were recorded. Efficacy of pollen beetle pollinationPollinator efficacy was evaluated based on the following four parameters: boll set, boll weight, seeds per boll, and lint quality. Two methods were used: caging experiments and flower bagging trials. Caging experiments allowed pollen beetles to visit multiple flowers, facilitating effective pollen transfer. However, the prolonged use of cages was observed to potentially impact plant growth due to restricted environmental conditions. On the other hand, flower-bagging trials did not interfere with plant development while limiting pollinator access. These methods provided complementary insights into the pollination dynamics influenced by C. aterrimus. Caging experimentsThe caging trial was conducted in Area 2 at peak flowering (February 1−7, 2007). Thirty strong honeybee colonies (Langstroth 8-frame, double chamber) were placed near Area 2 to allow natural pollination. The middle row of the plot was divided into 15 plots, which were arranged in a five-block completely randomized block design. Each block contained one replicate of the following treatments:
Each treatment plot consisted of a single 3-m-long cotton row (approximately 30 plants), separated by 10 m. Flowers and bolls were removed from plants a day before caging to eliminate the effects of prior pollination. The cages were tent-shaped (1.5 m high × 3 m long) and covered with 2 mm white cotton mesh. For treatment 2, pollen beetles were collected from nearby cotton flowers in the early morning and released into the cages. All cages were dismantled 7 days after they were set up to minimize any cage effects, and pollinated flowers were tagged to facilitate monitoring and subsequent data collection. Potential cage effects were assessed by measuring light intensity (photosynthetic photon flux density) using a light meter (COR LI-250, LI-COR BioSciences, Lincoln, NE, USA) at the top of the crop canopy at 11:00 AM (EADST) on four separate days during the investigation period. Mature bolls were harvested on 13th April and processed for yield and quality analysis. Standard lint quality measurements included fiber length, uniformity, strength, and micronaire, following USDA guidelines (Addis et al., 2024). The bagged flowers trialFour treatments were applied to individual flowers in the bagging experiments (Fig. 1):
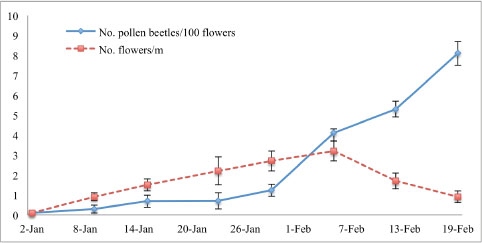
Fig. 1. Experimental setup for assessing the pollination efficacy of pollen beetles (Carpophilus aterrimus) on cotton flowers under four treatments: (a) bagged flowers without insects (control), (b) bagged flowers with 3 pollen beetles, (c) bagged flowers with 10 pollen beetles, and (d) unbagged flowers exposed to natural pollinators, including pollen beetles and honeybees. Eighty randomly selected flowers were tagged, and 60 were bagged before opening (07:00−08:00). Each treatment received 20 flowers, and the procedure was repeated on five dates: January 29 and 31, and February 5, 7, and 9, 2007. Custom-made organza bags (15×10 cm, mesh size ~0.5 mm) were used to exclude external pollinators, ensure airflow, and prevent condensation. The bags were secured with a drawstring around the peduncle, removed after one day, and mature bolls from tagged flowers were harvested on April 13 for yield and quality assessment, following cage experiment protocols. Effects of pollen beetles on honeybee visits to cotton flowersTo investigate whether pollen beetles deterred honeybee visits, the proximity of a nearby apiary was used to ensure a steady population of honeybees for observation and data collection. Observations were conducted in two rows, 60 and 80 m from the apiary, each consisting of 15 subplots (10 m long, 5 m apart). Flower visitation by honeybees was monitored during the flowering season at 12:00 and 14:00 EADT on seven dates: 29 and 31 January, and 5, 7, 9, 12, and 14 February. Each flower visited by a honeybee was inspected for the presence of pollen beetles, and the proportion of beetle-occupied flowers visited by honeybees was calculated. The number of freshly opened flowers was counted weekly across the 30 plots to track flowering dynamics and relate them to bee visitation rates. Statistical analysesData were analyzed using analysis of variance through the general linear model procedure in SPSS® for Windows™ Version 14. The normality of the variables was evaluated visually with P-P plots, while Levene’s test was used to assess the equality of variances. When significant differences were detected, treatment means were compared using Ryan’s Q test if variances were equal or Dunnett’s T3 test if variances were unequal. Statistical significance was set at p < 0.05. Pearson correlation analysis was performed to examine the relationship between pollen beetle density and the number of cotton flowers visited by bees. This analysis was conducted separately for two conditions: (1) in the presence and (2) in the absence of pollen beetles. This study aimed to assess whether increasing beetle abundance over time was associated with a decline in floral visitation rates of honeybees. ResultsAbundance of pollen beetlesThe presence of pollen beetles in the cotton field was first recorded during the second week of January, followed by a significant population increase (P=0.033). In Area 1, the number of beetles per 100 flowers increased weekly from 0 on January 2 to 4.11 by February 5. In Area 2, which was sown one month later to extend the flowering period for studying beetle biology, beetle numbers rose from 5.6 on January 15 to 8.1 by February 19 (Fig. 2).
