Open Veterinary Journal, (2026), Vol. 16(1): 427-436
Research Article
10.5455/OVJ.2026.v16.i1.39
Production, purification, and characterization of chicken immunoglobulin Y anti-Escherichia coli as a passive immunity candidate
Muhammad Farid Rizal1,2, I Wayan Teguh Wibawan3*, Huda S. Darusman3,4, Joko Pamungkas3,4,
Roostita L. Balia5, Denniswara Sibit6 and Hadrianus Lau2
1Doctorate Student of Primatology, Primatology program, School of Postgraduate IPB University, Bogor, Indonesia
2Urban Animal Clinic, Cibeunying Kolot, Cibeunying Kaler, Kota Bandung, Indonesia
3School of Veterinary Medicine and Biomedical Sciences, IPB University, Bogor, Indonesia
4Primate Research Center, IPB University, Bogor, Indonesia
5Veterinary Study Program, Faculty of Medicine, Universitas Padjajaran, Sumedang, Indonesia
6Departement Research and Development, PT. Biomol Nusantara Mandiri, Panyileukan, Bandung, Indonesia
*Corresponding Author: I. Wayan T. Wibawan. School of Veterinary Medicine and Biomedical Sciences, IPB University, Bogor, Indonesia. Email: wayanwi [at] apps.ipb.ac.id
Submitted: 02/05/2025 Revised: 30/11/2025 Accepted: 20/12/2025 Published: 31/01/2026
© 2025 Open Veterinary Journal
This is an Open Access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited, and is not altered, transformed, or built upon in any way.
Abstract
Background: Escherichia coli bacteria are common in the environment, and the immune systems of humans and animals are essential for fighting these pathogens.
Aim: This study aimed to produce, purify, and characterize chicken immunoglobulin Y (IgY) specific against E. coli as a passive immunity candidate.
Methods: A single E. coli was isolated from the enteric tract of fascicularis. In this study, two groups of 22-week-old hens, each consisting of three hens, with a body weight range of 2–3 kg, were vaccinated with E. coli inactivated either with 4% formaldehyde overnight (Group A) or at 90°C for 2 hours (Group B). In the first week (priming), each chicken group was injected intramuscularly with 0.5 ml of vaccine containing E. coli at a concentration of 109 Colony Forming Unit without Montanide ISA 70 as an adjuvant. In the second week (booster), each chicken group was injected with 0.5 ml of vaccine containing E. coli at the concentration 109 CFU mixed with Montanide ISA 70 at a volume ratio of 3:7. At the third week (final booster), each chicken group was injected with 0.5 ml of vaccine containing E. coli at the concentration 109 CFU mixed with Montanide ISA 70 in a volume ratio of 3:7. Serum and egg samples were collected before booster administration at week 1 and following booster administration at weeks 2, 3, and 6.
Results: Based on the agar gel precipitation test, the IgY titer on the chicken Group A step-by-step increase in antibody titer, with serum IgY titers of 22 and 32 at weeks 3 and 6, respectively egg yolk IgY titers of 21 and 22 at weeks 3 and 6, respectively. The IgY titer in group B had a high antibody titer, with serum IgY titers of 23 at both weeks 3 and 6, and egg yolk IgY titers of 21 and 23 at weeks 3 and 6, respectively. The results of the sodium dodecyl sulfate-polyacrylamide gel electrophoresis analysis of purified serum and egg yolk at weeks 3rd and 6th , revealed protein bands with molecular weights of approximately 65 and 25 kDa. The 65 kDa protein band is hypothesized to correspond to the heavy chain of IgY, whereas the 25 kDa band is presumed to represent the light chain of IgY. Immunoblotting of E. coli proteins with various preparations revealed approximately 29 kDa, which can be recognized by IgY, an adhesin protein.
Conclusion: This study demonstrates that inactivated E. coli vaccination can increase IgY titers in chickens, which is a potential candidate for passive immunity against E. coli.
Keywords: Chicken immunoglobulin Y, Escherichia coli, Formaldehyde inactivation, Heat inactivation.
Introduction
The rising prevalence of antibiotic-resistant infections is a significant global health problem; therefore, new strategies for mitigating and curing bacterial infections must be developed (Salam et al., 2023). One of the most promising passive immunity, giving pre-formed antibodies to provide specific pathogens (Marcotte and Hammarström, 2015). Currently, chicken immunoglobulin Y (IgY), a potential candidate for passive immunity due to its high specificity, safety, and ease of production (Abbas et al., 2019).
Passive immunity conferred by antibodies is inherently transient its non-self-renewing nature and susceptibility to degradation over time. Consequently, periodic boosting is necessary to maintain sufficient antibody titers and ensure prolonged protection (Marcotte and Hammarström, 2015). The efficacy of passive immunity is contingent upon the specific antibodies employed, with IgY exemplifying a highly specific yet potentially immunogenic molecule that may necessitate meticulous formulation and delivery strategies (Slifka and Amanna, 2018). The optimal administration route for IgY is tissue-dependent, with mucosal surfaces posing a significant challenge for effective delivery due to their role as primary entry points for numerous pathogens (Pereira et al., 2019). Oral delivery of IgY requires surmounting the hostile gastrointestinal environment to achieve therapeutic efficacy (Vela Ramirez et al., 2017; Mahalakshmi et al., 2024).
