| Review Article | ||
Open Vet. J.. 2025; 15(10): 4834-4846 Open Veterinary Journal, (2025), Vol. 15(10): 4834-4846 Review Article Understanding bovine genital campylobacteriosis: A comprehensive review of its threat to global cattle reproductionSri Mulyati1*, Aswin Rafif Khairullah2, Rahmat Setya Adji2, Ikechukwu Benjamin Moses3, Nuha Fairusya2, Imam Mustofa1, Bambang Ngaji Utomo2, Rina Dewiyanti2, Widodo Suwito4, Harimurti Nuradji2, Sidna Artanto5, Bantari Wisynu Kusuma Wardhani6, Dea Anita Ariani Kurniasih7, Andi Thafida Khalisa8, Muhammad ‘Ahdi Kurniawan9, Adeyinka Oye Akintunde10, Syahputra Wibowo11 and Ainun Ganisia121Division of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 2Research Center for Veterinary Science, National Research and Innovation Agency (BRIN), Bogor, Indonesia 3Department of Applied Microbiology, Faculty of Science, Ebonyi State University, Abakaliki, Nigeria 4Research Center Processing Food Technology, National Research and Innovation Agency (BRIN), Gunungkidul, Indonesia 5Department of Microbiology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Sleman, Indonesia 6Research Center for Pharmaceutical Ingredients and Traditional Medicine, National Research and Innovation Agency (BRIN), Bogor, Indonesia 7Research Center for Public Health and Nutrition, National Research and Innovation Agency (BRIN), Bogor, Indonesia 8Faculty of Military Pharmacy, Universitas Pertahanan, Bogor, Indonesia 9Medical Biotechnology Research Group, Virtual Research Center for Bioinformatics and Biotechnology, Surabaya, Indonesia 10Department of Agriculture and Industrial Technology, Babcock University, Ilishan Remo, Nigeria 11Eijkman Research Center for Molecular Biology, National Research and Innovation Agency (BRIN), Bogor, Indonesia 12Faculty of Health Sciences, Universitas Dr. Soetomo, Surabaya, Indonesia *Corresponding Author: Sri Mulyati. Department of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: sri-m [at] fkh.unair.ac.id Submitted: 31/05/2025 Revised: 28/08/2025 Accepted: 10/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
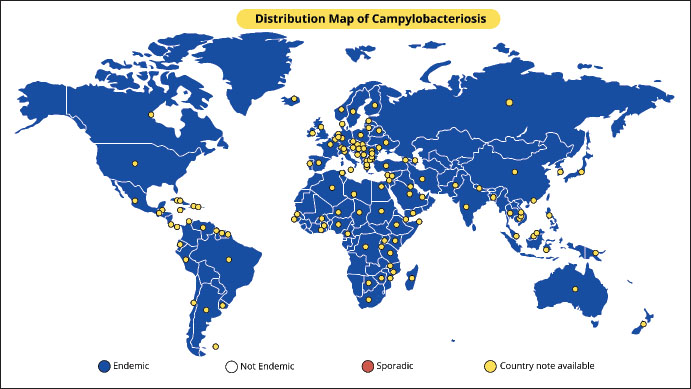
AbstractBovine genital campylobacteriosis (BGC) is a sexually transmitted disease caused by Campylobacter fetus subsp. venerealis (Cfv). Although BGC has been reported globally, its prevalence is significant in developing nations where natural breeding is commonly employed for cow reproduction. It is found worldwide. Female cattle with this disease may become temporarily infertile, and a tiny percentage (5%–10%) of affected animals may occasionally have abortions in the third trimester. The subclinical manifestation of this disease in bulls, where there are no overt clinical signs and a protracted possible carrier stage, is a significant feature. Sexually transmitted Cfv can lead to reproductive issues, including lower cow birth rates, return of estrus, and low pregnancy rates. Campylobacter fetus infections in the genital tract of cows and heifers typically only cause mild to moderate endometritis, moderate cervicitis, and salpingitis. The most widely used diagnostic techniques are polymerase chain reaction, fluorescent antibody test, enzyme-linked immunosorbent assay, and culture. Commercial vaccinations for BGC prevention are administered intramuscularly or subcutaneously and contain bacterin and adjuvant. BGC-positive bulls are treatable with both systemic and topical antimicrobial therapy. The disease is usually self-limiting, and cows may return to estrus after a few months. Control strategies include testing and culling of affected animals, vaccination of all bulls, cows, and heifers, and use of artificial insemination with controlled semen. Keywords: Abortion, BGC, Cow, Ruminants, Sexually transmitted disease. IntroductionBovine genital campylobacteriosis (BGC), also known as bovine venereal campylobacteriosis (BVC), is a sexually transmitted disease (STDs) in cattle (Mshelia et al., 2010). This disease is caused by Campylobacter fetus subsp. venerealis (Cfv) (Pena-Fernández et al., 2024). There are two closely related subspecies of C. fetus: C. fetus subsp. venerealis (Cfv) and C. fetus subsp. fetus (Cff). In cows, BGC can lead to fertility issues such as pregnancy failure, heat return, and lower birth rates (Seid, 2019). While Cfv is linked to endemic abortion and fertility problems in some countries, such as Argentina, Brazil, Uruguay, Japan, New Zealand, Spain, Germany, and the United Kingdom, Cff is linked to occasional incidences of bovine abortion (Cabell, 2007; Abdel-Glil et al., 2023). BGC is prevalent worldwide and is particularly common in underdeveloped nations where cattle reproduction is primarily accomplished by natural breeding (Sahin et al., 2017; Balzan et al., 2020). Furthermore, BGC is included on the World Organization for Animal Health’s (WOAH) list of diseases that must be reported and significantly affect global commerce in animals or animal products (WOAH, 2023). The main sources for isolating Cfv pathogens are the internal organs of aborted fetuses or the genital tract of affected cattle (such as the preputial smegma or vaginal mucus) (Truyers et al., 2014). Abortion can occur spontaneously after the initial infection; however, its chance of occurrence typically increases during the third trimester when the fetus is most rapidly growing because of increased nutritional factors (Seid, 2019). Bulls are considered the reservoir of the disease (European Food and Safety Authority et al., 2017). Sexual contact is the main way that the bacterial pathogen is transmitted, and it results in irritation of the female genital tract and infertility (Campos-Múzquiz et al., 2019). Every bull has a different level of vulnerability to illness; some seem to be immune to infection, while others develop into lifelong carriers. This heterogeneity is probably caused by age-related changes in the preputial and penile epithelial crypt depth (Balzan et al., 2020). BGC can be disseminated by artificial insemination (AI) using the semen of infected bulls or through contaminated equipment, although natural mating is the main way that the disease spreads (Morrell, 2006). Cows with BGC typically become sterile and require more services for fertilization (Bondurant, 2005). The main causes of cases or outbreaks are ineffective management of large-scale livestock movements across national boundaries and the introduction of bulls, cows, and heifers from endemic herds (El-Adawy et al., 2023). BGC is a finicky and slow-growing bacterium that is frequently overtaken by pollutants, making it challenging to diagnose by culture (Bastos et al., 2023). This disease is frequently overlooked until several heifers start exhibiting estrus. The most widely used diagnostic techniques are the fluorescent antibody test (FAT), ELISA, polymerase chain reaction (PCR), and culture (Aydin et al., 2025). Additionally, ELISA can be used to show that vaginal mucous contains particular antibodies (Aydin et al., 2025). Cfv can be directly detected in clinical samples using a real-time PCR technique (Chaban et al., 2012). Diagnosis for identifying the bacterial pathogen involves a culture of preputial washings, vaginal mucous, or fetal aborted material. Campylobacterios-related cases of reduced bovine reproductive performance, particularly in endemic regions, result in fertility issues and large financial losses (Seid, 2019). Designing effective and long-lasting control programs for contemporary livestock systems requires a thorough understanding of disease transmission traits and mechanisms. This article aims to summarize and analyze various scientific findings related to BGC epidemiology, pathogenesis, diagnosis, and control aspects. This review is anticipated to be a valuable resource for researchers, students, and practitioners in the field of animal health. This will help provide fast and accurate information for the early detection, prevention, and control of this disease to support the reproductive health and productivity of cattle. EtiologyThe word “Campylobacter” originated from Greek and means “curved bacillus” (Yousaf Kazmi, 2022). Campylobacter spp. are oxidase-positive, microaerophilic, catalase-variable, and Gram-negative curved rods with a diameter of 0.2 to 0.5 μm (Modi et al., 2015). These bacteria can grow at 25°C, and it takes at least 2 days to cultivate at 37°C in a microaerophilic environment (Wagenaar et al., 2014). A single polar flagellum at one or both ends of the cell rotates in a spiral motion for locomotion (Cohen et al., 2024). The colonies are slightly gray-pink, round, convex, shiny, smooth, and have uniform borders (Modi et al., 2015). The genus Campylobacter is a microaerophilic bacterium that typically requires oxygen concentrations