| Research Article | ||
Open Vet. J.. 2025; 15(10): 4949-4961 Open Veterinary Journal, (2025), Vol. 15(10): 4949-4961 Research Article Molecular detection of Anaplasma and Candidatus Rickettsia jingxinensis infections in domestic cattle and goats in Dejiang County, Guizhou Province, ChinaJiashun Yu1, Xiaopeng Yang2, Yisong Dai2,3, Jixia Tang2, Qiu Chen4, Jiao Meng3, Mingjiang Tao5, Xingxing Chen3, Chaomin Zhou3, Jiafu Jiang6, Wuchun Cao6, Fuxun Yu3, Kun Cai1 and Lin Zhan1,2,3,4*1Guizhou University of Traditional Chinese Medicine, Guiyang, China 2School of Public Health, The Key Laboratory of Environmental Pollution Monitoring and Disease Control, Ministry of Education, Guizhou Medical University, Guiyang, China 3NHC Key Laboratory of Pulmonary Immune-related Diseases, Guizhou Provincial People’s Hospital, Guiyang, China 4School of Public Health, Zunyi Medical University, Zunyi, China 5Epidemic Disease Prevention and Control Center of Dejiang County Department of Agriculture and Rural Affairs, Dejiang, China 6Beijing Institute of Microbiology and Epidemiology, Academy of Military Medical Sciences, Beijing, China *Corresponding Author: Lin Zhan. Guizhou Provincial People’s Hospital, Guiyang, China. Email: zhanlin300 [at] hotmail.com Submitted: 16/06/2025 Revised: 15/09/2025 Accepted: 30/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
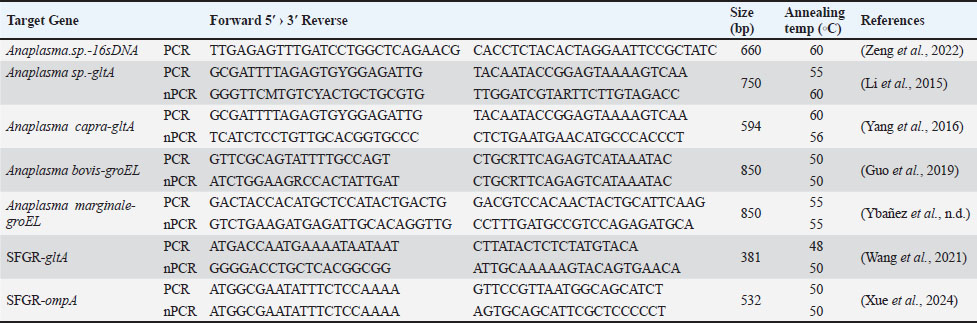
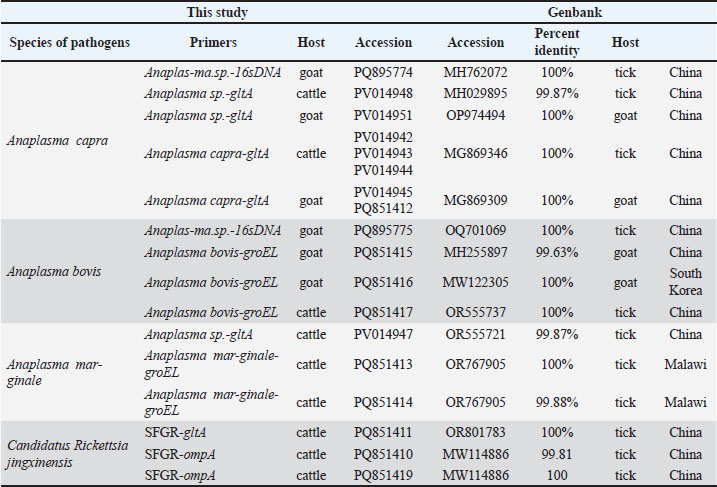
AbstractBackground: Anaplasma spp. and Candidatus Rickettsia jingxinensis have significant tick-borne zoonotic potential and cause diseases in both humans and domestic animals. Nevertheless, epidemiological data regarding their prevalence in cattle and goats within Dejiang County, China, are still limited. Understanding the local infections of these pathogens in domestic animals is crucial for assessing their potential spillover risk and informing public health interventions. Aim: This study aimed to determine the prevalence of Anaplasma and Candidatus R. jingxinensis in cattle and goats from Dejiang County, China, and assess the current infection rates and potential public health risks associated with these pathogens. Methods: A total of 170 domestic animal samples were collected. Following DNA extraction, polymerase chain reaction was employed to amplify pathogen-specific gene fragments of Anaplasma spp. and Candidatus R. jingxinensis, confirming their presence and constructing phylogenetic trees of the detected genotypes. Results: In this study, 35 out of 119 cattle were infected with Anaplasma, of which three species, including A. capra, A. bovis, and A. marginale, were identified with infection rates of 9.24% (95% CI:5.24%–15.80%,17.65%(95% CI: 11.84%–25.48%), and 26.89% (95% CI: 19.74%–35.49%), respectively. A total of 48 goats (82.35%, 69.75%–90.43%) were infected by Anaplasma, of which two species, A. capra and A. bovis, were identified with infection rates of 62.75% (95% CI: 49.03%–74.68% and 66.67% (95% CI: 52.97%–78.03%), respectively. No goats were infected with A. marginale. Furthermore, 4 (3.36%, 1.31%–8.32%) cattle were infected with Ca. R. jingxinensis. This is the first time that molecular evidence of this organism has been detected in cattle. Notably, double and triple infections with these agents were found in cattle, and coinfections were detected in goats. Conclusion: Anaplasmosis is highly prevalent in local ruminants, particularly in goats. The pathogen species distribution differs between hosts, with A. marginale showing host specificity for cattle. The detection of Ca. R. jingxinensis in cattle suggests an expansion of its host range. These findings highlight the need for enhanced surveillance and control measures for these pathogens, especially considering the potential spillover risks and public health threat. Keywords: Dejiang, Tick-borne zoonotic pathogens, PCR, Cattle, Goat. IntroductionAnaplasma and Spotted Fever Group Rickettsiae (SFGR) are key members of the Rickettsiae group, primarily transmitted by hard ticks to animal hosts. Infections caused by these pathogens can lead to rickettsial diseases, which have substantial implications for both veterinary and public health (Rar and Golovljova, 2011; Salje, 2021). Anaplasma refers to a group of small, highly diverse, intracellular, gram-negative bacteria that primarily parasitize eukaryotic cell cytoplasm, forming mulberry-like inclusions (Rar et al., 2021). At least seven species of Anaplasma, including A. phagocytophilum, A. bovis, A. marginale, A. centrale, A. platys, A. ovis, and A. capra, are recognized as causative agents of anaplasmosis in hosts. Five of these species (A. phagocytophilum, A. bovis, A. ovis, A. capra, and A. platys) can cause disease in humans (Akwongo and Byaruhanga, 2024). Anaplasma infections are highly infectious to ruminant livestock, such as cattle and sheep, potentially leading to anaplasmosis. The presence of anaplasmosis within a herd can result in a 20%–30% loss in body weight, abortion, or even maternal death, as well as a 30% increase in natural culling rate of the herd, causing significant economic losses (Goodger et al., 1979). The financial burden of Anaplasma infections on cattle farms has been estimated at $352.70 to $727.70 per cow (Akwongo and Byaruhanga, 2024). In the United States, the economic impact of Anaplasma infections on California’s livestock industry has been estimated at $11 million (Goodger et al., 1979). Notably, anaplasmosis in humans often presents with mild or nonspecific symptoms, which can lead to misdiagnosis as other infectious diseases (Sanchez et al., 2016), frequently resulting in the disease being overlooked. However, an increasing number of cases have been reported in countries such as China, South Korea, Japan, Poland, and Belgium (Yang et al., 2017; Wang et al., 2020; Schudel et al., 2024). Therefore, clinicians and veterinarians working in endemic areas should remain vigilant in recognizing and managing this disease. Candidatus Rickettsia jingxinensis is a newly discovered SFGR species with potential zoonotic risks. It was first identified in 2016 in Haemaphysalis longicornis ticks (Liu et al., 2016). Research on Ca. R. jingxinensis has involved a wide range of hosts, including humans, companion animals (e.g., dogs and cats), and pangolins. In contrast, reports of Ca. R. jingxinensis in domesticated, economically important