| Research Article | ||
Open Vet. J.. 2025; 15(10): 4984-4996 Open Veterinary Journal, (2025), Vol. 15(10): 4984-4996 Research Article Evaluation of the antifungal activity of coconut oil/chitosan nanoparticles against Microsporum canisIsraa Ibrahim Khalil1*, Hawraa F.H. Al-Abedi2, Alyaa Sabti Jasim3 and Junaid Ghanim Younus21Department of Microbiology, College of Veterinary Medicine, University of Mosul, Mosul, Iraq 2Department of Biology, College of Education for Pure Sciences, University of Al-Hamdaniya, Al-Hamdaniya, Nineveh, Iraq 3Department of Microbiology, College of Veterinary Medicine, University of Basrah, Basrah, Iraq *Corresponding Author: Israa Ibrahim Khalil. Department of Microbiology, College of Veterinary Medicine, University of Mosul, Mosul, Iraq. Email: israibrahim [at] uomosul.edu.iq Submitted: 12/06/2025 Revised: 09/09/2025 Accepted: 19/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: Many dermatophyte infections have become health problems in humans and animals and require treatment with chemical antifungals whose adverse effects cannot be ignored and are difficult to obtain in many countries. Therefore, research has begun to find biological alternatives and load them onto fine nanoparticles that have proven effective in many fields. Aim: This study investigated the antifungal efficacy of chitosan nanoparticle-coated coconut oil (CO/CsNPs) against Microsporum canis, which was collected from cattle suspected to be infected with ringworm. Methods: Microsporum canis was isolated using Sabouraud dextrose agar (SDA), and colony coloration (white/yellow front, deep yellow/orange reverse). Spindle-shaped macroconidia and septate hyphae were identified microscopically. Distinct phytochemical compound analysis using GC-MS revealed that COEx is predominantly phenolic compounds (96.63%), with minor alkaloids and resins. After biosynthesizing the CO/CsNPs with the sol-gel technique, chitosan and sodium tri-poly-phosphate (STPP), the characterization of CO/CsNPs with UV-visible absorption spectroscopy confirmed successful nanoencapsulation with peaks indicating phenolic, terpenoid, and flavonoid interactions. The field emission scanning electron microscopy (SEM) analysis showed that nanoparticles averaged 39.7–50.06 nm in size, with rough surfaces enhancing biological interactions and sharp edges potentially disrupting microbial membranes. Energy dispersive X-ray explained the elemental composition (C, O, and N) and reduced crystallinity in nanocomposites, suggesting improved polymer flexibility. Results: The antifungal activity of CO/CsNPs exhibited concentration-dependent, with 527.40 µg/ml achieving 100% fungicidal activity against M. canis. Lower concentrations (263.7 and 174.04 µg/ml) exhibited 99.99% inhibition. The study highlights CO/CsNPs as a potent antifungal agent, urging further research into clinical trials, genetic engineering for enhanced properties, sustainable production, and veterinary applications. Conclusion: This work underscores the promise of nanotechnology-integrated coconut oil in combating fungal infections in animals while emphasizing the need for expanded safety and efficacy studies. Keywords: Antifungal, Coconut oil/chitosan nanoparticles, Microsporum canis, Ringworm, Nanotechnology. IntroductionMicrosporum canis is one of the most prevalent fungi that infects various animal species, including cattle, buffalo, sheep, goats, puppies, and cats. M. canis is a dermatophyte, causing ringworm (Alhasan et al., 2022). Microsporum canis is a zoophilic dermatophyte that usually has symptoms such as alopecia (hair loss) around the head, especially near the nose, eyes, and ears. Infected sites flash yellow-green under UV light (Agarwal et al., 2018). Broken hairs bearing M. canis spores spread the disease to people by contact with affected animals or contaminated things (e.g., grooming tools). Due to the difficulty of dealing with chemical treatments, the increasing resistance, and the side effects caused by the antifungals used, leading to the search for therapeutic alternatives (Kumar Nigam, 2015; Keshwania et al., 2023). Nanotechnology, which emerged in the 1970s, involves manipulating materials at the nanoscale (1–100 nm) to create innovative solutions (Al-Abedi et al., 2023). It has transformative applications in medicine, particularly for drug delivery, antimicrobial therapies, and wound treatment (Mohammadi et al., 2015). Key nanoparticle systems include liposomes, metallic nanoparticles, carbon nanotubes, and chitosan-based structures. Nanoparticles enhance drug solubility, bioavailability, and targeted delivery while minimizing adverse effects (Youssef et al., 2019). In antimicrobial therapy, nanoparticles, such as florfenicol, neomycin, silver, and chitosan, are engineered to combat microbial resistance (Kolo et al., 2018). Chitosan, a biocompatible biopolymer derived from chitin, stands out for its antifungal/antimicrobial properties and versatility. For instance, coconut oil-loaded chitosan nanoparticles (synthesized via ionic gelation) combine natural antifungal agents with nanotechnology for enhanced efficacy in treating skin infections (Boulaiz et al., 2011). Biogenic synthesis using bacteria, fungi, or plants offers sustainable, cost-effective methods to produce nanoparticles. These biologically capped NPs exhibit improved stability and biocompatibility compared with their chemically synthesized counterparts (Guigler-Casagrande and Lima, 2019). In veterinary medicine, nanotechnology improves drug delivery precision, wound healing, and disease management. Additionally, chitosan’s adaptability allows its use in bone tissue engineering, where it forms 3D scaffolds or composites with ceramics/polymers to support regenerative therapies (Gholami-Shabani et al., 2014). The absence of any harmful side effects caused by chemical treatments for coconut oil makes it the ideal choice for treating fungal skin infections, as it has been used in the field of cosmetics for a long time. This has attracted the attention of researchers, especially in the field of alternative treatments loaded with nanoparticles. Overall, nanotechnology bridges natural and synthetic approaches, offering scalable