| Research Article | ||
Open Vet. J.. 2025; 15(10): 5355-5360 Open Veterinary Journal, (2025), Vol. 15(10): 5355-5360 Research Article Oxidation-antioxidation biomarkers and histopathological brain damage induced by sodium benzoate exposureFatima Aziz Mahdi Al-badry*Department of Biology, College of Education for Pure Sciences, University of Thi-Qar, Thi-Qar, Iraq *Corresponding Author: Fatima Aziz Mahdi Al-badry. Department of Biology, College of Education for Pure Sciences, University of Thi-Qar, Thi-Qar, Iraq. Email: fatimaaziz.bio [at] utq.edu.iq; fatima.biohis [at] gmail.com Submitted: 08/05/2025 Revised: 10/09/2025 Accepted: 21/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
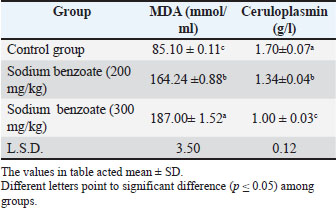
AbstractBackground: Sodium benzoate, a preservative, can cause adverse histological and physiological impacts when administered at different doses. Aim: This study aimed to investigate the negative impact of sodium benzoate on the oxidation-anti-oxidation system and histopathological changes in the brain of female rats. Methods: Thirty-six mature female rats were divided into two main groups: the control group received only water and diet, whereas the second group was divided into two subgroups, each receiving two doses of sodium benzoate (200 and 300 mg/kg body weight) for 8 weeks. Oxidative stress biomarkers [malondialdehyde, ceruloplasmin, catalase (CAT), glutathione (GSH), and albumin] and histopathological sections of the brain were examined. Results: The results showed a significant excess in the malondialdehyde level at two doses compared with the control group. Significant declines in ceruloplasmin, CAT, GSH, and albumin levels were observed in the two treated groups. Histological lesions in the brain included vacuolation, degeneration, and hypertrophy of neurons, accumulation of inflammatory cells, bleeding, edema, and congestion, in addition to the occurrence of nuclei arranged in a pair and encompassed with clear space. Conclusion: Sodium benzoate intake was linked to adverse oxidative stress biomarkers and brain damage; these damages were more intensive at high concentrations than at low concentrations. Keywords: Brain, Catalase, Glutathione, Rats, Sodium benzoate. IntroductionPreservatives are substances that prevent chemical and biological deterioration. They are of three types: antioxidants, anti-enzymatic, and antimicrobial (Seetarmaiah et al., 2011). Sodium benzoate is an antimicrobial preservative that inhibits the growth of microorganisms that produce toxins, leading to changes in the test and nutritional contents of food (Wroblewska, 2009). It is used as a flavoring agent and a preservative in acidic food, beverages, and milk products (Shahid et al., 2018) as well as in pharmaceuticals, tooth paste, and cosmetics (Piper and Piper, 2017). Sodium benzoate is a white crystalline powder that dissolves in water and ethanol (Hadi and Mahdi, 2019). Although it is used in preventing food spoilage caused by pathogens, it has negative effects resulting from chronic and sub-chronic consumption. Excessive intake causes physiological and hematological harm, acute poisoning, teratogenicity, aggression, and renal-hepatic damage, (Sinha and D’Souza, 2012; Jalil et al., 2022). In addition to agitation of the skin, respiratory channel damages with gastric ulcers (Alwan et al., 2022). Furthermore, when it is mixed with vitamin C in carbonated beverages, it can form carcinogens that destroy DNA in mitochondria (Nair, 2001). Therefore, the current study aimed to investigate the negative impact of sodium benzoate on oxidation-anti-oxidation system and histopathological changes in the brain of female rats. Materials and MethodsExperimental designedThirty-six healthy female rats (Rattus norvegicus) were used in this study, weighting (220–235 g) and aged (3 months). Animals were obtained