| Research Article | ||
Open Vet. J.. 2025; 15(10): 5041-5047 Open Veterinary Journal, (2025), Vol. 15(10): 5041-5047 Research Article Effects of boiled fenugreek seed extract on testicular histology and reproductive parameters in adult male miceSamira Musa Sasi1*, Nagia Musa Alghoul2, Ragil Angga Prastiya3 and Khalid Salem41Department of Zoology, Faculty of Science, University of Tripoli, Tripoli. Libya 2Libyan Center of Biotechnology Researches, Tripoli, Libya 3Faculty of Health, Medicine and Life Sciences (FIKKIA), Universitas Airlangga, Indonesia 4Department of Plant Biotechnology, Genetic Engineering and Biotechnology Research Institute, University of Sadat City, Sadat City, Egypt *Corresponding Author: Evan L. Khlef. Department of Physiology and Microbiology, College of Medicine, Hawler Medical University, Erbil, Iraq. Email: evan.latef [at] hmu.edu.krd Submitted: 15/05/2025 Revised: 15/09/2025 Accepted: 28/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
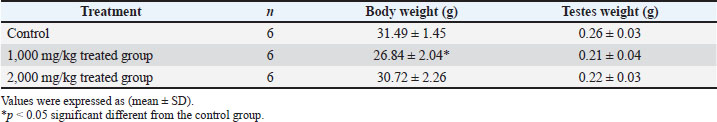
AbstractBackground: Fenugreek is a medicinal plant widely used in food and traditional medicine in several countries, including Libya. Aim: This study aimed to evaluate the effects of boiled aqueous fenugreek seed extract on sperm parameters, testicular histology, and embryo morphology in male mice. Method: Adult male mice were randomly divided into three groups (n=6 per group). The control group received distilled water orally, whereas the treatment groups received boiled fenugreek seed extract at doses of 1,000 and 2,000 mg/kg body weight for 21 consecutive days. After treatment, body and testis weights were recorded, and serum testosterone levels, sperm motility, and morphology were evaluated. Testicular histology was examined, and embryo morphology was assessed by mating treated males with untreated females. Results: A significant decrease in body weight was observed in the 1,000 mg/kg treated group. However, no significant differences in testis weight, testosterone levels, sperm motility, or morphology were detected between the treated and control groups. Histological examination revealed no pathological changes in the testicular tissues. In addition, embryos sired by treated males showed no external abnormalities or significant differences in body or tail length compared with controls. Conclusion: Boiled aqueous fenugreek seed extract at doses of 1,000 and 2,000 mg/kg did not exert adverse effects on male reproductive parameters or embryo morphology. Further studies are recommended to evaluate its safety on other body organs and its potential therapeutic applications. Keywords: Trigonella foenum-graecum, Male mice, Embryo morphology, Sperm parameters, Testes. IntroductionFenugreek (Trigonella foenum-graecum) is an annual plant belonging to the Fabaceae family, known for its potential nutraceutical and medicinal properties (Laila et al., 2022; Tewari et al., 2024). It is cultivated in various regions, including Turkey, Egypt, Canada, North Africa, Western Asia, and India, and is highly adaptable to different environmental and geographical conditions, which contributes to its widespread distribution (Ouzir et al., 2016; Tewari et al., 2024). Fenugreek is primarily produced in India, China, Pakistan, and Palestine (Ben Hameid et al., 2019). Its leaves are rich in vitamin C, minerals, and proteins, while the seeds contain various bioactive compounds, including saponins, alkaloids, oils, mucilage, galactomannan, essential amino acids (such as methionine, tryptophan, lysine, arginine, valine, and threonine), and vitamins A, C, D, and B1 (Elnaz et al., 2010; Tewari et al., 2024). Fenugreek seeds are also a good source of dietary fibers (Priyadarshini and Brar, 2022), steroidal compounds (Srivastava et al., 2022), proteins (Feyzi et al., 2015; Hassan Ali et al., 2021), and fatty acids (Aljuhaimi et al., 2018). Numerous studies have demonstrated pharmacological activities of the plant, including anti-inflammatory (Liu et al., 2012), antidiabetic (Shrivastava and Sharma, 2022), antioxidant (Srinivasan, 2014; Gawade et al., 2022), anticancer (Chatterjee et al., 2012), and anti-obesity effects (Reimer and Russell, 2008; Geetha et al., 2011). Additionally, it has been shown to exert protective