| Research Article | ||
Open Vet. J.. 2025; 15(10): 5361-5367 Open Veterinary Journal, (2025), Vol. 15(10): 5361-5367 Research Article Age-related serological response to H9N2 infection in southwest Tripoli, LibyaImad Benlashehr1*, Ahmed Shaban Kassem Agha2, Khalid Mohammed Naffati2, Salah Abdulhadi Bshina3, Ahmed Abdulmajed Khashkhosha2, Abdulatif A. Asheg1 and Abdulwahab M. Kammon1,41Department of Poultry and Fish Diseases, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 2Department of Microbiology, Libyan Center for Biotechnology Research, Tripoli, Libya 3Department of Medicine, Faculty of Veterinary Medicine, Azzaytuna University, Tarhuna, Libya 4The National Research Center for Tropical and Transboundary Diseases, Zintan, Libya *Corresponding Author: Imad Benlashehr. Department of Poultry and Fish Diseases, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya. Email: i.benlashehr [at] uot.edu.ly Submitted: 10/05/2025 Revised: 22/08/2025 Accepted: 10/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
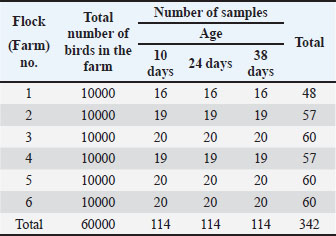
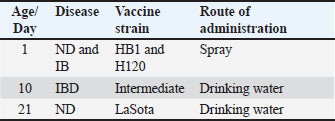
AbstractBackground: Avian influenza virus H9N2 is a low-pathogenic subtype that causes respiratory disease and economic losses in poultry and carries zoonotic potential. The virus is endemic across Asia, the Middle East, and North Africa, often exacerbated by co-infections with other pathogens. In Libya, H9N2 was first detected in 2006, but data on age-related immune responses in broilers remain limited. Aim: This study investigated the age-related serological response to H9N2 in southwest Libyan broiler flocks to provide baseline data for vaccination and control programs. Methods: A total of 342 blood samples were collected from six unvaccinated broiler flocks. Antibodies against influenza A were detected by indirect enzyme-linked immunosorbent assay, and H9N2-specific antibodies were measured by hemagglutination inhibition assay. Results: The seropositivity for influenza A increased with age (30%, 52%, and 100% at 10, 24, and 38 days, respectively). The positivity rates for H9N2 were 0%, 22%, and 72%. The progressive increase confirms the active field circulation and endemicity of H9N2. The absence of detectable antibodies at 10 days indicates strong maternal antibody interference. Conclusion: H9N2 is endemic in Libyan broiler flocks, with continuous exposure reflecting age-dependent seroconversion. Vaccination after the age of 10 days may optimize immune protection by reducing maternal antibody interference. Continuous surveillance and molecular monitoring are essential for guiding effective national control measures and assessing zoonotic risks. Keywords: H9N2, Seroprevalence, ELISA, HI, Libya. IntroductionAIVs belong to the family Orthomyxoviridae, genus Influenza virus A. These viruses cause highly contagious respiratory disease in avian species, particularly aquatic and wild migratory birds (St. Paul et al., 2012; Tong et al., 2013; Swayne et al., 2013). Influenza A viruses consist of 18 hemagglutinin (HA, H1–H18) and 11 neuraminidase (NA, N1–N11) subtypes, with waterfowl and seabirds as their natural reservoirs (Wu et al., 2014). Based on pathogenicity, AIVs are classified as highly pathogenic avian influenza viruses (HPAIVs), including certain H5 and H7 subtypes, which cause severe respiratory symptoms with a high mortality rate. In contrast, low pathogenic avian influenza viruses (LPAIVs), such as H9N2 subtypes, cause mild respiratory symptoms, low mortality rate, and a drop in egg production (Dhama et al., 2005). H9N2 has become endemic in poultry across Asia, the Middle East, and North Africa since the 1990s, resulting in considerable economic losses in the poultry industry. Once introduced, H9N2 is more difficult to eradicate than HPAI viruses (Sagong et al., 2023; Kareche et al., 2024). The pathogenicity of H9N2 can significantly increase in the presence of live attenuated vaccines or when co-infections occur with other pathogens, such as infectious bronchitis virus (IBV), Newcastle disease virus (NDV), and bacteria, such as E. coli and Mycoplasma (Kammon et al., 2015; Ismail et al., 2018; Ellakany et al., 2018; Wang et al., 2021). Vaccination programs have been adopted in several countries, such as China, Korea, Morocco, Pakistan, Egypt, and Iran, to control H9N2 infection in poultry farms and reduce the