| Case Report | ||
Open Vet. J.. 2025; 15(10): 5415-5419 Open Veterinary Journal, (2025), Vol. 15(10): 5415-5419 Case Report Platelet-rich plasma application for the management of early-stage Canine Legg-calvé-perthes disease: A clinical case reportViktoras Sinkevičius1, Matas Klupšas2*, Dmitrij Kvitka2 and Martinas Jankauskas21R. Sinkevičius Veterinary Clinic, Telšiai, Lithuania 2Dr. L. Kriaučeliūnas Small Animals Clinic, Faculty of Veterinary, Veterinary Academy, Lithuanian University of Health Sciences, Kaunas, Lithuania *Corresponding Author: Matas Klupšas. Dr. L. Kriaučeliūnas Small Animals Clinic, Faculty of Veterinary, Veterinary Academy, Lithuanian University of Health Sciences, Kaunas, Lithuania. Email: matas.klupsas [at] lsmu.lt Submitted: 24/05/2025 Revised: 10/09/2025 Accepted: 22/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: Legg-Calvé-Perthes disease (LCPD) is characterized by ischemic necrosis of the femoral head, primarily affecting small-breed dogs. Case Description: An 11-month-old female toy poodle presented with hindlimb lameness due to LCPD. The dog was treated with intra-articular injection of platelet-rich plasma (PRP), and clinical improvement was evaluated using lameness scoring, range of motion, thigh girth measurements, and radiographic assessment. Conclusion: PRP administration resulted in significant clinical improvement, including reduction in lameness, minimal requirement of pain relief, cessation of bone lysis, and increased muscle mass by day 44 post-treatment. Keywords: Canine, Legg-Calvé-Perthes disease, Platelet-rich plasma, PRP, Orthopedic. IntroductionAseptic necrosis of the femoral head, also known as Legg-Calvé-Perthes disease (LCPD), is characterized by ischemic necrosis of the femoral head (Crovace et al., 2020). The first reports in humans were in 1910 (Perthes, 1910) and dogs (Tutt, 1935). This disease arises due to impaired blood flow, leading to necrosis and subsequent degradation of the articular and bony tissues of the femoral head. Although the exact etiology remains unclear, several contributing factors have been proposed. These include excess sex hormones, hereditary factors in certain breeds (particularly in breeds such as the Manchester Terrier, West Highland White Terrier, Yorkshire Terrier, and Toy Poodle following a recessive monogenic model), anatomical abnormalities, increased susceptibility to intracapsular tamponade, vascular embolism, and potential coagulation disorders, although the latter remains controversial (Aguado and Goyenvalle, 2020). Clinically, the disease typically affects small breed dogs weighing less than 10 kg and aged between 5 and 8 months (Crovace et al., 2020; Eto et al., 2024). Clinical signs of the disease often include lameness, dogs typically exhibit progressive, unilateral lameness, which may worsen over time. In bilateral cases, weight shifting to the front limbs may be observed (Fossum et al., 2013; Harper, 2017). Affected dogs may struggle to rise from a lying position, climb stairs, or jump. They may also exhibit a shortened stride length and a noticeable limp (Jankovits et al., 2012; Crovace et al., 2020). During orthopedic examination, the palpation and manipulation of the hip joint often elicit pain, particularly during limb extension or abduction. In extension, the hind limb may appear shorter than the opposite limb. Crepitus may be present in advanced cases (Isola et al., 2005; Aguado and Goyenvalle 2021). A definite diagnosis of LCPD can be made using radiographs in a hip-extended ventro-dorsal position. Radiographic findings may include joint space widening, femoral head deformation, decreased bone density, and osteophyte formation, correlating with clinical severity (Ljunggren, 1967; Isola et al., 2005). Conservative therapy consists of rest, restricted exercise, and nonsteroidal anti-inflammatory drugs for analgesia, with a resolution of lameness reported in <25% of affected dogs. In rare instances where lucencies are seen without femoral head collapse, an Ehmer sling may be used for 3–4 weeks. This allows the necrotic bone to remodel without weight bearing, causing the head to collapse (Piermattei et al., 2006). The most common and most beneficial treatment for patients with Legg-Calvé-Perthes disease is surgery, which includes femoral head and neck osteotomy or total hip replacement (Jankovits et al 2012). A previous study showed that when autologous bone marrow mononuclear cells were injected into the femoral head and neck, 28 (87.5%) treated dogs showed reduced pain and improved gait within 3 weeks after a single injection (Crovace et al., 2020). Platelet-rich plasma (PRP) is a blood-derived biological product with a high platelet concentration