effects against hepatic (Ibrahim and Osman, 2017; Fatima Zaidi and Masood, 2020) and gastric damage (Ghosal et al., 2016; Sankarapillai et al., 2018; Selmi et al., 2020). Fenugreek has also been reported to modulate antioxidant enzyme activity and reduce oxidative stress (Varshney and Siddique, 2023), improve female reproductive health (Singh, N et al., 2022), regulate sex hormone levels, and enhance sperm morphology in male volunteers (Maheshwari et al., 2017). Furthermore, it stimulates immune function by increasing the phagocytic capacity of macrophages in mice (Palka et al., 2021), is traditionally used to treat leg edema (Varshney and Siddique, 2023), and is known to promote lactation (Yassin, 2012). Both aqueous and methanolic extracts of fenugreek seeds have demonstrated antimicrobial activity against fungi and bacteria (Haouala et al., 2008; Al-Timimi, 2019; Faisal, 2022). Several studies have investigated the reproductive effects of fenugreek. For example, seed extracts have been shown to decrease estrogen and progesterone levels in female rabbits (Kassem et al., 2006), retard postnatal pup growth in mice (Khalki et al., 2010), and increase sperm count and motility in male mice (Al-Yahya, 2019). Administration of 100 mg/day of steroidal fenugreek extract resulted in decreased alkaline and acid phosphatase activity in the ovary and uterus in female rats (Sharma and Bhinda, 2005). Additionally, Oufquir et al. (2020) reported that oral administration of lyophilized aqueous extract of germinated fenugreek at 500, 800, and 1,000 mg/kg/day from gestation day 0 to postnatal day 0 induced spontaneous abortion and neurobehavioral disorders in offspring. Despite its wide use and reported biological effects, there is a lack of data on the influence of fenugreek seed extract on male reproductive function in Libya. Therefore, this study was conducted to evaluate the effects of boiled aqueous fenugreek seed extract on sperm parameters, testicular histology, serum testosterone levels, and embryo morphology in adult male mice. Materials and MethodsPreparation of boiled fenugreek seeds extractThe boiled fenugreek seed extract was prepared by adding 16 g of seeds to 100 ml of distilled water and boiling the mixture for 5 minute. The decoction was allowed to cool at room temperature and then filtered using Whatman filter paper No. 4. The resulting filtrate was used to prepare treatment doses of 1,000 and 2000 mg/kg body weight ( BW). Experimental animalsThis study was conducted using 18 adult Swiss albino male mice and 18 adult females weighing between 25 and 30 g. All animals were housed in plastic cages under standard laboratory conditions (22°C–25°C, 12-hour light/dark cycle) at the Animal house, Department of Zoology, Faculty of Science, University of Tripoli, Libya. The animals had free access to a standard pellet diet and water ad libitum. Treatment designMice were divided into three groups; each group is made up of six males. The first group (control) received distilled water orally, and the second and third groups were administered the aqueous extract of boiled fenugreek seeds at doses of 1,000 and 2,000 mg/kg BW, respectively, for 21 days. Samples collectionAt the end of the experiment, the mice were weighed, killed by cervical dislocation, and dissected. Testes were removed, weighed, and preserved immediately in 10% formalin solution for histological examination. Preparation of sperm suspensionThe vas deferens was excised and gently teased into a Petri dish containing 1 ml of 0.9% saline solution. The suspension was incubated at 37°C for 10 minute. After thorough mixing, a drop of the suspension was placed on a counting slide and left to settle for 5 minute before examination under a light microscope at 400× magnification. Sperm motility was assessed according to the method of Soheir and Haya (2013). Sperm morphology was assessed using smears stained with hematoxylin and eosin, and the percentage of abnormal spermatozoa was recorded (Bernardino et al., 2019). Histological examination of testisThe testicular tissues were processed for histological examination. The samples were dehydrated in ascending concentrations of ethanol, cleared in xylene, and embedded in paraffin wax. Sections