ruminants (e.g., cattle and goats) are scarce. Given the potential health risks to ruminant livestock and the associated agricultural losses, continued surveillance and focused investigations are necessary. Dejiang County, located in the northeastern part of Guizhou Province in southwestern China, lies at the confluence of the Wuling and Daloushan Mountain Ranges. The region possesses a diverse and resource-rich ecosystem, providing an ideal environment for cattle and goat farming. According to data from the People’s Government of Dejiang County, by the end of 2024, there were 65 livestock farms in the county, each with more than 100 cattle. Additionally, the number of cattle-raising households reached 43,800. The total cattle inventory is expected to be 175,600 head, with 61,400 head slaughtered annually, generating an annual output value exceeding $ 209 million (Dejiang County People’s Government Portal, 2024). Previous studies have reported that Guizhou Province is home to at least 19 tick species across 5 genera and 2 families. Haemaphysalis longicornis and Rhipicephalus microplus are the predominant tick species in the region and are recognized vectors of Anaplasma spp. and Ca. R. jingxinensis, which can be transmitted to livestock through tick bites (Shen and Shen, 1997; Liu and Guo, 2011. These ticks thrive in the region's subtropical climate, dense vegetation, and the presence of both domestic and wild animal hosts—conditions that create an optimal environment for tick survival, reproduction, and pathogen transmission. However, epidemiological data on Anaplasma and Ca. R. jingxinensis infections in domestic ruminants, particularly cattle and goats, in Dejiang County are still lacking. Given the potential zoonotic risks posed by these pathogens, this study aims to assess their prevalence in local livestock by collecting and analyzing blood samples from cattle and goats at designated monitoring sites. Materials and MethodsSample sourcesIn May 2024, with the assistance of the Dejiang County Bureau of Agriculture and Rural Affairs, a convenience sampling approach was used to collect anticoagulated whole blood samples (5 ml per animal) from clinically healthy goats (Capra hircus) and cattle (Bos taurus) on local farms. Blood was obtained from the jugular vein of goats and the tail vein of cattle using ethylenediaminetetraacetic acid-treated vacuum tubes. All samples were stored at 4°C and transported to the laboratory on the same day, and genomic DNA was extracted within 24 hours. Genomic DNA extractionNucleic acids were extracted from 200 μl of blood using the qEx-DNA/RNA Virus Kit (Xi’an Tianlong Science and Technology, Xi’an, China) according to the manufacturer’s instructions and then stored in a refrigerator at −20°C until PCR. Detection of Anaplasma and SFGRAnaplasma spp. amplification was first performed using universal primers targeting the 16S rDNA and gltA genes. Subsequent amplifications used species-specific primers for individual Anaplasma species. To detect Ca. R. jingxinensis, the SFGR ompA and gltA genes were amplified. Table 1 provides a complete list of the primers used in this study. Anaplasma and Ca. R. jingxinensis positive samples obtained from our previous study were used as positive controls, and sterile double-distilled water was used as negative controls. PCR amplification was performed in a 25 µl reaction mixture containing 2.5 µl of 10 × PCR buffer, 2 µl of a 2.5 mM dNTP mixture, 0.5 µl of each primer (10 µM), 0.125 µl of Taq polymerase (5 units/µl) (Takara Biotechnology, China), and 1 µl of template DNA (40–60ng/µl). The final volume was adjusted to 25 µl with double-distilled water. Table 1. Anaplasma and Ca. R. jingxinensis primers.
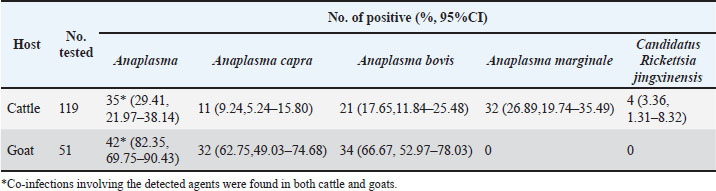
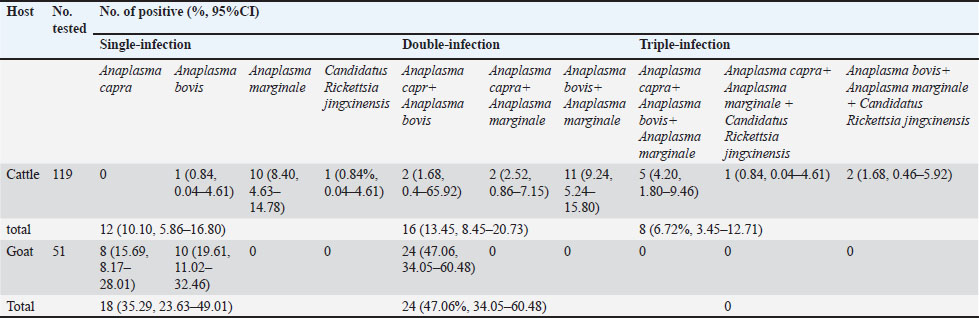
Genetic and evolutionary analysisThe PCR products were electrophoresed on a 1.5% agarose gel, and the UV imaging system was used to visualize the bands. If more than 10 positive bands were detected, 50% were randomly selected and sent to Chongqing BGI for Sanger sequencing. If fewer than 5 positive bands were detected, all were subjected to sequencing. The obtained sequences were compared with gene sequences registered in the GenBank database using the BLAST tool from the National Center for Biotechnology Information (NCBI). Representative sequences were selected, and a phylogenetic tree was constructed using the neighbor-joining method with 1,000 bootstrap replicates in MEGA 11 software. Statistical analysisCount data are presented as numbers (%, 95% CI). The 95% confidence intervals (95% CI) were calculated using the Wilson score method, which provides more accurate interval estimates, especially for small sample sizes or proportions close to 0 or 1. The chi-square test (χ²) was used to compare differences in pathogen positivity rates between and within the cattle and goat groups. Fisher’s exact test was applied when the chi-square test assumptions were not met. A significance level of 0.05 was used for all analyses. Statistical analyses were conducted using the SPSS 27 software. Ethical approvalThe Animal Care Welfare Committee of Guizhou Medical University approved this study (Ethical Approval No. 2305072). All animals were handled in accordance with the Animal Ethics Procedures and Guidelines of the People’s Republic of China. ResultsRates of Anaplasma and SFGR infectionsOut of the 119 cattle screened by PCR, 35 (29.41%, 95% CI: 21.97%–38.14%) were infected with Anaplasma and 4 (3.36%, 1.31%–8.32%) were infected with Ca. R. jingxinensis. Sequencing identified the genotypes of Anaplasma: A. capra in 11 cattle (9.24%, 95% CI: 5.24%–15.80%), A. bovis in 21 cattle (17.64%, 95% CI: 11.84%–25.48%) and A. marginale in 32 cattle (26.89%, 95% CI: 19.74%–35.49%) (Table 2). Among the infected cattle, 12 (10.10%, 95% CI: 5.86%–16.80%) were infected with a single pathogen, 16 (13.45%, 95% CI: 8.45%–20.73%) were co-infected with two pathogens, and 8 (6.72%, 95% CI: 3.45%–12.71%) were infected with three pathogens (Table 3). The infection rate of A. marginale was significantly higher than that of A. capra (χ2=12.52, p < 0.001), whereas no significant differences were observed between A. marginale and A. bovis (χ2=2.94, p=0.087) or between A. bovis and A. capra (χ2=3.61, p=0.057). Table 2. Overall Prevalence of Anaplasma and Ca. R. jingxinensis in cattle and goat.
