| Research Article | ||
Open Vet. J.. 2026; 16(1): 437-444 Open Veterinary Journal, (2026), Vol. 16(1): Research Article Growth of Octopus vulgaris cuvier, 1797 along the Mediterranean Sea’s western Libyan coastMagfera A. Wail1, Mohamed L. Showehdi2, Hamida A. Jbr3, Widien Khoufi4, Chedia Jabeur5 and Taher Shaibi3*1Biology Department, Faculty of Education, University of Tripoli, Tripoli, Libya 2Department of Poultry and Fish Diseases, Faculty of Veterinary Medicine,University of Tripoli, Tripoli, Libya 3Zoology Department, Faculty of Science, University of Tripoli, Tripoli, Libya 4Laboratory of Analysis, Treatment and Valorization of the Pollutants of the Environment and Products, Faculty of Pharmacy, University of Monastir, Monastir, Tunisia 5High Institute of Biotechnology of Monastir, Monastir, Tunisia *Corresponding Author: Taher Shaibi. Zoology Department, Faculty of Science, University of Tripoli, Tripoli, Libya. Email: t.shaibi [at] uot.edu.ly Submitted: 18/05/2025 Revised: 20/11/2025 Accepted: 11/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: The common octopus, Octopus vulgaris, is an ecologically and economically vital cephalopod species in the Mediterranean; however, its biological characteristics remain unstudied in Libyan waters. Aim: This study provides the first comprehensive analysis of growth patterns for this species along the western Libyan coast. Methods: A total of 390 specimens (172 males, 218 females) were collected monthly from January 2021 to January 2022. Results: Dorsal mantle length (DML) ranged from 5 to 25 cm, and body weight (BW) ranged from 63 to 3376 g, with no significant difference between the sexes. A strong seasonal pattern was observed, with the minimum average size recorded in August (~11 cm, ~340 g) and the maximum size recorded in January (~18 cm, ~1450 g). The length–weight relationship revealed negative allometric growth (b=2.85) for both sexes, with a strong correlation (R² > 0.94) and no significant sexual dimorphism. The von Bertalanffy growth function estimated asymptotic lengths (L∞) of 30 and 28 cm for males and 28 cm for females, consistent with other Mediterranean populations but lower than those of the Atlantic ones, suggesting biogeographical constraints. The condition index showed significant seasonal variation, with lows in March and peaks in June, likely linked to reproductive cycles. Conclusion: These results characterize a population with typical Mediterranean growth dynamics. A pronounced seasonal recruitment pulse in late summer highlights the vulnerability of juveniles to fishing pressure. We recommend the implementation of fisheries management strategies, such as seasonal closures during this critical period, to ensure the sustainability of this valuable Libyan resource. Keywords: Octopus vulgaris, Length–weight relationship, Fisheries management, Libya, Mediterranean Sea. IntroductionOctopus vulgaris Cuvier, 1797, commonly known as the common octopus or Mediterranean octopus, is a widely distributed cephalopod species throughout the Mediterranean Sea (Bilecenoglu et al., 2013). Octopus vulgaris inhabits benthic habitats from the shoreline to ~200 m depth, most commonly between 20 and 100 m, in waters with temperatures typically ranging from 15°C to 25°C, although it can tolerate lows near 7°C (Belcari et al., 2002). Growth and metabolic performance are considered optimal within this range (Sillero-Ríos et al., 2018). Understanding these growth dynamics is essential for the effective conservation and sustainable management of O. vulgaris populations within the Mediterranean marine ecosystem (Repolho et al., 2014; Sillero-Ríos et al., 2018). The range of O. vulgaris includes the Mediterranean Sea and the Eastern North Atlantic, with additional records from the subtropical and temperate East and West Atlantic, the Indian