of 5 µm thickness were obtained using a rotary microtome, mounted on slides, and stained with hematoxylin and eosin. Sections were examined using a light microscope at 400× magnification for structural evaluation and photomicrography (Bancroft and Gamble, 2002). Determination of the testosterone levelBlood samples were collected from the facial vein into Ethylene diamine tetra-acetic acid tubes and centrifuged at 3,000 rpm for 10 minute. The serum was separated and stored for testosterone analysis, which was conducted using enzyme-linked immunosorbent assay kits according to the manufacturer’s instructions (Bio String). Assessment of embryo morphologyIn the second week of treatment, untreated female mice were put with treated males at a ratio (two females: one male) for mating. The presence of a vaginal plug was recorded as day 0 of pregnancy. On day 18 of gestation, pregnant females were euthanized, and embryos were collected. The body and tail lengths were measured, and external malformations were examined and recorded. Statistical analysisData were expressed as mean ± SD. Statistical comparisons among groups were performed using one-way analysis of variance (ANOVA), followed by Duncan’s post hoc test using SPSS software (version 25). A p-value of <0.05 was considered statistically significant. Ethical approvalThe ethics committee of University of Tripoli has issued an ethical clearance (SREC 16-2024). ResultsBody and testis weights of male mice exposed to boiled fenugreek seed extractA significant decrease in mean body weight was observed in the group treated with 1,000 mg/kg of boiled fenugreek seed extract compared with the control group (p < 0.05). However, no significant difference was found between the control and the 2,000 mg/kg group. No significant differences in testis weight were observed across all groups (Table 1). Table 1. Body and testis weights of male mice exposed to boiled fenugreek seed extract.
Sperm motility and morphologySperm motility showed a slight increase but was non-significant in both treated groups compared with the control group (p > 0.05). Similarly, the percentage of abnormal spermatozoa slightly decreased in the treated groups, but the differences were not statistically significant (p > 0.05) (Table 2). Table 2. Sperm motility and morphology in male mice exposed to boiled fenugreek seed extract.
Serum testosterone levelNo significant differences in serum testosterone levels were observed between the control and treated groups (p > 0.05), although a non-significant elevation was noted in both treatment groups (Table 3). Table 3. Serum testosterone level in male mice exposed to boiled fenugreek seed extract.
Testicular histologyHistological examination of testicular sections revealed normal seminiferous tubules in all groups. Seminiferous tubules in the control group displayed well-organized spermatogenic cells, including spermatogonia, spermatocytes, spermatids, and abundant spermatozoa in the lumen, with Leydig cells present in the interstitial spaces (Fig. 1A). Testes from both treatment groups (1,000 and 2,000 mg/kg) exhibited similar histoarchitecture without any observable pathological alterations (Fig. 1B and C).
Fig. 1. Photomicrographs of testicular sections from adult male mice. (A) Control group showing normal seminiferous tubules lined with intact spermatogenic epithelium, including spermatogonia, spermatocytes, and spermatids (double headed arrow), with visible spermatozoa in the lumen. Interstitial spaces contain Leydig cells (indicated by S). (B) The treated group receiving 1,000 mg/kg of boiled fenugreek seed extract showing similar testicular architecture as the control group. (C) The treated group receiving 2,000 mg/kg of boiled fenugreek seed extract also showing normal histoarchitecture comparable to the control. Sections were stained with hematoxylin and eosin, magnification 400×. Embryo morphologyExternal morphological evaluation of embryos from untreated females mated with treated males revealed no abnormalities. No significant differences were observed in the body or tail lengths of embryos across the groups (p > 0.05) (Table 4). Table 4. Body and tail length of embryos sired by male mice received boiled fenugreek seed extract.