An immunoglobulin is a globulin, also called an antibody. The antibody is secreted by plasma cells to specifically respond to the antigen. IgY is a nickname for an antibody that comes from chicken. IgY is found on egg yolk, as well as a third protein characteristic difference compared with mammalian Ig (Amro et al., 2018). The emergence of antibiotic resistance constitutes a severe threat, as it can lead to the development of multidrugs resistant in specific bacterial strains. IgY has demonstrated efficacy in treating infections by reducing mortality rates in vivo studies, thereby conferring a beneficial effect in mitigating the transmission of resistance genes (Esmaeili et al., 2024). IgY is an antibody produced by poultry immunized with a certain antigen (Radwan et al., 2023). This kind of antibody is transferred by the parent to the offspring that was inherited through egg yolk (through the transfer of maternal antibodies through the egg yolk.), so that IgY that was cheap, easy, and can be produced on a massive scale can be achieved. IgY was reported to have effectivity in the inhibition and neutralization of pathogens and ruled as passive immunity, so that IgY can ruled as candidate therapy against Escherichia coli infection (El-Kafrawy et al., 2023).
Escherichia coli is a gram-negative bacteria that usually causes many health issues; however, E. coli is generally commensal to the host cell (Braz et al., 2020). Certain strains of E. coli are classified as intestinal or extraintestinal pathogens. Generally, the treatment of E. coli infection involves the use of suitable antibiotics; however, antibiotic effectiveness decreases with time, which correlates with the increasing bacterial resistance. Antibiotic resistance can lead to treatment failure with serious consequences, such as economic loss and even death (Anjum et al., 2021).
Chicken-derived IgY has been proposed as a promising treatment for E. coli infection without contributing to antibiotic resistance (Esmaeili et al., 2024). IgY as a candidate for passive immunity was an alternative method for prophylaxis and therapy of gastrointestinal infections caused by bacterial and viral infections such as Salmonella, E. coli, Campylobacter, Streptococcus mutans, Rotavirus, and coronavirus (Esmailnejad et al., 2019; Mahalakshmi et al., 2024). The utility of specific antibodies was aimed to decrease conventional treatment, such as the use of antivirals or antibiotics, which correlated with an increase in bacterial resistance (Hidayat et al., 2024). IgY confers protectivity through antimicrobial activity, binding, neutralization, growth inhibition, and pathogen replication and spreading (Setthawong et al., 2025). This study aimed to evaluate whether immunization with heat- or formaldehyde-inactivated E. coli can increase IgY titers in chickens.
Materials and Methods
Preparation of the E. coli antigen
In this research, diarrheagenic E. coli was isolated from an anal swab of Macaca fascicularis suffering from severe diarrhea in the monkey breeding, which is located on the Bogor Agricultural Institute Dramaga, Bandung, Indonesia, and was performed by the microbiology and immunology laboratory, Bogor Agricultural Institute. The researcher used E. coli from M. fascicularis because M. fascicularis demonstrates a high degree of genetic homology with humans, which facilitates the extrapolation of the research result from M. fascicularis to humans (Tan et al., 2018). Escherichia coli was inoculated on brain heart infusion agar and then incubated on temperature 37°C with duration 24 hours. The culture was harvested and washed three times using phosphate-buffered saline (PBS) (pH 7.4) and centrifuged at 2,000 rpm for 15 minutes. The resulting pellet was washed three times using sterile PBS (pH 7.4) (Low et al., 2013). The E. coli pellets that were washed were divided into two groups (Group A and Group B) according to how the E. coli pellet was inactivated.
In Group A, E. coli was inactivated using 4% formaldehyde overnight (Arshadi et al., 2020). The inactivation result was subjected to a sterility test on nutrient agar, incubated at 37°C with a duration 24-hour, and colony growth was observed on the media. The sterility was declared suitable when no colony grew (Wescot et al., 2023). The E. coli pellet that had been inactivated using formaldehyde 4% overnight was then added with PBS pH 7.4, measured by UV-Vis spectrophotometer Thermo Fisher Scientific® to determine turbidity and the mixing procedure was carried out with inactivated E. coli at the concentration 109 CFU mixed with Montanide ISA 70 (Seppic, France) in a volume ratio of 3:7 (Matlock and Wilmington, 2019).
In Group B, E. coli was inactivated at a temperature 90°C at 2 hour in a water bath (Biswas et al., 2019). The inactivation result was subjected to a sterility test on nutrient agar, incubated at 37°C with duration 24-hour, colony growth was observed on the media. The sterility was declared suitable when no colony grew. The E. coli pellet was inactivated at temperature 90°C for 2-hour then added with PBS (pH 7.4). The turbidity was measured using a UV-Vis spectrophotometer (Thermo Fisher Scientific®) and the mixing procedure was carried out with inactivated E. coli at the concentration 109 CFU mixed with Montanide ISA 70 (Seppic, France) in a volume ratio of 3:7 (Matlock and Wilmington, 2019).
Chicken vaccination
Six pathogen-free leghorn chickens, 22 weeks old, with a body weight range of 2–3 kg provided by Perseroan Terbatas. Biomol Nusantara Mandiri Tbk. The chickens were used divided into two treatment groups: Group A and Group B. In Group A, the chicken given vaccine contained an E. coli isolate inactivated by 4% formaldehyde, and in Group B, the chicken given vaccine contained an E. coli isolate inactivated at 90°C for 2 hours.
