| Research Article | ||
Open Vet. J.. 2025; 15(10): 5246-5258 Open Veterinary Journal, (2025), Vol. 15(10): 5246-5258 Research Article Lithium reduces cerebral ischemia/reperfusion injury in dexamethasone-treated rats: The role of the PI3K/Akt pathwayMohamed M. Askar*, Islam A. A. E.-H. Ibrahim and Mona F. MahmoudDepartment of Pharmacology and Toxicology, Faculty of Pharmacy, Zagazig University, Zagazig, Egypt *Corresponding Author: Mohamed M. Askar. Department of Pharmacology and Toxicology, Faculty of Pharmacy, Zagazig University, Zagazig, Egypt. Email: m.m.askar86 [at] gmail.com; m.askar22 [at] pharmacy.zu.edu.eg Submitted: 10/05/2025 Revised: 15/08/2025 Accepted: 01/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: Insufficient blood flow to the brain or loss of vascular integrity and bleeding within the brain tissue can damage neuronal cells and cause stroke. Aim: This study aimed to investigate the possible protective effect of lithium against cerebral ischemia reperfusion injury (CIRI) in dexamethasone-treated rats by focusing on changes in the phosphatidylinositol-3 kinase (PI3K)/protein kinase B (Akt) pathway. Methods: Dexamethasone (10 mg/kg) was intraperitoneally injected into Wistar albino rats for 5 days. On the fifth day of the experiment, CIRI was triggered by bilateral carotid artery occlusion for one hour, followed by reperfusion for 1 hour. Three groups were investigated: sham, CIRI, and CIRI + Li groups. Rats in the CIRI + Li group were treated with lithium (50 mg/kg) via intraperitoneal injection 30 minutes before the induction of cerebral ischemia. At the end of the experiment, the serum blood glucose levels and body weights were measured. The levels of malondialdehyde, myeloperoxidase, glutathione, nuclear factor erythroid 2, heme oxygenase-1, and superoxide dismutase and Akt in the cerebral tissue were recorded. PI3K, phosphorylated Akt (Ser473), phosphorylated glycogen synthase kinase 3β (Ser9), and silent mating-type information regulation 2 homolog 1 levels were measured. Furthermore, histopathological and P-Akt (Ser473) immunohistochemical investigations of cerebral tissues were performed. Results: CIRI suppressed the PI3K/Akt pathway and increased oxidative stress markers in dexamethasone-treated rats. Lithium pre-injection ameliorated all pathological changes observed with CIRI and upregulated the PI3K/Akt pathway. Conclusion: Lithium can protect against CIRI even in the presence of metabolic disturbances, such as those induced by high-dose dexamethasone. The ameliorative effects of lithium were associated with PI3K/Akt pathway upregulation and oxidative stress downregulation. Keywords: Lithium, Cerebral ischemia, Phosphoinositide 3-kinase, Dexamethasone. IntroductionInsufficient blood flow to the brain or loss of vascular integrity and bleeding within the brain tissue can damage neuronal cells and cause stroke (Turc et al., 2023). Stroke is the second leading cause of mortality worldwide (Bonkhoff et al., 2021). Approximately 13.7 million new stroke cases and 5.5 million related deaths occur annually (Feigin et al., 2019). The incidence of stroke in Egypt is approximately 0.963%, which is approximately 150,000–210,000 yearly cases, according to Aref et al. (2021). Insufficient blood flow to the brain or brain ischemic injuries can be caused by a sudden blockage in one or more cerebral arteries or by cerebral hypoperfusion secondary to a severe drop in blood pressure. Notably, arteriosclerosis of the cerebral vessels is one of the leading causes of ischemic injury and represents approximately 50% of the recorded cases. Cardiogenic cerebral infarction, vasculitis, and extracranial arterial dissection may be other causes. Cerebral stroke is classified as focal or multifocal, triggered by complete closure or noticeable narrowing of the artery supplying a part of the brain (Formisano et al., 2020). Currently, a limited number of pharmacological interventions, such as aspirin, statins, and β-blockers, can be used to protect against cerebral ischemic injuries before cardiac and cerebral surgical operations. In the same context, well-defined pharmacological interventions, such as aspirin and alteplase (tPA, recombinant tissue plasminogen activator), are used to treat acute ischemic injuries. Alteplase is considered the cornerstone of ischemic brain management. Although tPA may be lifesaving, it may cause severe hemorrhage. Furthermore, this approach is only suitable for 10% of stroke cases. Therefore, there is a continuous need to find new therapeutic alternatives. The activation of the phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt) pathway is considered a critical factor in preventing neuronal injury (Zhu et al., 2013). Cellular survival and metabolism in mammals are regulated via PI3K signaling (Bellacosa et al., 1998). During ischemic insult, the level and activity of PI3K and Akt in the brain decrease, leading to increased GSK3β activity and apoptotic signal stimulation (Endo et al., 2006; Sasaki et al., 2006; Pang et al., 2016; Yang et al., 2016; Ruan et al., 2021). Silent mating type information regulation 2 homolog 1 (SIRT1) is a histone-deacetylating protein. Previous studies have shown that Sirt1 upregulation has a protective effect against ischemic insults (Yang et al., 2013) and that SIRT1 expression decreases in ischemic brain mice (Wang et al., 2020). Lithium is a monovalent cation with mood-stabilizing properties. Therefore, it is used for the treatment of bipolar disorders (BDs), a disease that results in disturbed neuronal development (Chiu et al., 2013; Diniz et al., 2013). Preclinical and clinical studies have shown lithium-mediated neuroprotective and neurotrophic effects (Forlenza et al., 2014), suggesting that lithium is a potential agent for the management of acute (e.g., stroke) or prolonged (e.g., Alzheimer’s, Parkinson’s, and Huntington’s disease) neurodegenerative diseases (Forlenza et al., 2014). Notably, the PI3K/Akt pathway mediates several beneficial effects of lithium, including metabolic and other cytoprotective effects. Dexamethasone, a synthetic glucocorticoid, is used in research to simulate metabolic syndrome due to its profound effects on metabolic processes. According to previous studies, dexamethasone exposure can result in insulin resistance, hyperglycemia (Oche et al., 2023), and dyslipidemia (Bowles et al., 2015)—all of which are characteristics of the metabolic syndrome. It is noteworthy that metabolic syndrome is a risk factor for cerebrovascular disorders, especially ischemic stroke (Holay, 2015). Although the neuroprotective effects of lithium have been explored in stroke models, its impact under glucocorticoid-induced metabolic stress has not been investigated. We hypothesized that lithium pretreatment can mitigate cerebral ischemia reperfusion injury (CIRI) via modulation of the PI3K/Akt signaling pathway and oxidative stress response in rats with dexamethasone-induced metabolic syndrome. Therefore, this study aimed to investigate the possible neuroprotective impact of lithium on CIRI in dexamethasone-treated rats, focusing on the role of the PI3K/Akt pathway. Materials and MethodsAnimalsAdult male Wistar albino rats (185 ± 18 g, 2 months old) were obtained from the Faculty of Veterinary Medicine, Zagazig University, Egypt. Rats were housed in suitable plastic cages with wood shavings as bedding material within the animal care facility at the Faculty of Pharmacy, Zagazig University. The animals were maintained under regulated environmental conditions, including a temperature of (23°C ± 2°C) and humidity (60% ± 10%) and a 12-h/12-h light/dark cycle. Rats were acclimatized for at least 15 days prior to the experiments and had free access to a pellet chow diet and water. DrugsLithium chloride and dexamethasone were purchased from AMRIYA Co. (10th of Ramadan, Egypt) and Sigma-Aldrich (Munich, Germany), respectively. Experimental designAfter acclimatization, rats received intraperitoneal dexamethasone (10 mg/kg/day) (Mahendran and Devi, 2001; Barel et al., 2010) for 5 days (Saad et al., 1993) to induce metabolic syndrome (Askar et al., 2022). Rats were randomly distributed into three experimental groups: sham, disease, and lithium treatment groups (n=6 each). Rats in the sham group were subjected to an identical surgical procedure, but the carotid arteries were not occluded, whereas rats in the disease group were subjected to a surgical procedure including bilateral carotid artery blockage. In the lithium treatment group, rats intraperitoneally received lithium (50 mg/kg) (Sandoughdaran et al., 2016) 30 minutes before ischemia induction (Fig. 1a).