DiscussionThis study demonstrated that oral administration of 1,000 mg/kg boiled fenugreek seed extract for 21 days resulted in a significant reduction in body weight in male mice. This finding contrasts with previous studies that reported no significant effect of fenugreek seed extract on body weight in rats and mice (Singh and Verma, 2021; DeSouza-Ortolan et al., 2022). The discrepancy may be attributed to differences in extract preparation, dose, duration of exposure, or individual metabolism variation. The presence of galactomannan fibers in fenugreek, which are known to induce satiety and reduce food intake, is one possible explanation for the observed weight loss (Bahnasy et al., 2020). Despite the change in body weight, the testis weights remained unaffected in both treatment groups. This result aligns with the findings of DeSouza-Ortolan et Faisal (2022), who reported no significant impact of fenugreek on testicular mass. In contrast, some studies have shown a reduction in testicular weight following exposure to fenugreek (Kassem et al., 2006; Singh and Verma, 2021). Variations among these findings may be due to differences in extraction methods, duration, or animal species used. Testis weight is often used as a biomarker of reproductive health, given its correlation with spermatogenesis and steroidogenesis (Geeta and Jain, 2017). Evaluation of sperm parameters revealed no significant effects of boiled fenugreek seed extract on sperm motility or morphology. These findings are consistent with those of Hind et al. (2017) and DeSouza-Ortolan et al. (2022), who also observed no significant alterations in sperm characteristics after treatment with aqueous fenugreek extracts. However, other studies have reported detrimental effects, such as reduced motility or increased abnormal sperm morphology, following prolonged or higher-dose administration (Abdulaziz, 2019; Singh, A et al., 2022). The lack of significant changes in our study may reflect the relatively short exposure period or the stability of the boiled aqueous extract. Sperm motility and morphology must be evaluated because of their influence on the fertilization process. According to Yamanaka et al. (2024), fertilization can only occur if the sperm can meet the ovum at the appropriate location, time, and condition to fuse with the egg. Therefore, sperm must have a normal morphology and progressive motility (straight-line movement) that allows them to navigate through the cervical mucus until the fallopian tube, where fertilization takes place by penetration of the outer surface of the ovum (Prastiya et al., 2024; Yamanaka et al., 2024). Serum testosterone levels in the treated groups showed a non-significant increase compared with controls. This observation supports the findings of Singh, A et al. (2022), who reported no hormonal disruption following aqueous extract administration of fenugreek seeds. Conversely, several studies have documented both increases (Rao et al., 2015; Nagulapalli Venkata et al., 2017; Badry et al., 2021) and decreases (Kassem et al., 2006) in testosterone levels, possibly due to different extraction solvents (aqueous vs. ethanolic or steroidal), dosages, or treatment durations. Since testosterone plays a crucial role in regulating male fertility, further investigation is warranted to elucidate the precise mechanisms of fenugreeks action on endocrine function. Histological analysis of the testes revealed normal seminiferous tubules and interstitial architecture in all groups, with no signs of degeneration, atrophy, or necrosis. These results are in agreement with those of Hind et al. (2017), who reported no histopathological lesions following treatment with aqueous extract of fenugreek. However, Singh, A et al. (2022), Singh and Verma (2021), and Kassem et al. (2006) documented structural abnormalities in testicular tissues, possibly due to different doses or extract