| Case Report | ||
Open Vet. J.. 2025; 15(10): 5408-5414 Open Veterinary Journal, (2025), Vol. 15(10): 5408-5414 Case Report Successful treatment with recombinant human GH in a kitten with suspected congenital hyposomatotropismPauline L Corre* and Mathieu R. FaucherSmall Animal Alliance Clinic, Bordeaux, France *Corresponding Author: Pauline Le Corre. Small Animal Alliance Clinic, Bordeaux, France. Email: pauline.le-corre [at] hotmail.fr Submitted: 20/05/2025 Revised: 09/09/2025 Accepted: 22/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
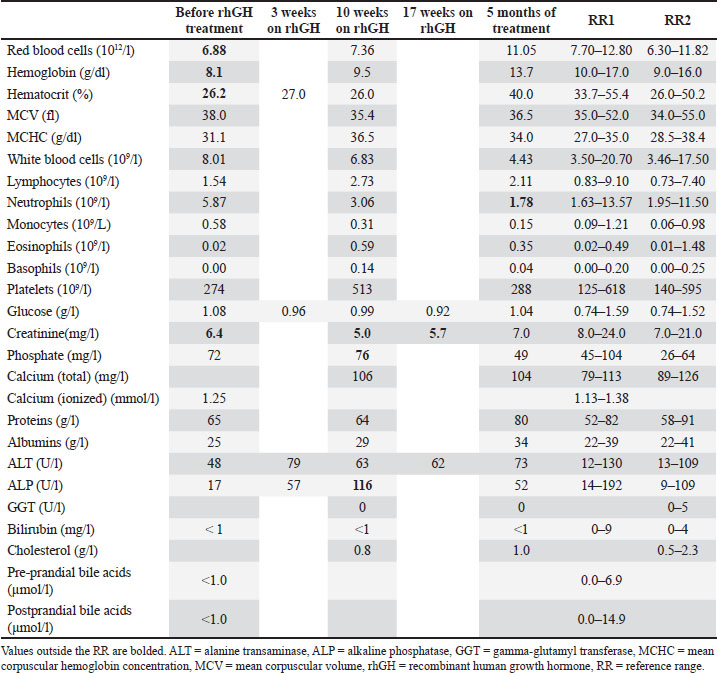
AbstractBackground: Hyposomatotropism is an extremely rare disease in cats. It is associated with stunted growth, and a severely decreased life expectancy is suspected based on the very few cases that have been reported in this species. To date, no treatment has been recommended in cats. Case description: A 4-month-old female British Longhair was presented for stunted growth without any other clinical signs reported. All other causes of growth retardation were excluded, and persistently low circulating insulin growth factor-1 levels were documented, leading to a diagnosis of presumed isolated GH-deficiency. This cat was subcutaneously treated with recombinant human GH (rhGH) for 20 weeks. At the time of treatment discontinuation, the cat had reached the weight of its littermates. No treatment adverse effects were observed. Serum IGF-1 levels were measured 5 months after treatment discontinuation and were within the reference range. Conclusion: This case report describes the successful treatment of a presumed GH-deficient cat with rhGH without adverse effects. rhGH might be a reasonable treatment option in GH-deficient cats, considering the grave prognosis associated with this disease in the few cases described. Keywords: Endocrine disease, GH, IGF-1, Pituitary dwarf, Stunted growth. IntroductionHyposomatotropism is an extremely rare disease in cats and is characterized by a primary deficiency of growth hormone (GH). Its etiology in cats is unknown due to the scarcity of reported cases. Stunted growth is a hallmark of this disease. GH is synthesized in the adenohypophysis. Its secretion is regulated by the GH-releasing hormone (GHRH) and somatostatin, both produced by the hypothalamus. The potential causes of GH-deficiency in cats can be separated into congenital defects, as described in German Shepherd dogs, or acquired disorders (trauma, neoplasia) (Greco, 2012; Feldman et al., 2015). Only a few cases of GH-deficient cats have been described (Smith and Elwood, 2004; Donaldson et al., 2008; König et al., 2018; Degregori et al., 2020; Načeradská et al., 2021; Paulin et al., 2023). Replacement therapy with recombinant human GH (rhGH) has been reported in only one case; the remaining cases were not treated either because of non-availability of veterinary products or concerns about rhGH immunogenicity (Načeradská et al., 2021). To date, little data are available regarding the prognosis of GH-deficient cats. However, this condition is likely