Ocean, and the West Pacific (Al-Maliky et al., 2021). This species can be found throughout the Mediterranean, and it is caught in several ways, including pottery pots, trawl nets, and direct diving (Aguado Giménez and García García, 2002). Owing to its ecological and economic significance, the biology and growth of O. vulgaris have been extensively studied in Mediterranean and adjacent Atlantic regions. Growth assessments often focus on length–weight relationships (Quetglas et al., 1998; Riad and Gabr, 2007; Gonzalez et al., 2011) and length–age relationships (Guerra, 1979a; Zghidi-Barraj, 2002; Jabeur et al., 2012). Studies have been conducted in diverse locations, including the Balearic Sea, Egyptian coasts, Gulf of Alicante, eastern Tunisia, Andalusia, Gulf of Cádiz, and the northeastern Atlantic (Otero et al., 2007; Jabeur et al., 2012; Kivengea, 2014; Ajana et al., 2018). Moreover, physiological condition indices have been highlighted as important indicators of growth and health in octopus populations (Gonzalez et al., 2011; Kivengea, 2014). In Libya, O. vulgaris represents a valuable component of artisanal and commercial fisheries, providing coastal communities with a source of income and an export commodity of growing demand. Recent increases in fishing pressure and limited management measures highlight the need for biological data to support sustainable exploitation. Despite this socioeconomic importance, scientific information on the species in Libyan waters remains scarce. Previous studies have only briefly noted its presence among commercial benthic species (Rawag et al., 2004) or addressed taxonomic aspects along the western Libyan coast (Alaswad and Eisay, 2020). To date, no detailed studies have examined the biological characteristics or growth patterns of O. vulgaris in Libya. Such information is vital for establishing appropriate fishing regulations, defining biological rest periods, supporting conservation efforts, and enriching regional marine biodiversity databases. The findings of this study will fill a critical gap in central Mediterranean cephalopod biology. Therefore, this study investigates the growth patterns of O. vulgaris along the western Mediterranean coast of Libya, providing the first detailed biological data from this region and a foundation for local fisheries management and conservation strategies. Materials and MethodsSpecimen collectionA total of 390 O. vulgaris specimens (172 males and 218 females) were collected at a rate of 30 individuals per month from January 2021 to January 2022. Samples were directly obtained from local fishermen and fish markets in Tripoli, Libya. According to the fishermen, the octopuses were captured by free and scuba diving from Zuwara, Az-Zāwiyah, and Tripoli. Immediately after collection, the specimens were placed in plastic bags containing crushed ice and transported for further analysis to the Laboratory of the Biology Department, Faculty of Education, University of Tripoli. Morphological measurementsMorphological measurements were taken following the methodologies described in previous studies (Fig. 1) (Kivengea, 2014; Drerup and Cooke, 2019). The total body weight (BW) was measured to the nearest 0.1 g. Dorsal mantle length (DML) to the nearest 0.1 cm: measured from the anterior end of the dorsal mantle to the midpoint between the eyes.
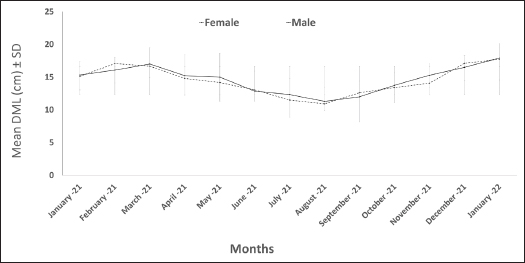