of 3%–15% and carbon dioxide concentrations of 3%–5% (Kaakoush et al., 2007). Few species can grow in the presence of oxygen because they are aerotolerant. There are now 33 species of Campylobacter known to exist, including C. fetus (http://www.bacterio.net/campylobacter.html). There are three known subspecies of C. fetus: C. fetus subsp. testudinum (Cft), C. fetus subsp. fetus (Cff), and C. fetus subsp. venerealis (Cfv). Cft is linked to reptiles, whereas Cff and Cfv are linked to mammals (Emele et al., 2019). Although the clinical indicators linked to infection by both viruses overlap, both Cfv and Cff were originally classified according to habitat features, transmission, clinical manifestations, and laboratory phenotypic identification (Emele et al., 2019). According to Holt et al. (1994), these subspecies were phenotypically identified by testing for tolerance to 1% glycine and hydrogen sulfide (H2S) generation; Cff and Cfv showed positive and negative results, respectively. Cfv is still found as a biovar, known as Cfv biovar intermedius, and is categorized according to its capacity to generate H2S but is insensitive to 1% glycine (Holt et al., 1994). HistoryIn 1909, McFadyean and Stockman recovered the first spirilla from abomasum fluid samples from aborted sheep and cow fetuses in England (Louwen et al., 2012). Smith identified the same germ in bovine abortions in the United States in 1918 and called it Vibrio fetus in Taylor’s stain (Smith et al., 1920). This same agent was shown to be the cause of enzootic sterility in cattle in the Netherlands in 1949 (Vinzent and R, 1949). In 1956, it was shown that the bacteria that cause enzootic sterility and sporadic abortion in sheep and cattle belonged to separate subspecies (Vandeplassche et al., 1976). In 1959, a distinction was made between intestinal vibriosis, which was caused by V. fetus (intestinalis) and manifested as enzootic abortions in sheep and sporadic abortions in cattle, and venereal vibriosis, which caused enzootic sterility with abortion (Florent, 1959). Additionally, he discovered strains with a vaginal and uterine tropism that produced hydrogen sulfide and were less tolerant of the presence of glycine. This bacterium, V. fetus (intermedius), was believed to cause enzootic sterility disease in cattle (van Bergen et al., 2005). In 1953, Florent isolated a microbe with traits of the genus “Vibrio” but saprophytic, which he named Vibrio bubulus, from the genitals of bulls (Córdova-Izquierdo et al., 2016). It was subsequently referred to as Campylobacter sputorum subsp. bubulus. Sebald and Véron (1963) created a new genus named “Campylobacter”, which included the species C. fetus and its subspecies (Véron and Chatelain, 1973). The bacteria in the genus “Vibrio” ferment glucose and have between 40% and 53% guanine and cytosine (G + C) in their DNA, whereas the bacteria in the genus “Campylobacter” are asaccharolytic and have between 29% and 36% G + C (Hugh and Leifson, 1953). EpidemiologyBGC, caused by Cfv, is found worldwide, although many countries do not have accurate data (Pena-Fernández et al., 2021). Older bulls have a higher frequency of BGC (Pena-Fernández et al., 2021). The persistence of C. fetus colonization in the preputial and penile mucosa of adult bulls is associated with larger and more irregular crypts compared with younger bulls (Silveira et al., 2018). Nonetheless, Mai et al. (2013) noted that bulls of all ages may continue to be carriers following infection. Cows that have not been infected by natural mating can naturally become infected by the bull’s prepuce (Truyers et al., 2014). The disease is mainly observed in newly brought cows and heifers in herds where BGC is endemic, but the addition of new carrier bulls presents a special risk to disease-free herds (Balzan et al., 2020). Although infected bulls are the main source of infection for susceptible cows, contamination can also occur during AI using contaminated insemination equipment or infected semen (Hoque et al., 2023). Figure 1 depicts the global distribution of BGC. The farming system, disease surveillance, and reproductive management techniques affect the distribution of BGC in different countries and areas. The prevalence of BGC disease is higher in developing nations where cattle are commonly bred naturally, whereas infection rates are lower in wealthy nations because natural breeding is mostly limited to herds of beef cattle (El-Adawy et al., 2023). The incidence of BGC has significantly decreased in dairy herds after the implementation of AI (Julie Gard, 2016). As evidenced by the underreporting of illness incidence in most nations, the lack of regular disease monitoring systems may result in a significant underestimation of BGC in Africa (Mai et al., 2015).
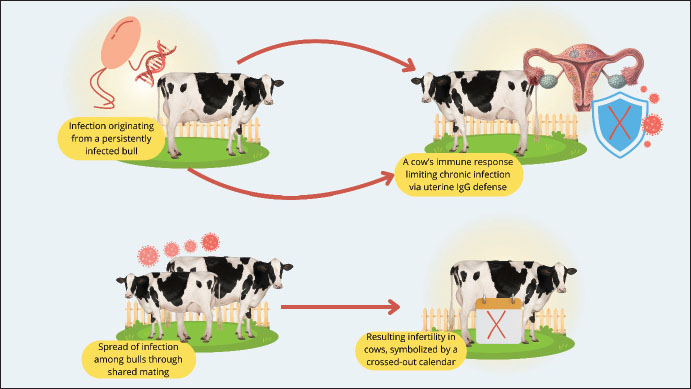
Fig. 1. Global endemicity map of Bovine genital campylobacteriosis (Source: This study). The prevalence of BGC is comparatively low in industrialized nations such as the United States, Canada, Mexico, and many Western European nations (Tremblay et al., 2003; Iraola et al., 2017; El-Adawy et al., 2023; Raúl et al., 2023). This is a result of stringent animal health monitoring programs and regulations, as well as the extensive use of reproductive technologies such as artificial intelligence (AI). However, isolated incidents are still possible, particularly on farms that continue to practice mating. However, BGC is still widely prevalent in regions with more extensive and traditional livestock systems, including several Latin American nations, Sub-Saharan Africa, and a large portion of South and Southeast Asia (Madoroba et al., 2011; Silveira et al., 2018; Hoque et al., 2023). Furthermore, the lack of national control programs, laboratory infrastructure, animal health monitoring, and farmer awareness of infectious illnesses are the primary barriers to reducing BGC. BGC is highly endemic in locations with sizable cattle populations and less intensive livestock systems, such as Brazil, Argentina, India, and Indonesia (Pellegrin et al., 2002; Beniawan et al., 2020; Ishtifaq et al., 2020; Oyhenart, 2024). Although the prevalence statistics are not always clear, similar illnesses have been found in several parts of the Middle East and North Africa (Mai et al., 2013; Hoque et al., 2023). However, BGC is not commonly endemic; however, cases have been reported in Australia and New Zealand (Juli et al., 2024). Its expansion has been slowed by a well-established system for identifying and controlling animal diseases. Low frequency of reproductive systems in East Asian nations with high biosecurity regulations and technology-based reproductive systems, such as South Korea and Japan (Yamazaki et al., 2009; (Seong et al., 2021). PathogenesisFollowing sexual contact with a carrier bull, C. fetus remains in the vaginal fundus of the female until completion of estrus (Mshelia et al., 2010). This could be because the reproductive tract’s enhanced blood flow of IgG1 and activated polymorphonuclear cells during this phase inhibits microbial adherence and colonization (Campos-Múzquiz et al., 2019). Campylobacter fetus that is deposited in the vagina during sexual activity reproduces, makes its way to the uterine corpuscle by day 5, reaches the uterine horns between days 12 and 14, and is found in some animals’ oviducts by day twenty after infection. During the progestational phase, the thick cervical mucus that clogs the cervix becomes liquid, and the cervix’s muscles relax, allowing microorganisms to enter the uterus, especially due to the lack of a uterine defense mechanism (Campos-Múzquiz et al., 2019). Once the bacterial pathogen infects the uterus, it sticks to the mucosa, causing endometrial inflammation and occasionally cervicitis, salpingitis, and vaginitis. This lowers the partial tension of oxygen and vital nutrients for the embryo, thus preventing the fertilized egg from implanting and affecting the developing embryo’s survival (Silveira et al., 2018). Bacterial elimination starts 40–60 days after the uterus and vagina begin to produce immunoglobulins (Córdova-Izquierdo et al., 2016). Bacteria can begin to act on days 12–14 of the embryo’s development. The female will repeat estrus on day 21 if this continues until day 14 because pregnancy will not be recognized, and the estrus cycle will not be impacted. However, depending on how long it takes for the uterus to regain its function during embryo development, it enters the estrous phase between 62 and 90 days; thus, extending the estrous cycle. As a result, the body and tail will rise in the distribution of the birth curve (Catena et al., 2003). The fetus or embryo usually dies during the first 90 days of pregnancy. Female cows may continue to conceive