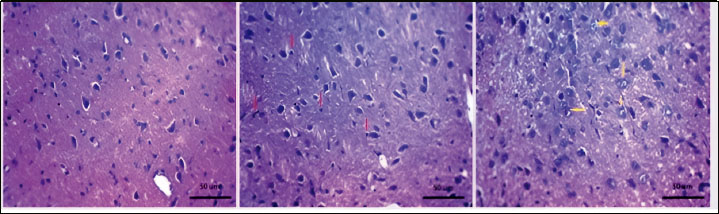
Fig. 1. Experiment timeline and effect of dexamethasone on body weight and blood glucose level. (a) Diagram of the experimental timeline. Dexa: A dose of 10 mg/kg dexamethasone was injected intraperitoneally for 5 days. Before DEXA: day zero. After DEXA: day 5 Graphical presentation of body weight (b) and blood glucose levels (c). Statistical analysis was performed using the paired t-test. Values are presented as mean ± SE (n=6 per group). *p < 0.05. Surgical procedure for bilateral occlusion of common carotid arteriesRats were anesthetized via intraperitoneal administration of thiopental (30 mg/kg) (Wang-Fischer, 2009). Lidocaine HCl (2%; 0.1–0.2 ml) (Eappen and Kissin, 1998) was injected into the surgical area before initiating treatment to prevent thiopental-associated hyperalgesia (Archer et al., 1994). The bilateral common carotid arteries were exposed and ligated through a midline incision in the anterior neck area to prevent cerebral blood flow. This occlusion was applied for 1 hour, followed by reperfusion for another hour. During the surgery, the animals’ temperature was maintained at 37°C using a heating blanket. Body weight and blood glucose level determinationRat body weight and postprandial blood glucose levels were measured before and five days after dexamethasone injection. A drop of blood from the tail vein was applied to ACCU-CHEK active strips®, and blood glucose levels were measured using a glucometer (Roche, Switzerland). Blood and tissue samplesAt the end of the experiment, the rats were euthanized by decapitation, and trunk blood was collected from the area of decapitation. Serum samples were separated by centrifugation (4,000 rpm for 15 minutes at 4°C), then stored for subsequent analyses at −80°C. The brains were also collected and sectioned longitudinally into two halves; one half was fixed via formalin for further histological and immunohistochemical analysis, while the other half was rapidly frozen in liquid nitrogen and stored at −80°C for further biochemical assessments. Histopathological examinationBrain tissue was fixed with 10% formalin, dehydrated with ethanol gradient, cleared with xylene, and finally implanted in paraffin blocks. These blocks were sectioned at 5 μm thickness in a coronal plane pattern and stained with hematoxylin and eosin (H&E). Immunohistochemical studyImmunohistochemical analysis was conducted using the avidin-biotin-peroxidase method, following the procedure outlined by Butler et al. (2019) and Askar et al. (2022). The monoclonal primary antibody P-Akt (Ser473) (D9E) XP® Rabbit mAb catalog No. 4060; Cell Signaling, Danvers, USA) was used to localize the Ser473-phosphorylated form of P-Akt in the analyzed tissues. Immunohistochemical staining for P-Akt revealed brownish discoloration of the cytoplasm. The immunostained segments were morphometrically examined. The ratio of P-Akt-related immune response area was measured in cerebral cortex sections from three rats in every group at 100× magnification utilizing ImageJ v.1.51d version (NIH & LOCI, Wisconsin University, USA). In brief, stain-positive (brown colored area) samples were selected, masked by red binary coloring, and measured relative to a standard measurement frame. Measurement of biochemical changes in brain tissuesMalondialdehyde (MDA), myeloperoxidase (MPO), glutathione (GSH), nuclear factor erythroid 2 (Nrf-2), and heme oxygenase 1 (HO-1) levels and superoxide dismutase (SOD) activity were determined using colorimetric kits supplied by BioVision, USA (Cat. No. #K739, #E4581, #K264, #nwknrf2rev070116, #E4525, and #K335, respectively). All procedures and processing were executed according to the manufacturer’s instructions. The levels of phosphoinositide 3-kinase (PI3K), phosphorylated glycogen synthase kinase 3β (P-GSK3β) (Ser9), silent mating-type information regulation 2 homolog 1 (SIRT1), and P-Akt (Ser473) were determined using enzyme-linked immunosorbent assay kits supplied by MyBiosource San Diego, USA, Cat. No. (MBS260381, MBS730623, and MBS2023434), and SigmaAldrich (Cat. No. RAB0012), respectively. Collagen expression measurement in brain tissuesTotal RNA was extracted by homogenizing brain tissue using an RNeasy cleansing agent (Qiagen, Valencia, CA). cDNA was produced from (5 μg) of entire RNA removed with 1 μl (20 pmol) of antisense primer and (0.8 μl) of superscript AMV reverse transcriptase at 37°C for 60 minutes. For the PCR process, cDNA (4 μl) was incubated with water (30.5 μl), MgCl2 (4 μl of 25 mM), dNTPs (1 μl of 10 mM), 10×PCR buffer (5 μl), Taq polymerase [0.5 μl (2.5 U)], and (2.5 μl) of each primer, for a total of 10 pmol. The primer sequences used for collagen measurement were as follows: forward, 5′-GAACTTGGGGCAAGACAGTCA-3′; reverse, 5′-GTCACGTTCAGTTGGTCAA-3′ (UniGene Rn.2953). The reaction mixture was exposed to 40 cycles of PCR amplification: denaturation (1 minute at 95°C), annealing (1 minute at 67°C), and extension (2 minutes at 72°C). A standard control sample was used to measure gene expression. The RQ of each target gene was measured according to the calculation of the delta-delta Ct (ΔΔCt) value. The RQ of the genes was calculated using the 2-∆∆ method. Statistical analysisAll data are presented as mean ± SEM. Means were compared using one-way analysis of variance followed by Bonferroni post hoc correction for selected pairs using GraphPad Prism v. 7 (GraphPad Software, Inc., USA). A p < 0.05 was considered statistically significant. Ethical approvalAll experimental procedures were conducted in compliance with both national and international regulations regarding the care and use of laboratory animals. The study received approval from the Institutional Animal Care and Use Committee of Zagazig University (ZU-IACUC) and was assigned the approval number ZU-IACUC/3/F/108/2019. ResultsDexamethasone impact on body weight and blood glucose concentrationsAs shown in Fig. 1, the body weight of the rats after dexamethasone injection (10 mg/kg, IP, 5 days) was significantly lower (141.83 ± 6.31 vs. 162 ± 7.73 g; Fig. 1b). On the other hand, compared with the original level, intraperitoneal injection of dexamethasone (10 mg/kg, 5 days) significantly increased blood glucose levels (157.75 ± 6.98 is 119 ± 6.18 mg/dl; Fig. 1c). Effect of lithium on brain tissue histopathology in dexamethasone-treated rats exposed to cerebral ischemia–reperfusion injuryAs shown in Fig. 2, the sham group exhibited nearly normal neuronal structure of the cerebral cortex. In contrast, in the cerebral ischemia reperfusion group, astrocytes with small, darkly stained pyknotic nuclei were observed within the fibrillary background, reflecting apoptosis and damage (red arrow; Fig. 2). In contrast, astrocytes with large vesicular nuclei and prominent nucleoli were observed in the lithium group, reflecting the potent neuroprotective effects of lithium (yellow arrow, Fig. 2).