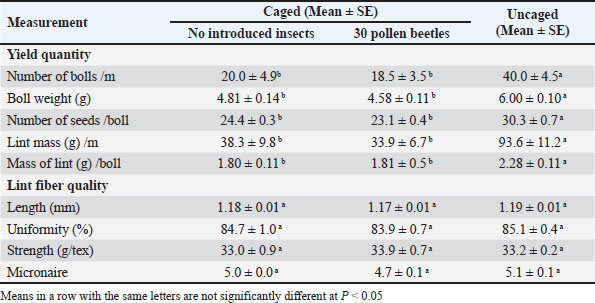
Fig. 2. Flowering phenology and field abundance of pollen beetles (Carpophilus aterrimus): The dotted line indicates the average number of open cotton flowers per meter of row from January 29, 2007 to February 14, 2007, at ACRI, Narrabri. Although flower density declined sharply after peaking on February 1, the number of pollen beetles per 100 flowers continued to increase. This trend may result from beetle concentration on the fewer remaining flowers as food sources became limited in the field. Efficacy of pollen beetle pollinationCaging experimentsOne-way ANOVA revealed significant differences among the three pollination treatments for all cotton yield parameters. The number of bolls per meter differed significantly among treatments (F2,12=8.186, P=0.012), with the highest boll count observed in the uncaged treatment. Similarly, boll weight (F2,12=42.379, P < 0.001), number of seeds per boll (F2,12=43.147, P < 0.001), lint mass per meter (F2,12=12.816, P=0.003), and lint mass per boll (F2,12=22.351, P < 0.001) were all significantly higher in the uncaged treatment than in the caged treatment (Table 1). No significant differences between caged plots with and without pollen beetles were observed in post-hoc tests. Table 1. Effects of the presence of pollen beetles (Carpophilus aterrimus) on cotton yield components and lint fiber quality in transgenic cotton (Bollgard II® Sicot 71BR) under three pollination treatments: caged without insects, caged with 30 pollen beetles, and uncaged plants exposed to natural pollinators.
In contrast, lint fiber quality traits did not show significant differences among treatments: fiber length (F2,12=0.51, P=0.612), uniformity (F2, 12=0.84, P=0.456), fiber strength (F2, 12=0.31, P=0.740), and micronaire (F2, 12=2.17, P=0.157). These results indicate that exposure to natural pollinators significantly enhanced cotton yield, whereas the presence of pollen beetles alone did not significantly improve yield. Moreover, none of the treatments had a significant effect on lint fiber quality. The study used cages for a short duration (7 days) to minimize potential effects. Light intensity inside cages (1650–1750 μmol/m2/s) was reduced to 81.1%–83.7% compared to uncaged conditions (2035–2100 μmol/m2/s), yet remained well above levels reported to reduce cotton yields (Moffett et al., 1972; Zhao and Oosterhuis, 1988). These cages, which are routinely used by breeders at the Australian Cotton Research Institute, showed no adverse effects on plant physiology or yield. The bagged flower trialThe bagged flower trial data could not be statistically analyzed because bolls from each treatment were mistakenly combined into a single bag, thereby eliminating the possibility of replicates. Nonetheless, a trend of decreasing yield metrics was observed as the number of pollen beetles in flowers increased (Table 2). Flowers with 10 beetles produced no bolls, demonstrating detrimental effects at high beetle densities. Table 2. Effects of pollen beetles (Carpophilus aterrimus) on cotton yield components and lint fiber quality in transgenic cotton (Bollgard II® Sicot 71BR) under four pollination treatments: bagged flowers without insects, bagged flowers with 3 pollen beetles, bagged flowers with 10 pollen beetles, and uncaged flowers exposed to natural pollinators.
