| Research Article | ||
Open Vet. J.. 2025; 15(10): 5058-5068 Open Veterinary Journal, (2025), Vol. 15(10): 5058-5068 Research Article Prevalence of subclinical mastitis in dairy cows in Kabul, AfghanistanMuhammad Ajmal Saidee and Mohammad Monir Tawfeeq*Department of Clinic, Faculty of Veterinary Science, Kabul University, Kabul, Afghanistan *Corresponding Author: Mohammad Monir Tawfeeq, Department of Clinic, Faculty of Veterinary Science, Kabul University, Kabul, Afghanistan. Email: monir.tawfeeq [at] gmail.com Submitted: 15/05/2025 Revised: 27/08/2025 Accepted: 11/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
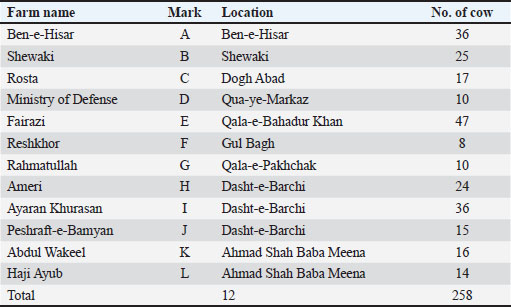
AbstractBackground: Mastitis is a significant and economically burdensome disease in dairy cattle, leading to considerable production losses. Generally, it has been divided into two types: clinical and subclinical. The clinical type of mastitis is easily recognizable, whereas the subclinical type requires improved laboratory tests for its detection. Aim: This study aimed to evaluate the prevalence of subclinical mastitis (SCM) in dairy farms in Kabul, Afghanistan. Methods: A cross-sectional study was conducted from August to October 2023 to estimate the prevalence and identify the major etiological agents of SCM in dairy farms. A total of 258 lactating cows were randomly selected from 12 farms. Diagnostic methods, including the California mastitis test (CMT), somatic cell count (SCC), and bacteriological culture, were employed to detect SCM. Results: Among 258 cows and 1,032 quarters, 63 cows (24.41%) and 86 quarters (8.33%) tested positive for SCM using CMT. CMT-positive samples were further analyzed for SCC and subjected to bacterial culture (BC) for etiological identification. Of the 63 cows and 86 quarters, 55 (21.32%) cows and 74 (7.17%) quarters tested positive for SCC. The mean SCC was highest for Staphylococcus aureus (S. aureus), followed by (S. agalactiae), Bacillus spp., coagulase-negative staphylococci (CNS), other Streptococcus spp., other Staphylococcus spp., coliforms, and Escherichia coli (E. coli). BC was positive in 56 cows (21.70%) and 74 quarters (7.17%). The predominant bacterial isolates were S. aureus (36.48%), CNS (16.21%), and other (5.40%). S. agalactiae (22.97%) and other Streptococcus spp. (10.81%) The other isolates included Bacillus spp. (4.05%), Coliforms (2.70%), and E. coli (1.35%). Conclusion: SCM is highly prevalent among lactating cows in Kabul, posing a significant challenge to dairy farming. Keywords: Bacteriological culture, California mastitis test, Dairy cows, Prevalence, Subclinical mastitis. IntroductionMastitis is one of the most significant diseases affecting dairy cows, leading to substantial economic losses in milk production. It adversely impacts milk yield, profit margins, and milk and dairy product quality in dairy-producing regions worldwide (Birhanu et al., 2017; Kaki et al., 2019). Mastitis is broadly classified into two types: subclinical mastitis (SCM) and clinical mastitis (CM). CM is characterized by visible physical, chemical, and microbiological changes in milk, accompanied by clinical signs such as dehydration, hyperthermia, and lethargy (Mpatswenumugabo et al., 2017). SCM is an asymptomatic condition identified primarily through elevated somatic cell counts (SCC) in milk (Qayyum et al., 2016). Unlike CM, SCM does not present with overt clinical signs, such as abnormal milk appearance, udder swelling, tenderness, or systemic symptoms, like fever and depression. However, it is associated with a significant increase in SCC, which is a key diagnostic indicator (Kayesh et al., 2014). SCM is responsible for 3–4 times greater economic losses than clinical mastitis, with milk production decreasing by up to 17.2% in SCM-affected cows, even in the absence of visible clinical signs (Abed et al., 2021). Furthermore, SCM is 15–40 times more prevalent than CM (Islam et al., 2011), and infected cows serve as a major reservoir of infection for healthy herd members, particularly in the absence of effective preventive measures (Ndahetuye et al., 2019). Numerous studies have documented the prevalence of SCM across various countries, with rates varying significantly depending on geographic location, farming practices, and diagnostic methods. For example, a study in Pakistan involving 2,000 quarters from 500 crossbred cows free from clinical mastitis, using the Surf Field Mastitis Test (SFMT), reported a quarter-level prevalence of 35.25% and an animal-level prevalence of 36% for SCM (Bachaya et al., 2011). Similarly, a study in Ethiopia conducted on 262 crossbred lactating cows from 12 intensively managed dairy farms, using the California Mastitis Test (CMT) for diagnosis, found that 105 cows (40.1%) and 170 quarters (16.2%) tested positive for SCM (Birhanu et al., 2017). Other studies have reported varying SCM prevalence rates in cattle. A study in Bangladesh performed by Sayeed et al. (2020) documented SCM prevalence rates of 71.9%, 67.9%, and 29.5% at the farm, animal, and quarter levels, respectively, using the SFMT. Mpatswenumugabo et al. (2017) from Rwanda, reported a cow-level prevalence of 50.4% using CMT and bacterial culture (BC). Naghshineh et al. (2015) observed an overall prevalence of SCM of 20.83% in Holstein cows, with 23.71% of cases affecting a single quarter in Iran. Qayyum et al. (2016) reported cow-level and quarter-level prevalences of 21.96% and 7.88%, respectively, in Pakistan’s Cholistani cows. SCM remains a critical challenge in dairy farming globally, necessitating continuous efforts for its effective control and management. This study aimed to determine the quarter- and cow-level prevalence of SCM and identify the primary etiological agents responsible for SCM in dairy farms in Kabul, Afghanistan, using multiple diagnostic methods. Materials and MethodsStudy areaThe present study was conducted from August to October 2023 on 12 dairy farms (Table 1) located in various districts (Char Asyab and Bagrami) and regions (6th, 7th, 13th, and 22nd) of Kabul, Afghanistan. These farms operated under an intensive management system, with milking performed twice daily using milking machines. Routine mastitis screening and dry cow therapy were not performed; only cases of clinical mastitis received treatment. Basic hygiene practices, such as udder washing, drying, and post-milking teat dipping, were followed on the farms. Table 1. Sampling farms and its locations.