solutions to global health challenges such as AMR and chronic infections. It is critical to optimize these systems for clinical and veterinary use. This study aimed to determine phytochemical content and biosynthesis of chitosan nano-encapsulated CO/CsNPs and the antifungal activity of coconut oil against M. canis. Materials and MethodsThis study included five experiments, four of which were conducted in the laboratories of the Scientific Research Commission/ Research and Technology Center of Environment, Water and Renewable Energy Department, Baghdad, Iraq. The isolation of M. canis and in vitro evaluation of the antifungal efficacy of coconut oil/chitosan nanoparticles were performed in the Microbiology Laboratories of the College of Veterinary Medicine, University of Mosul, Mosul, Iraq. Fungal species used in the studyThe tested M. canis strain was isolated from cattle with skin desquamation and hair lesions, which were suspected to be ringworm. Identification was confirmed based on colony morphology (SDA) and microscopic characteristics of macro and microconidia using lactophenol cotton blue staining (Alhasan et al., 2022; Al-Abedi et al., 2023). Gas chromatograph mass analysis for phytochemical determinationThe text describes the methodology for analyzing the chemical components of coconut oil extract using gas chromatography-mass spectrometry (GC-MS) according to the method described by Brito et al., (2017) including the following steps: Preparation 0.2 ml of the coconut oil sample was dissolved in 10 ml of pure methanol at 60°C and, filtered through a 0.45 µm filter. GC-MS analysis via an Agilent 7820A GC-MS system. Components were identified by comparing chromatograms and retention times with the Wiley W9N11 mass spectra library (Amelia et al., 2017). Biosynthesis of coconut oil extract/chitosan nanoparticles (CO/CsNPs)Synthesis of chitosan-Cs-adductThe text outlines the synthesis and characterization of chitosan-based NPs using coconut oil and ionic gelation with tripolyphosphate (Guilger-Casagrande et al.,2019). 1. Chitosan adduct preparationChitosan and coconut oil (equimolar ratio) undergo a condensation reaction using a Dean-Stark apparatus with xylene to remove water. The product was filtered, washed with (methanol, hot water, or ethanol), and dried at 50°C (Agrawal et al., 2018). 2. Nanoparticle formation (ionic gelation)Cs-TPP NPs: Chitosan was dissolved in 1% acetic acid, mixed with TPP (1:2.5 w/w ratio), and stirred for 6 hours. A similar process is used with a chitosan-coconut oil adduct (25 mg/ml). The NPs are separated, washed, and dried (Brito et al., 2017). Characterization techniquesThis protocol ensures the systematic synthesis and multifaceted characterization of CCO nanoparticles for structural, morphological, and functional analysis (Ch et al., 2010; Pinlap et al., 2023).
Where D is the crystal size, λ is the wavelength of x-ray, θ is the diffraction angle (Braggs angle) in radians, and β is the full width at half maximum (FWHM) of the peak in radians (Sarvamangala et al., 2013).
This experiment was conducted for antifungal activity detection of coconut oil extract-COEx and CO/CsNPs against M. canis isolates (in vitro). The minimum fungal inhibition concentration (MIC) and minimal fungicidal concentration (MFC) depend on the spreading plate method (Wasien et al., 2019; Alnuaimi et al., 2023). Preparation of the Microsporum canis suspensionThe sensitivity of M. canis to coconut oil extract (COEx) and chitosan-coconut oil nanoparticles (CO/CsNPs) was assessed using the CLSI M51-S guidelines (Castro Méndez et al., 2008). Key steps included:
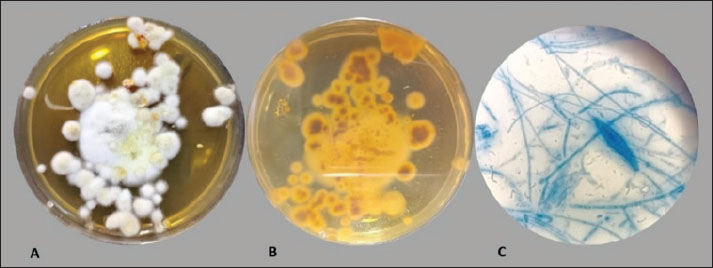
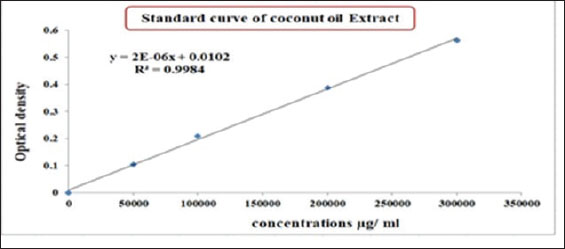
Determination of the CO/CsNP concentrationCoconut oil extract/chitosan nanoparticle concentration was determined by drawing the standard curve of CO extract using a UV-Vis spectrophotometer (Shimadzu, Japan) at an optical density of 284 nm of (50,000, 10,000, 20,000, and 3,00,000) µg/ml at 284 nm. The COEx standard curve equation was applied to determine the CO/Cs NPs concentration after dilution of the CO/Cs NPs 100 times. Statistical analysisStatistical analysis software was used to determine how the variables affected the research parameters. The least significant difference (LSD) test, analysis of variance (ANOVA), and t test were used to compare means. This study compared 0.05 and 0.01 probability percentages using the chi-square test (Cary, 2018). Ethical approvalThe Animal Welfare Committee of Mosul University-College of Veterinary Medicine, reference number UM.VET. UM.VET.2024.123, directed the ethical euthanasia of the samples after owner agreement. ResultsIdentification of M. canisThe morphological characterization of colonies after culturing of hair and skin samples on SDA and incubation at 25oC for at least 5 days and microscopically by noticing micro and macroconidia after staining with Lactophenol cotton blue (Alhasan et al., 2022; Al-Abedi et al., 2023) has been verified. Macroscopically, colonies of M. canis appear as flat to slightly groove with a fuzzy to cottony feel, the color ranges from white to yellowish on the front and from deep yellow to orange on the reverse. Microscopically, staining with Lactophenol cotton blue showed septate hyphae, long spindle-shaped macroconidia with thick walls, and stiff macroconidia with strong exterior cell walls (Fig. 1).
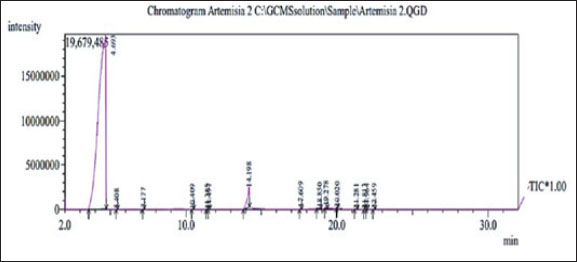
Fig. 1. Macroscopic appearance (A) Top view, (B) Reverse view of Microsporum canis colonies on SD agar, (C) Microscopic observation: M. canis macroconidia stained with lactophenol cotton blue under (100 x) Lens. Determination of active compounds in coconut oil extraction by GC-MSTable 1 shows the gas chromatography-mass spectrometry-determined COEx chemical compositions, retention time, area percentage, and compound structures. Figure 2 shows a typical GC-MS histogram with five notable peaks showing 96.63 phenolic chemicals (2.7, 0.16, and 0.11 alkaloids), and 0.17 resins. Three retention periods for alkaloids were found in GC-MS, and 96.63% of CO Ex is terpenes. The retention duration of the three techniques was 14.198, 18.850, and 17.609 minutes (Hasab Allh and Alhasan, 2023).