in 5%–10% of cases; however, infection may result in an abortion in the fifth or sixth month (Córdova-Izquierdo et al., 2016). However, research at INTA Balcarce showed that campylobacteriosis infections can linger for up to 14 months, indicating that the phenomenon of “autoimmunity” which helps cows avoid infection, should not be undervalued. The herd’s “carrier females,” who carry the illness in their vaginal system, may be explained by this fact (Terzolo et al., 1996). Since semen’s ability to fertilize remains unaffected, the sperm fertilizes the egg effectively, and pregnancy starts until bacterial invasion of the uterus (beginning on day 20) alters the environment and creates an unfavorable condition for the developing embryo. The animal endures infertile heat and inflammation until the endometrium returns to its normal form, and the microbes have been eradicated from the uterus (Córdova-Izquierdo et al., 2016). Competition for nutrients in the uterus and fallopian tubes, the habitat of the conceptus, is one of the virulence mechanisms of C. fetus. This is believed to be a reduction in the oxygen and nutrition pressure that the embryo needs during the preimplantation phase (Córdova-Izquierdo et al., 2016). Virulence factorsIn C. fetus, virulence factors include flagella, mucinase, lipopolysaccharide (LPS), cytolethal toxin (CLDT), K antigen (microcapsule), peritrichous pili, and flagella, the latter two of which are involved in adhesion (Catena et al., 2003). Flagella provide the bacterial pathogen mobility, but mucinase breaks down the protective mucus, delaying implantation and ultimately leading to the embryo’s death (Sprenger et al., 2012). The cell walls of gram-negative bacteria contain LPS, which causes inflammatory responses (Sprenger et al., 2012). Although its synthesis has been confirmed in cell culture in a strain of C. fetus obtained from cattle, its impact on the embryo is unknown (Dworkin et al., 1995). Regarding adhesion, which is a crucial “step” in colonization, electron microscopy has described structures resembling flagellar adhesions and peritrichous pili (Wassenaar, 1997). Somatic or thermostable (O), flagella (H), and thermolabile surface antigen (K), also known as the S-Layer, which covers the O antigen, are the three types of antigens found in Campylobacter (Inglis et al., 2004). The existence of a microcapsule, a structure composed of high molecular weight proteins, is another virulence feature linked to specific strains of C. fetus known as the S-Layer (+) (Thompson, 2002). These proteins are heterogeneous in terms of size, shape, and conserved and divergent sequences. Antigenic variation is linked to this heterogeneity, enabling the agent to elude the immune system and endure in the reproductive system (Thompson, 2002). Microcapsules play a significant role in the resistance of the bovine genital tract to Cfv infection (Sprenger et al., 2012). This protects the vaginal tract from microbial colonization. Additionally, during infection, the antigenic structure of this layer changes, enabling the bacteria to evade the host immune response and create a carrier state in both male and female (Córdova-Izquierdo et al., 2016). Macrophages can easily phagocytose S (−) layer strains, but S (+) layer strains are immune to the two most crucial defenses against extracellular Gram-negative bacteria: phagocytosis and serum bactericidal activity (Thompson, 2002). The complement component C3b’s incapacity to adhere to the surface of S-layer (+) cells and the resulting antigenic diversity are the causes of this. However, because of its poor invasiveness and immunological response, the antigenic diversity of C. fetus enables subpopulations to evade detection and continue to proliferate, leading to persistent infection ((Bénéjat et al., 2025). Antigenic variation causes evasion until the host response reaches the level of specificity and magnitude required to eradicate infection (Corbeil et al., 2003). Immune responseThe immunological response to C. fetus is mostly local. IgG1 immunoglobulins, which are strong opsonins that cooperate to quickly eradicate infection, are the most prevalent in the uterus (Cipolla et al., 2001). Immunoglobulin A (IgA), the most prevalent type of immunoglobulin in cervical mucus, attacks bacteria and stops them from infecting the uterus; however, IgA might not be able to effectively eliminate the bacterial pathogen as they have poor opsonization ability (Monroy et al., 2010). IgA can immobilize C. fetus, preventing it from adhering to the epithelium (de Vargas et al., 2002), whereas immunoglobulin G (IgG) opsonizes it and immobilizes it, enabling phagocytosis. Infectious bacteria must adhere to the mucosal surface for the disease to manifest. IgA stops infection by blocking binding (de Vargas et al., 2002). IgG opsonization ability enables the removal of bacteria from the uterus and explains why fertility is restored in both sick and vaccinated individuals, even when isolated bacteria are present in the vagina (Inglis et al., 2004). Previous research indicates that IgA antibodies are more effective than IgG antibodies at inactivating C. fetus; however, IgA antibodies do not trigger the opsonization and death of intracellular C. fetus by neutrophils and macrophages, unlike IgG antibodies, which can trigger opsonization and intracellular death (Corbeil et al., 2003). PathologyInfections in the genital tract of cows and heifers cause mild to moderate endometritis, cervicitis, and salpingitis (Sheldon and Owens, 2017). The uterus may contain a tiny quantity of mucopurulent exudate that may spread through the cervix into the vagina (Hermadi et al., 2024). Microscopically, these alterations are hardly noticeable and mostly comprise a mild diffuse infiltration of inflammatory cells and minimal superficial desquamation of the epithelium (Campos-Múzquiz et al., 2021). Although there is little endometrial glandular involvement, a few cystic glands with mild periglandular fibrosis can be observed at resolution (Campos-Múzquiz et al., 2021). Plasma cell infiltration and lymphocyte foci in the stroma are indicators of mild endometritis (Hermadi et al., 2025). Bulls typically show no obvious preputial or penile mucosa abnormalities (Hermadi et al., 2025). The lamina propria exhibits diffuse mononuclear cell infiltration. Plasma cells are typically found in clusters at the apex of the dermal papillae in older bulls (Chen et al., 2021). Clinical signsThe clinical signs of BGC include transient infertility and occasional abortions in cows (Silveira et al., 2018). The subclinical character of this disease in bulls, where there are no overt clinical symptoms and a protracted possible carrier stage, is a significant feature (Hoque et al., 2023). No discernible clinical symptoms other than an unusually protracted estrous cycle or a late resumption of estrus are observed in females. Subclinical endometritis, cervicitis, and vaginitis can occasionally cause vaginal discharge within a few weeks after infection (Campos-Múzquiz et al., 2021). Most infected cows do not exhibit any clinical signs while the infection is active. Infected cows typically experience irregular abortions in the third trimester of pregnancy, many months after infection (Owusu-Ansa et al., 2025). Heifers and cows that have never been exposed to Cfv are extremely prone to infection, and this vulnerability does not seem to decrease with age (European Food and Safety Authority et al., 2017). Cattle that have recovered from the illness become immune to disease recurrence. The immunological state of recovered cattle deteriorates over time and, in many cases, completely disappears 3–4 years after the initial infection (Raúl et al., 2023). Most cows in outbreaks among recently infected herds go back to work, and only 25%–50% of them become pregnant after a few weeks. It frequently takes more than 60 days on average from the start of labor to pregnancy (Campos-Múzquiz et al., 2019). A percentage of infected females may miscarry, but most will eventually become pregnant (Gribble et al., 1981). Although the majority are infection-free after 3 months of delivery, up to 10% may still be infected after delivery (de Vargas et al., 2002). DiagnosisDark-field or phase-contrast microscopy of placental scrapings and fresh fetal abomasum contents is a well-known technique for diagnosing BGC (Paisley et al., 1982). Campylobacter species are identified by the presence of thin spiral or curved rods that measure 0.2–0.9 μm × 0.5–5.0 μm, exhibit corkscrew-like activity, and moving quickly (Chon et al., 2020). Prior to examination, this approach can also be used to identify putative Campylobacter colonies suspended in saltwater; however, the culture media may