from the National Organization for Drug Control and Research/Baghdad Province. Rats were housed under control conditions with a 12:12 hour light: dark cycle and 22 ± 2°C, and they were adapted for 2 weeks in the animal facility and provided with a standard diet and water during the acclimation period. The rats were divided into two main groups: the control group (n=12) which received water and diet only without sodium benzoate, and the second group (n=24) rats treated with sodium benzoate, which was divided into two subgroups: group A and group B, which received 200 and 300 mg/kg of sodium benzoate, respectively, orally by stomach tube for 2 months. Sera collectionA mixture of xylazine and ketamine hydrochloride (1:8 mg/kg body weight) was used for euthanasia that was injected intramuscularly. The animals were sacrificed at the end of the experimental period, and blood was collected by cardiac puncture. The blood was placed in gel tubes for sera separation with centrifugation (10,000 rpm/10 minutes). A micropipette was used to draw the sera, and oxidative stress parameters were measured. Oxidation-antioxidation biomarkers measurementThe biomarkers of oxidation, including malondialdehyde (MDA) and ceruloplasmin, were evaluated in serum samples. MDA levels were determined using the color method of thiobarbituric acid. The measurement was based on the reaction between MDA and TBA (Muslih et al., 2002). Menden et al. (1977) method was used for ceruloplasmin level estimation. In addition, the oxidation enzymes catalase (CAT), glutathione (GSH), and albumin were analyzed. The CAT activity in the serum was determined according to the procedure of Hadwan and Abed (2016), while the GSH activity was measured by Heibashy (2000). The method of Doumas et al. (1971) was used for Albumin assessment. Histopathological examinationThe brains of rats in the control group and both groups treated with sodium benzoate were processed and fixed in 10% formalin as the first step for section preparation. Histological sections were obtained according to the method of Bancroft and Gamble (2008). The procedure included washing the samples with Tab water to remove the fixative solution and dehydration by ascending concentrations of ethanol. Xylene was used for clearing, and tissues were embedded in paraffin wax. Sections were stained with hematoxylin and eosin. An experienced pathologist examined these sections microscopically. Statistical analysisThe current data were analyzed using the SPSS software (version 20). All values are presented as mean ± SD for all parameters studied. A one-way ANOVA was used, and the least significant difference test was used to show significant differences among groups at (p ≤ 0.05) level (Moder, 2010). Ethical approvalThis study has been accepted by the Ethics Committee of Animal Research at the College of Education for Pure Sciences/University of Thi-Qar, which awarded a positive agreement at number 7/54/3500 on 12/10/2024. ResultsOxidative stress parametersThe effect of sodium benzoate at the two doses (200 and 300 mg/kg) in female rats resulted in a significant increase (p ≤ 0.05) in serum malondialdehyde levels compared with the control group. The higher dose of sodium benzoate (300 mg/kg) caused a significant elevation in MDA levels significantly (p ≤ 0.05) compared with the lowest dose (200 mg/kg). Moreover, the oxidation biomarkers in Tables 1 and 2, which included (ceruloplasmin, CAT, GSH, and albumin) showed a significant decline (p ≤ 0.05) in both treated groups compared to the control group. The higher dose group displayed a significant treated reduction (p ≤ 0.05) in these biomarkers of oxidation compared with the lowest-dose group. Table 1. Effect of sodium benzoate in malondialdehyde and Ceruloplasmin levels (n=12).
Table 2. Effect of sodium benzoate in other oxidation enzymes levels (n=12).