in a small plasma volume. Methodologies to prepare PRP can rely on single centrifugation, double centrifugation, or selective blood-filtration procedures using a manual or automatic system operated in open or closed circuits (Gato-Calvo et al., 2019). After activation with calcium chloride, several growth and differentiation factors are released from alpha granules stored inside platelets, including platelet-derived growth factor, transforming growth factor β1, transforming growth factor β2, growth factor vascular endothelial growth, fibroblast growth factor and epidermal growth factor, insulin-like growth factors 1 and 2, interleukin 8, keratinocyte growth factor, and connective tissue growth factor (Cuervo et al., 2020; Catarino et al., 2020). In addition, PRP contains adhesion molecules, such as fibrinogen, fibronectin, and vitronectin (Sánchez-González et al., 2012). These bioactive molecules influence mitosis, chemotaxis, cell differentiation, undifferentiated mesenchymal cell growth, and extracellular matrix production, thereby promoting tissue regeneration (De La Mata, 2013). If PRP treatment can accelerate angiogenesis, as suggested by previous studies, it could increase blood supply to the necrotic bone in the femoral head of dogs with Legg-Calvé-Perthes disease. In this study, we aimed to evaluate the clinical outcomes of PRP therapy for treating canine Legg-Calvé-Perthes disease in a dog. Case DetailsThe study was conducted in 2025 at the R. Sinkevičius Veterinary Clinic and the LSMU Dr. L. Kriaučeliūnas University Clinic. The patient enrolled in the study was an 11-month-old female Toy Poodle weighing 2.5 kg who presented with a 3-week history of progressive left hindlimb lameness. No traumatic events were reported. On physical examination, the dog showed muscle atrophy of the left thigh, a limited range of motion, and hip joint manipulation pain. Radiographic evaluation in a ventrodorsal extended view revealed flattening and focal lysis of the left femoral head, with indistinct margins of the femoral neck, consistent with Grade 1 LCPD according to the Ljunggren classification. Subsequently, the dog underwent an experimental intra-articular PRP treatment as part of the study. The procedure, including PRP preparation and treatment administration, was performed under general anesthesia, and data were collected before and after PRP treatment. During orthopedic examinations, the dog’s posture, gait, lameness severity, range of motion (ROM) at the point of pain during hip joint flexion and extension, thigh girth measurements, and radiographic evaluations were assessed before PRP injection and subsequently on days 0, 14, and 44. The dog’s activity was restricted for 2 weeks following the initial PRP injection. The dog was confined to a strict cage rest and allowed only brief, leash-controlled walks of up to 10 minutes for elimination purposes. Before anesthesia, the dog underwent a general clinical examination, including monitoring of vital signs, such as rectal temperature, respiratory rate, mucosal color, capillary refill time, and pulse rate. (Duprey and Huff 2013). Before the anesthesia, the dog was fasted for at least 8 hours and was restricted from water for 2 hours. Sedation was achieved using 30 mcg/kg medetomidine hydrochloride (Cepetor 1 mg/ml, CP-Pharma Handelsgesellschaft mbH, Germany) and 0.2 mg/kg butorphanol (Butomidor 10 mg/ml, Richter Pharma AG, Wels) administered intramuscularly. General anesthesia was maintained with intermittent intravenous propofol top-ups (Propoven 1 %, Fresenius Kabi, Germany) at 2–4 mg/kg. Blood samples were collected on days 0, 14, and 44 to prepare autologous PRP, which was injected intra-articularly into the affected hip joint on each occasion. Therefore, the dog received three PRP treatments during the study period. Autologous whole blood (15 ml) was aseptically collected from the jugular vein into a 15-ml ACP double syringe (Arthrex, Inc., Naples, FL, USA). The double syringes were centrifuged at 1,500 rpm (350 g, soft spin) for 5 minutes. The injection site for PRP was prepared aseptically. The femoral head and neck were located by rotating the femur medially or laterally and placing an index finger on the greater trochanter. A 0.7 ml dose of PRP was injected using a 0.9 × 40 mm, 20G needle inserted between the femoral head and neck, against the bone. The limb was flexed and extended through the hip joint 10 times after injection. Radiographic examination (EcoRay 400, Germany): The hips were radiographically assessed using an extended ventro-dorsal projection (Fig. 1). The severity of LCPD affecting the hip joint was graded according to the Ljunggren classification scale (Ljunggren, 1967).