to carry a long-term guarded to grave prognosis as seen in GH-deficient dogs and humans. No guidelines currently exist for diagnosis of GH-deficient cats or possible treatment regimens. Concerns have been raised about the side effects of GH administration to cats, particularly the development of auto-antibodies; however, their prevalence and clinical relevance are unknown. This case report describes the successful treatment of a cat with presumed GH-deficiency with rhGH without adverse effects. Case DetailsAn intact 4-month-old female British Longhair cat was referred for the evaluation of stunted growth. No digestive, respiratory, neurological, or urinary symptoms were observed. The owners deemed the appetite and general condition appropriate. No history of trauma was reported. Littermates were almost twice the weight of the cat at the time of presentation (1.1–1.2 kg against 0.615 kg). The kitten gained only 100 g between 2 and 4 months of age. A routine examination by the referring veterinarian showed harmonious stunted growth and a low body condition score 10 days before presentation. The physical examination revealed harmonious stunted growth, a body condition score of 3/9, and an unkempt, fluffy, and fine haircoat (Fig. 1). Routine serum biochemistry, electrolytes levels, complete blood count (CBC), serum pre- and postprandial bile acid concentrations were within normal physiological limits (mild anemia was attributed to the patient’s age) (Table 1). Thoracic X-rays and abdominal ultrasound were unremarkable. Limb X-rays did not reveal any abnormalities in bone density or epiphyseal dysplasia. Urinalysis revealed a mild decrease in urine specific gravity (1.028) without any other abnormality.
Fig. 1. (A) Patient at 4 months of age (first visit) (B) Patient at 21 months of age (10 months after treatment discontinuation). At initial presentation, the kitten displayed a harmonious stunted growth (half the size and weight of the littermates) and a dull and fluffy haircoat. After treatment, the cat had reached a normal size compared to its littermates. Table 1. Routine hematologic and serum biochemistry parameters before, during, and after rhGH treatment.
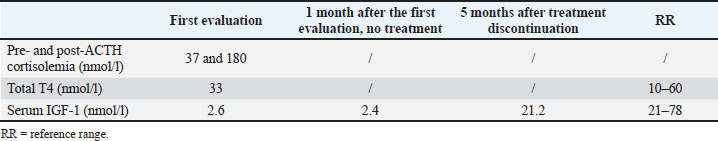
Given the stunted growth, serum total thyroxin level was measured and was within the reference range (RR), ruling out hypothyroidism (Table 2). Hypoadrenocorticism was excluded after pre- and post-adrenocorticotropic hormone cortisolemia (Table 2). Circulating serum IGF-1 was measured twice, revealing a persistently low concentration compatible with hyposomatotropism (2.6 and 2.4 nmol/l, 1 month apart; RR: 21–78 nmol/l) (Table 2). A GH-stimulation test could not be performed due to the non-availability of GHRH in France. Brain magnetic resonance imaging was also advocated, but was declined by the owner. After excluding other causes of growth retardation and the persistently very low IGF-1 concentrations, congenital hyposomatotropism was presumably diagnosed. Table 2. Endocrine testing results before and after rhGH treatment.