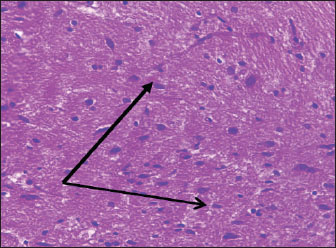
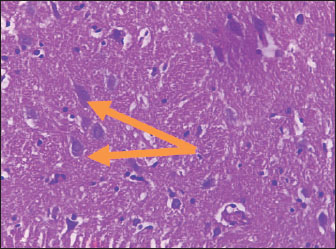
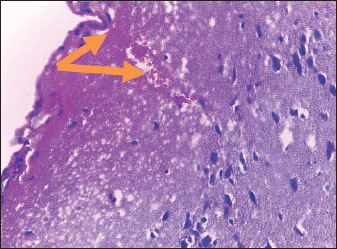
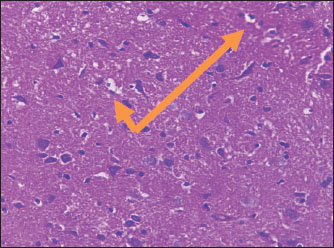
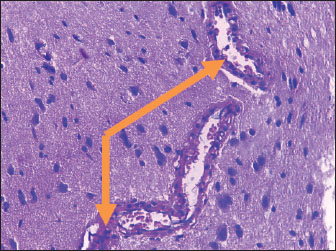
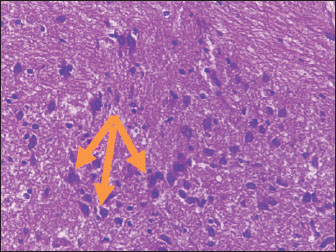
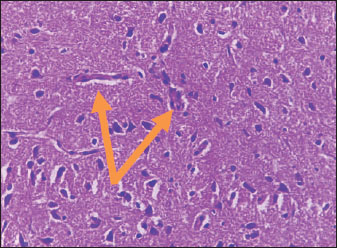
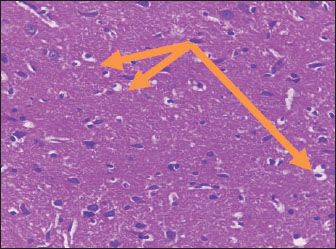
Histological studyBrain of control group appeared normal structure of tissue with manifestation of Purkenji cells (Fig. 1), whereas sodium benzoate administration at low dose (200 mg/kg) occurred several histological lesions as hypertrophy of neurons (Fig. 2), pair nuclei arranged encompassed with clear space (Fig. 3), bleeding with degeneration of nerve cells (Figs. 4 and 5), respectively. In addition, lesions such as vacuolation, congestion, and accumulation of inflammatory cells were also evident (Figs. 6 and 7). However, severe histopathological lesions were observed following high-dose of sodium benzoate exposure, such as degeneration of neurons stained in dark blue (Fig. 8), prominent vascular duct with inflammation (Fig. 9), vacuolation, edema, and clear congestion as seen in Figures 10–12, respectively.
Fig. 1. Points to the brain of control group: normal structure and manifestation of purkinje cells (H&E) (100 ×).
Fig. 2. Points to the brain of sodium benzoate group (200 mg/kg): hypertrophy of neurons (H&E) (100×).
Fig. 3. Points to the brain of sodium benzoate group (200 mg/kg): pair of nuclei ranking enclitic by clear area (H&E) (100×).
Fig. 4. Points to the brain of sodium benzoate group (200 mg/kg): heavy hemorrhage (H&E) (100×).
Fig. 5. Points to the brain of sodium benzoate group (200 mg/kg): degeneration of neurons which stained in dark blue (H&E) (100×).
Fig. 6. Points to the brain of sodium benzoate group (200 mg/kg): vacuolation of neurons (H&E) (100×).
Fig. 7. Points to the brain of sodium benzoate group (200 mg/kg): vacuolation of neurons (H&E) (100×).
Fig. 8. Points to the brain of sodium benzoate group (300 mg/kg): degeneration of neurons which stained in dark blue (H&E) (100×).
Fig. 9. Points to the brain of sodium benzoate group (300 mg/kg): vascular prominent duct with assemblage of inflammatory cells in the lumen (H&E) (100×).
Fig. 10. Points to the brain of sodium benzoate group (300 mg/kg): vacuolation of nerve cell at different stages (H&E) (100×).
Fig. 11. Points to the brain of sodium benzoate group (300 mg/kg): perivascular edema (H&E) (100×).
