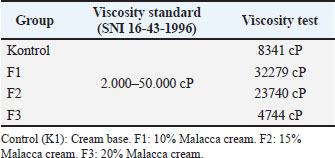| Research Article | ||
Open Vet. J.. 2025; 15(10): 4972-4983 Open Veterinary Journal, (2025), Vol. 15(10): 4972-4983 Research Article Topical anti-inflammatory potential of ethanol extract of Phyllanthus emblica leaves in a carrageenan-induced mouse modelNuzul Asmilia1*, Amalia Sutriana2, Henni Vanda2, Frengki2, Mazayya Qanita Manan1, Dwi Salsha Reyno1and Muhammad Rifqi Al Ikhzan21Department of Clinic Veterinary, Faculty of Veterinary Medicine, Universitas of Syiah Kuala, Banda Aceh, Indonesia 2Department of Pharmacology Veterinary, Faculty of Veterinary Medicine, Universitas of Syiah Kuala, Banda Aceh, Indonesia *Corresponding Author: Nuzul Asmilia. Department of Clinic Veterinary, Faculty of Veterinary Medicine, Universitas of Syiah Kuala, Banda Aceh, Indonesia. Email: nuzulasmilia [at] usk.ac.id Submitted: 14/06/2025 Revised: 23/08/2025 Accepted: 02/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
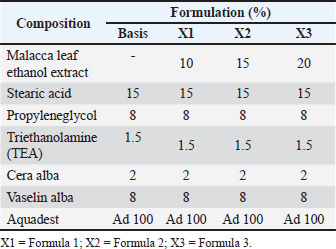
AbstractBackground: Inflammation is a protective mechanism by which the body neutralizes and eradicates harmful agents at the site of injury and conditions tissue repair. Malacca plants contain active metabolites that are believed to be able to cure inflammation. Aim: This study aims to test the formula of ethanol extract cream of Malacca leaves 10%, 15%, and 20% with a negative control group (without treatment) and a positive control group (treated with betamethasone cream) and its anti-inflammatory activity through observation of edema of the feet of male mice induced by carrageenan. Methods: The physical stability of the cream formula was evaluated every week for 3 weeks at room temperature storage (±29°C) by observing organoleptic, pH, viscosity, adhesiveness, and spreadability. Furthermore, an in vivo test consisting of five treatments and five repetitions using the mouse hind paw edema method. The volume of edema was measured using a plestimometer 30 minutes after carrageenan administration and every day for 4 days of treatment. Results: The results of the study showed the most significant decrease in edema volume using 10% (F1) Malacca cream. Differential leukocyte counts showed that the percentage of eosinophils in group K1 was the highest and significantly different (p < 0.05) compared to other groups (K2, F1, F2, and F3). The percentage of neutrophils in groups F2 and F3 increased, whereas that in group K2 decreased, and that in group F1 decreased significantly (p < 0.05). The percentage of monocytes in the groups treated with K2, F1, F2, and F3 was higher than that in group K1. The percentage of lymphocytes in groups F1, F2, and F3 was higher than that in group K1. However, basophils were not found in all treatment groups. Conclusion: Ethanol extract of Malacca cream (Phyllanthus emblica) has been proven to have anti-inflammatory effects in mice after being induced by carrageenan, with mechanisms such as inhibition of the NF-κB pathway, modulation of pro-inflammatory cytokines, activation of the antioxidant pathway, and inhibition of inflammatory enzymes. The most optimal potential as an anti-inflammatory and leukocyte regulation agent is shown in the administration of 10% Malacca cream. Keywords: Malacca leaf ethanol extract, Inflammation, Leukocyte differential, Plethysmometer, Phyllanthus emblica. IntroductionInflammation is a body defense reaction that occurs when tissue injury or infection occurs. Inflammation plays a role in killing microorganisms or substances considered foreign by the body. Inflammation also plays a role in the elimination of dead or damaged cells (Haque and Sugihartini, 2015). Common signs of inflammation include calor (heat), dolor (pain), rubor (red), and tumor (swelling) (Ciaccia, 2011). Inflammation can be treated with steroids and non-steroidal anti-inflammatory drugs. However, both often cause stomach pain due to prostaglandin production suppression as a protector of the gastric mucosal layer. Other frequently reported side effects include liver damage due to toxic metabolite products and kidney problems (suppression of prostaglandins causes reduced blood flow to the kidneys) (Takeuchi, 2012; Bindu et al., 2020). The use of herbs as an alternative is increasingly popular for several reasons, including wide availability and easy availability, affordable prices, flexible use, and minimal side effects (Ekor, 2014; Welz et al., 2018). Herbs, such as turmeric and temulawak, also have multicomplex effects. In addition to being anti-inflammatory, they are also reported to be effective as antimicrobials, antioxidants, and immunostimulants that work synergistically (Parham et al., 2020; Rahmad et al., 2021; Nainggolan et al., 2023; Nainggolan et al., 2025). Topical formulas are often more effective and safe to treat non-systemic inflammation, such as muscle pain, mild arthritis, or skin irritation, than oral medications because they can work directly at the site of inflammation, minimizing systemic exposure and reducing the risk of side effects on the stomach (Shi et al., 2022). Cream formulas are chosen because of the balance among absorption, stability, and comfort of use. Cream formulas are more optimally absorbed into the skin than ointments, are more stable than gels, and are suitable for both dry and oily skin types (Barnes et al., 2021; Tania et al., 2022). Malacca (Phyllanthus emblica) is a plant that is often used as a traditional medicine for various diseases in Aceh Besar Regency (Asmilia et al., 2019). The malacca plant is known in Indonesia as himalaya (Malay), balaka (Minangkabau), malaya (Sundanese and Betawi), keloid (Javanese), and malakah (Madura). This plant is commonly known as Indian gooseberry, Amla, or Myrobalan (Holthoon, 2011). Dhale and Mogle (2011) identified the phytochemicals of this plant containing alkaloid, flavonoid, saponin, tannin, terpenoid, glycerol, benzene, phenolic, lignin, furanolactone, and triterpene groups. The main groups of polyphenol-flavonoid compounds contained in this plant are gallic acid, astragalin, corilagin, quercetin, kaempferol, apigenin, epigallocatechin gallate, and rutin (Prananda et al., 2023). Embellican A and B, lupeol, punicalagin, and minerals such as calcium, phosphorus, and iron in small amounts were also identified. This plant has been reported to be effective as an analgesic, hepatoprotective, antipyretic, anti-inflammatory, anticancer, antihyperlipidemic, antidiarrheal (Kaur et al., 2013), chemoprotective and antioxidant (Charoenteeraboon et al., 2010), antitumor (Sumalatha, 2013), able to lower blood sugar levels (Qureshi et al., 2009), antiviral, antimutagenic, antiallergic (Khopde et al., 2001), antimicrobial, antifungal, and antibacterial (Elangovan et al., 2015; Singh et al., 2015). Several preclinical studies have reported the effects of extract of Malacca leaf (P. emblica). An ethanolic extract of P. emblica leaves at doses of 200–400 mg/kg demonstrated edema inhibition comparable to that of 10 mg/kg diclofenac in Wistar rats (Golechha et al., 2011). Administration of an aqueous extract at 500 mg/kg/day reduced joint swelling by nearly 50% by day 14 (Yokozawa et al., 2007). A methanol extract (300 mg/kg) showed 2–3 times greater pain reduction in the Hot-Plate and Tail-Flick tests (Perianayagam et al., 2004). Acute toxicity tests indicated the safety of the extract with an LD50 > 5,000 mg/kg, while subchronic toxicity studies revealed no hepatic or renal damage at doses of 1,000 mg/kg/day over 28 days of administration (Rao et al., 2011; Patel et al., 2013). The analgesic-anti-inflammatory mechanism of this plant metabolite, such as luteolin, has also been reported in the form of inhibition of COX-2 enzyme activity, which suppresses the formation of prostaglandins (PGE2) and pro-inflammatory cytokines (TNF-α, IL-6) and pain inhibition through modulation of pain receptors (TRPV1) (Palazzo et al., 2008; Ntolouka et al., 2023). The combination interaction with antidiabetic drugs may potentially cause excessive hypoglycemia, while concurrent use with antiplatelet agents increases bleeding risk (Qureshi et al., 2009). Therefore, testing the extract of the Malacca plant in the form of a topical formula, such as a cream, is very promising because it has been supported by a lot of pre-clinical test data. Based on the potential of active compounds in the malacca plant (P. emblica), which exhibit anti-inflammatory mechanisms through COX-2 inhibition, PGE2, and pro-inflammatory cytokines (TNF-α, IL-6), as well as pain receptor modulation (TRPV1), we hypothesized that malacca leaf extract cream will show significant anti-inflammatory effects in a carrageenan-induced mouse paw edema model, showing reduction in swelling and inflammatory parameters (calor, dolor, rubor, and tumor) comparable or superior to standard drugs (topical NSAIDs/steroids). The rat paw edema model is a classical pharmacological approach for evaluating the anti-inflammatory activity of test compounds through induction by carrageenan, histamine, or other pro-inflammatory agents (Winter and Nuss, 1962). This model offers several key advantages, including simplicity, cost-effectiveness, and physiological relevance for studies on acute inflammation (Di Rosa et al., 1971). However, it has limitations because it primarily reflects the early phase of inflammation (vasodilation and