Fig. 2. Typical GC histogram of the coconut extract. Table 1. Chemical composition of the coconut oil extract.
Biosynthesis of chitosan nano-encapsulated coconut oil extractCoconut oil has demonstrated promising results in the production of CNPs. Because of changes in its molecular structure during manufacture, CS is no longer soluble in acid solutions and can instead exist in liquid forms (Franklin et al., 2024) (Fig. 3).
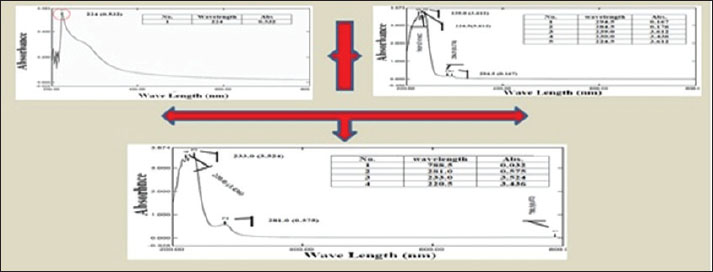
Fig. 3. Biosynthesis process of CO/CS nanoencapsulation. Characterization of coconut oil extract/chitosan nanoparticlesA UV-visible spectrophotometerUse UV-Vis spectroscopy at wavelengths after dilution with deionized water to optimize absorption. UV-Vis study of the absorption spectra of the CO/chitosan nano-encapsulated nanoparticles showed broad resonances, indicating effective nanoparticle encapsulation in solution (Fig. 4). The absorption spectra for CO Ex show peaks at 294, 284.5, 239.0, 230.0, and 224.5 nm with absorbance values of 0.167, 0.176, 3.612, 3.436, and 3.612, respectively. In addition, chitosan-Cs has a 0.532 absorbance peak 0.532 at 224 nm. The CO/CsNPs exhibited peaks at 778.5, 281.0, 233.0, and 220.5 nm and absorbance values of 0.032, 0.575, 3.524, and 3.436.
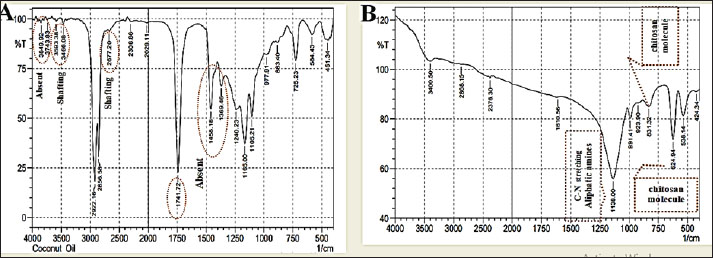
Fig. 4. UV-Vis Spectroscopy of A- Coconut oil Extract –COEx, B- CO/CsNPs. B-FTIR spectra of CO Ex and CO/CsNPsThe nanoparticle wavenumber functional types are shown in Fig. 5. The lack of wavenumbers between the CO Ex and CO/CsNPs spectra and the amide I peak (1,610.56) cm−1 is significant, showing CO Ex and nano-encapsulated CO/CsNPs. Alcohol and phenol (2,029.11–2,677.2) cm−1 are detected in CO Ex spectra (Fig. 5A). These findings match the predicted chemical makeup of CO Ex and CS molecules. The spectra of the biosynthesized nano-encapsulated particles are close to the CO Ex spectrum, showing that the biomolecules of the extract have been linked to the nanoparticles (Fig. 5B). Biomolecules, such as water, phenol, terpenoids, and flavonoids, may be implicated. O-H groups from the synthesis process may also explain the reported peaks. On the other hand, the absence of some peaks and shifts may be due to the interaction between coconut oil extract and chitosan groups in biosynthesis of nano-encapsulated nanoparticles.
Fig. 5. FTIR spectroscopy of A- significant shows CO Ex and nano-encapsulated CO/CsNPs. Alcohol and phenol (2029.11–2677.2) cm-1 are detected in the CO Ex spectra. B- The spectra of the biosynthesized nano-encapsulated particles are close to the CO Ex spectrum, showing that the biomolecules of the extract have been linked to the nanoparticles. C-Fe-SEM, EDX, and AFM analysis of CO/CsNPsNano-encapsulated Fe-SEM and AFM analysis of CO/CsNPs: the shot displays shape variations; however, the nanoparticles were 29.34–50.06 nm, smooth, rough, and protruding granules. This supports high stability. Rough surfaces may allow biological processes, such as active plant groups, to engage more with their targets than smoother surfaces with smaller contact areas (Fig. 6). The image shows an average particle size of 39.7 nm. The size distribution matches the AFM results. Handling samples before inspection may cause nanoparticle clumping. The H-C=O and C-H stretch revealed the structure of chitosan, confirming that the particles are stable and efficient in cells (Shaghati et al., 2023). They used AFM to study CsNPs-loaded clove extract. Their 3D image showed a homogeneous population of particles with uniform surfaces, which confirmed our findings. AFM imaging showed intelligent interaction of clove-loaded chitosan nanoparticles, forming distinct aggregates.
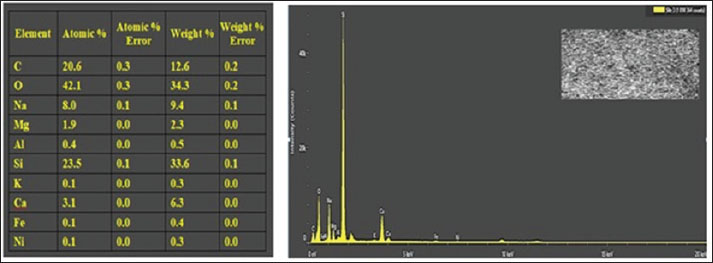
Fig. 6. A- AFM results and B- Fe-SEM image of CO/CsNPs D-energy dispersive X-ray (EDX) microanalysis of CO/CsNPsThe elemental analysis of CO/CsNPs by EDX shows C, O, and N. The EDX investigation confirms chitosan in Figure 7 by finding a large signal at 1.5 and 2.3 keV due to carbon and oxygen. Particles average 50.06 nm, while surface protrusions are 2.14 nm. In addition to their elongated structure and parallel plates, NPs exhibit sharp edges.