Effects of pollen beetle on honeybee visit to cotton flowersA total of 9,864 cotton flowers were observed during the 15-day peak flowering period. Among the 338 flowers visited by the honeybee foragers, 191 (56.5%) were free of adult pollen beetles, whereas 147 flowers were infested, with beetle densities ranging from one to over 20 individuals per flower. Early in the observation period, honeybees strongly avoided beetle-infested flowers; however, over time, the frequency of visits to these flowers gradually increased (Fig. 3).
Fig. 3. Effect of the presence of pollen beetles (Carpophilus aterrimus) in cotton flowers on the number of floral visits by honeybees (A. mellifera) during the peak flowering period (January 29, 2007; to February 14, 2007) at ACRI, Narrabri, Australia. The number and proportion of bee-visited flowers that were occupied by beetles on successive sampling dates were as follows: 1 (2.7%) on 29 January (F1,6=15.756, P=0.007), 2 (2.9%) on 31 January (F1,6=49.00, P < 0.001), 79 (44.3%) on 5 February (F1,6=0.358, P=0.572), 46 (43.4%) on 7 February (F1,6=0.771, P=0.414), 53 (77.4%) on 9 February (F1,6=42.763, P=0.001), 37 (83.8%) on 12 February (F1,6=3.888, P=0.096), and 18 (100%) on February 14 (F1,6=7.385, P=0.035). The correlation between pollen beetle density (number per 100 flowers) and the number of bee-visited flowers was weakly negative when beetles were present (r=–0.55, P=0.628) and strongly positive when beetles were absent (r=0.92, P=0.252). However, neither correlation was statistically significant, likely because of the limited sample size. Bees strongly avoided flowers with pollen beetles when possible (Fig. 3). Both nectar foragers (collecting from extrafloral nectaries) and pollen-gatherers (e.g., hovering near infested flowers) exhibited this avoidance behavior. However, as beetle density increased throughout the study, bees were increasingly forced to visit infested flowers, resulting in shorter handling times. Initial avoidance behaviors diminished as uninfested flowers became scarce. By the end of the study (14/2/07), all flowers were beetle-infested, eliminating the bees’ avoidance option. DiscussionPollen beetles, identified as Carpophilus aterrimus, measured approximately 4–5 mm in length. These beetles remained predominantly inside cotton flowers during the day, feeding on pollen by climbing anthers. They often stayed within the wilted flower overnight and foraged for new flowers the following day. Occasionally, they attempted to enter the flowers before they had fully opened. The beetles were occasionally observed mating; however, no egg-laying behavior was observed during the study period. They were never seen on leaves or extrafloral nectaries, indicating that their activity was restricted to cotton flowers as shown in Figure 4.