types used. The preserved histological integrity observed in our study suggests that boiled fenugreek seed extract does not compromise spermatogenesis or testicular microstructure at the tested concentrations. Finally, the absence of teratogenic or morphological abnormalities in embryos sired by treated males indicates that fenugreek extract at the given doses does not adversely affect male fertility or embryonic development. These findings contrast with reports of fetal toxicity and neurodevelopmental alterations in offspring from fenugreek-treated females (Khalki et al., 2010; Abdulaziz, 2019; Oufquir et al., 2020). Another study revealed that administration of female rabbits with 30% of fenugreek capsules as diet orally for 3 months led to small sizes of fetuses (Kassem et al., 2006). The differences could stem from maternal versus paternal exposure routes and physiological responses. Nevertheless, the safety of fenugreek for reproductive health remains a subject of ongoing concern and should be further studied. Overall, the present study suggests that boiled fenugreek seed extract at doses of 1,000 and 2,000 mg/kg for 21 days, does not adversely affect male reproductive function or embryo morphology in mice. However, to confirm the safety profile of the extract. Further studies involving longer treatment periods, different extract types, and broader systemic evaluations are recommended. ConclusionIn conclusion, oral administration of 1,000 and 2,000 mg/kg of boiled fenugreek seed extract for 21 days did not produce adverse effects on testicular histology, sperm parameters, serum testosterone levels, or embryo morphology in male mice. Although a significant reduction in body weight was observed at the lower dose, the extract appeared to be well-tolerated in terms of reproductive function. These findings suggest that boiled fenugreek seed extract may be safe for short-term use at the tested concentrations. However, to confirm its broader safety and therapeutic potential, further studies are warranted, including longer treatment durations, a comprehensive evaluation of systemic toxicity, and assessment of its effects on other organs. AcknowledgmentsThe authors are grateful to University of Tripoli for providing the laboratory facilities necessary to achieve this research. Conflict of interestThe authors declare no conflict of interest. FundingNone. Authors’ contributionSMS and NMA designed, organized, and performed the experiments. SMS and NMA wrote the manuscript and conducted. All authors have read, reviewed, and approved the final manuscript. Data availabilityAll data are provided in the manuscript. ReferencesAbdulaziz, A.A.Y. 2019. Reproductive, cytological and biochemical toxicity of fenugreek in male Swiss albino mice. African J. Pharmacy Pharmacol. 7(29), 2072 -2080. Aljuhaimi, F., Simsek, S., Ozcan, M.M., Ghafoor, K. and Babiker, E.E. 2018. Effect of location on chemical properties, amino acid and fatty acid compositions of fenugreek (Trigonella foenum graecum L) seed and oils. J. Food Process Preservation 42(1), e13569. Al-Timimi, L.A.N. 2019. Antibacterial and anticancer activities of fenugreek seed extract. Asian Pac. J. Cancer Prev. 20(12), 3771–3776. Al-Yahya, A. 2019. Reproductive, cytological and biochemical toxicity of fenugreek in male Swiss albino mice. Afr. J. Pharm. Pharmacol. 7(29), 2072–2080. Badry, A., Elghait, A.A., Abo-youssef, A., Abdelwahab, N.S. and Helaly, H. 2021. A comparative study on the effects of the fenugreek seeds’ powder and its aqueous and oil extracts on the male reproductive system in albino rats. Bull. Pharm. Sci. Assuit. Univ. 44(2), 623–635. Bahnasy, R.M., Abd-Elhady, E.E.S. and Abd-Elfatah, M.H. 2020. The protective effect of seeds and sprouts of fenugreek and alfalfa on rats exposed to lead poisoning. Egypt. J. Nutr. Health. 15(1), 85–100. Bancroft, J. and Gamble, M. 2002. Theory and practice of histological techniques. 