Treatment with rhGH was discussed with the owner and accepted. Following the protocol of a previous case report and based on extrapolation of human treatment of GH-deficiencies, a daily injection of 0.330 mg/kg of rhGH was initiated (Norditropine®) (Načeradská et al., 2021). The daily rhGH dosage was increased to 0.390 mg/kg at 2 weeks after telephone contact with the owner because of initial weak growth, which was evaluated based on the weight gain that was closely monitored by the owners. The first in-hospital re-evaluation was performed 3.5 weeks after treatment initiation. A single self-limiting vomiting episode without associated dysorexia or lethargy was reported. No clinical adverse effects were reported other than this mild vomiting episode. Given the risk of diabetes mellitus and the potential hepatotoxicity associated with rhGH treatment, serum liver enzymes alkaline phosphatase and alanine aminotransferase (ALP and ALT) and blood glucose levels were rechecked and remained within the RR (Table 1). The total weight gain observed (120 g) was still modest, and the daily rhGH dosage was further increased to 0.490 mg/kg. Email or telephone contact was maintained with the owners over the following weeks to adapt the dosage to the weight gain and screen for adverse events. Weight gain was deemed great during the remaining treatment period. Therefore, the daily dosage was maintained, with an actual range of 0.417–0.495 mg/kg/day, reflecting stepwise adjustments of injection volume to body weight (Fig. 2). The cat was reevaluated after 10 weeks on rhGH. The decidual teeth had been lost, and permanent incisors were visible; the hair coat displayed areas of adult hairs with patches of still retained kitten coat. To monitor the adverse effects of rhGH, a full serum biochemistry analysis was performed, which revealed a mild increase in ALP activity, low creatinine level, and hyperphosphatemia compatible with the cat’s age and body condition score (Table 1). The CBC was unremarkable. The rhGH dosage was left unchanged, and weight gain was progressive until the last reexamination 17 weeks after treatment initiation (Fig. 2). On this follow-up, the cat’s weight had reached 2.6 kg, the adult dentition had totally developed, and the hair coat was normal. Serum ALT activity, blood glucose, and creatinine levels were measured and were within the RR (Table 1). Owing to the costs associated with the treatment and considering the good results that had been achieved, the owners discontinued supplementation 20 weeks after treatment initiation.
Fig. 2. Weight and rhGH dose before, during, and after rhGH supplementation. The owners reported no clinical signs after the discontinuation of treatment. Five months later, the cat’s body weight reached 3.2 kg, and the physical examination was unremarkable (Fig. 1). In comparison, the weight of the 2 littermates was reported to be 3.4 kg at the time of this follow-up. Serum biochemistry and CBC were unremarkable (Table 1). The IGF-1 level was rechecked and was within the RR (21.2 nmol/l) despite treatment discontinuation. Two weeks after the last reexamination, the cat exhibited signs of sexual maturity. DiscussionThis case report describes the successful treatment of a presumed GH-deficient kitten with rhGH. This is only the second description of a GH-deficient cat treated with rhGH. The treatment regimen was chosen empirically based on a single previous case report describing clinical response along with IGF-1 increase following treatment with rhGH at an initial dosage of 1.6 mg/kg twice a week, not modified later during treatment (Načeradská et al., 2021). No studies have evaluated rhGH metabolism in cats; therefore, this treatment regimen might not be optimal in terms of safety and efficacy. A daily injection protocol with an initial dosage of 0.330 mg/kg/day (2.310 mg/kg/week) was chosen in our cat. This dosage was initially chosen lower than what was reported to monitor the onset of potential adverse effects. A daily injection protocol was selected because it may produce a more physiological effect. Furthermore, the treatment of GH-deficient children is more successful when the same weekly GH dose is administered in seven daily doses rather than in 3 or 6 injections (Feldman et al., 2015; Grimberg et al., 2016; Richmond and Rogol, 2016). IGF-1 measurements during treatment were offered to the owner to adapt the rhGH dosage, but the owner declined for financial reasons. In humans, IGF-1 concentration has recently been proposed as a tool for monitoring the response to rhGH administration in GH-deficient children and adapting rhGH dosage for optimal growth (Grimberg et al., 2016; Richmond and Rogol, 2016). However, no consensus has been