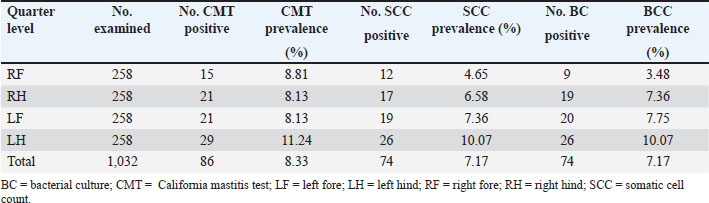
Kabul, the capital city of Afghanistan, is located in the eastern part of the country. Its geographic coordinates are approximately 34.5553° N latitude and 69.2075° E longitude, with an elevation of 1,790 m (5,873 feet) above sea level. The city experiences a semi-arid climate characterized by hot summers and cold winters. The annual precipitation is relatively low, averaging around 300 mm (11.8 inches), with dry conditions prevailing throughout much of the year. Seasonal temperature variations are significant, reflecting the region’s climatic extremes (Ahmadi et al., 2023). Animals and sample sizesA total of 258 lactating dairy cows were included in this study, selected from 12 dairy farms located in Kabul, Afghanistan. The study population comprised 218 Holstein Friesian, 32 crossbred, and 8 lactating cows of the local breed. Among the studied cows, 64 were in early lactation (1–3 months), 79 in mid-lactation (4–6 months), and 115 in late lactation (>6 months). Daily milk production was between 1 and 15 l in 89 cows, 16–30 l in 149 cows, and greater than 30 l in 20 cows. In terms of parity, 213 cows were in their first to third parity, 39 were in their fourth to sixth parity, and 6 were beyond the sixth parity. Regarding age distribution, 183 cows were between 3 and 6 years old, 56 were between 7 and 9 years old, and 19 were over 9 years of age. Based on the body condition score (BCS), 41, cows had a poor BCS, 205 had a moderate BCS, and 12 had a good BCS. The sample size was determined based on an estimated prevalence of 20.83% from a previous report (Naghshineh et al., 2015), with a 95% confidence level and a desired absolute precision of 5%. A simple random sampling technique was employed for sample selection. The sample size calculation was performed using the formula provided by Tripura et al. (2014): N=Z2 × Pexp (1−Pexp)/d2 N=required sample size, Z=the alpha value of 95%, Pexp=expected prevalence, and d=desired absolute precision. SamplingA thorough clinical examination was conducted on each lactating cow before collecting milk samples for mastitis screening. The udder was palpated to detect any abnormalities, such as fibrosis, swelling, or other mastitis-related clinical signs. Milk characteristics, including watery consistency, the presence of pus or clots, and non-functional (blind) quarters, were also assessed. The presence of at least one of these signs was sufficient to classify the mammary quarter as positive for CM, and such animals were excluded from the study (Mpatswenumugabo et al., 2017). Before sample collection, the udder and teats were cleaned with water and dried using sterile towels. The teat orifice and surrounding skin were disinfected with 70% alcohol and allowed to dry. Initially, 2–3 ml of milk was collected from each quarter for CMT. If the CMT result was positive, a second sample of 30–50-ml milk was promptly collected from the same quarter for SCC and BC. Samples were transported to the laboratory within 1–2 hours using a cooler box (Mpatswenumugabo et al., 2017). California mastitis testThe prevalence of SCM was initially assessed using the CMT, with scores ranging from 0 to 4 based on the modified Scandinavian scoring system. A score of 0 indicated a negative result (no gel formation), a score of 1 represented a trace result (suggesting a possible infection), and scores of 2 or 3 indicated a positive result, with a score of 4 corresponding to the thickest gel formation. A mammary quarter was considered positive for SCM if it exhibited a CMT score of ≥2+. Milk samples from all four quarters of each cow were individually analyzed using the CMT to detect SCM, following the methodology outlined by Mpatswenumugabo et al. (2017). Somatic cell countCMT-positive samples were further analyzed for SCC and BC. Additional milk samples were collected aseptically from CMT-positive quarters using sterile universal bottles. The samples