Fig. 4. Foraging behavior and floral interaction between honeybees and pollen beetles (Carpophilus aterrimus): A honeybee forager cautiously lands on a cotton petal, avoiding contact with C. aterrimus individuals (left). A cotton flower heavily infested with more than 20 pollen beetles clustered around the anthers and near the floral nectaries (right). The field ecology of C. aterrimus and its potential harmful effects on cotton crops remain largely unexplored. Similarly, limited research has been conducted on its flight behavior, especially regarding the environmental and biological factors influencing its migratory movements across different landscapes. The results demonstrated a significant increase in C. aterrimus populations, particularly in late-sown crops, during the flowering period, reaching a peak density of 8.1 beetles per 100 flowers. This aligns with the findings of Llewellyn et al. (2007), who observed similar beetle accumulations during flowering in a nearby region. They reported that these beetles migrate late in the season from surrounding grasslands, where cotton becomes one of the few available floral resources. The observed aggregation behavior of the beetles may be influenced by pheromones or kairomones, as similar patterns have been reported in Nitidulidae species (Lin et al., 1992; Bartelt et al., 1994), indicating that chemical signals likely drive the clustering behavior of C. aterrimus. To shed light on these aspects, researchers have proposed employing a combination of established methodologies, such as suction traps, vertical-looking radar, and high-altitude aerial netting (Mauchline et al., 2017). These techniques can help demonstrate that C. aterrimus exhibits flight activity at varying altitudes throughout different stages of its active season, potentially revealing critical insights into its ecological dynamics and informing pest management strategies. Both the caging and bagging experiments showed that C. aterrimus did not contribute to cotton pollination. Flowers accessible to honeybees and other insects had significantly higher boll set and quality than those of other insects. This result is consistent with Steiner (1998), who noted that smooth-bodied beetles, such as C. aterrimus, are less effective in transferring pollen than hairy-bodied insects, such as honeybees. However, pollinators’ effectiveness depends not only on body surface structure but also on between-flower movement, which is a crucial factor in pollination success. Although studies, such as Åhman et al. (2009), have reported pollination by beetles in the absence of other pollinators, this study found no evidence supporting such a role for C. aterrimus. The absence of specialized adaptations, such as structures for pollen gathering or hairy bodies, which are typical of true pollinators, further reinforces the conclusion that C. aterrimus cannot function as a true pollinator. Flowers exposed to high densities of beetles (10 beetles per flower) failed to produce any bolls, indicating that C. aterrimus severely damaged the stamens and stigmas of the flowers. Previous studies, such as that by Young (1988), show similar floral damage by beetles, interfering with pollination processes. Similarly, Sakai et al. (1999) found that certain beetles contribute to pollination at low densities but become harmful at higher populations due to cumulative floral damage. However, in this study, C. aterrimus showed no evidence of being beneficial to cotton fruit yields at any density, instead acting as a consistent threat to boll set. Pollen beetles (Meligethes spp.) have a harmful impact on various agricultural crops. For example, Williams (2010) reported that M. aeneus causes significant damage to oilseed rape (Brassica napus), particularly during flowering, by feeding on pollen and damaging buds, leading to reduced seed production. Similarly, Ekbom and Borg (1996) showed that M. viridescens affects strawberry (Fragaria × ananassa) crops, resulting in a reduced fruit set. Duriez et al. (2009) reported that Meligethes spp. in sunflower (Helianthus annuus) hinder seed development by feeding on floral structures, which subsequently reduces overall yield. Honeybees displayed a preference for unoccupied flowers during the early flowering season, avoiding flowers with beetles. These findings indicate that honeybee foragers initially discriminate against beetle-infested flowers, due to interference or competition for floral resources. However, as pollen beetle densities increase over the flowering period and the availability of un-infested flowers