reached regarding this method. In our case, the rhGH dose was regularly modified (every 2 weeks) during the treatment duration according to the weight gain, and the dosage was initially modified subjectively according to the weight gain velocity. This resulted in an overall weight and height status similar to littermates at the end of the treatment course. Because of the scarcity of case reports regarding GH-deficient cats and rhGH treatments in this species, potential adverse effects are unknown. Our cat did not show any adverse effects other than one self-limiting episode of vomiting early in the treatment course. In the previous case report, the cat showed a mild asymptomatic increase in liver enzyme activity, which spontaneously resolved after treatment cessation and could have represented an adverse effect of rhGH (Načeradská et al., 2021). This was not presented in our case report. The mild increase in ALP activity observed at the 2.5-month follow-up was attributed to growth and resolved during the follow-up period. This asymptomatic increase in liver enzyme levels has also been described in humans (4% of the cases in a study) (Salerno et al., 2000). The mechanism is yet unknown but might be related to GH-secondary increase in liver size and increased tissue transaminase content. It has been reported to occur 6–9 months after the initiation of GH supplementation and to spontaneously resolve within 3–6 months without discontinuing the treatment (Salerno et al., 2000). Overzealous GH supplementation may lead to diabetes mellitus because of the diabetogenic effects of GH (reduced muscular glucose uptake, decreased glucose oxidation, increased gluconeogenesis, and competition with IGF-1 on insulin receptors) (Scudder and Church, 2024). In two studies, the incidence of type 2 diabetes mellitus in GH-deficient children and adolescents supplemented with rhGH was increased approximately six times compared with the general population, especially when predisposing factors were present (i.e., obesity, family history of diabetes mellitus, glucocorticoid treatment, and Turner syndrome). The development of type 2 diabetes mellitus did not seem to be associated with the dose or duration of rhGH treatment (Cutfield et al., 2000; Child et al., 2011). The incidence of type 2 diabetes mellitus was similar between the two studies, with 29.3 and 34.4 cases per 100,000 person-years of GH-treatment, respectively. Some patients showed hyperglycemia resolution with GH discontinuation, some showed hyperglycemia resolution despite GH continuation, and some others remained hyperglycemic at the last follow-up under GH treatment. There are no guidelines regarding GH treatment adjustment or discontinuation after the onset of type 2 diabetes mellitus in children or adults. Long-term consequences of GH supplementation on glucose metabolism are still under investigation. In our case, the kitten did not show any clinical signs of diabetes mellitus during the treatment course and follow-up. Glucose levels were also within the RR during the follow-up period. However, it is possible that GH supplementation could lead to diabetes mellitus in some cats, especially during the treatment course. There is an approximately 33% difference in the amino acid sequence of human GH and canine or feline GH: potential immunogenicity of rhGH in cats and dogs is, therefore, a concern. In dogs, porcine GH is preferred for the treatment of hyposomatotropism because it has an identical amino-acid sequence compared with canine GH (Van Herpen et al., 1994; Feldman et al., 2015). Feline and porcine GH have only one amino-acid difference; thus, porcine GH may be a more suitable choice in this species too, but very limited availability limits its use (Feldman et al., 2015). The development of rhGH antibodies has been described and associated with collapse in one dog; this has been a motive for not treating GH-deficient dwarf dogs with rhGH (Van Herpen et al., 1994). Thus, the potential adverse effects of treatment with rhGH in dogs are largely unknown. In the cat previously reported to have been treated with rhGH and in our cat, no serious adverse effects have been observed; however, the presence of antibodies against rhGH has not been evaluated; therefore, it is not possible to draw any conclusion about potential immunogenicity in this species. More studies will be needed to evaluate the risks associated with rhGH treatment in cats, against its potential benefits, namely on survival. Among the few feline cases described with available follow-up, one 1-year-old GH-deficient cat died of congestive heart failure due to dilated cardiomyopathy, one 6-month-old GH-deficient cat died of unknown causes, and another 2-year-old