Table 3. Composition of single infection and co-infection pathogens.
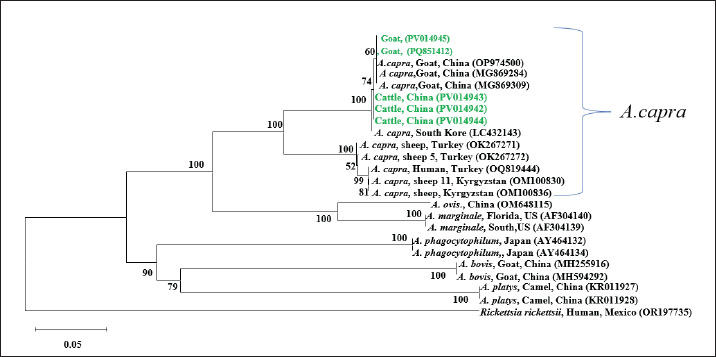
Among the 51 goats, 42 (82.35%, 95% CI: 69.75%–90.43%) were infected with Anaplasma, a rate that was significantly higher than that in cattle (χ2=40.38, p < 0.001). No Ca. R. jingxinensis was detected in the goats. Specifically, 32 (62.75%; 95% CI: 49.03%–74.68%) goats were infected with A. capra and 34 (66.67%; 95% CI: 52.97%–78.03%) with A. bovis (Table 2). Among the infected goats, 18 (35.29%; 95% CI: 23.63%–49.01%) were infected with a single pathogen, and 24 (47.06%; 95% CI: 34.05%–60.48%) were co-infected with both pathogens (Table 3). No significant difference was observed between the infection rates of A. capra and A. bovis in goats (χ2=0.17, p=0.68). However, the infection rates of both A. capra (cattle vs. goat, χ2=54.08, p < 0.001) and A. bovis (cattle vs. goat, χ2=39.20, p < 0.001) were significantly higher in goats than in cattle. Infection pathogen compositionAmong the single-infected cattle, A. marginale was the most prevalent (10/119, 8.40%, 95% CI: 4.63%–14.78%), followed by A. bovis and Ca. R. jingxinensis (1/119, 0.84%, 95% CI: 0.04%–4.61%). No cattle were found to be singly infected with A. capra. Regarding double infections, the most common combination was A. marginale + A. bovis (11/119, 9.24%, 95% CI: 5.24%–15.80%), followed by A. marginale + A. capra (3/119, 2.52%, 95% CI: 0.86%–7.15%) and A. bovis + A. capra (2/119, 1.68%, 95% CI: 0.46%–5.92%). Triple infections occurred most frequently with A. capra + A. bovis + A. marginale (5/119, 4.20%, 95% CI: 1.80%–9.46%), followed by A. bovis + A. marginale + Ca. R. jingxinensis (2/119, 1.68%, 95% CI: 0.46%–5.92%) and A. capra + A. marginale + Ca. R. jingxinensis (1/119, 0.84%, 95% CI: 0.04%–4.61%) (Table 3). In goats, single infections with A. capra (8/51, 15.69%, 95% CI: 8.17%–28.01%) and A. bovis (10/51, 19.61%, 95% CI: 11.02%–32.46%) were observed. In addition, only one mixed infection involving A. capra +A. bovis (24/51, 47.06%, 95% CI: 34.05%–60.48%) was detected (Table 3). Phylogenetic analysisThe sequences (Table S1) were compared to previously published data in the NCBI GenBank, and different sequences were selected to construct the phylogenetic tree. The gltA gene (PQ895774, PV014942–PV014945, PV014948–PV014951) and 16S rDNA sequence (PQ895774) showed 99.87%–100% sequence identity with A. capra (S1). Phylogenetic analysis showed that the sequences from cattle (PV014942–PV014944) and goat (PV014945, PQ851412) were closely related to those from Chinese goats (A. capra strains OP97450, MG869284, and MG869309), although the cattle-derived sequences formed a distinct subclade within this group. In contrast, all sequences remained genetically distinct from the human pathogenic strain from Turkey (OQ819444) (Fig. 1).
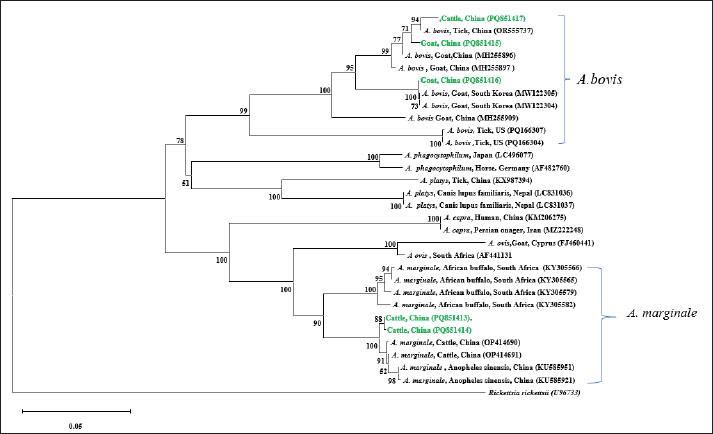
Fig. 1. Phylogenetic tree constructed based on A. capra gltA (594bp). The tree was constructed using the Neighbor-Joining method in MEGA 11, with 1000 bootstrap replicates. Rickettsia rickettsii (OR197735) was used as the outgroup. The sequences obtained in this study are highlighted in green in the figure. The groEL gene sequences (PQ851415, PQ851416, and PQ851417) and 16S rDNA sequence (PQ895775) shared 99.63%–100% identity with reference A. bovis sequences available in GenBank (S1). Phylogenetic analysis based on groEL sequences divided A. bovis into three clusters. The goat-derived sequence PQ851416 was grouped with isolates from South Korean black goats (MW122304, MW122305), while the cattle-derived sequence PQ851417 clustered with HbYS56 (OR555737), isolated from the Chinese H. longicornis. The goat-derived PQ851415 sequence was more closely related to PQ851417, forming a distinct branching node with it (Fig. 2).