contain coccoid forms. The gold standard technique for the diagnosis of BGC is isolation by culture (García et al., 2023). Since C. fetus are picky eaters, the accuracy of culture procedures (such as collection of samples, transportation from collection source to the laboratory for analysis, and cultivation/recovery of C. fetus) is dependent on the size of the inoculum, the presence of competing microflora in the sample, the conditions in which the sample was collected and transported to the laboratory, and the experience of the laboratory technician (Schulze et al., 2006). Bacteria need an environment that contains 85% nitrogen, 10% carbon dioxide, and 5% oxygen for survival and reproduction (Van Alst et al., 2022). Filtration or selective agar should be used to support Campylobacter growth while preventing the overgrowth of other bacteria (Kim et al., 2016). Although a variety of media have been described for the isolation of Campylobacter spp., some are unsuitable for C. fetus culture because they include specific antimicrobials, such as cephalosporins (McGready et al., 2010). Skirrow medium is the suggested culture medium for C. jejuni and C. fetus (Neyaz et al., 2024). However, Monke et al. (2002) deduced that the recovery of C. fetus subsp. venerealis is improbable if the transit period is longer than 24 h based on the difference in culture findings between 4 and 24 hours. Cfv and Cff can be distinguished from one another using the distinct DNA banding patterns that each subspecies exhibits on an agarose gel following electrophoresis (Monke et al., 2002). Although there are several distinct identifiers, Cfv and Cff share high sequence identity (Monke et al., 2002). Pathogenicity islands and mobility genes, such as transposable insertion sequences, are distinguishing features that have been incorporated into the design of multiple PCR primer sequences. The MG3F/MG4R primers for the carbon-deficient protein gene found in both subspecies of C. fetus and the VenSF/VenSR primers, a portion of the parA gene peculiar to Cfv, are the most reliable and quantifiable (Chaban et al., 2012). Amplification or restriction fragment length polymorphism (RFLP) is an additional molecular technique that can be used in conjunction with PCR to boost its sensitivity (Hashim and Al-Shuhaib, 2019). Since the full Cfv genome was previously published, better diagnostic tools have been developed, such as real-time PCR, which reduces the requirement for previous culture by directly detecting Cfv from clinical samples (Pena-Fernández et al., 2024). In addition to being helpful for the direct detection of Cff in abomasum samples from aborted fetuses, the multiplex assay is frequently used for the identification and distinction of Cff and Cfv isolates. The 16-multiplex assay, which can distinguish between C. fetus and C. jejuni cultures as well as other Campylobacter species, was developed using an adaptation of this assay, which targets the cstA gene (Ricke et al., 2019). ELISA was created to identify antigen-specific secretory IgA antibodies in the vaginal mucus of calves that may have had a prior infection with Cfv. Presently, IgA ELISA seems to be the preferred test for the retroactive diagnosis of bovine campylobacteriosis, with a reported specificity of 98.5% (Hum et al., 1994). Microorganisms in samples can be directly diagnosed using immunofluorescence assays, or their identity can be verified after isolation (Miller et al., 2018). FAT is frequently employed as a preliminary sample screening test (Cipolla et al., 2001). Immunofluorescence assays have not been thoroughly tested, and issues have been noted with poor conjugate availability, nonspecific fluorescence that can lead to false-positive results, and the inability to distinguish between distinct C. fetus subspecies (Silveira et al., 2018). Additionally, technicians need to be skilled and knowledgeable to ensure that subjectivity does not affect test results. In 2009, surveys on the presence of C. fetus in various parts of Brazil made extensive use of the Direct Flourescent Antibody Test, which had a high positive rate (Balzan et al., 2020). Due to the commercial unavailability of conjugates, the application of this technology is now restricted to research laboratories that create the input. Differential diagnosisCattle herds experience reproductive issues due to other STDs, which should be considered for an accurate diagnosis. Serum samples are typically used to diagnose reproductive diseases, although samples including preputial smegma, semen, fetal fluid, placenta, and vaginal fluid can also be used to provide a conclusive diagnosis (Givens, 2006). Bovine genital trichomoniasis is caused by the protozoan Tritrichomonas fetus, which also produces a disease that resembles BGC (which also causes early embryonic death in cattle) and should be investigated as a differential diagnosis (Hermadi et al., 2024). For differentiation, PCR and culture are typically used to diagnose trichomoniasis, and the same sample type is used for BGC diagnosis (Michi et al., 2016). Additionally, viral infections, including bovine viral diarrhea (BVD) and infectious bovine rhinotracheitis, can induce infertility or abortion in calves, as can Haemophilus somnus, Ureaplasma, and other Mycoplasmas, as well as, in certain cases, Leptospira spp. and Brucella abortus (Givens, 2018). Laboratory test findings, clinical symptoms, and epidemiological traits must all be considered in these situations (Givens, 2006). Arcobacter spp. is another microbe that resembles Campylobacter in microbiological culture (Shange et al., 2019). The application of a selected broth for enrichment can eradicate these bacteria that thrive in aerobiosis. Although Arcobacter spp. was identified in cases of abortion in cows, it was also discovered in cow preputial fluid (also known as smegma) and vaginal swabs from cows that did not exhibit any reproductive issues (Collado and Figueras, 2011). Thus, Arcobacter spp. should be considered for differential diagnosis when samples from animals suspected of having BGC are cultured microbiologically (Shange et al., 2019). TransmissionCfv transmission is entirely through breeding (Sahin et al., 2017). Persistently infected bulls are the most common source of this infection in cow herds, although cows are less common (McFadden et al., 2005). A bull may carry the Cfv for a long time after being infected by caring for a carrier female, which could spread the infection to other bulls during mating (Dawson, 1986). Figure 2 illustrates the transmission process, which demonstrates the initiation of infection from a persistently infected bull, subsequent horizontal transmission among bulls through shared natural mating, and eventual transmission to susceptible cows. Genetic alterations in the infectious strain as it attempts to elude the immune system may be factors influencing the development of chronic infections (Wagenaar et al., 2014). Bulls differ widely in their capacity to harbor C. fetus and the duration of time they can remain carriers, and this antigenic change has not been thoroughly described (Balzan et al., 2020). The incidence of the carrier stage in females is not well documented; however, persistent infection is less common. This is believed to be caused by a robust immune response dominated by IgG in response to bacterial colonization of the uterus and cervix (Shen et al., 2023).
Fig. 2. Transmission of Bovine genital campylobacteriosis (Source: This study). Management strategies have a significant impact on the frequency of subsequent infertility episodes once an infection has entered a herd (Owusu-Ansa et al., 2025). Pregnancy rates of 40%–60% in recently infected herds are regularly reported in field observations conducted in Australia and abroad (Owusu-Ansa et al., 2025). Infertility is more severe in herds where mixing restrictions are in place because it frequently takes more than 60 days from the start of treatment to the development of pregnancy after infection (Cagnoli et al., 2020). In contrast to multiple sire mating, individual breeding units in sensitive herds always experience significant infertility, which allows for the availability of both infected and uninfected sires (Cagnoli et al., 2020). Pregnancy rates are typically higher and may seem good in herds where the disease is endemic. Acquired immunity usually prevents previously infected females from having reduced fertility (Cagnoli et al., 2024); however, the percentage and quantity of vulnerable heifers or replacement virgin heifers added to an endemically contaminated breeding unit will significantly affect the level of infertility (Pena-Fernández et al., 2021). Risk factorsThe greater infection rate in bulls aged >5 years may be