risk of interspecies transmission (Lee and Song, 2013; Bahari et al., 2015; Kilany et al., 2016; Lau et al., 2016). However, these vaccination programs face limitations due to maternally derived antibodies in chicks and antigenic differences between the vaccine and circulating field strains. These factors significantly contribute to the failure of H9N2 vaccination efforts (Kapczynski and Swayne, 2009; Balish et al., 2010; Cattoli et al., 2011; Capua and Cattoli, 2013; Bahari et al., 2015; Alizadeh et al., 2016; Peacock et al., 2019; Cui et al., 2021). Libyan poultry are exposed to multiple pathogens, including IBV, NDV, Escherichia coli, Salmonella, and Mycoplasma, which can cause high mortality and reduced egg production, either alone or through co-infections (Kammon et al., 2013, 2015, 2017 and 2018; Asheg et al., 2023; Agha et al., 2022; Benlashehr et al., 2024). Serological studies have confirmed the circulation of H9 AIV in broiler and layer flocks in Libya (Fares et al., 2010; Al-Garib et al., 2016). In 2013, field investigations showed that co-infection of H9N2 with NDV was associated with high mortality outbreaks in 2013 (Kammon et al., 2015). More recently, H9N2 was detected in broiler flocks with elevated mortality in southwestern Libya, although the virus alone was unlikely to be the sole cause, suggesting roles for co-infections or management-related stressors (Agha et al., 2023). Despite these findings, information regarding the age-related serological response to H9N2 in broiler flocks in Libya and the monitoring of viral evolution remains limited. Understanding age-related immunity is essential for evaluating flock susceptibility and guiding preventive strategies. Therefore, the present study was conducted to investigate the age-related serological response to H9N2 in broiler flocks in southwest Libya, providing essential baseline data to inform future vaccination and control strategies. Materials and MethodsField surveySix broiler farms located in the southwest region of Libya were included in the current study, having sixty thousand broilers. All flocks were obtained from the same parents. A total of 342 blood samples were randomly collected from the six broiler farms at ages of 10, 24, and 38 days (Table 1). Serum samples were separated and used for indirect enzyme-linked immunosorbent assay (ELISA) and hemagglutination inhibition (HI) tests. All flocks were vaccinated against ND, IB, and IBD based on the program approved by the National Center for Animal Health (NCAH) (Table 2). However, the broilers were not vaccinated against AI. The clinical signs and postmortem lesions were recorded. Table 1. Samples collected from six broiler flocks of different ages.
Table 2. Vaccination program used in the tested farms.
Serological assaysIndirect ELISA to detect antibodies against avian influenza type A was used for the analysis of sera at the NCAH in Tripoli, Libya. The ELISA was conducted according to the manufacturer’s (X-OvO Limited, United Kingdom) instructions. An ELx800 ELISA reader with a 550 nm filter (BIO-TEK Instruments, Inc., Winooski, VT, USA) was used. The sera were also tested for AIV serotype H9N2 using HI test. HA and HI were performed according to the procedure described by the OIE (2009) using 96 “V-shaped” well microtiter plates, 1% v/v blood cells (RBCs), and 4 HA units of AIV H9N2 antigen (OIE, 2009). Statistical analysisData were managed and analyzed using SPSS, version 27.0 (SPSS Inc., Chicago, IL, USA). Analysis was performed using the chi-square test. Statistical significance was designated for differences with p-values less than or equal to 0.05. Ethical approvalAll samples were collected according to local and international standards for the care and use of research animals. ResultsClinical signs and postmortem findingsBroiler flocks included in this study showed mild to moderate respiratory symptoms. The most frequent observations were sneezing, nasal discharge, and mild breathing difficulty. No lesions that could be attributed to a specific respiratory disease were found on postmortem examination. The findings suggest that the signs were nonspecific and could have been influenced by other infections or management-related factors. ELISA assay for influenza A antibodiesSerum samples from six broiler flocks were tested by indirect ELISA at 10, 24, and 38 days of age (Table 3).