declines, honeybees appear to relax their avoidance behavior, potentially indicating a shift in foraging thresholds or a tolerance to beetle presence. This behavior is similar to findings reported earlier by Kirk et al. (1995) and Murawski (1987) where bees could visually identify and avoid insect-occupied flowers. Honeybees also spent less time on beetle-infested flowers, likely due to reduced nectar availability. Such dynamic interplay, as highlighted by Gagic et al. (2019), reveals the intricate balance between pollinators and non-pollinators and their combined impact on crop productivity. This aligns with the findings of Gori (1983) who noted that nectar competition and beetle contamination can reduce effective pollination. These findings demonstrate that C. aterrimus not only fails to contribute to pollination but also interferes with the activity of effective pollinators such as honeybees, particularly during the crucial early flowering period. The results align with most prior research, confirming that C. aterrimus does not play a positive role in cotton pollination. Instead, it may harm the crop by physically damaging flowers and deterring honeybee visits. Although some studies, such as Åhman et al. (2009), suggest that beetles can act as secondary pollinators, the interaction between C. aterrimus and cotton flowers appears to be fundamentally different, likely due to variations in the biology and behavior of the species. Given the lack of any beneficial role in pollination, the hypothesis that C. aterrimus serves as a pollinator is rejected. Instead, the beetle should be viewed primarily as a pest species that causes physical damage to flowers and disrupts the activity of honeybees, further reinforcing its role as a pest rather than a pollinator. The results of this study were consistent with most prior research, confirming that C. aterrimus did not play a positive role in cotton pollination. Instead, it appeared to harm the crop by physically damaging the flowers and deterring honeybee visits. As noted by Gagic et al. (2019), understanding such interactions is essential for developing strategies that optimize pollination services while mitigating the impacts of pest species. The need for controlling C. aterrimus became particularly significant when cotton was planted late, as in this study, because beetle populations tended to be higher than those planted at standard times. Under regular planting schedules, beetle numbers were often lower, and intervention might not have been required. However, in late-season crops, integrated control methods, such as regular beetle monitoring, habitat management, use of beetle-resistant cultivars, or employing biological control agents — could be necessary to reduce beetle numbers, minimize competition with honeybees for floral resources, and enhance pollination efficiency. Future research should replicate this study across multiple locations and apply robust randomization procedures in the experimental design to enhance the applicability of these findings. Such efforts would strengthen the understanding of pollinator interactions and inform better agricultural practices. ConclusionTo the best of our knowledge, this study is the first to specifically demonstrate that the pollen beetle C. aterrimus does not significantly contribute to G. hirsutum pollination and may negatively affect yield parameters by reducing boll set and deterring honeybee visitation. Unlike A. mellifera, which enhanced cotton productivity, C. aterrimus showed no measurable pollination efficacy benefit, especially at higher densities. These findings contributed new insights to the existing body of knowledge by providing the first detailed evaluation of C. aterrimus in cotton pollination systems under field conditions in Australia. The results highlighted the importance of distinguishing between effective and non-effective insect visitors in crop ecosystems and stressed the potential of certain beetles to act more as floral antagonists than as beneficial pollinators. Thus, this study informs future integrated pest and pollinator management strategies in cotton agroecosystems. AcknowledgmentsThe authors would like to express their gratitude to the Australian Cotton Research Institute (ACRI), Narrabri, New South Wales, for hosting the experiment, providing technical assistance, and conducting the quality assessment. Special thanks are also extended to Dr. Robert Mensah for his valuable comments, which greatly enhanced the presentation of this article. Conflict of interestThe authors declare that there