caused by the pathogen’s ability to thrive in the deeper epithelial crypts found in the prepuce and penis of older bulls (Silveira et al., 2018; Cagnoli et al., 2020). It is most likely the result of consecutive (or discontinuous) alterations in the organism’s surface antigens that C. fetus infection persists in the bovine genital canal. Rearrangement of the homologous sapA gene genome is linked to the occurrence of this antigenic shift in vivo (Wagenaar et al., 2014). Economic impactSince infection in a herd is readily undetected, campylobacteriosis has been dubbed the “silent profiteer” (Thompson, 2002). Usually, infection is not recognized until the herd exhibits poor birth rates, at which point significant losses have already taken place (van Bergen et al., 2005). Reduced birth rates, postponed births, sterile animal culling, and abortions are the primary causes of farmers’ financial losses (Underwood et al., 2015). The disease is more likely to occur in regions where natural breeding and intensive livestock management are prevalent (Bondurant, 2005). In BGC, abortion rates can be as high as 10% and pregnancy rates as low as 20%. Infertility can occur in up to 11% of infected heifers (Seid, 2019). A growing embryo in the uterus is more frequently lost because of this illness, but it can also prevent the implantation of a fertilized egg. The animal typically returns to estrus after this, although frequently with lengthy and erratic cycles (Sprenger et al., 2012). Cattle with campylobacteriosis may experience erratic heat due to early embryonic death, irregular abortions, temporary or permanent sterility, and disruptions in the breeding schedule (Delano et al., 2002). After months of being out of production, leading to financial losses, animals that recover from illness are regularly put back to work. Although this was not quantified, Akhtar et al. (1993) demonstrated that Cfv infection in dairy herds had a detrimental effect on milk output. According to Hum (2004), campylobacteriosis causes a considerable amount of reproductive waste in infected dairy and beef herds in Australia. According to estimates, gross profit margins may drop by as much as 66% in the first year of infection, resulting in a considerable financial loss for the producer. Profit margins in a disease-affected herd might decrease by up to 36% compared with a herd that is not affected by the disease (McMillen et al., 2006). The OIE presently lists campylobacteriosis as a “List B” notifiable disease because it is thought to have public health and socioeconomic repercussions (Oyarzabal et al., 1997). Consequently, the disease has an impact on global commerce in animals and animal products. Many nations demand that bovine semen, embryos, and cows be free from CfV infection to import and export. They also require that bulls at semen production and distribution facilities be certified healthy (Balzan et al., 2020). Consequently, it is crucial to use precise and trustworthy diagnostic procedures to identify campylobacteriosis (Khairullah et al., 2024). TreatmentBGC-positive bulls and cows are treatable (Underwood et al., 2015). Given the high cost of therapy and the requirement to care for the animal for several days, decisions about bull treatment must be made with the animal’s age and worth in mind (Balzan et al., 2020). Systemic and topical antimicrobial therapy have been used with differing degrees of success. Oxytetracycline, dimetridazole chlorhydrate, and streptomycin were among the antimicrobials examined (Campero et al., 1993). Erickson et al. (2017) tested a combination of antimicrobial treatment with injectable oxytetracycline and vaccination with two doses of a commercial monovalent oil-based Cfv vaccine in a bull with BGC. None of the steers under study had BGC eradicated by two treatments with commercial monovalent bacterin and long-acting oxytetracycline at prescribed dosages. Consequently, this is not advised as a successful management tactic. However, due to recent reports of antibiotic resistance and laws in some nations, therapy should be administered in compliance with laws pertaining to the use of antibiotics in livestock production and on a veterinarian’s advice. VaccinationPossible prevention of recurrent estrus and abortion caused by Cfv is achieved with vaccination (Julie Gard, 2016). Commercial vaccinations for BGC prevention are administered intramuscularly or subcutaneously and contain bacterin and adjuvant (Balzan et al., 2020). Vaccination is effective in protecting against experimental vaginal infections, at least partially, when administered to females (Cobo et al., 2004). It is recommended that every year, approximately 30 days before the onset of breeding, females of reproductive age in herds infected with Cfv should be vaccinated (Bondurant, 2005). Heifers and cows should receive two doses of the initial immunization approximately 60 and 30 days before the start of the reproductive season. BGC outbreaks are sometimes difficult to control with vaccination alone, and bulls that test positive must be culled (Erickson et al., 2017). Bryner et al. (1988) found that the effectiveness of 10 commercial vaccinations in preventing abortion following exposure to C. fetus ranged from 0% to 89%. Bryner et al. (1988) also documented immune response deficiencies to certain commercial vaccines, indicating that the mass of bacteria used in a vaccine directly correlates with the percentage of protection provided and that the vaccine’s sample or adjuvant type’s immunogenicity may be involved. ControlControl strategies include testing and culling affected animals, vaccinating bulls, cows, and heifers, and using artificial intelligence with controlled semen (Givens, 2018). Since Cfv has been isolated from cows for over six months after calving, infected herds are advised to use AI exclusively for two breeding seasons (Haas et al., 2020). However, it is impractical to apply AI on cattle raised for food. Therefore, the main methods of control used by most producers are vaccination, management, culling of problematic animals, and adoption of bulls (European Food and Safety Authority et al., 2017). ConclusionThe fertility and reproductive performance of animals are seriously threatened by BGC, particularly in underdeveloped nations where natural breeding is prevalent. Developing efficient management measures to lessen the financial impact of the disease on livestock output requires an understanding of the epidemiology and transmission processes. AcknowledgmentsThe authors thank Universitas Airlangga and Badan Riset dan Inovasi Nasional. FundingThe authors would like to thank Universitas Airlangga for their managerial support and Salma Firdausya Qurrotunnada Noor, Eunice Wong Hui Wen, Joo Jia Yin, and Rahma Novhira for their technical support. This research was funded by the Directorate of Research and Community Service, Deputy for Strengthening Research and Technology, Ministry of Research and Technology/National Research and Innovation Agency for the 2022 fiscal year (Chancellor’s Decree number: 770/UN3.14/PT/2022). Author’s contributionsSM, ARK, RSA, and BWKW drafted the manuscript. M’AK, DAAK, AOA, and SW revised and edited the manuscript. WS, ATK, IBM, HN, and NF participated in the preparation and critical checking of the manuscript. IM, BNU, RD, AG, and SA edited the references. All authors have read and approved the final version of the manuscript. Conflict of interestThe authors declare no conflict of interest. Data availabilityAll references are open access, so data can be obtained from the internet. ReferencesAbdel-Glil, M.Y., Hotzel, H., Tomaso, H., Didelot, X., Brandt, C., Seyboldt, C., Linde, J., Schwarz, S., Neubauer, H. and El-Adawy, H. 2023. Genomic epidemiology of Campylobacter fetus subsp. venerealis from Germany. Front. Vet. Sci. 9, 1069062. Akhtar, S., Riemann, H.P., Thurmond, M.C. and Franti, C.E. 1993. The association between antibody titers against Campylobacter fetus and reproductive efficiency in dairy cattle. Vet. Res. Commun. 17(3), 183–191. Aydin, S., Emre, E., Ugur, K., Aydin, M.A., Sahin, I., Cinar, V. and Akbulut, T. 2025. An overview of ELISA: a review and update on best laboratory practices for quantifying peptides and proteins in biological fluids. J. Int. Med. Res. 53(2), 3000605251315913. Balzan, C., Ziech, R.E., Gressler, L.T. and Vargas, A.P.C.D. 2020. Bovine genital Campylobacteriosis: main features and perspectives for diagnosis and control. Cienc. Rural 50(3), e20190272. Bastos, L., Gomes, R., Pocinho, S., Baptista, T. and Mansinho, K. 2023. Campylobacter fetus Cellulitis. Cureus 15(2), e35328. Bénéjat, L., Ducournau, A., Gebhart, J., Bessede, E., Becker, J., Jauvain, M. and Lehours, P. 2025. Evaluation of a rapid fluorescence immunoassay for detecting Campylobacter antigens in stool samples. Gut Pathog. 