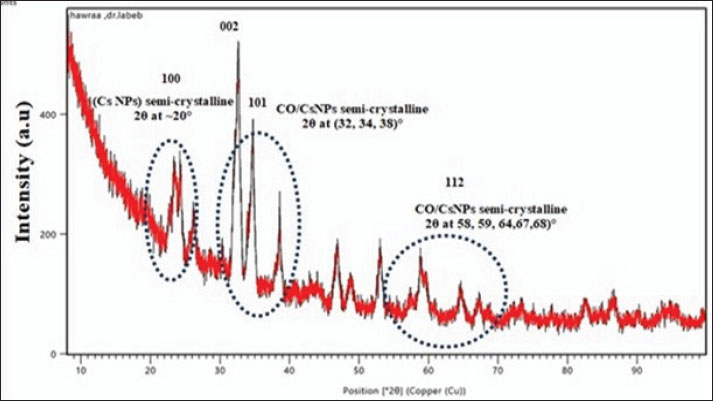
Fig. 7. EDX of CO/CsNPs. XRD of A Ex/CsNPsAttenuated semi-crystalline CO/CsNP semi-crystalline peaks: crystal plane reflection creates XRD peaks. Figure 8 shows the XRD patterns of CO/CsNPs and natural chitosan. The additional diffraction peaks correspond to the (1 0 0), (0 0 2), (1 0 1), and (1 1 2) planes of the CO/CsNPs. The data show that nanocomposite membranes integrated CO and CsNPs. By influencing steric interactions and intermolecular hydrogen bonding, CO NPs rearranged polymer chains. Existence of COEx components, e.g., N and other elements. Raza and Anwar (2017) found CsNPs on treated fabric using scanning electron microscopy with EDX. According to Omidi and Kakanejadifard (2019), the EDX spectra of CO/CsNPs contain C, O, Na, Ca, Mg, Al, N, and others in high weight percent.
Fig. 8. XRD diagram of the CO/CsNPs. Determination of CO/CsNP concentrationThe concentration of coconut oil extract/chitosan nanoparticles was determined using a UV-Vis spectrophotometer at an optical density of 284 nm and the standard curve equation of coconut extract (Fig. 9).
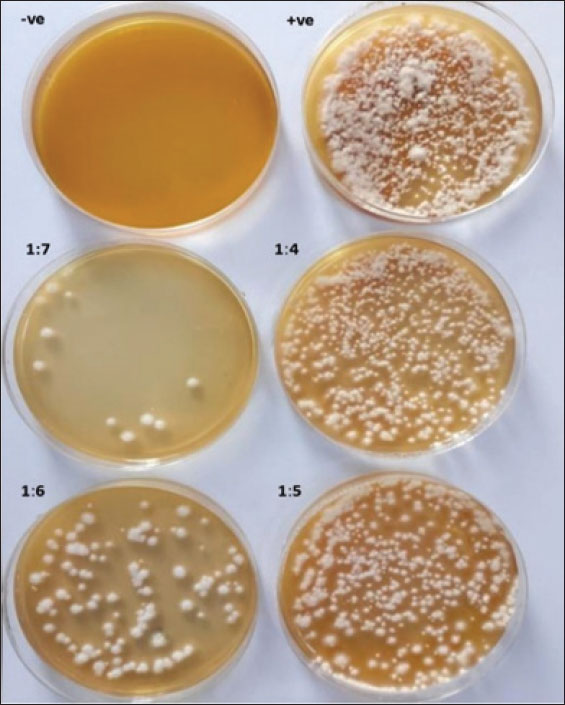
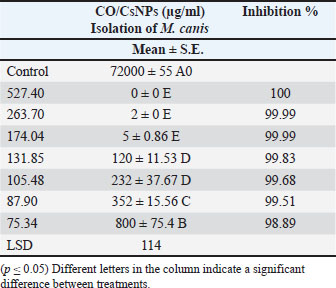
Fig. 9. Standard curve of the CO/CsNPs. (Y=2E-06x + 0.0102). Y represents the OD of the CO/CsNPs sample, and X represents the CO/CsNP concentration. Results of nanoparticles concentration equal 0.527 µg/ml. The final CO/ CsNP concentration equals 527.400 µg/ml. Evaluation of CO/CsNP activity against M. canis isolatesThe antifungal assay of coconut oil extract and CO/CsNPs at various concentrations was conducted on the M. canis isolate, which was inoculated onto the surface of freshly gelled sterile SDA plates using a sterilized swab stick for spreading. Table 2 presents the antifungal activity of CO/CsNPs at varying concentrations against the M. canis isolate, with the activity levels increasing with the concentration. These results are presented for three replicates. The concentrations of 527.40 µg/ml, 263.7 µg/ml, and 174.04 µg/ml exhibited minimum fungicidal activity, corresponding to 100%, 99.99%, and 99.99% inhibitory activity, respectively. These values demonstrated significant differences (p ≤ 0.05) when compared to the control and other CO/CsNP concentrations (131.85, 105.48, 87.90, and 75.34 µg/ml), which showed inhibitory activities of 99.83%, 99.68%, 99.51%, and 98.89%, respectively. Conversely, the activity of the NP indicated higher efficacy, as illustrated in Fig. 10.
Fig. 10. Antifungal activity of CO/CsNPs against M. canis isolates. Table 2. Antifungal activity of CO/CsNPs against M. canis isolates.
