| Research Article | ||
Open Vet. J.. 2025; 15(10): 4936-4948 Open Veterinary Journal, (2025), Vol. 15(10): 4936-4948 Research Article Therapeutic efficacy of liposomal caffeic acid in high-fat diet-induced obesity in rats: Targeting hedgehog signalling pathway, adipokines, and insulin resistanceWafa S. Alansari1, Mahran Mohamed Abd El-Emam2*, Rawan Altalhi3, Safaa I. Khater2, Tarek Khamis4, Hoda S. Sherkawy5, Abdelhamid M. Fares6, Marwa M. Lotfy2, Ola Mohammed Youssef7 and Engy Mohamed Mohamed Yassin21Biochemistry Department, Faculty of Science, University of Jeddah, Jeddah, Saudi Arabia 2Department of Biochemistry and Molecular Biology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt 3Department of Biological Sciences, College of Science, University of Jeddah, Jeddah, Saudi Arabia 4Department of Pharmacology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt 5Medical Biochemistry and Molecular Biology Department, Faculty of Medicine, Aswan University, Aswan, Egypt 6Department of Pathology, Faculty of Veterinary Medicine, University of Sadat City, Monofiya, Egypt 7Department of Human Anatomy and Embryology, Faculty of Medicine, Mansoura University, Mansoura, Egypt *Corresponding Author: Mahran Mohamed Abd El-Emam. Department of Biochemistry and Molecular Biology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt. Email: MMMahran [at] vet.zu.edu.eg Submitted: 16/06/2025 Revised: 24/08/2025 Accepted: 03/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: Obesity spurred by a high-fat diet (HFD) has emerged as a significant worldwide health issue. Caffeic acid (CA) is a plant-derived phenolic compound found in various foods such as coffee, wine, and tea. It exhibits antioxidant, anti-inflammatory, and anticarcinogenic properties. However, the therapeutic potential of this substance is hindered by its poor water solubility and low oral bioavailability. Aim: In this study, CA was loaded into liposomes Caffeic Acid -Loaded Liposomes (CA-Lip) , and the potential role of CA-Lip in mitigating HFD-induced obesity in rats was examined. Methods: Forty male Wistar rats were used in this investigation. They were allocated into four groups: the first was fed a regular diet; the second received a normal diet plus CA-Lip; the third received an HFD; and the fourth received both an HFD and CA-Lip. Results: The developed CA-Lip showed a particle size of 125 nm, zeta potential of −15 mV, and entrapment efficiency of 82%. The HFD+CA-Lip group's body weight was considerably lower than that of the HFD group. The HFD+CA-Lip group also showed a significant improvement in their serum lipid profiles. CA-Lip lowered levels of adipokines, which encourage inflammation, including leptin, interleukin-6, and tumor necrosis factor-α. It also reduced the size of the adipocytes when compared to the HFD control group. Furthermore, it reversed the HFD-induced upregulation of Hh genes [protein patched homolog 1 (PTCH1), Hh-interacting protein (HHIP), glioma-associated oncogene homolog 1 (Gli1), and smoothened (Smo) receptor] in adipose tissue. Conclusion: In summary, CA-Lip may reduce obesity and related inflammation by activating the Hh signaling pathway. Keywords: Caffeic acid, Liposomes, Obesity, Lipid, Adipokines, Hhog pathway. IntroductionObesity is a multifaceted health concern impacted by various factors such as genetics, lifestyle choices, and dietary habits (McCafferty et al., 2020). Nowadays, the rates of obesity have doubled in many countries (WHO, 2023). By 2035, over half of the global population is predicted to be overweight due to various lipid problems. This condition is marked by various lipid issues, including high triglyceride (TG) and total cholesterol (TC) levels, low high-density lipoprotein cholesterol (LDL-C) levels, and irregular LDL-C composition (Vekic et al., 2019). Moreover, consuming a high-fat diet (HFD) regularly leads to the body accumulating extra energy, which is stored as TGs in white adipose tissue (Bastías-Pérez et al., 2020). Furthermore, Obesity leads to malfunctioning white adipose tissue, lipid overflow, metabolic problems, and ectopic fat accumulation in muscle and liver (Cioffi et al., 2022). Additionally, abnormal metabolic processes are intimately associated with dementia, cancer, heart disease, and type 2 diabetes (Yan et al., 2023). As well, obesity impairs organ and system physiological processes, leading to increased mortality