| Research Article | ||
Open Vet. J.. 2025; 15(10): 5238-5245 Open Veterinary Journal, (2025), Vol. 15(10): 5238-5245 Research Article Comparison of normal adiponectin levels among animal population: Prospective health and metabolic physiologySinan Thanoon Abdullah1*, Ayman Mohamed Albanna2and Reem Marwan Mousa31Department of Basic Science, College of Dentistry, University of Mosul, Mosul, Iraq 2Department of Environmental Science, College of Environmental Sciences, University of Mosul, Mosul, Iraq 3Basic Nursing Sciences Branch, Nineveh University, Mosul, Iraq *Corresponding Author: Sinan Thanoon Abdullah. Department of Basic Science, College of Dentistry, University of Mosul, Mosul, Iraq. Email: sinantag2016 [at] uomosul.edu.iq Submitted: 11/05/2025 Revised: 22/08/2025 Accepted: 04/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: Public health in animals is increasingly being recognized due to its role in elucidating metabolic and physiological disorders in different species. Adiponectin, a hormone secreted by adipocytes, is a significant marker of glucose and lipid metabolism and metabolic health. However, baseline serum adiponectin levels in different animal species are not well characterized. Aim: This study aimed to measure and compare baseline serum adiponectin levels among nine animal species as a reference for physiological and metabolic investigations in veterinary science. Methods: We analyzed 90 sera samples (10/species) from healthy fish, chicken, rabbit, cat, dog, goat, sheep, cow, and rat. Adiponectin levels were assessed using a validated enzyme-linked immunosorbent assay-based method. Results: Large interspecies differences were observed. The highest adiponectin levels were found in rats (7.22 ± 0.25 ng/ml), followed by cows (6.94 ± 0.23 ng/ml) and goats (6.35 ± 0.25 ng/ml). Adiponectin levels were lower in rabbits (3.89 ± 0.18 ng/ml), chickens (2.43 ± 0.17 ng/ml), and fish (1.62 ± 0.11 ng/ml). Conclusion: These findings highlight the role of species-specific metabolic adaptations in adiponectin secretion, with higher levels in mammals overall. This information provides a foundation for future research into metabolic health, animal husbandry, and the physiological functions of adiponectin. Keywords: ELISA, Hormone, Animal populations, Public health. IntroductionAdiponectin is a pleiotropic adipokine that is predominantly secreted by adipose tissue and is crucial in the regulation of glucose metabolism, lipid oxidation, insulin sensitivity, and systemic inflammation (Fang and Judd, 2018). Adiponectin is inversely correlated with body fat mass, making it a unique biomarker in metabolic physiology (Frühbeck et al., 2019). Reducing circulating adiponectin levels are associated with a range of metabolic diseases, including obesity, insulin resistance, type 2 diabetes mellitus, cardiovascular disease, and non-alcoholic fatty liver disease (Pandey et al. 2015). Although adiponectin has been thoroughly investigated in clinical and epidemiological environments, its physiological baseline concentrations in a range of animal species have been poorly investigated (Swarbrick and Havel, 2008). Species-specific adiponectin dynamics are essential for not only comparative physiology but also translational research using animal models (Ebert et al., 2021). Variability of adiponectin among animal populations is influenced by a range of factors, including species, breed, age, sex, diet, and environmental factors (Kaminska et al., 2020). For instance, rodents, such as mice and rats, are widely used in metabolic studies due to their relatively high adiponectin expression and sensitivity to dietary and drug interventions (Barnea et al., 2006). However, interspecies differences in the molecular makeup, receptor binding, and secretion characteristics of adiponectin may limit its direct extrapolation to humans or other animals (Maeda et al., 2020). Hence, establishing normal reference ranges of adiponectin in a variety of animal populations is important for the interpretation of experimental data and the validation of animal models used in metabolism studies (Peake et al., 2005). Comparative studies of adiponectin levels can elucidate species-specific adaptations to metabolism (Rak et al., 2017). For instance, ruminants such as sheep and cattle have different lipid metabolism and insulin dynamics from monogastric animals, which may be expressed in their adipokine profiles. Similarly, variations in the regulation of energy balance between carnivores and herbivores may impact baseline adiponectin levels and their physiological significance (Al-Thuwaini, 2022). These comparisons could shed more light on evolutionary adaptations of energy homeostasis and help identify animal species that are resistant or susceptible to metabolic syndromes (Tsatsoulis et al., 2013). Another complexity in measuring adiponectin levels is its structure isoforms. Adiponectin circulating in the blood exists in multiple oligomeric structures: low-molecular-weight (trimer), medium-molecular-weight (hexamer), and high-molecular-weight (HMW) multimers. The HMW isoform has the highest biological