17(1), 12. Beniawan, A., Indrawati, A. and Pasaribu, F.H. 2020. The development of transport media for prepuce sample collection, for bovine genital Campylobacteriosis detection. J. Sain Vet.38(2), 126–134. Bondurant, R.H. 2005. Venereal diseases of cattle: natural history, diagnosis, and the role of vaccines in their control. Vet. Clin. North Am. Food Anim. Pract. 21(2), 383–408. Bryner, J.H., Firehammer, B.D. and Wesley, I.V. 1988. Vaccination of pregnant guinea pigs with Campylobacter fetus: effects of antigen dose, Campylobacter strain, and adjuvant type. Am. J. Vet. Res. 49(4), 449–455. Cabell. 2007. Bovine abortion: aetiology and investigations. Pract 29(1), 455–463. Cagnoli, C.I., Chiapparrone, M.L., Cacciato, C.S., Rodríguez, M.G., Aller, J.F. and Catena, M.D.C. 2020. Effects of Campylobacter fetus on bull sperm quality. Microb. Pathog. 149(1), 104486. Cagnoli, C.I., Chiapparrone, M.L., Cacciato, C.S., Rodríguez, M.G., Catena, M.D.C. and Aller, J.F. 2024. Effect of Campylobacter fetus on in vitro fertilization and embryonic development of preimplantation bovine embryos. Vet. Microbiol. 288(1), 109925. Campero, C.M., Cipolla, A.L., Odriozola, E., Medina, D., Morsella, C.G. and Saubidet, M. 1993. Tratamientos sistémicos en toros con infección genital a Campylobacter fetus subsp. fetus. Vet. Arg. 10(1), 303–309. Campos-Múzquiz, L.G., Méndez-Olvera, E.T., Arellano-Reynoso, B. and Martínez-Gómez, D. 2019. Campylobacter fetus is Internalized by Bovine Endometrial Epithelial Cells. Pol. J. Microbiol. 68(2), 217–224. Campos-Múzquiz, L.G., Méndez-Olvera, E.T., Martínez, M.P. and Martínez-Gómez, D. 2021. Campylobacter fetus Induced Proinflammatory Response in Bovine Endometrial Epithelial Cells. Pol. J. Microbiol. 70(1), 99–106. Catena, M., Callejas, S., Soto, P., Aba, M., Echevarría, H., Monteavaro, C. and Mazzolli, A. 2003. Efectos de la infección experimental con Campylobacter fetus venerealis sobre la preñez temprana en vaquillonas. Invest. Vet. 5(1), 37–44. Chaban, B., Chu, S., Hendrick, S., Waldner, C. and Hill, J.E. 2012. Evaluation of a Campylobacter fetus subspecies venerealis real-time quantitative polymerase chain reaction for direct analysis of bovine preputial samples. Can. J. Vet. Res. 76(3), 166–173. Chen, Y., Chen, W., Wang, Y., Jiang, L. and Liu, J. 2021. What is the role of plasma cell in the lamina propria of terminal ileum in Good’s syndrome patient?. Open Med. (Wars). 16(1), 1681–1682. Chon, J.W., Seo, K.-H., Kim, B., Jeong, D. and Song, K.-Y. 2020. Advanced Methods for Isolating from and Confirming Campylobacter spp. in Milk and Dairy Products: review. J. Dairy Sci. Biotechnol. 38(3), 121–133. Cipolla, A., Cordeviola, J., Terzolo, H., Combessies, G., Bardón, J., Ramón, N., Martínez, A., Medina, D., Morsella, C. and Malena, R. 2001. Campylobacter fetus diagnosis: direct immunofluorescence comparing chicken IgY and rabbit IgG conjugates. ALTEX 18(3), 165–170. Cobo, E.R., Morsella, C., Cano, D., Cipolla, A. and Campero, C.M. 2004. Immunization in heifers with dual vaccines containing Tritrichomonas foetus and Campylobacter fetus antigens using systemic and mucosal routes. Theriogenology 62(8), 1367–1382. Cohen, E.J., Drobnič, T., Ribardo, D.A., Yoshioka, A., Umrekar, T., Guo, X., Fernandez, J.J., Brock, E.E., Wilson, L., Nakane, D., Hendrixson, D.R. and Beeby, M. 2024. Evolution of a large periplasmic disk in Campylobacterota flagella enables both efficient motility and autoagglutination. Dev. Cell 59(24), 3306–3321. Collado, L. and Figueras, M.J. 2011. Taxonomy, epidemiology, and clinical relevance of the genus Arcobacter. Clin. Microbiol. Rev. 24(1), 174–192. Corbeil, L.B., Campero, C.M., Rhyan, J.C. and BonDurant, R.H. 2003. Vaccines against sexually transmitted diseases. Reprod. Biol. Endocrinol. 1(1), 118. Córdova-Izquierdo, A., Reyes, A.E.I., Lang, C.G.R., Liera, J.E.G., Castro, J.F.I., Mancera, E.A.V., Mendoza, M.M., Crispín, R.H., Mosqueda M de., Vázquez, A.G., Pérez, J.O., Ordóñez, V.V. and Cervantes, R.E. 2016. Effect of mastitis and stress on cow reproduction. Int. J. Curr. Res. 8(4), 29584–29588. Dawson, L.J. 1986. Diagnosis, Prevention, and Control of Campylobacteriosis and Trichomoniasis. Bovine Pract. 21(1), 180–183. De Vargas, A.C., Costa, M.M., Vainstein, M.H., Kreutz, L.C. and Neves, J.P. 2002. Campylobacter fetus subspecies venerealis surface array protein from bovine isolates in Brazil. Curr. Microbiol. 45(2), 111–114. Delano, M.L., Mischler, S.A. and Underwood, W.J. 2002. Biology and Diseases of Ruminants: sheep, Goats, and Cattle. Lab. Anim. Med. 1(1), 519–614. Dworkin, J., Tummuru, M.K. and Blaser, M.J. 1995. A lipopolysaccharide-binding domain of the Campylobacter fetus S-layer protein resides within the conserved N terminus of a family of silent and divergent homologs. J. Bacteriol. 177(7), 1734–1741. El-Adawy, H., Hotzel, H., Tomaso, H. and Neubauer, H. 2023. The History of Bovine Genital Campylobacteriosis in the Face of Political Turmoil and Structural Change in Cattle Farming in Germany. Vet. Sci. 10(12), 665. Emele, M.F., Karg, M., Hotzel, H., Bloois, L.G., Groß, U., Bader, O. and Zautner, A.E. 2019. Differentiation of Campylobacter fetus Subspecies by Proteotyping. Eur. J. Microbiol. Immunol. (Bp). 9(2), 62–71. Erickson, N.E.N., Lanigan, E., Waugh, T., Gesy, K. and Waldner, C. 2017. Evaluation of long-acting oxytetracycline and a commercial monovalent vaccine for the control of Campylobacter fetus subsp. venerealis infection in beef bulls. Can. Vet. J. 58(10), 1051–1058. European Food and Safety Authority (EFSA) Panel on Animal Health and Welfare (AHAW), More, S., Bøtner, A., Butterworth, A., Calistri, P., Depner, K., Edwards, S., Garin-Bastuji, B., Good, M., Gortázar Schmidt, C., Michel, V., Miranda, M.A., Nielsen, S.S., Raj, M., Sihvonen, L., Spoolder, H., Stegeman, J.A., Thulke, H.H., Velarde, A., Willeberg, P., Winckler, C., Baldinelli, F., Broglia, A., Candiani, D., Beltrán-Beck, B., Kohnle, L. and Bicout, D. 2017. Assessment of listing and categorisation of animal diseases within the framework of the Animal Health Law (Regulation (EU) No 2016/429): bovine genital Campylobacteriosis. EFSA. J. 15(10), e04990. Florent, A. 1959. Les deux vibrioses génitales de la bête bovine : La vibriose vénérienne, due à Vibrio fœtus venerialis, et la vibriose d’origine intestinale due à V. fœtus intestinalis. Proceedings 10th International Veterinary Congress Madrid. 2(1), 953–957. García, J.A., Farace, P., Gioffré, A.K., Morsella, C., Méndez, M.A., Acuña, J., Aller, J.F., Signorini, M. and Paolicchi, F.A. 2023. Bovine Campylobacteriosis in bulls: insights in the conventional and molecular diagnosis. Braz. J. Microbiol. 54(1), 459–467. Givens, M.D. 2006. A clinical, evidence-based approach to infectious causes of infertility in beef cattle. Theriogenology 66(3), 648–654. Givens, M.D. 2018. Review: risks of disease transmission through semen in cattle. Animal 12(s1), s165–s171. Gribble, M.J., Salit, I.E., Isaac-Renton, J. and Chow, A.W. 1981. Campylobacter infections in pregnancy. Case report and literature review. Am. J. Obstet. Gynecol. 140(4), 423–426. Haas, D.J., Miranda-Guimarães, K.L., Dorneles, E.M.S. and Lage, A.P. 2020. Prevalence of Bovine Genital Campylobacteriosis in beef cattle in Brazil. EC. Vet. Sci. 5(12), 42–53. Hashim, H.O. and Al-Shuhaib, M.B. 2019. Exploring the Potential and Limitations of PCR-RFLP and PCR-SSCP for SNP Detection: a Review. J. Appl. Biotechnol. Rep. 6(4), 137–144. Hermadi, H.A., Khairullah, A.R., Damayanti, Y., Safitri, E., Tyasningsih, W., Warsito, S.H., Fauzia, K.A., Wardhani, B.W.K., Ekawasti, F., Wibowo, S., Fauziah, I., Moses, I.B., Kurniasih, D.A.A., Kusala, M.K.J. and Julaeha, J. 2024. Bovine trichomoniasis: a hidden threat to reproductive efficiency. Open Vet. J. 14(11), 2722–2730. Hermadi, H.A., Khairullah, A.R., Damayanti, Y., Safitri, E., Tyasningsih, W., Warsito, S.H., Kusala, M.K.J., Wibowo, S., Moses, I.B., Kurniawan, M.A., Wardhani, B.W.K., Prasetyo, S.T., Kurniasih, D.A.A., Rahajeng, A.D.R., Fauziah, I., Vidhianty, C.B., Ansori, A.N.M., Wasito, W. and Ahmad, R.Z. 2025. Endometritis in Cattle: a Review of Current Understanding and Practical Causes of Repeat Breeding. Trends. Sci. 22(6), 9587. Holt, J.G., Krieg, N.R., Sneath, P.H.A., Staley, J.T. and Williams, S.T. 1994. Bergey’s manual of determinative bacteriology in facultatively anaerobic gram-negative rods. Williams & Wilkins, Baltimore, MD. pp 175–190. Hoque, N., Islam, S.S., Saddam, M.J.I., Rafikuzzaman, M., Sikder, M.H., Castellan, D.M. and Kabir, S.M.L. 2023. Investigation of Campylobacter fetus in breeding bulls of private farms in Bangladesh. Vet. Med. Sci. 9(1), 417–428. Hugh, R. and Leifson, E. 1953. The taxonomic significance of fermentative versus oxidative metabolism of carbohydrates by various Gram-negative bacteria. J. Bacteriol. 