Table 3. ELISA assay results.
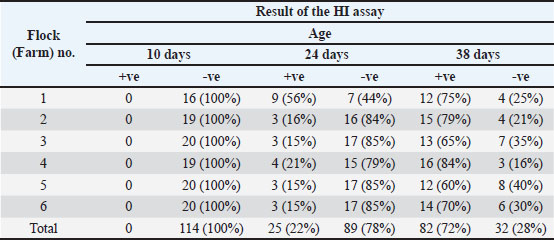
These results confirm that the influenza A virus is circulating in broiler flocks in the study area and that the antibody levels progressively increase as the birds get older. HI assay for H9N2 antibodiesELISA-positive samples were further examined for H9N2-specific antibodies using the HI test (Table 4).
Table 4. Result of the HI assay.
The absence of detectable antibodies at 10 days, followed by their appearance and sharp increase later, suggests a role for maternal antibody interference and supports the view that broilers were exposed to H9N2 virus during the growing period. Overall serological trendsTaken together, the ELISA and HI results show that the prevalence of antibodies increased steadily with age. This pattern is consistent with continuous exposure to influenza viruses under field conditions. The results also provide useful information for vaccination programs, indicating that protective immunity may be more effective when vaccination is performed after the decline of maternal antibodies, usually beyond the first week of age. DiscussionAIVs cause significant economic losses in poultry and pose public health risks. In this study, all broiler flocks tested positive for AIV type A antibodies by ELISA, confirming natural field exposure. These findings align with previous reports from Libya, Jordan, and Lebanon, demonstrating the endemic circulation of AIVs in poultry populations (Al-Natour and Abo-Shehada, 2005; Barbour et al., 2006; Al-Garib et al., 2007). HI testing confirmed the presence of H9N2-specific antibodies in several flocks in southwest Libya, indicating continued circulation of this subtype. Comparable findings from Iran, Tunisia, and Egypt highlight its established endemicity in the region (Pazani et al., 2008; Ghaniei et al., 2013; Nagy et al., 2017; Hassan et al., 2016; Naguib et al., 2017). Some ELISA-positive sera tested negative by HI, suggesting the presence of other AIV subtypes or interference from maternally derived antibodies (de Wit et al., 2004; Barbour et al., 2006). At 10 days of age, no H9N2 antibodies were detected, which is consistent with the high levels of maternal antibodies. By day 24, 22% of birds had detectable antibodies, rising to 72% at day 38. Meanwhile, AIV type A seropositivity increased from 30% to 100% over the same ages. These trends highlight the influence of maternal antibodies on early immune responses and support vaccination after the first week of life to achieve effective seroconversion (Amer et al., 2010; Talat et al., 2020; Pan et al., 2022). The flocks were not vaccinated against AIV, confirming that the observed seropositivity reflected natural infection. However, they were vaccinated against ND, IB, and IBD. Co-infections with these viruses or bacterial pathogens, such as E. coli, can enhance the susceptibility and severity of H9N2 infection (Kammon et al., 2015; Wang et al., 2021; Huang et al., 2024). Molecular studies have also shown that co-infection with H9N2 can suppress key innate immune responses, promoting viral replication (Huang et al., 2017). Although our study confirmed H9N2 circulation serologically, ELISA alone cannot identify specific viral variants or monitor genetic changes. Therefore, future studies will employ PCR and sequencing to characterize