activity in causing insulin sensitivity and anti-inflammatory effects. Some species specifically secrete other isoforms and thus influence the measurement of total adiponectin concentration (Liu and Liu, 2014). Therefore, qualitative and quantitative assessments are essential when comparing species’ adiponectin physiology. Furthermore, tissue- and species-specific expression of adiponectin receptors—AdipoR1 and AdipoR2—is also essential in specifying the metabolic effect of adiponectin (Rak et al., 2017). The skeletal muscle retains predominantly AdipoR1, whereas the liver has high AdipoR2 expression. Elucidation of receptor expression and regulation in different animal models can also help in understanding the systemic functions of adiponectin and resistance or susceptibility to metabolic disease (Zha et al., 2017). More recently, however, the role of adiponectin in animal health beyond metabolism has also been an area of interest, for example, immune function, reproduction, heat regulation, and aging (Khoramipour et al., 2021). Increased adiponectin levels have been linked to increased fertility in some animal breeds, and decreased levels have been linked to increased vulnerability to infection and oxidative stress (Rak et al., 2017). These results highlight the physiological relevance of adiponectin and the need to establish normal ranges and context-dependent meanings in various animal systems (Fantuzzi, 2013). Despite the expanding body of data, the gap between normative information on adiponectin concentration between various animal populations remains extremely large (Ricci and Bevilacqua, 2012). Most available studies are performed on laboratory animals under laboratory conditions, with scarce data for wild, farm, or companion animal populations (National Research Council, 2010). In addition, methodological differences in sample handling, storage, and assay techniques introduce variability into reported adiponectin levels, rendering interstudy comparison problematic. Therefore, systematic investigations are needed to normalize adiponectin measurement procedures and establish valid reference ranges across various species (Muñoz-Prieto et al., 2020). The current study aimed to compare normal adiponectin values across selected animal populations, that is, rodents, farm animals, and domestic pets, under physiologically stable conditions (Ricci and Bevilacqua, 2012). By measuring serum adiponectin concentrations using standardized immunoassays and comparing them against species-specific physiological values (e.g., body mass index, metabolic rate, and diet), this work will provide normative data that can be used to sustain both veterinary health monitoring and experiment model validation (Radin et al., 2009) . The findings of this study would assist in shedding light on comparative biology, establish standards for pathological alterations in metabolic wellness, and determine appropriate species for animal models of singular adipokine profiles for translational metabolic studies (Mora et al., 2024). This study aimed to determine adiponectin levels across various animal types to bridge the disparity between comparative physiology and biomedical relevance. Establishing normal levels of adiponectin and their biological relevance in different species not only improves our knowledge of energy metabolism but also consolidates the foundation for utilizing animals as models in studies on obesity, diabetes, and metabolic syndrome. This comparative approach paves the way to enhancing diagnostic tools and therapeutic targets that can be translated to answer questions regarding animal public health and veterinarian physiology for animal welfare. Materials and MethodsAnimal selection and collection of samplesNinety healthy animals representing nine species were used in this study: fish, chicken, rabbit, cat, dog, goat, sheep, cow, and rat. Ten animals of each species were selected, providing 10 samples for each species. The animals were housed in a controlled environment under standard husbandry conditions with free access to food and water ad libitum. Animal care and handling were performed in compliance with ethical standards, and experiments were not performed on any animals. Preparation of serum samplesBlood was sampled from all animal models by routine venipuncture. Blood samples were allowed to clot at room temperature for 30 minute after collection, and serum was separated by centrifugation at 3,000 rpm for 10 minute. The last serum samples were stored at –20°C until further analysis (Harikrishnan et al., 2018; Husain et al., 2024). Adiponectin quantificationSerum adiponectin levels were measured using a commercially available, validated enzyme-linked immunosorbent assay (ELISA) kit from Reedbiotech Co., Ltd. The ELISA was performed following the manufacturer’s guidelines with the required minor adjustments for the samples of certain species. Briefly, serum samples were appropriately diluted, and the quantity of adiponectin present was quantitated based on the standard curve created with provided amounts of recombinant adiponectin (Zeyad et al., 2024) (Fig. 1).