Group A (given an E. coli isolate inactivated by formaldehyde 4%), at the first week, was injected with 0.5 ml of the vaccine consisting of E. coli with a concentration of 109 CFU without adjuvant intramuscularly at the chicken breast that ruled as priming vaccine (Keita et al., 2022). The first booster administered at the second week was injected with 0.5 ml of the vaccine consisting of E. coli with concentration 109 CFU mixed with Montanide ISA 70 in a volume ratio of 3:7. The second booster (final booster) was injected with 0.5 ml of the vaccine consisting of E. coli with concentration 109 CFU mixed with Montanide ISA 70 in a volume ratio of 3:7 (Khabazzadeh et al., 2016).
Group B (given an E. coli isolate inactivated at 90°C for 2 hours) was injected with 0.5 ml vaccine consist of E. coli with concentration 109 CFU without adjuvant intramuscularly at the chicken breast with the major goal of priming vaccine (Biswas et al., 2019). The first booster was done at the second week, injected with 0.5 ml vaccine consisting of E. coli with a concentration 109 CFU mixed with Montanide ISA 70, in a volume ratio of 3:7 (Meng et al., 2024). The final booster was done at the third week, injected with 0.5 ml vaccine consist of E. coli at the concentration 109 CFU mixed with Montanide ISA 70, in a volume ratio of 3:7 (Khabazzadeh et al., 2016). Serum and egg samples were collected before booster administration at week 1 and following booster administration at weeks 2, 3, and 6.
Extraction and purification of IgY
The purification procedure was carried out using the titration water-soluble fraction method with polyethylene glycol (PEG) (Amro et al., 2018). Separate the egg yolk from the egg white with filter paper, tear the egg yolk membrane, and pour the egg yolk into a measuring cup (Volume 1), and add PBS at a concentration of 0.01 M (pH 7.2) as much as twice (V1) (Dhruvi et al., 2025). Mix and stir using a spatula, then pour to the centrifuge tube (V2), add PEG-6,000 as much as 3.5% (b/v) of (V2). Vortex until homogeneous, then roll using a rotator for 10 minutes at a velocity of 20/min. Centrifuge for 20 minutes at a temperature of 4°C and velocity 13,000 × g, filter the super natant using filter paper, and measure the volume. Then, 50 ml (V3) is poured into the falcon. Add PEG-6,000 as much as 8.5% (b/v) of (V3). Vortex until homogeneous, then roll using a rotator for 10 minutes at a velocity of 20/min. Centrifuge for 20 minutes at 4°C and velocity 13,000 × g, dispose the supernatant and resuspension the pellet using PBS 0.01M (pH 7.2) as much as 5 ml stir using a stirring rod, and vortex at the same time. Add 0.01 M PBS (pH 7.2) until the end volume reaches 10 ml (V4). Add PEG-6,000 to as much as 12% (b/v) of V4. Vortex until homogeneous, then roll using a rotator for 10 minutes at a velocity of 13.000 g, dispose the supernatant, and resuspension of the pellet using PBS 0.01M (pH 7.2) as much as 600 µl, and simultaneously stir with a stirring rod and vortex. After homogenization, add PBS 0.01M pH7.2 as much as 600 µl at the same time, vortexed, and stirred with a stirring rod (Siriya et al., 2013).
Storage conditions for soluble E. coli antigen
Escherichia coli was grown on brain heart infusion media (Thermo Fisher Scientific®) and incubated at 37°C for 18 hours (Tamai et al., 2025). The suspension was centrifuged at 20,000 rpm for 10 minutes. The pellet was washed, then 0.5 ml HCL 0.2 nN was added, and then centrifuged again at the same velocity for 10 minutes. To dispose the supernatant, 0.5 ml HCL 0.2 N was added to the pellet and heated at 52°C for 1 hour. The homogenat was titrated with 1 N NaOH using a phenol red indicator (the homogenat color was yellow and red). Then, the mixture was centrifuged at 10.000 rpm (PEC series) for 10 minutes. The supernatant was used as a soluble antigen (HCL extract) (Tuttle et al., 2021).
Specific antibody detection using the agar gel precipitation test
A 1% agarose solution was prepared by dissolving 0.4 g of agarose and 1.2 g of PEG 6,000 in a mixture of 20 ml of 0.5 M PBS (pH 7.4) and 20 ml of distilled water (pH 7.4). The solution was then heated using a hot plate stirrer until boiling and subsequently stirred until the agarose was fully dissolved and the solution became clear (Okwor et al., 2011). The liquid agar was poured into the surface of the object glass and waited until it became solid. A 1% agarose gel was punched with a gel puncher to create a central well surrounded by six peripheral wells in a circular pattern. As much as 50 μl of chicken serum and IgY that have been extracted, then dissolved with PBS. Dilution with multiple of 2 log 2 as much as five times dilution, 45 μl serum and IgY that have been dissolved pour into circle hole at the edge sequentially, started with lower dilution, antigen HCl extract as much as 50 μl pour into circle hole that located in the center, then incubated at 25°C in period less than 18 hours and observe the agar gel precipitation test (AGPT) result (Putri et al., 2018).