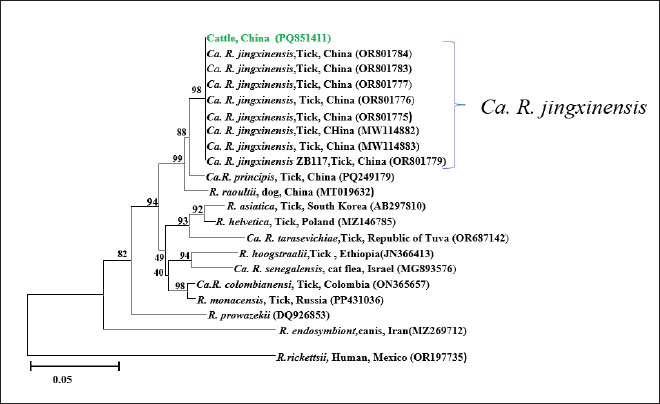
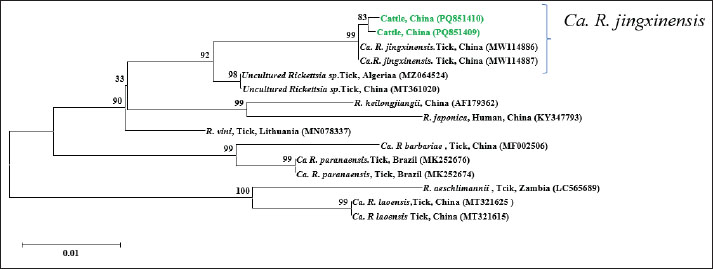
Fig. 2. Phylogenetic tree constructed based on Anaplasma groEL (850bp). The tree was constructed using the Neighbor-Joining method in MEGA 11, with 1,000 bootstrap replicates. Rickettsia rickettsii (U96733) was used as the outgroup. The sequences obtained in this study are highlighted in green in the figure. The groEL (PQ851413, PQ851414) and gltA (PV014947) sequences of A. marginale exhibited 99.87%–100% identity with the reference sequences (S1). In the phylogenetic tree, both PQ851413 and PQ851414 clustered with A. marginale isolates from Chinese cattle (OP414690, OP414691), indicating a close genetic relationship (Fig. 2). The SFGR gltA sequence (PQ851411) showed 100% identity with Ca. R. jingxinensis isolate BJ5 (OR801783), obtained from ticks in Guizhou, China. The SFGR ompA sequences (PQ851410 and PQ851409) showed 99.81% and 100% identity with Ca. R. jingxinensis isolate Hl2 (MW114886) from southwest China, respectively. Phylogenetic trees were constructed separately based on the gltA and ompA gene sequences. In the gltA-based tree, PQ851411 clustered with Ca. R. jingxinensis isolates from Guizhou (OR801775, OR801776, OR801777, OR801779, OR801783, OR801784) and southwest China (MW114882, MW114883), indicating a high degree of genetic relatedness (Fig. 3). In the ompA-based phylogenetic tree, PQ851410 and PQ851409 formed a distinct cluster but shared a branch with Ca. R. jingxinensis isolates from ticks in southwest China (MW114886, MW114887), supported by a high bootstrap value (Fig. 4).
Fig. 3. Phylogenetic tree constructed based on SFGR gltA (381bp). The tree was constructed using the Neigh-bor-Joining method in MEGA 11, with 1,000 bootstrap replicates. The sequences obtained in this study are highlighted in green in the figure.
Fig. 4. Phylogenetic tree constructed based on SFGR ompA (532bp). The tree was constructed using the Neigh-bor-Joining method in MEGA 11, with 1,000 bootstrap replicates. The sequences obtained in this study are highlighted in green in the figure. DiscussionIn this study, we screened cattle and goats from the Dejiang region for Anaplasma spp. and Ca. R. jingxinensis, identifying several important Anaplasma species and providing the first global molecular evidence of Ca. R. jingxinensis infection in cattle. These findings highlight the need for sustained surveillance of Anaplasma and SFGR in local ruminant populations to prevent the emergence and spread of tick-borne diseases, which pose risks to both animal and human health. In this study, the overall infection rate of Anaplasma in cattle (29.41%) was found to be higher than previously reported across 12 provinces in China (13.2%) (Qiu et al., 2016). The overall infection rate in local goats (82.35%) was also markedly higher than that reported in Anhui (14.7%) (Wu et al., 2024), and Hainan (30.1%), but was consistent with a previously reported prevalence in Guizhou goats (79.31%) (Han et al., 2024). This regionally elevated prevalence may be attributed to the high abundance of R. microplus and H. longicornis in the region. Previous studies have shown that cattle and goats in this area are frequently infested with these tick species, facilitating Anaplasma transmission (Wang et al., 2021). Among the identified Anaplasma species, A. capra and A. bovis, goats exhibited significantly higher infection rates than cattle. Previous studies have not identified any host preference between these two pathogens in cattle and goats (Chimnoi et al., 2025), which may be related to local animal migration patterns and management practices. Locally, goats are typically grazed in the mountains for extended periods, resulting in a more complex range of activities, time, and habitats. In contrast, cattle generally graze on grasslands around farms, and their grazing time is shorter than that of goats because some are supplemented with artificial feeding. This difference in grazing behavior increases the likelihood of goats coming into contact with blood-sucking tick vectors. Anaplasma capra is a recently discovered zoonotic pathogen in China (Seo et al., 2020). It has been reported in several provinces across China, including Gansu, Guangxi, Guizhou, Chongqing, Shaanxi, Henan, Shandong, Hubei, Sichuan, Guangdong, Liaoning, Hainan, and the Qinghai-Tibet Plateau (Yang et al., 2017; Altay et al., 2024; Ma et al., 2024; Tang et al., 2024; Xue et al., 2024). Additionally, A. capra has been detected in various countries worldwide, including the Republic of Angola, Brazil, Kyrgyzstan, Greece, France, Sweden, Spain, South Korea, and Turkey (Calchi et al., 2020; Barradas et al., 2021; Jouglin et al., 2022; Remesar et al., 2022; Saratsis et al., 2022; Lysholm et al., 2023; Seo et al., 2023; Addo et al., 2024; Kim et al., 2024; Oguz et al., 2024). Anaplasma capra is transmitted by various species of hard ticks, which are widely distributed and can infect zoonotic hosts and domestic animals (Peng et al., 2018). It has been detected in 14%–18% of small ruminants, such as goats and sheep (Yang et al., 2017; Peng et al., 2018), and has also been reported to infect dogs, water buffalo, and cattle (Sahin et al., 2022). The gltA gene of A. capra appears to be particularly susceptible to variation driven by host factors. In this study, A. capra strains derived from cattle clustered into a distinct subclade, a phenomenon previously observed in other studies (Peng et al., 2018; Peng et al., 2021). However, the mechanisms underlying this variation and their potential biological impacts on the host require further investigation for a clearer understanding. In 2015, human cases of A. capra infection were first reported in China (Li et al., 2015). Infected individuals may present with symptoms such as fever, headache, malaise, dizziness, chills, nausea, vomiting, and diarrhea, as well as abnormal laboratory findings, including elevated liver transaminase levels, leukopenia, and thrombocytopenia (Altay et al., 2024). Although A. capra isolated in this study is distantly related to human isolates in the phylogenetic tree, there is currently no evidence to exclude its potential to infect humans. The potential risk of cross-species transmission should not be overlooked. Cattle and goats are primary reservoir hosts of A. bovis (Atif, 2016). Previous studies have reported