cat was diagnosed with azotemic chronic kidney disease (International Renal Interest Society stage 2). Even if a cause-and-effect relationship between GH deficiency and poor outcome in the published cases cannot be ascertained, hyposomatotropism is suspected to carry a poor prognosis in felines, as reported in dogs. Indeed, a severely reduced survival in GH-deficient dogs has been reported, likely due to progressive loss of pituitary functions, expansion of pituitary cysts, renal failure, or infections (Greco, 2012; Feldman et al., 2015). In humans, GH treatment in GH-deficient individuals has been shown to improve body composition, bone mineral density, exercise capacity, and psychological well-being and has a positive impact on cardiovascular health (Grimberg et al., 2016). Moreover, anti-GH antibody development has been described in humans and does not seem to interfere with growth or have other adverse effects (Pfäffle and Kiess, 2020). Even with the possibility of anti-GH antibody development, treating GH-deficient cats with rhGH might be a reasonable choice due to the shortened survival and late consequences of GH deficiency. The most common adverse effects associated with GH replacement therapy in humans include headaches, idiopathic intracranial hypertension, increased intraocular pressure, slipped capital femoral epiphysis, and worsening of existing scoliosis. Pancreatitis, edema, arthralgia, development of insulin resistance, and disorders of glucose tolerance happen to be much less frequent (Pfäffle and Kiess, 2020). Long-term adverse effects are discordant between studies, particularly regarding the development of neoplasms or cardiovascular diseases. GH treatment is still considered safe in humans (Ranke, 2021). Owners in this case decided to discontinue the treatment due to financial concerns. Concerns are present about the need for long-term GH treatment in human medicine, particularly at adult age. A study revealed that 40%–60% of young adults who completed GH replacement therapy in childhood have a degree of GH deficiency for which treatment should be considered in adult life (Nicolson et al., 1996). In our case, an IGF-1 measurement was performed 5 months after treatment discontinuation and was within the normal range. At this time, the cat had reached a weight considered normal for its age. Therefore, the treatment was not reimplemented. In this case, a definitive diagnosis of hyposomatotropism based on stimulation tests (pre- and post-GHRH serum GH measurements) could not be performed due to the unavailability of GHRH in Europe. However, diagnosis of hyposomatotropism can be highly suspected when all other causes of stunted growth have been excluded and persistently low IGF-1 levels are documented. Traumatic and neoplasia-related GH deficiency were deemed much less likely due to the lack of other hormonal deficiencies; however, a brain MRI would have enabled the exclusion of these hypotheses. ConclusionThis is the first report of a successful rhGH treatment in a cat with presumed GH deficiency without associated adverse effects. Therefore, rhGH might be a reasonable option in GH-deficient cats compared to the poor prognosis suspected in this disease. However, the short- and long-term possible adverse effects are unknown, and there are no guidelines regarding the optimal treatment regimen in the feline species. AcknowledgmentsSpecial thanks to Dr Vet Alisée Delteil (Small Animal Mon Veto Clinic, Lescar), who initially evaluated the case. Conflict of interestNone. FundingNone. Author’s contributionBoth authors contributed equally in this case. Data availabilityThe data that support the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request. ReferencesChild, C.J., Zimmermann, A.G., Scott, R.S., Cutler, G.B., Battelino, T. and Blum, W.F. 2011. Prevalence and incidence of diabetes mellitus in GH-treated children and adolescents: analysis from the GeNeSIS observational research program. J. Clin. Endocrinol. Metab. 96, E1025–E1034. Cutfield, W.S., Wilton, P., Bennmarker, H., Albertsson-Wikland, K., Chatelain, P., Ranke, M.B. and Price, D.A. 2000. Incidence of diabetes mellitus and impaired glucose tolerance in children and adolescents receiving growth-hormone treatment. Lancet 355, 610–613. Degregori, E.B., Pippi, M.D.R., De Moura, A.L.O.N., Soila, R., Furtado, P.V. and Pöppl, A.G. 2020. Evidence of pituitary hypoplasia associated with partial central diabetes insipidus in a young persian cat. Acta Scientiae. Vet. 48, 1–7; doi: 10.22456/1679-9216.104148 Donaldson, D., Billson, F.M., Scase, T.J., Sparkes, A.H., Mcconnell, F., Mould, J.R.B. and Adams, V. 2008. Congenital hyposomatotropism in a domestic shorthair cat presenting with congenital corneal oedema. J. Small Anim. Pract. 