is no conflict of interest. FundingNo external funding for this study. Authors contributionsThe authors contributed to this study. All authors read and approved the final manuscript. Data availabilityAll data are provided in the manuscript. ReferencesAddis, T., Fentahun, B., Genetu, S. and Sisay, B. 2024. Cotton quality testing and characterization: trends and grading. In Cotton Sector Development in Ethiopia: challenges and Opportunities. Eds. Singapore, Ethiopia: Springer Nature Singapore, pp: 219–39. Åhman, I., Lehrman, A. and Ekbom, B. 2009. Impact of herbivory and pollination on performance and competitive ability of oilseed rape transformed for pollen beetle resistance. Arthropod-Plant Interact. 3(3), 105–113. Bartelt, R.J., Vetter, R.S., Carlson, D.G. and Baker, T.C. 1994. Responses to aggregation pheromones for five Carpophilus species (Coleoptera: nitidulidae) in a California date garden. Environ. Entomol. 23, 1534–1543. Blanche, R. and Cunningham, S. 2005. Rain forest provides beetles for Atemoya crops. J. Econ. Entomol. 98, 1193–1201. Compton, S.G., Ellwood, M.D.F., Davis, A.J. and Welch, K. 1994. Pollination and fig wasp selection in fig trees. Oecologia 97, 567–573. Crowson, R.A. 1981. The biology of the Coleoptera. New York, NY: Academic Press. Duriez, P., Lecomte, P. and Nénon, J.P. 2009. Pollen Beetles (Meligethes spp.) and Sunflower Seed Production: Impact on yield and management practices. J. Oilseed Res. 22, 178–184. Ekbom, B. and Borg, A. 1996. Pollen beetles (Meligethes spp.) in strawberry crops: effects on fruit development and potential for control. Ann. Appl. Biol. 129, 189–197. El-Sarrag, M.S., Ahmed, H.M. and Siddig, M.A. 1993. Insect pollinators of certain crops in the Sudan and the effect of pollination on seed yield and quality. J. King Saud Univ. Agric. Sci. 5, 253–262. Faegri, K. and van der Pijl, L. 1979. The principles of pollination ecology. Pergamon Press. Free, J.B. 1993. Insect pollination of crops. 2nd ed., Academic Press, London, UK. Gagic, V., Marcora, A. and Howie, L. 2019. Additive and interactive effects of pollination and biological pest control on crop yield. J. Appl. Ecol. 56, 2528–2535. García-Robledo, C., Kattan, G., Murcia, C. and Quintero-Marín, P. 2004. Beetle pollination and fruit predation of Xanthosoma daguense (Araceae) in an Andean cloud forest in Colombia. J. Trop. Ecol. 20, 459–469. George, A.P., Nissen, R.J., Ironside, D.A. and Anderson, P. 1989. Effects of nitidulid beetles in natural pollination of fruit set of Annona spp. hybrids. J. Am. Soc. Hortic. Sci. 39, 289–299. Gori, D.F. 1983. Post-pollination phenomena and adaptive floral changes. In: Handbook of Experimental Pollination Biology, Ed., Little, R.J. New York: Van Nostrand Reinhold, pp: 31–45. Gottsberger. and G. 2016. Beetle pollination and flowering ecology in tropical ecosystems. Biotropica 48, 342–356. Henderson, A. 1986. Pollination biology of economically important palms. Adv. Econ. Bot. 2, 37–57.Jordano, P. 1987. Patterns of mutualistic interactions in pollination and seed dispersal: Connectance, dependence asymmetries, and coevolution. Am. Nat. 129, 657–677. Keshlaf, M.A. 2009. An assessment of honeybee foraging activity and pollination in Australian Bt cotton. Ph. D. thesis, Univ. Western Sydney, Sydney, Australia. Kirk, W.D.J., Ali, M. and Breadmore, K.N. 1995. The effects of pollen beetles on the foraging behaviour of honeybees. J. Apic. Res. 34, 15–22. Kono, M. and Tobe, H. 2007. Is Cycas revoluta (Cycadaceae) wind- or insect-pollinated?. Am. J. Bot. 94, 847–855. Levan, K.E. and Holway, D.A. 2015. Ant-aphid interactions increase ant floral visitation and reduce plant reproduction via decreased pollinator visitation. Ecology 96, 1620–1630. Lin, H., Phelan, P.L. and Bartelt, R.J. 1992. Synergism between synthetic food odors and the aggregation pheromone for attracting Carpophilus lugubris in the field (Coleoptera: nitidulidae). Environ. Entomol. 21, 156–159. Llewellyn, D. and Fitt, G. 1996. Pollen dispersal from two field trials of transgenic cotton in the Namoi Valley, Australia. Mol. Breed. 2, 157–166. Llewellyn, D., Tyson, C., Constable, G., Duggan, B., Beale, S. and Stell, P. 2007. Contamination of regulated genetically modified cotton in the field. Agric. Ecosyst. Environ. 