| Research Article | ||
Open Vet. J.. 2025; 15(10): 5032-5040 Open Veterinary Journal, (2025), Vol. 15(10): 5032-5040 Research Article Whole blood cholinesterase activity in humans and ruminants: Comparison using a modified electrometric methodMaha Ahmed Ramadhan1 and Fouad Kasim Mohammad2,3*1Department of Pharmacology, College of Pharmacy, University of Duhok, Kurdistan Region, Iraq 2Department of Physiology, Biochemistry and Pharmacology, College of Veterinary Medicine, University of Mosul, Mosul, Iraq 3College of Nursing, The American University of Kurdistan, Duhok, Kurdistan Region, Iraq *Corresponding Author: Fouad Kasim Mohammad. Department of Physiology, Biochemistry and Pharmacology, College of Veterinary Medicine, University of Mosul, Mosul, Iraq. Email: fkmohammad [at] uomosul.edu.iq; fouadmohammad [at] yahoo.com Submitted: 17/05/2025 Revised: 11/09/2025 Accepted: 21/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: Determination of normal whole blood cholinesterase (WBChE) activity is required in the assessment of exposure to organophosphate and carbamate pesticides in humans and animals. Aim: Using a modified electrometric method to comparatively measure normal WBChE activities in humans, sheep, goats, and cattle with their true Cholinesterase (ChE) components by inhibiting pseudo ChE activity in vitro using quinidine. Methods: The WBChE activities in humans, sheep, goats, and cattle were measured by a modified electrometric method using 7.1% acetylcholine iodide as a substrate in the reaction mixture. The mixture also contained whole blood samples (0.2 ml), distilled water (3 ml), and the pH 8.1 barbital-phosphate buffer (3 ml). The incubation period was 20 minutes in humans and 30 minutes in ruminants at 37ºC. Whole blood samples (8–10/species) were subjected to in vitro pseudo ChE inhibition using 50 µl of 1% quinidine sulfate with the objective of estimating the remaining true ChE in the blood. Results: The overall WBChE activity in humans (1.049 ± 0.411 Δ pH/20 minute) was found to be significantly higher than that observed in the sheep (0.595 ± 0.324 Δ pH/30 minute), goats (0.432 ± 0.226 Δ pH/30 minute), and cattle (0.693 ± 0.328 Δ pH/30 minute). The comparative median WBChE activities in humans and ruminants were 1.12, 0.565, 0.395, and 0.665, respectively. True ChE activity of humans was found to be significantly higher than that of ruminants, whereas the pseudo ChE of humans was found to be significantly higher than that of sheep and goats. Within each species, the pseudo ChE activity in the whole blood was consistently low (19%–26%) and significantly below the corresponding value of true ChE (74%–81%). Conclusion: The study presents a comparative modified electrometric measurement of normal WBChE activities between humans and ruminants. The results obtained demonstrate that human blood exhibits higher WBChE, pseudo ChE, and true ChE activities compared to those of ruminants. A comparative determination of WBChE activity using a simple modified electrometric method in humans and farm animals could serve as a valuable tool for collectively assessing cross-species exposure to pesticides. Keywords: Acetylcholinesterase, Pseudocholinesterase, Sheep, Goat, Cattle. IntroductionCholinesterase (ChE) activity in the blood is measured in three forms: plasma ChE (PChE), also called butyrylcholinesterase or pseudo ChE (E.C. 3.1.1.8), erythrocyte ChE (EChE), also known as acetylcholinesterase or true ChE (E.C. 3.1.1.7), and whole blood ChE (WBChE), which represents the combined activity of both enzymes (PChE and EChE) (Strelitz et al., 2014; Wilson, 2014). Cholinesterase catalyzes the hydrolysis of acetylcholine into choline and acetic acid to end the action of this neurotransmitter at neuronal endings (Taylor et al., 2009; Wilson, 2014). However, it is important to note that acetylcholinesterase rapidly hydrolyzes acetylcholine, whereas butyrylcholinesterase has the capacity to hydrolyze a wide range of substrates, including acetylcholine, butyrylcholine, succinylcholine, organophosphates, and local anesthetics containing esters (Wilson, 2014; Sridhar and Gumpeny, 2024). In addition to the blood (acting a sensitive biomarker of erythrocyte ageing), true ChE is mainly found in the nervous system and neuromuscular junctions, and its function is regulating cholinergic neurotransmission by rapid hydrolysis of acetylcholine after its release into the synaptic clefts (Taylor et al., 2009; Lazarevic-Pasti et al., 2017; Saldanha, 2017; Korabecny and Soukup, 2021). In contrast, the liver synthesizes pseudo ChE to be released into the plasma in order to modulate functional activities related to growth, neuronal integrity, cellular adhesions, and inflammatory response during disease conditions that involve the cardiovascular