circulating H9N2 variants and track viral evolution. This approach will complement serological findings and guide targeted vaccination strategies. Overall, these findings underline the complex ecology of H9N2 in broiler production systems. Effective control requires vaccination strategies timed to the decline of maternal antibodies, improved biosecurity, and ongoing molecular surveillance. Based on age-related serological trends, vaccination after 10 days of age appears optimal for inducing protective immunity while minimizing interference from maternal antibodies. ConclusionOur findings provide clear evidence that the avian influenza virus, particularly H9N2, is endemically circulating in broiler flocks in southwest Libya. In the absence of vaccination, the consistent rise in antibody prevalence with age reflects ongoing natural exposure under field conditions. The role of maternal antibodies in delaying seroconversion and reducing early vaccine effectiveness was evident. On the basis of these dynamics, vaccination after the first 10 days of life appears to be the most appropriate window for inducing protective immunity. Co-infections with other respiratory viruses and bacteria remain an important complicating factor, highlighting the need for integrated vaccination strategies alongside strict biosecurity and regular monitoring. Finally, future work should move beyond serology to include molecular approaches such as polymerase chain reaction (PCR) and sequencing. This will allow the characterization of circulating variants, a better understanding of viral evolution, and informed decision-making for national control programs. AcknowledgmentsThe authors sincerely acknowledge the Libyan Center for Biotechnology Research, the National Center of Animal Health, and the Faculty of Veterinary Medicine, University of Tripoli, for their invaluable support and collaboration. The authors also extend their gratitude to all individuals who contributed directly or provided technical or moral support during sample collection and laboratory work, as their assistance was instrumental in the completion of this study. FundingThe authors did not receive support from any organization for the submitted work (self-fund). Authors’ contributionsImad Benlashehr and Ahmed Shaban Kassem Agha conceptualized the study, designed the experiments, and supervised the project. Ahmed Shaban Kassem Agha and Khalid Mohammed Naffati performed the laboratory experiments and collected the samples. Salah Abdulhadi Bshina and Ahmed Abdulmajed Khashkhosha contributed to the field investigation and sample collection. Khalid Mohammed Naffati and Abdulatif A. Asheg curated and analyzed the data. Imad Benlashehr and Abdulwahab M. Kammon drafted the initial manuscript. Ahmed Shaban Kassem Agha and Khalid Mohammed Naffati reviewed and edited the manuscript. All authors read and approved the final manuscript. Conflict of interestThe authors declare that they have no conflict of interest. Data availabilityThe data are available upon request. ReferencesAgha, A., Benlashehr, I., Naffati, K., Bshina, S. and Khashkhosha, A. 2023. Correlation of avian influenza-H9N2 with high mortality in broiler flocks in the southwest of Tripoli, Libya. Open Vet. J. 13(6), 715–722. Agha, A.S.K., Belgasim, A.A., Nwegi, M.A., Khashkhosha, A.A., Milad, H.S., Alghzewi, H.A., Salem, K.M. and