66(1), 24–26. Hum, S. 2004. Vibriosis of cattle. Agfact A2 9(7), 1–2. Hum, S., Brunner, J., Mcinnes, A., Mendoza, G. and Stephens, J. 1994. Evaluation of cultural methods and selective media for the isolation of Campylobacter fetus subsp. venerealis from cattle. Aust. Vet. J. 71(6), 184–186. Inglis, G.D., Kalischuk, L.D. and Busz, H.W. 2004. Chronic shedding of Campylobacter species in beef cattle. J. Appl. Microbiol. 97(2), 410–420. Iraola, G., Forster, S.C., Kumar, N., Lehours, P., Bekal, S., García-Peña, F.J., Paolicchi, F., Morsella, C., Hotzel, H., Hsueh, P.R., Vidal, A., Lévesque, S., Yamazaki, W., Balzan, C., Vargas, A., Piccirillo, A., Chaban, B., Hill, J.E., Betancor, L., Collado, L., Truyers, I., Midwinter, A.C., Dagi, H.T., Mégraud, F., Calleros, L., Pérez, R., Naya, H. and Lawley, T.D. 2017. Distinct Campylobacter fetus lineages adapted as livestock pathogens and human pathobionts in the intestinal microbiota. Nat. Commun. 8(1), 1367. Ishtifaq, A., Qureshi, S., Farooq, S., Kashoo, Z.A., Malik, M.Z., Alam, M.R., Wani, S.A., Bhat, M.A., Hussain, M.I., Dar, R.A. and Shah, S.M. 2020. Genotyping and antibiotic resistance patterns of Campylobacter fetus subsp. venerealis from cattle farms in India. Lett. Appl. Microbiol. 71(6), 627–636. Juli, M.S.B., Raza, A., Forutan, M., Siddle, H.V., Fordyce, G., Muller, J., Boe-Hansen, G.B. and Tabor, A.E. 2024. Characterisation of reproductive tract microbiome and immune biomarkers for bovine genital campylobacteriosis in vaccinated and unvaccinated heifers. Front. Microbiol. 15(1), 1404525. Julie Gard, B. 2016. Bovine Genital Campylobacteriosis - A Review. Int. J. Vet. Sci. Res. 2(1), 29–31. Kaakoush, N.O., Miller, W.G., De Reuse, H. and Mendz, G.L. 2007. Oxygen requirement and tolerance of Campylobacter jejuni. Res. Microbiol. 158(8–9), 644–650. Khairullah, A., Yanestria, S., Effendi, M., Moses, I., Kusala, M., Fauzia, K., Ayuti, S., Fauziah, I., Silaen, O., Riwu, K., Aryaloka, S., Dameanti, F., Raissa, R., Hasib, A. and Furqoni, A. 2024. Campylobacteriosis: a rising threat in foodborne illnesses. Open Vet. J. 14(8), 1733–1750. Kim, J., Oh, E., Banting, G.S., Braithwaite, S., Chui, L., Ashbolt, N.J., Neumann, N.F. and Jeon, B. 2016. An Improved Culture Method for Selective Isolation of Campylobacter jejuni from Wastewater. Front. Microbiol. 7(1), 1345. Louwen, R., Van Baarlen, P., Van Vliet, A.H., Van Belkum, A., Hays, J.P. and Endtz, H.P. 2012. Campylobacter bacteremia: a rare and under-reported event?. Eur. J. Microbiol. Immunol. 2(1), 76–87. Madoroba, E., Gelaw, A., Hlokwe, T. and Mnisi, M. 2011. Prevalence of Campylobacter foetus and Trichomonas foetus among cattle from Southern Africa. Afr. J. Biotechnol. 10(50), 10311–10314. Mai, H.M., Irons, P.C. and Thompson, P.N. 2015. Brucellosis, genital campylobacteriosis and other factors affecting calving rate of cattle in three states of Northern Nigeria. BMC Vet. Res. 11(1), 7. Mai, H.M., Irons, P.C., Kabir, J. and Thompson, P.N. 2013. Prevalence of bovine genital campylobacteriosis and trichomonosis of bulls in northern Nigeria. Acta Vet. Scand. 55(1), 56. McFadden, A., Heuer, C., Jackson, R., West, D. and Parkinson, T. 2005. Investigation of bovine venereal campylobacteriosis in beef cow herds in New Zealand. N. Z. Vet. J. 53(1), 45–52. Mcgready, R., Wuthiekanun, V., Ashley, E.A., Tan, S.O., Pimanpanarak, M., Viladpai-Nguen, S.J., Jesadapanpong, W., Blacksell, S.D., Proux, S., Day, N.P., Singhasivanon, P., White, N.J., Nosten, F. and Peacock, S.J. 2010. Diagnostic and treatment difficulties of pyelonephritis in pregnancy in resource-limited settings. Am. J. Trop. Med. Hyg. 83(6), 1322–1329. Mcmillen, L., Fordyce, G., Doogan, V.J. and Lew, A.E. 2006. Comparison of culture and a novel 5 Taq nuclease assay for direct detection of Campylobacter fetus subsp. venerealis in clinical specimens from cattle. J. Clin. Microbiol. 44(3), 938–945. Michi, A.N., Favetto, P.H., Kastelic, J. and Cobo, E.R. 2016. A review of sexually transmitted bovine trichomoniasis and Campylobacteriosis affecting cattle reproductive health. Theriogenology 85(5), 781–791. Miller, J.M., Binnicker, M.J., Campbell, S., Carroll, K.C., Chapin, K.C., Gilligan, P.H., Gonzalez, M.D., Jerris, R.C., Kehl, S.C., Patel, R., Pritt, B.S., Richter, S.S., Robinson-Dunn, B., Schwartzman, J.D., Snyder, J.W., Telford, S., Theel, E.S., Thomson, R.B., Weinstein, M.P. and Yao, J.D. 2018. A Guide to Utilization of the Microbiology Laboratory for Diagnosis of Infectious Diseases: 2018 Update by the Infectious Diseases Society of America and the American Society for Microbiology. Clin. Infect. Dis. 67(6), e1–e94. Modi, S., Brahmbhatt, M.N., Chatur, Y.A. and Nayak, J.B. 2015. Prevalence of Campylobacter species in milk and milk products, their virulence gene profile and anti-bio gram. Vet. World 8(1), 1–8. Monke, H.J., Love, B.C., Wittum, T.E., Monke, D.R. and Byrum, B.A. 2002. Effect of transport enrichment medium, transport time, and growth medium on the detection of Campylobacter fetus subsp. venerealis. J. Vet. Diagn. Invest. 14(1), 35–39. Monroy, O.L., Aguilar, C., Lizano, M., Cruz-Talonia, F., Cruz, R.M. and Rocha-Zavaleta, L. 2010. Prevalence of human papillomavirus genotypes, and mucosal IgA anti-viral responses in women with cervical ectopy. J. Clin. Virol. 47(1), 43–48. Morrell, J. 2006. Update on semen technologies for animal breeding. Reprod. Domest. Anim. 41(1), 63–67. Mshelia, G., Amin, J., Woldehiwet, Z., Murray, R. and Egwu, G. 2010. Epidemiology of bovine venereal campylobacteriosis: geographic distribution and recent advances in molecular diagnostic techniques. Reprod. Domest. Anim. 45(5), e221–e230. Neyaz, L.A., Arafa, S.H., Alsulami, F.S., Ashi, H., Elbanna, K. and Abulreesh, H.H. 2024. Culture-Based Standard Methods for the Isolation of Campylobacter spp. in Food and Water. Pol. J. Microbiol. 73(4), 433–454. Owusu-Ansa, I., Ramadas, M., Jacob, N. and Ayeni, F.E. 2025. Campylobacter fetus subsp. fetus: an unforeseen cause of abortion in regional Australia. Access Microbiol. 7(2), 889. Oyarzabal, O.A., Wesley, I.V., Harmon, K.M., Schroeder-Tucker, L., Barbaree, J.M., Lauerman, L.H., Backert, S. and Conner, D.E. 1997. Specific identification of Campylobacter fetus by PCR targeting variable regions of the 16S rDNA. Vet. Microbiol. 58(1), 61–71. Oyhenart, J. 2024. Positive cases of bovine genital campylobacteriosis and bovine trichomonosis in the eradication program of La Pampa, Argentina: 2008–2021. Trop. Anim. Health Prod. 56(1), 96. Paisley, J.W., Mirrett, S., Lauer, B.A., Roe, M. and Reller, L.B. 1982. Dark-field microscopy of human feces for presumptive diagnosis of Campylobacter fetus subsp. jejuni enteritis. J. Clin. Microbiol. 15(1), 61–63. Pellegrin, A.O., Lage, A.P., Sereno, J.R.B., Ravaglia, E., Costa, M.S. and Leite, R.C. 2002. Bovine Genital Campylobacteriosis in Pantanal, State of Mato Grosso do Sul, Brazil. Revue Élev. Méd. Vét. Pays Trop. 55(3), 169–173. Pena-Fernández, N., Cano-Terriza, D., García-Bocanegra, I., Horcajo, P., Vázquez-Arbaizar, P., Cleofé-Resta, D., Pérez-Arroyo, B., Ortega-Mora, L.M. and Collantes-Fernández, E. 2021. Prevalence of Bovine Genital Campylobacteriosis, Associated Risk Factors and Spatial Distribution in Spanish Beef Cattle Based on Veterinary Laboratory Database Records. Front. Vet. Sci. 8(1), 750183. Pena-Fernández, N., Kortabarria, N., Hurtado, A., Ocejo, M., Fort, M., Pérez-Cobo, I., Collantes-Fernández, E. and Aduriz, G. 2024. Biochemical and molecular characterization of Campylobacter fetus isolates from bulls subjected to bovine genital campylobacteriosis diagnosis in Spain. BMC Vet. Res. 20(1), 131. Raúl, S.B., Faustino, L.G., Jorge, A.-P., José, V.V., Luis, L.Z. and Hugo, B, G. 2023. Frequency of Campylobacter fetus in bulls in the central zone of Tamaulipas Mexico. Abanico Vet. 13(1), 1–10. Ricke, S.C., Feye, K.M., Chaney, W.E., Shi, Z., Pavlidis, H. and Yang, Y. 2019. Developments in Rapid Detection Methods for the Detection of Foodborne Campylobacter in the United States. Front. Microbiol. 9(1), 3280. Sahin, O., Yaeger, M., Wu, Z. and Zhang, Q. 2017. Campylobacter-associated diseases in Animals. Annu. Rev. Anim. Biosci. 5(1), 21–42. Schulze, F., Bagon, A., Müller, W. and Hotzel, H. 2006. Identification of Campylobacter fetus subspecies by phenotypic differentiation and PCR. J. Clin. Microbiol. 