A. bovis infection rates in animal hosts in China ranging from 4.5% to 49% (Guo et al., 2020), with infection rates in cattle in Japan (53%) (Ooshiro et al., 2008), South Korea (18.2%), and goats (0.5%) (Miranda et al., 2021). In this study, the infection rate of A. bovis in cattle (17.64%) falls within this range, although it is notably lower than that in goats (66.67%). Phylogenetic analysis revealed regional differences in A. bovis, suggesting the presence of distinct circulating strains in the area. We hypothesize that this diversity may be linked to the region’s intensive livestock trade, which hosts the largest cattle and goat trading market in northeastern Guizhou. The movement of animals likely introduces ticks carrying diverse A. bovis strains from different regions. Consequently, it is crucial to enhance public awareness of tick-borne pathogen control in livestock trade markets. Further epidemiological research is required to explore the relationship between livestock trade, strain transmission, and A. bovis genetic diversity. A. bovis has been identified as a zoonotic pathogen, with reported cases in regions such as Anhui, Jiangxi, and Ganzhou in China, as well as in the United States (Karpathy et al., 2023; Lu et al., 2019). Although most animals infected with A. bovis show no clinical symptoms, human infections may present with fever (up to 39°C), chills, headache, myalgia, anorexia, rash, cold sweats, diarrhea, thrombocytopenia, and lymphadenopathy (Tang et al., 2024). This study detected A. marginale exclusively in cattle but not in goats, which could be attributed to the high host specificity of A. marginale for cattle (Kocan et al., 2010). Although A. marginale is not considered pathogenic to humans, it remains the most prevalent and dangerous bovine anaplasmosis pathogen, causing significant morbidity and mortality in cattle worldwide (Kocan et al., 2010; El-Alfy et al., 2023). Therefore, the potential health risks associated with A. marginale in cattle should not be underestimated. Our study also identified 4 cattle infected with Ca. R. jingxinensis. To the best of our knowledge, this study provides the first molecular evidence of Ca. R. jingxinensis detection in cattle worldwide. This finding suggests a potential expansion of its host range; however, further experimental and serological studies are necessary to determine whether cattle are competent hosts and to elucidate its transmission dynamics. Ca. R. jingxinensis is primarily transmitted by H. longicornis and R. microplus. In southwestern China, the overall infection rate of Ca. R. jingxinensis in H. longicornis and R. microplus reaches up to 62% (Wang et al., 2021). Notably, the infection rate is even higher in R. microplus in Guizhou, with rates of 88.89% in Qiannan, 100% in Liupanshui, and 100% in Bijie. Ca. R. jingxinensis has demonstrated zoonotic potential. It has been detected in cats and dogs in Tianjin (Jian et al., 2024). Ca. R. jingxinensis was identified in pangolins that died despite treatment efforts, and it is likely that the infection contributed to the deterioration of their condition and eventual death (Li et al., 2024). A human-derived sequence from China was retrieved from the NCBI GenBank database (Accession number: PP922931). A report from South Korea indicated that Ca. R. jingxinensis infection may be associated with fever, erythema, and eschar (Seo et al., 2024). Although only a few cattle were found to be infected in this study, increased surveillance of local animal hosts and humans is warranted, given the zoonotic potential of Ca. R. jingxinensis. In this study, we identified multiple co-infections of pathogens in both cattle and goats, a common phenomenon among ruminants. These co-infections may be attributed to the presence of various tick species or the ability of certain ticks to harbor multiple hemopathogens (Mohanta et al., 2023; Nahal and Ben Said, 2024). In goats, the most prevalent co-infection pattern was A. bovis + A. capra, whereas in cattle, the predominant patterns were A. marginale + A. bovis and A. capra + A. bovis + A. marginale, consistent with previous studies (Nahal and Ben Said, 2024). The concurrent presence of these pathogens in hosts may complicate both diagnosis and treatment strategies (Nahal and Ben Said, 2024). Furthermore, the co-infection of Ca. R. jingxinensis with Anaplasma spp. was detected for the first time in cattle, highlighting the need for further research to explore the potential implications of this mixed infection. This study has several limitations. First, the absence of individual animal metadata (e.g., sex and age) precluded stratified risk factor analyses and limited our ability to assess the impact of host-specific variables on infection rates. Second, although we detected the potential circulation of distinct pathogen strains in the study area, our interpretation of transmission dynamics and evolutionary relationships was constrained by the lack of phylogeographic and genetic clustering analyses. Although species-specific PCR was appropriate for detecting the target pathogens, it may have failed to identify co-infections with rare or novel agents. Not all positive amplicons were sequenced, which could have introduced false positives in a subset of cases. Future studies should incorporate detailed epidemiological metadata, utilize metagenomic or multiplex high-throughput sequencing to capture broader pathogen diversity, and conduct comprehensive genetic analyses to clarify the origin and dissemination of these tick-borne agents. ConclusionAnaplasmosis is highly prevalent in local ruminants, particularly in goats. The distribution of pathogen species differs between hosts, with A. marginale showing host specificity for cattle. The presence of Ca. R. jingxinensis in cattle in this study suggests an expansion of its host range. These findings highlight the need for enhanced surveillance and control measures for these pathogens, especially considering the potential spillover risks and public health threat. AcknowledgmentsThe authors would like to extend their sincere gratitude to all the staff at the Epidemic Disease Prevention and Control Center of the Dejiang County Department of Agriculture and Rural Affairs for their invaluable support. Conflicts of interestThe authors have no conflicts of interest to declare. FundingThis research was funded by