were transported to the laboratory for SCC testing and bacteriological analysis (Birhanu et al., 2017). The SCC test was conducted at the Central Diagnostic Veterinary Laboratory, Bacteriology Department, Darulaman, Kabul Province, Afghanistan. All CMT-positive samples were evaluated for SCC using the NucleoCounter® SCC-100™ (ChemoMetec). Quarters with an SCC exceeding 200 × 10³ cells/ml were classified as positive for SCM (Williamson et al., 2022). The SCC was graded as negative for (0–200,000 cells/ml), trace for (>200,000–400,000 cells/ml), (1+) for (>400,000–1,200,000 cells/ml), (2+) for (>1,200,000–2,000,000 cells/ml), and (3+) (>2,000,000 cells/ml). Bacterial isolation and identificationAll BC and isolation tests were conducted at the Central Diagnostic Veterinary Laboratory, Bacteriology Department, Darulaman, Kabul Province, Afghanistan. Bacteriological examination of CMT-positive samples was performed following the procedures outlined by Abed et al. (2021). A loopful of milk sample from each infected quarter was inoculated onto MacConkey agar (Oxoid, England) and blood agar base (Oxoid, England) enriched with 7% defibrinated sheep blood. The inoculated plates were incubated aerobically at 37°C for 24–48 hours. Staphylococci were identified based on colony morphology, catalase and tube coagulase test results, and sugar fermentation patterns on mannitol salt agar (Oxoid, England). Streptococci were identified using the catalase test and the BBL™ Streptocard™ Latex Agglutination Test (El-Behiry et al., 2015). Among gram-negative bacteria, the primary focus was on Escherichia coli. Isolation was based on growth characteristics and lactose fermentation on MacConkey agar. Identification was confirmed using colony reactions on selective eosin methylene blue agar (Oxoid, England) and the Indole, Methyl Red, Voges-Proskauer, and Citrate biochemical test series (Birhanu et al., 2017). Statistical analysisAll data were analyzed descriptively using SPSS version 20. The association of the different variables with the prevalence of SCM at the cow or quarter levels was analyzed using the chi-squared test. The level of significance was set at p < 0.05 (Yimam et al., 2020). The independent samples t-test and one-way ANOVA were used to analyze the mean SCC, and the group means were compared using Duncan’s test (Mohammad Goli, 2012). Ethical approvalThis study was conducted in accordance with the research and postgraduate board guidelines of the veterinary science faculty of Kabul University (Protocol No. 31). ResultsCMT resultsThe CMT screening test indicated that 86 out of 1032 (8.33%) quarters (Table 2) and 63 out of 258 (24.41%) cows (Table 3) were positive for SCM. Table 2. Quarter-level prevalence of SCM using CMT, SCC, and BC (n=1,032).
Quarter-level prevalenceThe quarter-wise prevalence of SCM using CMT was recorded as (8.81%), (8.13%), (8.13%), and (11.24 %) in lactating cow right fore (RF), right hind (RH), left fore (LF), and left hind (LH) quarters, respectively (Table 2). Cow level prevalenceThe prevalence of SCM related to cow level, using CMT, was recorded as (33.33%), (44%), (29.41%), (30%), (14.89%), (25%), (20%), (29.16%), (13.88%), (6.66%), (25%), and (28.57%) in lactating cows of A, B, C, D, E, F, G, H, I, J, K, and L farms, respectively. The higher prevalence based on lactating cow level was (44%) in Farm B, and the lower prevalence was (6.66%) in Farm J (Table 3). Table 3. Cow level prevalence of SCM using CMT, SCC, and BC (n=258).
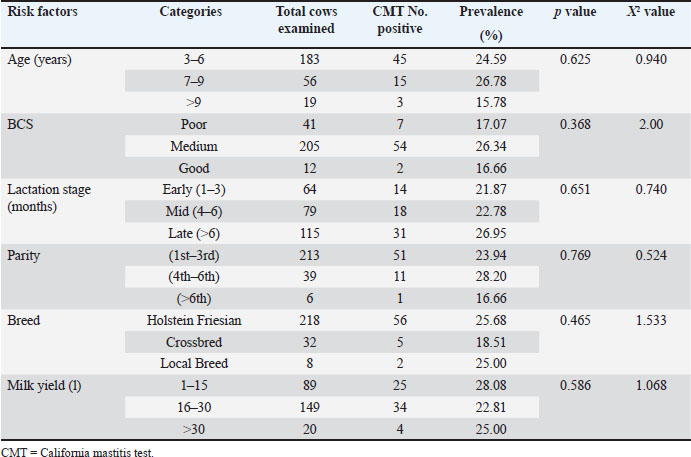
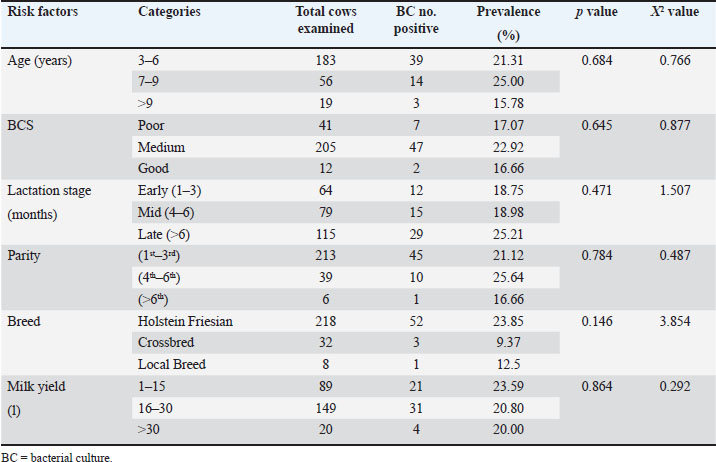
Risk factorsThe association of several risk factors, including age, BCS, lactation stage, breed, milk yield, and parity, with SCM was analyzed. Age-wise prevalenceThe prevalence of SCM across different age groups was 24.59%, 26.78%, and 15.78% for cows aged 3–6, 7–9, and >9 years, respectively (Table 4). Table 4. Risk factors related to the prevalence of SCM using CMT.