Naffati, K.M. 2022. Evaluation of bacteriological and chemical quality of drinking water used in chicken house in Libya. World J. Adv. Res. Rev. 13(03), 413–420; doi:10.30574/wjarr.2022.13.3.0090 Al-Garib, O.S., Kammon, A., Asheg, A., Hamid, M., Fathalla, H. and Lawala, A. 2007. Detection of antibodies to avian influenza H9N2 in broiler flocks clinically expressing a respiratory disease syndrome. In: Proceedings of the 15th Congress & Exhibition of the World Veterinary Poultry Association. Beijing, China, World Veterinary Poultry Association (WVPA), Vol. 130, p 1–108. Al-Garib, S., Agha, A. and Al-Mesilaty, L. 2016. Low pathogenic avian influenza H9N2: world-wide distribution. World’s. Poult. Sci. J. 72, 125–136. Alizadeh, M., Munyaka, P., Yitbarek, A., Echeverry, H. and Rodriguez-Lecompte, J.C. 2016. Maternal antibody decay and antibody-mediated immune responses in chicken pullets fed prebiotics and synbiotics. Poult. Sci. 96(1), 58–64. Al-Natour, M.Q. and Abo-Shehada, M.N. 2005. Sero-prevalence of avian influenza among broiler-breeder flocks in Jordan. Prev. Vet. Med. 70, 45–50. Amer, M.M., Hamouda, A.S. and EL-Bayomi, K.M. 2010. Studies on maternal antibodies to avian influenza H9N2 vaccine. BS. Vet. Med. J. 6th Sci. Conf. 20(1), 268–274. Asheg, A., Otman, M., Benlashehr, I., Kraim, E., Almashri, R. and Kammon, A. 2023. Prevalence of Salmonella in poultry slaughterhouses located in Tripoli. Libya. Open Vet. J. 13(5), 638–644; doi:10.5455/OVJ. 2023.v13.i5.17 Bahari, P., Pourbakhsh, S.A., Shoushtari, H. and Bahmaninejad, M.A. 2015. Molecular characterization of H9N2 avian influenza viruses isolated from vaccinated broiler chickens in northeast Iran. Trop. Anim. Health &. Prod. 47(6), 1195–1201; doi:10.1007/s11250-015-0848-x Balish, A.L., Davis, C.T., Saad, M.D., El-Sayed, N., Esmat, H., Tjaden, J.A., Earhart, K.C., Ahmed, L.E., El-Halem, M.A., Ali, A.H.M., Nassif, S.A., El-Ebiary, E.A., Taha, M., M. Aly, M., Arafa, A., O’Neill, E., Xiyan, X., Cox, N.J., Donis, R.O. and Klimov, A.I. 2010. Antigenic and genetic diversity of highly pathogenic avian influenza A (H5N1) viruses isolated in Egypt. Avian Dis. 54(s1), 329–334; doi:10.1637/8903-042909-Reg.1 Barbour, E.K., Sagherian, V.K., Sagherian, N.K., Dankar, S.K., Jaber, L.S., Usayran, N.N. and Farran, M.T. 2006. Avian influenza outbreak in poultry in the Lebanon and transmission to neighbouring farmers and swine. Vet. Italiana 42(2), 77–85. Benlashehr, I., Elmasri, K., Kammon, A. and Asheg, A. 2024. Evaluation of Salmonella Enteritidis Isolated from Layer Hens and Murine Fecal Pellets in Poultry Farms of Libya. J. World Poult. Res. 14(2), 211–218. Capua, I. and Cattoli, G. 2013. Prevention and control of highly pathogenic avian influenza with particular reference to H5N1. Virus Res. 178(1), 114–120; doi:10.1016/j.virusres.2013.04.002 Cattoli, G., Milani, A., Temperton, N., Zecchin, B., Buratin, A., Molesti, E., Aly, M.M., Arafa, A. and Capua, I. 2011. Antigenic drift in H5N1 avian influenza virus in poultry is driven by mutations in major antigenic sites of the hemagglutinin molecule analogous to those for human influenza virus. J. Virol. 