the Guizhou Provincial Basic Research Program (Natural Science zk [2025] (No.497); grants from the National Science Foundation of China (Nos. 82160633 and 81760605); Foundation of State Key Laboratory of Pathogen and Biosecurity of China (Grant No. SKLPBS2442) and the GZPH-NSFC-2021-17. Authors' contributionJiashun Yu: Conceptualization; methodology, validation, investigation, formal analysis, writing-original draft preparation. Xiaopeng Yang: validation, investigation. Yisong Dai: validation, Jixia Tang and Qiu Chen: investigation. Jiao Meng and Mingjiang Tao: formal analysis, Lin zhan: funding acquisition, writing—review and editing. Kun Cai and Funxun Yu: writing—review and editing. Xingxing Chen and Chaomin Zhou: visualization. Jiafu Jiang and Wuchun Cao: supervision. Data availabilityAll the data can be found in Supplementary Table S1. ReferencesAddo, S.O., Amoah, S., Unicorn, N.M., Kyeremateng, E.T., Desewu, G., Obuam, P.K., Malm, R.O.T., Osei-Frempong, E., Torto, F.A., Accorlor, S.K., Baidoo, P.K., Dadzie, S.K. and Larbi, J.A. 2024. Molecular detection of tick-borne pathogens in kumasi: with a first report of zoonotic pathogens in abattoir workers. Biomed. Res. Int. 2024, 4848451; doi:10.1155/2024/4848451 Akwongo, C.J. and Byaruhanga, C. 2024. Epidemiology of Anaplasma species amongst cattle in Africa from 1970 to 2022: a systematic review and meta-analysis. Prev. Vet. Med. 228, 106214; doi:10.1016/j.prevetmed.2024.106214 Altay, K., Erol, U. and Sahin, O.F. 2024. Anaplasma capra: a new emerging tick-borne zoonotic pathogen. Vet. Res. Commun. 48, 1329–1340; doi:10.1007/s11259-024-10337-9 Atif, F.A. 2016. Alpha proteobacteria of genus Anaplasma (Rickettsiales: anaplasmataceae): Epidemiology and characteristics of Anaplasma species related to veterinary and public health importance. Parasitology 143, 659–685; doi:10.1017/S0031182016000238 Barradas, P.F., Mesquita, J.R., Ferreira, P., Gärtner, F., Carvalho, M., Inácio, E., Chivinda, E., Katimba, A. and Amorim, I. 2021. Molecular identification and characterization of Rickettsia spp. and other tick-borne pathogens in cattle and their ticks from Huambo, Angola. Ticks Tick. Borne. Dis. 12, 101583; doi:10.1016/j.ttbdis.2020.101583 Calchi, A.C., Vultão, J.G., Alves, M.H., Yogui, D.R., Desbiez, A.L.J., De Santi, M., Santana, M.D.S., Da Silva, T.M.V., Werther, K., Teixeira, M.M.G., Machado, R.Z. and André, M.R. 2020. Ehrlichia spp. and Anaplasma spp. in Xenarthra mammals from Brazil, with evidence of novel “Candidatus Anaplasma spp.”. Sci. Rep. 10, 12615; doi:10.1038/s41598-020-69263-w Chimnoi, W., Jhaiaun, P., Phasuk, J., Kaewnoi, D., Inpankaew, T., Nimsuphan, B., Ngasaman, R. and Kamyingkird, K. 2025. Application of molecular detection, phylogenetic analysis, and risk factor evaluation for combating Anaplasma infection in small-scale livestock farms in Thailand. Parasitology 152, 321–329; doi:10.1017/S0031182025000277 Dejiang County People's Government Portal. 2024. Annual output value exceeds 1.5 billion yuan! This industry in Dejiang is truly impressive. Dejiang County People's Government Portal. Available from: https://www.dejiang.gov.cn/xwzx/xzcz/202412/t20241212_86349970.html. El-Alfy, E., -S.., Abbas, I., Elseadawy, R., Saleh, S., Elmishmishy, B., El-Sayed, S.A.E., -S.. and Rizk, M.A. 2023. Global prevalence and species diversity of tick-borne pathogens in buffaloes worldwide: a systematic review and meta-analysis. Parasit. Vectors. 16, 115; doi:10.1186/s13071-023-05727-y Goodger, W.J., Carpenter, T. and Riemann, H. 1979. Estimation of economic loss associated with anaplasmosis in California beef cattle. J. Am. Vet. Med. Assoc. 174, 1333–1336. Guo, W., -P.., Tie, W., -F.., Meng, S., Li, D., Wang, J., -L.., Du, L., -Y.., Xie, G. and -C. 2020. Extensive genetic diversity of Anaplasma bovis in ruminants in Xi’an, China. Ticks. Tick. Borne. Dis. 11, 101477; doi:10.1016/j.ttbdis.2020.101477 Guo, W.P., Wang, X., Li, Y.N., Xu, G., Wang, Y.H. and Zhou, E.M. 2019. GroEL gene typing and genetic diversity of Anaplasma bovis in ticks in Shaanxi, China. Infect. Genet. Evol. 74, 103927; doi:10.1016/j.meegid.2019.103927 Han, X., -Y.., Du, L., -F.., Lin, Z., -T.., Li, C., Xiong, T., Zhu, W., -J.., Ye, R., -Z.., Wang, N., Wang, Y., -F.., Gao, W., -Y.., Zhao, L., Cui, X., -M.., Cao, W. and -C. 2024. Genomic characters of Anaplasma bovis and genetic diversity in China. Emerg. Microbes. Infect. 13, 2323153; doi:10.1080/22221751.2024.2323153 Jian, R., Xue, J., Xu, Z.Y., Chen, S.S., Wang, F.N., Du, L., Xie, G.C. and Guo, W.P. 2024. Genetic diversity of vector-borne zoonotic pathogens in companion dogs and cats, Tianjin, China. Front. Vet. Sci. 11, 1373178; doi:10.3389/fvets.2024.1373178 Jouglin, M., Rispe, C., Grech-Angelini, S., Gallois, M. and Malandrin, L. 2022. Anaplasma capra in sheep and goats on Corsica Island, France: a European lineage within A. capra clade II?. Ticks. Tick. Borne. Dis. 13, 101934; doi:10.1016/j.ttbdis.2022.101934 Karpathy, S.E., Kingry, L., Pritt, B.S., Berry, J.C., Chilton, N.B., Dergousoff, S.J., Cortinas, R., Sheldon, S.W., Oatman, S., Anacker, M., Petersen, J. and Paddock, C.D. 2023. Anaplasma bovis–Like Infections in Humans, United States, 2015–2017. Emerg. Infect. Dis. J. 29(9), 1904–1907; doi: 10.3201/eid2909.230559 Kim, Y.J., Seo, J.Y., Park, J.S., Kim, S.Y., Aknazarov, B., Atabekova, N. and Lee, H.I. 2024. Molecular Analysis of Tick-Borne Bacterial Pathogens from Ticks Infesting Animal Hosts in Kyrgyzstan, 2021. Microorganisms 12, 1046; doi:10.3390/microorganisms12061046 Kocan, K.M., De La Fuente, J., Blouin, E.F., Coetzee, J.F. and Ewing, S.A. 2010. The natural history of Anaplasma marginale. Vet. Parasitology. Ticks Tick-borne Pathogens 167, 95–107; doi:10.1016/j.vetpar.2009.09.012 Li, B., Zhai, J.Q., Wu, Y.J., Shan, F., Zou, J.J., Hou, F.H., Que, T.C. and Chen, W. 2024. Molecular identification of tick-borne Rickettsia, Anaplasma, Ehrlichia, Babesia, and Colpodella in confiscated Malayan pangolins. PLos Negl. Trop. Dis. 18, 12667; doi:10.1371/journal.pntd.0012667 Li, H., Zheng, Y.C., Ma, L., Jia, N., Jiang, B.G., Jiang, R.R., Huo, Q.B., Wang, Y.W., Liu, H.B., Chu, Y.L., Song, Y.D., Yao, N.N., Sun, T., Zeng, F.Y., Dumler, J.S., Jiang, J.F. and Cao, W.C. 2015. Human infection with a novel tick-borne Anaplasma species in China: a surveillance study. Lancet Infect. Dis. 15, 663–670; doi:10.1016/S1473-3099(15)70051-4 Liu, H., Li, Q., Zhang, X., Li, Z., Wang, Z., Song, M., Wei, F., Wang, S. and Liu, Q. 2016. Characterization of rickettsiae in ticks in northeastern China. Parasites. &. Vectors. 