Fig. 12. Points to thebrain of sodium benzoate group (300 mg/kg): sever congestion (H&E) (100×). DiscussionSodium benzoate is a widely used food preservative. There are recommended limits for safe daily intake of this substance, as excessive consumption can cause serious health effects. Therefore, the current study aimed to investigate the negative impact of sodium benzoate on oxidation-anti-oxidation system and histopathological changes in the brain of female rats. The indicators of oxidation showed high levels of malondialdehyde in the sodium benzoate groups, which can be attributed to lipid peroxidation by this substance. Al-Ameen et al. (2022) referred to free radicals fashioning and reduction of antioxidants as super oxide dismutase (SOD), leading to damage in organic molecules in cells and lipid peroxidation. The antioxidants (ceruloplasmin, CAT, GSH, and albumin) showed a decline. This would have been linked to the effect of sodium benzoate and its exhaustion for the suppression of free radicals. A previous study noted that sodium, benzoate reduced the levels of antioxidant enzymes such as GSH (Langlands et al., 2024). Similarly, El-Shennawy et al. (2020) observed oxidation stress and imbalance of antioxidant enzymes (decline of CAT, GSH, and SOD) resulting from sodium benzoate. The treatment of rats with sodium benzoate at varying doses ranging (100–1,000 mg/kg) caused an elevation of MDA with a decline of GSH (Osman et al., 2025). Similarly, Olofinnade et al. (2021) found an elevation of MDA in the brains of mice after sodium benzoate exposure. The histopathological lesions observed in the brain can be associated with two doses of sodium benzoate that cause defects in brain architecture. This agrees with the findings of Al-Ameen et al. (2022), who pointed to brain damage in the cortex, such as vacuolation, satellitosis, edema, and focal gliosis. The same study also described mild to severe changes according to the sodium benzoate ingestion dose. Similarly, a previous study by Mohammed and Al-Gnami (2022) referred to the passive histological effects of sodium benzoate in many tissues, causing vacuolation, degeneration, and infiltration of inflammatory cells. Furthermore, histopathological lesions may be due to DNA damage of neurons by sodium benzoate exposure. This is similar to the findings of Mohiuddin et al. (2022), who showed DNA fragments of rats treated with a high dose (120 mg/kg) of sodium benzoate. The structural damage to the brain may be correlated with oxidative stress induced by sodium benzoate intake and the smashing of nucleic acids by reactive oxygen species, which are considered responsible for nervous changes. Khan et al. (2022) reported oxidative stress and toxicological impacts in rats treated with sodium benzoate. This is consistent with the findings of Cantuti et al. (2000) who viewed neurodegenerative flaws and, morphological and functional malformations in the brain caused by oxidative stress. Al-Badry (2024) also linked the adverse histological lesions and physiological changes to lipid peroxidation that resulted from the intake of Puma supplements. Excessive oxidation damage causes loss of cell functionality, resulting in cell death. Sabour and Ibrahim (2019) mentioned Similarly, mentioned oxidative stress elevation in erythrocytes of rat exposed to different doses of sodium benzoate . The pathological changes recorded in the current results following sodium benzoate administration may be associated with the effects of free free radicals that cause the breakdown of polyunsaturated in the membranes of cells, leading to functional disorders. Furthermore, Al-Badry (2023) demonstrated that the brain was histologically affected by topiramate exposure. ConclusionIn conclusion, we conclude that the exposure of rats to sodium benzoate at two doses (200 and 300 mg/kg) led to disturbances in oxidation parameters, causing malondialdehyde elevation and ceruloplasmin decline. In addition to lowering of oxidation enzymes, such as CAT, GSH, and albumin. Furthermore, histological damages in the brain were