edema), necessitating the use of complementary chronic models (such as rheumatoid arthritis in rats) for assessing long-term effects (Yokozawa et al., 2007). Despite this, the rat paw edema test remains the gold standard for preliminary screening of anti-inflammatory compounds due to its reliable predictive value for COX inhibition activity and lysosomal membrane stabilization (Perianayagam et al., 2004). The topical malacca extract cream formulation will locally modulate the immune response, as reflected by changes in the differential leukocyte profiles during therapy. The cream will show minimal skin irritation or systemic toxicity due to its localized action and synergistic composition of natural compounds. The cream from malacca (P. emblica) extract has potential for further development in treating inflammation in livestock such as cattle and sheep, as its natural compounds directly act on inflamed areas (e.g., mastitis and foot lesions), with fewer side effects than chemical drugs. Materials and MethodsMaterialsSamples of Malacca leaves (P. emblica), Aquades, 96% ethanol solvent, filter paper, 1% carrageenan, standard feed, drinking water, 0.1 mg betamethasone cream, and cream-making materials [stearic acid, propylene glycol, triethanolamine, cera alba, and vaseline alba (Table 1)] were used in this study. The equipment used included analytical scales, knives, measuring cups, glass jars, funnels, stirring rods, rotatory vacuum evaporators, water baths, beakers, mice cages, and mercury plethysmometer. Table 1. Formulation of the Malacca cream.
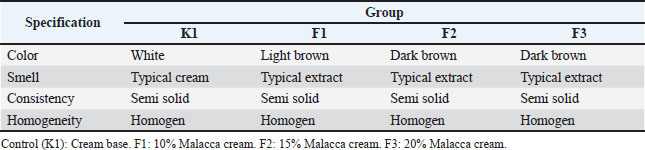
Ethanol extract of Malacca leaves (P. emblica)Malacca leaf samples were collected from Lambirah Bha Ulee Tutu Village, Simpang Tiga District, Aceh Besar Regency. The sample was rinsed and dried under a steady flow of clean water. The dried leaves were ground using a blender and sieved to obtain a smooth simplicia. The powder was then macerated with 5 l of 96% ethanol solution for 5 days with occasional stirring, filtered, and macerated again with 96% ethanol using the same procedure. The solvent was then evaporated using a vacuum rotary evaporator to obtain a thick extract of Malacca leaves. The extract was dried in an oven at a temperature of 50°C to obtain a thick extract. Preparation and dosage determination of Malacca cream (P. emblica)The stages of cream preparation refer to the report of Rikomah et al. (2021). The preparation of the cream base begins with the preparation of a fat phase containing stearic acid, cera alba, and vaseline alba, heated at a temperature of 70°C until melted. Then, the liquid phase containing distilled water, triethanolamine, and methyl propylene glycol was heated to a temperature of 70°C. The fat phase was put into a mortar, which was then gradually added to the liquid phase and ground until homogeneous. The ethanol extract of Malacca leaves is then mixed with the finished cream base and ground until homogeneous. Then, a series of concentrations of ethanol extract cream of Malacca leaves are made at 10%, 15%, and 20%. Table 1 shows the formulation for making Malacca cream. Physical properties evaluation of Malacca creamThe cream formulation’s physical quality was evaluated for 3 weeks. Each formulation was stored at a temperature of ±29°C. The parameters evaluated included organoleptic examination (color, odor, shape, and homogeneity), pH measurement, viscosity measurement, spreadability examination, and adhesion examination. OrganolepticOrganoleptic tests include the direct examination of texture, odor, color, and homogeneity using the five senses. A good cream formula has a semi-solid form, pleasant odor, and attractive color that is homogeneous and does not contain coarse granules (Elya et al., 2013). Determination of pHTo determine its pH, 1 g of cream was weighed and mixed with 10 ml of distilled water. The measurement process was then carried out using a pH meter by immersing its sensor into the mixture, allowing the pH value to be observed on the monitor. The reference for evaluating the cream's pH is SNI 16-4399-1996 (pH 4.5–8.0), which serves as the quality standard for skin moisturizing cream. ViscosityThe viscosity was determined using a Brookfield viscometer. The cream formulation sample was placed in a spindle-shaped cup 4 was installed, and the rotor was activated at a speed of 12 rpm. After the Brookfield viscometer showed a stable reading, the findings were recorded and multiplied by a factor of 500 (Sundar et al., 2022). The viscosity range commonly used for cream formulations is 2,000–50,000 cP, in accordance with SNI 16-43-1996. SpreadabilityThe spread-ability test was conducted to evaluate the ease of cream application (Lumentut et al., 2020). A sample of each cream formulation (0.5 g) was weighed and placed in the center of the glass plate. After 1 minute, it was covered with another glass plate, and the cream's diameter was measured. Then, it was given a weight of 50 g for 1 minute, and its diameter was measured again. Furthermore, it was given a weight of 100 g for 1 minute, and the diameter was measured again. Good spreadability increases the contact area between the cream and the skin and increases the adsorption of the active substance. AdhesionThis test is carried out by attaching a 0.5 g sample of each cream formulation to a glass object. Furthermore, it is covered with another glass object and pressed with a load of 250 g for 5 minutes. Next, the time is recorded until the two objects are released. The value of good cream adhesion ranges from 2 to 300 seconds (Yacobus et al., 2019). In vivo anti-inflammatory test resultsAnimal preparationThe experimental animals used in this study were five groups of male white mice (Mus musculus) aged 2–3 months weighing 25–30 g that received the same treatment. The mice had fasted for 18 hours before being treated but had been acclimatized a week before. Preparation of 1% carrageenan solution and edema induction in mouse pawsPreparation of 1% carrageenan solution based on Lallo et al. (2020), namely 50 mg of carrageenan was weighed and then dissolved in physiological saline solution (0.9% NaCl) to obtain a volume of 5 ml. 30 minutes before treatment, each mouse was subplantarly injected with 1% carrageenan solution into the sole of its left foot as much as 0.05 ml. Anti-inflammatory testThe anti-inflammatory effect test was performed using a plethysmometer (Ugo Basile) (Lallo et al., 2020), which is an edema induction method with carrageenan on the soles of the mice's feet. The volume of the soles of each test animal was first measured using a plethysmometer. The edema volume was measured at the 30th minute after carrageenan administration and every day for 4 days of treatment. During the measurement, the fluid volume must be the same for each measurement, the boundary marks on the mouse’s feet must be clear, and the mouse’s feet must be immersed to the limit made. Furthermore, each mouse’s foot was smeared with extract cream and control material every day, 2 times a day for 4 days of treatment. K1 was given a cream base as a negative control, and K2 was given betamethasone cream as a positive control. Meanwhile, F1, F2, and F3 were given Malacca cream with 10%, 15%, and 20% topically, respectively. The parameters observed were the decrease in edema volume in the soles of the mice's feet before and after being induced by carrageenan to determine the effectiveness of the Malacca cream concentration. Differential leukocyte countDifferential leukocyte counting was performed 4 days after treatment. Blood was collected from the caudal vein, and then one drop of blood was dripped onto a glass object and smoothed using another glass object to obtain a thin blood smear, which was then dried. After drying, each preparation was soaked in methanol for 1–2 seconds. Then, it is dipped in the eosin solution for 20–30 seconds. Next, for 15–30 seconds, it is dipped in methylene blue solution for 15–30 seconds and rinsed with water. The preparation was dried and then observed under a microscope with 100× magnification using immersion oil. Then, 100 cells of all types of leukocytes, including neutrophils, eosinophils, monocytes, lymphocytes, and basophils, were counted (Bellwood and Andrasik-Catton, 2014). Ethical approvalThis research was conducted after obtaining a research permit from the Ethics Commission for the Use of Experimental Animals, Faculty of Veterinary Medicine, Syiah Kuala University (No. 123/KEPH/VIII/2021). ResultsExtracting Malacca leafExtraction of 1 kg of Malacca leaf powder produces a thick extract with a distinctive Malacca leaf odor, semi-solid texture, and blackish brown color, as much as 125.56 g. Evaluation of the Malacca cream formulationThe results of organoleptic observations of all cream formulas showed that the Malacca leaf ethanol extract cream on day 0 had the same characteristics, namely the distinctive smell of Malacca leaf extract in each formulation, brown color with different levels of color concentration [faded brown (F1), dark brown (F2), and darker brown (F3)], semi-solid shape, soft and homogeneous texture. After three weeks of storage at room temperature (±29°C), re-observations were performed, and F1, F2, and F3 maintained their color, odor, shape, and homogeneity (Table 2). Table 2. Results of organoleptic observation.