Fig. 1. Octopus measurements (Kivengea, 2014; Drerup and Cooke, 2019). DML: dorsal mantle length; ED: eye diameter; TL: total length. Data analysisRelationship between length and weightGrowth was calculated manually using the length–weight relationship, based on the equation between DML and total BW: BW=a × DMLᵇ, where BW is the total BW, DML is the DML, and a and b are regression constants (Ricker, 1973). Nonlinear regression analysis was performed to estimate the parameters of this relationship. Additionally, analysis of covariance (ANCOVA) was used to test for significant differences in the length–weight relationship between males and females. Age determinationAge was determined according to the method of Bhattacharya (1967) using the FISAT program (Gayanilo et al., 1996). This method analyzes length–frequency data by decomposing the overall length distribution into normal components, allowing the estimation of age groups based on length cohorts over the study period. To determine the growth parameters (t0, L∞ and k) of the von Bertalanffy equation, the relationship between length and age was extracted (Von Bertalanffy, 1938): L=L∞(1 – exp(−K(t – t0))), where L=length at a certain age, L∞ = the asymptotic average length of the dorsal mantle, K=growth rate, and t0 = the default lifetime at which length=0. Condition indexThe condition index (CI) was calculated using the equation CI=(BW/DML³) × 100 (Ricker, 1975), where CI is the condition index, BW is the total BW, and DML is the dorsal mantle length. Data were analyzed using IBM SPSS Statistics v23. Statistical significance was set at p=0.05. The independent samples t-test was used to determine the differences between the average ages of males and females. One-way analysis of variance was used to test for differences in monthly condition indices between sexes, followed by post hoc pairwise comparisons using the least significant difference test. Ethical approvalNot needed for this study. ResultsThe dorsal mantle length of the common octopus ranged from 5 to 25 cm in males and from 6 to 24 cm in females. The BWs ranged from 63 to 3,376 g in males and from 78 to 3,294 g in females. Figure 2 shows a clear seasonal pattern in the average size of O. vulgaris. The smallest average dorsal mantle lengths were recorded in August 2021, measuring 11.29 ± 1.40 cm for males and 10.92 ± 2.40 cm for females. In contrast, the largest averages were observed in January 2022, reaching 17.93 ± 3.30 cm for males and 17.73 ± 2.10 cm for females.
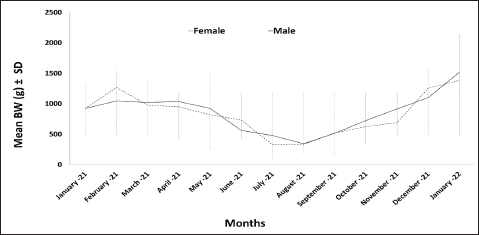
Fig. 2. Monthly mean dorsal mantle length of O. vulgaris off the western coast of Libya. Statistical analysis revealed no significant difference in size between the sexes (p=0.804). However, a highly significant difference was found among the monthly average sizes for each sex (p < 0.001), indicating that the captured population size varied considerably throughout the study period. The monthly distribution of average total BW in the common octopus followed a similar pattern to body length, with the lowest values recorded in August 2021 for both males (343.53 ± 143.80 g) and females (333.00 ± 232.30 g). The highest values were observed in January 2022 for males (1517.27 ± 776.7 g) and females (1384.20 ± 611.20 g) (Fig. 3). Statistical analysis revealed no significant overall difference in weight between the sexes (p=0.921). However, a significant difference was found among the monthly average weights for each sex (p < 0.001), indicating a substantial monthly variation in the weight of the population. The only exception was in November, when a significant difference was observed between males and females (p=0.038).
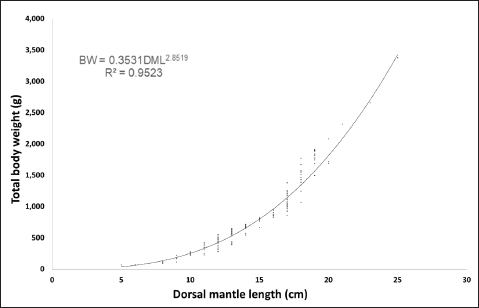
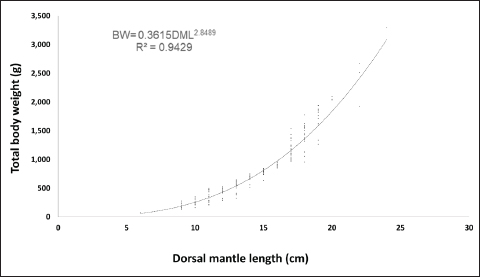
Fig. 3. Monthly mean total BW of O. vulgaris off the western coast of Libya. The allometric coefficient (b) was 2.85 for males and 2.85 for females, indicating negative allometric growth in both sexes. The coefficient of determination (R²) showed a strong positive correlation between length and weight, with 0.95 and 0.94 for males and 0.94 for females (Figs. 4 and 5). The ANCOVA test indicated homogeneous slopes in the length–weight relationship between sexes (p=0.90), suggesting that sex had no significant effect on this relationship.