Body condition scoreThe prevalence of SCM varied with the BCS, with the highest prevalence observed in cows with a medium BCS (26.78%), followed by those with a poor BCS (24.59%) and a good BCS (15.78%); however, these differences were not statistically significant (Table 4). Lactation stageThe overall prevalence of SCM based on lactation stage revealed that cows in all three stages were affected. The prevalence was observed in late lactation (26.95%), followed by mid-lactation (22.78%) and early lactation (21.87%) (Table 4). ParityThe prevalence of SCM across parity groups was 28.20% in cows with 4th–6th parity, followed by those with 1st–3rd parity (23.93%) and those with more than 6th parity (16.66%) (Table 4). Breed-wise prevalenceAmong the 218 Holstein Friesian cows, 56 (25.68%) tested positive for SCM using CMT. Of the 32 crossbred cows, 5 (18.51%) were CMT-positive, and of the 8 local breed cows, 2 (25%) were CMT-positive (Table 4). Daily milk yieldThe prevalence of SCM based on daily milk yield was 28.08% in cows producing 1–15 l, 22.81%, and 25% in cows producing 1–15, 16–30, and >30 l, respectively (Table 4). None of the potential risk factors showed a statistically significant association (p > 0.05) with the occurrence of SCM as detected by the CMT (Table 4). SCC resultQuarter levelFrom 86 CMT-positive quarters, 74 (7.17%) quarters (Table 2) were positive, and from 63 CMT-positive cows, 55 (21.32%) cows (Table 3) were positive using SCC. The prevalence of SCM related to quarter-wise, using SCC, was recorded as 4.65%, 6.58%, 7.36%, and 10.07% in RF, RH, LF, and LH, respectively. The highest prevalence based on the lactating cow quarter was 10.07% in the LH quarter, and the lowest prevalence was 4.65% in the RF quarter (Table 2). Cow-level prevalenceThe prevalence of SCM related to cow level, using SCC, was recorded as (22.22%), (40%), (29.41%), (30%), (12.76%), (12.50%), (10%), (29.16%), (11.11%), (6.66%), (18.75%), and (28.57%) in lactating cows of A, B, C, D, E, F, G, H, I, J, K, and L farms, respectively. The highest prevalence at the lactating cow level was found in Farm B (40%), while the lowest was recorded in Farm J (6.66%) (Table 3). All 86 CMT-positive milk samples at the quarter level were examined, and 80 (7.75%) were counted as positive for somatic cells (Table 5). Table 5. Number of somatic cells determined by the somatic cell count test.
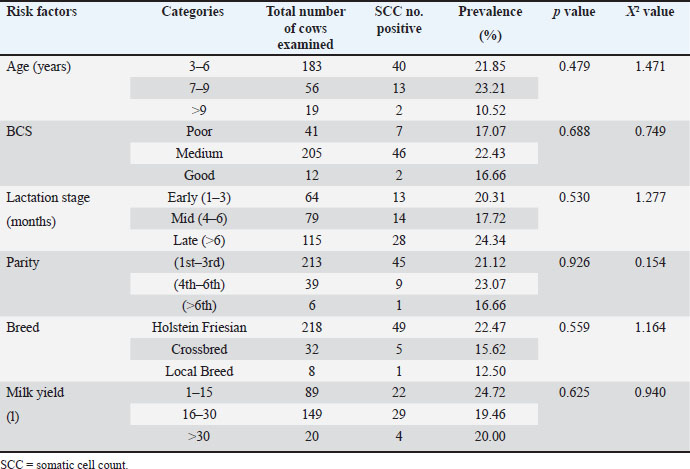
Risk factorsSeveral risk factors, including age, BCS, lactation stage, breed, milk yield, and parity, were analyzed for their association with SCM using SCC. Age-wiseThe prevalence of SCM across different age groups was recorded as 21.85% in the 3–6 years age group, 23.21% in the 7–9 years age group, and 10.52% in the >9 years age group, respectively (Table 6). Table 6. Risk factors related to the prevalence of SCM using SCC.
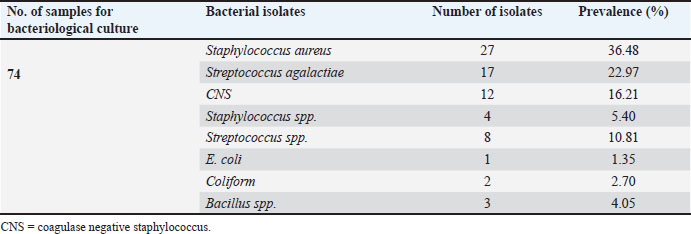
Body condition scoreThe prevalence of SCM based on the BCS was highest in cows with a medium BCS (22.43%), followed by those with poor (17.07%) and good (16.66%) BCSs (Table 6). Lactation stageThe overall prevalence of SCM based on lactation stages showed that all three stages were affected. It was 24.34%, 20.31%, and 17.72% in late, early, and mid-lactation, respectively (Table 6). ParityThe prevalence of SCM by parity was 23.07% in the 4th–6th parity group, followed by 21.12% in the 1st–3rd parity group, and 16.66% in the >6th parity group (Table 6). BreedAmong 218 Holstein Friesian cows, 49 (22.47%) tested positive for SCC. Out of 32 Crossbred cows, 5 (15.62%) tested positive for SCC, and of the 8 local breed cows, 1 (12.50%) tested positive (Table 6). Daily milk yieldBased on daily milk yield, the prevalence of SCM was 24.72% in cows producing 1–15 l per day, 19.46% in cows producing 16–30 l per day, and 20% in cows producing >30 l per day, respectively (Table 6). Statistical significanceNone of the potential risk factors showed a statistically significant association (p > 0.05) with the occurrence of SCM as measured by SCC (Table 6). Bacteriological culture (BC) resultOut of 86 CMT-positive quarters, 74 (7.17%) quarters were confirmed positive by BC (Table 9). Similarly, of the 63 CMT-positive cows, 56 (21.70%) were confirmed positive using BC (Table 3). Table 7. Prevalence of bacterial agents isolated from CMT positive cases.