rates and a 5–7-year decrease in human lifespan (Tam et al., 2020). It significantly impacts public health, making prevention and treatment crucial yet still a significant challenge. The Hedgehog (Hh) signaling pathway is a conserved regulator of tissue development and cellular function. Activation of this pathway inhibits adipocyte differentiation in various cell models, including 3T3-L1 and NIH-3T3 (Shi and Long, 2017). The Hh ligand binds to the Patched (PTC1/PTC2) receptor complex, relieving inhibition of the Smoothened (Smo) receptor, which then transduces signals to Glibenclamide transcription factors in the nucleus, modulating genes involved in cell fate, polarity, and proliferation (Sigafoos et al., 2021). Notably, Hh activation suppresses adipogenesis, whereas its inhibition promotes fat cell formation, suggesting a potential therapeutic role for Hh signaling modulation in metabolic diseases such as type 2 diabetes and obesity (Wei et al., 2019; Chou et al., 2021). There are several approaches to managing obesity and/or overweight, such as medication, exercise, and nutrition (“Expert Panel Report: Guidelines (2013) for the management of overweight and obesity in adults,” 2014). Orlistat and sibutramine are anti-obesity drugs with modest clinical efficacy, but they also have serious side effects (Tak and Lee, 2021). Natural products, such as caffeic acid (CA), are often associated with minimal side effects, which enhances their appeal as alternative therapeutic options (Wang et al., 2023). CA, also known as 3,4-dihydroxycinnamic acid, is a type of phenolic acid characterized by a hydroxybenzene acrylic acid structure. It is a catechol-based secondary metabolite found in plants like Melissa officinalis, various fruits, and certain traditional Chinese medicinal herbs (Chen et al., 2019). CA and its analogs exhibit various pharmacological activities such as anti-inflammatory, anti-cancer, anti-oxidant, anti-mutagenic, and anti-virus properties (Bounegru and Apetrei, 2020). Additionally, CA can enhance lipid metabolism and help reduce obesity (Kim et al., 2018). However, CA's therapeutic effects in vivo are hindered by its limited solubility, absorption, bioavailability, instability, lack of specificity, and high systemic clearance (Stasiłowicz-Krzemień et al., 2023). Drug delivery systems (DDSs) help address limitations by protecting compounds from degradation and improving their physicochemical properties (Chen et al., 2024). Liposomes are nanocarriers with phospholipid bilayers, accommodating hydrophilic agents and lipophilic molecules due to their amphiphilic nature (Ramalho et al., 2018). Liposomes offer excellent biocompatibility and bioavailability, enabling controlled, sustained drug release. This allows for effective treatment with lower doses, reducing potential side effects (Chelliah et al., 2025). This investigation aims to develop and evaluate a novel liposomal CA formulation Caffeic Acid -Loaded Liposomes (CA-Lip) as a targeted anti-obesity therapy, investigating its multifaceted effects on metabolic health—specifically glycaemic status, lipid profile, adipokine secretion, and the molecular regulation of the Hh signaling pathway in adipose tissue of HFD-induced obese rats. Materials and MethodsMaterialsCA (98% purity) and cholesterol were supplied by Sigma–Aldrich (St. Louis, MO). Saturated phosphatidylcholine from soybean was provided by Lipoid GmbH (Ludwigshafen, Germany). Furthermore, the study employed only the highest-quality reagents and other compounds. Preparation of liposomal characterizationCA-Lip has been developed using the ethanol injection method, as outlined previously (Alaaeldin et al., 2021). In this approach, a mixture of saturated phosphatidylcholine from soybean, cholesterol, and CA, in a molar ratio of 10:1:1, was dissolved in a minimal volume of anhydrous alcohol to form the organic phase, which was subsequently heated to 60°C–70°C. In parallel, phosphate-buffered saline (PBS) at pH 7.4 was maintained at the same temperature and agitated at 1,000 rpm within a closed system. The heated organic phase was then injected into the PBS solution through a 23G syringe, resulting in the formation of a liposomal suspension. This mixture was continuously stirred at 60°C–70°C until any residual ethyl alcohol was completely evaporated. After the completion of the process, the CA-Lip was stored at 4°C for later analysis