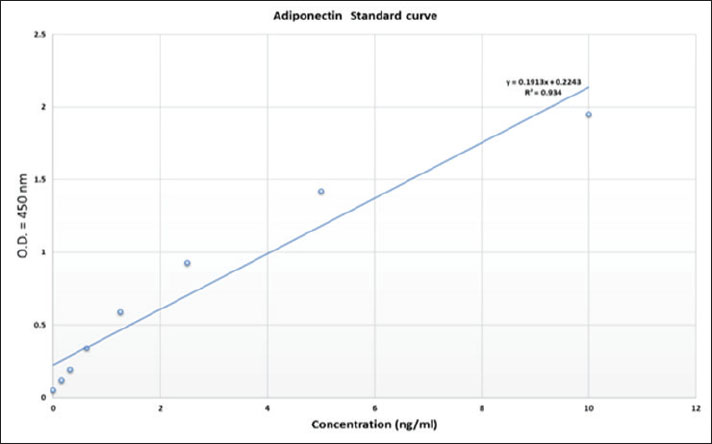
Fig. 1. The Reed Biotech ELISA kit for animal adiponectin detection has been developed for sensitive and species-compatible quantitation of serum adiponectin. The ELISA was composed of pre-coated plates, standard solution, and reagents optimized for consistent assay performance. Standard curve and assay validationA calibration curve with eight calibration points from 0 to 10 ng/ml (0.000, 0.156, 0.312, 0.625, 1.25, 2.5, 5.0, and 10.0 ng/ml) was constructed. The Optical density (OD) values for these points were determined at 450 nm using a microplate reader. The standard curve was used to interpolate the adiponectin concentration in the unknown samples. The assay’s linearity, reproducibility, and precision were checked by ensuring that all sample OD values were within the dynamic range of the standard curve, and intra- and inter-assay CV% were maintained below 10%. Statistical analysisValues are presented as mean ± SD by species. A one-way analysis of variance was used to compare adiponectin levels among species, followed by post hoc comparisons between pairs using Tukey’s test. Statistical significance was set at p < 0.05. The Shapiro-Wilk test was used to confirm data normality. Statistical analyses were performed using GraphPad Prism software (version X, GraphPad Software, San Diego, CA). Ethical approvalThe Institutional Animal Care and Use Committee, College of Veterinary Medicine, University of Mosul, approved this study (reference number: (UM.VET.2025) based on national and international guidelines of the ethical treatment of animals. All animal treatments were performed with utmost care according to the principles of humanenes and welfare. ResultsThis study aimed to compare baseline serum adiponectin levels among different animal species using a quantification and qualification method. Adiponectin levels were also determined successfully using ELISA kit, and values were interpolated from a standard curve made from known concentrations of recombinant adiponectin. Performance of the standard curve and assay validationThe ELISA standard curve had eight calibration points between 0 and 10 ng/ml (0.000, 0.156, 0.312, 0.625, 1.25, 2.5, 5.0, and 10.0 ng/ml). The corresponding OD values ranged from 0.050 at the zero standard to 1.950 for the highest concentration, demonstrating excellent positive linear correlation (R2=0.934), demonstrating excellent assay linearity (Fig. 2). The curve facilitated useful interpolation of adiponectin concentration for all sample values. All unexpected sample OD readings were also in this predefined dynamic range, and intra- and inter-assay CV% were less than 10% to ensure reproducibility and assay precision.