DiscussionCoconut products are valued for their antioxidant, anticancer, and nutritional qualities. This study examined the qualitative phytochemical content and biosynthesis of chitosan nano-encapsulated CO/CsNPs and the antifungal activity of CO against M. canis. Alkaloid, resin, phenol, and total phenolic compounds were found (Raza and Anwar, 2017; Omidi and Kakanejadifard, 2019). Coconut oil nonnutrient (phytochemical) content, which fights harmful free radicals in the body, is used medicinally (Obidoa et al., 2010. According to Platero et al. (2020), alkaloids have been utilized as central nervous system stimulants, topical anesthetics in ophthalmology, potent painkillers, and anti-puritics. Neurons, on the other hand, reduce inflammation, modify immunological responses, lower cholesterol, combat cancer, prevent diabetes, and destroy bacterial and fungal germs (Njoku et al., 2010; Celenk et al., 2023). Biosynthesis of coconut oil nanoencapsulation using chitosan yields a positively charged complex carbohydrate natural chitosan from chitin deacetylation. CS is pH-sensitive, chemically changeable, and breaks down, making it ideal for nanocarrier systems (Rohman et al., 2019). Ionic gelation is the most popular way to make chitosan nanoparticles because of its versatility. The method is simple and delicate, and it boosts compound activity (Purwanti et al., 2018). Comparing UV-Vis spectrophotometer data to COEx data, wavelength peaks and absorbance demonstrate that surface plasmon resonance (SPR) actions form nanoparticles. The SPR pattern depends on particle size, shape, synthesis media dielectric characteristics, and nanoparticle interactions. After extended interaction, the SPR band grew, indicating CsNP formation (Wang et al., 2014). Chitosan, phosphate, sodium, and potassium were confirmed by carbon and oxygen analysis and EDX analysis. Antunes Filho et al. (2023) found that biosynthesis-encapsulated nanoparticles with plant extracts and Cs molecules have attachment sites not filled by plant extract/CsNPs (665–910, 2376.3, 2868.15) cm1. The structural stability of chitosan nanoparticles following bioconversion is confirmed by FTIR. Nanocomposite particles with good surface protrusions, elongated structure, parallel plates, and sharp edges may damage microbial membranes and increase antibacterial effectiveness. EDX shows COEx components such as N and other elements in coconut oil extract with various active chemicals such as alkaloids and phenol. The peak at 2θ=~20° fits the characteristic chitosan peaks in the CsNP XRD patterns (Litvin and Minaev, 2013). The study exhibits the well-organized crystal structure of chitosan. Adding peaks at 2θ values of ~20° for chitosan lowered peak intensity. The XRD patterns of chitosan nanocomposite membranes reinforced with CO/CsNPs had lower peak intensity. The CO nanoparticles reorganized the polymer chains via steric and intermolecular hydrogen bonding. We agree with Filho et al. (2023) that chitosan NPs reduced composite membrane crystallinity and increased polymer chain flexibility. The 2θ value of ~20° aligns with (Li et al., 2010; Alnuaimi et al., 2023). XRD is used for composite material crystallinity and structural interactions. XRD of coconut oil with chitosan nanoparticles, a semi-crystalline biopolymer, exhibits peaks at ~10° and ~20° (2θ angles) due to its hydrated structure and inter-chain hydrogen bonding, indicating a curve with elements of both components. Coconut oil may reduce crystallinity if 10° and 20° chitosan peaks are weaker or wider than pure chitosan. Damage to the hydrogen-bonded network by oil can cause amorphous chitosan. Encapsulation with weak coconut oil peaks prevents crystallization by evenly distributing the oil in the chitosan matrix. Amorphous phase dominance allows the production of nanoparticles with a broken long-range structure and a large 15°–30° halo. The structural implications showed that encapsulation efficiency and crystalline peak suppression indicate chitosan encapsulation of coconut oil. Lowering chitosan crystallinity may improve nanoparticle stability, flexibility, and drug-loading for regulated release (Litvin and Minaev, 2013; Alnuaimi et al., 2023). If the peak shifts indicate chemical interactions (hydrogen bonding between the amino group of chitosan and the ester groups of oil) and complementary FTIR analysis shows a composite structure, the XRD curve may show reduced crystallinity and amorphous dominance due to chitosan nanoparticles encapsulating the oil. Further characterization would confirm these findings. Coconut oil has promising antifungal effects in vitro compared with COEx and nanoparticles. Coconut oil is tested on fungal cultures in controlled laboratories. Inhibition zone formed by VCO in M. canis culture. These results are in accordance with previous studies (Pangesti et al., 2023). The main findings of these studies are as follows. Coconut oil breaks cell membranes and inhibits Candida, Aspergillus, and Malaysian fungi in vitro. Coconut oil contains lauric acid, which destroys fungal cells membranes, killing them (Kaur et al., 2013). Coconut oil contains 92% saturated fatty acids, 50% of which are lauric acid, and is antimicrobial. Monolaurin and other medium-chain monoglycerides can change microbial cell walls, infiltrate and tear cell membranes, and kill microorganisms by inhibiting energy production and nutrition transfer enzymes (Haron et al., 2018). According to Quindos et al. (2019), coconut oil and chlorhexidine have antifungal action like ketoconazole. Coconut oil is effective against Candida albicans. Coconut oil had the highest inhibitory effect at 100 µl (Shino et al., 2016). These antioxidants stabilize coconut oil. The highest inhibitory percentage was observed at 527.4 µg/ml (minimum fungicidal concentration-MFC) for COEx/CsNPs, while the lowest inhibition was observed at 75.34 µg/ml (minimum inhibitory concentration, MIC), supporting the findings of some researchers (Kannan and Mohammed, 2014). Some fungal