and experimentation. Blank niosomal systems were prepared following the same protocol without CA. The liposomal systems were characterized by evaluating various parameters, including entrapment efficiency, drug loading efficiency, mean particle size, and zeta potential, as described in earlier work (Abd El-Emam et al., 2023). Experimental animalsForty male Sprague–Dawley rats weighing 250 ± 5 g were acquired from the research animal farm located at Zagazig University's Faculty of Veterinary Medicine. Each rat was housed in a stainless-steel cage that was maintained at a temperature between 21°C and 24°C, offering a clean, pathogen-free environment. To achieve ideal living circumstances, the rats were exposed to a 12-hour light-dark cycle and a 60% relative humidity. Commercial food pellets and unlimited water were provided to the rats. The standard rat diet and the HFD were crafted based on the research conducted by Lasker and colleagues in their study (Lasker et al., 2019). The composition of the HFD used in this study is based on protocols reported in previous studies (Salem et al., 2023). Animals and experimental designFour groups of 10 rats each were randomly assigned based on their body weights. The first one is the control group, which received a 20-week standard rat diet. The second group, referred to as the CA-Lip group, was maintained on a normal diet for the entire 20-week period. During the final 8 weeks, the animals received CA-Lip (1.8 mg/kg body weight) via oral gavage once daily while continuing on the same diet (Shahin et al., 2022). The third one was the HFD-fed group, which was given an HFD for 12 weeks for the induction of obesity and fed with a standard rat diet for the last 8 weeks. During the final 8 weeks, the animals also received normal saline via oral gavage once daily. The fourth group was fed an HFD for the initial 12 weeks, followed by a standard rat diet for the final 8 weeks. During the final 8 weeks, the animals also received CA-Lip (1.8 mg/kg body weight) via oral gavage once daily (Shahin et al., 2022). Body weight measurementBody weight was measured at the beginning of the experiment to ensure proper randomization and equal distribution of animals into experimental groups. Weight measurements relevant to the treatment protocol were then initiated at week 12, following the obesity induction phase, and final body weights were recorded at the end of the 20-week study. SamplingUpon the conclusion of the experimental period, the rats were anesthetized, and blood was drawn from the medial canthus. Part of the blood was put in test tubes without anticoagulants, which were centrifuged at 3,000 rpm for 15 minutes at room temperature, and a serum sample was used for biochemical analysis. The remaining portion was resuspended in tubes containing sodium fluoride, centrifuged at 3,000 rpm for 15 minutes, and then used for blood glucose and insulin analysis. The adipose tissue sample was dissected into two parts for gene expression [real-time quantitative PCR (RT-qPCR)] and histopathological examination. Biochemical analysisThe bioMed diagnostic kit from Egypt was used to detect blood glucose and insulin levels, and Randox assay kits were used to evaluate the serum lipid profile following the manufacturer's instructions. Furthermore, Thermofisher Scientific CO's KRC3012 and ERA31RB ELISA kits were used to measure the levels of TNF-α and IL-6 in serum. The serum's adiponectin and leptin levels were ascertained using an ELISA kit from Millipore in the US. RT-qPCR analysisPrevious documentation of the real-time analysis was done by Mansour et al., 2025. At the beginning, we utilized the Trizol Reagent from Thermo Fisher Scientific in Massachusetts to extract total RNA from adipose tissue. The concentration of the extracted RNA was then measured using the nanoDrop® ND-1000 Spectrophotometer from NanoDrop Technologies in North Carolina. Subsequently, first-strand cDNA synthesis was conducted using the HiSenScriptTM RH (-) cDNA Synthesis Kits from iNtRON Biotechnology in the Republic of Korea. This process was performed in a Veriti 96-well heat cycler using the cDNA synthesis kit from Applied Biosystems in California. The primer sequences are presented in Table 1. The real-time PCR amplification was performed following the established protocol devised by Livak and Schmittgen (2001). Table 1. The sequence of primers used for RT-PCR.