Fig. 4. Relationship between the length and weight of male O. vulgaris off the western coast of Libya.
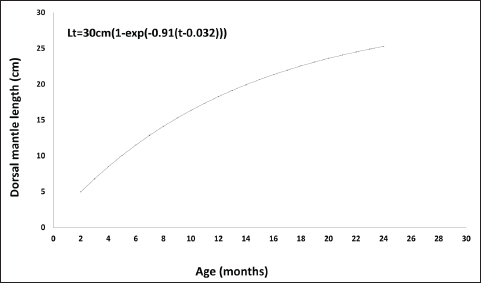
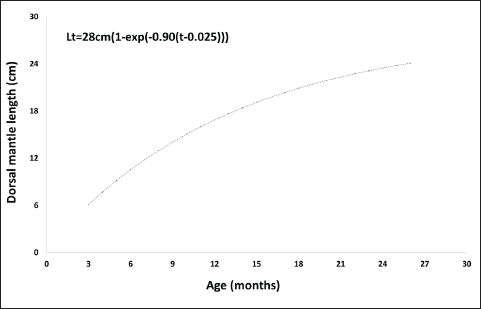
Fig. 5. Relationship between the length and weight of O. vulgaris females off the western coast of Libya. The age of O. vulgaris individuals ranged from 2 to 26 months. Male ages ranged from 2 to 24 months, whereas female ages ranged from 3 to 26 months. A positive relationship between dorsal mantle length and age was observed using the Von Bertalanffy growth model, with parameter values shown in Figures 6 and 7. No significant differences in age were observed between the sexes (p=0.46).
Fig. 6. Growth curve of O. vulgaris males by applying the Von Bertalanffy equation.
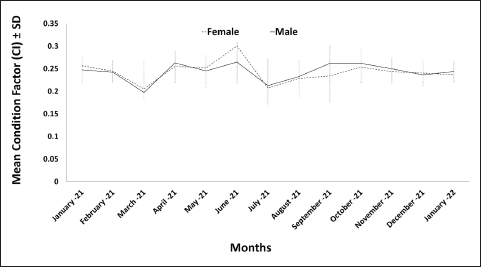
Fig. 7. Growth curve of O. vulgaris females by applying the Von Bertalanffy equation. The monthly average CI for O. vulgaris showed the highest values for both males and females in June, with females exhibiting higher index values than males. The lowest CI values were recorded for both sexes in March (Fig. 8). Significant differences were found between males and females (p=0.035), as well as between the monthly averages for both sexes (p=0.000).
Fig. 8. Average CI of male and female O. vulgaris off the western coast of Libya. DiscussionThis study provides the first comprehensive biological data for O. vulgaris from the western coast of Libya, filling a critical knowledge gap for the region. Our findings on size distribution, growth patterns, and seasonal dynamics align with established Mediterranean benchmarks and offer specific insights for local fisheries management. The recorded size range (DML: 5–25 cm) and weight (63–3,376 g) of individuals indicate that the fishery is accessing a broad population spectrum. The smallest individuals (5–6 cm) are consistent with findings from the Balearic Sea, the Gulf of Cádiz, and Italian waters (Quetglas et al., 1998; Belcari et al., 2002; Silva et al., 2002). However, smaller sizes reported in Egypt (4.5 cm) and Catalonia (3 cm) suggest potential regional variations in recruitment or fishing pressure (Guerra, 1979b; Riad and Gabr, 2007). The maximum size (25 cm) recorded here is slightly smaller than the 26.5–27 cm reported in Tunisia and Spain (Belcari et al., 2002; Jabeur et al., 2012) but larger than the maxima found in Egyptian and Balearic populations (Quetglas et al., 1998; Riad and Gabr, 2007). This global variation in size extremes is likely attributable to a complex interplay of factors, including differential fishing pressure (e.g., overfishing by trawlers selectively removing larger individuals), fishing gear selectivity (e.g., diving vs. pots vs. trawling), and environmental gradients in nutrient availability, temperature, and salinity (Tsangridis et al., 2002; Vargas-Yáñez et al., 2009). It is important to acknowledge a potential limitation inherent in the sampling methodology used in this study. As the specimens were sourced exclusively from fish markets in Tripoli, the fisher behavior and gear selectivity may influence the population representativeness. The observed size range (5–25 cm DML) necessarily reflects the sizes targeted by and available to the local fishery, which may use specific gear types that are selected for certain size classes. The smallest juveniles or the largest, potentially reclusive adults