species were susceptible to coconut oil due to their mycotoxin production (Bassey et al., 2016; Anzaku, 2017). Coconut oil can treat fungal infections. The phytochemicals of coconut oil inhibit the tested fungal isolates (Ogbolu et al., 2007). The tiny diameter of nanoparticles allows active chemicals to adsorb onto microorganisms, improving their therapeutic effects (Mikhailova, 2020; Burchacka et al., 2023). Nanoparticles also have high porosity, specific surface area, and required surface functionality (Alvarez-Galvan et al., 2022; Franklin et al., 2024). Few threshold values exist for the efficacy of antimicrobial nanomaterials, but some authors suggest highly active and active plant extracts should have MIC values < 100 μg/ml and 100 ≤ MIC ≤ 512 μg/ml, respectively (Brice et al., 2023). Many studies have proven the effectiveness of nanoparticles, especially chitosan particles, in the manufacture of biological alternatives for the treatment of many diseases. Coconut oil, which is considered an antioxidant material that is used in many vital fields such as cooking and skin cosmetics to treat dermatophyte infections caused by M. canis in vitro, was chosen as a first step to manufacture harmless biological topical ointments as an alternative to chemical antifungals. ConclusionThis study examined coconut oil particles coated with chitosan nanoparticles as a promising antifungal agent, particularly for dermatophytosis (Ringworm) caused by M. canis. Coconut oil, which is rich in fatty acids such as lauric acid, exhibits antifungal properties by damaging fungal cell membranes and leveraging antioxidant activity. In vitro experiments demonstrate its efficacy in inhibiting fungal growth. The preparation and extraction process of virgin coconut oil CO is outlined. This study and its results are encouraging for upgrading the work to the in vivo stage. AcknowledgmentSpecial thanks go to Labeeb Ahmed Kadhim Alzubaidi, Scientific Research Commission/Research and Technology Center of Environment, Water and Renewable Energy, Baghdad, Iraq. FundingThe authors, participants in preparing this manuscript, confirm that we have not received any financial funding. Authors' contributionsAll researchers participated in preparing this manuscript, whether in the practical part or in writing and final preparation. Conflict of interestThe authors have no conflicts of interest to declare. Data availabilityAll data related to this manuscript are included. ReferencesAgarwal, M., Agarwal, M.K., Shrivastav, N., Pandey, S., Das, R. and Gaur, P. 2018. Preparation of chitosan nanoparticles and their in vitro characterization. Int. J. Life Sci. Scientific Res. 4(2), 1713–1720; doi: 10.21276/ijlssr.2018.4.2.17 Al-Abedi, H.F.H., Khalil, I.I., Ali, A.Q.M. and Al-Mayah, W.T. 2023. Morphological, molecular investigation of fungi isolated from layers poultry and their environment with their mycotoxins profile in Wasit province. J. Glob. Innov. Agric. Sci. 11(2), 141–146; doi:10.1016/j.jgi.2016.09.010 Alhasan, D.A., Al-Abedi, H.F., Hussien, T.J. and Mohammad Ali, A.Q. 2022. Morphological detection of dermatophytes isolated from cattle in Wasit province. Iraqi J. Vet. Sci. 36(I), 167–172; doi:10.33899/ijvs.2022.135833.2530 Alnuaimi, M.T.A., Al-Hayanni, H.S.A. and Aljanabi, Z.Z. 2023. Green synthesis of gold nanoparticles from Sophora flavescens extract and their antibacterial effect against some pathogenic bacteria. Malaysian J. Microbiol. 19(1), 74–82; doi:10.21161/mjm.2200 Alvarez-Galvan, Y., Minofar, B., Futera, Z., Francoeur, M., Jean-Marius, C., Brehm, N., Yacou, C., Jauregui-Haza, U.J. and Gaspard, S. 2022. Adsorption of hexavalent chromium using activated carbon produced from Sargassum ssp.: comparison between lab experiments and molecular dynamics simulations. Molecules 27, 6040; doi:10.3390/molecules27186040 Amelia, B., Saepudin, E., Cahyana, A.H., Rahayu, D.U., Sulistyoningrum, A.S. and Haib, J. 2017. GC-MS analysis of clove (Syzygium aromaticum) bud essential oil from Java and Manado. In AIP Conference Proceedings, American Institute of Physics, College Park, MD, 1862(1), p 30082. doi: 10.1063/1.4991186 Antunes Filho, S., Dos Santos, M.S., Dos Santos, O.A.L., Backx, B.P., Soran, M.L., Opriş, O., Lung, I., Stegarescu, A. and Bououdina, M. 2023. Biosynthesis of nanoparticles using plant extracts and essential oils. Molecules 28(7), 3060; doi:10.3390/molecules28073060 Anzaku, A.A. 2017. Antimicrobial activity of coconut oil and its derivative (Lauric acid) on some selected clinical isolates. Int. J. Med. Sci. Clin. Inventions 4(8), 3173–3177; doi:10.18535/ijmsci/v4i8.12 Bassey, E.E., Gwana, A.M., Halima, M.B., Mohammed, A.B., Bagudu, B.Y., Okeke, I., Umeh, S.O., Abubakar, M. and Mbanu, B. 2016. Comparative phytochemical screening and in vitro antimicrobial activity of aqueous, ethanolic and ethyacetate extracts of stem bark and leaves of horse radish (Moringa oleifera) plant. Academic J. Life Sci. 2(9), 61–76; doi:10.30574/wjarr.2019.1.3.0006 Boulaiz, H., Alvarez, P.J., Ramirez, A., Marchal, J.A., Prados, J., Rodríguez-Serrano, F., Perán, M., Melguizo, C. and Aranega, A. 2011. Nanomedicine application areas and development prospects. Int. J. Mol. Sci. 12(5), 3303–3321; doi:10.3390/ijms12053303 Brice, S. 2023. Making space for aradical trans imagination: Towarda kinder, more vulnerable, geography. EPD Soc, Space 41(4), 592 -599. Brito, R.E., González-Rodríguez, J., Montoya, M.R. and Rodríguez Mellado, J.M. 2017. Comparison of the volatile antioxidant contents in the aqueous and methanolic extracts of a set of commercial spices and condiments. Eur. Food Res. Technol. 