Histopathological examinationThe adipose tissue was excised and handled with caution to minimize any damage to the tissue using a 21-gauge needle before immersion in modified Davidson’s solution for 24 hours (Bancroft and Layton, 2013). Next, the adipose tissue was processed for paraffin infiltration and embedding, and 4–5 µm thick transverse tissue sections from the middle portion of the testicles were prepared, stained with hematoxylin and eosin (Erfan, 2017), and examined microscopically. Statistical analysisWe utilized GraphPad INSTAT software (Version 2) to conduct the statistical analysis. The statistical assessment involved applying a one-way analysis of variance and Tukey's multiple range test. The results comprise the presentation of SE and SEM differences. Significance was determined by considering a p-value of 0.05 or less. Ethical approvalThe ethics committee at Zagazig University's Faculty of Veterinary Medicine in Egypt carefully reviewed and gave its approval to the research protocol. All steps of the study were conducted by the relevant laws and regulations (approval number: ZU-IACUC/2/F/187/2022). The research was also carried out following the guidelines set forth by ARRIVE to ensure the highest standards of research conduct. ResultsCharacterization of CA-LipThe ethanol injection method was employed to fabricate CA-Lip, which was subsequently characterized using several analytical techniques, including particle size analysis, polydispersity index (PDI), zeta potential measurement, and encapsulation efficiency. The average particle size of the CA-Lip, determined via Zetasizer Nano, was found to be 125.1 ± 12.6 nm (Fig. 1A). The PDI of 0.27 indicated a relatively narrow size distribution, suggesting a uniform dispersion of the liposomal particles. The zeta potential of the niosomal systems was measured at −15 ± 0.84 mV (Fig. 1B), reflecting a moderate negative surface charge, which is indicative of the stability of the formulations. The encapsulation efficiency of the CA-Lip was determined to be 82% ± 3.6%, indicating a substantial entrapment of the active compound within the vesicles.
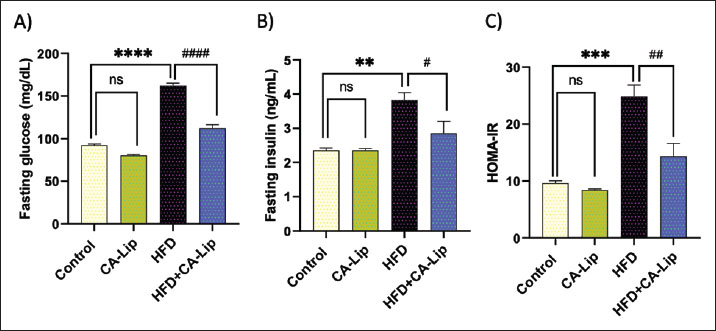
Fig. 1. Characterization of CA-Liposomes: (A) Particle size (125.1 ± 12.6 nm), and (B) zeta potential (−15 ± 0.84 mV). Data are presented as mean ± SEM. CA-Lip administration induces body weight loss in HFD-fed ratsWe examined the effect of HFD on body weight to assess the impact of CA-Lip. There was no significant difference in body weight among the experimental groups at the start of the experiment (Day 0) (Fig. 1A). On the other hand, HFD significantly increased body weight compared to the control group. However, there was no significant difference in body weight between the CA-Lip-treated group and the control group, nor between the HFD group and the HFD + CA-Lip group at week 12. Conversely, HFD markedly increased body weight by 44% compared to the control group at the end of the experiment (week 20th). HFD-fed rats’ body weight significantly dropped by 26% after CA-Lip injection. Also, there was a decrease in body weight in the CA-Lip group compared to the control group (Fig. 1C). CA-Lip restores glucose and insulin levels, as well as Homeostatic Model Assessment of Insulin Resistance (HOMA-IR) scores in HFD-fed ratsThe impact of CA-Lip was assessed by monitoring the blood levels of fasting glucose and insulin, as well as the HOMA-IR scores. Compared to the control group, it was observed that there was a significant increase of 76% in fasting glucose levels, 46% in insulin levels, and 158% in HOMA-IR after consuming HFD. However, when the HFD was accompanied by the administration of CA-Lip, there was a reduction of 30% in fasting glucose levels, 15% in insulin levels, and 40% in HOMA-IR. The CA-Lip group exhibited similar results to the control group (see Fig. 2A–C).