5th ed., Philadelphia: Churchill Livingstone, pp: 125–38. Ben Hameid, A., Al-Sindi, T., Allow, A., Nafie, E., Alahmad, B. and Faisal, G. 2019. Substantial effect of fenugreek seeds aqueous extract on serum estradiol level in ovarian hyper-stimulation syndrome rat model. Oman. Med. J. 34(3), 238–243. Bernardino, R.L., D’Souza, W.N., Rato, L., Rothstein, J.L., Dias, T.R., Chui, D., Wannberg, S., Alves, M.G. and Oliveira, P.F. 2019. Knockout of MCT1 results in total absence of spermatozoa, sex hormones dysregulation, and morphological alterations in the testicular tissues. Cell. Tissue. Res. 378(2), 333–339. Chatterjee, S., Kumar, M. and Kumar, A. 2012. Chemomodulatory effect of Trigonella foenum graecum (L) seed extract on two stage mouse skin carcinogenesis. Toxicol. Int. 19(3), 287–294. DeSouza-Ortolan, L., Vicentine, K.F.D. and Acrani, S. 2022. Effects of fenugreek (Trigonella foenum-graecum L) on daily sperm production, epididymal transit and sperm morphology of pubertal Wistar. Braz. J. Sci. 1(11), 65–72. Elnaz, H., Shamsali, R., Sayed, F., Reza, D.L. and Jalal, Z. 2010. Review on fenugreek therapy and phytochemical benefits. Med. Pants Magas. 2, 34. Faisal, Z.G. 2022. Evaluation of antibacterial and antifungal activity of Trigonella foenugræcum seeds and leaves against some of clinical bacteria and fungi. J. Edu. Sci. Stud. 2(19), 175–180. Fatima Zaidi, S.N. and Masood, J. 2020. The protective effect of fenugreek seeds extract supplementation on glucose and lipid profile in thioacetamide induced liver damage in rats. Pak. J. Pharm. Sci. 33(5), 2003–2008. Feyzi, S., Varidi, M., Zare, F. and Varidi, M.J. 2015. Fenugreek (Trigonella foenum graecum) seed protein isolate: extraction optimization, amino acid composition, thermos and functional properties. J. Sci. Food. Agric. 95, 3165–3176. Gawade, M.B., D. Shejul, D. and Jagtap, S. 2022. Trigonella foenum graecum (Fenugreek): an herb with impressive health benefits and pharmacological therapeutic effects. Asian Food Sci. J. 21(3),19–28. Geeta, P. and Jain, G.C. 2017. Aluminium chloride-induced testicular effects in rats: a histomorphometrical study. Asian. J. Appl. Sci. Technol. 1(9), 46–52. Geetha, M., Reddy, S.K., Krupanidhi, A.M., Muralikrishna, K.S. and Patil, N.A. 2011. Effect of fenugreek on total body and organ weights. A study on mice. PharmacoL 3, 747–752. Ghosal, N., Firdaus, S.B., Naaz, S., Paul, S., Ghosh, A.K., Chattopadhyay A Mohan., Thakurdesai, P., Bhaskaran, S., Pattari, S. and Bandyopadhyay, D. 2016. Gastroprotective effect of fenugreek 4-hydroxyisoleuine and trigonelline enriched fraction (TF4H 28%) Sugaheal against indomethacin induced ulcer in male wister rats. J. Pharm. Res. 10(6), 351–364. Haouala, R., Hawala, S., El-Ayeb, A., Khanfir, R. and Boughanmi, N. 2008. Aqueous and organic extracts of Trigonella foenum graecum L. inhibit the mycelia growth of fungi. J. Environ. Sci. 20(12), 1453–1457. Hassan Ali, A., Yeasmin, T., Mohamed, Y.A., Mohamud, A.I. and Mishra, P. 2021. Evaluation of dietary supplementation of fenugreek seed (Trigonella foenum-graecum L.) as a growth promoter in broiler diet and its impacts on growth performance, carcass quality and cost effectiveness. J. Istanbul Vet. Sci. 5(1), 6–12. Hind, B., Zineb, M., Elbachir, H., Najat, E., Siham, A. and Driss. 2017. Evaluation of potential effects of the aqueous extract of fenugreek seeds on fertility in male rats. J. Ayurveda Herbal Med. 3(4), 210–215. Ibrahim, A.A. and Osman, A.A. 2017. Fenugreek downregulating caspase-3 VEGF expression as a therapeutic agent in a acetaminophen-induced hepatotoxicity in male rats. Merit. Res. J. Med. Sci. 5(3), 177–186. Kassem, A., Al-Aghbari, A., Al-Habori, M. and Al-Mamary, M. 2006. Evaluation of the potential antifertility effect of fenugreek seeds in male and female rabbits. Contraception 73(3), 301–306. Khalki, L., M’hamed, S.B., Bennis, M., Chait, A. and Sokar, Z. 2010. Evaluation of the developmental toxicity of the aqueous extract from Trigonella foenum-graecum (L) in mice. J. Ethnopharmacol. 