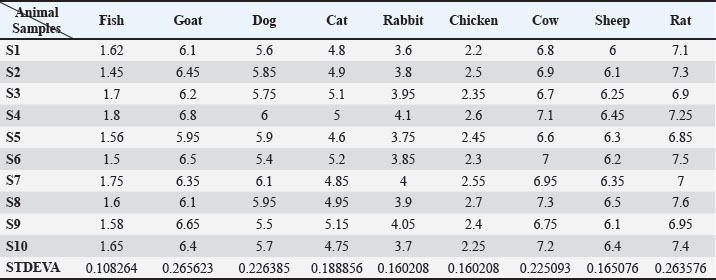
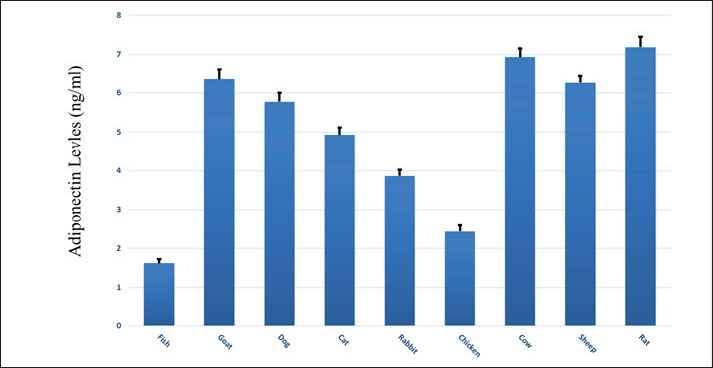
Fig. 2. The ELISA standard curve of adiponectin was eight calibration points between 010 ng/ml (0.000, 0.156, 0.312, 0.625, 1.25, 2.5, 5.0, and 10.0 ng/ml). Adiponectin levels across speciesThere existed a clear interspecies variation in adiponectin levels. Rats had the highest mean serum adiponectin concentration, at 7.22 ± 0.25 ng/ml, followed by cows at 6.94 ± 0.23 ng/ml, then goats at 6.35 ± 0.25 ng/ml and sheep at 6.28 ± 0.16 ng/ml. These concentrations were consistent with the high metabolic rate and lipid oxidation rates that are usually set in rodents and ruminants. Most importantly, adiponectin levels were more comparable across animals, as standard deviations minimal interspecies variation (Table 1). Table 1. Adiponectin concentrations (ng/ml) were measured via ELISA in different animals for comparison; each animal group was represented by 10 individual samples.
Felines and canines had mean adiponectin levels at intermediate ranges with means of 5.78 ± 0.22 and 4.93 ± 0.19 ng/ml, respectively. This value is an intermediary metabolic status characteristic of omnivorous domestic animals and very well matches the data presented in earlier veterinary research. Rabbits, as small hindgut fermenters and with a high-fiber herbivorous diet, also recorded a lower mean adiponectin value of 3.89 ± 0.18 ng/ml. Chickens and fish subsequently recorded the lowest serum adiponectin levels, at 2.43 ± 0.17 and 1.62 ± 0.11 ng/ml, respectively. These results may be due to profound physiological differences, such as body temperature regulation, lipid metabolic pathways, and evolutionary divergence from mammals (Fig. 3).
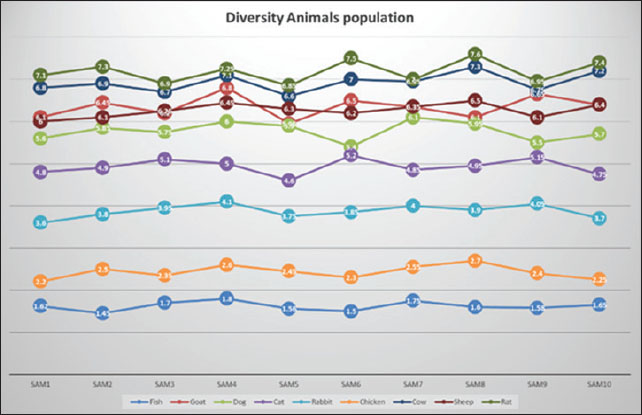
Fig. 3. Microplate ELISA for determining adiponectin levels for nine different animal samples and different standard concentrations for blotting unknown samples for comparison. Range and distributionThe within-group variability was minimal across all species. For example, rats and cows varied between 6.85 and 7.60 ng/ml and 6.60 and 7.30 ng/ml, respectively. Similarly, fish ranged between 1.45 and 1.80 ng/ml, and chickens between 2.20 and 2.70 ng/ml, demonstrating tight biological regulation of adiponectin during basal physiological states. These tight ranges confirm the validity of the data of each species as a reference baseline for use in subsequent physiological or pathophysiological studies (Fig. 4).