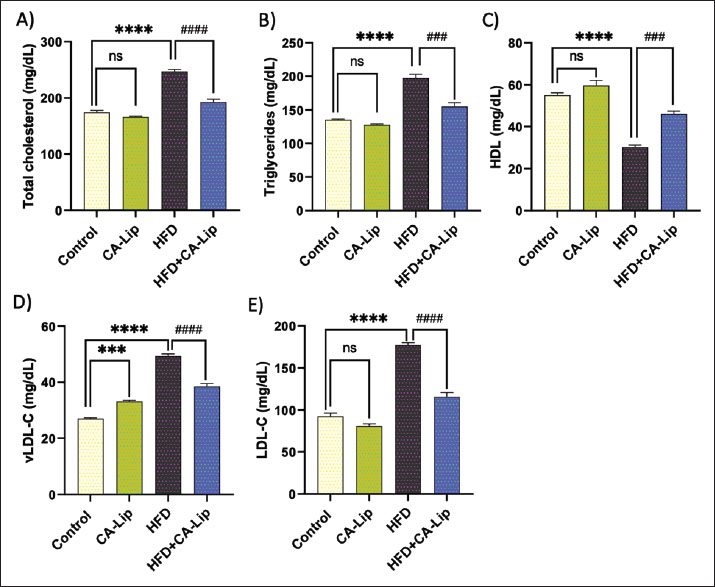
Fig. 2. Effect of CA-Lip on the body weight in HFD-fed rats. Data are presented as the mean ± SEM (n=10). ****p < 0.0001, *p < 0.05, ####p < 0.0001 versus HFD group, ns indicates nonsignificant. CA-Lip modulates changes in serum levels of lipid profile in HFD-fed ratsRats fed an HFT showed a notable increase in serum TC, TG, very low-density lipoprotein (VLDL-C), and LDL-C levels by 41%, 45%, 82%, and 92%, respectively, and a significant decrease in high-density lipoprotein (HDL) levels by 44% in comparison to the control group. CA-Lip injection induced a significant decline in TC, TG, VLDL-C, and LDL-C levels by 22%, 21%, 22%, and 34%, and an elevation in HDL by 51% in CA-Lip-fed HFD rats versus the HFD group. Serum levels of lipid profile were insignificantly different in the CA-Lip group compared to the control rats (Fig. 3A–E).
Fig. 3. Influence of CA-Lip on fasting glucose, insulin, and HOMA-IR levels in HFD-fed rats. The levels of fasting glucose (A), insulin (B), and the scores for HOMA-IR (C) were measured for the different groups. ****p < 0.0001, ***p < 0.001, **p < 0.01, #p < 0.05 versus HFD group, ns indicates nonsignificant. Data are presented as the mean ± SEM (n=10). Effect of CA-Lip on adipokines changes in HFD-fed ratsWhen compared to the negative control, HFD dramatically decreased the levels of blood adiponectin by 61% and increased serum leptin by 75%. CA-Lip administration significantly decreased serum leptin levels by 34% and significantly inhibited serum leptin levels by 100% in the HFD-fed group. CA-Lip group showed almost similar effects on adipokine levels compared to the control rats (Fig. 4A and B).
Fig. 4. Influence of CA-Lip on serum lipid profile. The levels of TC (A), TGs (B), HDL (C), VLDL-C (D), and LDL-C (E) were measured for the different groups. ****p < 0.0001, ***p < 0.001, ####p < 0.0001 versus HFD group, ns indicates nonsignificant. Data are presented as the mean ± SEM (n=10). CA-Lip suppresses proinflammatory cytokines in HFD-fed ratsThe present research identified several significant proinflammatory cytokines associated with obesity. The pro-inflammatory cytokines TNF-α and IL-6 had significantly higher (p < 0.05) levels in rats on an HFD. They were significantly suppressed (p < 0.05) by CA-Lip administration (Fig. 5A and B). These findings imply that CA-Lip could reduce systemic inflammation driven by obesity.