9, 498; doi:10.1186/s13071-016-1764-2 Liu, L.K. and Guo, J.J. 2011. Tick species and dominant species in poultry and livestock in Guizhou. J. Southwest. Univ. (Nat. Sci. Ed). 36, 98–101; doi: 10.13718/j.cnki.xsxb.2011.01.040 Lu, M., Chen, Q., Qin, X., Lyu, Y., Teng, Z., Li, K., Yu, L., Jin, X., Chang, H., Wang, W., Hong, D., Sun, Y., Kan, B., Gong, L. and Qin, T. 2022. Anaplasma bovis infection in fever and thrombocytopenia Patients - Anhui Province, China, 2021. China. CDC. Wkly. 4, 249–253; doi:10.46234/ccdcw2022.053 Lu, M., Li, F., Liao, Y., Shen, J.J., Xu, J.M., Chen, Y.Z., Li, J.H., Holmes, E.C. and Zhang, Y.Z. 2019. Epidemiology and diversity of rickettsiales bacteria in humans and animals in Jiangsu and Jiangxi provinces, China. Scientific. Rep. 9, 13176; doi:10.1038/s41598-019-49059-3 Lysholm, S., Ådén, F., Aspán, A., Högberg, A., Wensman, J.J. and Omazic, A. 2023. Presence of Anaplasma spp. and their associated antibodies in the swedish goat population. Animals. (Basel). 13, 333; doi:10.3390/ani13030333 Ma, Y., Jian, Y., Wang, G., Zafar, I., Li, X., Wang, G., Hu, Y., Yokoyama, N., Ma, L. and Xuan, X. 2024. Epidemiological investigation of tick-borne bacterial pathogens in domestic animals from the Qinghai-Tibetan Plateau Area, China. Pathogens 13, 86; doi:10.3390/pathogens13010086 Miranda, E.A., Han, S.W., Cho, Y.K., Choi, K.S. and Chae, J.S. 2021. Co-infection with anaplasma species and novel genetic variants detected in cattle and goats in the Republic of Korea. Pathogens 10, 28; doi:10.3390/pathogens10010028 Mohanta, U.K., Chikufenji, B., Galon, E.M., Ji, S., Ma, Z., El-Sayed, S.A.E.S., Amer, M.M., Do, T.T. and Xuan, X. 2023. Molecular characterization and phylogeny of Anaplasma marginale, A. phagocytophilum and A. bovis in livestock of Bangladesh. Parasitol. Int. 97, 102790; doi:10.1016/j.parint.2023.102790 Nahal, A. and Ben Said, M. 2024. Systematic Review and Meta-Analysis on Piroplasma spp. Infection and Co-infection with Anaplasma marginale in Domestic Ruminants from Algeria. Acta Parasitol. 69, 135–151; doi:10.1007/s11686-023-00768-w Oguz, B., Deger, M.S., Al-Olayan, E. and El-Ashram, S. 2024. Molecular Survey of Anaplasma capra in Goats in Van Province, Eastern Türkiye. Acta Parasitol. 69, 370–374; doi:10.1007/s11686-023-00758-y Ooshiro, M., Zakimi, S., Matsukawa, Y., Katagiri, Y. and Inokuma, H. 2008. Detection of Anaplasma bovis and Anaplasma phagocytophilum from cattle on Yonaguni Island, Okinawa, Japan. Vet. Parasitol. 154, 360–364; doi:10.1016/j.vetpar.2008.03.028 Peng, Y., Lu, C., Yan, Y., Shi, K., Chen, Q., Zhao, C., Wang, R., Zhang, L., Jian, F. and Ning, C. 2021. The first detection of Anaplasma capra, an emerging zoonotic Anaplasma sp., in erythrocytes. Emerg. Microbes Infect. 10, 226–234; doi:10.1080/22221751.2021.1876532 Peng, Y., Wang, K., Zhao, S., Yan, Y., Wang, H., Jing, J., Jian, F., Wang, R., Zhang, L. and Ning, C. 2018. Detection and phylogenetic characterization of Anaplasma capra: an emerging pathogen in sheep and goats in China. Front. Cell. Infect. Microbiol. 8, 283; doi:10.3389/fcimb.2018.00283 Qiu, H., Kelly, P.J., Zhang, J., Luo, Q., Yang, Y., Mao, Y., Yang, Z., Li, J., Wu, H., Wang, C., 2016. Molecular detection of Anaplasma spp. and Ehrlichia spp. in Ruminants from Twelve Provinces of China. Can. J. Infect. Dis. Med. Microbiol 2016, 9183861; doi:10.1155/2016/9183861 Rar, V. and Golovljova, I. 2011. Anaplasma, Ehrlichia, and “Candidatus Neoehrlichia” bacteria: pathogenicity, biodiversity, and molecular genetic characteristics, a review. Infection. Genet. Evol. 11, 1842–1861; doi:10.1016/j.meegid.2011.09.019 Rar, V., Tkachev, S. and Tikunova, N. 2021. Genetic diversity of Anaplasma bacteria: twenty years later. Infect. Genet. Evol. 91, 104833; doi:10.1016/j.meegid.2021.104833 Remesar, S., Prieto, A., García-Dios, D., López-Lorenzo, G., Martínez-Calabuig, N., Díaz-Cao, J.M., Panadero, R., López, C.M., Fernández, G., Díez-Baños, P., Morrondo, P. and Díaz, P. 2022. Diversity of Anaplasma species and importance of mixed infections in roe deer from Spain. Transbound. Emerg. Dis. 69, e374–e385; doi:10.1111/tbed.14319 Sahin, O.F., Erol, U. and Altay, K. 2022. Buffaloes as new hosts for Anaplasma capra: molecular prevalence and phylogeny based on gtlA, groEL, and 16S rRNA genes. Res. Vet. Sci. 152, 458–464; doi:10.1016/j.rvsc.2022.09.008 Salje, J. 2021. Cells within cells: rickettsiales and the obligate intracellular bacterial lifestyle. Nat. Rev. Microbiol. 19, 375–390; doi:10.1038/s41579-020-00507-2 Sanchez, E., Vannier, E., Wormser, G.P. and Hu, L.T. 2016. Diagnosis, treatment, and prevention of lyme disease, human granulocytic anaplasmosis, and babesiosis: a review. JAMA 315, 1767–1777; doi:10.1001/jama.2016.2884 Saratsis, A., Ligda, P., Aal, F., Jelicic, M., Polgar, J., De Vries, M., Mastranestasis, I., Musella, V., Rinaldi, L., Jongejan, F. and Sotiraki, S. 2022. The Scenario of Ticks and Tick-Borne Pathogens of Sheep on a Mediterranean Island. Microorganisms 10, 1551; doi:10.3390/microorganisms10081551 Schudel, S., Gygax, L., Kositz, C., Kuenzli, E. and Neumayr, A. 2024. Human granulocytotropic anaplasmosis—A systematic review and analysis of the literature. PLos Negl. Trop. Dis. 18, 12313; doi:10.1371/journal.pntd.0012313 Seo, J.Y., Kim, Y.J., Kim, S.Y. and Lee, H.I. 2023. Molecular Detection of Anaplasma, Ehrlichia and Rickettsia Pathogens in Ticks Collected from Humans in the Republic of Korea, 2021. Pathogens 12, 802; doi:10.3390/pathogens12060802 Seo, J.Y., Park, J.S., Lee, H.I. and Ju, J.W. 2024. Molecular Identification of Spotted Fever Group Rickettsiae in Ticks in the Republic of Korea. Pathogens 13, 575; doi:10.3390/pathogens13070575 Seo, M.G., Kwon, O.D. and Kwak, D. 2020. Genotypic analysis of piroplasms and associated pathogens from ticks infesting cattle in Korea. Microorganisms 8, 728; doi:10.3390/microorganisms8050728 Shen, X.R. and Shen, D.R. 1997. Ticks in Guizhou and their relationship with diseases. Chin. J. Vector. Biol. Control. 