Fig. 4. Comparison of adiponectin levels (ng/ml) among nine different animals. Statistical analysis (one-way ANOVA) figure out a significantly high variance among animal groups (F=972.76, p < 0.0001). Tukey’s HSD post-hoc test was also employed to compare groups, with p < 0.05 being the measure of significance. Comparative visualizationA histogram was plotted for the visualization of species-specific adiponectin profiles (Fig. 5), showing mean concentrations with SD represented by error bars. Graphic comparison emphasized the dramatic difference between mammals (rodents and ruminants in particular) and non-mammals (avian and aquatic). The histogram clearly shows a tiered pattern: highest in rats and cows, mid-level in goats, sheep, dogs, and cats, and lowest in rabbits, chickens, and fish. This pictorial illustration identifies how metabolic health, body composition, and evolutionary heritage affect circulating adiponectin concentration.
Fig. 5. Adiponectin concentrations (ng/ml) from 10 samples were mean for each species in nine animal species: fish, chicken, rabbit, cat, dog, goat, sheep, cow, and rat. Error bars represent the SD, illustrating interspecies variability and homogeneity within each group. DiscussionThis work contributes to the elucidation of interspecies differences in adiponectin levels in several animals (Rak et al., 2017). The observed variations in serum adiponectin levels might reflect, in part, differences in metabolism among animal species related to their specific physiological adaptations (Liu et al., 2009). Adiponectin is involved in the control of glucose homeostasis, lipid metabolism, and immune response, and the knowledge of its naturally occurring level can provide valuable insight into animal species-specific health and metabolic status (Ji et al., 2021). Rats have the highest adiponectin levels of the species we have examined, consistent with their high metabolic rate and rapid fat oxidation rates, which may drive their elevated adiponectin concentrations (Ribot et al., 2008). The characteristic high levels of plasma adiponectin in ruminant animals, such as cows and goats, are consistent with their ability to maintain long-term fermentation in their complex stomachs and stable energy flux (Chilliard et al., 2005). These species are specialized to efficiently obtain and metabolize nutrients for extended durations and presumably benefit from elevated levels of adiponectin to promote energy homeostasis and lipid metabolism (Kalamaras, 2012). On the other hand, the argument that lowered adiponectin levels in rabbits, chickens, and fish are simply a reflection of their individual metabolic rates needs to be significantly changed. Although each of these creatures also exhibits unique energy usage patterns, medical data confirm that adiponectin is the dominant metabolic regulator for all three groups (Fu et al., 2009). Contrary to the initial hypothesis, adiponectin actions have an important role in energy balance and lipid metabolism in chickens. It is inversely related to fat storage and actively modulates fat and glucose storage, especially in response to dietary energy requirements. Ectothermic status for fish means having diverse metabolic processes, yet adiponectin remains a crucial hormone in regulating energy. It is also a satiety signal for some fish, an imperfectly established function among mammals. The existence of multiple forms of adiponectin receptors in fish implies a complex and critical regulatory mechanism. In rabbits, the idea that hindgut fermentation abolishes the need for adiponectin regulation has no scientific basis. Studies show dynamic variation in rabbit adiponectin, particularly as a response to insulin and the high metabolic demands of lactation, which suggests its role in energy regulation, similar to other mammals (Fischer et al., 2010). The consistency of serum adiponectin levels in individuals of the same species with relatively small fluctuations in the majority of individuals indicates that adiponectin secretion is tightly regulated under physiological conditions (Lihn et