85(17), 8718–8724; doi:10.1128/JVI.02403-10 Cui, H., De Jong, M.C., Beerens, N., Van Oers, M.M., Teng, Q., Li, L., Li, X., Liu, Q. and Li, Z. 2021. Vaccination with inactivated virus against low pathogenic avian influenza subtype H9N2 does not prevent virus transmission in chickens. J. Virus Eradication 7(3), 100055. de Wit, J.J., Koch, G., Fabri, T.H.F. and Elbers, A.R.W. 2004. A crosssectional serological survey of the Dutch commercial poultry population for the presence of low pathogenic avian influenza virus infections. Avian Pathol. 33(6), 565–570. Dhama, K., Chauhan, R.S., Kataria, J.M., Mahendran, M. and Tomar, S. 2005. Avian Influenza: the Current Perspectives. J. Immunol. Immunopathol. 7, 1–33. Ellakany, H.F., Gado, A.R., Elbestawy, A.R., Abd El-hamid, H.S., Hafez, H.M., Abd El-hack, M.E., Swelum, A.A., Al-Owaimer, A. and Saadeldin, I.M. 2018. Interaction between avian influenza subtype H9N2 and Newcastle disease virus vaccine strain (LaSota) in chickens. BMC Vet. Res. 14, 358. Fares, A.R., Elhafi, G., Alzaletny, R. and Gerish, E. 2010. Seroprevalence of Avian influenza in broilers and layers, Northwestern area of Libya. 27ème Congrès Vétérinaire Maghrébin, Hammamet, Tunisia. pp 10–11. Ghaniei, A., Allymehr, M. and Moradschendi, A. 2013. Seroprevalence of avian influenza (H9N2) in broiler chickens in Northwest of Iran. Asian Pac. J. Trop. Biomed. 10, 822–824; doi:10.1016/S2221-1691(13)60162-8 Hassan, K.E., Shany, S.A.S., Ali, A., Dahshan, A.H.M., El-Sawah, A.A. and El-Kady, M.F. 2016. Prevalence of avian respiratory viruses in broiler flocks in Egypt. Poult. Sci. 95(6), 1271–1280; doi:10.3382/ps/pew068 Huang, Q., Wang, K., Pan, L., Qi, K., Liu, H. and Chen, H. 2017. Co-infection of H9N2 subtype avian influenza virus and infectious bronchitis virus decreases SP-A expression level in chickens. Vet. Microbiol. 203, 110–116. Huang, Q., Yang, X., Zhao, X., Han, X., Sun, S., Xu, C., Cui, N. and Lu, M. 2024. Co-infection of H9N2 subtype avian influenza virus and QX genotype live attenuated infectious bronchitis virus increase the pathogenicity in SPF chickens. Vet. Microbiol. 295, 110163; doi:10.1016/j.vetmic.2024.110163 Ismail, Z.M., El-Deeb, A.H., El-Safty, M.M. and Hussein, H.A. 2018. Enhanced pathogenicity of low-pathogenic H9N2 avian influenza virus after vaccination with infectious bronchitis live attenuated vaccine. Vet. World 11(7), 977–985. Kammon, A., Heidari, A., Dayhum, A., Eldaghayes, I., Sharif, M., Monne, I., Cattoli, G., Asheg, A., Farhat, M. and Kraim, E. 2015. Characterization of avian influenza and Newcastle disease viruses from Poultry in Libya. Avian Dis. 59(3), 422–430. Kammon, A., Monne, I., Asheg, A. and Cattoli, G. 2018. Molecular Detection and Characterisation of Avian Paramyxovirus type 1 in Backyard Chickens and Pigeons in Alzintan City of Libya. Open Vet. J. 8(4), 401–405. Kammon, A., Mulatti, P., Lorenzetto, M., Ferre, N., Sharif, M., Eldaghayes, I. and Dayhum, A. 2017. Biosecurity and geospatial analysis of mycoplasma infections in poultry farms at Al-Jabal Al-Gharbi region of Libya. Open Vet. J. 7(2), 81–85; doi:10.4314/ovj.v7i2.1 Kammon, A., Wei, T.S., Omar, A.R., Dayhum, A., Eldghayes, I. and Sharif, M. 2013. Molecular detection and characterization of infectious bronchitis virus from Libya. World Acad. Sci. Eng. Technol. Int. J. Med. Health Pharm. Biomed. Eng. 7(12), 2634–2637. Kapczynski, D.R. and Swayne, D.E. 2009. Influenza Vaccines for Avian Species. Curr. Topics Microbiol. &. Immunol. 