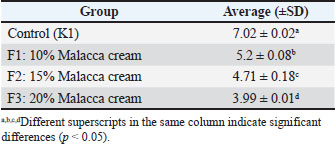
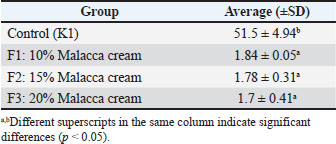
The pH test data showed that the average pH value of the cream decreased as the concentration of the ethanol extract from Malacca leaves increased. The highest average pH value of the cream was found in F1, which was given a concentration of 10% ethanol extract from Malacca leaves, while the lowest average pH value of the cream was found in F3, which was given 20% ethanol extract from Malacca leaves. The results showed that the treatment was significantly different (p < 0.05) from that of K1. The Duncan test showed that all formulations (F1, F2, and F3) were significantly different (p < 0.05) from the control (K1) and from each other. These results indicate that there are significant differences between each variation of the formulation of ethanol extract of Malacca leaves (Table 3). Table 3. Average pH value of the cream.
This test aims to determine each cream formulation’s viscosity using a Brookfield viscometer. The highest viscosity of Malacca cream was found in the Malacca leaf extract formulation of 10% (F1) with a viscosity of 32.279 cP, followed by 15% (F2) with a viscosity of 23.740 cP, and 20% (F3) with a viscosity of 4.744 cP (Table 4). Table 4. Results of the cream viscosity test using a Brookfield viscometer.
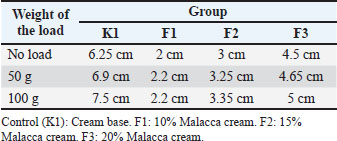
A spreadability test was conducted to determine the cream’s ability to spread in the desired application area and the cream formula’s softness. Table 5 shows that F3 has a wider spreadability compared to cream formulations F1 and F2. Tabel 5. Results of the cream spreadability test.
The examination of the adhesion strength of the cream aims to determine the length of time the cream adheres to the skin surface. The highest average cream adhesion value was found in F3, which was given a concentration of 20% Malacca leaf ethanol extract, while the lowest average cream adhesion value was found in F1, which was given 10% Malacca leaf ethanol extract. ANOVA analysis showed that the treatment was significantly different (p < 0.05) from that of K1. Duncan’s analysis showed that K1 was significantly different (p < 0.05) from F1, F2, and F3, whereas F1 was not significantly different (p > 0.05) from F2 and F3 (Table 6). Table 6. Average value of the cream adhesion test.
Anti-inflammatory testFigure 1 shows the results of the anti-inflammatory effectiveness test of the cream formula using ethanol extract of Malacca leaves.
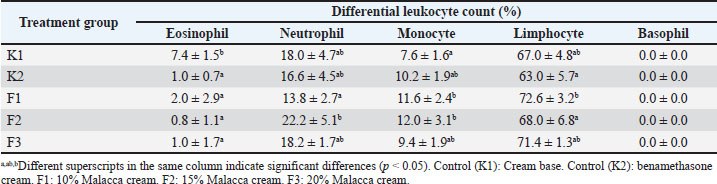
Fig. 1. Changes in paw edema volume over time. The research results indicate that the differences in treatment or days were significantly different (p < 0.05) compared with those in K1. The Duncan test showed that the administration of betamethasone cream (K2) and all formulations (F1, F2, and F3) significantly reduced inflammation (p < 0.05) compared with the control (K1) and all formulations. Thus, increasing the dosage of the treatment resulted in a significant reduction in inflammation (p < 0.05). Similarly, the duration of treatment also showed a significant decrease (p < 0.05). The condition of the mice’s paws before carrageenan induction on the first day and after carrageenan induction on the second day showed the same condition, indicating that the mice’s paws had returned to normal by the second day. A decrease in edema volume occurred in the legs of mice 30 minutes after carrageenan injection and daily administration of a cream containing ethanol extract from Malacca leaves for 4 days. Edema results from the action of inflammatory mediators such as histamine, serotonin, and bradykinin on local inflammation through three phases of edema formation induced by carrageenan (Borsani et al., 2021). Histamine and serotonin are released at 90 minutes in the first phase. The second phase is the release of bradykinin, which occurs 1.5–2.5 hours after induction. Prostaglandin is released in the third phase, 3 hours after induction. Then, the edema develops rapidly and persists at a maximum volume of approximately 5 hours after induction (Mansouri et al., 2015). At the beginning of the observation, the volume of the mouse feet in group K1 and group K2 showed values of 0.16 and 0.128 ml, respectively. Meanwhile, the treatment groups of 10% (F1), 15% (F2), and 20% (F3) Malacca cream obtained the volume of the mouse feet at values of 0.176, 0.256, and 0.180 ml, respectively. On the first day of treatment after carrageenan induction, the volume of edema in all groups of mice increased. In groups K1 and K2, the volume of the mouse feet was obtained at values of 0.256 and 0.22 ml, respectively. Meanwhile, the treatment groups of Malacca leaf extract cream (F1, F2, and F3) obtained the volume of the mouse feet at values of 0.316, 0.276, and 0.184 ml, respectively. On the second day of treatment, group K1 showed a decrease in the volume of edema at a value of 0.208 ml. Group K2 showed a constant edema volume of 0.22 ml. Treatment groups F1 and F2 each showed a decrease in edema volume at 0.22 ml. In contrast, treatment group F3 showed an increase in the edema volume at 0.212 ml. On the third day of treatment, group K1 showed a decrease in edema volume at 0.196 ml. Group K2 also showed a decrease at 0.192 ml. Meanwhile, treatment groups F1, F2, and F3 also experienced a decrease in edema volume at 0.216, 0.168, and 0.14 ml, respectively. On observation on day 4, group K1 showed a decrease in edema volume at 0.144 ml. Group K2 also showed a decrease at 0.160 ml. Likewise, treatment group F1 experienced a decrease in edema volume at 0.16 ml. Meanwhile, group F2 showed a constant value of 0.168 ml and group F3 showed a slight increase in edema volume to 0.148 ml. Leukocyte differentialLeukocyte differential is a dynamic indicator that reflects the effects of anti-inflammatory compounds on the immune system. Changes in the percentage of leukocytes, both granulocyte types (neutrophils, eosinophils, and basophils) and agranulocyte types (lymphocytes and monocytes), can be used to monitor the response to anti-inflammatory therapy. Table 7 shows the results of the calculation of leukocyte differentials in male white mice. Table 7. Differential leukocyte counts of male white mice.