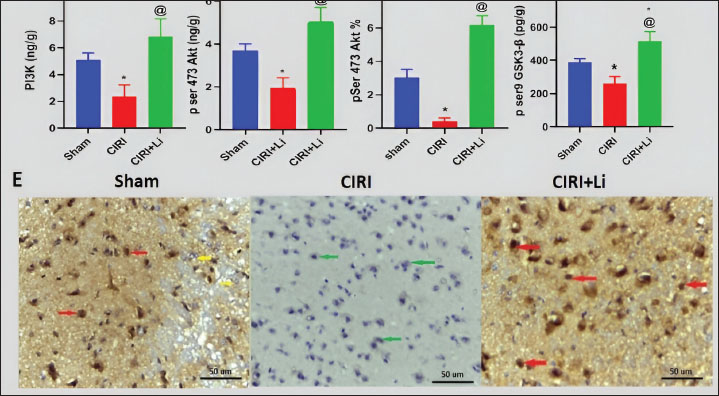
Fig. 2. Changes in brain histopathology. Photomicrographs of cerebral cortex tissue stained with H&E (400×). All groups received a daily dose of 10 mg/kg dexamethasone intraperitoneally for 5 days. CIRI: cerebral ischemia-reperfusion injury (60 minutes of ischemia/60 minutes of reperfusion). The sham group: Rats were exposed to the same procedure as CIRI except for vessel occlusion. In the CIRI + Li group, rats were exposed to the same procedure as CIRI plus lithium (50 mg/kg/IP) 30 minutes before cerebral ischemia induction. Statistical analysis was conducted using one-way analysis of variance along with the Bonferroni post hoc test for specific pairs. The results are displayed as the means ± SEs (n=3 per group). *p < 0.05 compared with the sham group. [at] p < 0.05 compared with the CIRI group. The red arrow indicates apoptosis and damage, while the yellow arrow indicates astrocytes with large vesicular nuclei. Effect of lithium on brain PI3K levels and p ser473 Akt and p ser9 GSK3-β in dexamethasone-treated rats exposed to CIRIAs illustrated in Fig. 3, cerebral ischemia-reperfusion following the intraperitoneal administration of dexamethasone (10 mg/kg) for 5 days significantly decreased the cerebral cortex PI3K protein level (2.33 ± 0.52 vs. 5.10 ± 0.31 ng/g, (54%); Fig. 3a), phosphorylated Akt protein at ser473 (1.93 ± 0.28 vs. 3.67 ± 0.2 ng/g, (47%); Fig. 3b), P-AKT immunostaining (86%) (Fig. 3c,e), and phosphorylated GSK3-β protein at ser 9 (261.33 ± 24.63 vs. 302.33 ± 24.33 pg/g, (32%); Fig. 3d).
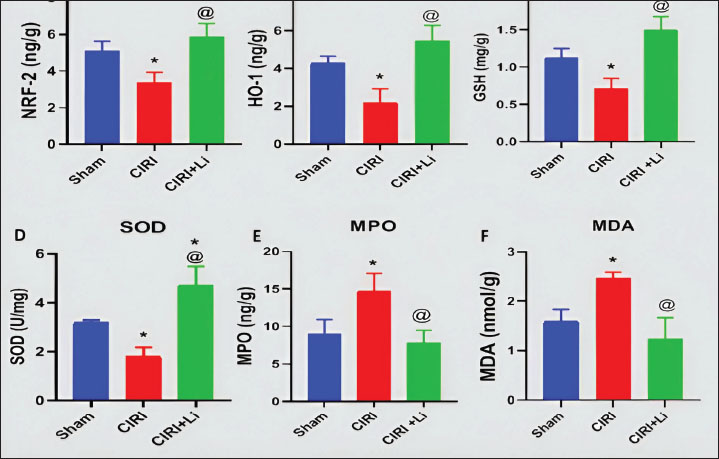
Fig. 3. Changes in the brain PI3K/Akt/GSK3-β pathway. Graphical representation of PI3K (a), phospho-Ser473-Akt (b), the % immune-stained area of phospho-Ser473-Akt (c), and phospho-Ser9-GSK3β (d). Photomicrographs of phospho-S473-Akt-immunostained cerebral cortex tissue sections (400X) (e). All groups received a daily dose of 10 mg/kg dexamethasone intraperitoneally for 5 days. CIRI: cerebral ischemia-reperfusion injury (60 minutes of ischemia/60 minutes of reperfusion). The sham group: Rats were exposed to the same procedure as CIRI except for vessel occlusion. In the CIRI+ Li group, rats were exposed to the same procedure as CIRI plus lithium (50 mg/kg/IP) 30 minutes before cerebral ischemia induction. Statistical analysis was performed using one-way analysis of variance and the Bonferroni post hoc test for selected pairs. Values are presented as the means ± SEs (n=3 per group). *p < 0.05 compared with the sham group. [at] p < 0.05 compared with the CIRI group. The red arrows indicate positive pAKT immunostaining, whereas the green and yellow arrows indicate negative pAKT immunostaining. On the other hand, pretreatment with lithium significantly increased cerebral cortex PI3K (293%, Fig. 3a), phospho-Akt (Ser473) (261%, Fig. 3b & 1460%, Fig. 3c,e), and phospho-GSK3β (ser9) (197%, Fig. 3d) levels compared with those in the cerebral ischemia reperfusion group. Effect of lithium on brain oxidative stress in dexamethasone-treated rats with cerebral ischemia–reperfusion injuryCerebral ischemia reperfusion after intraperitoneal administration of dexamethasone at a dosage of 10 mg/kg over a period of 5 days significantly decreased the levels of Nrf2 (3.40 ± 0.31 vs. 5.14 ± 0.29 ng/g, Fig. 4a), HO-1 (2.23 ± 0.41 vs. 4.29 ± 0.21 ng/g, Fig. 4b), GSH (0.72 ± 0.08 vs. 1.13 ± 0.07 mg/g, Fig. 4c), and SOD activity (1.83 ± 0.2 vs. 3.21 ± 0.05 U/mg, Fig. 4d), while significantly increased cerebral cortex content of MPO (14.77 ± 1.33 vs. 9.07 ± 1.07 ng/g, Fig. 4e) and MDA (2.47 ± 0.07 vs. 1.60 ± 0.14 nmol/g, Fig. 4f) compared with the sham group.
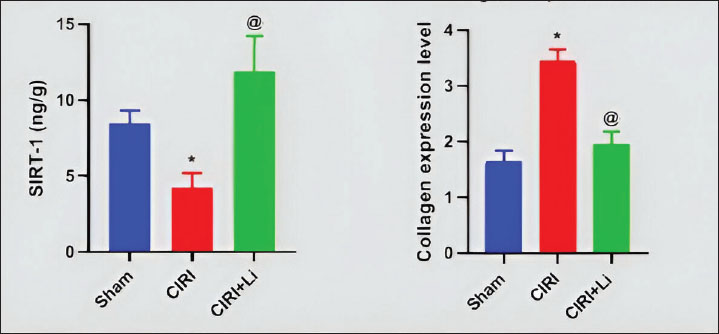
Fig. 4. Changes in oxidative stress markers in the brain. Graphical representation of Nrf-2 (a), HO-1 (b), GSH (c), SOD (d), MPO (e), and MDA (f) levels. All groups received a daily dose of 10 mg/kg dexamethasone intraperitoneally for 5 days. CIRI: cerebral ischemia-reperfusion injury (60 minutes of ischemia/60 minutes of reperfusion). The sham group: Rats were exposed to the same procedure as CIRI except for vessel occlusion. In the CIRI + Li group, rats were exposed to the same procedure as CIRI plus lithium (50 mg/kg/IP) 30 minutes before cerebral ischemia induction. Statistical analysis was performed using one-way analysis of variance and the Bonferroni post hoc test for selected pairs. Values are presented as the means ± SEs (n=3 per group). *p < 0.05 compared with the sham group. [at] p < 0.05 compared with the CIRI group. On the other hand, preLi injection caused a significant increase in the cerebral cortex levels of Nrf-2 (172%, Fig. 4a), HO-1 (245%, Fig. 4b), and GSH (208%, Fig. 4c) and the activity of SOD (258%, Fig. 4d) compared with those in the cerebral ischemia reperfusion group. In addition, the cerebral cortex MPO (47.19%, Fig. 4e) and MDA (49.79%, Fig. 4f) levels in lithium-treated rats were significantly lower than those in the cerebral ischemia reperfusion group. Effect of lithium on brain sirtuin-1 levels in dexamethasone-treated rats exposed to cerebral IRICerebral ischemia reperfusion after intraperitoneal injection of dexamethasone (10 mg/kg) for 5 days significantly decreased SIRT-1 levels (4.2 ± 0.58 vs. 8.43 ± 0.5 ng/g; Fig. 5a) compared with those in the sham group.