333, 133–152. Kareche, H., Elbohy, O. and Daly, J.M. 2024. Epidemiological situation and spread of avian influenza H9N2 in poultry in North Africa. J. Virol. Sci. 12(1), 10–25. Kilany, W.H., Ali, A., Bazid, A.H.I., El-Deeb, A.H., El-Abideen, M.A.Z., Sayed, M.E. and El-Kady, M.F. 2016. A Dose-Response Study of Inactivated Low Pathogenic Avian Influenza H9N2 Virus in Specific-Pathogen-Free and Commercial Broiler Chickens. Avian Dis. 60(1s), 256–261. Lau, S.Y., Joseph, S., Chan, K.H., Chen, H., Patteril, N.A.G., Elizabeth, S.K., Muhammed, R., Baskar, V., Lau, S.K.P., Kinne, J., Wernery, U. and Woo, P.C.Y. 2016. Complete Genome Sequence of Influenza Virus H9N2 Associated with a Fatal Outbreak among Chickens in Dubai. Genome Announcements 4(4), e00752–e00716. Lee, D.H. and Song, C.S. 2013. H9N2 avian influenza virus in Korea: evolution and vaccination. Clin. Exp. Vaccine Res. 2(1), 26–33. Naguib, M.M., El-Kady, M.F., Lüschow, D., Hassan, K.E., Arafa, A.S., El-Zanaty, A., Hassan, M.K., Hafez, H.M., Grund, C. and Harder, T.C. 2017. New real time and conventional RT-PCRs for updated molecular diagnosis of infectious bronchitis virus infection (IBV) in chickens in Egypt associated with frequent co-infections with avian influenza and Newcastle Disease viruses. J. Virol. Methods 245, 19–27; doi:10.1016/j.jviromet.2017.02.018 Nagy, A., Mettenleiter, T.C. and Abdelwhab, E.M. 2017. A brief summary of the epidemiology and genetic relatedness of avian influenza H9N2 virus in birds and mammals in the Middle East and North Africa. Epidemiolinfection 145(16), 3320–3333; doi:10.1017/s0950268817002576 Pan, X., Su, X., Ding, P., Zhao, J., Cui, H., Yan, D., Teng, Q., Li, X., Beerens, N., Zhang, H., Liu, Q., De Jong, M.C.M. and Li, Z. 2022. Maternal-derived antibodies hinder the antibody response to H9N2 AIV inactivated vaccine in the field. Anim. Dis. 2(1), N2–H9. Peacock, T.P., James, J., Sealy, J.E. and Iqbal, M. 2019. A Global Perspective on H9N2 Avian Influenza Virus. Viruses 11(7), 620. Sagong, M., Lee, K.N., Lee, E.K., Kang, H., Choi, Y.K. and Lee, Y.J. 2023. Current situation and control strategies of H9N2 avian influenza in South Korea. J. Vet. Sci. 24(1), 5; doi:10.4142/jvs.22216 St. Paul, M., Mallick, A.I., Read, L.R., Villanueva, A.I., Parvizi, P., Abdul-Careem, M.F., Nagy, E. and Sharif, S. 2012. Prophylactic treatment with Toll-like receptor ligands enhances host immunity to avian influenza virus in chickens. Vaccine 30, 4524–4531. Swayne, D.E., Suarez, D.L. and Sims, L.D. 2013. Influenza. 13th Ed. In: Diseases of poultry. Eds., Glisson, J.R., Swayne, D.E., McDougald, L.R., Nair, V., Nolan, L.K. and Suarez, D.L. Ames, IA: Wiley-Blackwell, pp: 181–218; doi: 10.1002/9781119421481.ch6 Talat, S., Abouelmaatti, R.R., Almeer, R., Abdel-Daim, M.M. and Elfeil, W.K. 2020. Comparison of the Effectiveness of Two Different Vaccination Regimes for Avian Influenza H9N2 in Broiler Chicken. Animals 10(10), 1875; doi:10.3390/ani10101875 Tong, S., Zhu, X., Li, Y., Shi, M., Zhang, J., Bourgeois, M., Yang, H., Chen, X., Recuenco, S., Gomez, J., Chen, L.M., Johnson, A., Tao, Y., Dreyfus, C., Yu, W., Mcbride, R., Carney, P.J., Gilbert, A.T., Chang, J., Guo, Z., Davis, C.T., Paulson, J.C., Stevens, J., Rupprecht, C.E., Holmes, E.C., Wilson, I.A. and Donis, R.O. 2013. New world Bats Harbor Diverse Influenza A Viruses. PLos Pathogens 9(10), e1003657. Wang, S., Jiang, N., Shi, W., Yin, H., Chi, X., Xie, Y., Hu, J., Zhang, Y., Li, H. and Chen, J.L. 2021. Co-infection of H9N2 Influenza A Virus and Escherichia coli in a BALB/c Mouse Model Aggravates Lung Injury by Synergistic Effects. Front. Microbiol. 