131(2), 321–325. Laila, U., Albina, T., Zuha, S.S. and Tamang, H. 2022. Fenugreek seeds: nutritional composition and therapeutic properties. J. Pharm. Innov. 11(6S), 2417–2425. Liu, Y., Kakani, R. and Nair, M.G. 2012. Compounds in functional food fenugreek spice exhibit anti-inflammatory and antioxidant activities. Food. Chem. 131, 1187–1192. Maheshwari, A., Verma, N., Swaroop A Bagchi., Preuss, H.G., Tiwari, K. and Bagchi, D. 2017. Efficacy of furosap TM, a novel Trigonella foenum-graecum seed extract, in enhancing testosterone level and improming sperm profil in male volunteers. Int. J. Med. Sci. 14(1), 58–66. Nagulapalli Venkata, K.C., Swaroop, A., Bagchi, D. and Bishayee, A. 2017. A small plant with big benefits: fenugreek (Trigonella foenum-graecum Linn.) for disease prevention and health promotion. Mol. Nutr. Food Res. 61, 243–254. Oufquir, S., Ait Laaradia, M., El Gabbas, Z., Bezza, K., Laadraoui, J., Aboufatima, R., Sokar, Z. and Chait, A. 2020. Trigonella foenum-graecum L. Sprouted seed extract: its chemical HPLC analysis, abortive effect, and neurodevelopmental toxicity on mice. Evi. Based. Complement. Alter. Med. 3, 1–10. Ouzir, M., El Bairi, K. and Amzazi, S. 2016. Toxicological properties of fenugreek (Trigonella foenum graecum). Food Chem. Toxicol. 96, 145–154. Palka, S.E., Otwinowska-Mindur, A., Migdal, L., Kmiecik, M. and Wojtysiak, D. 2021. Effect of a diet supplement with nettle (Urtica dioica L.) or fenugreek (Trigonella foenum-graecum L) on the post-slaughter traits and meat quality parameters of termond white rabbits. Anim. (Basel). 11(6), 1566. Prastiya, R., Sardjito, T., Saputro, A., Budi, D., Maxdhameta, S., Sulistiyawati, E., Sulistyowati, D., Amaliya, A., Sasi, S. and Haryuni, N. 2024. Quality and kinematic characteristics of Bali bulls frozen semen with purified green tea extract epigallocatechin-3-gallate antioxidant addition in diluent. Open. Vet. J. 14(8), 2040–2048. Priyadarshini, S. and Brar, J.K. 2022. Biofortification of chromium in fenugreek seeds. J. Trace Elem. Med. Biol. 61, 126521. Rao, A., Steels, E., Beccaria, G., Inder, W.J. and Vitetta, L. 2015. Influence of a specialized Trigonella foenum graecum seed extract (Libifem), on testosterone, estradiol and sexual function in healthy menstruating women, a randomized placebo controlled study: libifem, influence on hormones and sexual function. Phytother. Res. 8, 1123–1130. Reimer, R.A. and Russell, J.C. 2008. Glucose tolerance, lipids, and GLP-1 secretion in JCR: lA-cp rats fed a high protein fiber diet. Obesity 16, 40–46. Sankarapillai, J., Chandrakumari, A.S. and Sinha, P. 2018. Effect of Trigonella foenum-graecum (fenugreek) seeds extract in experimental induced gastric ulcer in wister rats. Pharmacogen. J. 10(6), 1169–1173. Selmi, S., Alimi, D., Rtibi, K., Jedidi, S., Grami, D., Marzouki, L., Hosni, K. and Sebai, H. 2020. Gastro-protective and antioxidant properties of Trigonella foenum graecum seeds aqueous extract (fenugreek) and omeprazole against ethanol-induced peptic ulcer. J. Med. Food. 21, 1115–1112. Sharma, J.D. and Bhinda, A. 2005. Antifertility activity of steroidal extract of Trigonella foenum-graecum (seeds) in female rats. Asian. J. Exp. Sci. 19, 115–120. Shrivastava, A. and Sharma, N. 2022. Antidiabetic effect of fenugreek seeds in selected non-insulin dependent diabetic patients. Pharma Innov. J. 11, 1780–1883. Singh, A., Sarkar, D. and Singh, S.K. 2022. Effect of Trigonella foenum-graecum L. seed extract on the reproductive system of male mice and possible mechanism of its action on spermatogenesis. Andrologia 54(7), 1643–1659. Singh, M. and Verma, G.N. 2021. Investigating the effect of ethanolic extract of Trigonella foenum-graecum L. seeds on reproductive system of male albino rats. Highlights Med. Med. Res. 7, 