121, 419–429. Mauchline, A.L., Cook, S.M., Powell, W., Chapman, J.W. and Osborne, J.L. 2017. Migratory flight behaviour of the pollen beetle Meligethes aeneus. Pest Manag. Sci. 73, 1076–1082. Mcgregor, E.S. 1976. Insect pollination of cultivated crop plants.In USDA Agricultural Research Service (ed), USDA Agricultural Handbook 496. Washington, DC: U.S. Department of Agriculture, pp: 171–89. Moffett, J.O., Jones, C.E. and Little, R.J. 1983. Hybrid cotton.In Handbook of Experimental Pollination Biology. Jones, C.E. and Little, R.J New York, NY: Van Nostrand Reinhold, pp: 508–14. Moffett, J.O., Lloyd, H.A. and Maki, T.E. 1972. Effect of shade on flowering and fruiting of cotton. Agron. J. 64, 644–645. Moffett, J.O., Waller, G.D. and Rummel, R.D. 1981. Study on insect pollinators and hybrid cotton production in the High Plains of Texas. In the Proceedings of the Beltwide Cotton Production Research Conference, Dallas, Texas. National Cotton Council of America, Memphis, TN, p: 88. Murawski, D.A. 1987. Floral resource variation, pollinator response and potential pollen flow in Psiguria warscewiczii. Ecol 68, 1273–1282. Nadel, H. and Peña, J.E. 1994. Identity, behaviour, and efficacy of nitidulid beetles (Coleoptera: nitidulidae) pollinating commercial Annona species in Florida. Environ. Entomol. 23, 878–886. Proctor, M., Yeo, P. and Lack, A. 1996. The natural history of pollination. Portland: Timber Press. Rhodes, J. 2002. Cotton pollination by honeybees. Aust. J. Exp. Agric. 42, 513–518. Sakai, S., Momose, K., Yumoto, T., Kato, M. and Inoue, T. 1999. Beetle pollination of Shorea parvifolia (section Mutica, Dipterocarpaceae) in a general flowering period in Sarawak, Malaysia. Am. J. Bot. 86, 62–69. Steiner, K.E. 1998. Beetle pollination of peacock moraeas (Iridaceae) in South Africa. Plant Syst. Evol. 209, 47–65. Tanda, A.S. 1983. Assessing the role of honeybees in a field of Asiatic cotton (Gossypium arboreum L.). Am. Bee J. 123, 593–594. Williams, I.H. 2010. The major insect pests of oilseed rape in Europe and their management: an overview.In Biocontrol-Based Integrated Management of Oilseed Rape Pests. Ed. and Williams, I.H Dordrecht, The Netherlands: Springer, pp: 1–43. Young, H.J. 1988. Differential importance of beetle species pollinating Dieffenbachia longispatha (Araceae). Ecol 69, 832–844. Zhao, D. and Oosterhuis, D.M. 1988. Physiological response of cotton to high temperatures and water stress. Field Crops Res. 56, 47–52. | ||
| How to Cite this Article |
| Pubmed Style Keshlaf MM, Spooner-hart R, Mirwan HB. Role of pollen beetles (Carpophilus aterrimus: Nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae). Open Vet. J.. 2025; 15(10): 5069-5077. doi:10.5455/OVJ.2025.v15.i10.25 Web Style Keshlaf MM, Spooner-hart R, Mirwan HB. Role of pollen beetles (Carpophilus aterrimus: Nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae). https://www.openveterinaryjournal.com/?mno=254000 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.25 AMA (American Medical Association) Style Keshlaf MM, Spooner-hart R, Mirwan HB. Role of pollen beetles (Carpophilus aterrimus: Nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae). Open Vet. J.. 2025; 15(10): 5069-5077. doi:10.5455/OVJ.2025.v15.i10.25 Vancouver/ICMJE Style Keshlaf MM, Spooner-hart R, Mirwan HB. Role of pollen beetles (Carpophilus aterrimus: Nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae). Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5069-5077. doi:10.5455/OVJ.2025.v15.i10.25 Harvard Style Keshlaf, M. M., Spooner-hart, . R. & Mirwan, . H. B. (2025) Role of pollen beetles (Carpophilus aterrimus: Nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae). Open Vet. J., 15 (10), 5069-5077. doi:10.5455/OVJ.2025.v15.i10.25 Turabian Style Keshlaf, Marwan M., Robert Spooner-hart, and Hamida B. Mirwan. 2025. Role of pollen beetles (Carpophilus aterrimus: Nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae). Open Veterinary Journal, 15 (10), 5069-5077. doi:10.5455/OVJ.2025.v15.i10.25 Chicago Style Keshlaf, Marwan M., Robert Spooner-hart, and Hamida B. Mirwan. "Role of pollen beetles (Carpophilus aterrimus: Nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae)." Open Veterinary Journal 15 (2025), 5069-5077. doi:10.5455/OVJ.2025.v15.i10.25 MLA (The Modern Language Association) Style Keshlaf, Marwan M., Robert Spooner-hart, and Hamida B. Mirwan. "Role of pollen beetles (Carpophilus aterrimus: Nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae)." Open Veterinary Journal 15.10 (2025), 5069-5077. Print. doi:10.5455/OVJ.2025.v15.i10.25 APA (American Psychological Association) Style Keshlaf, M. M., Spooner-hart, . R. & Mirwan, . H. B. (2025) Role of pollen beetles (Carpophilus aterrimus: Nitidulidae) in cotton pollination and their interaction with honeybees (Apis mellifera: Apidae). Open Veterinary Journal, 15 (10), 5069-5077. doi:10.5455/OVJ.2025.v15.i10.25 |