may be undersampled or absent from the market supply, potentially biasing growth parameter estimates and affecting the accurate determination of the true population size structure. Future studies should supplement market sampling with direct, scientific surveys employing a variety of methods (e.g., scuba diving, trawling, and benthic grabs) across different habitats to capture a more complete picture of the population, including early benthic stages and very large individuals that may evade standard fishing practices. Despite this limitation, the data presented here provide an invaluable baseline for the exploited component of the population, which is most relevant for the management of fisheries. A core finding of this study is the highly significant seasonal fluctuation in average size and weight, with minima recorded in August (~11 cm, ~340 g) and maxima in January (~18 cm, ~1450 g). This pattern mirrors the species’ typical annual life cycle across the Mediterranean (Pierce, 2010), where a peak in spawning leads to a massive recruitment pulse of juveniles (the small August cohort), followed by a period of rapid growth leading to larger winter sizes. The absence of a significant overall difference in size or weight between sexes is a common trait of O. vulgaris (Silva et al., 2002; Gonzalez et al., 2011) and suggests parallel growth and mortality rates. The significant difference observed in November is likely a sampling artifact rather than a biological trend. The length–weight relationship revealed negative allometric growth (b=2.85 for both sexes), confirming that weight increases at a slower rate than length. This pattern is widespread for O. vulgaris and is consistent with studies from the Balearic Sea, the eastern Tunisian coast, and the Gulf of Alicante (Quetglas et al., 1998; Gonzalez et al., 2011; Jabeur et al., 2012). The homogeneous slopes between sexes, as confirmed by ANCOVA, indicate identical energy allocation strategies for growth in males and females. This finding contrasts with populations of Morocco and Mauritania, where sexual dimorphism in growth (isometric in males, negative allometric in females) has been reported (Jurado-Ruzafa et al., 2014; Ajana et al., 2018). Growth modeling using the Von Bertalanffy function yielded asymptotic lengths (L∞) of 30 and 28 cm for male and female rates, respectively. These estimates closely align with values reported for other Mediterranean regions, such as the Gulf of Gabes (L∞=29.6 cm) and the eastern Tunisian coast (L∞=28.3 cm) (Zghidi-Barraj, 2002; Jabeur et al., 2012), but are markedly lower than those for Atlantic populations (L∞=40 cm) (Guerra, 1979b). This discrepancy underscores the profound influence of biogeographical factors. The Mediterranean Sea, a semi-enclosed basin with higher salinity, warmer temperatures, and lower nutrient availability, likely imposes different physiological constraints on growth compared with the more productive, cooler Atlantic Ocean (Ezzeddine and El Abed, 2004; Peña, 2024). The condition factor data further illuminate the reproductive cycle. The lowest values in March suggest a period of energy depletion associated with gametogenesis and pre-spawning, whereas the peak in June indicates a post-spawning recovery phase fueled by active feeding, a pattern also observed on the Moroccan Mediterranean coast and the Gulf of Alicante (Gonzalez et al., 2011; Ajana