As shown in Table 7, each number of leukocyte cells in male mice that have been given Malaka cream with different concentrations showed varying results. The results of statistical analysis showed a significant difference (p < 0.05) in each leukocyte type, except for basophils, which were not detected in the blood of mice in all treatment groups. EosinophilThe percentage of eosinophils in Table 7 shows that group K1 was significantly different (p < 0.05) compared to other groups (K2, F1, F2, and F3). The high eosinophils in group K1 indicate that the inflammatory process is still ongoing. According to Guyton (1997), eosinophils will increase in the blood in response to parasitic infections, allergies, and inflammation. NeutrophilThe number of neutrophils in the blood increases rapidly during the inflammation process by four to five times the normal number (Hall, 2016). The increase in neutrophils aims to increase the phagocytosis process of damaged or dead cells (Aria et al., 2015). It causes a decrease in neutrophils along with the therapy given. Table 7 shows that the percentage of neutrophils in groups F2 and F3 increased. On the other hand, group K2 showed a significant decrease, even group F1 decreased significantly (p < 0.05). MonocyteThe lowest percentage of monocytes was significantly (p < 0.05) seen in the group given the cream base (K1). This is because the inflammatory process is still occurring, which causes monocytes to concentrate in the area of inflammation so that the percentage is low in the blood. In contrast, the groups treated with K2, F1, F2, and F3 showed suppression of activation and migration of inflammatory cells out of circulation to inflammatory tissue, so the percentage was higher in the blood (Table 7). Monocytes play an important role in digesting damaged and dead cells and phagocytosing bacteria (Guan et al., 2025). LymphocyteThe percentage of lymphocytes in the Malacca cream therapy group (F1, F2, and F3) was higher than that in the K1 group. The increase in the number of lymphocytes in the blood of mice indicates that Malacca leaf ethanol extract cream can stimulate the formation of immunity in mice. An increase in lymphocytes typically coincides with a decrease in neutrophils (Buonacera et al., 2022). BasophilMicroscopic examination of blood smears in each treatment group revealed no basophils. Basophils are easily found in cases of systemic allergic inflammation by releasing histamine and heparin (Voigt and Swist, 2011). Heparin is released by basophils in the area of inflammation to prevent blood clotting and lymph stasis (Nuroini and Faruq, 2018). DiscussionCharacteristics of the Malacca cream formulationThe color, odor, and texture of the cream formula of Malaka Cream did not change after several weeks of room-temperature storage. Thus, the resulting cream formula meets the requirements for good stability during three weeks of storage (Purwaningsih et al., 2020). The decrease in pH in Malaka cream was significant (p < 0.05) (Table 3), which was influenced by the increase in extract concentration. This is because the metabolites in Malacca leaf extract are predominantly acidic, particularly those from the phenolic acid and vitamin C groups (Juwita et al., 2013). However, this high acidity is not ideal for the skin's pH. Skin that is too acidic becomes easily irritated, while skin that is too alkaline becomes dry (Hwang et al., 2022). Likewise, the evaluation of cream viscosity showed a decrease with increasing concentration. Increasing the extract content causes the pH to become more acidic, destabilizing the thickener, disrupting polymer bonds so that the thickener is less hydrated, and competing with the emulsifier. However, the three cream formulations still meet the requirements of SNI 16-43-1996 in the normal cream viscosity in the range 2,000–50,000 cP (Table 4). A high viscosity is a good viscosity; the higher the viscosity, the more stable the particle movement will be (Erwiyani et al., 2018). On the other hand, increasing the extract concentration increases the spreading power. In this study, there was an increase in the spreading power of Malacca cream along with an increase in the extract concentration (Table 5). The structure of the cream base is disrupted by organic acids (vitamin C, phenolic acids) and polar compounds in Malacca leaf extract, resulting in the system becoming more liquid. The higher saponin content reduces the surface tension between the oil-water phase and increases particle mobility, facilitating the spread of the emulsion. The spread of the cream on the skin surface is wider along with the increase in spreading power (Elcistia and Zulkarnain, 2018). Ideally, an increase in spreading power is followed by strong adhesive power so that the skin absorbs more of the active substances released. A good duration for topical formulations is >4 seconds (Ulean et al., 2012). These results show that all Malacca leaf extract cream formulations had low adhesiveness, implying that 15 g stearic acid and 1.5 g TEA (as bioadhesive components) are either incompatible with the extract or ineffective at forming a skin-adhesive matrix. Incorporating bioadhesive polymers (e.g., Carbomer 940) and optimizing pH for skin compatibility may improve adhesion (Zakzak et al., 2024). However, this study will proceed to clinical trials on furry animals such as cats and dogs. Stearic acid (hydrophobic) and TEA (which forms soap with stearic acid) may enhance adhesion by penetrating/binding to hair follicles or interacting with keratin. Meanwhile, the adhesion test follows methods referring to human skin with little to no hair, where mechanical adhesion may be weaker. This aligns with the findings of Youssef et al. (2019) who stated that formulations containing hydrophobic components (e.g., stearic acid) are more effective on animal skin due to interactions with hair follicles/skin lipids. Thus, stearic acid and TEA were used in this study. Anti-inflammatoryTherapy with 10% Malacca cream (F1) showed a consistent reduction in edema volume for 4 days of treatment compared to groups F2 and F3. At a dose of 10%, the anti-inflammatory metabolites contained in Malacca leaves are suspected to reach the saturation point for inflammatory receptors. The dose-effect relationship is non-linear after the receptor saturation point is reached (Peper, 2009). Alves et al. (2013) report showed that the anti-inflammatory effect of ellagic acid reached a maximum at a dose of 10 mg/kg in mice, but a dose of 20 mg/kg did not increase the effect further. The bioactive compounds in P. emblica exhibit anti-inflammatory effects through complementary mechanisms. Gallic acid inhibits COX-2 and LOX enzymes (Sharanya et al., 2023) and suppresses NF-κB expression (Bai et al., 2021), whereas quercetin modulates