Fig. 5. Changes in SIRT1 and collagen levels in the brain. Graphical representation of (a) sirtuin-1 levels and (b) collagen expression. All groups received a daily dose of 10 mg/kg dexamethasone intraperitoneally for 5 days. CIRI: cerebral ischemia-reperfusion injury (60 minutes of ischemia/60 minutes of reperfusion). The sham group: Rats were exposed to the same procedure as CIRI except for vessel occlusion. In the CIRI + Li group, rats were exposed to the same procedure as CIRI plus lithium (50 mg/kg/IP) 30 minutes before cerebral ischemia induction. Statistical analysis was performed using one-way analysis of variance and the Bonferroni post hoc test for selected pairs. Values are presented as the means ± SEs (n=3 per group). *p < 0.05 compared with the sham group. [at] p < 0.05 compared with the CIRI group. Conversely, compared with cerebral ischemia reperfusion, preinjection of lithium significantly increased SIRT-1 protein levels (283%, Fig. 5a). Effect of lithium on the expression of brain collagen in dexamethasone-treated rats exposed to cerebral ischemia–reperfusion injuryCerebral ischemia‒reperfusion after intraperitoneal injection of dexamethasone (10 mg/kg) for 5 days caused a significant increase in cerebral cortex collagen expression (3.45 ± 0.12 vs. 1.64 ± 0.11; Fig. 5b). In contrast, preinjection of lithium significantly decreased cerebral cortex collagen expression (44%, Fig. 5b) compared with that in the cerebral ischemia‒reperfusion group. DiscussionThe incidence of stroke and its associated neurological complications is increasing annually, constituting a high economic and health burden on people and governments. Notably, the risk factors for stroke include diabetes (Feigin and Brainin, 2022), hypertension, hypercholesterolemia, obesity, and atherosclerosis (Yang et al., 2017), which are considered the main risk factors for metabolic syndrome. Therefore, this study aimed to investigate the neuroprotective effects of lithium against cerebral ischemia‒reperfusion injury in dexamethasone-treated rats, focusing on the role of the PI3K/Akt pathway. The dexamethasone-induced metabolic syndrome model was used because it is a well-established experimental model characterized by a short period of induction (Askar et al., 2022). Body weight changes and glycemic abnormalities were confirmed in the present study, as dexamethasone injection significantly reduced body weight. Fasting blood glucose levels increased compared with preinjection values, reflecting impaired glucose metabolism. The reduction in body weight may be attributed to dexamethasone-induced muscle atrophy (Jhuo et al., 2023). However, elevated blood glucose levels may be related to dexamethasone-induced impairment of glucose metabolism, insulin resistance, and pancreatic damage (Mahmoud et al., 2022; da Silva et al., 2023). In this study, CIRI was induced in rats treated with dexamethasone. Our results showed that astrocytes in the cerebral cortex of rats subjected to CIRI have small, darkly stained pyknotic nuclei within the fibrillary background, reflecting apoptosis and damage, and confirming the successful induction of CIRI. In addition, the present study showed significant decreases in PI3K and Ser473 phospho-Akt levels in the cerebral cortex of rats exposed to CIRI compared with those in the sham group. The PI3K/Akt pathway is a key regulator of several cellular functions, including survival, metabolism, and proliferation (Bellacosa et al., 1998). PI3K activates Akt by mediating its phosphorylation at Ser473 (Vivanco and Sawyers, 2002). In nerve cells, the PI3K/Akt pathway has been demonstrated to possess neuroprotective properties on cortical neurons in a glutamate-induced neuronal injury model (Gao et al., 2015). In addition, PI3K/Akt activation suppresses neuronal damage (Hetman et al., 1999) and promotes poststroke neuroprotective activity (Endo et al., 2006). In a similar context, the expression level of PI3K has been observed to diminish in a microglial cell line following exposure to an ischemic insult (Ruan et al., 2021). Conversely, the suppression of Akt activity may reduce the neuroprotective benefits associated with preconditioning ischemia (Fukunaga and Kawano, 2003). GSK3-β is a versatile serine-threonine kinase that is especially prevalent in the CNS (Leroy and Brion, 1999). Akt can phosphorylate GSK3-β at the serine 9 (Ser9) residue and block its activity, thereby promoting resistance to apoptotic stimuli (Endo et al., 2006). In contrast, GSK3-β phosphorylation at the tyrosine 216 residue, which increases GSK3-β activity, has been found to increase after middle cerebral artery occlusion, mediating neuronal injury (Pang et al., 2016). Conversely, the inhibition of GSK3-β activity through Akt phosphorylation facilitates neuroprotective effects against glutamate-induced neural apoptosis (Cross et al., 1995). Consistent with these findings, the current study demonstrated notable reductions in Ser9 phospho-GSK3-β levels within the cerebral cortex of rats subjected to CIRI compared with those in the sham group. Nrf2 is a crucial transcription factor that promotes the transcription of genes that harbor antioxidant response elements, such as HO-1 and glutathione peroxidase (Milani et al., 2013; Gan and Johnson, 2014). Notably, the PI3K/Akt/GSK3-β pathway may organize Nrf2 activity. The activation of GSK3-β promotes Fyn kinase, leading to nuclear export and subsequent Nrf2 degradation (Niture et al., 2014). In the same context, the downregulation of Nrf2 is associated with heightened neuronal injury (Alural et al., 2015). Our findings indicated a notable decrease in Nrf2 levels within the cerebral cortex of rats subjected to CIRI when compared with those in the sham group. HO-1 is a widely present redox-sensitive enzyme that provides beneficial effects, including anti-inflammatory and antioxidant properties. The anti-inflammatory effects of HO-1 have been attributed to its carbon monoxide product, which is generated by heme degradation (Lee et al., 2003; Kao et al., 2008). It also plays a role in the transformation of heme into BV (Chen, 2014). HO-1 is generated and promoted via Nrf2 (Satoh et al., 2013). This antioxidant protein is essential for mitigating neuronal damage and preventing cell death, as well as protecting against excitotoxicity and subarachnoid hemorrhage (Chen, 2014). Similarly, HO-1 expression is decreased in the cerebral cortex of rats with temporal middle cerebral artery occlusion (Pang et al., 2016; Wang et al., 2022). Consistent with previous research, the current study demonstrated a notable reduction in HO-1 levels within the cerebral cortex of rats subjected to CIRI compared with those in the sham group. Furthermore, GSH plays a crucial role in preserving cellular redox balance, facilitating conjugation and detoxification processes, and neutralizing free radicals and electrophilic intermediates (Savolainen et al., 1998; Zhang et al., 2013; Pang et al., 2016). Notably, activation of GSK3-β during transient middle cerebral artery occlusion has been found to decrease Nrf2 and, subsequently, GSH levels (Rojo et al., 2008; Pang et al., 2016). Nrf2 initiates the transcription of glutamate cysteine ligase, a key enzyme that regulates the rate of GSH biosynthesis. Consistent with earlier research, our investigation revealed a notable reduction in GSH levels within the cerebral cortex of rats subjected to CIRI compared to the sham group. SOD is a mitochondrial antioxidant enzyme that can scavenge free radicals and convert superoxide