Table 8. Bacterial isolates and mean SCC of SCM mammary quarters according to the bacterial type causing SCM in dairy cows.
Table 9. Risk factors related to the prevalence of SCM of BC.
Quarter-level prevalenceThe quarter-wise prevalence of SCM, as determined by BC, was 3.48%, 7.36%, 7.75%, and 10.07% in the RF, RH, LF, and LH quarters, respectively (Table 2). Cow-level prevalenceThe prevalence of SCM related to cow level, by using BC, was recorded as (30.55%), (40%), (29.41%), (10%), (14.89%), (12.50%), (20%), (29.16%), (11.11%), (6.66%), (18.75%), and (28.57%) in lactating cows of A, B, C, D, E, F, G, H, I, J, K, and L farms, respectively (Table 3). Bacterial isolationAll 86 CMT-positive milk samples at the quarter level were examined, and 74 (86.05%) were BC-positive. All BC-positive cases exhibited single-growth isolates. The isolated bacteria included Staphylococcus aureus (36.48%), Streptococcus agalactiae (22.97%), coagulase-negative staphylococci (CNS) (16.21%), Staphylococcus spp. (5.40%), Streptococcus spp. (10.81%), E. coli (1.35%), coliforms (2.70%), and Bacillus spp. (4.05%). The predominant isolated bacterium was S. aureus (Table 7). Effect of bacterial isolates on the mean SCCMean SCC of subclinical mastitic mammary quarters according to the type of bacteria are presented in Table 8. The mean SCC for E. coli and Coliform was (2,000 × 103 cells/ml, followed by S. aureus (1923.56 × 103 cells/ml), S. agalactiae (1617.25 × 103 cells/ml), Bacillus spp. (1,537×103 cells/ml), CNS (9506.36 × 103 cells/ml), Streptococcus spp. (9057.50 × 103 cells/ml), Staphylococcus spp. (621 × 103 cells/ml), and Unknown (1373.66 × 103 cells/ml) (Table 8). Risk factorsSeveral risk factors, including age, BCS, lactation stage, breed, milk yield, and parity, were analyzed for their association with SCM using BC. AgeIn the case of age-wise prevalence, the recorded prevalence of SCM in different age groups was 21.31% in the 3–6 years group, 25% in the 7–9 years group, and 15.78% in the >9-year-old group (Table 9). Body condition scoreThe prevalence of SCM was 22.92% in cows with a medium BCS, followed by those with a poor BCS (17.07%) and those with a good BCS (16.66%) (Table 9). Lactation stageThe prevalence of SCM based on the lactation stage in cows showed that all three lactation stages were affected; it was recorded (25.21%) in late lactation, followed by mid-lactation (18.98%) and early lactation (18.75%) (Table 9). ParityThe prevalence of SCM was recorded (25.64%) in the 4th–6th parity group, followed by the 1st–3rd parity group (21.12%) and the >6th parity group (16.66%) (Table 9). BreedAmong the 218 Holstein Friesian cows, 52 (23.85%) tested positive for SCM. Out of 32 crossbred cows, 3 (9.37%) tested positive, and out of 8 local breed cows, 1 (12.5%) tested positive (Table 9). Milk yieldBased on daily milk yield, the prevalence of SCM was 23.59% in cows producing 1–15 lper day, 20.80% in cows producing 16–30 l per day, and 20% in cows producing >30 l/day. None of the potential risk factors showed a statistically significant association (p > 0.05) with the occurrence of SCM as detected by the BC (Table 9). DiscussionSCM causes severe economic losses and decreases milk production on farms (Anueyiagu et al., 2022). The prevalence of SCM in different countries varies. Qayyum et al. (2016) and Khan et al. (2019) reported SCM prevalence rates of 21.96% and 67.3% in dairy farms in Pakistan, respectively. In Iran, it ranges from 20.83% (Naghshineh et al., 2015) to 42.5% (Hashemi et al., 2011), whereas in Bangladesh, it ranges from 28.50% (Kayesh et al., 2014) to 51.8% (Tripura et al., 2014); in Ethiopia 38.5% (Birhanu et al., 2017); in Rwanda, it ranges from 50.4 (Mpatswenumugabo et al., 2017) to 70% (Iraguha et al., 2017). In this study, the overall prevalence of SCM at the cow level using CMT was 24.41%, whereas that of SCC and BC was 21.70%. This is the first report on the prevalence of SCM in Kabul, Afghanistan. This report is in line with the findings of Naghshineh et al. (2015), Qayyum et al. (2016), and Yimam et al. (2020), who reported overall SCM prevalence rates of 20.83%, 21.8%, and 21.96%, respectively. In this study, the quarter-level prevalence of SCM for CMT was 8.33%, whereas it was 7.17% for SCC and BC. These findings are consistent with those of Yimam et al. (2020) and Qayyum et al. (2016), who reported almost the same percentages of quarter-level SCM prevalence. Other reports, such as Hashemi et al. (2011) and Bachaya et al. (2011), reported comparatively higher quarter-level prevalence of 21.6% and 35.25%, respectively. In this study, several risk factors, such as age, influenced the prevalence of SCM. The highest prevalence was observed in the age group of 7–9 years using the CMT or BC. This finding supports the results of Ayano et al. (2013) and Birhanu et al. (2017), who reported the higher prevalence of SCM in older cows (≥6 years). The increased prevalence of SCM in older cows may be attributed to their heightened susceptibility, which is likely associated with age-related structural alterations in the teat canal, such as dilation or incomplete closure due to repeated milking (Kibebew, 2017). Moreover, the mammary epithelium in older cows