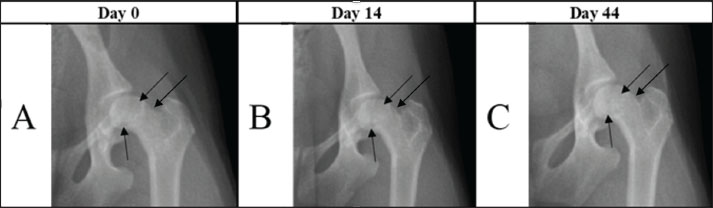
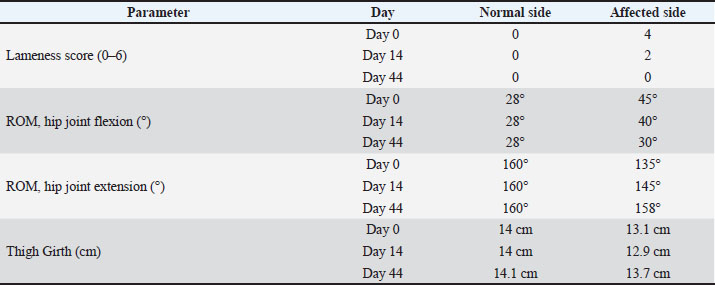
Fig. 1. Images showing reduction in femoral head and neck lysis on days 0, 14, and 44. Radiographs A and B show a decrease in the radiopaque contrast of the bone tissue (black arrows). In C, the same bone areas are homogeneous compared with other bone regions. The degree of lameness in the animal was assessed at each visit (days 0, 14, and 44) using the previously described six-point lameness scoring system (0–6) outlined by Carr DL Dycus (2016). ROM: The animals were positioned in lateral recumbency, and a conventional goniometer was placed with its fulcrum aligned over the hip joint. The pelvic axis served as the reference line, and the longitudinal axis of the femur was utilized to measure the ROM of the hip joint, following the method described by Jaegger et al. (2002). Thigh girth analysis: thigh circumference was measured using a Gulick II measuring tape. Measurements were performed at the mid-point of the femur while the animal was positioned in lateral recumbency with the limb fully extended and under sedation. DiscussionThe case involved an 11-month-old female Toy Poodle weighing 2.5 kg. The owners reported observing hindlimb lameness for approximately 1 month. Despite the previous treatment with oral meloxicam suspension (Melosus 1,5 mg/ml, CP-Pharma Handelsgesellschafts mbH, Germany) at 0.1 mg/kg for 3 weeks, the left hindlimb lameness did not improve. Upon clinical examination, the dog exhibited lameness in the affected limb, thigh muscle atrophy, hip joint crepitus, and painful flexion and extension movements. Radiographic examination confirmed grade 1 LCPD according to the Ljunggren scale (Ljunggren, 1967). On the Day-1 radiograph, there was a reduction in bone density (focal lysis) in the femoral head (Fig. 1A), and the distal margin of the femoral neck was not clearly delineated. 14 days after the PRP injection, follow-up radiographs showed diminished femoral-head lysis, but the head itself had become markedly irregular (Fig. 1B). The distal border of the femoral neck was now distinguishable. In Chinnabrut’s 2022 study, surface deterioration of the femoral head was likewise noted in two of the three cases, although the control radiographs were taken only at 8 weeks. Parra and Silva (2017) study reported a similar worsening of the femoral head surface as early as 15 days post-treatment, and this change persisted throughout the study. By day 44, the bone lysis had stopped, and the distal outline of the femoral neck was still clearly defined (Fig. 1C). In addition, the mild surface irregularity previously seen on the femoral head was partially resolved. In Chinnabrut’s 2022 study, one of the three cases exhibited no femoral head changes; however, radiographs were obtained only twice, in the first 24 hours and again at 8 weeks, so it is impossible to determine whether a transient surface deterioration similar to that observed here developed and later subsided in the interim. Clinically, lameness improved significantly by day 14, although muscle atrophy persisted, and hip flexion and extension pain-free ROM increased. By day 44, lameness had completely resolved, muscle atrophy had ceased with evidence of muscle regeneration, and both hip flexion and extension were pain-free (Table 1). Chinnabrut and Soontornvipart (2022) reported that lameness resolved in two of three dogs and improved in the third by eight weeks posttreatment. In contrast, Parra and Silva (2017) reported a transient worsening at Day 15, followed by full resolution by Day 30. These findings align with our own results, in which we documented significant clinical improvement—including resolution of lameness—was documented by Day 44. Collectively, the evidence suggests that measurable clinical benefits after intraarticular PRP may require at least 30 days to become apparent. The transient inflammation and pain seen after PRP administration may stem from proteins released by platelet α-granules: these proteins up-regulate inflammatory receptors that enhance platelet adhesion to other cells and release multiple chemokines (Blair and Flaumenhaft, 2009). Hip-joint range of motion improved in both the Chinnabrut and Soontornvipart (2022) and Parra and Silva (2017) series, although direct comparison is impossible because different scoring systems were used. Thigh-girth measurements in this case showed progressive muscle-mass gain from day 1 to day 44, despite a transient setback on day 14. Similarly, Chinnabrut and Soontornvipart (2022) recorded increased thigh muscle mass in two dogs that became sound, whereas the dog with residual lameness exhibited no muscular improvement. Table 1. Comparative results of lameness scoring, hip joint range of motion (ROM) during flexion and extension, and thigh girth measurements of both hind limbs on different assessment days following PRP injection.