MAPK and NF-κB pathways (Naso et al., 2025) and reduces COX-2 and iNOS activity (Xiao et al., 2011). Kaempferol decreases NO production by inhibiting iNOS (Chang et al., 2023) and blocks the inflammasome of NLRP3 (Tian et al., 2021). Apigenin inhibits STAT3 and NF-κB inhibition (Ai et al., 2017) and activates Nrf2 (Huang et al., 2023). EGCG inhibits TLR4/NF-κB (Joo et al., 2012) and NLRP3 inflammasome (Zhang et al., 2021), whereas corilagin reduces microglial activation (Liu et al., 2020). Lupeol inhibits NF-κB and STAT3 pathways (Liu et al., 2021), whereas vitamin C functions as an antioxidant (Gęgotek and Skrzydlewska, 2022). Emblicanin A/B demonstrates potent antioxidant activity (Usharani et al., 2013; Prananda et al., 2023). Collectively, these compounds work synergistically through: inhibition of key inflammatory pathways (NF-κB, NLRP3), reducing inflammatory mediators (PGE2, NO, cytokines), enhancing antioxidant defenses, and modulating immune response, making them effective anti-inflammatory therapeutic candidates with favorable safety profiles. Malaka cream administration also affects the differential number of leukocytes in the blood of male white mice. The low percentage of eosinophils in the K2 control group and the Malaka cream group (F1, F2, and F3) is likely because topical administration is concentrated in the area of inflammation. Previous reports (Asmilia et al., 2019) have shown different results, where quite high levels of eosinophils were found in the blood after oral administration of the drug. Topical administration of anti-inflammatory drugs can reduce the number of neutrophils in the blood of mice suffering from inflammation after being induced with carrageenan (Latief et al., 2021; de Siqueira Patriota et al., 2022). Group F1 has a lower percentage of neutrophils than group K1 (Table 7). This indicates that 10% Malacca cream is effective in reducing inflammation by suppressing the production of cytokines that stimulate the bone marrow so that the number of neutrophils decreases (Tsioumpekou et al., 2023). On the other hand, administration of 15% and 20% Malacca leaf extract cream did not increase neutrophils. It is suspected that at a dose of 10%, the saturation point of drug contact with inflammatory receptors has been reached so that increasing the dose does not result in an increase in effect (Tan et al., 2021; Redelmeier and Zipursky, 2023). A slightly different condition was seen in the percentage of monocytes, all groups treated (K2, F1, F2, and F3) showed a significantly higher increase in the number of monocytes (p < 0.05).This is thought to be due to drug therapy being able to suppress the activation and migration of inflammatory cells out of circulation into inflammatory tissue. Similarly, the administration of Malaka cream (F1, F2, and F3) increased the percentage of lymphocytes. Interestingly, group K2 (betamethasone cream administration) actually decreased the number of lymphocytes because this group of steroids is immunosuppressive. Betamethasone suppresses the immune system by directly suppressing T lymphocyte production (through inhibition of proliferation and differentiation of T helper cells (Th1/Th17) and the production of β lymphocytes (Mori et al., 2016; Perna-Barrull et al., 2020). Phyllanthus emblica is generally not immunosuppressive but is known as an immunomodulator that can increase or balance the immune response (Jantan et al., 2019; Nguse et al., 2022). This study provides significant contributions including: evidence of the effectiveness of a topical cream formulation of P. emblica leaf extract with adequate physicochemical stability (pH, viscosity, and spreading ability), though requiring optimization of adhesiveness through cream base modification, and demonstration of anti-inflammatory effects through dual mechanisms, including inhibition of carrageenan-induced edema (optimal at 10% concentration) and modulation of leukocyte differential counts. ConclusionThis study demonstrates that the ethanolic extract cream of P. emblica leaves maintains good stability over a 3-week observation period, despite a reduction in pH and viscosity changes with increasing extract concentration. The 10% formulation exhibited optimal anti-inflammatory effects, reducing edema by 62% (p < 0.01 vs. control) and significantly modulated leukocyte profiles by decreasing neutrophils while increasing monocytes and lymphocytes, in contrast to the immunosuppressive effects of betamethasone. However, the study limitations include preliminary adhesion tests requiring optimization with suitable adhesive components and the fact that the active compounds’ mechanisms of action were inferred from literature sources (NF-κB and COX-2 inhibition) rather than direct molecular analyses. These findings support the potential of P. emblica cream as a topical anti-inflammatory agent. However, further studies are needed to validate the mechanisms in hairy animal models and evaluate long-term stability. AcknowledgmentsThe authors would like to express gratitude to the Faculty of Veterinary Medicine, Universitas Syiah Kuala, for supporting this research and to the members of the Research Laboratory who provided technical assistance in conducting this research. Conflict of interestAll authors declare no conflict of interest. FundingNone. Authors’ contributionsAll authors contributed to the study’s ideas, design, and development. All authors have contributed to this study and have read and approved the final manuscript. Data availabilityAll data are provided in the revised manuscript. ReferencesAi, X.Y., Qin, Y., Liu, H.J., Cui, Z.H., Li, M., Yang, J.H., Zhong, W.L., Liu, Y.R., Chen, S., Sun, T., Zhou, H.G. and Yang, C. 2017. Apigenin inhibits colonic inflammation and tumorigenesis by suppressing STAT3-NF-κB signaling. Oncotarget 8(59), 100216–100226; doi:10.18632/oncotarget.22145 Alves C deF., Angeli, G.N., Favarin, D.C., De Andrade, E.L., Chica, J.E., Faccioli, L.H., Da Silva, P.R. and Rogerio A deP. 2013. Effects of ellagic acid proresolution in an experimental model of allergic airway inflammation. Mediators Inflammation 2013, 863198; doi:10.1155/2013/863198 Aria, M., Arel, V. and Monika. 2015. Anti-inflammatory effect of the piladang leaf fraction (Solenostemons cutellarioides (L.) Codd) on female white mice. Scientia 5(2), 84–91; doi:10.36434/SCIENTIA.V5I2.27 Asmilia, N., Sutriana, A. and Azhari, L. 2019. Efek Antiinflamasi Ekstrak Etanol Daun Malaka (Phyllanthus emblica) Terhadap Differensial Leukosit Pada Mencit Jantan. Jurnal Bioleuser 3(3), 55–58. Bai, J., Zhang, Y., Tang, C., Hou, Y., Ai, X., Chen, X., Zhang, Y., Wang, X. and Meng, X. 2021. Gallic acid: pharmacological activities and molecular mechanisms involved in inflammation-related diseases. Biomed. Pharmacotherapy=Biomedecine & Pharmacotherapie 133, 110985; doi:10.1016/j.biopha.2020.110985 Barnes, T.M., Mijaljica, D., Townley, J.P., Spada, F. and Harrison, I.P. 2021. Vehicles for Drug Delivery and Cosmetic Moisturizers: review and Comparison. Pharmaceutics 13(12), 12012; doi:10.3390/pharmaceutics13122012 Bellwood, B. and Andrasik-Catton, M. 2014. Handbook of laboratory procedures for veterinary technicians. New York: John Wiley & Sons, Inc.. Bindu, S., Mazumder, S. and Bandyopadhyay, U. 2020. Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: a current perspective. J. Surgical Oncol. 33(12), 114147; doi:10.1016/j.bcp.2020.114147 Borsani, B., De Santis, R., Perico, V., Penagini, F., Pendezza, E., Dilillo, D., Bosetti, A., Zuccotti, G.V. and D'Auria, E. 2021. Role of Carrageenan in Inflammatory Bowel Diseases and Allergic Reactions: where Do We Stand?. Nutrients 13(10), 3402; doi:10.3390/nu13103402 Buonacera, A., Stancanelli, B., Colaci, M. and Malatino, L. 2022. Neutrophil-to-lymphocyte ratio: an emerging marker of the relationship between the immune system and diseases. Int. J. Mol. Sci. 23(7), 3636; doi:10.3390/ijms23073636 Chang, S.Y., Ko, Y. and Kim, M.J. 2023. Regulatory mechanisms of kaempferol on iNOS expression in RINm5F β-cells under exposure to interleukin-1β. Heliyon 9(4), e14818; doi:10.1016/j.heliyon.2023.e14818 Charoenteeraboon, J., Ngamkitidechakul, C., Soonthornchareonnon, N., Jaijoy, K. and Sireeratawong, S. 2010. Antioxidant activities of the standardized water extract from fruit of Phyllanthus emblica Linn. Sonklanakarin J. Sci. Technol. 32(6), 599–604; doi:10.14456/sjst-psu.2010.80 Ciaccia, L. 2011. The Fundamentals of Inflammation. Yale J. Biol. Med. 84(1), 64–65; doi:10.3410/B3-64 de Siqueira Patriota, L.L., De Brito Marques Ramos, D., E Silva, M.G., Dos Santos, A.C.L.A., Silva, Y.A., Paiva Pmgp, M.G., Pontual, E.V., De Albuquerque, L.P., Mendes, R.L. and Napoleão, T.H. 2022. Inhibition of carrageenan-induced acute inflammation in mice by Micrograma vacciniifolia frond lectin polymers. Polymers 14(8), 1609; doi:10.3390/polym14081609 Dhale, D.A. and Mogle, U.P. 2011. Phytochemical screening and antibacterial activity of Phyllanthus emblica (L.). Sci. Res. Rep. 1(3), 138–142. Di Rosa, M., Giroud, J.P. and Willoughby, D.A. 1971. Studies on the acute inflammatory response. Involvement of the nervous system in inflammatory responses. J. Pathol. 103(1), 15–29; doi:10.1002/path.1711030103 Ekor, M. 2013. The growing use of herbal medicines: issues related to adverse reactions and safety monitoring challenges. Front. Pharmacol. 4, 177; doi:10.3389/fphar.2013.00177 Elangovan, N.M., Dhanarajan, M.S. and Elangovan, I. 2012. Evaluation of antibacterial and antifungal activity of Phyllanthus emblica leaf extract. Int. Res. J. Pharm. Biosci. 2(2), 59–66; doi:10.22207/JPAM.13.4.11 Elcistia, R. and Zulkarnain, A.K. 2018. Optimization of the o/w cream preparation formula of a combination of oxybenzone and titanium dioxide and its sunscreen activity test in vivo. Pharm. Mag. 14(2), 63–78; doi:10.22146/farmaseutik.v14i2.42596 Elya, B., Dewi, R. and Budiman, M.H. 2013. Antioxidant cream of Solanumlycopersicum L. Int. J. PharmTech. Res. 5(1), 233–238. Erwiyani, A.S., Destianti, D. and Kabelen, S.A. 2018. Effect of storage time on the physical preparation of avocado leaf cream (Persea americana Mill) and betel leaf (Piper bettle Linn). Indonesian J. Pharm. Naturnal. Product. 1(1), 26–27; doi:10.35473/ijpnp.v1i1.31 Guan, R., Wang, Z., Yi, P., Luo, W., Liu, Y., Xie, Z., Liu, Z., Xia, Z., Zhang, H. and Cheng, Q. 2025. Tissue macrophages: origin, heterogeneity, biological functions, diseases, and therapeutic targets Signal transduction and targeted therapy, 10(1), 93; doi: 10.1038/s41392-025-02124-Y Gęgotek, A. and Skrzydlewska, E. 2022. Antioxidative and anti-inflammatory activity of ascorbic acid. Antioxidants (Basel. Switzerland). 1993(10), 11; doi:10.3390/antiox11101993 Golechha, M., Bhatia, J. and Arya, D.S. 2011. Effects of P. emblica extract on inflammatory and oxidative stress markers in a rat model of rheumatoid arthritis. J. Environ. Pathol. Toxicol. Oncol. 30(2), 163–171. Guyton, A.C. 1997. Medical physiology. Jakarta, EGC. Hall, J.E. 2016. Textbook of Medical Physiology. 13th ed., Singapura: Elsevier. Haque, A.F. and Sugihartini, N. 2015. Evaluation of irritation test and physical properties test on cream preparation of clove essential oil with various concentrations. Pharmacology 12(2), 131–139. Holthoon, L. 2011. Phytochemistry, Traditional Uses and Cancer Chemopreventive Activity of Amla (Phyllanthus emblica). J. Appl. Pharm. Sci. MedPlant. 12(1), 388–389. Huang, W., Zhong, Y., Gao, B., Zheng, B. and Liu, Y. 2023. Nrf2-mediated therapeutic effects of dietary flavones in different diseases. Front. Pharmacol. 14, 1240433; doi:10.3389/fphar.2023.1240433 Hwang, J.H., Lee, S., Lee, H.G., Choi, D. and Lim, K.M. 2022. Evaluation of Skin Irritation of Acids Commonly Used in Cleaners in 3D-Reconstructed Human Epidermis Model, KeraSkinTM. Toxics 10(10), 558; doi:10.3390/toxics10100558 Jantan, I., Haque, M.A., Ilangkovan, M. and Arshad, L. 2019. Modulatory Effects and Mechanisms of Action of Phyllanthus species and their bioactive metabolites on the immune system. Front. Pharmacol. 10, 878; doi:10.3389/fphar.2019.00878 Joo, S.Y., Song, Y.A., Park, Y.L., Myung, E., Chung, C.Y., Park, K.J., Cho, S.B., Lee, W.S., Kim, H.S., Rew, J.S., Kim, N.S. and Joo, Y.E. 2012. Epigallocatechin-3-gallate inhibits LPS-induced NF-κB and MAPK Signaling Pathways in Bone Marrow-Derived Macrophages. Gut Liver 6(2), 188–196; doi:10.5009/gnl.2012.6.2.188 Juwita, N.K., Djajadisastra, J. and Azizahwati, A. 2013. Tyrosinase inhibition test and physical stability of whitening cream preparations containing jackfruit bark extract. Pharm. Sci. Mag. 8(3), 127–140; doi:10.7454/psr.v8i2.3477 Kaur, J., Kaur, D., Singh, H. and Khan, M.U. 2013. Emblica officinalis: a meritocratic drug for treating various disorders. Indo Am. J. Pharm. Res. 3(6), 4477–4496; doi:10.30604/jika.v5i1.756 Khopde, S.M., Priyadarsini H Mohan., Gawandi JG Satav., Yakhmi. and Mittal. 2001. Characterizing the antioxidant activity of amla (Phyllanthus emblica) extract. Curr. Sci. India 81(2), 185–190. Lallo, S., Hardiyanti, H. and Suhendi, A. 2020. Evaluation of anti-inflammatory activity using plethysmometer in animal models: a methodological approach. J. Ethnopharmacol. 258, 112876; doi:10.1016/j.jep.2020.112876 Latief, M., Fisesa, A.T., Sari, P.M. and Taringan, I.L. 2021. Anti-inflammatory activity of ethanol extract of sugai leaves (Peronema canescens Jack) in mice induced by carrageenan. J. Pharm. Sci. Pract. 