into the less reactive form of hydrogen peroxide (Yang et al., 2011; Zhang et al., 2013). Notably, middle cerebral artery occlusion decreases SOD activity (Pang et al., 2016). Moreover, the Nrf2/HO-1 pathway is an essential regulator of SOD activity (Zhao et al., 2022). Consistent with the results of earlier research, we observed a notable reduction in SOD activity within the cerebral cortex of rats subjected to CIRI compared with that in the sham group. In the same context, MPO is an inflammatory enzyme involved in the generation of reactive oxygen species. The main sources of MPO are infiltrated neutrophils, activated microglia, neurons, and astrocytes within the ischemic brain (Chen et al., 2020). Moreover, MPO facilitates the synthesis of HOCl, which exhibits significant diffusivity and oxidative properties, resulting in the peroxidation of proteins, lipids, and DNA molecules, as well as the attraction of inflammatory cells to the ischemic region (Üllen et al., 2013). Consistent with earlier research, our results indicated a significant rise in MPO levels within the cerebral cortex of rats subjected to CIRI compared with that in the sham group. Moreover, MDA is among various low-molecular-weight end products generated through lipid peroxidation (Janero, 1990) during stress conditions such as middle cerebral artery occlusion (Du et al., 2020; Oztanir et al., 2022; Wang et al., 2022). Our research demonstrated a significant rise in MDA levels within the cerebral cortex of rats subjected to CIRI compared with the sham group. The silent mating type information regulation 2 homolog 1 (SIRT1) protein is a histone deacetylase that is extensively expressed in various tissues (Dali-Youcef et al., 2007). SIRT1 has a crucial physiological role in the brain, as it influences brain structure at several levels, including axon growth through the Akt/GSK3-β pathway (Koronowski and Perez-Pinzon, 2015), in addition to protecting neurons from ischemic insults (Yang et al., 2013). Previous studies have demonstrated that SIRT1 overexpression increases cell viability and decreases apoptosis and proinflammatory cytokine release. SIRT1 is crucial for maintaining glucose homeostasis and regulating fat metabolism (Elibol and Kilic, 2018). Knockdown or inhibition of p53 activity enhances the activation of Sirt1 (Poulsen et al., 2014). Conversely, the activation of GSK3-β reduces the expression of SIRT1 (Xu et al., 2021). In the same context, Sirt1 significantly suppresses myofibroblast differentiation and collagen synthesis in experimental lung fibrosis models (Chu et al., 2018). Our results indicated a notable decrease in SIRT1 levels within the cerebral cortex of rats subjected to CIRI when compared with those in the sham group. Collagen is a structural protein that maintains integrity and provides mechanical support for body tissues (Friess, 1998). It is also involved in cell attachment, migration, proliferation, and differentiation (Ricard-Blum, 2011). Collagen is a fundamental element of the extracellular matrix and is predominantly synthesized by fibroblasts. Fibroblast-like cells have been discovered in the meninges and perivascular Virchow–Robin spaces in both humans and mice (Xu and Yao, 2021). Moreover, collagen concentrations in brain tissue generally increase in a time-dependent fashion following permanent occlusion of the middle cerebral artery (Michalski et al., 2020). Consistent with earlier research, this study demonstrated a markedly increased collagen expression in the cerebral cortex of rats subjected to CIRI compared with the sham group. Lithium chloride is a monovalent ion known for its mood-stabilizing effects and is commonly used in the treatment of BD (Diniz et al., 2013). Numerous preclinical and clinical investigations have documented the neuroprotective and neurotrophic properties of lithium, indicating that lithium represents a promising approach for the short-term treatment of cerebral ischemia (Forlenza et al., 2014). Lithium has a remarkable effect on cells. It appears to be designed to activate multiple cellular mechanisms and genes that protect cells against stress, ischemia, and injury. Lithium is surprisingly nontoxic in the therapeutic range of 0.6–1.0 mM (500–1,200 mg of lithium per day), while toxic levels occur at 1.4 mM or higher. Acute toxicity is rare and generally reversible. Because toxic levels are close to therapeutic levels, careful dose titration with serum measurements is recommended. Oral doses of 500–1,200 mg will bring most patients into the therapeutic range (Young, 2009). Histopathological examination of cerebral cortex tissue from lithium-treated rats revealed astrocytes with large vesicular nuclei and prominent nucleoli, reflecting the potent neuroprotective effects of lithium. Lithium can directly activate the PI3K/Akt pathway while also directly inhibiting GSK3-β (Nishimoto et al., 2008; Ates et al., 2022). Both effects potentially contribute to mood stabilizing and neuroprotective effects. In alignment with earlier research, the current study demonstrated that, compared to CIRI, the preinjection of lithium markedly elevated PI3K levels and Akt activity while significantly reducing GSK3-β activity. Importantly, prior studies have indicated that lithium's inhibition of GSK3-β offers protection to neurons against apoptosis in a glutamate-induced model of neural injury (Leng et al., 2008). In addition, preinjection of lithium significantly increased the cerebral cortex levels of antioxidant molecules such as Nrf2, HO-1, GSH, and SOD compared with the CIRI group. Compared with the CIRI group, lithium treatment significantly decreased the levels of oxidative stress markers, such as MPO and MDA, in the cerebral cortex. All these changes support the potent neuroprotective effect of lithium against CIRI. Notably, the impact of lithium on oxidative stress is probably due to the enhanced PI3K/Akt pathway and the suppression of GSK3-β activity, as previously stated. Consistent with our results, clinical studies have indicated that lithium can protect against complications from ischemic stroke by reducing oxidative stress (Xu et al., 2007). Furthermore, our results indicated that lithium treatment led to a notable rise in SIRT1 levels and a significant reduction in collagen expression within the cerebral cortex compared with CIRI. Additionally, this alteration in SIRT1 levels may be associated with the changes observed in the PI3K/Akt/GSK3-β pathway in the lithium group. Moreover, the decrease in collagen expression could be a consequence of the elevated SIRT1 levels in lithium-treated rats, as previously discussed (Chu et al., 2018). Notably, hyperglycemia has been associated with poor outcomes in patients with stroke receiving thrombolytic therapy (Wang et al., 2023). Moreover, dexamethasone use has been correlated with worsening postischemic outcomes (Wass et al., 1996). Interestingly, our study showed the potent neuroprotective effects of lithium against CIRI even in the presence of a severe form of impaired glucose metabolism. Considering the aforementioned findings, we determined that CIRI is linked to a notable downregulation of the PI3K/Akt/GSK3-β pathway, upregulation of oxidative stress signals, and downregulation of SIRT1 levels in brain tissue. Moreover, preinjection of lithium, a potent activator of the PI3K/Akt/GSK-3β pathway, significantly reduced all pathological changes associated with CIRI, including oxidative stress signals, collagen expression, and histopathological changes, in addition to upregulating SIRT1 levels in brain tissue. The neuroprotective effects of lithium were significant even though the CIRI rats were hyperglycemic. Lithium may be a good choice for treating challenging conditions in CIRIs. AcknowledgmentsNone. Conflict of interestThe authors declare no conflict of interest. FundingThe authors declare that no funds, grants, or other support were received during the preparation of this manuscript. The authors have no relevant financial or nonfinancial interests to disclose. Authors' contributionsMF and IA designed and supervised the experiments and revised and approved the manuscript. MM performed the experiments, collected the samples, measured the parameters, analyzed the data, and wrote the manuscript. Data availabilityAll data used are available. ReferencesAlural, B., Ozerdem, A., Allmer, J., Genc, K. and Genc, S. 2015. Lithium protects against paraquat neurotoxicity by NRF2 activation and miR-34a inhibition in SH-SY5Y cells [Original Research]. Front. Cellular Neurosci. 