49, 306–309. Feldman EC, Nelson RW, Reusch C, Scott-Moncrieff JCR, Berhend EN. Canine & feline endocrinology. 4th edition. St. Louis, MI: Elsevier Saunders. Greco, D.S. 2012. Pituitary deficiencies. Top. Comp. Anim. Med. 27, 2–7. Grimberg, A., Divall, S.A., Polychronakos, C., Allen, D.B., Cohen, L.E., Quintos, J.B., Rossi, W.C., Feudtner, C. and Murad, M.H. 2016. Guidelines for growth hormone and insulin-like growth factor-I treatment in children and adolescents: growth hormone deficiency, idiopathic short stature, and primary insulin-like growth factor-I deficiency. Horm. Res. Paediatr. 86, 361–397. König, M.L., Henke, D., Adamik, K. and Pérez Vera, C. 2018. Juvenile hyposomatotropism in a Somali cat presenting with seizures due to intermittent hypoglycaemia. J. Feline Med. Surg. Open Rep. 4, 205511691876144. Načeradská, M., Návojová Horáčková, K. and Fridrichová, M. 2021. Case Report: human Recombinant Growth Hormone Therapy in a DSH Cat Presented With Dwarfism. Front. Vet. Sci. 8, 773355. Nicolson, A., Toogood, A.A., Rahim, A. and Shalet, S.M. 1996. The prevalence of severe growth hormone deficiency in adults who received growth hormone replacement in childhood. Clin. Endocrinol. 44, 311–316. Paulin, M.V., Gleasure, S. and Snead, E.C. 2023. Multiple pituitary hormone deficiencies in a kitten: hyposomatotropism, hypothyroidism, central diabetes insipidus and hypogonadism. Can. Vet. J. 64, 245–251. Pfäffle, R. and Kiess, W. 2020. GH and IGF-1 Replacement in Children.In Pediatric Pharmacotherapy. Kiess, W., Schwab, M. and Van Den Anker J Cham, Germany: Springer International Publishing, pp: 67–86. Ranke, M.B. 2021. Short and long-term effects of growth hormone in children and adolescents with GH deficiency. Front. Endocrinol. 12, 720419. Richmond, E. and Rogol, A.D. 2016. Treatment of growth hormone deficiency in children, adolescents and at the transitional age. Best Pract. Res. Clin. Endocrinol. & Metab. 30, 749–755. Salerno, M., Maio, S.D. and Ferri, P. et al. 2000. Liver abnormalities during growth hormone treatment. J. Pediatr. Gastroenterol. Nutr. 31, 149–151. Scudder, C. and Church, D. 2024. Feline comorbidities: hypersomatotropism-induced diabetes in cats. J. Feline Med. Surg. 26, 1098612X. Smith, J.R. and Elwood, C.M. 2004. Traumatic partial hypopituitarism in a cat. J. Small Anim. Pract. 45, 405–409. Van Herpen, H., Rijnberk, A. and Mol, J. 1994. Production of antibodies to biosynthetic human growth hormone in the dog. Vet. Rec. 134, 171. | ||
| How to Cite this Article |
| Pubmed Style Corre PL, Faucher MR. Successful treatment with recombinant human GH in a kitten with suspected congenital hyposomatotropism. Open Vet. J.. 2025; 15(10): 5408-5414. doi:10.5455/OVJ.2025.v15.i10.59 Web Style Corre PL, Faucher MR. Successful treatment with recombinant human GH in a kitten with suspected congenital hyposomatotropism. https://www.openveterinaryjournal.com/?mno=259565 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.59 AMA (American Medical Association) Style Corre PL, Faucher MR. Successful treatment with recombinant human GH in a kitten with suspected congenital hyposomatotropism. Open Vet. J.. 2025; 15(10): 5408-5414. doi:10.5455/OVJ.2025.v15.i10.59 Vancouver/ICMJE Style Corre PL, Faucher MR. Successful treatment with recombinant human GH in a kitten with suspected congenital hyposomatotropism. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5408-5414. doi:10.5455/OVJ.2025.v15.i10.59 Harvard Style Corre, P. L. & Faucher, . M. R. (2025) Successful treatment with recombinant human GH in a kitten with suspected congenital hyposomatotropism. Open Vet. J., 15 (10), 5408-5414. doi:10.5455/OVJ.2025.v15.i10.59 Turabian Style Corre, Pauline Le, and Mathieu R. Faucher. 2025. Successful treatment with recombinant human GH in a kitten with suspected congenital hyposomatotropism. Open Veterinary Journal, 15 (10), 5408-5414. doi:10.5455/OVJ.2025.v15.i10.59 Chicago Style Corre, Pauline Le, and Mathieu R. Faucher. "Successful treatment with recombinant human GH in a kitten with suspected congenital hyposomatotropism." Open Veterinary Journal 15 (2025), 5408-5414. doi:10.5455/OVJ.2025.v15.i10.59 MLA (The Modern Language Association) Style Corre, Pauline Le, and Mathieu R. Faucher. "Successful treatment with recombinant human GH in a kitten with suspected congenital hyposomatotropism." Open Veterinary Journal 15.10 (2025), 5408-5414. Print. doi:10.5455/OVJ.2025.v15.i10.59 APA (American Psychological Association) Style Corre, P. L. & Faucher, . M. R. (2025) Successful treatment with recombinant human GH in a kitten with suspected congenital hyposomatotropism. Open Veterinary Journal, 15 (10), 5408-5414. doi:10.5455/OVJ.2025.v15.i10.59 |