44(6), 2019–2024. Sebald, M. and Véron, M. 1963. Teneur en bases de l’adn et classification des vibrions [base dna content and classification of Vibrios]. Ann. Inst. Pasteur. (Paris) 105(1), 897–910. Seid, A. 2019. Bovine Campylobacteriosis and its economic importance: a review. J. Dairy Vet. Sci. 11(2), JDVS.MS.ID.555807. Seong, Y.J., Lee, S.H., Kim, E.J., Choi, Y.H., Kim, T.J., Lee, W.G. and Heo, J.Y. 2021. Campylobacter fetus subspecies venerealis meningitis associated with a companion dog in a young adult: a case report. BMC Infect. Dis. 21(1), 1280. Shange, N., Gouws, P. and Hoffman, L.C. 2019. Campylobacter and Arcobacter species in food-producing animals: prevalence at primary production and during slaughter. World J. Microbiol. Biotechnol. 35(9), 146. Sheldon, I.M. and Owens, S.E. 2017. Postpartum uterine infection and endometritis in dairy cattle. Anim. Reprod. 14(3), 622–629. Shen, L., Wang, W., Hou, W., Jiang, C., Yuan, Y., Hu, L. and Shang, A. 2023. The function and mechanism of action of uterine microecology in pregnancy immunity and its complications. Front. Cell. Infect. Microbiol. 12(1), 1025714. Silveira, C.D.S., Fraga, M., Giannitti, F., Macías-Rioseco, M. and Riet-Correa, F. 2018. Diagnosis of bovine genital Campylobacteriosis in South America. Front. Vet. Sci. 5(1), 321. Smith, T., Little, R.B. and Taylor, M.S. 1920. Further studies on the etiological role of Vibrio fetus. J. Exp. Med. 32(6), 683–689. Sprenger, H., Zechner, E.L. and Gorkiewicz, G. 2012. So close and yet so far - Molecular Microbiology of Campylobacter fetus subspecies. Eur. J. Microbiol. Immunol. (Bp). 2(1), 66–75. Terzolo, M., Osella, G., Alì, A., Borretta, G., Magro, G.P., Termine, A., Paccotti, P. and Angeli, A. 1996. Different patterns of steroid secretion in patients with adrenal incidentaloma. J. Clin. Endocrinol. Metab. 81(2), 740–744. Thompson, S.A. 2002. Campylobacter surface-layers (S-layers) and immune evasion. Ann. Periodontol. 7(1), 43–53. Tremblay, C., Gaudreau, C. and Lorange, M. 2003. Epidemiology and antimicrobial susceptibilities of 111 Campylobacter fetus subsp. fetus strains isolated in Québec, Canada, from 1983 to 2000. J. Clin. Microbiol. 41(1), 463–466. Truyers, I., Luke, T., Wilson, D. and Sargison, N. 2014. Diagnosis and management of venereal campylobacteriosis in beef cattle. BMC Vet. Res. 10(1), 280. Underwood, W.J., Blauwiekel, R., Delano, M.L., Gillesby, R., Mischler, S.A. and Schoell, A. 2015. Biology and Diseases of Ruminants (Sheep, Goats, and Cattle). Lab. Anim. Med. 1(1), 623–694. Van Alst, A.J., Demey, L.M. and Dirita, V.J. 2022. Vibrio cholerae requires oxidative respiration through the bd-I and cbb3 oxidases for intestinal proliferation. PLos Pathog. 18(5), e1010102. Van Bergen, M.A.P., Dingle, K.E., Maiden, M.C.J., Newell, D.G., Van Der Graaf-van Bloois, L., Van Putten, J.P.M. and Wagenaar, J.A. 2005. Clonal nature of Campylobacter fetus as defined by multilocus sequence typing. J. Clin. Microbiol. 43(12), 5888–5898. Vandeplassche, M., Coryn, M., Spincemaille, J., Bouters, R. and Bonte, P. 1976. Die prophylaxe von abortus und frühgeburt beim rind und pferd. Dtsch. Tierarztl. Wochenschr. 83(12), 554–556. Vinzent, R. 1949. Une affection méconnue de la grossesse. L’infection placentaire à Vibrio foetus. Presse Méd. 81(1), 1230–1232. Véron, M. and Chatelain, R. 1973. Taxonomic study of the genus Campylobacter Sebald and Vu00e9ron and designation of the neotype strain for the type species, Campylobacter fetus (Smith and Taylor) Sebald and Vu00e9ron. Int. J. Syst. Evol. Microbiol. 23(2), 122–134. Wagenaar, J.A., Van Bergen, M.A.P., Blaser, M.J., Tauxe, R.V., Newell, D.G. and Van Putten, J.P.M. 2014. Campylobacter fetus infections in humans: exposure and disease. Clin. Infect. Dis. 58(11), 1579–1586. Wassenaar, T.M. 1997. Toxin production by Campylobacter spp. Clin. Microbiol. Rev. 10(3), 466–476. WOAH. 2023. Bovine genital campylobacteriosis. In Manual of diagnostic tests and vaccines for terrestrial animals. Paris, France: World Organization for Animal Health (WOAH), p: 1–4. Yamazaki, W., Taguchi, M., Ishibashi, M., Nukina, M., Misawa, N. and Inoue, K. 2009. Development of a loop-mediated isothermal amplification assay for sensitive and rapid detection of Campylobacter fetus. Vet. Microbiol. 136(3–4), 393–396. Yousaf Kazmi, S. 2022. The etymology of microbial nomenclature and the diseases these cause in a historical perspective. Saudi J. Biol. Sci. 29(11), 103454. | ||
| How to Cite this Article |
| Pubmed Style Mulyati S, Khairullah AR, Adji RS, Moses IB, Fairusya N, Mustofa I, Utomo BN, Dewiyanti R, Suwito W, Nuradji H, Artanto S, Wardhani BWK, Kurniasih DAA, Khalisa AT, Kurniawan M�, Akintunde AO, Wibowo S, Ganisia A. Understanding bovine genital campylobacteriosis: A comprehensive review of its threat to global cattle reproduction. Open Vet. J.. 2025; 15(10): 4834-4846. doi:10.5455/OVJ.2025.v15.i10.4 Web Style Mulyati S, Khairullah AR, Adji RS, Moses IB, Fairusya N, Mustofa I, Utomo BN, Dewiyanti R, Suwito W, Nuradji H, Artanto S, Wardhani BWK, Kurniasih DAA, Khalisa AT, Kurniawan M�, Akintunde AO, Wibowo S, Ganisia A. Understanding bovine genital campylobacteriosis: A comprehensive review of its threat to global cattle reproduction. https://www.openveterinaryjournal.com/?mno=261959 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.4 AMA (American Medical Association) Style Mulyati S, Khairullah AR, Adji RS, Moses IB, Fairusya N, Mustofa I, Utomo BN, Dewiyanti R, Suwito W, Nuradji H, Artanto S, Wardhani BWK, Kurniasih DAA, Khalisa AT, Kurniawan M�, Akintunde AO, Wibowo S, Ganisia A. Understanding bovine genital campylobacteriosis: A comprehensive review of its threat to global cattle reproduction. Open Vet. J.. 2025; 15(10): 4834-4846. doi:10.5455/OVJ.2025.v15.i10.4 Vancouver/ICMJE Style Mulyati S, Khairullah AR, Adji RS, Moses IB, Fairusya N, Mustofa I, Utomo BN, Dewiyanti R, Suwito W, Nuradji H, Artanto S, Wardhani BWK, Kurniasih DAA, Khalisa AT, Kurniawan M�, Akintunde AO, Wibowo S, Ganisia A. Understanding bovine genital campylobacteriosis: A comprehensive review of its threat to global cattle reproduction. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4834-4846. doi:10.5455/OVJ.2025.v15.i10.4 Harvard Style Mulyati, S., Khairullah, . A. R., Adji, . R. S., Moses, . I. B., Fairusya, . N., Mustofa, . I., Utomo, . B. N., Dewiyanti, . R., Suwito, . W., Nuradji, . H., Artanto, . S., Wardhani, . B. W. K., Kurniasih, . D. A. A., Khalisa, . A. T., Kurniawan, . M. �., Akintunde, . A. O., Wibowo, . S. & Ganisia, . A. (2025) Understanding bovine genital campylobacteriosis: A comprehensive review of its threat to global cattle reproduction. Open Vet. J., 15 (10), 4834-4846. doi:10.5455/OVJ.2025.v15.i10.4 Turabian Style Mulyati, Sri, Aswin Rafif Khairullah, Rahmat Setya Adji, Ikechukwu Benjamin Moses, Nuha Fairusya, Imam Mustofa, Bambang Ngaji Utomo, Rina Dewiyanti, Widodo Suwito, Harimurti Nuradji, Sidna Artanto, Bantari Wisynu Kusuma Wardhani, Dea Anita Ariani Kurniasih, Andi Thafida Khalisa, Muhammad ‘ahdi Kurniawan, Adeyinka Oye Akintunde, Syahputra Wibowo, and Ainun Ganisia. 2025. Understanding bovine genital campylobacteriosis: A comprehensive review of its threat to global cattle reproduction. Open Veterinary Journal, 15 (10), 4834-4846. doi:10.5455/OVJ.2025.v15.i10.4 Chicago Style Mulyati, Sri, Aswin Rafif Khairullah, Rahmat Setya Adji, Ikechukwu Benjamin Moses, Nuha Fairusya, Imam Mustofa, Bambang Ngaji Utomo, Rina Dewiyanti, Widodo Suwito, Harimurti Nuradji, Sidna Artanto, Bantari Wisynu Kusuma Wardhani, Dea Anita Ariani Kurniasih, Andi Thafida Khalisa, Muhammad ‘ahdi Kurniawan, Adeyinka Oye Akintunde, Syahputra Wibowo, and Ainun Ganisia. "Understanding bovine genital campylobacteriosis: A comprehensive review of its threat to global cattle reproduction." Open Veterinary Journal 15 (2025), 4834-4846. doi:10.5455/OVJ.2025.v15.i10.4 MLA (The Modern Language Association) Style Mulyati, Sri, Aswin Rafif Khairullah, Rahmat Setya Adji, Ikechukwu Benjamin Moses, Nuha Fairusya, Imam Mustofa, Bambang Ngaji Utomo, Rina Dewiyanti, Widodo Suwito, Harimurti Nuradji, Sidna Artanto, Bantari Wisynu Kusuma Wardhani, Dea Anita Ariani Kurniasih, Andi Thafida Khalisa, Muhammad ‘ahdi Kurniawan, Adeyinka Oye Akintunde, Syahputra Wibowo, and Ainun Ganisia. "Understanding bovine genital campylobacteriosis: A comprehensive review of its threat to global cattle reproduction." Open Veterinary Journal 15.10 (2025), 4834-4846. Print. doi:10.5455/OVJ.2025.v15.i10.4 APA (American Psychological Association) Style Mulyati, S., Khairullah, . A. R., Adji, . R. S., Moses, . I. B., Fairusya, . N., Mustofa, . I., Utomo, . B. N., Dewiyanti, . R., Suwito, . W., Nuradji, . H., Artanto, . S., Wardhani, . B. W. K., Kurniasih, . D. A. A., Khalisa, . A. T., Kurniawan, . M. �., Akintunde, . A. O., Wibowo, . S. & Ganisia, . A. (2025) Understanding bovine genital campylobacteriosis: A comprehensive review of its threat to global cattle reproduction. Open Veterinary Journal, 15 (10), 4834-4846. doi:10.5455/OVJ.2025.v15.i10.4 |