9, 209; doi: 10.3389/fncel.2015.00209 Archer, D.P., Ewen, A., Roth, S.H. and Samanani, N. 1994. Plasma, brain, and spinal cord concentrations of thiopental associated with hyperalgesia in the rat. Anesthesiology 80(1), 168–176; doi:10.1097/00000542-199401000-00024 Aref, H., Zakaria, M., Shokri, H., Roushdy, T., El Basiouny, A. and El Nahas, N. 2021. Changing the Landscape of Stroke in Egypt. Cerebrovasc. Dis. Extra. 11(3), 155–159; doi:10.1159/000521271 Askar, M., Ibrahim, I. and Mahmoud, M. 2022. Dexamethasone-induced metabolic syndrome: re-evaluation of an underestimated experimental model. Bull. Pharm. Sci. Assiut. 45, 989–1004; doi: 10.21608/BFSA.2022.271784 Ates, N., Caglayan, A., Balcikanli, Z., Sertel, E., Beker, M.C., Dilsiz, P., Caglayan, A.B., Celik, S., Dasdelen, M.F., Caglayan, B., Yigitbasi, T., Ozbek, H., Doeppner, T.R., Hermann, D.M. and Kilic, E. 2022. Phosphorylation of PI3K/Akt at Thr308, but not phosphorylation of MAPK kinase, mediates lithium-induced neuroprotection against cerebral ischemia in mice. Exp. Neurol. 351, 113996; doi:10.1016/j.expneurol.2022.113996 Barel, M., Perez, O.A., Giozzet, V.A., Rafacho, A., Bosqueiro, J.R. and Do Amaral, S.L. 2010. Exercise training prevents hyperinsulinemia, muscular glycogen loss and muscle atrophy induced by dexamethasone treatment. Eur. J. Appl. Physiol. 108(5), 999–1007; doi:10.1007/s00421-009-1272-6 Bellacosa, A., Chan, T.O., Ahmed, N.N., Datta, K., Malstrom, S., Stokoe, D., Mccormick, F., Feng, J. and Tsichlis, P. 1998. Akt activation by growth factors is a multiple-step process: the role of the PH domain. Oncogene 17(3), 313–325; doi:10.1038/sj.onc.1201947 Bonkhoff, A.K., Schirmer, M.D., Bretzner, M., Hong, S., Regenhardt, R.W., Brudfors, M., Donahue, K.L., Nardin, M.J., Dalca, A.V., Giese, A.K., Etherton, M.R., Hancock, B.L., Mocking, S.J.T., Mcintosh, E.C., Attia, J., Benavente, O.R., Bevan, S., Cole, J.W., Donatti, A., Griessenauer, C.J., Heitsch, L., Holmegaard, L., Jood, K., Jimenez-Conde, J., Kittner, S.J., Lemmens, R., Levi, C.R., Mcdonough, C.W., Meschia, J.F., Phuah, C.L., Rolfs, A., Ropele, S., Rosand, J., Roquer, J., Rundek, T., Sacco, R.L., Schmidt, R., Sharma, P., Slowik, A., Söderholm, M., Sousa, A., Stanne, T.M., Strbian, D., Tatlisumak, T., Thijs, V., Vagal, A., Wasselius, J., Woo, D., Zand, R., Mcardle, P.F., Worrall, B.B., Jern, C., Lindgren, A.G., Maguire, J., Bzdok, D., Wu, O. and Rost, N.S. 2021. Outcome after acute ischemic stroke is linked to sex-specific lesion patterns. Nature Commun. 12(1), 3289; doi:10.1038/s41467-021-23492-3 Bowles, N.P., Karatsoreos, I.N., Li, X., Vemuri, V.K., Wood, J.A., Li, Z., Tamashiro, K.L., Schwartz, G.J., Makriyannis, A.M., Kunos, G., Hillard, C.J., Mcewen, B.S. and Hill, M.N. 2015. A peripheral endocannabinoid mechanism contributes to glucocorticoid-mediated metabolic syndrome. Proc. Natl. Acad. Sci. U. S. A. 112(1), 285–290; doi:10.1073/pnas.1421420112 Butler, J.L., Barham, B.J. and Heidenreich, B.A. 2019. Comparison of indirect peroxidase and avidin-biotin-peroxidase complex (ABC) immunohistochemical staining procedures for c-fos in rat brain. J. Anat. 234(6), 936 -942; doi: 10.1111/joa.12967 Chen, S., Chen, H., Du, Q. and Shen, J. 2020. Targeting Myeloperoxidase (MPO) Mediated Oxidative Stress and Inflammation for Reducing Brain Ischemia Injury: potential Application of Natural Compounds. Front. Physiol. 11, 433; doi:10.3389/fphys.2020.00433 Chen, J. 2014. Heme oxygenase in neuroprotection: from mechanisms to therapeutic implications. Rev. Neurosci. 25(2), 269–280; doi: 10.1515/revneuro-2013-0046 Chiu, C.T., Wang, Z., Hunsberger, J.G. and Chuang, D.M. 2013. Therapeutic potential of mood stabilizers lithium and valproic acid: beyond bipolar disorder. Pharmacol. Rev. 65(1), 105–142; doi:10.1124/pr.111.005512 Chu, H., Jiang, S., Liu, Q., Ma, Y., Zhu, X., Liang, M., Shi, X., Ding, W., Zhou, X., Zou, H., Qian, F., Shaul, P.W., Jin, L. and Wang, J. 2018. Sirtuin1 Protects against Systemic Sclerosis-related Pulmonary Fibrosis by Decreasing Proinflammatory and Profibrotic Processes. Am. J. Respir. Cell. Mol. Biol. 58(1), 28–39; doi:10.1165/rcmb.2016-0192OC Cross, D.A., Alessi, D.R., Cohen, P., Andjelkovich, M. and Hemmings, B.A. 1995. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 378(6559), 785–789; doi:10.1038/378785a0 Da Silva, F.N., Zimath, P.L., Do Amaral, T.A., Martins, J.R.N. and Rafacho, A. 2023. Coadministration of olanzapine causes minor impacts on the diabetogenic outcomes induced by dexamethasone treatment in rats. Life Sci. 322, 121660; doi:10.1016/j.lfs.2023.121660 Dali-Youcef, N., Lagouge, M., Froelich, S., Koehl, C., Schoonjans, K. and Auwerx, J. 2007. Sirtuins: the 'magnificent seven', function, metabolism and longevity. Ann. Med. 39(5), 335–345; doi:10.1080/07853890701408194 Diniz, B.S., Machado-Vieira, R. and Forlenza, O.V. 2013. Lithium and neuroprotection: translational evidence and implications for the treatment of neuropsychiatric disorders. Neuropsychiatr. Dis. Treat. 9, 493–500; doi:10.2147/ndt.s33086 Du, J., Yin, G., Hu, Y., Shi, S., Jiang, J., Song, X., Zhang, Z., Wei, Z., Tang, C. and Lyu, H. 2020. Coicis semen protects against focal cerebral ischemia-reperfusion injury by inhibiting oxidative stress and promoting angiogenesis via the TGFβ/ALK1/Smad1/5 signaling pathway. Aging (Albany. NY). 13(1), 877–893; doi:10.18632/aging.202194 Eappen, S. and Kissin, I. 1998. Effect of subarachnoid bupivacaine block on anesthetic requirements for thiopental in rats. Anesthesiology 88(4), 1036–1042; doi:10.1097/00000542-199804000-00024 Elibol, B. and Kilic, U. 2018. High Levels of SIRT1 Expression as a Protective Mechanism Against Disease-Related Conditions. Front. Endocrinol. (Lausanne) 9, 614; doi:10.3389/fendo.2018.00614 Endo, H., Nito, C., Kamada, H., Nishi, T. and Chan, P.H. 2006. Activation of the Akt/GSK3β signaling pathway mediates survival of vulnerable hippocampal neurons after transient global cerebral ischemia in rats. J. Cerebral Blood Flow Metab. 26(12), 1479–1489; doi:10.1038/sj.jcbfm.9600303 Feigin, V.L. and Brainin, M. 2022. World Stroke Organization (WSO): global Stroke Fact Sheet 2022. Stroke 17(1), 18–29; doi:10.1177/17474930211065917 Feigin, V.L., Nichols, E., Alam, T., Bannick, M.S., Beghi, E., Blake, N., Culpepper, W.J., Dorsey, E.R., Elbaz, A., Ellenbogen, R.G., Fisher, J.L., Fitzmaurice, C., Giussani, G., Glennie, L., James, S.L. and Johnson, C.O. 2019. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 18(5), 459–480; doi:10.1016/S1474-4422(18)30499-X Forlenza, O.V., De-Paula, V.J. and Diniz, B.S. 2014. Neuroprotective effects of lithium: implications for the treatment of Alzheimer's disease and related neurodegenerative disorders. ACS Chem. Neurosci. 5(6), 443–450; doi:10.1021/cn5000309 Formisano, L., Guida, N., Mascolo, L., Serani, A., Laudati, G., Pizzorusso, V. and Annunziato, L. 2020. Transcriptional and epigenetic regulation of ncx1 and ncx3 in the brain. Cell Calcium 87, 102194; doi:10.1016/j.ceca.2020.102194 Friess, W. 1998. Collagen–biomaterial for drug delivery. Eur. J. Pharmaceutics Biopharmaceutics 45(2), 113–136; doi: 10.1016/s0939-6411(98)00017-4 Fukunaga, K. and Kawano, T. 2003. Akt is a molecular target for signal transduction therapy in brain ischemic insult. J. Pharmacol. Sci. 92(4), 317–327; doi:10.1254/jphs.92.317 Gan, L. and Johnson, J.A. 2014. Oxidative damage and the Nrf2-ARE pathway in neurodegenerative diseases. Biochimica Et Biophysica Acta (BBA). - Mol. Basis Dis. 1842(8), 1208–1218; doi:10.1016/j.bbadis.2013.12.011 Gao, Y., Xu, X., Chang, S., Wang, Y., Xu, Y., Ran, S., Huang, Z., Li, P., Li, J., Zhang, L., Saavedra, J.M., Liao, H. and Pang, T. 2015. Totarol prevents neuronal injury in vitro and ameliorates brain ischemic stroke: potential roles of Akt activation and HO-1 induction. Toxicol. Appl. Pharmacol. 289(2), 142–154; doi:10.1016/j.taap.2015.10.001 Hetman, M., Kanning, K., Cavanaugh, J.E. and Xia, Z. 1999. Neuroprotection by brain-derived neurotrophic factor is mediated by extracellular signal-regulated kinase and phosphatidylinositol 3-kinase. J. Biol. Chem. 274(32), 22569–22580; doi:10.1074/jbc.274.32.22569 Holay, M.P. 2015.Metabolic Syndrome and Intracranial Atherosclerosis. Vidarbha J. Intern. Med. 34, 108664. Available via https://api.semanticscholar.org/CorpusID:12108664 Janero, D.R. 1990. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic. Biol. Med. 9(6), 515–540; doi:10.1016/0891-5849(90)90131-2 Jhuo, C.F., Hsieh, S.K., Chen, W.Y. and Tzen, J.T.C. 2023. Attenuation of skeletal muscle atrophy induced by dexamethasone in rats by teaghrelin supplementation. Molecules 28(2), 688; doi:10.3390/molecules28020688 Kao, S., -H., Yuan, M., -P.., Tzen, C., -Y.., Liang, Y., -C.., Kuo, P., Chen, T., -H.., Lee, H. and -M. 2008. Lithium Induces Heme Oxygenase1 Expression and Suppresses Lipopolysaccharide-Induced Inducible Nitric Oxide Synthase Expression in C6 Glioma Cells. Clin. Mol. Med. 1, 1. Koronowski, K.B. and Perez-Pinzon, M.A. 2015. Sirt1 in cerebral ischemia. Brain Circ. 1(1), 69–78; doi:10.4103/2394-8108.162532 Lee, T.S., Tsai, H.L. and Chau, L.Y. 2003. Induction of heme oxygenase-1 expression in murine macrophages is essential for the anti-inflammatory effect of low dose 15-deoxy-Delta 12,14-prostaglandin J2. J. Biol. Chem. 278(21), 19325–19330; doi:10.1074/jbc.M300498200 Leng, Y., Liang, M.H., Ren, M., Marinova, Z., Leeds, P. and Chuang, D.M. 2008. Synergistic neuroprotective effects of lithium and valproic acid or other histone deacetylase inhibitors in neurons: roles of glycogen synthase kinase-3 inhibition. J. Neurosci. 28(10), 2576–2588; doi:10.1523/jneurosci.5467-07.2008 Leroy, K. and Brion, J.P. 1999. Developmental expression and localization of glycogen synthase kinase-3beta in rat brain. J. Chem. Neuroanat. 16(4), 279–293; doi:10.1016/s0891-0618(99)00012-5 Mahendran, P. and Devi, C.S. 2001. Effect of Garcinia cambogia extract on lipids and lipoprotein composition in dexamethasone administered rats. Indian J. Physiol. Pharmacol. 45(3), 345–350. Mahmoud, M.F., Elmaghraby, A.M., Ali, N., Mostafa, I., El-Shazly, A.M., Abdelfattah, M.A.O. and Sobeh, M. 2022. Black pepper oil (Piper nigrum L.) mitigates dexamethasone induced pancreatic damage via modulation of oxidative and nitrosative stress. Biomed. Pharmacother 153, 113456; doi:10.1016/j.biopha.2022.113456 Michalski, D., Spielvogel, E., Puchta, J., Reimann, W., Barthel, H., Nitzsche, B., Mages, B., Jäger, C., Martens, H., Horn, A.K.E., Schob, S. and Härtig, W. 2020. Increased immunosignals of collagen IV and fibronectin indicate ischemic consequences for the neurovascular matrix adhesion zone in various animal models and human stroke tissue. Front. Physiol. 11, 575598; doi:10.3389/fphys.2020.575598 Milani, P., Ambrosi, G., Gammoh, O., Blandini, F. and Cereda, C. 2013. SOD1 and DJ-1 converge at Nrf2 pathway: a clue for antioxidant therapeutic potential in neurodegeneration. Oxid. Med. Cell. Longev. 2013, 836760; doi:10.1155/2013/836760 Nishimoto, T., Kihara, T., Akaike, A., Niidome, T. and Sugimoto, H. 2008. Alpha-Amino-3-hydroxy-5-methyl-4-isoxazole propionate attenuates glutamate-induced caspase-3 cleavage via regulation of glycogen synthase kinase 3beta. J. Neurosci. Res. 86(5), 1096–1105; doi:10.1002/jnr.21567 Niture, S.K., Khatri, R. and Jaiswal, A.K. 2014. Regulation of Nrf2-an update. Free Radic. Biol. Med. 66, 36–44; doi:10.1016/j.freeradbiomed.2013.02.008 Oche, J., Olorundare, O., Afolabi, S., Ologe, M., Njan, A. and Akanbi, O. 2023. Comparative therapeutic effect of single/combined administration of saxagliptin, metformin and intranasal insulin on dexamethasone induced insulin resistance in albino wistar rat model. Niger. J. Physiol. Sci. 38(1), 37–46; doi:10.54548/njps.v38i1.7 Oztanir, M.N., Dogan, M.F., Turkmen, N.B., Taslidere, A., Sahin, Y. and Ciftci, O. 2022. Secukinumab ameliorates oxidative damage induced by cerebral ischemia-reperfusion in rats. Turk. Neurosurg. 32(5), 732–739; doi:10.5137/1019-5149.jtn.33606-21.3 Pang, T., Wang, Y.J., Gao, Y.X., Xu, Y., Li, Q., Zhou, Y.B., Xu, L., Huang, Z.J., Liao, H., Zhang, L.Y., Gao, J.R., Ye, Q. and Li, J. 2016. A novel GSK-3β inhibitor YQ138 prevents neuronal injury induced by glutamate and brain ischemia through activation of the Nrf2 signaling pathway. Acta Pharmacol. Sinica 37(6), 741–752; doi:10.1038/aps.2016.3 Poulsen, R.C., Watts, A.C., Murphy, R.J., Snelling, S.J., Carr, A.J. and Hulley, P.A. 2014. Glucocorticoids induce senescence in primary human tenocytes by inhibition of sirtuin 1 and activation of the p53/p21 pathway: in vivo and in vitro evidence. Ann. Rheumatic Dis. 73(7), 1405; doi:10.1136/annrheumdis-2012-203146 Ricard-Blum, S. 2011. The collagen family. Cold Spring Harbor Perspect. Biol. 3(1), 4978; doi:10.1101/cshperspect.a004978 Rojo, A.I., Sagarra, M.R.D. and Cuadrado, A. 2008. GSK‐3β down‐regulates the transcription factor Nrf2 after oxidant damage: relevance to exposure of neuronal cells to oxidative stress. J. Neurochem. 105(1), 192–202; doi:10.1111/j.1471-4159.2007.05124 Ruan, C., Guo, H., Gao, J., Wang, Y., Liu, Z., Yan, J., Li, X. and Lv, H. 2021. Neuroprotective effects of metformin on cerebral ischemia-reperfusion injury by regulating PI3K/Akt pathway. Brain Behav. 11(10), 2335; doi:10.1002/brb3.2335 Saad, M.J., Folli, F., Kahn, J.A. and Kahn, C.R. 1993. Modulation of insulin receptor, insulin receptor substrate-1, and phosphatidylinositol 3-kinase in liver and muscle of dexamethasone-treated rats. J. Clin. Invest. 92(4), 2065–2072; doi:10.1172/jci116803 Sandoughdaran, S., Sadeghipour, H. and Sadeghipour, H.R. 2016. Effect of acute lithium administration on penile erection: involvement of nitric oxide system. Int. J. Reprod. Biomed. 