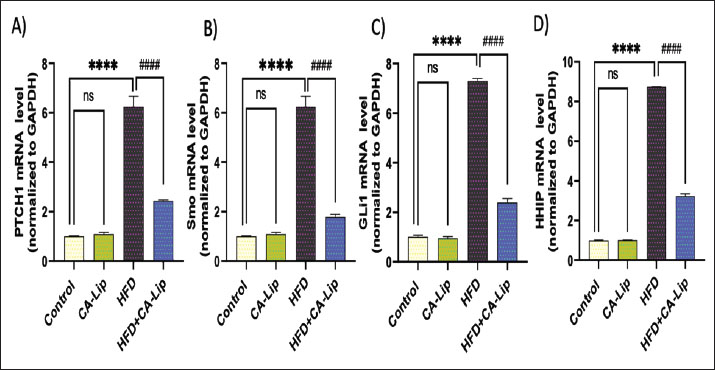
Fig. 5. Effect of CA-Lip on leptin and adiponectin in HFD-fed rats. Data are presented as the mean ± SEM (n=10).****p < 0.0001, **p < 0.0, ####p < 0.0001, ##p < 0.01 versus HFD group, ns indicates nonsignificant. CA-Lip regulates the Hh pathway in adipose tissue of HFD-fed ratsProtein patched homolog 1 (PTCH1), Smo, glioma-associated oncogene homolog 1 (Gli1), and Hh-interacting protein (HHIP) relative mRNA expression were significantly upregulated in obese rats by 470%, 450%, 580%, and 725%, respectively, compared to the control group. The treated rats showed a considerable decrease in PTCH-1, Smo, Gli-1, and HHIP expression by 57%, 73%, 64%, and 60%, correlated with the obese group. There was no obvious statistically significant difference between the CA-Lip group and the control group. The CA-Lip group showed a decrease in PTCH-1, Smo, Gli-1, and HHIP expression by 10%, 11%, 6%, and 2%, compared to the control group (Fig. 7 A–D).
Fig. 6. Effects of CA-Lip on serum inflammation (n=10 per group). (a) TNF-α and (b) IL-6. ****p < 0.0001, ####p < 0.0001 versus HFD group, ns indicates nonsignificant. Data are presented as the mean ± SEM (n=10).
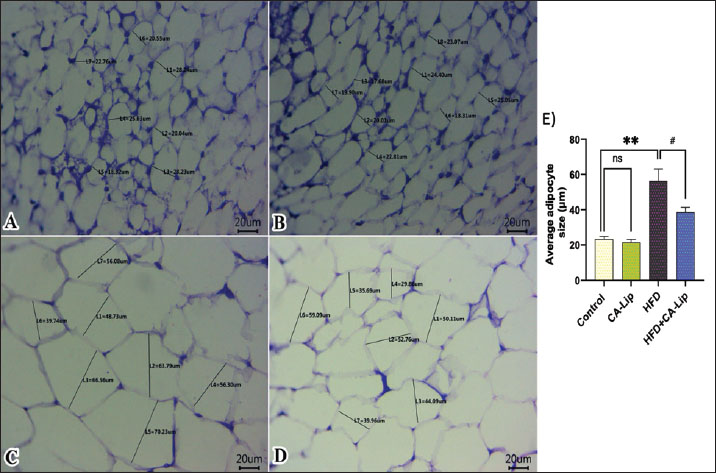
Fig. 7. Effect of CA-Lip on the mRNA expression of (A) PTCH1, (B) Smo, (C) GLI-1, and (D) HHIP genes in adipose tissue of the HFD group. ****p < 0.0001, ####p < 0.0001 versus HFD group, ns indicates nonsignificant. Data are presented as the mean ± SEM (n=10). Histopathological findingsNormal histological structures of adipocytes, represented by clear cytoplasm, peripherally located nuclei, and intact cell membranes, were noticed in both the control (Fig. 8A) and the CA-Lip group alone (Fig. 8B) of abdominal fat. However, increased volumes of fat cells due to more lipid deposits in adipocytes were seen in the HFD group (Fig. 8C) when compared to the control group. On the other hand, there are reductions in the sizes of adipocytes in most examined sections in the treated obese group by CA-Lip (Fig. 8D) in comparison with the obese rats’ group. Representative figures from three sections per mouse (n=5 rats) were examined to determine average adipocyte sizes by counting 50 cells using ImageJ software (Fig. 8E).