recorded. Moreover, increasing the consumption of sodium benzoate at high doses had a severe impact compared with those lower doses. AcknowledgmentsThe author is grateful to the University of Thi-Qar/College of Education for Pure Sciences, especially the Department of Biology, for their help and support in the current work achievements. My sincere thanks and appreciation to Prof. Dr. Ansam Ali Flefil Al-Hussinawy for reviewing the language of the manuscript. Conflict of interestThe author declares that there is no conflict of interest. FundingThis study received no specific grant. Authors’ contributionFatima Aziz Mahdi contributed to the conceptualization, data curation, methodology, analysis of results, and writing manuscript. Data availabilityAll data supporting this study's findings are available within the manuscript. ReferencesAl-Ameen, S.A., Jirjees, E.H. and Tawfeeq, F.K. 2022. Effect of sodium benzoate on some biochemical, physiological and histopathological aspects in adult male rats. Iraqi J. Vet. Sci. 36(2), 267–272. Al-Badry, F.A. 2023. Histopathological study of brain by different doses of Topiramate intake in male rats. Indian J. Ecol. 50(21), 253–258. Al-Badry, F.A.M. 2024. Adverse histo-physiological damages of increasing consummation of Puma (Super Fat) on female rats. Adv. Anim. Vet. Sci. 12(7), 1335–1340. Alwan, N.A., Kudayer, A.M. and Al-Masoudi, E.A. 2022. The toxic effect of oral gavaged of sodium benzoate (SB) on adult male rabbits. Mal. J. Med. Health Sci. 18, 201–205. Bancroft, J.D. and Gamble, M. 2008. Theory and practices of histological technique. 2nd ed. Churchill Elsevier. London, UK, p: 56. Cantuti, C.I., Shukitt-Hale, B. and Joseph, J.A. 2000. Neurobehavioural aspects of antioxidants in aging. Int. J. Dev. Neurosci. 8, 367–381. Doumas, B.T., Watson, W.A. and Biggs, H.G. 1971. Albumin standards and the measurement of serum albumin with bromocresol green. Clin. Chim. Acta. 31(1), 87–96. El-Shennawy, L., Kamel, M.A.E.N., Khalaf, A.H.Y. and Yousef, M.I. 2020. Dose-Dependent Reproductive Toxicity of Sodium Benzoate in Male Rats: inflammation, Oxidative Stress and Apoptosis. Reprod. Toxicol. 98, 92–98. Hadi, M.M. and Mahdi, W.T. 2019. The effect of sodium benzoate as a preservative on the reproductive system of male rats. Int. J. Res. Sci. 10(1), 98–105. Hadwan, M.H. and Abed, H.N. 2016. Data supporting the spectrophotometric method for the estimation of catalase activity. Data Brief 6, 194–199. Heibashy, M.A. 2000. Hypolipidimia effect of taurine and L-carnine on rats fed a high cholesterol diet. J. Union Arab Biol. Cairo. 46(6), 114–118. Jalil, S.M., Alwan, N.A. and Al-Masoudi, E.A. 2022. Preservative sodium benzoate as a food additive alone and in combination with vitamin c and its toxic effects on the liver and kidney function of adult male rabbits. Bas. J. Vet. Res. 21(S1), 214 -223. Khan, I.S., Dar, K.B., Ganie, S.A. and Ali, M.N. 2022. Toxicological impact of sodium benzoate on inflammatory cytokines, oxidative stress and biochemical markers in male Wistar rats. Drug Chem. Toxicol. 45, 1345–1354. Langlands, H.D., Shoemark, D.K. and Toye, A.M. 2024. Modulation of antioxidant enzyme expression of in vitro culture-derived reticulocytes. Antioxidants 13(9), 1070. Menden, E.E., Boiano, J.M., Murthy, L. and Petering, H.G. 1977. Modification of phenylene diamine oxidase method to permit non-automated ceruloplasmin determination in batches of rat serum or plasma micro samples. Analytical 10, 197–204. Moder, K. 2010. Alternatives to F-test in one way ANOVA in case of heterogeneity of variances (a simulation study). Psychol. Test Assessment Model. 52(4), 343–353. Mohammed, H.J. and Al-Gnami, S.A. 2022. Nanopiperine reduces the toxicity of sodium benzoate on female rat's reproductive system. QJVMS 21(1), 39–46. Mohiuddin, M., Rokeya, B., Al Shoeb, M.A. and Kabir, Y. 2022. Sodium benzoate in locally available soft drinks and its effect on DNA damage and liver function in rats. Dhaka Univ. J. Biol. Sci. 30(3 CSI), 371–383. Muslih, R.K., Al-Nimer, M.S. and Al-Zamely, O.M. 2002. The study level of malondialdehyde after activation with (H2O2 and CuSO4) and inhibition of desferoxamine and molsidomine in the serum of patients with acute myocardial infection. Nat. J. Chem. 5, 139–143. Nair, B. 2001. Final report on the safety assessment of benzyl alcohol, benzoic acid, and sodium benzoate. Int. J. Toxicol. 