al., 2005). This stability of circulating adiponectin supports the suggestion that adiponectin participates stably and significantly in the metabolic equilibrium of each species and may serve as a biomarker of healthy status in different animal populations (Baldelli et al., 2024). These findings contribute to animal public health in a positive light, especially concerning cross-species differences regarding the manifestation of MD (Oh et al., 2007). For example, specific metabolic disorders can be diagnosed or the likelihood of developing obesity, diabetes, or cardiovascular diseases can be predicted based on the specific adiponectin profiles of a particular species (Matsuda and Shimomura, 2014). Furthermore, the interspecies variability demonstrated in this study may help shape future research directions on the physiological functions of adiponectin and its possible applications in veterinary and human medicine (Khoramipour et al., 2021). ConclusionThis study demonstrates large interspecies differences in baseline serum adiponectin concentrations among nine animal species, consistent with their wide variety of metabolic and physiological adaptations. Higher fat mass in all mammal species (rats, goats, and cows) was reflected by higher adiponectin levels, which corresponded to higher metabolic rate and specific energy regulation systems in these animals. On the other hand, non-mammalian animals including fish, chickens, and rabbits, showed lower adiponectin levels, probably due to variations in metabolic mechanisms and evolutionary adaptations. Consistent intraspecific differences and interspecific variation serve as an essential background for interpreting the role of adiponectin in metabolic health. These findings are not only significant for our understanding of adiponectin regulation but, also provide invaluable information in the field of animal public health to facilitate the development of monitoring and control methodologies. AcknowledgmentsThe authors would like to thank the Department of Physiology, Pharmacology, and Biochemistry of the College of Veterinary Medicine and the Department of Dental Basic Sciences of the College of Dentistry of Mosul University for their support in completing the research. Conflict of interestNo conflict to declare. FundingNo fund. Authors' contributionsS.T.A.: Conceptualization, methodology, writing, review, and editing. M.A.: Methodology, software, writing, review and editing. R.M.M.: Writing, review and editing. Data availabilityAll data were provided in the manuscript. ReferencesAl-Thuwaini, T.M. 2022. Adiponectin and its physiological function in ruminant cattle. Res. Agricult. Sci. 10, 115–122; doi:10.7831/ras.10.0_115 Baldelli, S., Aiello, G., Mansilla Di Martino, E., Campaci, D., Muthanna, F.M. and Lombardo, M. 2024. The role of adipose tissue and nutrition in the regulation of adiponectin. Nutrients 16(15), 2436; doi:10.3390/nu16152436 Barnea, M., Shamay, A., Stark, A.H. and Madar, Z. 2006. A high-fat diet has a tissue-specific effect on adiponectin and related enzyme expression. Obesity 14(12), 2145–2153; doi:10.1038/oby.2006.251 Chilliard, Y., Delavaud. and Bonnet. 2005. Leptin expression in ruminants: nutritional and physiological regulation of energy metabolism. Domestic Anim. Endocrinol. 29(1), 3–22; doi: 10.1016/j.domaniend.2005.02.026 Ebert, M.L., Schmidt, V.F., Pfaff, L., von Thaden, A., Kimm, M.A. and Wildgruber, M. 2021. Animal models of neointimal hyperplasia and restenosis: species-specific differences and implications for translational research. JACC 6(11), 900–917; doi: 10.1016/j.jacbts.2021.06.006 Fang, H. and Judd, R.L. 2018. Adiponectin regulation and function. Compreh. Physiol. 8(3), 1031–1063; doi:10.1002/cphy.c170046 Fantuzzi, G. 2013. Adiponectin in Inflammatory and Immune-Mediated Diseases. Cytokine 64, 1–10; doi:10.1016/j.cyto.2013.06.317 Fischer, S., Navarrete Santos, R. Thieme., Ramin. and Fischer. 2010. Adiponectin stimulates glucose uptake in rabbit blastocysts. Biol. Reprod. 