14(2), 109–116. Sasaki, T., Han, F., Shioda, N., Moriguchi, S., Kasahara, J., Ishiguro, K. and Fukunaga, K. 2006. Lithium-induced activation of Akt and CaM kinase II contributes to its neuroprotective action in a rat microsphere embolism model. Brain Res. 1108(1), 98–106; doi:10.1016/j.brainres.2006.06.009 Satoh, T., Mckercher, S.R. and Lipton, S.A. 2013. Nrf2/ARE-mediated antioxidant actions of pro-electrophilic drugs. Free Radic. Biol. Med. 65, 645–657; doi:10.1016/j.freeradbiomed.2013.07.022 Savolainen, K.M., Loikkanen, J., Eerikäinen, S. and Naarala, J. 1998. Glutamate-stimulated ROS production in neuronal cultures: interactions with lead and the cholinergic system. Neurotoxicology 19(4-5), 669–674. Turc, G., Bhogal, P., Fischer, U., Khatri, P., Lobotesis, K., Mazighi, M., Schellinger, P.D., Toni, D., De Vries, J., White, P. and Fiehler, J. 2023. European Stroke Organisation (ESO)-European Society for Minimally Invasive Neurological Therapy (ESMINT) guidelines on mechanical thrombectomy in acute ischemic stroke. J. Neurointerventional Surg. 15(8), e8. Üllen, A., Singewald, E., Konya, V., Fauler, G., Reicher, H., Nusshold, C., Hammer, A., Kratky, D., Heinemann, A., Holzer, P., Malle, E. and Sattler, W. 2013. Myeloperoxidase-derived oxidants induce blood-brain barrier dysfunction in vitro and in vivo. PLos One 8(5), e64034; doi:10.1371/journal.pone.0064034 Vivanco, I. and Sawyers, C.L. 2002. The phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nat. Rev. Cancer 2(7), 489–501; doi:10.1038/nrc839 Wang, H., Yao, X., Huang, K., Zhang, J., Xiao, J., Guo, J., Wei, D. and Xiang, B. 2022. Low-dose dexamethasone in combination with luteolin improves myocardial infarction recovery by activating the antioxidative response. Biomed. Pharmacother. 151, 113121; doi:10.1016/j.biopha.2022.113121 Wang, K.J., Zhang, W.Q., Liu, J.J., Cui, Y. and Cui, J.Z. 2020. Piceatannol protects against cerebral ischemia/reperfusion-induced apoptosis and oxidative stress via the Sirt1/FoxO1 signaling pathway. Mol. Med. Rep. 22(6), 5399–5411; doi:10.3892/mmr.2020.11618 Wang, Y., Jiang, G., Zhang, J., Wang, J., You, W. and Zhu, J. 2023. Blood glucose level affects prognosis of patients who received intravenous thrombolysis after acute ischemic stroke? A meta-analysis. Front. Endocrinol. (Lausanne). 14, 1120779; doi:10.3389/fendo.2023.1120779 Wang-Fischer, Y. 2009. Manual of Stroke Models in Rats (1st Edition ed.). Boca Raton: CRC Press. Wass, T.C., Scheithauer, B.W., Bronk, J.T., Wilson, R.M. and Lanier, W.L. 1996. Insulin treatment of corticosteroid-associated hyperglycemia and its effect on outcome after forebrain ischemia in rats. Anesthesiology 84(3), 644–651; doi:10.1097/00000542-199603000-00020 Xu, L. and Yao, Y. 2021. Central nervous system fibroblast-like cells in stroke and other neurological disorders. Stroke 52(7), 2456–2464; doi: 10.1161/STROKEAHA.120.033431 Xu, T., Wang, S., Li, X., Li, X., Qu, K., Tong, H., Zhang, R., Bai, S. and Fan, J. 2021. Lithium chloride represses abdominal aortic aneurysm via regulating GSK3β/SIRT1/NF-κB signaling pathway. Free Radical Biol. Med. 166, T1–10; doi:10.1016/j.freeradbiomed.2021.02.007 Xu, X.H., Hua, Y.N., Zhang, H.L., Wu, J.C., Miao, Y.Z., Han, R., Gu, Z. and Qin, Z.H. 2007. Greater stress protein expression enhanced by combined prostaglandin A1 and lithium in a rat model of focal ischemia. Acta Pharmacol. Sinica 28, 1097–1104. Yang, M.C., Zhang, H.Z., Wang, Z., You, F.L. and Wang, Y.F. 2016. The molecular mechanism and effect of cannabinoid-2 receptor agonist on the blood-spinal cord barrier permeability induced by ischemia-reperfusion injury. Brain Res. 1636, 81–92; doi:10.1016/j.brainres.2016.01.047 Yang, T., Sun, Y., Lu, Z., Leak, R.K. and Zhang, F. 2017. The impact of cerebrovascular aging on vascular cognitive impairment and dementia. Ageing Res. Rev. 34, 15–29; doi:10.1016/j.arr.2016.09.007 Yang, Y., Duan, W., Li, Y., Yan, J., Yi, W., Liang, Z., Wang, N., Yi, D. and Jin, Z. 2013. New role of silent information regulator 1 in cerebral ischemia. Neurobiol. Aging 34(12), 2879–2888; doi:10.1016/j.neurobiolaging.2013.06.008 Yang, Y.C., Lii, C.K., Lin, A.H., Yeh, Y.W., Yao, H.T., Li, C.C., Liu, K.L. and Chen, H.W. 2011. Induction of glutathione synthesis and heme oxygenase 1 by the flavonoids butein and phloretin is mediated through the ERK/Nrf2 pathway and protects against oxidative stress. Free Radical Biol. Med. 51(11), 2073–2081; doi:10.1016/j.freeradbiomed.2011.09.007 Young, W. 2009. Review of lithium effects on brain and blood. Cell Transplantation 18(9), 951–975; doi:10.3727/096368909x471251 Zhang, Q., Ding, M., Cao, Z., Zhang, J., Ding, F. and Ke, K. 2013. Pyrroloquinoline quinine protects rat brain cortex against acute glutamate-induced neurotoxicity. Neurochemical Res. 38(8), 1661–1671; doi:10.1007/s11064-013-1068-2 Zhao, Y. -J., Qiao, H., Liu, D. -F.., Li, J., Li, J.-X., Chang, S.-E., Lu, T., Li, F.-T., Wang, D., Li, H.-P., He, X. J. and Wang, F. 2022. Lithium promotes recovery after spinal cord injury. Neural Regeneration Res. 17(6), 1324–1333; doi:10.4103/1673-5374.327348 Zhu, Y.M., Wang, C.C., Chen, L., Qian, L.B., Ma, L.L., Yu, J., Zhu, M.H., Wen, C.Y., Yu, L.N. and Yan, M. 2013. Both PI3K/Akt and ERK1/2 pathways participate in the protection by dexmedetomidine against transient focal cerebral ischemia/reperfusion injury in rats. Brain Res. 1494, K1–K8; doi:10.1016/j.brainres.2012.11.047 | ||
| How to Cite this Article |
| Pubmed Style Askar MM, Ibrahim IAAE, Mahmoud MF. Lithium reduces cerebral ischemia/reperfusion injury in dexamethasone-treated rats: The role of the PI3K/Akt pathway. Open Vet. J.. 2025; 15(10): 5246-5258. doi:10.5455/OVJ.2025.v15.i10.41 Web Style Askar MM, Ibrahim IAAE, Mahmoud MF. Lithium reduces cerebral ischemia/reperfusion injury in dexamethasone-treated rats: The role of the PI3K/Akt pathway. https://www.openveterinaryjournal.com/?mno=257335 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.41 AMA (American Medical Association) Style Askar MM, Ibrahim IAAE, Mahmoud MF. Lithium reduces cerebral ischemia/reperfusion injury in dexamethasone-treated rats: The role of the PI3K/Akt pathway. Open Vet. J.. 2025; 15(10): 5246-5258. doi:10.5455/OVJ.2025.v15.i10.41 Vancouver/ICMJE Style Askar MM, Ibrahim IAAE, Mahmoud MF. Lithium reduces cerebral ischemia/reperfusion injury in dexamethasone-treated rats: The role of the PI3K/Akt pathway. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5246-5258. doi:10.5455/OVJ.2025.v15.i10.41 Harvard Style Askar, M. M., Ibrahim, . I. A. A. E. & Mahmoud, . M. F. (2025) Lithium reduces cerebral ischemia/reperfusion injury in dexamethasone-treated rats: The role of the PI3K/Akt pathway. Open Vet. J., 15 (10), 5246-5258. doi:10.5455/OVJ.2025.v15.i10.41 Turabian Style Askar, Mohamed M., Islam A. A. E.-h. Ibrahim, and Mona F. Mahmoud. 2025. Lithium reduces cerebral ischemia/reperfusion injury in dexamethasone-treated rats: The role of the PI3K/Akt pathway. Open Veterinary Journal, 15 (10), 5246-5258. doi:10.5455/OVJ.2025.v15.i10.41 Chicago Style Askar, Mohamed M., Islam A. A. E.-h. Ibrahim, and Mona F. Mahmoud. "Lithium reduces cerebral ischemia/reperfusion injury in dexamethasone-treated rats: The role of the PI3K/Akt pathway." Open Veterinary Journal 15 (2025), 5246-5258. doi:10.5455/OVJ.2025.v15.i10.41 MLA (The Modern Language Association) Style Askar, Mohamed M., Islam A. A. E.-h. Ibrahim, and Mona F. Mahmoud. "Lithium reduces cerebral ischemia/reperfusion injury in dexamethasone-treated rats: The role of the PI3K/Akt pathway." Open Veterinary Journal 15.10 (2025), 5246-5258. Print. doi:10.5455/OVJ.2025.v15.i10.41 APA (American Psychological Association) Style Askar, M. M., Ibrahim, . I. A. A. E. & Mahmoud, . M. F. (2025) Lithium reduces cerebral ischemia/reperfusion injury in dexamethasone-treated rats: The role of the PI3K/Akt pathway. Open Veterinary Journal, 15 (10), 5246-5258. doi:10.5455/OVJ.2025.v15.i10.41 |