and endocrine systems, as well certain drug metabolism such as anesthetics (Oda, 2016; Andersson et al., 2019; Sridhar and Gumpeny, 2024; Mohammad et al., 2025). Both enzymes are variably inhibited by different pesticides (WHO, 1986; Worek et al., 1999; Wilson et al., 2005; Wilson, 2010; 2014; Ahmad et al., 2024). Organophosphates and carbamates are still used in many countries, including Iraq, as outdoor and indoor insecticides in public health, agriculture, and veterinary clinical practice (Odisho and Mohammad, 2022; Ahmad et al., 2024). Organophosphate pesticides have been demonstrated to irreversibly inhibit ChE activities in the blood and nervous tissues, whereas carbamate pesticides have been shown to inhibit them reversibly (Wilson, 2014; Ahmad et al., 2024). For this reason, the blood ChE inhibition is used to monitor exposure to these pesticides (Strelitz et al., 2014; Mohammad et al., 2024(. For this purpose, the determination of blood ChE activities for monitoring pesticide exposure is crucial for the health of agricultural workers (including farmers and veterinarians), individuals with occupational exposure, and farm animals such as sheep, goats, and cattle (Mohammad et al., 2007a; Furman, 2010; Strelitz et al., 2014; Wilson, 2014; Mohammad et al., 2024). A decline in human blood ChE activity by 20% is indicative of exposure to pesticides, while a 50% necessitates immediate action (Furman, 2010; Strelitz et al., 2014; Wilson, 2014; Ahmad et al., 2024). A variety of methods are available for the measurement of blood and tissue ChE activities, particularly the spectrophotometric method of Ellman and the electrometric method of Michel (Wills, 1972; Fairbrother et al., 1991; Mohammad, 2007; Mohammad et al., 2007b; Arrieta et al., 2009). The latter electrometric method has been modified and advocated by many investigators for rapidity and ease of application in humans, various animal species, and insects (Mohammad and St. Omer, 1982; Mohammad, 2007; Mohammad et al., 2007 b; Alias et al., 2011; Askar et al., 2011; Garmavy et al., 2023; Wongta et al., 2024, Kyi et al., 2025). The modified electrometric method was initially introduced by Mohammad and St. Omer (Mohammad and St. Omer, 1982), and it has been ever since used for monitoring exposure of humans and various animal species to organophosphate and carbamate pesticides (Ahmed and Mohammad, 2007; Mohammad et al., 2007 a; Mohammad et al., 2008; Mohammad et al., 2012; Mohammed and Mohammad, 2022; Odisho and Mohammad, 2022). The original and modified electrometric methods have been shown to correlate well with those of the Ellman method (Haigh et al., 2008; Askar et al., 2011; Kyi et al., 2025), thus making the modified electrometric method, currently advocated, a desirable choice for use due to its simplicity, accuracy, and considerable cost-effectiveness (Mohammad, 2007; Mohammad et al., 2007 b, 2007c, Garmavy et al., 2023). For this reason, the modified electrometric method has become widely used in Iraq and has also attracted international interest for measuring blood ChE activity in humans and various animal species (Mohammad 2007; Askar et al., 2011; Garmavy et al., 2023; Wongta et al., 2024; Kyi et al., 2025). This method has recently been the focus of several investigations (Tayseer and Jassim, 2022; Al-Abdaly et al., 2023; Garmavy et al., 2023; Mohammad et al., 2024, Raouf et al., 2024; Wongta et al., 2024; Kyi et al., 2025). A crucial consideration in the context of exposure of humans and animals to pesticides is the knowledge of baseline or reference blood ChE activities (Wilson et al., 2005; Mohammad et al., 2007 b; Furman, 2010; Lionetto et al., 2013; Wilson, 2014; Strelitz et al., 2014; Garmavy et al., 2023). Concurrent measurement of blood ChE activities of non-exposed subjects is an added advantage for the assessment of pesticide exposure (Wilson et al., 2005; Furman, 2010; Mohammad et al., 2023; 2024). Furthermore, EChE activity is higher than that of PChE in ruminants, contributing > 90% to WBChE activity, making the whole blood a suitable target for ChE determination in ruminants when evaluating exposure to pesticides (Mohammad and St. Omer, 1982; Abdelsalam and Ford, 1985; Cerón et al., 1999; Tecles et al., 2001; Al-Qarawi et al., 2003; Mohammad et al., 2006; Mohammad, 2007; Mohammad et al., 2007 c; Askar et al., 2011). As EChE activity functionally reflects synaptic acetylcholinesterase, WBChE, which contains the bulk of true ChE activity, could be the preferred enzyme source for measuring blood ChE activity under field conditions (Worek et al., 1999; Haigh et al., 2008; Furman, 2010; Rajapakse et al., 2011), especially when considering the possibility of differential ChE inhibition by pesticides (WHO, 1986; Wilson 2014; Moon et al., 2015; Ahmad et al., 