exhibits increased permeability, primarily due to tissue damage caused by previous inflammatory episodes (Król et al., 2013). The BCS has also been correlated with the prevalence of SCM, which affects the occurrence of mastitis. Increased SCM prevalence rates were found in the group of cattle with medium BCS. This finding contrasts with the results of Khan et al. (2019), who reported the highest prevalence in cows with a good BCS. The precise mechanisms underlying the increased SCM susceptibility of animals with medium BCS remain unclear and warrant further investigation. In this study, the lactation stage of cows influenced the prevalence of SCM. Increased prevalence was observed in cows in the late stage of lactation (>6 months), as determined by the CMT, SCC, and BC. The increased prevalence of mastitis during late lactation may result from cumulative and recurrent exposure to different microorganisms throughout the lactation stages (Almaw et al., 2008). Furthermore, the influence of the lactation phase on SCM could arise from unresolved chronic intramammary infections that were likely undiagnosed in prior lactation periods (Silva et al., 2021). This finding supports the results of Ndahetuye et al. (2019), who reported the highest prevalence in cows in the late stage of lactation, and aligns with the findings of Kayesh et al. (2014), who reported the highest prevalence in cows in the mid-stage of lactation. Parity was identified as an important factor influencing SCM prevalence. In this study, a higher prevalence was observed in cows with 4th–6th parity. This finding contrasts with the results of Birhanu et al. (2017), who reported the highest prevalence in cows with ≥7 parity. In this study, only six cows were included in the group with >6 parity. Therefore, the limited sample size may have affected the reliability of the findings, and the results may differ with a larger and more representative population. Breed was also correlated with the prevalence of SCM in the present study. A higher prevalence rate was observed in Holstein Friesian cows compared to crossbred cows using CMT, SCC, or BC. This finding supports the results of Ndahetuye et al. (2019), who reported similar trends. Holstein-Friesian cows appear to exhibit a higher genetic predisposition to mastitis than other breeds (Washburn et al., 2002). This increased vulnerability is thought to be linked to specific genetic traits associated with milk production and udder conformation, which may compromise their natural defense mechanisms against intramammary infections (Shaheen et al., 2016). The daily milk yield was another factor that influenced the prevalence of SCM. In this study, a higher prevalence was observed in cows with a daily milk yield of 1–15 l using CMT, SCC, or BC. The findings of our study contrast with those of Sanotharan et al. (2016), Biswas and Sarker (2017), and Taponen et al. (2006), who reported a higher prevalence of SCM in high-producing cattle. In our study, cows with a milk yield of 1–15 l may suffer from chronic SCM, which could negatively affect their production. The most frequently isolated pathogens were Staphylococcus species [S. aureus, CNS, and other Staphylococcus spp.], followed by Streptococcus species (S. agalactiae and other Streptococcus spp.), Bacillus spp., coliforms, and E. coli. These findings are consistent with those of Birhanu et al. (2017) and Kayesh et al. (2014), who also identified Staphylococcus spp. as the most prevalent pathogen, followed by Streptococcus spp. and E. coli. Reports from Bortolami et al. (2015) and Sztachańska et al. (2016) provide support for the results of this study. The current study revealed a high prevalence of S. aureus (36.48%). Khan and Khan (2006) reported that S. aureus is the leading cause of mastitis in dairy cattle. Furthermore, S. aureus can be transmitted from cow to cow (Zadoks et al., 2011) or via the milking process (Khasapane et al., 2023). In contrast, the prevalence of E. coli in this study was relatively low (1.35%), which aligns with the findings of Mpatswenumugabo et al. (2017), who reported a similarly low prevalence of E. coli (1.5%). The lower prevalence of SCM caused by E. coli may be attributed to its predominantly clinical nature and typically short infection duration (Smith, 1986). ConclusionThis study showed that SCM is highly prevalent among lactating dairy cows in Kabul City, using CMT, SCC, and BC. S. aureus, CNS, and S. agalactiae were the most frequently isolated pathogens, indicating a predominance of contagious agents. These findings highlight the urgent need for routine screening, improved hygiene during milking, and effective mastitis control programs to reduce the economic impact of this disease on Kabul’s dairy farms. AcknowledgmentsThe authors are especially grateful to the members of the Central Diagnostic Veterinary Laboratory for providing a conducive academic environment in the laboratory and resources that facilitated the completion of this research. Conflict of interestThe authors declare no conflict of interest. FundingThe authors did not receive any financial support or funding for this work. Authors’ contributionsMMT conceptualized the research framework and prepared