The owners observed a notable improvement as early as 3 days following the initial PRP injection, an outcome not previously achieved with nonsteroidal anti-inflammatory drug treatment. Intraarticular PRP injections successfully alleviated pain and resolved lameness over the 44-day study period. No adverse effects were observed. The improvement in lameness was confirmed by increased ROM and thigh girth measurements. Although the radiographic changes on day 14 were not considered significant, clinical improvement was evident. By day 44, radiographs revealed halted lysis and increased bone tissue density (Fig. 1C). Similar observations of radiographic changes alongside clinical improvement were noted by Chinnabrut and Soontornvipart (2022). PRP is an autologous fluid concentrate primarily composed of platelets and various growth factors. Recent studies have demonstrated the role of PRP in promoting healing through the provision of growth factors, cytokines, chemokines, and other bioactive compounds (Boswell et al., 2012; Abrams et al., 2013; Pelletier et al., 2013; Hsu et al., 2013). These cytokines and growth factors effectively mitigate inflammation and initiate anabolic processes and tissue regeneration (Arnoczky and Shebani-Rad, 2013), as demonstrated by the cessation of femoral head and neck lysis observed in this study. In the Parra and Silva (2017) and the Chinnabrut study (2022), most animals demonstrated a positive clinical outcome after PRP injection, regardless of age or LCPD severity, as reflected by improvements in gait, range of motion, and pain. However, not all animals exhibited significant radiographic changes (Chinnabrut and Soontornvipart, 2022). Disease severity significantly influences treatment plans, and intraarticular PRP injections may be beneficial in early or mild stages of Legg-Calvé-Perthes disease but are not recommended for advanced or severe cases because of limited efficacy (Harper 2017; Crovace et al., 2020). The limitation of this study was the small number of suitable LCPD cases for PRP injection. LCPD is an uncommon disease and is sometimes misdiagnosed, leading to delayed diagnosis and rapid disease progression. ConclusionIntraarticular PRP injection effectively managed early-stage LCPD in a dog, demonstrating clinical improvement and bone healing promotion. The results of our study suggest that surgical treatment is a promising alternative treatment in early-stage LCPD. AcknowledgmentsNone reported. Conflict of interestThe authors have no conflicts of interest to declare. FundingThe authors have nothing to disclose. Authors’ contributionDVM Viktoras Sinkevičius: investigation and writing; DVM Matas Klupšas: investigation, writing, and preparation of the manuscript; PhD DVM Dmitrij Kvitka: revision and correction of the manuscript; DVM Martinas Jankauskas: supervision, revision, and correction of the manuscript. All authors have contributed to the study and approved the submitted version. Data availabilityAll relevant data are included in the revised manuscript. ReferencesAbrams, G.D., Frank, R.M., Fortier, L.A. and Cole, B.J. 2013. Platelet-rich plasma for articular cartilage repair. Sports Med. Arthroscopy Rev. 21, 213–219. Aguado, E. and Goyenvalle, E. 2020. Legg-Calvé-Perthes disease in the dog. Morphologie 105, 143–147; doi:10.1016/j.morpho.2020.11.011 Arnoczky, S.P. and Shebani-Rad, S. 2013. The basic science of platelet-rich plasma (PRP): what clinicians need to know. Sports