2024). Measuring WBChE does not require the separation of plasma and erythrocytes, and it would save time for the initiation of the ChE assay, especially under urgent needs in the field (Worek et al., 1999; Tecles et al., 2001; Worek et al., 2016). The WBChE activity, measured by a modified electrometric method, has been documented in humans (Mohammad et al., 2007 b; Garmavy et al., 2023) and in the ruminants sheep, goats, and cattle (Mohammad et al., 2007 c). The present study builds upon existing literature by comparatively determining normal WBChE activity values in humans, sheep, goats, and cattle, utilizing the established modified electrometric method (Mohammad et al., 2005, 2006; Mohammad, 2007; Mohammad et al., 2007 b, 2007c). In view of the lack of information concerning the use of the current electrometric method for in vitro quinidine inhibition of WBChE in ruminants, the present study applied the pseudo ChE inhibition technique by quinidine sulfate to determine the percentage of true ChE activity comparatively across the species (Barenghi et al., 1986; Worek et al., 1999; Mohammad et al., 2007 b). Utilizing a consistent and simple modified electrometric methodology (Mohammad et al., 2007 b, 2007c), and incorporating the concept of conducting periodic ChE determinations to ensure data replicability (Wilson et al., 2005; Furman, 2010), this cross-species comparative analysis offers novel data regarding WBChE activities in humans and ruminants. This information would fill a gap in the current literature and contribute to our understanding of the enzyme by providing essential and updated normal baseline WBChE activity values, especially in the context of potential differential ChE inhibition by pesticides (WHO, 1986; Wilson 2014; Moon et al., 2015; Ahmad et al., 2024). Materials and MethodsBlood samplingWhole blood samples were collected from adult healthy Iraqi male (aged 25–45 years) and female (aged 25–45 years) volunteers (non-alcoholic, non-smokers) with no prior history of exposure to organophosphate or carbamate pesticides. In addition, blood samples were obtained from healthy, approximately 1-year-old, Awassi sheep and mixed-breed goats, as well as approximately 1.5–year-old local breed cattle. Access to the animals was granted through regular visits to the Dohuk Abattoir from 1 October 2024 to 25 February 2025, since only healthy animals fit for human consumption were slaughtered at this location. At the abattoir, blood was collected from both sexes of sheep and goats, and only from male cattle because of local regulations for economic reasons that prohibit slaughtering cows. Prior to the blood sampling, the animals had not been exposed to agricultural or veterinary pesticides for at least 3 months, as certified by the abattoir’s veterinary authority and the certified case history of the animal flock. Blood samples were obtained by venipuncture into heparinized test tubes under sterile conditions. All samples were transferred in cooling boxes to the laboratory and frozen at −20ºC before analysis within 3 weeks. Subjects deemed to be in poor health, both human and animal, were excluded from the study. The sample size of each species was determined by the online tool G*Power 3.1.9.7 (https://www.psychologie.hhu.de/arbeitsgruppen/allgemeine-psychologie-und-arbeitspsychologie/gpower/), taking into consideration population effect size ρ=0.4, α=0.05, power (1-β)=0.95 (Faul et al., 2009). Additional blood samples (8 to 13) from each species were obtained for the in vitro ChE inhibition experiment. Cholinesterase assay and enzymatic reaction mixtureThe modified electrometric method was used to measure WBChE activity in humans and ruminants (Mohammad et al., 2007 a, b, c; Odisho and Mohammad, 2022; Garmavy et al., 2023). As shown in Table 1, the enzymatic reaction mixture used to measure WBChE activity was prepared in a 10 ml glass beaker by adding distilled water, a whole blood sample, a barbital-phosphate buffer, and acetylcholine iodide as the substrate (Mohammad et al., 2007 a, b, c; Odisho and Mohammad, 2022; Garmavy et al., 2023). The pH of the reaction mixture was measured before and after the incubation using a glass electrode of a pH meter (pH700, Eutech Instruments, Singapore). The incubation time was 20 minutes for human samples and 30 minutes for ruminants, as optimized for the assay before (Mohammad et al., 2007 b, c; Odisho and Mohammad, 2022; Garmavy et al., 2023). The unit of ChE activity was Δ pH/incubation time. Table 1. The enzymatic reaction mixtures and steps for measuring cholinesterase (ChE) activity in human and animal whole blood.
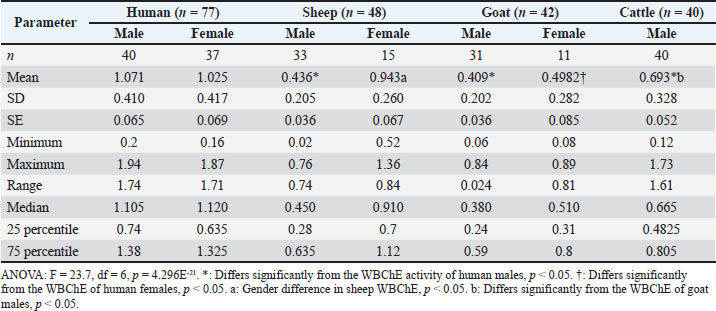
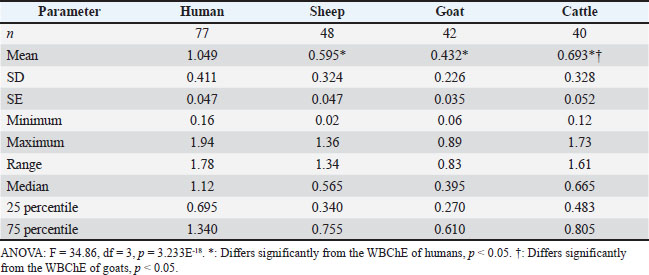