et al., 2018). The significant difference between sexes during this peak likely reflects females’ greater energetic investment in gamete production. In conclusion, the population of O. vulgaris along the western Libyan coast exhibits characteristically Mediterranean growth dynamics. This study’s key managerial insight is the clear seasonal pulse in recruitment. The wider continental shelf and lower pollution levels of the western Libyan coast (Said et al., 2010; El-Adl et al., 2022) may support this healthy population. Management strategies must account for the high vulnerability of the juvenile cohort in late summer to ensure its sustainability. Implementing a seasonal closure or gear restrictions during this period would protect this recruiting year-class, ultimately increasing the fishery’s long-term ecological resilience and economic yield. Future research should expand to the middle and eastern Libyan coasts to determine if these population characteristics are consistent across the country’s varying marine habitats. AcknowledgmentsWe would like to express our deep gratitude to all who contributed to the completion of this research. Conflict of interestThe authors declare no conflicts of interest. FundingThis research received no specific grant. Authors’ contributionsMAW and TS contributed to the design and implementation of the research and wrote the manuscript with the input of all authors. MAW and HAJ collected specimens, while MLS, WK, and CJ contributed to data analysis. Data availabilityAll data supporting this study’s findings are available within the manuscript. ReferencesAguado Giménez, F. and García García, B. 2002. Growth and food intake models in Octopus vulgaris Cuvier (1797): influence of body weight, temperature, sex and diet. Aquacult. Int. 10(5), 361–377. Ajana, R., Techetach, M. and Saoud, Y. 2018. Length-weight relationship and Fulton’s condition factor of the common octopus Octopus vulgaris in the Moroccan Mediterranean coast. J. Biol. Nat. 9(3), 95–100. Alaswad, H. and Eisay, A. 2020. Classification of Octopus in the Libyan west coast (Coast of the cities of Sorman and Sabratha). J. Sci. (A special issue of the fourth annual conference on theories and applications of basic and biological sciences), 6,158–166. Al-Maliky, A.M.J., Al-Khafaji, K.K. and Al-Maliky, T.H. 2021. First record of Octopus vulgaris (Cuvier, 1797) (Octopodidae) in the Iraqi coastal waters, NW Arabian Gulf. J. Appl. Natural Sci. 13(3), 1048–1051. Belcari, P., Cuccu, D., González, M., Srairi, A. and Vidoris, P. 2002. Distribution and abundance of Octopus vulgaris Cuvier, 1797 (Cephalopoda: octopoda) in the Mediterranean sea. Sci. Mar. 66(S2), 157–166. Bhattacharya, C.G. 1967. A Simple method of resolution of a distribution into gaussian components. Biometrics 23(1), 115–135. Bilecenoglu, M., Alfaya, J., Azzurr, E., Baldacconi, R., Boyaci, Y., Circosta, V., Compagno, L., Coppola, F., Deidun, A. and Durgham, H. 2013. New Mediterranean marine biodiversity records. Mediterr. Mar. Sci. 14(2), 463–480. Drerup, C. and Cooke, G. (2019). Cephalopod ID guide for the Mediterranean Sea. Genève, Zenodo. El-Adl, M.F., El-Katony, T.M. and Saleh, A.M. 2022. Pollution and substrate characteristics are correlated with intertidal macroalgal community structure on the eastern Libyan coast. Phycologia 61(5), 528–538. Ezzeddine, S. and El Abed, A. 2004. Potential biological and