7(2), 144–153. Liu, F.C., Yu, H.P., Chou, A.H., Lee, H.C. and Liao, C.C. 2020. Corilagin reduces acetaminophen-induced hepatotoxicity through the MAPK and NF-κB signaling pathways in a mouse model. Am. J. Transl. Res. 12(9), 5597–5607. Liu, K., Zhang, X., Xie, L., Deng, M., Chen, H., Song, J., Long, J., Li, X. and Luo, J. 2021. Lupeol and its derivatives as anticancer and anti-inflammatory agents: molecular mechanisms and therapeutic efficacy. Pharmacol. Res. 164, 105373; doi:10.1016/j.phrs.2020.105373 Lumentut, N., Tilaar, W. and Rottie, J. 2020. Evaluation of the spreadability and physical stability of herbal cream formulations containing grape seed extract. Int. J. Appl. Pharmaceutics 12(3), 123–129; doi:10.22159/ijap.2020.v12s3.39456 Mansouri, M.T., Hemmati, A.A., Naghizadeh, B., Mard, S.A., Rezaie, A. and Ghorbanzadeh, B. 2015. Mechanisms underlying the anti-inflammatory effect of ellagic acid in carrageenan-induced paw edema in rats. Indian J. Pharmacol. 47(3), 292–298; doi:10.4103/0253-7613.157127 Mori, H., Arita, K., Yamaguchi, T., Hirai, M. and Kurebayashi, Y. 2016. Effects of topical betamethasone application on mice with imiquimod-induced psoriasis-like skin inflammation. Kobe J. Med. Sci. 62(4), E79–E88. Nainggolan, S.I., Rajuddin, R., Kamarlis, R.K., Hambal, M. and Frengki, F. 2025. In silico study of the potential of curcumin and its derivatives for increasing wild-type p53 expression and improving the function of R273H p53 mutant. Vet. World 18(3), 715–730; doi:10.14202/vetworld.2025.715-730 Nainggolan, S.R., Rajuddin, R., Hasanuddin, H., Keumalazia, R., Hambal, M. and Frengki, F. 2023. In silico analysis of anticancer curcumin and its metabolites in increasing paclitaxel effectiveness. Res. J. Pharm. Technol. 16(2), 885–892; doi:10.52711/0974-360X.2023.00150 Naso, M., Trincianti, C., Tosca, M.A. and Ciprandi, G. 2025. Quercetin and its lecithin-based formulation: potential applications for allergic diseases based on a narrative review. Nutrients 17(9), 1476; doi:10.3390/nu17091476 Nguse, M., Yang, Y., Fu, Z., Xu, J., Ma, L. and Bu, D. 2022. Phyllanthus emblica (Amla) fruit powder as a supplement to improve preweaning dairy calves’ health: effect on antioxidant capacity, immune response, and gut bacterial diversity. Biology 11(12), 1753; doi:10.3390/biology11121753 Ntalouka, F. and Tsirivakou, A. 2023. Luteolin: a promising natural agent in management of pain in chronic conditions. Front. Pain Res. 4, 4; doi:10.3389/fpain.2023.1114428 Nuroini, F. and Faruq, Z.H. 2018. Leukocyte count in inflammation of the feet of mice (Mus musculus) induced by carrageenan with white bird's nest (Collocali fuciphaga). Biomedica 11(2), 57–62. Palazzo, E., Rossi. and Maione. 2008. Role of TRPV1 receptors in descending pain modulation. Mol. Cellular Endocrinol. 286(1–2 Suppl 1), S79–S83; doi:10.1016/j.mce.2008.01.013 Parham, S., Kharazi, A.Z., Bakhsheshi-Rad, H.R., Nur, H., Ismail, A.F., Sharif, S., RamaKrishna, S. and Berto, F. 2020. Antioxidant, antimicrobial and antiviral properties of herbal materials. J. Appl. Nutr. 69(12), 69–79; doi:10.3390/antiox9121309 Patel, S.S., Shah, R.S. and Goyal, R.K. 2013. Antihyperglycemic, antihyperlipidemic, and antioxidant effects of Phyllanthus emblica in streptozotocin-induced diabetic rats. J. Clin. Med. Physiol. Pharm. Biol. 51(6), 781–790; doi:10.3109/13880209.2013.764329 Peper, A. 2009. Relationship between drug dose and drug effect. Dose-response. A Publication. Int. Hormesis Soc. 7(2), 172–192; doi:10.2203/dose-response.08-019.Peper Perianayagam, J.B., Sharma, S.K., Joseph, A. and Christina, A.J.M. 2004. Evaluation of the antipyretic and analgesic activities of Emblica officinalis Gaertn. J. Ethnopharmacology 95(1), 83–85; doi:10.1016/j.jep.2004.06.020 Perna-Barrull, D., Gieras, A., Rodriguez-Fernandez, S., Tolosa, E. and Vives-Pi, M. 2020. Immune system remodeling by prenatal betamethasone: effects on β-cells and type 1 diabetes. Front. Endocrinol. 11, 540; doi:10.3389/fendo.2020.00540 Prananda, A.T., Dalimunthe, A., Harahap, U., Simanjuntak, Y., Peronika, E., Karosekali, N.E., Hasibuan, P.A.Z., Syahputra, R.A., Situmorang, P.C. and Nurkolis, F. 2023. Psychiatry, 69, e279–e279. Phyllanthus emblica: a comprehensive review of its phytochemical composition and pharmacological properties. Front. Pharmacol. 14, 1288618. doi: 10.3389/fphar.2023.1288618 Purwaningsih, N.S., Romlah, S.N. and Choirunnisa, A. 2020. Literature review of cream preparation evaluation. Edu Masda J. 4(2), 109–119. Qureshi, S.A., Asad, W. and Sultana, V. 2009. Effect of Phyllanthus emblica Linn on type II diabetes, triglycerides, and liver–specific enzyme. Pak. J. Nutr. 8(2), 125–128; doi:10.3923/pjn.2009.125.128 Rahmat, E., Lee, J. and Kang, Y. 2021. Javanese turmeric (Curcuma xanthorrhiza Roxb.): ethnobotany, phytochemistry, biotechnology, and pharmacological activities. Evid.-Based Complem. Altern. Med. ECAM. 2021, 9960813; doi:10.1155/2021/9960813 Rao, T.P., Okamoto, N. Akita., Hayashi. and Kato-Yasuda. 2011. Acute and subchronic toxicity of Phyllanthus emblica extract in rodents. J. MedFood. 14(7–8), 721–728; doi:10.1089/jmf.2010.1209 Redelmeier, D.A. and Zipursky, J.S. 2023. A dose of reality about dose-response relationships. J. Gen. Internal Med. 38(16), 3604–3609; doi:10.1007/s11606-023-08395-x Rikomah, S.E., Dharmayanti, L. and Sakinah, M.D. 2021. Test of the effectiveness of anti-inflammatory cream type m/a from ethanol extract of kapok leaves (Ceiba pentandra.(L) Gaertn) on male white mice as test animals (Mus musculus. L). Oceana Biomed. J. 4(1), 66–75; doi:10.30649/obj.v4i1.89 Sharanya, C.S., Abhithaj, J., Arun, K.G., Eeda, K.R., Bhat, V., Variyar, E.J., Sabu, A. and Haridas, M. 2023. Lipoxygenase inhibitory synthetic derivatives of methyl gallate regulate COX-2 and cytokine gene expression to reduce arthritis in an animal model. Scientific Rep. 13(1), 10644; doi:10.1038/s41598-023-37613-z Shi, C., Ye, Z., Shao, Z., Fan, B. and et al. 2022. Multidisciplinary guidelines for the rational use of topical non-steroidal anti-inflammatory drugs for musculoskeletal pain (2022). J. Clin. Med. 12(4), 1544; doi:10.3390/jcm12041544 Singh, N, Mathur, C, Sase, N.A., Rai, S. and Abraham, J. 2015. Pharmaceutical properties of Emblica officinalis and Phyllanthus emblica extracts. Res. J. Pharm. Biol. Chem. Sci. 6, 1007–1016. Sumalatha, D. 2013. Antioxidant and antitumor activity of Phyllanthus emblica in colon cancer cell lines. Int. J. Curr. Microbiol. Appl. Sci. 2(5), 189–195; doi:10.1016/j.egyr.2019.08.088 Sundar M, Suresh S, Lingakumar K.. 2022. Preparation and optimization of medicated cold cream using Caralluma adscendens var. attenuata for treating Candida skin infection. Biotechnologia 103(3), 249–260; doi:10.5114/bta.2022.118668 Takeuchi, K. 2012. Pathogenesis of nonsteroidal anti-inflammatory drug-induced gastric damage: importance of cyclooxygenase inhibition and gastric hypermotility. World J. Gastroenterol. 