Fig. 8. Representative photomicrographs of H&E-stained sections from abdominal fat (Scale bar 20 μm) in rats (A–D) and a representative graph illustrating average adipocyte sizes among different groups (E). DiscussionThe global obesity epidemic, influenced by genetics, fast food, and urban decline, is causing limited mainstream remedies due to side effects and restrictions, prompting an urgent need for a safe, effective natural alternative (Payab et al., 2020; Aron-Wisnewsky et al., 2021). This research investigates the potential of CA-Lip in combating obesity and promoting a healthy lifestyle. When rats are fed HFD, their body fat percentage increases linearly with their body weight. Our findings indicated that HFD significantly raises body weight levels. The results obtained matched those of Huang et al. (2023), who found a rise in the body weight of HFD-induced obese recipient mice. However, the administration of CA-Lip to obese rats resulted in a notable enhancement in weight loss, suggesting a promising therapeutic avenue for obesity management. A study by Xu et al. (2020) found that administrated CFA at 50 mg/kg BW by daily gavage for 12 weeks significantly reduced body weight in HFD mice. The bioavailability of CA is increased considerably when delivered in liposomal form, allowing for more pronounced biological effects at lower doses. Our results were supported by histopathological examinations, which revealed that CA-Lip could effectively suppress the HFD-associated increase in abdominal fat accumulation and adipocyte size. These findings imply that consuming CA-Lip may be a viable approach for managing and preventing overweight and obesity. Glucose homeostasis encompasses all metabolic processes that help regulate blood sugar levels. Obesity can lead to insulin resistance (IR) due to chronic hyperinsulinemia and inflammatory mediator interference with insulin signaling molecules (Rohm et al., 2022). HFD-fed rats exhibited a rise in fasting blood glucose, insulin, and HOMA-IR. The same findings were found by Huang et al. (2024). Fasting blood glucose, insulin, and HOMA-IR were markedly decreased in CA-Lip in HFD-fed rats. The reduction in HOMA-IR suggests an improvement in insulin sensitivity. CA treatment in chronic restraint stress-induced insulin-resistance mice reduced fasting blood sugar, oxidative stress, and improved insulin sensitivity (Choudhary et al., 2016). Castro et al. (2021) found that treatment with CA (50 mg/kg) managed to lower glucose levels in diabetic rats. These findings support the high therapeutic potential of nanoliposome-encapsulated CA in managing obesity-related hyperglycemia and IR. HFD-induced hyperlipidemia is a suitable model due to its ability to mimic pathological conditions in humans and rats. Feeding with an HFD caused a significant elevation of serum TC, TG, VLDL-C, and LDL-C levels and a marked decrease in HDL-C levels compared to rats fed a normal diet. According to Eynde et al. (2023) , feeding rats HFD results in hyperlipidemia, as demonstrated by a considerable drop in serum HDL-C levels compared to the normal group and a significant increase in serum levels of TC, LDL-C, and TG. HFD can alter fat composition and impact fat breakdown, absorption, and stomach enzyme activation. Elevated TC and LDL-C levels increase the risk of coronary heart disease, while higher HDL-C promotes cholesterol removal from the liver (Saravanan and Ponmurugan, 2012). Dietary cholesterol increases TG levels, which reduces fatty acid oxidation, thereby increasing hepatic and plasma TG levels (Huang et al., 2024). The administration of CA-Lip to HFD rats restored the increase in TG, TC, and LDL-C levels and the drop in HDL-C levels. The observed lipid-lowering effects are consistent with the established role of polyphenolic substances, such as CA, in modifying lipid profiles through inhibiting lipid peroxidation, reducing lipid accumulation, and promoting adipocytes' white-to-brown transition (Mariana et al., 2018). CA down-regulates lipogenesis gene expression of sterol regulatory element-binding protein-1 and target genes, fatty acid synthase, while promoting lipolysis through up-regulation