In vitro inhibition of WBChE activity by quinidine sulfateIn this experiment, whole blood samples were obtained from 13 humans, 10 sheep, 10 goats, and 8 cattle. The aim was to estimate true ChE activity in the whole blood by inhibiting its pseudo ChE component using quinidine sulfate. In order to achieve this objective, blood samples were subjected to pseudo ChE inhibition by adding 50 µl of 0.1% aqueous solution of quinidine sulfate (Sigma, St. Louis, USA) and then incubating for 10 minutes at 37ºC (Mohammad et al., 2006; 2007b, 2007c; Alias et al., 2011) prior to the addition of the substrate and the measurement of WBChE activity, as outlined in Table 1. Thereafter, the measured ChE activity represents the true ChE activity in the whole blood, and its percentage (%) was as follows: % of true ChE activity in the whole blood=[ChE activity (with quinidine) / ChE activity (without quinidine)]*100, since pseudo ChE activity=ChE (without quinidine) – ChE (with quinidine) (Mohammad et al., 2007 b). StatisticsThe statistical software Past version 5.1 (https://www.nhm.uio.no/english/research/resources/past/) was used for the statistical analysis of WBChE activity data from humans and ruminants (Hammer et al., 2001). The statistical differences among WBChE, pseudo, and true ChE activities across the species were analyzed using one-way analysis of variance followed by Tukey’s multiple comparison test. The level of statistical significance was set at p-value < 0.05. Ethical approvalThis cross-sectional comparative study was conducted on whole blood samples obtained from healthy humans and three ruminants (sheep, goats, and cattle). The management of human subjects participating in the present study was in accordance with the principles outlined in the Declaration of Helsinki, which presents ethics guidelines for medical research (https://www.wma.net/policies-post/wma-declaration-of-helsinki/). The use of ruminants in the present study was in accordance with the institutional regulations and ethics, in compliance with the guidelines of Animal Research: Reporting of in vivo experiments (https://arriveguidelines.org/) (Percie du Sert et al., 2020). The Directorate of Postgraduate Studies and Scholarships, at the University of Duhok in the Kurdistan Region of Iraq (Number 9327, September 10, 2024), and the Research Ethics Committee at Duhok Directorate General of Health, in the Kurdistan Region of Iraq (Reference No. 25092024-8-31, September 25, 2024), have kindly approved the present study protocols. Free written consent was obtained from all eligible Iraqi participants, following a detailed explanation of both the purpose of the study and the blood sampling for the WBChE assay. All information regarding participants was maintained under strict confidentiality. ResultsAs shown in Table 2, the electrometric measurement of WBChE activities in humans (Δ pH/20 minute) and ruminants (Δ pH/30 minute) is presented. The means ± SD of male and female WBChE activities of humans (1.071 ± 0.410 and 1.025 ± 0.417) were found to be significantly higher than those of similar sexes in sheep (0.436 ± 0.205 and 0.943 ± 0.260), goats (0.409 ± 0.202 and 0.4982 ± 0.282), or cattle (males only 0.693 ± 0.328). The gender effect was found to be minor and not consistent across the species. The overall result of the combined WBChE activities of males and females also showed significantly higher human WBCHE activity (1.049 ± 0.411) compared to those of sheep (0.595 ± 0.324), goats (0.432 ± 0.226), and cattle (0.693 ± 0.328) (Table 3). The table also presents median WBChE activities and related statistics, showing consistently higher enzyme activity in humans (1.120) compared to ruminants (0.565, 0.395, 0.665). Table 2. Comparison of whole blood cholinesterase (WBChE) activity among humans (Δ pH/20 minute) and ruminants (Δ pH/30 minute).
Table 3. The overall whole blood cholinesterase (WBChE) activity among humans (Δ pH/20 minute) and ruminants (Δ pH/30 minute).
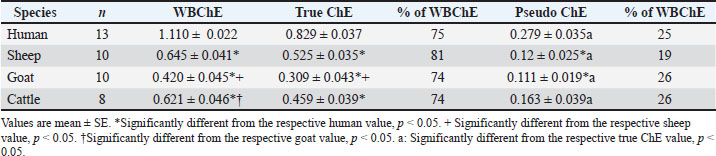
As illustrated in Table 4, the use of quinidine sulfate as a specific pseudo ChE inhibitor, enabled a comparative analysis of true and pseudo ChE activities in humans (0.829 ± 0.037 and 0.279 ± 0.035), sheep (0.525 ± 0.035 and 0.12 ± 0.025), goats (0.309 ± 0.043 and 0.111 ± 0.019), and cattle (0.459 ± 0.039 and 0.163 ± 0.039). The true ChE activity of humans was found to be significantly higher than that of ruminants, and the pseudo ChE of humans was also found to be significantly higher than that of sheep and goats (Table 4). Within each species, the pseudo ChE activity in the whole blood was consistently low (19%-26%) and significantly below the corresponding value of true ChE (74%–81%). Table 4. Comparison of true and pseudocholinesterase (ChE) activities among humans (Δ pH/20 minute) and ruminants (Δ pH/30 minute) in relation to whole blood ChE (WBChE).