1997, 287–288; doi:cnki:sun:zmsk.0.1997-04-052 Tang, J., Xu, J., Liu, X.H., Lv, F.Z., Yao, Q.J., Zhou, X.F., Lu, H.Y., Yu, T.M., Jiang, Z.Z., Jin, X.Z., Guo, F. and Yu, X.J. 2024. Prevalence and genetic diversity of Anaplasma and Ehrlichia in ticks and domesticated animals in Suizhou County, Hubei Province, China. Scientific. Rep. 14, 12621; doi:10.1038/s41598-024-63267-6 Wang, F., Yan, M., Liu, A., Chen, T., Luo, L., Li, L., Teng, Z., Li, B., Ji, Z., Jian, M., Ding, Z., Wen, S., Zhang, Y., Yue, P., Cao, W., Xu, X., Zhou, G. and Bao, F. 2020. The seroprevalence of Anaplasma phagocytophilum in global human populations: a systematic review and meta-analysis. Transbound. Emerg. Dis. 67, 2050–2064; doi:10.1111/tbed.13548 Wang, Q., Guo, W.B., Pan, Y.S., Jiang, B.G., Du, C.H., Que, T.C., Zhan, L., Wu, J.H., Yu, M.H., Cui, X.M., Zhao, L., Xu, D.L., Xia, L.Y., Ye, R.Z., Li, J., Li, L.F., Wei, W., Zhou, Y.H., Jiang, J.F., Jia, N. and Cao, W.C. 2021. Detection of Novel Spotted Fever Group Rickettsiae (Rickettsiales: rickettsiaceae) in Ticks (Acari: Ixodidae) in Southwestern China. J. Med. Entomol. 58, 1363–1369; doi:10.1093/jme/tjaa294 Wu, X., Wang, J., Gao, S., Luo, X., Li, Q., Chen, D., Liu, X., Gu, Y. and Li, W. 2024. Prevalence of Anaplasma infections in sheep and goats in Anhui Province in 2020. Zhongguo. Xue. Xi. Chong. Bing. Fang. Zhi. Za. Zhi. 36, 620–625; doi:10.16250/j.32.1374.2023232 Xue, J., Chen, S., -S.., Xu, Z., -Y.., Wang, F., -N.., Wang, J., Diao, D., Du, L., Xie, G., -C.., Guo, W. and -P. 2024. Anaplasma, Bartonella, and Rickettsia infections in Daurian ground squirrels (Spermophilus dauricus), Hebei, China. Front. Microbiol. 15, 1359797; doi:10.3389/fmicb.2024.1359797 Yang, J., Liu, Z., Niu, Q., Liu, J., Han, R., Guan, G., Hassan, M.A., Liu, G., Luo, J. and Yin, H. 2017. A novel zoonotic Anaplasma species is prevalent in small ruminants: potential public health implications. Parasites & Vectors 10, 264; doi:10.1186/s13071-017-2182-9 Yang, J., Liu, Z., Niu, Q., Liu, J., Han, R., Liu, G., Shi, Y., Luo, J. and Yin, H. 2016. Molecular survey and characterization of a novel Anaplasma species closely related to Anaplasma capra in ticks, northwestern China. Parasit. Vectors. 9, 603; doi:10.1186/s13071-016-1886-6 Ybañez, A.P., Sivakumar, T., Ybañez, R.H.D., Ratilla, J.C., Perez, Z.O., Gabotero, S.R., Hakimi, H., Kawazu, S., Matsumoto, K., Yokoyama, N., Inokuma, H., n.d. First Molecular Characterization of Anaplasma marginale in Cattle and Rhipicephalus (Boophilus) microplus Ticks in Cebu, Philippines. Zeng, W., Li, Z., Jiang, T., Cheng, D., Yang, L., Hang, T., Duan, L., Zhu, D., Fang, Y. and Zhang, Y. 2022. Identification of bacterial communities and tick-borne pathogens in Haemaphysalis spp. collected from Shanghai, China. Trop. Med. Infect. Dis. 7, 413; doi:10.3390/tropicalmed7120413 Supplementery MaterialsTable S1. BLAST results of the sequences in this study compared with the sequences in GenBank.
| ||
| How to Cite this Article |
| Pubmed Style Yu J, Yang X, Dai Y, Tang J, Chen Q, Meng J, Tao M, Chen X, Zhou C, Jiang J, Cao W, Yu F, Cai K, Zhan L. Molecular detection of Anaplasma and Candidatus Rickettsia jingxinensis infections in domestic cattle and goats in Dejiang County, Guizhou Province, China. Open Vet. J.. 2025; 15(10): 4949-4961. doi:10.5455/OVJ.2025.v15.i10.14 Web Style Yu J, Yang X, Dai Y, Tang J, Chen Q, Meng J, Tao M, Chen X, Zhou C, Jiang J, Cao W, Yu F, Cai K, Zhan L. Molecular detection of Anaplasma and Candidatus Rickettsia jingxinensis infections in domestic cattle and goats in Dejiang County, Guizhou Province, China. https://www.openveterinaryjournal.com/?mno=264648 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.14 AMA (American Medical Association) Style Yu J, Yang X, Dai Y, Tang J, Chen Q, Meng J, Tao M, Chen X, Zhou C, Jiang J, Cao W, Yu F, Cai K, Zhan L. Molecular detection of Anaplasma and Candidatus Rickettsia jingxinensis infections in domestic cattle and goats in Dejiang County, Guizhou Province, China. Open Vet. J.. 2025; 15(10): 4949-4961. doi:10.5455/OVJ.2025.v15.i10.14 Vancouver/ICMJE Style Yu J, Yang X, Dai Y, Tang J, Chen Q, Meng J, Tao M, Chen X, Zhou C, Jiang J, Cao W, Yu F, Cai K, Zhan L. Molecular detection of Anaplasma and Candidatus Rickettsia jingxinensis infections in domestic cattle and goats in Dejiang County, Guizhou Province, China. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4949-4961. doi:10.5455/OVJ.2025.v15.i10.14 Harvard Style Yu, J., Yang, . X., Dai, . Y., Tang, . J., Chen, . Q., Meng, . J., Tao, . M., Chen, . X., Zhou, . C., Jiang, . J., Cao, . W., Yu, . F., Cai, . K. & Zhan, . L. (2025) Molecular detection of Anaplasma and Candidatus Rickettsia jingxinensis infections in domestic cattle and goats in Dejiang County, Guizhou Province, China. Open Vet. J., 15 (10), 4949-4961. doi:10.5455/OVJ.2025.v15.i10.14 Turabian Style Yu, Jiashun, Xiaopeng Yang, Yisong Dai, Jixia Tang, Qiu Chen, Jiao Meng, Mingjiang Tao, Xingxing Chen, Chaomin Zhou, Jiafu Jiang, Wuchun Cao, Fuxun Yu, Kun Cai, and Lin Zhan. 2025. Molecular detection of Anaplasma and Candidatus Rickettsia jingxinensis infections in domestic cattle and goats in Dejiang County, Guizhou Province, China. Open Veterinary Journal, 15 (10), 4949-4961. doi:10.5455/OVJ.2025.v15.i10.14 Chicago Style Yu, Jiashun, Xiaopeng Yang, Yisong Dai, Jixia Tang, Qiu Chen, Jiao Meng, Mingjiang Tao, Xingxing Chen, Chaomin Zhou, Jiafu Jiang, Wuchun Cao, Fuxun Yu, Kun Cai, and Lin Zhan. "Molecular detection of Anaplasma and Candidatus Rickettsia jingxinensis infections in domestic cattle and goats in Dejiang County, Guizhou Province, China." Open Veterinary Journal 15 (2025), 4949-4961. doi:10.5455/OVJ.2025.v15.i10.14 MLA (The Modern Language Association) Style Yu, Jiashun, Xiaopeng Yang, Yisong Dai, Jixia Tang, Qiu Chen, Jiao Meng, Mingjiang Tao, Xingxing Chen, Chaomin Zhou, Jiafu Jiang, Wuchun Cao, Fuxun Yu, Kun Cai, and Lin Zhan. "Molecular detection of Anaplasma and Candidatus Rickettsia jingxinensis infections in domestic cattle and goats in Dejiang County, Guizhou Province, China." Open Veterinary Journal 15.10 (2025), 4949-4961. Print. doi:10.5455/OVJ.2025.v15.i10.14 APA (American Psychological Association) Style Yu, J., Yang, . X., Dai, . Y., Tang, . J., Chen, . Q., Meng, . J., Tao, . M., Chen, . X., Zhou, . C., Jiang, . J., Cao, . W., Yu, . F., Cai, . K. & Zhan, . L. (2025) Molecular detection of Anaplasma and Candidatus Rickettsia jingxinensis infections in domestic cattle and goats in Dejiang County, Guizhou Province, China. Open Veterinary Journal, 15 (10), 4949-4961. doi:10.5455/OVJ.2025.v15.i10.14 |