18, 2147–2160; doi:10.3748/wjg.v18.i18.2147 Tan, Y.M., Barton, H.A., Boobis, A., Brunner, R., Clewell, H., Cope, R., Dawson, J., Domoradzki, J., Egeghy, P., Gulati, P., Ingle, B., Kleinstreuer, N., Lowe, K., Lowit, A., Mendez, E., Miller, D., Minucci, J., Nguyen, J., Paini, A., Perron, M. and Embry, M. 2021. Opportunities and challenges related to toxicokinetic process saturation: implications for risk assessment. Regulatory Toxicol. Pharmacol. RTP. 127, 105070; doi:10.1016/j.yrtph.2021.105070 Tania, B.L., Dwiastuti, R. and Lestari ABS Setyaningsih. 2022. Sunscreen cream formulation of noni leaf extract (Morinda citrifolia L.) with tween 80 and lecithin emulsifier. J. Farmasi Dan Ilmu Kefarm. Indonesia 9(3), 262–271; doi:10.20473/jfiki.v9i32022.262-271 Tian, H., Lin, S., Wu, J., Ma, M., Yu, J., Zeng, Y., Liu, Q., Chen, L. and Xu, J. 2021. Kaempferol alleviates corneal transplant rejection by inhibiting NLRP3 inflammasome activation and macrophage M1 polarization by promoting autophagy. Exp. Eye Res. 208, 108627; doi:10.1016/j.exer.2021.108627 Tsioumpekou, M., Krijgsman, D., Leusen, J.H.W. and Olofsen, P.A. 2023. Role of cytokines in neutrophil development, tissue homing, function, and plasticity in health and disease. Cells 12(15), 1981; doi:10.3390/cells12151981 Ulean, S., Banne, Y. and Suatan, R. 2012. Pembuatan salep anti jerawat dari ekstrak rimpang temulawak (C. xanthorriza Roxb). J. Ilmiah Farmasi 3(2), 45–49. Usharani, P., Fatima, N. and Muralidhar, N. 2013. Effects of Phyllanthus emblica extract on endothelial dysfunction and biomarkers of oxidative stress in patients with type 2 diabetes mellitus: a randomized, double-blind, controlled study. Diabetes Metabolic Syndr. Obesity. Targets Ther. 6, 275–284; doi:10.2147/DMSO.S46341 Voigt, G.L. and Swist, S.L. 2011. Hematologic techniques and concepts for veterinarians Edisi 2. London, UK: John Wiley & Sons, Inc. Welz, A.N., Emberger-Klein, A. and Menrad, K. 2018. Why people use herbal medicine: insights from a focus-group study in Germany. BMC Complement. Alternat. Med. 18(1), 92; doi:10.1186/s12906-018-2160-6 Winter, C.A., Risley, E.A. and Nuss, G.W. 1962. Carrageenan-induced edema in hind paw of the rat as an assay for anti-inflammatory drugs. Proc. Soc. For Exp. Biol. Med. 111(3), 544–547; doi:10.3181/00379727-111-27849 Xiao, X., Shi, D., Liu, L., Wang, J., Xie, X., Kang, T. and Deng, W. 2011. Quercetin suppresses cyclooxygenase-2 expression and angiogenesis through inactivation of P300 signaling. PLos One 6(8), e22934; doi:10.1371/journal.pone.0022934 Yacobus, A.R., Lau, S.H.A. and Syawal, H. 2019. Formulasi dan uji stabilitas krim ekstrak ethanol daun beluntas (Pluchea indica L.) dari Kota Benteng Kabupaten Kepulauan Selayar Provinsi Sulawesi Selatan. J. Farmasi Sandi Karsa 5(1), 19–25; doi:10.36060/jfs.v5i1.44 Yokozawa T Kim., Kim, H.Y., Tanaka, H.J., Sugino, T., Okubo, H. and Chu, D.C. 2007. Amla (Phyllanthus emblica L.) attenuates oxidative stress-induced age-related renal dysfunction. J. Agricult. Food Chem. 55(19), 7744–7752; doi:10.1021/jf072105s Youssef, F.S., El-Banna, H.A., Elzorba, H.Y. and Galal, A.M. 2019. Application of nanoparticles in veterinary medicine. Int. J. Vet. Sci. Med. 7(1), 78–93; doi:10.1080/23144599.2019.1691379 Zakzak K Semenescu., Moac, A.D., ă EA Predescu., Dr, I., ăghici G Vlaia., Vlaia, L., Borcan, V. and Dehelean, C.A. 2024. Comprehensive Biosafety Profile of Carbomer-Based Hydrogel Formulations Including Phosphorus Derivatives Gels. (Basel. Switzerland). 10(7), 477; doi:10.3390/gels10070477 Zhang, C., Li, X., Hu, X., Xu, Q., Zhang, Y., Liu, H., Diao, Y., Zhang, X., Li, L., Yu, J., Yin, H. and Peng, J. 2021. Epigallocatechin-3-gallate prevents inflammation and diabetes -induced glucose tolerance through inhibition of NLRP3 inflammasome activation. J. Clin. Pharmacol. Pharmacol. Int. Immunopharmacol. 93, 107412; doi:10.1016/j.intimp.2021.107412 | ||
| How to Cite this Article |
| Pubmed Style Asmilia N, Sutriana A, Vanda H, Frengki F, Manan MQ, Reyno DS, Ikhzan MRA. Topical anti-inflammatory potential of ethanol extract of Phyllanthus emblica leaves in a carrageenan-induced mouse model. Open Vet. J.. 2025; 15(10): 4972-4983. doi:10.5455/OVJ.2025.v15.i10.16 Web Style Asmilia N, Sutriana A, Vanda H, Frengki F, Manan MQ, Reyno DS, Ikhzan MRA. Topical anti-inflammatory potential of ethanol extract of Phyllanthus emblica leaves in a carrageenan-induced mouse model. https://www.openveterinaryjournal.com/?mno=264422 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.16 AMA (American Medical Association) Style Asmilia N, Sutriana A, Vanda H, Frengki F, Manan MQ, Reyno DS, Ikhzan MRA. Topical anti-inflammatory potential of ethanol extract of Phyllanthus emblica leaves in a carrageenan-induced mouse model. Open Vet. J.. 2025; 15(10): 4972-4983. doi:10.5455/OVJ.2025.v15.i10.16 Vancouver/ICMJE Style Asmilia N, Sutriana A, Vanda H, Frengki F, Manan MQ, Reyno DS, Ikhzan MRA. Topical anti-inflammatory potential of ethanol extract of Phyllanthus emblica leaves in a carrageenan-induced mouse model. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4972-4983. doi:10.5455/OVJ.2025.v15.i10.16 Harvard Style Asmilia, N., Sutriana, . A., Vanda, . H., Frengki, . F., Manan, . M. Q., Reyno, . D. S. & Ikhzan, . M. R. A. (2025) Topical anti-inflammatory potential of ethanol extract of Phyllanthus emblica leaves in a carrageenan-induced mouse model. Open Vet. J., 15 (10), 4972-4983. doi:10.5455/OVJ.2025.v15.i10.16 Turabian Style Asmilia, Nuzul, Amalia Sutriana, Henni Vanda, Frengki Frengki, Mazayya Qanita Manan, Dwi Salsha Reyno, and Muhammad Rifqi Al Ikhzan. 2025. Topical anti-inflammatory potential of ethanol extract of Phyllanthus emblica leaves in a carrageenan-induced mouse model. Open Veterinary Journal, 15 (10), 4972-4983. doi:10.5455/OVJ.2025.v15.i10.16 Chicago Style Asmilia, Nuzul, Amalia Sutriana, Henni Vanda, Frengki Frengki, Mazayya Qanita Manan, Dwi Salsha Reyno, and Muhammad Rifqi Al Ikhzan. "Topical anti-inflammatory potential of ethanol extract of Phyllanthus emblica leaves in a carrageenan-induced mouse model." Open Veterinary Journal 15 (2025), 4972-4983. doi:10.5455/OVJ.2025.v15.i10.16 MLA (The Modern Language Association) Style Asmilia, Nuzul, Amalia Sutriana, Henni Vanda, Frengki Frengki, Mazayya Qanita Manan, Dwi Salsha Reyno, and Muhammad Rifqi Al Ikhzan. "Topical anti-inflammatory potential of ethanol extract of Phyllanthus emblica leaves in a carrageenan-induced mouse model." Open Veterinary Journal 15.10 (2025), 4972-4983. Print. doi:10.5455/OVJ.2025.v15.i10.16 APA (American Psychological Association) Style Asmilia, N., Sutriana, . A., Vanda, . H., Frengki, . F., Manan, . M. Q., Reyno, . D. S. & Ikhzan, . M. R. A. (2025) Topical anti-inflammatory potential of ethanol extract of Phyllanthus emblica leaves in a carrageenan-induced mouse model. Open Veterinary Journal, 15 (10), 4972-4983. doi:10.5455/OVJ.2025.v15.i10.16 |