83(5), 859–865; doi:10.1095/biolreprod.110.085346 Frühbeck, G., Catalán, V., Rodríguez, A., Ramírez, B., Becerril S Salvador., J, G., ómez. and Ambrosi, J. 2019. Adiponectin-leptin ratio is a functional biomarker of adipose tissue inflammation. Nutrients 11, 454; doi:10.3390/nu11020454 Fu, J.F., Fang, Y.L., Liang, L., Wang, C.L., Hong, F. and Dong. 2009. A rabbit model of pediatric nonalcoholic steatohepatitis: the role of adiponectin. World J. Gastroenterol. 15(8), 912; doi:10.3748/wjg.15.912 Harikrishnan, V., Hansen, A.K., Abelson, K.S. and Sørensen, D.B. 2018. Comparison of various blood sampling methods in mice and rats: effects on animal welfare. Lab. Animals 52(3), 253–264; doi:10.1177/0023677217741332 Husain, E.O., Albanna, A.M. and Asim, R.A. 2024. Ameliorative effect of adiponectin on experimentally induced arthritis in local rabbits. Egypt. J. Vet. Sci. 55(4), 1101–1109; doi:10.21608/ejvs.2024.264769.1803 Ji, R., Xu, X., Turchini, G.M., Mai, K. and Ai, Q. 2021. Roles of adiponectin in lipid and glucose metabolism modulation in fish: mechanisms and perspectives. Rev. Aquac. 13(4), 2305–2321; doi:10.1111/raq.12574 Kalamaras, K. 2012. The role of adiponectin in metabolism regulation in dairy cows (Doctoral dissertation, University of Nottingham). Available via https://eprints.nottingham.ac.uk/id/eprint/12588 Kaminska, B., Czerwinska, J., Bogacka, I., Chojnowska, K., Smolinska, N. and Nowakowski, J. 2020. Sex-and season-dependent differences in the expression of adiponectin and adiponectin receptors (AdipoR1 and AdipoR2) in the hypothalamic-pituitary-adrenal axis of the Eurasian beaver (Castor fiber L.). J. Physiol. Evol. 27(12), 113575; doi:10.1016/j.ygcen.2020.113575 Khoramipour, K., Chamari, K., Hekmatikar, A.A. and Ziyaiyan, A. 2021. Adiponectin: structure, physiological functions, role in diseases, and effects of nutrition. Nutrients 13(4), 1180; doi:10.3390/nu13041180 Lihn, A.S., Pedersen, S.B. and Richelsen, B. 2005. Adiponectin: action, regulation and association to insulin sensitivity. J. Physiol. Pharmacol. Obesity Rev. 6(1), 13–21; doi:10.1111/j.1467-789X.2005.00159.x Liu, M. and Liu, F. 2014. Regulation of adiponectin multimerization, signaling, and function. Best Pract. Res. Clin. Endocrinol. Metab. 28(1), 25–31; doi:10.1016/j.beem.2013.06.003 Liu, Y., Chewchuk, S., Lavigne, C., Brûlé, S., Pilon, G., Houde V., Sweeney. and G. 2009. Functional significance of skeletal muscle adiponectin production, changes in animal models of obesity and diabetes, and regulation by rosiglitazone treatment in vivo. Am. J. Physiology-Endocrinology Metab. 297(3), E657–E664; doi:10.1152/ajpendo.00132.2009 Maeda, N., Funahashi, T., Matsuzawa, Y. and Shimomura, I. 2020. Adiponectin, a unique adipocyte-derived factor beyond hormones. Atherosclerosis 292, 1–9; doi:10.1016/j.atherosclerosis.2019.11.011 Matsuda, M. and Shimomura, I. 2014. Adiponectin and oxidative stress roles in obesity-associated metabolic and cardiovascular diseases. Rev. Endocrine & Metabolic Disord. 15, 1–10; doi:10.1007/s11154-013-9278-6 Mora, I., Puiggròs, F., Serras, F., Gil-Cardoso, K. and Escoté, X. 2024. Emerging models for studying adipose tissue metabolism. Biochimica Et Biophysica Acta. (BBA). Molecula. Cell. Biol. Lipids , 116123; doi:10.1016/j.bbalip.2024.116123 Muñoz-Prieto, A., Cerón, J.J., Martínez-Subiela, S., Mrljak, V. and Tvarijonaviciute, A. 2020. A systematic review and meta-analysis of serum adiponectin measurements in the context of dog obesity. Animals 10(9), 1650; doi:10.3390/ani10091650 National Research Council. 2011. Guide for the care and use of laboratory animals. National Washington, DC: Academies Press; doi:10.17226/12910 Oh, D.K., Ciaraldi. and Henry. 2007. Adiponectin in health and disease. Diabetes. Obesity & Metab. 9(3), 282–289; doi:10.1111/j.1463-1326.2006.00623.x Pandey, A.K., Kalra, J.D. and Prabhu, M.D. 2015. Estimation of adiponectin levels in patients with diabetic and non-diabetic fatty liver diseases and healthy controls. Int. J. Res. Med. Sci. 3(1), 140–146; doi:10.5455/2320-6012.ijrms20150126 Peake, P.W., Kriketos, A.D., Campbell, L.V., Shen, Y. and Charlesworth, J.A. 2005. The metabolism of human adiponectin isoforms: studies in human subjects and experimental animals. Eur. J. Endocrinol. 