DiscussionSince WBChE activity is the preferred ChE measurement for monitoring pesticide exposure under field conditions (Worek et al., 1999; Tecles et al., 2001; Haigh et al., 2008; Rajapakse et al., 2011; Worek et al., 2016), this study compares WBChE activities in humans and three ruminant species using a modified electrometric method. This comparative finding, being the first of its kind with this modified electrometric approach, further enhances and complements previous studies that employed the same methodology conducted separately on different blood components of humans (Mohammad et al., 2007 b; Garmavy et al., 2023) and ruminants (Mohammad et al., 2005, 2006, 2007c; Askar et al., 2011). The WBChE activities presented in the current study and in accordance with previous ones (Mohammad et al., 2005, 2006, 2007c) would be an added advantage in the evaluation of exposure of humans and ruminants to ChE inhibiting pesticides such as organophosphates and carbamates (Mohammad et al., 2007 a; Furman, 2010; Odisho and Mohammad, 2022; Mohammad et al., 2024). Cholinesterase measurement is used for biomonitoring pesticide exposure)Mohammad et al., 2007 a; Furman, 2010; Strelitz et al., 2014; Wilson, 2014; Mohammad et al., 2024), and it has been demonstrated that true ChE and pseudo ChE show differential susceptibility to pesticides (WHO, 1986; Wilson, 2010; Moon et al., 2015; Ahmad et al., 2024). Consequently, WBChE activity, which constitutes a practical and fast approach, is recommended in field conditions, where immediate separation of plasm and erythrocytes is not feasible, particularly when the specific pesticide is unknown. Another compelling reason to advocate the use of whole blood in the electrometric measurement of blood ChE is the inherently low levels of ChE found in the plasma fraction of the blood in ruminants, in contrast to humans. In separate studies, human PChE activity was found to be considerably higher (1.078 Δ pH/ 20 minute) (Mohammad et al., 2007 b) compared to sheep (0.133, 0.121 Δ pH/ 30 minute), goats (0.135, 0.175), and cattle (0.135, 0.137) (Mohammad et al., 2007 c). Furthermore, while the specific effects of quinidine on ChE activity in farm animals have not been extensively detailed in the available literature, in the present study, quinidine sulfate was applied in vitro to specifically inhibit pseudo ChE in humans and the three ruminant species. The findings of the present study revealed a lower pseudo ChE activity (19%–26%) within the whole blood component when compared to true ChE (74%–81%) across the species. However, in humans, the percentage of true ChE in the plasma was estimated to be 8.2% (Mohammad et al., 2007 b). The present findings in ruminants support the hypothesis of a low level of true ChE existing as a mixture with the pseudo ChE in the plasma (Mohammad et al., 2007 b, 2007c). Therefore, the presence of a small percentage of active true ChE in ruminant plasma, even under quinidine inhibition, cannot be ruled out (Al-Jobory and Mohammad, 2005; Mohammad et al., 2007 b). Given the inherently low PChE activity in ruminants, as reported herein, future studies will likely focus on comparative quantification by the quinidine inhibition technique of the percentages of true ChE inhibition in ruminant plasma following exposure to pesticides. This is because organophosphates and carbamates differentially inhibit blood ChE activities (WHO, 1986; Arrieta et al., 2009; Furman, 2010; Strelitz et al., 2014; Wilson, 2014; Moon et al., 2015). Low ChE activity in the plasma of ruminants highlights the importance of using whole blood, among other ChE measurements, for accurate assessments of pesticide exposure in ruminants. However, as demonstrated in this study and supported by previous research (Mohammad et al., 2007 b, 2007c), baseline WBChE activity significantly differs between humans and farm animals. This variation has important implications for the pharmacological effects of drugs and the toxicity of pesticides. The levels of blood ChE in workers and animals exposed to pesticides are influenced by several factors. These factors include differential enzyme inhibition by various pesticides, which differ in their intrinsic toxicity potential, as well as age, gender, body weight, diet, inter-individual and species variations that can be expected during pesticide exposure (Wills, 1972; Mohammad and St. Omer, 1982; WHO, 1986; Fairbrother et al., 1991; Arrieta et al., 2009; Furman, 2010; Strelitz et al., 2014; Wilson, 2014; Moon et al., 2015). The present findings, which include additional cross-species normal values for WBChE activities, lend support to the call for the establishment of national guidelines to measure blood ChE levels in monitoring pesticide exposure among farmers and their animals (Mohammad et al., 2007 c; Odisho and Mohammad, 2022; Mohammad et al., 2024). ConclusionThis study presents a comparative