AGPT has one interpretation result, namely, positive or negative. A positive result in AGPT is characterized by the formation of visible, white lines of precipitation between the wells containing the antigen and the corresponding antibody (Chismirina et al., 2023). These lines form as a result of the antigen and antibody diffusion into the agar gel, where they interact to create insoluble complexes that precipitate out of solution. The validity of the test is confirmed when the lines of precipitation between the test sample and the reference antigen merge with the reference antigen-antiserum precipitin lines, indicating a positive identification. Negative AGPT results are characterized by the absence of visible lines of precipitation, suggesting that the antigen and antibody did not interact to form detectable complexes. This outcome may indicate the absence of specific antibodies to the tested antigen, a low concentration of the antigen, or a technical issue with the test procedure (Hutomo et al., 2018). The precipitation line pattern can be analyzed to elucidate the relationship between different antigens. Specifically, a continuous line at the junction of two lines indicates serological identity, suggesting that the antigens share similar epitopes. Crossed lines indicate non-identity, whereas a continuous line with a spur indicates partial identity, implying that the antigens possess some common epitopes but also exhibit distinct epitopes.
Purity of IgY isolated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis
Analysis of IgY molecule weight determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) using 12% separating gel and 4% stacking gel. Before entering into the gel well, the supernatant samples were added with a buffer containing 62.5 mM Tris-HCl (pH 11 6.8), 10% glycerol, and 0.001% bromophenol blue with 5% (v/v) mercaptoethanol and heated for 5 minutes. As much as 20 μl of antigen-soluble E. coli entered the gel well. Electrophoresis was performed in 60 minutes at 125 V at 25°C in the buffer electrode (pH 8.3). Then, gel colorization was carried out using Coomassie brilliant blue for 30 minutes, and destaining was continued using a solution containing 20% methanol and 10% glacial acetate acid (Rizkiantino et al., 2020).
The purity of the isolated IgY was verified through SDS-PAGE analysis, which typically revealed two distinct bands corresponding to the heavy chain (HC) and light chain (LC) of the molecule. A highly purified IgY preparation is characterized by the prominent presence of these two bands, with minimal or negligible contamination by other proteins (Bergmann-Leitner et al., 2008). Specifically, the HC of IgY exhibits a molecular weight of approximately 70 kDa, constituting the primary component of the molecule, while the LC has a molecular weight of approximately 25 kDa, playing a crucial role in maintaining the structural integrity of IgY (Hodek et al., 2013). The predominance of these two bands on the gel, with minimal background or extraneous bands, indicates the purification process’s efficacy in separating IgY from other proteins present in the original sample. This suggests that the isolated IgY is of high purity and suitable for further applications (Bergmann-Leitner et al., 2008).
Immunobloting
The E. coli pellet sample that has been harvested, proceeded with many treatments including: protein extraction of E. coli by HCl, protein extraction of E. coli inactivated by 90°C at 2 hours, protein extraction of E. coli inactivated by 4% formaldehyde, and protein extraction of freeze-thaw E. coli and whole E. coli cells without any treatment. Escherichia coli samples were separated by SDS-PAGE 15% gel and then stained using Coomassie Brilliant Blue R-250. Detection of E. coli protein by IgY was performed using the colorimetric western blot technique, as described by Towbin et al. (1979). Briefly, the separated E. coli proteins were transferred onto a nitrocellulose membrane via electroblotting using a mini Trans-Blot System (Bio-Rad) at a constant current of 30 mA for 2 hours. Following electroblotting, the nitrocellulose membrane was blocked with blocking buffer at 4°C. The membrane was incubated for 1 hour at 4°C in IgY anti-E. coli diluted using PBS-T in a volume ratio of 1:1,000 and washed with PBS-T solution. After washing three times, the nitrocellulose membrane was incubated for 1 hour in anti-chicken antibody conjugated with peroxidase (Katalog #A9046; Sigma-Aldrich) diluted with PBS-T in a 1:500 volume ratio. Subsequently, the membrane was washed with PBS-T and incubated for 5 minutes with a chromogenic solution (10 mg diaminobenzidine, 10 ml PBS dan 6,4 µl H2O2). Throughout the incubation process, if the E. coli protein binds with IgY in the nitrocellulose membrane, a chemical reaction occurs between the anti-chicken antibody conjugated with peroxidase (anti-chicken antibody conjugated with peroxidase that has been bind with IgY in which IgY binds with E. coli protein before) and 3,3′-Diaminobenzidine and H2O2 that compose the chromogenic solution and cause the color to change to bluish–purple at the nitrocellulose membrane located by E. coli protein (Tarsalewska et al., 2025).
Ethical approval
This research protocol was approved by the Ethics Commission of the School of Veterinary Medicine and Biomedicine, Bogor Agricultural Institute (260/KEH/ SKEx/2025).
Results
Antigen preparation results
Group A, E. coli inactivated using 4% formaldehyde overnight. Based on the sterility test, no colony growth was observed on nutrient agar; therefore, it is suitable for the antigen to be the candidate vaccine for the chicken in Group A.
Group B, E. coli inactivated using temperature 90°C for 2 hours. Based on the sterility test, no colony growth was observed on nutrient agar; therefore, it is suitable for the antigen to be the candidate vaccine for the chicken in Group B.
Chicken vaccination result
Vaccinated chickens grow normally without any signs of sickness. Vaccinated chickens did not change their body weight, but their egg production stopped several days after vaccination.
The examination of serum and egg yolk at the chicken group that have been vaccinated with the temperature-inactivated E. coli and chicken group that have been vaccinated with the formaldehyde-inactivated E. coli, Ig Y detected at week 3rd and 6th post-vaccination.