153(3), 409–417; doi:10.1530/eje.1.01977 Radin MJ, Sharkey LC, Holycross BJ. Adipokines: a review of biological and analytical principles and an update in dogs, cats, and horses. Vet. Clin. Pathol. 38(2), 136–56; doi: 10.1111/j.1939-165X.2009.00133.x Rak, A., Mellouk, N., Froment, P. and Dupont. 2017. Adiponectin and resistin: potential metabolic signals affecting hypothalamo-pituitary gonadal axis in females and males of different species. Reproduction 153(6), R215–R226. Ribot, J., Rodríguez, A.M., Rodríguez, E. and Palou, A. 2008. Adiponectin and resistin response in the onset of obesity in male and female rats. Obesity 16(4), 723–730; doi:10.1038/oby.2007.123 Ricci, R. and Bevilacqua, F. 2012. The potential role of leptin and adiponectin in obesity: a comparative review. Vet. J. 191(3), 292–298; doi:10.1016/j.tvjl.2011.03.012 Swarbrick, M.M. and Havel, P.J. 2008. Physiological, pharmacological, and nutritional regulation of circulating adiponectin concentrations. Metabolic Syndrome Rel. Disord. 6(2), 87–102; doi:10.1089/met.2007.0012 Tsatsoulis, A., Mantzaris, M.D., Bellou, S. and Andrikoula, M. 2013. Insulin resistance: an adaptive mechanism becomes maladaptive in the current environment—an evolutionary perspective. Metabolism 62(5), 622–633; doi:10.1016/j.metabol.2012.11.004 Zeyad, H., Albanna, A. and Alomari, A. 2024. Combating dioxin and furan pollution in Mosul City's industrial water effluents: the enjcacy of ELISA detection and tannic acid treatment. Egypt. J. Aquatic Biol. Fisheries 28(4), 65–79; doi:10.21608/ejabf.2024.362118 | ||
| How to Cite this Article |
| Pubmed Style Abdullah ST, Albanna AM, Mousa RM. Comparison of normal adiponectin levels among animal population: Prospective health and metabolic physiology. Open Vet. J.. 2025; 15(10): 5238-5245. doi:10.5455/OVJ.2025.v15.i10.40 Web Style Abdullah ST, Albanna AM, Mousa RM. Comparison of normal adiponectin levels among animal population: Prospective health and metabolic physiology. https://www.openveterinaryjournal.com/?mno=257454 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.40 AMA (American Medical Association) Style Abdullah ST, Albanna AM, Mousa RM. Comparison of normal adiponectin levels among animal population: Prospective health and metabolic physiology. Open Vet. J.. 2025; 15(10): 5238-5245. doi:10.5455/OVJ.2025.v15.i10.40 Vancouver/ICMJE Style Abdullah ST, Albanna AM, Mousa RM. Comparison of normal adiponectin levels among animal population: Prospective health and metabolic physiology. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5238-5245. doi:10.5455/OVJ.2025.v15.i10.40 Harvard Style Abdullah, S. T., Albanna, . A. M. & Mousa, . R. M. (2025) Comparison of normal adiponectin levels among animal population: Prospective health and metabolic physiology. Open Vet. J., 15 (10), 5238-5245. doi:10.5455/OVJ.2025.v15.i10.40 Turabian Style Abdullah, Sinan Thanoon, Ayman Mohamed Albanna, and Reem Marwan Mousa. 2025. Comparison of normal adiponectin levels among animal population: Prospective health and metabolic physiology. Open Veterinary Journal, 15 (10), 5238-5245. doi:10.5455/OVJ.2025.v15.i10.40 Chicago Style Abdullah, Sinan Thanoon, Ayman Mohamed Albanna, and Reem Marwan Mousa. "Comparison of normal adiponectin levels among animal population: Prospective health and metabolic physiology." Open Veterinary Journal 15 (2025), 5238-5245. doi:10.5455/OVJ.2025.v15.i10.40 MLA (The Modern Language Association) Style Abdullah, Sinan Thanoon, Ayman Mohamed Albanna, and Reem Marwan Mousa. "Comparison of normal adiponectin levels among animal population: Prospective health and metabolic physiology." Open Veterinary Journal 15.10 (2025), 5238-5245. Print. doi:10.5455/OVJ.2025.v15.i10.40 APA (American Psychological Association) Style Abdullah, S. T., Albanna, . A. M. & Mousa, . R. M. (2025) Comparison of normal adiponectin levels among animal population: Prospective health and metabolic physiology. Open Veterinary Journal, 15 (10), 5238-5245. doi:10.5455/OVJ.2025.v15.i10.40 |