243(8), 1439–1445; doi: 10.1007/s00217-017-2854-x Burchacka, E., Pstrowska, K., Bryk, M., Maciejowski, F., Kułażyński, M. and Chojnacka, K. 2023. The properties of activated carbons functionalized with an antibacterial agent and a new SufA protease inhibitor. Materials 1263, 1–18; doi: 10.3390/ma16031263 Cary, N.C. 2018. Statistical analysis system, user’s guide. Statistical. Version 9.6th. SAS. Inst. Inc. USA. Castro Méndez, C., Carmen Serrano, M., Valverde, A., Pemán, J., Almeida, C. and Martín-Mazuelos, E. 2008. Comparison of E-test®, disk diffusion and a modified CLSI broth microdilution (M 38-A) method for in vitro testing of itraconazole, fluconazole and voriconazole against dermatophytes. Med. Mycology 46(2), 119–123; doi:10.1080/13693780701670491 Celenk, V.U., Gumus, Z.P. and Argon, Z.U. 2023. Bioactive phytochemicals from coconut (Cocos nucifera) oil processing by-products. Ref. Work Entry 6, 309–321. Doi: 10.1007/978-3-030-91381-6_14 Ch, O., Reisine, S.L. and Cherniack, M. 2010. How does the SF-36 perform in healthy populations? a structured review of longitudinal studies. J. Social Evol. Cultural Psychol. 4(1), 1–18; doi: 10.5590/JSBHS.2010.04.1.02 Dimitrijevic, R., Cvetkovic, O., Miodragović, Z., Simic, M., Manojlović, D. and Jovic, V. 2013. SEM/EDX and XRD characterization of silver nanocrystalline thin film prepared from organometallic solution precursor. J. Min. Metall. Sect. B-Metall. 49(1), 91–95; doi:10.2298/JMMB120111041D Du, W.L., Xu, Z.R., Han, X.Y., Xu, Y.L. and Miao, Z.G. 2008. Preparation, characterization and adsorption properties of chitosan nanoparticles for eosin Y as a model anionic dye. J. Hazardous Mater. 153(1–2), 152–156; doi:10.1016/j.jhazmat.2007.08.040 Franklin, L.T.T., Boniface, P.K., Vincent, N., Zuriatou, Y.T., Yimgang JN Ndi. and Paul FB Fabrice. 2024. Biological synthesis and characterization of silver-doped nanocomposites: antibacterial and mechanistic studies. Drugs Drug Candidates 3, 13–32; doi:10.3390/ddc3010002 Gholami-Shabani, M., Akbarzadeh, A., Norouzian, D., Amini, A., Gholami-Shabani, Z., Imani, A., Chiani, M., Riazi, G., Shams-Ghahfarokhi, M. and Razzaghi-Abyaneh, M. 2014. Antimicrobial activity and physical characterization of silver nanoparticles green synthesized using nitrate reductase from Fusarium oxysporum. Appl. Biochem. Biotechnol. 172, 4084–4098; doi:10.1007/s12010-014-0809-2 Guilger-Casagrande., M. and Lima, D.R. 2019. Synthesis of silver nanoparticles mediated by fungi: a review. Front. Bioeng. Biotechnol. 7, 287; doi: 10.3389/fbioe.2019.00287 Haron, U.A., Abllah, Z. and Nasir, N.A. The comparative antimicrobial effect of activated virgin coconut oil (AVCO) and virgin coconut oil (VCO) against dental caries-related pathogens. International Dental Conference of Sumatera Utara 2017 (IDCSU 2017),2018, Medan, Indonesia, Atlantis Méndez Press, doi: 10.2991/idcsu-17.2018.79 Hasab Allh, H.F. and Alhasan, D.A. 2023. Detection of the compounds produced from Rhizopus oryzae. Iraqi J. Vet. Sci. 37(3), 611–617; doi: 10.33899/ijvs.2023.135462.2484 Kannan, N. and Mohammed, A. 2014. Comparative evaluation of antifungal activity of Cocos nucifera oil against candida albicans. Int. J. Phytother. Res. 4, 27–31; doi:10.4103/jiaomr.jiaomr_137_21 Kaur, P., Choudhary, A. and Thakur, R. 2013. Synthesis of Chitosan-silver nanocomposites and their antibacterial activity. Int. J. Sci. Eng. Res. 4(4), 869–872. Available via https://www.researchgate.net/publication/273455396 Keshwania, P., Kaur, N., Chauhan, J., Sharma, G., Afzal, O., Alfawaz Altamimi, A.S. and Almalki, W.H. 2023. Superficial dermatophytosis across the world’s populations: potential benefits from nanocarrier-based therapies and rising challenges. ACS. Omega 8(35), 31575–31599. Kolo, M.T., Khandaker, M.U., Amin, Y.M., Abdullah, W.H.B., Bradley, D.A. and Alzimami, K.S. 2018. Assessment of health risk due to the exposure of heavy metals in soil around mega coal-fired cement factory in Nigeria. Results Phys. 11(11), 755–762. Kumar Nigam, P. 2015. Antifungal drugs and resistance: current concepts. Our Dermatology. Online 6(212) 212–221, doi: 10.7241/ourd.20152.58 Kumar, A. and Bali, A. 2025. Development of advanced instruments for spectroscopic analyses of nanoparticles/nanobioparticles. In Das, S., Mukundrao Khade, S., Barman Roy, D. and Trivedi, K. (eds). Synthesizing and characterizing plant-mediated biocompatible metal nanoparticles. IGI Global (Scientific Publishing), Pune, India. p. 24, https://doi.org/10.4018/979-8-3693-6240-2.ch011 Li, L.H., Deng, J.C., Deng, H.R., Liu, Z.L. an`d Xin, L. 2010. Synthesis and characterization of chitosan/ZnO nanoparticle composite membranes. Carbohydr. Res. 345(8), 994–998; doi:10.1016/j.carres.2010.03.019 Litvin, V.A. and Minaev, B.F. 2013. Spectroscopy study of silver nanoparticles fabrication using synthetic humic substances and their antimicrobial activity. Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 108, 115. Mikhailova, E.O. 2020. Silver Nanoparticles: mechanism of action and probable bio-application. J. Funct. Biomater. 11, 84; doi:10.3390/jfb11040084 Mohammadi, A., Hashemi, M. and Hosseini, S.M. 2015. Nanoencapsulation of Zataria multiflora essential oil preparation and characterization with enhanced antifungal activity for controlling Botrytis cinerea, the causal agent of gray mould disease. Innov. Food Sci. Emerg. Technol. 28, 73–80; doi: 10.1016/j.ifset.2014.12.011 Njoku, O.U., Boniface, J.A.E., Obitte, N.C., Odimegwu, D.C. and Ogbu, H.I. 2010. Some nutriceutical potential of beniseed oil. Int. J. Appl. Res. Natural Product 2(4), 11–19; https://www.semanticscholar.org/paper Obidoa, O., Joshua, P.E. and Eze, N.J. 2009. Phytochemical analyses of Cocos nucifera L. Arch. Pharm. Sci. Res. 1(1), 87 -96. Ogbolu, D.O., Oni, A.A., Daini, O.A. and Oloko, A.P. 2007. In vitro antimicrobial properties of coconut oil on Candida species in Ibadan, Nigeria. J. Med. Food 10, 384–387; doi:10.1089/jmf.2006.1209 Oh, J.W., Chun, S.C. and Chandrasekaran, M. 2019. Preparation and in vitro characterization of chitosan nanoparticles and their broad-spectrum antifungal action compared to antibacterial activities against phytopathogens of tomato. Agronomy 9(1), 1-12; doi: 10.3390/agronomy9010021 Omidi, S. and Kakanejadifard, A. 2019. Modification of chitosan and chitosan nanoparticle by long chain pyridinium compounds: synthesis, characterization, antibacterial, and antioxidant activities. Carbohydrate Polym. 