Chicken vaccination result by the AGPT
The detection of IgY presented every week using the AGPT is shown in Table 1. Based on the AGPT, IgY in chicken group A (containing E. coli inactivated with 4% formaldehyde overnight) showed a step-by-step increase in antibody titer at week 3rd namely the serum IgY titer 22; week 6 serum IgY titer 32; week 3 egg yolk IgY titer 21 and at week 6th the egg yolk IgY titer 22). The IgY titer at chicken group B (containing E. coli inactivated at 90°C for 2 hours) have the antibody titer higher and stable, at week 3rd the serum IgY titer 23, at week 6th the serum IgY titer 23 and at week 3rd the egg yolk IgY titer 21, at week 6th the egg yolk IgY titer 23. The examination result of the IgY titer is shown in Table 2.
Table 1. Results of IgY detection in the serum and egg of chickens vaccinated with inactivated E. coli using the AGPT with dilution.
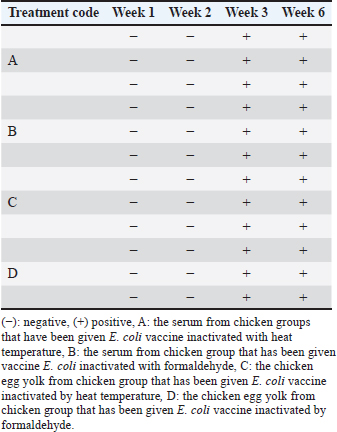
Table 2. Results of the AGPT conducted on serum and egg yolk samples from 22-week-old hens with a body weight range of 2–3 kg.

Chicken vaccination results using SDS-PAGE
The SDS-PAGE examination result on purified serum and egg yolk (Fig. 1) showed a protein band of 65 and a weight of 25 kDa. Protein band 65 was suspected to be a HC of IgY and the protein band 25 was a LC of IgY.
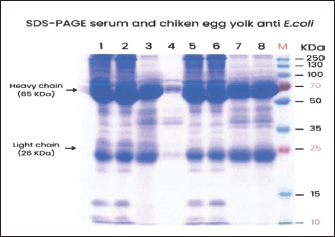
Fig. 1. SDS-PAGE of serum and chicken egg yolk anti-E. coli. Samples: Chicken serum heat inactivation (90°C) sampled at week-3 (1) and week-6 (2); egg yolk heat inactivation at week-3 (3) and week-4 (4); chicken serum formaldehyde inactivation at week-3 (5) and week-6 (6); egg yolk formaldehyde inactivation at week-3 (7) and week-4 (8); M: marker.
Immunobloting
Immunoblotting examination with the colorimetric western blotting method (Fig. 2) revealed a color change on the nitrocellulose membrane band to a bluish-purple color, indicating that IgY can recognize E. coli protein with a molecular weight of 29 kDa.
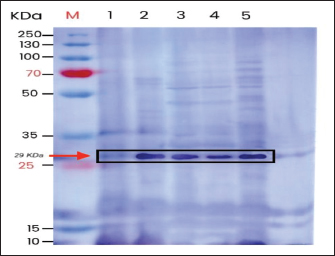
Fig. 2. Immunoblotting test. E. coli proteins from various preparations recognized by IgY anti-E. coli are in the 29-kDa band, which is an adhesin protein. M: marker, (1) whole E. coli protein; (2) E. coli protein extraction by HCl; (3) protein extraction of E. coli inactivated by 90ºC; (4) E. coli protein extraction inactivated by 4% formaldehyde; (5) E. coli protein extraction by freeze-thaw.
Discussion
The AGPT result on the serum and egg yolk showed a precipitation line in both groups. The presence of a precipitation line indicated a specific interaction between the antibody and E. coli, confirming the antibody’s binding affinity for the bacterial antigen. In the treatment group, the specific antibody against E. coli began to emerge at week 3rd, the antibody titer in both serum and egg yolk in Group B (heat temperature-inactivated E. coli) had a higher titer concentration than that in Group A (formaldehyde-inactivated E. coli). This phenomenon can be attributed to the preservation of the native antigenic structure by heat inactivation, thereby facilitating recognition by the immune system. In contrast, formaldehyde alters the antigenic structure, resulting in reduced recognition by the immune system (Elveborg et al., 2022). A similar phenomenon occurs when Salmonella spp. inactivated with heat induce higher IgY production by Lohmann brown chickens vaccinated with Salmonella spp. vaccine than immunogens inactivated with formaldehyde (Gordon et al., 2016). However, this finding is inconsistent with that of Esmailnejad et al. (2019), who notably demonstrated that formalin-inactivated immunogens (Salmonella typhimurium and Salmonella enteritidis) induced relatively higher IgY responses than heat-inactivated immunogens in quail.
The E. coli vaccine used in this study was an emulsion accompanied by an adjuvant. The adjuvant was used on vaccination to trigger the immune response to maximize the formation of antibodies. Adjuvants can be categorized as immunostimulants and delivery systems. Immunostimulant is a dangerous signaling molecule that causes the maturation and activation of the Antigen-Presenting Cell by targeting the receptor toll-like receptor and another receptor, the pattern recognition receptor, to increase the production of antigen and costimulation signals, which in turn increases the adaptive immune response (Zhao et al., 2023). This study used Montanide ISA 70 as an adjuvant in the chicken E. coli vaccine. Rizkiantino et al. (2020) reported that the chicken vaccinated with an adjuvant, Montanide ISA 70, helped to increase IgY production more quickly than the chicken vaccinated with Freund adjuvant and mineral oil.
Protein characterization on IgY was carried out using SDS-PAGE to recognize protein molecule weight. Generally, the weight of IgY was 66–67 kDa, HC weight was 60–70 KDa, and LC weight was 22–30 kDa (Li et al., 2009).