environmental influences on the Octopus vulgaris population of the Gulf of Gabès (south-eastern Tunisian coast). MedSudMed Tech. Doc. 2, 42–49. Gayanilo, F.C., Jr Sparre, P. and Pauly, D. 1996. FiSAT: FAO-ICLARM stock assessment tools. User’s manual. Computerized Information Series: Fisheries (FAO). Rome, FAO. Gonzalez, M., Barcala, E., Perez-Gil, J.L., Carrasco, M.N. and Garcia-Martinez, M.C. 2011. Fisheries and reproductive biology of Octopus vulgaris (Mollusca: cephalopoda) in the Gulf of Alicante (Northwestern Mediterranean). Mediterr. Mar. Sci. 12(2), 369–389. Guerra, A. 1979a. Estructura de la población de Octopus vulgaris del Atlántico Centro-Oriental (26º10’N-23º30’N) (Ad hoc Working Group of assessment of cephalopod stocks. S/C de Tenerife (Spain), Issue. Guerra, A. 1979b. Fitting a von Bertalanffy expression to Octopus vulgaris growth. Invest. Pesq. 43(1), 319–326. Jabeur, C., Nouira, T., Khoufi, W., Mosbahi, D.S. and Ezzeddine-Najai, S. 2012. Age and growth of Octopus vulgaris Cuvier, 1797, along the east coast of Tunisia. J. Shellfish. Res. 31(1), 119–124. Jurado-Ruzafa, A., Duque, V. and Carrasco, M.N. 2014. Reproductive aspects of Octopus vulgaris, Cuvier 1797 (Cephalopoda: octopodidae), caught in Mauritanian waters by the industrial Spanish fleet (NW Africa). Vieraea 42, 149–164. Kivengea, G.M. 2014. The biology and fishery of common octopus (Octopus vulgaris, Cuvier 1797) in the Kenyan South Coast. PhD, University of Nairobi, Nairobi, Kenya. Otero, J., González, A.F., Sieiro, M.P. and Guerra, A. 2007. Reproductive cycle and energy allocation of Octopus vulgaris in Galician waters, NE Atlantic. Fish. Res. 85(1-2), 122–129. Peña, M. 2024. Atlantic versus Mediterranean deep scattering layers around the Iberian Peninsula. Prog. Oceanogr. 221, 103211. Pierce, G.J. 2010. Fisheries biology of Octopus vulgaris in European waters (ICES) Coop. Res. Rep., Issue. Quetglas, A., Alemany, F., Carbonell, A., Merella, P. and Sánchez, P. 1998. Biology and fishery of Octopus vulgaris Cuvier, 1797, caught by trawlers in Mallorca (Balearic Sea, Western Mediterranean). Fisheries Res. 36(2-3), 237–249. Rawag, A.A., Haddoud, D.A. and Zgozi, S.W. 2004. Commercial demersal marine species of Libya (MedSudMed Tech. Doc.), Issue. Repolho, T., Baptista, M., Pimentel, M.S., Dionísio, G., Trübenbach, K., Lopes, V.M., Lopes, A.R., Calado, R., Diniz, M. and Rosa, R. 2014. Developmental and physiological challenges of octopus (Octopus vulgaris) early life stages under ocean warming. J. Comp. Physiol. B 184(1), 55–64. Riad, R. and Gabr, H.R. 2007. Comparative study on Octopus vulgaris (Cuvier, 1797) from the Mediterranean and Red Sea coasts of Egypt. Egypt. J. Aquat. Res. 33(3), 140–146. Ricker, W.E. 1973. Linear regressions in fishery research. Fish. Res. Board. Can. 30(3), 409–434. Ricker, W.E. 1975. Computation and interpretation of biological statistics of fish populations. Bull. Fish. Res. Board Can. 191, 1-382. Said, A., Godeh, M. and El-Menifi, F. 2010. Marine algal survey of Derna, Susa and Tolmeta at Libya coasts. Egypt. J. Phycology 11(1), 1–13. Sillero-Ríos, J., Sureda, A., Capó, X., Oliver-Codorniú, M. and Arechavala-Lopez, P. 2018. Biomarkers of physiological responses of Octopus vulgaris to different coastal environments in the western Mediterranean Sea. Mar. Pollut. Bull. 128, 240–247. Silva, L., Sobrino, I. and Ramos, F. 2002. Reproductive biology of the