208, 477–485; doi:10.1016/j.carbpol.2018.12.097 Pangesti, R.R., Rahmahani, S.J. and Indrawati, A. 2023. Antifungal activity test of virgin coconut oil (VCO) against Microsporum canis. YMER 22(11), 311–321. Available via http://ymerdigital.com Pinlap, B.R., Pone Kamdem, B., Kamto, E.L.D., Ngouana, V., Melogmo Dongmo, Y.K., Lunga, P.K. and Fekam Boyom, F. 2023. Extracts from Cardiospermum grandiflorum and Blighia welwitschii (Sapindaceae) reveal antibacterial activity against Shigella species. S. Afr. J. Bot. 164, 419–428; doi: 10.1016/j.sajb.2023.12.010 Platero, J.L., Cuerda-Ballester, M., Ibáñez, V., Sancho, D., Lopez-Rodríguez, M.M., Drehmer, E. and De La Rubia Ortí, J.E. 2020. The impact of coconut oil and epigallocatechin gallate on the levels of IL-6, anxiety and disability in multiple sclerosis patients. Nutrients 12, 305; doi:10.3390/nu12020305 Purwanti, N., Zehn, A.S., Pusfitasari, E.D., Khalid, N., Febrianto, E.Y., Mardjan, S.S., Andreas. and Kobayashi, I. 2018. Emulsion stability of clove oil in chitosan and sodium alginate matrix. Int. J. Food Properties 21(1), 566–581; doi:10.1080/10942912.2018.1454946 Quindos, G., Gil-Alonso, S., Marcos-Arias, C., Sevillano, E., Mateo, E., Jauregizar, N. and Eraso, E. 2019. Therapeutic tools for oral candidiasis: current and new antifungal drugs. Med. Oral Patol. Oral Cir. Bucal 24, e172–e180; doi:10.4317/medoral.22978 Raza, Z.A. and Anwar, F. 2017. Fabrication of chitosan nanoparticles and multi-response optimization in their application on cotton fabric by using a Taguchi approach. Nano-Structures Nano-Objects 10, 80–90; doi:10.1016/j.nanoso.2017.03.007 Rohman, A., Irnawati., Erwanto, Y., Lukitaningsih, E., Rafi, M., Fadzilah, N.A., Windarsih, A., Sulaiman, A. and Zakaria, Z. 2019. Virgin coconut oil: extraction, physicochemical properties, biological activities and its authentication analysis. Food Rev. Int. 37(1), 1–21; doi:10.1080/87559129.2019.1687515 Sarvamangala, D., Kondala, K., Murthy, U.S.N., Rao, B.N., Sharma, G.V.R. and Satyanarayana, R. 2013. Biogenic synthesis of AGNP’s using pomelo fruit – characterization and antimicrobial activity against Gram+ve and Gram-ve Bacteria. Int. J. Pharm. Sci. Rev. Res. 19(2), 30–35; https://www.researchgate.net/publication/276025370 Shaghati, H.A., Jassim, E.H. and Al-Zubaidi, L.A.K. 2023. Evaluation of the Inhibitory Activity of Syzygium aromaticum Extract-Chitosan Nanoparticles Against Biofilm Formation of Klebsiella pneumoniae. Bionatura 8(1), 1–14. Shino, B., Peedikayil, F.C., Jaiprakash, S.R., Ahmed, B.G., Kottayi, S. and Jose, D. 2016. Comparison of antimicrobial activity of chlorhexidine, coconut oil, probiotics, and ketoconazole on Candida albicans isolated in children with early childhood caries: an in vitro study. Scientifica 70, 61587. doi: 10.1155/2016/7061587 Wang, X., Chen, Y., Dahmani, F.Z., Yin, L., Zhou, J. and Yao, J. 2014. Amphiphilic carboxymethyl chitosan-quercetin conjugate with P-gp inhibitory properties for oral delivery of paclitaxel. Biomaterials 35, 7654–7665; doi:10.1016/j.biomaterials.2014.05.053 Wasien, S.M., Hatim, J.A. and Alzubaidi, L.A.K. 2019. Treatment of stored rice seeds contaminated with fungi by green synthesized silver nanoparticles produced from turmeric and cinnamon plants extracts. Baghdad: University of Baghdad/College of Science Department of Biology. pp. 46–47. Youssef, F.S., El-Banna, H.A., Elzorba, H.Y. and Galal, A.M. 2019. Application of some nanoparticles in the field of veterinary medicine. Int. J. Vet. Sci. Med. 7(1), 78–93. | ||
| How to Cite this Article |
| Pubmed Style Khalil II, Al-abedi HF, Jasim AS, Younus JG. Evaluation of the antifungal activity of coconut oil/chitosan nanoparticles against Microsporum canis. Open Vet. J.. 2025; 15(10): 4984-4996. doi:10.5455/OVJ.2025.v15.i10.17 Web Style Khalil II, Al-abedi HF, Jasim AS, Younus JG. Evaluation of the antifungal activity of coconut oil/chitosan nanoparticles against Microsporum canis. https://www.openveterinaryjournal.com/?mno=264140 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.17 AMA (American Medical Association) Style Khalil II, Al-abedi HF, Jasim AS, Younus JG. Evaluation of the antifungal activity of coconut oil/chitosan nanoparticles against Microsporum canis. Open Vet. J.. 2025; 15(10): 4984-4996. doi:10.5455/OVJ.2025.v15.i10.17 Vancouver/ICMJE Style Khalil II, Al-abedi HF, Jasim AS, Younus JG. Evaluation of the antifungal activity of coconut oil/chitosan nanoparticles against Microsporum canis. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4984-4996. doi:10.5455/OVJ.2025.v15.i10.17 Harvard Style Khalil, I. I., Al-abedi, . H. F., Jasim, . A. S. & Younus, . J. G. (2025) Evaluation of the antifungal activity of coconut oil/chitosan nanoparticles against Microsporum canis. Open Vet. J., 15 (10), 4984-4996. doi:10.5455/OVJ.2025.v15.i10.17 Turabian Style Khalil, Israa Ibrahim, Hawraa F.h. Al-abedi, Alyaa Sabti Jasim, and Junaid Ghanim Younus. 2025. Evaluation of the antifungal activity of coconut oil/chitosan nanoparticles against Microsporum canis. Open Veterinary Journal, 15 (10), 4984-4996. doi:10.5455/OVJ.2025.v15.i10.17 Chicago Style Khalil, Israa Ibrahim, Hawraa F.h. Al-abedi, Alyaa Sabti Jasim, and Junaid Ghanim Younus. "Evaluation of the antifungal activity of coconut oil/chitosan nanoparticles against Microsporum canis." Open Veterinary Journal 15 (2025), 4984-4996. doi:10.5455/OVJ.2025.v15.i10.17 MLA (The Modern Language Association) Style Khalil, Israa Ibrahim, Hawraa F.h. Al-abedi, Alyaa Sabti Jasim, and Junaid Ghanim Younus. "Evaluation of the antifungal activity of coconut oil/chitosan nanoparticles against Microsporum canis." Open Veterinary Journal 15.10 (2025), 4984-4996. Print. doi:10.5455/OVJ.2025.v15.i10.17 APA (American Psychological Association) Style Khalil, I. I., Al-abedi, . H. F., Jasim, . A. S. & Younus, . J. G. (2025) Evaluation of the antifungal activity of coconut oil/chitosan nanoparticles against Microsporum canis. Open Veterinary Journal, 15 (10), 4984-4996. doi:10.5455/OVJ.2025.v15.i10.17 |