12, 670688; doi:10.3389/fmicb.2021.670688 World Organization for Animal Health (OIE). 2009. Chapter 2.3.4. Avian Influenza. In: OIE Terrestrial Manual 2009. World Assembly of Delegates of the OIE. Wu, Y., Wu, Y., Tefsen, B., Shi, Y. and Gao, G.F. 2014. Bat-derived influenza-like viruses H17N10 and H18N11. Trends Microbiol. 22(4), 183–191. | ||
| How to Cite this Article |
| Pubmed Style Benlashehr I, Agha ASK, Naffati KM, Bshina SA, Khashkhosha AA, Asheg AA, Kammon AM. Age-related serological response to H9N2 infection in southwest Tripoli, Libya. Open Vet. J.. 2025; 15(10): 5361-5367. doi:10.5455/OVJ.2025.v15.i10.53 Web Style Benlashehr I, Agha ASK, Naffati KM, Bshina SA, Khashkhosha AA, Asheg AA, Kammon AM. Age-related serological response to H9N2 infection in southwest Tripoli, Libya. https://www.openveterinaryjournal.com/?mno=257324 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.53 AMA (American Medical Association) Style Benlashehr I, Agha ASK, Naffati KM, Bshina SA, Khashkhosha AA, Asheg AA, Kammon AM. Age-related serological response to H9N2 infection in southwest Tripoli, Libya. Open Vet. J.. 2025; 15(10): 5361-5367. doi:10.5455/OVJ.2025.v15.i10.53 Vancouver/ICMJE Style Benlashehr I, Agha ASK, Naffati KM, Bshina SA, Khashkhosha AA, Asheg AA, Kammon AM. Age-related serological response to H9N2 infection in southwest Tripoli, Libya. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5361-5367. doi:10.5455/OVJ.2025.v15.i10.53 Harvard Style Benlashehr, I., Agha, . A. S. K., Naffati, . K. M., Bshina, . S. A., Khashkhosha, . A. A., Asheg, . A. A. & Kammon, . A. M. (2025) Age-related serological response to H9N2 infection in southwest Tripoli, Libya. Open Vet. J., 15 (10), 5361-5367. doi:10.5455/OVJ.2025.v15.i10.53 Turabian Style Benlashehr, Imad, Ahmed Shaban Kassem Agha, Khalid Mohammed Naffati, Salah Abdulhadi Bshina, Ahmed Abdulmajed Khashkhosha, Abdulatif A. Asheg, and Abdulwahab M. Kammon. 2025. Age-related serological response to H9N2 infection in southwest Tripoli, Libya. Open Veterinary Journal, 15 (10), 5361-5367. doi:10.5455/OVJ.2025.v15.i10.53 Chicago Style Benlashehr, Imad, Ahmed Shaban Kassem Agha, Khalid Mohammed Naffati, Salah Abdulhadi Bshina, Ahmed Abdulmajed Khashkhosha, Abdulatif A. Asheg, and Abdulwahab M. Kammon. "Age-related serological response to H9N2 infection in southwest Tripoli, Libya." Open Veterinary Journal 15 (2025), 5361-5367. doi:10.5455/OVJ.2025.v15.i10.53 MLA (The Modern Language Association) Style Benlashehr, Imad, Ahmed Shaban Kassem Agha, Khalid Mohammed Naffati, Salah Abdulhadi Bshina, Ahmed Abdulmajed Khashkhosha, Abdulatif A. Asheg, and Abdulwahab M. Kammon. "Age-related serological response to H9N2 infection in southwest Tripoli, Libya." Open Veterinary Journal 15.10 (2025), 5361-5367. Print. doi:10.5455/OVJ.2025.v15.i10.53 APA (American Psychological Association) Style Benlashehr, I., Agha, . A. S. K., Naffati, . K. M., Bshina, . S. A., Khashkhosha, . A. A., Asheg, . A. A. & Kammon, . A. M. (2025) Age-related serological response to H9N2 infection in southwest Tripoli, Libya. Open Veterinary Journal, 15 (10), 5361-5367. doi:10.5455/OVJ.2025.v15.i10.53 |