analysis of normal WBChE activities, measured using a modified electrometric method, between humans and ruminants. The analysis reveals that human blood exhibits higher levels of both pseudo and true cholinesterase activities compared to ruminants. This information would fill a gap in the current literature and contribute to our understanding of the enzyme by providing essential and updated normal baseline WBChE activity values across the species, especially in the context of potential differential ChE inhibition by pesticides. Comparative determination of whole blood cholinesterase activity using a modified electrometric method in humans and farm animals could serve as a valuable tool for assessing collectively cross-species exposure to pesticides. AcknowledgmentsThis report represents a part of a thesis to be submitted by the first author to the University of Duhok, Iraq, in partial fulfillment of the requirements for the MSc degree in Toxicology. The authors are thankful to the College of Pharmacy, University of Duhok, for supporting and providing facilities and supplies to conduct this study. Conflict of interestThe authors declare that there is no conflict of interest. FundingNone. Authors' contributionsMAR: Dealt with the participants, obtained blood samples and executed laboratory assays; conducted literature search, statistical analyses, and shared in drafting the manuscript. FKM: Conceptualized and supervised the study, shared in the literature search, statistical analyses, and drafted the manuscript. Both authors have read the manuscript and approve it for publication. Data availabilityAll data were provided in the manuscript. ReferencesAbdelsalam, E.B. and Ford, E.J. 1985. Normal esterase activity in the plasma, whole blood and tissues of cattle. Zentralbl. Veterinarmed. A 32, 518–525. Ahmad, M.F., Ahmad, F.A., Alsayegh, A.A., Zeyaullah, M., Alshahrani, A.M., Muzammil, K., Saati, A.A., Wahab, S., Elbendary, E.Y., Kambal, N., Abdelrahman, M.H. and Hussain, S. 2024. Pesticides impacts on human health and the environment with their mechanisms of action and possible countermeasures. Heliyon 10, e29128. Al-Abdaly, Y.Z., Alfathi, M.Y. and Al-Mahmood, S.S. 2023. Comparison of azithromycin toxicity in chickens and quails. Iran. J. Vet. Med. 17, 321–332. Alias, A., Al-Zubaidy, M., Mousa, Y. and Mohammad, F. 2011. Plasma and whole brain cholinesterase activities in three wild bird species in Mosul, Iraq: in vitro inhibition by insecticides. Interdiscip. Toxicol. 4, 144–148. Al-Jobory, M.M. and Mohammad, F.K. 2005. Validation of an electrometric blood cholinesterase measurement in goats. J. Vet. Sci. 6, 299–303. Al-Qarawi, A.A. and Ali, B.H. 2003. Variations in the normal activity of esterases in plasma and liver of camels (Camelus dromedarius), cattle (Bos indicus), sheep (Ovis aries) and goats (Capra hircus). J. Vet. Med. A 50, 201–203. Andersson, M.L., Møller, A.M. and Wildgaard, K. 2019. Butyrylcholinesterase deficiency and its clinical importance in anaesthesia: a systematic review. Anaesthesia 74(4), 518–528. Arrieta, D.E., Mccurdy, S.A., Henderson, J.D., Lefkowitz, L.J., Reitstetter, R. and Wilson, B.W. 2009. Normal range of human red blood cell acetylcholinesterase activity. Drug Chem. Toxicol. 32, 182–185. Askar, K.A., Kudi, A.C. and Moody, A.J. 2011. Comparative analysis of cholinesterase activities in food animals using modified Ellman and Michel assays. Can. J. Vet. Res. 75, 261–270. Barenghi, L., Ceriotti, F., Luzzana, M., Ripamonti, M., Mosca, A. and Bonini, P.A. 1986. Measurement of erythrocyte acetylcholinesterase and plasma cholinesterase activity by a differential pH technique. Ann. Clin. Biochem. 23, 538–545. Cerón, J.J., Tecles, F. and Espín, J.C. 1999. Comparison of different diluents and chromophores for spectrophotometric determination of livestock blood cholinesterase activity. Res. Vet. Sci. 67, 261–266. Fairbrother, A., Marden, B.T., Bennett, J.K. and Hooper, M.J. 1991. Methods used in determination of cholinesterase activity. In Cholinesterase-inhibiting insecticides - their impact on wildlife and the environment - chemicals in agriculture. Mineau, P Amsterdam, The Netherlands: Elsevier, 2, pp: 35–71. Faul, F., Erdfelder, E., Buchner, A. and Lang, A.G. 2009. Statistical power analyses using G*Power 3.1: tests for correlation and regression analyses. Behav. Res. Methods 41, 1149–1160. Furman, J. 2010. Cholinesterase monitoring for agricultural pesticide handlers: guidelines for health care providers in Washington State. Washington, USA: Department of Labor and Industries, Division of Occupational Safety and Health. Garmavy, H.M.S., Mohammed, A.A., Rashid, H.M. and Mohammad, F.K. 2023. A meta-analysis of normal human blood cholinesterase activities determined by a modified electrometric method. J. Med. Life 16, 22–34. Haigh, J.R., Lefkowitz, L.J., Capacio, B.R., Doctor, B.P. and Gordon, R.K. 2008. Advantages of the WRAIR whole blood cholinesterase assay: comparative analysis to the micro-Ellman, Test-mate ChE, and Michel (DeltapH) assays. Chem. Biol. Interact. 