the final manuscript. MAS performed the experiments, collected the data, and drafted the preliminary manuscript. Both MMT and MAS conducted the data analysis. All authors reviewed and approved the final manuscript. Data availabilityAll data are provided in the manuscript. Any additional data that support the findings of this study are available from the corresponding author upon reasonable request. ReferencesAbed, A.H., Menshawy, A.M.S., Zeinhom, M.M.A., Hossain, D., Khalifa, E., Wareth, G. and Awad, M.F. 2021. Subclinical mastitis in selected bovine dairy herds in North Upper Egypt: assessment of prevalence, causative bacterial pathogens, antimicrobial resistance and virulence-associated genes. Microorganisms 9(6), 1175. Ahmadi, H., Sahak, A.S., Ayoobi, A.W., Pekkan, E., Inceo Glu, M. and Karsli, F. 2023. Application of GIS-Based AHP Model for the Impact Assessment of COVID-19 Lockdown on Environment Quality: the Case of Kabul City, Afghanistan. J. Indian Soc. Remote Sens. 51, 439–452. Almaw, G., Zerihun, A. and Asfaw, Y. 2008. Bovine mastitis and its association with selected risk factors in smallholder dairy farms in and around Bahir Dar, Ethiopia. Trop. Anim. Health Prod. 40(6), 427–432. Anueyiagu, K.N., Agu, G.C., Ayanbimpe, G.M. and Ike, E.I. 2022. Bacterial etiologies of subclinical mastitis in cows. Fol. Vet. 66, 49–55. Ayano, A.A., Hiriko, F., Simyalew, A.M. and Yohannes, A. 2013. Prevalence of subclinical mastitis in lactating cows in selected commercial dairy farms of Holeta district. J. Vet. Med. Anim. Health 5, 67–72. Bachaya, H.A., Raza, M.A., Murtaza, S. and Akbar, I.U.R. 2011. Subclinical bovine mastitis in Muzaffar Garh district of Punjab (Pakistan). J. Anim. Plant Sci. 21, 16–19. Birhanu, M., Leta, S., Mamo, G. and Tesfaye, S. 2017. Prevalence of bovine subclinical mastitis and isolation of its major causes in Bishoftu Town, Ethiopia. BMC. Res. Notes 10, 767. Biswas, D. and Sarker, T. 2017. Prevalence of sub-clinical mastitis at Banaripara Upazilla, Barisal. Bangladesh. J. Vet. Med. 15, 21–26. Bortolami, A., Flore, E., Gianesella, M., Corro, M., Catania, S. and Morgante, M. 2015. Evaluation of the udder health status in subclinical mastitis affected dairy cows through bacteriological culture, somatic cell count and thermographic imaging. Pol. J. Vet. Med. 18(4), 799–805. El-Behiry, A., Elsayed, M., Marzouk, E. and Bathich, Y. 2015. Detection of virulence genes in Staphylococcus aureus and Streptococcus agalactiae isolated from mastitis in the Middle East. Bir. Microbio. Res. J. 10(3), 1–9. Hashemi, M., Kafi, M. and Safdarian, M. 2011. The prevalence of clinical and subclinical mastitis in dairy cows in the central region of Fars province, south of Iran. Iran. J. Vet. Res. 12(3), 236–241. Iraguha, B., Hamudikuwanda, H., Mushonga, B., Kandiwa, E. and Mpatswenumugabo, J.P. 2017. Comparison of cow-side diagnostic tests for subclinical mastitis of dairy cows in Musanze district, Rwanda. J. S. Afr. Vet. Assoc. 21(88), e1–e6. Islam, M., Islam, M., Islam, M., Rahman, M. and Islam, M. 2011. Prevalence of subclinical mastitis in dairy cows in selected areas of Bangladesh. Bangl. J. Vet. Med. 9(1), 73–78. Kaki, A.A., Djebala, S., Latif, M.B. and Moula, N. 2019. Evaluation of the prevalence of subclinical mastitis in dairy cattle in the Soummam Valley (Bejaia, Algeria). Bull. UASVM. Vet. Med. 76(5), 143–148. Kayesh, M.E.H., Talukder, M. and Anower, A.K.M.M. 2014. Prevalence of subclinical mastitis and its association with bacteria and risk factors in lactating cows of Barisal district in Bangladesh. Int. J. Biol. Res. 2(2), 35–38. Khan, A., Durrani, A.Z., Yousaf, A., Khan, J.A., Chaudhry, M., Khan, M.A., Habibunnabi, H. and Khan, A. 2019. Epidemiology of Bovine Sub-Clinical Mastitis in Pothohar Region, Punjab, Pakistan in 2018. Pak. J. Zool. 51(5), 1667–1674. Khan, M.Z. and Khan, A. 2006. Basic facts of mastitis in dairy animals: review. Pak. Vet. J. 26, 204–208. Khasapane, N.G., Byaruhanga, C., Thekisoe, O., Nkhebenyane, S.J. and Khumalo, Z.T.H. 2023. Prevalence of subclinical mastitis, its associated bacterial isolates and risk factors among cattle in Africa: a systematic review and meta-analysis. BMC. Vet. Res. 12(1), 123. Kibebew. and K. 2017. Bovine mastitis: a review of causes and epidemiological point of view. J. Biol. Agric. Healthc. 7(2), 1–14. Król, J., Brodziak, A., Litwińczuk, Z. and Litwińczuk, A. 2013. Effect of age and stage of lactation on whey protein content in milk of cows of different breeds. J. Vet. Sci. 16(2), 395–397. M, S. and Ha, T. 2016. A treatise on bovine mastitis: disease and disease economics, etiological basis, risk factors, impact on human health, therapeutic management, prevention and control strategy. Adv. Dairy Res. 4(1), 1–10. Mohammad, G. 2012. Prevalence assessment of Staphylococcus aureus and Streptococcus agalactiae by multiplex polymerase chain reaction (M-PCR) in bovine sub-clinical mastitis and their effect on somatic cell count (SCC) in Iranian dairy cows. Afr. J. Microbiol. Res. 6(12), 3005–3010. Mpatswenumugabo, J.P., Bebora, L.C., Gitao, G.C., Mobegi, V.A., Iraguha, B., Kamana, O. and Shumbusho, B. 2017. Prevalence of subclinical mastitis and distribution of pathogens in dairy farms of Rubavu and