77–85. Singh, N., Yadav, S.S., Kumar, S. and Narashiman, B. 2022. Ethno-pharmacological, phytochemical and clinicalk studies on fenugreek (Trigonella foenum-graecum L). Food Bio. Sci. 46, 101546. Soheir, A.E.N. and Haya, A.S. 2013. Effect of sapogenin extract on anti-infertility induced by aluminum chloride in male rats. Net. J. 1, 77–85. Srinivasan, K. 2014. Antioxidant potential of spice and their constituents. Crit. Rev. Food. Sci. Nutr. 54(3), 352–372. Srivastava, A., Singh, Z., Verma, V. and Choedon, T. 2022. Potential health benefits of fenugreek with multiple pharmacological properties. In Research Anthology on Recent Advancements in Ethnopharmacology and Nutraceuticals. IGI Global, Hershey, PA, pp: 672–87. Tewari, A., Singh, R. and Brar, J.K. 2024. Pharmacological and therapeutic properties of fenugreek (Trigonella foenum-graecum) seeds: a review. J. Phytopharmacol. 13(2), 97–104. Varshney, H. and Siddique, Y.H. 2023. Medical properties of fenugreek: a review. Open Biol. J. 11, 187503622302220. Yamanaka, T., Xiao, Z., Tsujita, N., Awad, M., Umehara, T. and Shimada, M. 2024. Testosterone-induced metabolic changes in seminal vesicle epithelial cells alter plasma components to enhance sperm fertility. eLife 13, P95541. Yassin, S.A.T. 2012. Herbal remedy used by rural adolescent girls with menstrual disorders. J. Am. Sci. 8, 467–473. | ||
| How to Cite this Article |
| Pubmed Style Sasi SM, Alghoul NM, Prastiya RA, Salem K. Effects of boiled fenugreek seed extract on testicular histology and reproductive parameters in adult male mice. Open Vet. J.. 2025; 15(10): 5041-5047. doi:10.5455/OVJ.2025.v15.i10.22 Web Style Sasi SM, Alghoul NM, Prastiya RA, Salem K. Effects of boiled fenugreek seed extract on testicular histology and reproductive parameters in adult male mice. https://www.openveterinaryjournal.com/?mno=258540 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.22 AMA (American Medical Association) Style Sasi SM, Alghoul NM, Prastiya RA, Salem K. Effects of boiled fenugreek seed extract on testicular histology and reproductive parameters in adult male mice. Open Vet. J.. 2025; 15(10): 5041-5047. doi:10.5455/OVJ.2025.v15.i10.22 Vancouver/ICMJE Style Sasi SM, Alghoul NM, Prastiya RA, Salem K. Effects of boiled fenugreek seed extract on testicular histology and reproductive parameters in adult male mice. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5041-5047. doi:10.5455/OVJ.2025.v15.i10.22 Harvard Style Sasi, S. M., Alghoul, . N. M., Prastiya, . R. A. & Salem, . K. (2025) Effects of boiled fenugreek seed extract on testicular histology and reproductive parameters in adult male mice. Open Vet. J., 15 (10), 5041-5047. doi:10.5455/OVJ.2025.v15.i10.22 Turabian Style Sasi, Samira Musa, Nagia Musa Alghoul, Ragil Angga Prastiya, and Khalid Salem. 2025. Effects of boiled fenugreek seed extract on testicular histology and reproductive parameters in adult male mice. Open Veterinary Journal, 15 (10), 5041-5047. doi:10.5455/OVJ.2025.v15.i10.22 Chicago Style Sasi, Samira Musa, Nagia Musa Alghoul, Ragil Angga Prastiya, and Khalid Salem. "Effects of boiled fenugreek seed extract on testicular histology and reproductive parameters in adult male mice." Open Veterinary Journal 15 (2025), 5041-5047. doi:10.5455/OVJ.2025.v15.i10.22 MLA (The Modern Language Association) Style Sasi, Samira Musa, Nagia Musa Alghoul, Ragil Angga Prastiya, and Khalid Salem. "Effects of boiled fenugreek seed extract on testicular histology and reproductive parameters in adult male mice." Open Veterinary Journal 15.10 (2025), 5041-5047. Print. doi:10.5455/OVJ.2025.v15.i10.22 APA (American Psychological Association) Style Sasi, S. M., Alghoul, . N. M., Prastiya, . R. A. & Salem, . K. (2025) Effects of boiled fenugreek seed extract on testicular histology and reproductive parameters in adult male mice. Open Veterinary Journal, 15 (10), 5041-5047. doi:10.5455/OVJ.2025.v15.i10.22 |