Immunoblotting examination shows that IgY can recognize E. coli protein with a molecular weight of approximately 29 kDa, which is assumed to be an adhesin protein that is an important factor on bacterial colonization in the intestine mucosa, refering to the result of a study conducted by Hacker et al. (1993).
Several limitations of this study should be considered. The restricted sample size may have diminished the ability of the study to detect significant differences, whereas the absence of replication may have introduced variability in the results. Additionally, biases in antigen preparation may have influenced immune response outcomes. These limitations potentially impact the study’s generalizability and reliability, underscoring the importance of future investigations that incorporate larger sample sizes, experimental replication, and refined antigen preparation methods. Addressing these limitations can improve the validity and relevance of the study, ultimately advancing our understanding of the research topic.
Conclusion
This study provides concrete evidence for the beneficial effect of IgY as a prophylactic and therapeutic modality against E. coli. There are more pieces of evidence with standardized animal studies aimed at using IgY against E. coli infection. Further research on human subjects could open a new perspective on the application of IgY as a therapeutic agent.
Acknowledgments
The authors express their deepest gratitude and appreciation for the assistance and support received throughout the completion of this research paper. The authors want to take this opportunity to acknowledge the contributions of the Primates Research Center IPB University facilitated the laboratory facility.
Conflict of interest
The authors have no conflicts of interest to declare.
Funding
This study received financial and facility support from PT. Biomol Nusantara Mandiri Tbk.
Authors’ contributions
Muhammad Farid Rizal was responsible for the overall design and execution of the research. I. Wayan T. Wibawan was responsible for data collection and analysis. The manuscript was written and all necessary revisions were made by all authors. Roostita L. Balia and Denniswara Sibit conceptualized the study and contributed to the experimental design. Additionally, I. Wayan T. Wibawan helped with data analysis and interpretation. Huda S. Darusman and Joko Pamungkas are responsible as supervisors. Hadrianus Lau contributed to the literature review, interpretation of results, and final manuscript editing. All authors have read and approved the final version of the manuscript before its publication in the journal.
Data availability
The data to support the findings of this study are available upon reasonable request to the corresponding author.
References
Abbas, A.T., El-Kafrawy, S.A., Sohrab, S.S. and Azhar, E.I.A. 2019. IgY antibodies for immunoprophylaxis and respiratory infection therapy. Hum. Vaccines. Immunotherapeutics. 15(1), 264–275; doi:10.1080/21645515.2018.1514224
Amro, W.A., Al-Qaisi, W. and Al-Razem, F. 2018. IgY antibody production and purification from chicken egg yolk. J. Genet. Eng. Biotechnol. 16(1), 99–103; doi:10.1016/j.jgeb.2017.10.003
Anjum, M.F., Schmitt, H., Börjesson, S., Berendonk, T.U., Donner, E., Stehling, E.G., Boerlin, P., Topp, E., Jardine, C., Li, X., Li, B., Dolejska, M., Madec, J.Y., Dagot, C., Guenther, S., Walsh, F., Villa, L., Veldman, K., Sunde, M., Krzeminski, P., Wasyl, D., Popowska, M., Järhult, J., Örn, S., Mahjoub, O., Mansour, W., Thái, Đ.N., Elving, J. and Pedersen, K. 2021. The potential of using E. coli as an indicator for the surveillance of antimicrobial resistance (AMR) in the environment. Curr. Opin. Microbiol. 64, 152–158; https://www.sciencedirect.com/science/article/pii/S1369527421001302
Arshadi, N., Mousavi, S.L., Amani, J. and Nazarian, S. 2020. Immunogenic potency of formalin and heat-inactivated E. coli O157: h7 in a mouse model administered by different routes. Avicenna. J. Med. Biotechnol. 12(3), 194–200.