175, 417–420. Hammer., Harper, D.A.T. and Ryan, P.D. 2001. PAST: paleontological statistics software package for education and data analysis. Palaeontol. Electron. 4, 1. Korabecny, J. and Soukup, O. 2021. Cholinesterase research. Biomolecules 11(8), 1121. Kyi, N.E.M.M., Samar, M., Hongsibsong, S., Sawarng, N., Pata, S. and Wongta, A. 2025. Micro-electrometric method for assessing cholinesterase activity in human whole blood. Methods Protocols 8(2), 30. Lazarevic-Pasti, T., Leskovac, A., Momic, T., Petrovic, S. and Vasic, V. 2017. Modulators of acetylcholinesterase activity: from Alzheimer's disease to anti-cancer drugs. Curr. Med. Chem. 24(30), 3283–3309. Lionetto, M.G., Caricato, R., Calisi, A., Giordano, M.E. and Schettino, T. 2013. Acetylcholinesterase as a biomarker in environmental and occupational medicine: new insights and future perspectives. Biomed. Res. Int. 2013, 321213. Mohammad, F., Al-Badrany, Y. and Al-Jobory, M. 2008. Acute toxicity and cholinesterase inhibition in chicks dosed orally with organophosphate insecticides. Arch. Ind. Hyg. Toxicol. 59, 145–151. Mohammad, F.K. 2007. Review of a practical electrometric method for determination of blood and tissue cholinesterase activities in animals. VetScan 2, 1–12. Mohammad, F.K. and St. Omer, V.E. 1982. Modifications of Michel's electrometric method for rapid measurement of blood cholinesterase activity in animals: a minireview. Vet. Hum. Toxicol. 24, 119–121. Mohammad, F.K., Al-Baggou', B.K., Alias, A.S. and Faris, G.A.M. 2006. Application of an electrometric method for measurement of in vitro inhibition of blood cholinesterases from sheep, goats and cattle by dichlorvos and carbaryl. Vet. Med. 51, 45–50. Mohammad, F.K., Alias, A.S. and Ahmed, O.A.H. 2007b. Electrometric measurement of plasma, erythrocyte, and whole blood cholinesterase activities in healthy human volunteers. J. Med. Toxicol. 3(1), 25–30. Mohammad, F.K., Alias, A.S., Faris, D.A. and BKh, A.B. 2007a. Application of an electrometric method for measurement of blood cholinesterase activities in sheep, goats and cattle treated with organophosphate insecticides. J. Vet. Med. A. 54, 140–143. Mohammad, F.K., Al-Zubaidy, M.H.I. and Alias, A.S. 2007. Electrometric determination of erythrocyte, plasma and whole blood cholinesterase activities in sheep, goats and cattle and their in vitro inhibition by anticholinesterase insecticides. J. Pharmacol. Toxicol. 2, 131–141. Mohammad, F.K., Faris, G.A., -M.., Alias, A.S. and BKh, A.B. 2005. Blood cholinesterase activities in cattle, sheep and goats measured by a modified electrometric method. J. Anim. Vet. Adv. 4, 923–926. Mohammad, F.K., Mohammed, A.A. and Odisho, S.K. 2024. Changes of blood cholinesterase activities among pesticides-exposed agricultural workers in Iraq: a meta-analysis. Toxicol. Rep. 13, 101830. Mohammad, F.K., Mousa, Y.J., Al-Zubaidy, M.H.I. and Alias, A.S. 2012. Assessment of diphenhydramine effects against acute poisoning induced by the organophosphate insecticide dichlorvos in chicks. Hum. Vet. Med. 4(6), 6–13. Mohammad, F.K., Raouf, G.A.M. and Merza, M.A. 2025. Meta-analysis of decreased serum cholinesterase activity in COVID-19. Health Biotechnol. Biopharma. 9(1), 99–121. Mohammed, A.A. and Mohammad, F.K. 2022. Monitoring blood cholinesterase activity of farmworkers: in vitro inhibition by diphenhydramine and carbaryl. Malays. Appl. Biol. 51, 23–32. Moon, J., Chun, B. and Lee, S. 2015. Variable response of cholinesterase activities following human exposure to different types of organophosphates. Hum. Exp. Toxicol. 34, 698–706. Omar, H. and Fouad, K.M. 2007. Electrometric determination of blood cholinesterase activities in workers exposed to insecticides in Mosul, Iraq. Res. J. Environ. Toxicol. 1, 144–148. Oda, E. 2016. Serum cholinesterase (butyrylcholinesterase) may play a role in body weight homeostasis via the inactivation of ghrelin. Intern. Med. 55(9), 1245. Odisho, S.K. and Mohammad, F.K. 2022. Blood cholinesterase activities and oxidative stress status among farmworkers using pesticides in Duhok, KRG, Iraq. J. Ideas Health 5, 786–793. Percie Du Sert, N., Hurst, V., Ahluwalia, A., Alam, S., Avey, M.T., Baker, M., Browne, W.J., Clark, A., Cuthill, I.C., Dirnagl, U., Emerson, M., Garner, P., Holgate, S.T., Howells, D.W., Karp, N.A., Lazic, S.E., Lidster, K., Maccallum, C.J., Macleod, M., Pearl, E.J., Petersen, O.H., Rawle, F., Reynolds, P., Rooney, K., Sena, E.S., Silberberg, S.D., Steckler, T. and Würbel, H. 2020. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. PLos Biol. 18, e3000410. Rajapakse, B.N., Thiermann, H., Eyer, P., Worek, F., Bowe, S.J., Dawson, A.H. and Buckley, N.A. 2011. Evaluation of the Test-mate ChE (cholinesterase) field kit in acute organophosphorus poisoning. Ann. Emerg. Med. 58, 559–564. Raouf, G.A.M., Mohammad, F.K. and Merza, M.A. 2024. The potential risk of reduced serum cholinesterase activity in COVID-19 patients suffering from cytokine storm. Cureus 16, e60417. Saldanha, C. 2017. Human erythrocyte acetylcholinesterase in health and disease. Molecules 22(9), 1499. Sridhar, G.R. and Gumpeny, L. 2024. Emerging significance of butyrylcholinesterase. World J. Exp. Med. 14, 87202. Strelitz, J., Engel, L.S. and Keifer, M.C. 2014. Blood acetylcholinesterase and butyrylcholinesterase as biomarkers of cholinesterase depression among pesticide handlers. Occup. Environ. Med. 71, 842–847. Taylor, P., Camp, S. and Radić, Z. 2009. Acetylcholinesterase.In Encyclopedia of Neuroscience. Squire, L.R Oxford, UK: Academic Press, pp: 5–7. Tayseer, O.M. and Jassim, K. 2022. Serological and Biochemical Study for Neospora Caninum in Goats. Int. J. Health Sci. 6, 3406–3412. Tecles, F. and Cerón, J.J. 2001. Determination of whole blood cholinesterase in different animal species using specific substrates. Res. Vet. Sci. 70, 233–238. WHO. 1986. Environmental Health Criteria 63: Organophosphorus Insecticides: A General Introduction. WHO, Geneva, Switzerland. Wills, J.H. K.P. 1972. The measurement and significance of changes in the cholinesterase activities of erythrocytes and plasma in man and animals. CRC. Crit. Rev. Toxicol. 1, 153–202. Wilson, B.W. 2010. Cholinesterases. In: Hayes' handbook of pesticide toxicology. 3rd ed. Ed., Krieger, R., Amsterdam, The Netherlands: Academic Press, pp.1457-1147. Wilson, B.W. 2014. Cholinesterase inhibition. In: Encyclopedia of toxicology. 3rd ed. Ed., Wexler, P., Amsterdam, The Netherlands: Elsevier, pp: 942-951. Wilson, B.W., Arrieta, D.E. and Henderson, J.D. 2005. Monitoring cholinesterases to detect pesticide exposure. Chem. Biol. Interact. 157-158(158), 253–256. Wongta, A., Anand, P., Aning, N.A.A., Sawarng, N. and Hongsibsong, S. 2024. Advancing micro-electrometric techniques for the detection of organophosphate and carbamate residues using cricket cholinesterase. PLos One 19, 308112. Worek, F., Mast, U., Kiderlen, D., Diepold, C. and Eyer, P. 1999. Improved determination of acetylcholinesterase activity in human whole blood. Clin. Chim. Acta. 288, 73–90. Worek, F., Schilha, M., Neumaier, K., Aurbek, N., Wille, T., Thiermann, H. and Kehe, K. 2016. On-site analysis of acetylcholinesterase and butyrylcholinesterase activity with the ChE check mobile test kit-Determination of reference values and their relevance for diagnosis of exposure to organophosphorus compounds. Toxicol. Lett. 249, 22–28. | ||
| How to Cite this Article |
| Pubmed Style Ramadhan MA, Mohammad FK. Whole blood cholinesterase activity in humans and ruminants: Comparison using a modified electrometric method. Open Vet. J.. 2025; 15(10): 5032-5040. doi:10.5455/OVJ.2025.v15.i10.21 Web Style Ramadhan MA, Mohammad FK. Whole blood cholinesterase activity in humans and ruminants: Comparison using a modified electrometric method. https://www.openveterinaryjournal.com/?mno=258859 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.21 AMA (American Medical Association) Style Ramadhan MA, Mohammad FK. Whole blood cholinesterase activity in humans and ruminants: Comparison using a modified electrometric method. Open Vet. J.. 2025; 15(10): 5032-5040. doi:10.5455/OVJ.2025.v15.i10.21 Vancouver/ICMJE Style Ramadhan MA, Mohammad FK. Whole blood cholinesterase activity in humans and ruminants: Comparison using a modified electrometric method. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5032-5040. doi:10.5455/OVJ.2025.v15.i10.21 Harvard Style Ramadhan, M. A. & Mohammad, . F. K. (2025) Whole blood cholinesterase activity in humans and ruminants: Comparison using a modified electrometric method. Open Vet. J., 15 (10), 5032-5040. doi:10.5455/OVJ.2025.v15.i10.21 Turabian Style Ramadhan, Maha Ahmed, and Fouad Kasim Mohammad. 2025. Whole blood cholinesterase activity in humans and ruminants: Comparison using a modified electrometric method. Open Veterinary Journal, 15 (10), 5032-5040. doi:10.5455/OVJ.2025.v15.i10.21 Chicago Style Ramadhan, Maha Ahmed, and Fouad Kasim Mohammad. "Whole blood cholinesterase activity in humans and ruminants: Comparison using a modified electrometric method." Open Veterinary Journal 15 (2025), 5032-5040. doi:10.5455/OVJ.2025.v15.i10.21 MLA (The Modern Language Association) Style Ramadhan, Maha Ahmed, and Fouad Kasim Mohammad. "Whole blood cholinesterase activity in humans and ruminants: Comparison using a modified electrometric method." Open Veterinary Journal 15.10 (2025), 5032-5040. Print. doi:10.5455/OVJ.2025.v15.i10.21 APA (American Psychological Association) Style Ramadhan, M. A. & Mohammad, . F. K. (2025) Whole blood cholinesterase activity in humans and ruminants: Comparison using a modified electrometric method. Open Veterinary Journal, 15 (10), 5032-5040. doi:10.5455/OVJ.2025.v15.i10.21 |