common octopus, Octopus vulgaris Cuvier, 1797 (Cephalopoda: octopodidae) in the Gulf of Cádiz (SW Spain). Bull. Mar. Sci. 71(2), 837–850. Tsangridis, A., Sánchez, P. and Ioannidou, D. 2002. Exploitation patterns of Octopus vulgaris in two Mediterranean areas. Sci. Mar. 66(1), 59–68. Vargas-Yáñez, M., Moya, F., García-Martínez, M., Rey, J., González, M. and Zunino, P. 2009. Relationships between Octopus vulgaris landings and environmental factors in the northern Alboran Sea (Southwestern Mediterranean). Fisheries. Res. 99(3), 159–167. Von Bertalanffy, L. 1938. A quantitative theory of organic growth (inquiries on growth laws II). Hum. Biol. 10(2), 181–213. Zghidi-Barraj, W. 2002. Ecobiologie et exploitation du poulpe commun Octopus vulgaris Cuvier, 1797 (Cephalopoda, Octopoda) dans le Golfe de Gabès (Tunis, Méditerranée centrale). PhD, University of Tunis, Tunis, Tunisia. | ||
| How to Cite this Article |
| Pubmed Style Wail MA, Showehdi ML, Jbr HA, Khoufi W, Jabeur C, Shaibi T. Growth of Octopus vulgaris cuvier, 1797 along the Mediterranean Sea’s western Libyan coast. Open Vet. J.. 2026; 16(1): 437-444. doi:10.5455/OVJ.2026.v16.i1.40 Web Style Wail MA, Showehdi ML, Jbr HA, Khoufi W, Jabeur C, Shaibi T. Growth of Octopus vulgaris cuvier, 1797 along the Mediterranean Sea’s western Libyan coast. https://www.openveterinaryjournal.com/?mno=259174 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.40 AMA (American Medical Association) Style Wail MA, Showehdi ML, Jbr HA, Khoufi W, Jabeur C, Shaibi T. Growth of Octopus vulgaris cuvier, 1797 along the Mediterranean Sea’s western Libyan coast. Open Vet. J.. 2026; 16(1): 437-444. doi:10.5455/OVJ.2026.v16.i1.40 Vancouver/ICMJE Style Wail MA, Showehdi ML, Jbr HA, Khoufi W, Jabeur C, Shaibi T. Growth of Octopus vulgaris cuvier, 1797 along the Mediterranean Sea’s western Libyan coast. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 437-444. doi:10.5455/OVJ.2026.v16.i1.40 Harvard Style Wail, M. A., Showehdi, . M. L., Jbr, . H. A., Khoufi, . W., Jabeur, . C. & Shaibi, . T. (2026) Growth of Octopus vulgaris cuvier, 1797 along the Mediterranean Sea’s western Libyan coast. Open Vet. J., 16 (1), 437-444. doi:10.5455/OVJ.2026.v16.i1.40 Turabian Style Wail, Magfera A., Mohamed L. Showehdi, Hamida A. Jbr, Widien Khoufi, Chedia Jabeur, and Taher Shaibi. 2026. Growth of Octopus vulgaris cuvier, 1797 along the Mediterranean Sea’s western Libyan coast. Open Veterinary Journal, 16 (1), 437-444. doi:10.5455/OVJ.2026.v16.i1.40 Chicago Style Wail, Magfera A., Mohamed L. Showehdi, Hamida A. Jbr, Widien Khoufi, Chedia Jabeur, and Taher Shaibi. "Growth of Octopus vulgaris cuvier, 1797 along the Mediterranean Sea’s western Libyan coast." Open Veterinary Journal 16 (2026), 437-444. doi:10.5455/OVJ.2026.v16.i1.40 MLA (The Modern Language Association) Style Wail, Magfera A., Mohamed L. Showehdi, Hamida A. Jbr, Widien Khoufi, Chedia Jabeur, and Taher Shaibi. "Growth of Octopus vulgaris cuvier, 1797 along the Mediterranean Sea’s western Libyan coast." Open Veterinary Journal 16.1 (2026), 437-444. Print. doi:10.5455/OVJ.2026.v16.i1.40 APA (American Psychological Association) Style Wail, M. A., Showehdi, . M. L., Jbr, . H. A., Khoufi, . W., Jabeur, . C. & Shaibi, . T. (2026) Growth of Octopus vulgaris cuvier, 1797 along the Mediterranean Sea’s western Libyan coast. Open Veterinary Journal, 16 (1), 437-444. doi:10.5455/OVJ.2026.v16.i1.40 |