20, 23–50. Olofinnade, A.T., Onaolapo, A.Y. and Onaolapo, O.J. 2021. Anxiogenic, memory-impairing, pro-oxidant and pro-inflammatory effects of sodium benzoate in the mouse brain. Dusunen Adam J. Psychiatry Neurol. Sci. 34, 14. Osman, B.M., Abdel-Tawab, S.M., Khalaf, M.I., Maher, A., Kamel, M.A. and Shaker, S.A. 2025. The effect of sodium benzoate on the pathway of autophagy in the cardiac tissues of rats. JMRI 46(1), 47–60. Piper, J.D. and Piper, P.W. 2017. Benzoate and sorbate salts: a Systematic review of the potential hazards of these invaluable preservatives and the expanding spectrum of clinical uses for sodium benzoate. Comp. Rev. Food Sci. Food Safty. 16(5), 868–880. Sabour, A.N. and Ibrahim, I.R. 2019. Effect of sodium benzoate on corticosterone hormone level, oxidative stress indicators and electrolytes in immature male rats. Sci. J. Med. Res. 3, 101–106. Seetaramaih, K., Anton Smith, A., Murali, R. and Mana-Valan, R. 2011. Preservatives in Food products - review. Int. J. Phar. Biol. Arch. 2, 583–599. Shahid, M., Alwan, N.A. and Al-Masoudi, E.A. 2018. A study of toxic effect of sodium benzoate, vitamin C alone and their combination on reproductive functions of adult male rabbits. Bas. J. Vet. Res. 17(3), 533–543. Sinha, R. and D'Souza, D. 2012. Intraperitonially induced liver cell damage in Swiss albino mice. Asian J. Med. Clin. Sci. 1(2), 89–91. Wroblewska. 2009. Influence of food additives and contaminants (Nickel and Chromium) on Hypersensitivity and other adverse health reactions - a review. Pol. J. Food Nutr. Sci. 59(4), 287–294. | ||
| How to Cite this Article |
| Pubmed Style Fatima Aziz Mahdi Al-badry. Oxidation-antioxidation biomarkers and histopathological brain damage induced by sodium benzoate exposure. Open Vet. J.. 2025; 15(10): 5355-5360. doi:10.5455/OVJ.2025.v15.i10.52 Web Style Fatima Aziz Mahdi Al-badry. Oxidation-antioxidation biomarkers and histopathological brain damage induced by sodium benzoate exposure. https://www.openveterinaryjournal.com/?mno=256978 [Access: January 26, 2026]. doi:10.5455/OVJ.2025.v15.i10.52 AMA (American Medical Association) Style Fatima Aziz Mahdi Al-badry. Oxidation-antioxidation biomarkers and histopathological brain damage induced by sodium benzoate exposure. Open Vet. J.. 2025; 15(10): 5355-5360. doi:10.5455/OVJ.2025.v15.i10.52 Vancouver/ICMJE Style Fatima Aziz Mahdi Al-badry. Oxidation-antioxidation biomarkers and histopathological brain damage induced by sodium benzoate exposure. Open Vet. J.. (2025), [cited January 26, 2026]; 15(10): 5355-5360. doi:10.5455/OVJ.2025.v15.i10.52 Harvard Style Fatima Aziz Mahdi Al-badry (2025) Oxidation-antioxidation biomarkers and histopathological brain damage induced by sodium benzoate exposure. Open Vet. J., 15 (10), 5355-5360. doi:10.5455/OVJ.2025.v15.i10.52 Turabian Style Fatima Aziz Mahdi Al-badry. 2025. Oxidation-antioxidation biomarkers and histopathological brain damage induced by sodium benzoate exposure. Open Veterinary Journal, 15 (10), 5355-5360. doi:10.5455/OVJ.2025.v15.i10.52 Chicago Style Fatima Aziz Mahdi Al-badry. "Oxidation-antioxidation biomarkers and histopathological brain damage induced by sodium benzoate exposure." Open Veterinary Journal 15 (2025), 5355-5360. doi:10.5455/OVJ.2025.v15.i10.52 MLA (The Modern Language Association) Style Fatima Aziz Mahdi Al-badry. "Oxidation-antioxidation biomarkers and histopathological brain damage induced by sodium benzoate exposure." Open Veterinary Journal 15.10 (2025), 5355-5360. Print. doi:10.5455/OVJ.2025.v15.i10.52 APA (American Psychological Association) Style Fatima Aziz Mahdi Al-badry (2025) Oxidation-antioxidation biomarkers and histopathological brain damage induced by sodium benzoate exposure. Open Veterinary Journal, 15 (10), 5355-5360. doi:10.5455/OVJ.2025.v15.i10.52 |