Med. Arthroscopy Rev. 21, 180–185. Blair, P. and Flaumenhaft, R. 2009. Platelet α-granules: basic biology and clinical correlates. Blood Rev. 23, 177–189. Boswell, S.G., Cole, B.J., Sundman, E.A., Karas, V. and Fortier, L.A. 2012. Platelet-rich plasma: a milieu of bioactive factors. Arthroscopy 28, 429–439. Carr, D.L.D. 2016. Rehabilitation and gait analysis. In Clinician’s Brief, Tulsa, OK: Educational Concepts LLC, pp 93–100. Catarino, J., Carvalho, P., Santos, S., Martins, A. and Requicha, J. 2020. Treatment of canine osteoarthritis with allogeneic platelet-rich plasma: review of five cases. Open Vet. J. 10(2), 226–231; doi:10.4314/ovj.v10i2.12 Chinnabrut, P. and Soontornvipart, K. 2022. Preliminary: the platelet-rich plasma intra-articular injection application for canine Legg-Calvé-Perthes disease treatment. Thai J. Vet. Med. 52(Suppl.), 53–54. Crovace, A.M., Lacitignola, L., Staffieri, F., Francioso, E., Rossi, G. and Crovace, A. 2020. Treatment of monolateral Legg-Calvé-Perthes disease with autologous bone marrow mononuclear cells in 32 dogs. VCOT. Open 3, 1; doi:10.1055/s-0040-1701470 Cuervo, B., Rubio, M., Chicharro, D., Damiaá, E., Santana, A., Carrillo, J.M., Romero, A.D., Vilar, J.M., Cerón, J.J. and Sopena, J.J. 2020. Objective comparison between platelet rich plasma alone and in combination with physical therapy in dogs with osteoarthritis caused by hip dysplasia. Animals 10(2), E175. De La Mata, J. 2013. Platelet rich plasma. A new treatment tool for the rheumatologist?. Reumatología Clínica 9(3), 166–171. Duprey, L.P. and Huff, T.G. 2013. Fundamentals of physical rehabilitation, 4th ed. In: Small animal surgery. Ed., Fossum, T.W. St. Louis, MO: Elsevier / Saunders, pp: 114–130. Eto, H., Yamazaki, A., Tomo, Y., Tanegashima, K. and Edamura, K. 2024. Generation and characterization of mesenchymal stem cells from the affected femoral heads of dogs with Legg-Calvé-Perthes disease. Open Vet. J. 14(5), 1172–1181; doi:10.5455/OVJ.2024.v14.i5.12. Fossum, T.W., Dewey, C.W., Horn, C.V., Johnson, A.L., Schulz, K.S., Willard, M.D., Bahr, A. and Carroll, G.L. 2013. Small animal surgery, 4th ed. St. Louis, MO: Elsevier Mosby, p: 1321. Gato-Calvo, L., Magalhaes, J., Ruiz-Romero, C., Blanco, F.J. and Burguera, E.F. 2019. Platelet-rich plasma in osteoarthritis treatment: review of current evidence. Ther. Adv. Chronic Dis. 10, 1–18. Harper, T.A.M. 2017. Femoral head and neck excision. Vet. Clinics North Amer. Small Anim. Pract. 47(4), 885–897; doi:10.1016/j.cvsm.2017.03.002 Hsu, W.K., Mishra, A., Rodeo, S.R., Fu, F., Terry, M.A., Randelli, P., Canale, T.S. and Kelly, F.B. 2013. Platelet-rich plasma in orthopaedic applications: evidence-based recommendations for treatment. J. Am. Acad. Orthopaedic Surgeons 21, 739–748. Isola, M., Zotti, A., Carnier, P., Baroni, E. and Busetto, R. 2005. Dual-energy X-ray Absorptiometry in Canine Legg-Calvé-Perthes Disease. J. Vet. Med. Ser. A 52, 407–410. Jaegger, G., Marcellin-Little, D.J. and Levine, D. 2002. Reliability of goniometry in Labrador retrievers. Am. J. Vet. Res. 63, 979–986. Jankovits, D.A., Liska, W.D. and Kalis, R.H. 2012. Treatment of avascular necrosis of the femoral head in small dogs with micro total hip replacement. Vet. Surg. 41(2), 143–147; doi:10.1111/j.1532-950X.2011.00925.x Ljunggren, G. 1967. Legg -Perthes' disease. Acta Orthopaed. Scand. Suppl. 