Nyabihu Districts, Rwanda. J. Vet. Med. 8456713, pages. Naghshineh, S., Rafat, S.A., Shoja, J., Moghaddam, G.A. and Ebrahimi, M. 2015. Prevalence and risk factors of subclinical mastitis in Iranian Holstein cows. Iran. J. Appl. Anim. Sci. 5(3), 569–574. Ndahetuye, J.B., Persson, Y., Nyman, A.K., Tukei, M., Ongol, M.P. and Båge, R. 2019. Aetiology and prevalence of subclinical mastitis in dairy herds in peri-urban areas of Kigali in Rwanda. Trop. Anim. Health Prod. 51(7), 2037–2044. Qayyum, A., Khan, J.A., Hussain, R., Avais, M., Ahmed, N., Khan, A. and Khan, M.S. 2016. Prevalence and Association of Possible Risk Factors with Sub-Clinical Mastitis in Cholistani Cattle. Pak. J. Zool. 48, 519–525. Sanotharan, N., Pagthinathan, M. and Nafees, M.S.M. 2016. Prevalence of bovine subclinical mastitis and its association with bacteria and risk factors in milking cows of Batticaloa district in SriLanka. Int. J. Sci. Res. Innov. Technol. 3, 137–150. Sayeed, M.A., Rahman, M.A., Bari, M.S., Islam, A., Rahman, M.M. and Hoque, M.A. 2020. Prevalence of sub-clinical mastitis and associated risk factors at cow level in dairy farms in Southwestern part of Bangladesh. Adv. Anim. Vet. Sci. 8(s2), 12–121. Shaheen, M., Tantary, H. and Nabi, S. 2016. A treatise on bovine mastitis: disease and disease economics, etiological basis, risk factors, impact on human health, therapeutic management, prevention and control strategy. Adv Dairy Res. 4, 1–10. Silva, A.C., Laven, R. and Benites, N.R. 2021. Risk factors Associated with Mastitis in Smallholder dairy farms in Southeast Brazil. Ani 11(7), 2089. Smith, K.L. 1986. A practical look at environmental mastitis. Bov. Prac. 21, 73–76. Sztachańska, M., Barański, W., Janowski, T., Pogorzelska, J. and Zduńczyk, S. 2016. Prevalence and etiological agents of subclinical mastitis at the end of lactation in nine dairy herds in North-East Poland. Polish J. Vet. Sci. 19(1), 119–124. Taponen, S., Simojoki, H., Haveri, M., Larsen, H. and Pyorala, S. 2006. Clinical characteristics and persistence of bovine mastitis caused by different species of coagulase-negative staphylococci identified with API or AFLP. Vet. Microbiol. 115, 199–120. Tripura, T.K., Sarker, S.C., Roy, S.K., Parvin, M.S., Sarker, R.R., Rahman, A.K.M.A. and Islam, M.T. 2014. Prevalence of subclinical mastitis in lactating cows and efficacy of intra mammary infusion therapy. Bangladesh J. Vet. Med. 12(1), 55–61. Washburn, S.P., White, S.L., Green, J.T. and Benson, G.A. 2002. Reproduction, mastitis, and body condition of seasonally calved Holstein and Jersey cows in confinement or pasture systems. J. Dairy Sci. 85(1), 105–111. Williamson, J., Callaway, T., Rollin, E. and Ryman, V. 2022. Association of Milk Somatic Cell Count with Bacteriological Cure of Intramammary Infection—A Review. Agriculture 12(9), 1437. Yimam, T.M., Kasse, G.E. and Yitie, M.T. 2020. Study on Prevalence of Bovine Subclinical Mastitis and Associated Risk Factors in Smallholder Dairy Farms of Mecha District, West Gojam, Ethiopia. Int J. Epidemiol. 4(5), 00016. Zadoks, R.N., Middleton, J.R., McDougall, S., Katholm, J. and Schukken, Y.H. 2011. Molekular epidemiology of mastitis pathogens of dairy cattle and comparative relevance to humans. J. Mammary Gland Biol. 164, 357–372. | ||
| How to Cite this Article |
| Pubmed Style Saidee MA, Tawfeeq MM. Prevalence of subclinical mastitis in dairy cows in Kabul, Afghanistan. Open Vet. J.. 2025; 15(10): 5058-5068. doi:10.5455/OVJ.2025.v15.i10.24 Web Style Saidee MA, Tawfeeq MM. Prevalence of subclinical mastitis in dairy cows in Kabul, Afghanistan. https://www.openveterinaryjournal.com/?mno=258421 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.24 AMA (American Medical Association) Style Saidee MA, Tawfeeq MM. Prevalence of subclinical mastitis in dairy cows in Kabul, Afghanistan. Open Vet. J.. 2025; 15(10): 5058-5068. doi:10.5455/OVJ.2025.v15.i10.24 Vancouver/ICMJE Style Saidee MA, Tawfeeq MM. Prevalence of subclinical mastitis in dairy cows in Kabul, Afghanistan. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5058-5068. doi:10.5455/OVJ.2025.v15.i10.24 Harvard Style Saidee, M. A. & Tawfeeq, . M. M. (2025) Prevalence of subclinical mastitis in dairy cows in Kabul, Afghanistan. Open Vet. J., 15 (10), 5058-5068. doi:10.5455/OVJ.2025.v15.i10.24 Turabian Style Saidee, Muhammad Ajmal, and Mohammad Monir Tawfeeq. 2025. Prevalence of subclinical mastitis in dairy cows in Kabul, Afghanistan. Open Veterinary Journal, 15 (10), 5058-5068. doi:10.5455/OVJ.2025.v15.i10.24 Chicago Style Saidee, Muhammad Ajmal, and Mohammad Monir Tawfeeq. "Prevalence of subclinical mastitis in dairy cows in Kabul, Afghanistan." Open Veterinary Journal 15 (2025), 5058-5068. doi:10.5455/OVJ.2025.v15.i10.24 MLA (The Modern Language Association) Style Saidee, Muhammad Ajmal, and Mohammad Monir Tawfeeq. "Prevalence of subclinical mastitis in dairy cows in Kabul, Afghanistan." Open Veterinary Journal 15.10 (2025), 5058-5068. Print. doi:10.5455/OVJ.2025.v15.i10.24 APA (American Psychological Association) Style Saidee, M. A. & Tawfeeq, . M. M. (2025) Prevalence of subclinical mastitis in dairy cows in Kabul, Afghanistan. Open Veterinary Journal, 15 (10), 5058-5068. doi:10.5455/OVJ.2025.v15.i10.24 |