Bergmann-Leitner, E.S., Mease, R.M., Duncan, E.H., Khan, F., Waitumbi, J. and Angov, E. 2008. Evaluation of immunoglobulin purification methods and their impact on antigen-specific antibody quality and yield. Malar. J. 7, 129; doi:10.1186/1475-2875-7-129
Biswas, S., Nazmi, A., Pitesky, M., Gallardo, R. and Pandey, P. 2019. Thermal inactivation of Escherichia coli and Salmonella typhimurium in poultry carcass and litter at thermophilic temperatures. J. Appl. Poultry. Res. 28(2), 307–317; doi:10.3382/japr/pfy072
Braz, V.S., Melchior, K. and Moreira, C.G. 2020. Escherichia coli as a multifaceted pathogenic and versatile bacterium. Front. Cell. Infect. Microbiol. 10, 548492; doi:10.3389/fcimb.2020.548492
Chismirina, S., Sungkar, S., Adlim, M. and Darmawi, D. 2023. Potential of goats’ milk from Aceh hybrid and Etawa goats in producing specific anti-S. mutans antibodies as candidate antibodies for caries immunotherapy version 1; peer review: awaiting peer review. F1000Research 12, 944; doi:10.12688/f1000research.132668.1
Dhruvi, P., Gireesh, B.K., Sreenivasa, N., Amel, G., Pankaj, K., Apurva, S., Krishna, K.Y., Lamjed, M. and Haresh, S.K. 2025. Comprehensive analysis of the major IgY antibody extraction strategies from chicken egg yolk. Vet. Immunol. Immunopathol. 283, 110928; doi:10.1016/j.vetimm.2025.110928
El-Kafrawy, S.A., Abbas, A.T., Oelkrug, C., Tahoon, M., Ezzat, S., Zuml, A. and Azhar, E. 2023. IgY antibodies: the potential to overcome antibiotic resistance. Front. Immunol. 14, 10653; doi:10.3389/fimmu.2023.1065353
Elveborg, S., Monteil, V.M. and Mirazimi, A. 2022. Inactivation methods of highly pathogenic viruses for molecular, serological, or vaccine development purposes. Pathogens 11(2), 271; doi:10.3390/pathogens11020271
Esmaeili, Z., Shahsavar, S.K., Keikha, M. and Ghazvini, K. 2024. Chicken immunoglobulin (IgY) as an alternative treatment for bacterial infections: advantages, disadvantages, and mechanisms. J. Microbiol. Infect. Dis. 14(3), 95–102; doi:10.5455/JMID.2024.v14.i3.2
Esmailnejad, A., Abdi-Hachesoo, B., Hosseini, N.E. and Shakoori, M. 2019. Production, purification, and evaluation of quail immunoglobulin Y against S. typhimurium and S. enteritidis. Mol. Immunol. 107, 79–83; doi:10.1016/j.molimm.2019.01.012
Gordon, G., Moran, G., Ayala, L., Seqqat, R., Fernandez, R. and Torres, M. 2016. Generation and characterization of IgY antibodies from Lohmann Brown hens immunized with Salmonella spp. for their subsequent application in nanotherapy. Biol. Med. 8(3), 284; doi:10.4172/0974-8369.1000284
Hacker, J., Kestler, H., Hoschützky, H., Jann, K., Lottspeich, F. and Korhonen, T.K. 1993. Cloning and characterization of the S fimbrial adhesin II complex of an Escherichia coli O18: k1 meningitis isolate. InfectImmunity 61(2), 544–550; doi:10.1128/iai.61.2.544-550.1993
Hidayat, R., Pasaribu, F.H., Mulatsih, S. and Amin, A.A. 2024. Analysis of the sustainability of using immunoglobulin Y (IgY) as an antibiotic substitute. IOP Conf. Ser.: Earth Environ. Sci. 1359(1), 12–140; doi:10.1088/1755-1315/1359/1/012140
Hodek, P., Trefil, P., Šimůnek, J., Hudecek, J. and Stiborova, M. 2013. Optimized protocol for purification of chicken antibody (IgY) providing wlectrophoretically homogenous preparations. Int. J. Electrochemical. Sci. 8, 113–124; doi:10.1016/S1452-3981(23)14006-5
Hutomo, S., Susilowati, H., Agustina, D. and Asmara, W. 2018. Analysis of anti-S. sanguinis IgY ability to inhibit S. sanguinis adherence. Dental. J. 51(1), 33–36; doi:10.20473/j.djmkg.v51.i1.p33-36
Keita, A., Le Devendec, L., Amelot, M., Puterflam, J., Lucas, C., Bougeard, S., Delannoy, S., Schouler, C., Fach, P., Lucas, P., Souillard, R. and Kempf, I. 2022. Efficacy of passive immunization in broiler chicks via an inactivated Escherichia coli autogenous vaccine administered to broiler breeder hens. Avian Pathol. 51(5), 445–456; doi:10.1016/j.avianpathol.2022.02.045
Khabazzadeh, T.N., Mahdavi, M., Maleki, F., Zarrati, S. and Tabatabaie, F. 2016. Role of Montanide ISA 70 as an adjuvant in immune responses against Leishmania major induced by thiol-specific antioxidant-based protein vaccine. J. Parasit. Dis. 40(3), 760–767; doi:10.1007/s12639-014-0574-8
Li, X.Y., Jin, L.J., Lu, Y.N., Zhen, Y.H., Li, S.Y., Wang, L.H. and Xu, Y.P. 2009. Chitosan-alginate microcapsules for oral delivery of egg yolk immunoglobulin (IgY): effects of chitosan concentration. J. Immunoglobulin. Appl. Biochem. Biotechnol. 159(3), 778–787; doi:10.1007/s12010-009-8628-6
Low, S.X., Aw, Z.Q., Loo, B.Z., Lee, K.C., Oon, J.S., Lee, C.H. and Ling, M.H. 2013. Viability of Escherichia coli ATCC 8739 in nutrient broth, luria-bertani broth, and brain heart infusion over 11 weeks. Electron. Physician. 5(1), 576–581; doi:10.14661/2013.576-581
Mahalakshmi, K., Arangannal, P., Jayaprakash, J. and Muthukumaran, M. 2024. Efficacy of immunoglobulin Y chewable tablets on Streptococcus mutans count in patients undergoing orthodontic treatment. Int. J. Clin. Pediatr. Dentist. 17(3), 265–269. doi:10.5005/jpjournals-10005-2774
Marcotte, H. and Hammarström. 2015. Passive immunization: toward magic bullets. Mucosal. Immunol. 2015, 1403–1434.
Matlock, B.C. and Wilmington, D.E. 2019. Differences in bacterial optical density measurements between UV-Visible spectrophotometers. Technical note N. 52236, Thermo Fisher Scientific.