113, 23–26. Parra, E. and Silva, R. 2017. Autologous platelet concentrates as treatment for avascular necrosis of the femoral head in a dog. Top Companion Anim. Med. 32(1), 31–35; doi:10.1053/j.tcam.2017.05.001 Pelletier, M., Malhotra, A., Brighton, T., Walsh, W. and Lindeman, R. 2013. Platelet function and constituents of platelet-rich plasma. Int. J. Sports Med. 34, 74–80. Perthes, G. 1910. The classic: on juvenile arthritis deformans. Clin. Orthopaedics Rel. Res. 470(9), 2349–2368. Piermattei, D.L., Flo, G. and DeCamp, C., 2006. Handbook of small animal orthopedics and fracture repair. 4th ed. St. Louis, MO: Saunders Elsevier, 507–508. Sánchez-González, D.J., Méndez-Bolaina, E. and Trejo-Bahena, N.I. 2012. Platelet-rich plasma peptides: key for regeneration. Int. J. Peptides 2012, 10; 10 Tutt. and J. 1935. Tuberculosis of the hipjoint in a Cairn Terrier. Vet. Rec. 47, 428–431. | ||
| How to Cite this Article |
| Pubmed Style Sinkevičius V, Klupšas M, Kvitka D, Jankauskas M. Platelet-rich plasma application for the management of early-stage Canine Legg-calvé-perthes disease: A clinical case report. Open Vet. J.. 2025; 15(10): 5415-5419. doi:10.5455/OVJ.2025.v15.i10.60 Web Style Sinkevičius V, Klupšas M, Kvitka D, Jankauskas M. Platelet-rich plasma application for the management of early-stage Canine Legg-calvé-perthes disease: A clinical case report. https://www.openveterinaryjournal.com/?mno=260289 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.60 AMA (American Medical Association) Style Sinkevičius V, Klupšas M, Kvitka D, Jankauskas M. Platelet-rich plasma application for the management of early-stage Canine Legg-calvé-perthes disease: A clinical case report. Open Vet. J.. 2025; 15(10): 5415-5419. doi:10.5455/OVJ.2025.v15.i10.60 Vancouver/ICMJE Style Sinkevičius V, Klupšas M, Kvitka D, Jankauskas M. Platelet-rich plasma application for the management of early-stage Canine Legg-calvé-perthes disease: A clinical case report. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5415-5419. doi:10.5455/OVJ.2025.v15.i10.60 Harvard Style Sinkevičius, V., Klupšas, . M., Kvitka, . D. & Jankauskas, . M. (2025) Platelet-rich plasma application for the management of early-stage Canine Legg-calvé-perthes disease: A clinical case report. Open Vet. J., 15 (10), 5415-5419. doi:10.5455/OVJ.2025.v15.i10.60 Turabian Style Sinkevičius, Viktoras, Matas Klupšas, Dmitrij Kvitka, and Martinas Jankauskas. 2025. Platelet-rich plasma application for the management of early-stage Canine Legg-calvé-perthes disease: A clinical case report. Open Veterinary Journal, 15 (10), 5415-5419. doi:10.5455/OVJ.2025.v15.i10.60 Chicago Style Sinkevičius, Viktoras, Matas Klupšas, Dmitrij Kvitka, and Martinas Jankauskas. "Platelet-rich plasma application for the management of early-stage Canine Legg-calvé-perthes disease: A clinical case report." Open Veterinary Journal 15 (2025), 5415-5419. doi:10.5455/OVJ.2025.v15.i10.60 MLA (The Modern Language Association) Style Sinkevičius, Viktoras, Matas Klupšas, Dmitrij Kvitka, and Martinas Jankauskas. "Platelet-rich plasma application for the management of early-stage Canine Legg-calvé-perthes disease: A clinical case report." Open Veterinary Journal 15.10 (2025), 5415-5419. Print. doi:10.5455/OVJ.2025.v15.i10.60 APA (American Psychological Association) Style Sinkevičius, V., Klupšas, . M., Kvitka, . D. & Jankauskas, . M. (2025) Platelet-rich plasma application for the management of early-stage Canine Legg-calvé-perthes disease: A clinical case report. Open Veterinary Journal, 15 (10), 5415-5419. doi:10.5455/OVJ.2025.v15.i10.60 |