Okwor, E., Eze, D., Okonkwo, K. and Ibu, J. 2011. Comparative evaluation of agar gel precipitation test (AGPT) and indirect haemagglutination test (IHA) for detecting antibodies against infectious bursal disease (IBD) virus in village chickens. Afr. J. Biotechnol. 10, 16024–16027; doi:10.5897/AJB11.2006
Pereira, E.P.V., Van Tilburg, M.F., Florean, E.O.P.T. and Guedes, M.I.F. 2019. Egg yolk antibodies (IgY) and their applications in human and veterinary health: a review of the literature. Int. Immunopharmacol. 73, 293–303; doi:10.1016/j.intimp.2019.05.015
Putri, D., Handharyani, E., Soejoedono, R., Setiyono, A. and Poetri, O. 2018. Newcastle disease antibody production and characterization as a reagent to develop a rapid immunodiagnostic test tool. Vet. World 11, 895–901; doi:10.14202/vetworld.2018.895-901
Radwan, F., El-Shemy, A., Bastamy, M. and Amer, M. 2023. Review article: chicken immunoglobulin IgY Preparation, and Its Applications in Prevention and/or Control of Some Microbial Affections. Egypt. J. Vet. Sci. 54, 653–668; doi:10.21608/ejvs.2023.200854.1464
Rizkiantino, R., Wibawan, I.W., Pasaribu, F., Soejoedono, R.D., Poetri, O., Arnafia, W., Sasi, K. and Reisinta, D. 2020. Potential of adjuvant against anti-Streptococcal immunoglobulin Y (IgY) production in aquaculture. Jurnal Kedokteran Hewan-Indonesian J. Vet. Sci. 14, 66–72; doi:10.21157/j.ked.hewan.v14i3.16911
Salam, M.A., Al-Amin, M.Y., Salam, M.T., Pawar, J.S., Akhter, N., Rabaan, A.A. and Alqumber, M.A.A. 2023. Antimicrobial resistance: a serious threat to global public health. Health Care (Basel). 11, 12346; doi:10.3390/healthcare11131946
Setthawong, P., Yamkasem, J., Khemthong, M., Tattiyapong, P., Metheenukul, P., Prasertsincharoen, N., Lertwanakarn, T., Thengchaisri, N. and Surachetpong, W. 2025. Development of IgY-based passive immunization against Tilapia lake virus: development and in vitro neutralization assays. Viruses 17(3), 448; doi:10.3390/v17030448
Slifka, M.K. and Amanna, I.J. 2018. Passive immunization. Plotkin’s Vaccines. 2018, 84–95.e10; doi:10.1016/B978-0-323-35761-6.00008-0
Siriya , P., Chu, C., Chen, M.T., Lo, C.C., Huang, S.L. and Lien, T.F. 2013. Extraction and purification of anti-H. pylori IgY. J. Agricult. Sci. 5, 132; doi:10.5539/jas.v5n3p132
Tan, T., Xia, L., Tu, K. et al. 2018. Improved Macaca fascicularis gene annotation reveals evolution of gene expression profiles in multiple tissues. BMC Genomics, 19, 787. doi: 10.1186/s12864-018-5183-y
Tamai, S., Katafuchi, M., Hui, X. and Suzuki, Y. 2025. Detection and collection of shiga toxin-producing Escherichia coli using foam concentration without membrane filtration. Ecotoxicol. Environ. Saf. 291, 147; doi:10.1016/j.ecoenv.2025.117797
Tarsalewska C.P., Ponichtera, K., Rybicka, M., Sowa-Rogozińska, N., Sominka-Pierzchlewicz H Stodolna., Ogonowska, A., Kosciuk, P., Glosnicka, A. and Bielawski, R. 2025. Growth-inhibitory effect of chicken egg yolk polyclonal antibodies (IgY) on zoonotic pathogens Campylobacter jejuni, Salmonella spp., and Escherichia coli. Int. J. Mol. Sci. 26, 1040; doi:10.3390/ijms26031040
Towbin, H., Staehelin, T. and Gordon, J. 1979. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and applications. Proc. Nat. Acad. Sci. U. S. A. 76(9), 4350–4434; doi:10.1073/pnas.76.9.4350
Tuttle, A.R., Trahan, N.D. and Son, M.S. 2021. Growth and maintenance of Escherichia coli strains. Curr. Protoc. 1(1), 551; doi:10.1002/cpz1.20
Vela Ramirez, J.E., Sharpe, L.A. and Peppas, N.A. 2017. Current state and challenges in developing oral vaccines. Adv. Drug. Deliv. Rev. 114, 116–131; doi:10.1016/j.addr.2020.11.013
Westcott, M.M., Blevins, M., Wierzba, T.F., Morse, A.E., White, K.R., Sanders, L.A. and Sanders, J.W. 2023. The immunogenicity and properties of a whole-cell ETEC vaccine inactivated with psoralen and UVA light in comparison to formalin. Microorganisms 11(8), 2040; doi:10.3390/microorganisms11082040
Xia, L., Tu, K., Tang, J., Yin, F., Dai, L., Lei, P., Dong, B., Hu, H., Fan, Y., Yu, Y. and Xie, D. 2018. Improved fascicularis gene annotation reveals evolution of gene expression profiles in multiple tissues. BMC. Genomics. 5183, doi:10.1186/s12864-018-5183-y
Zhao, T., Cai, Y., Jiang, Y., He, X., Wei, Y., Yu, Y. and Tian, X. 2023. Vaccine adjuvants: mechanisms and platforms. Signal Transduct. Targeted Ther. 8, 283; doi:10.1016/j.sttt.2023