of AMP-activated kinase (Liao et al., 2014). Obesity caused by an HFD is well-known for altering the levels of adipokines in the body. Leptin and adiponectin are types of adipocytokines that originate from fat tissue. Thus, controlling these substances is crucial in preventing metabolic diseases such as obesity (Masuyama et al., 2016). Adiponectin, a hormone produced by adipocytes, has anti-inflammatory, anti-obesity, and antidiabetic properties, while leptin regulates satiety and energy homeostasis. Obesity is linked to lower levels of adiponectin and higher levels of leptin (Clemente-Suárez et al., 2023). Furthermore, HFD-induced mice displayed elevated leptin levels and hyperleptinemia compared to control mice (Yeh et al., 2017). Rats given CA-Lip as a supplement had lower leptin levels than the non-supplemented HFD group, but their adiponectin levels were higher. Specifically, CA-Lip was found to reduce the release of abnormal adipokines by controlling the levels of leptin and adiponectin in the body. Research showed that CA increased the levels of adiponectin receptors like AdipoR1 in the liver of rats (Tian et al., 2025). Additionally, Obesity increases the body's production of pro-inflammatory adipokines, disrupting the balance between pro-inflammatory and anti-inflammatory reactions (Kawai et al., 2021). This is evidenced by the rise in serum pro-inflammatory cytokines such as TNF-α and IL-6 in rats on an HFD. Tarbiah et al. (2024) found that rats given CA (0.8/100 g diet) resulted in significant adjustments of serum proinflammatory cytokines, including IFN-γ, TNF-α, IL-6, and NF-kB in rats fed HFD. Similar results were observed in our study as oral supplementation with CA-Lip alleviated the inflammatory condition, as evidenced by the marked reduction of proinflammatory cytokines. Our findings indicated that the liposomal encapsulation likely enhanced the bioavailability and stability of CA, contributing to its effectiveness at a relatively low dose. The Hh signaling pathway is crucial in adipose tissue regulation, inhibiting adipocyte growth in vivo and in vitro. It strongly regulates the adipogenesis of white and brown adipose tissue (Zhang et al., 2020 a). The Hh signaling pathway modifies cellular metabolism through an unconventional pathway, encouraging a glycolytic state resembling the Warburg effect (Teperino et al., 2012). The Gli-1 target genes are HHIP and PTCH-1. Both can lessen Hh signaling. SMO is constitutively depressed by PTCH, and HHI interacts with the Hh ligand to prevent it from binding to PTCH, hence blocking the activation of SMO (Sigafoos et al., 2021). The activation of Hh signaling leads to the inhibition of BAT production and suppression of brown preadipocyte differentiation (Zhang et al., 2020 b). The research findings showed a notable rise in the mRNA levels of HHIP, Gli-1, Ptch-1, and Smo in obese rats compared to the group of rats in a healthy weight range. These results were consistent with other findings (Khater et al., 2023). CA-Lip administration in our study can reduce obesity by inhibiting the activation of the Hh pathway. These findings align with prior research indicating that attenuation of Hh signaling blocked early brown-preadipocyte differentiation, inhibits BAT formation in vivo, and results in the replacement of neck BAT with poorly differentiated skeletal muscle (Nosavanh et al., 2015), highlighting the potential of CA-Lip as a novel therapeutic approach in obesity-related metabolic dysfunction. Several prior studies have reported similar mechanisms involving phenolic compounds. In a study by Aslam et al. (2022), quercetin could ameliorate liver fibrosis by antagonizing the key mediators of the Hh pathway and inflammation [Sonic Hedgehog (Shh), Indian hedgehog (Ihh), patched homolog 1 (Ptch-1), Smo, Hh-interacting protein (Hhip), Gli-3]. Additionally, CA-Lip group showed a decrease in expression of HHIP, Gli-1, Ptch-1, and Smo compared to the control group, suggesting that CA-Lip has regulatory effects even under standard diet conditions. In contrast, high fat exhibited a significant increase, reinforcing the limited impact of a standard diet alone without therapeutic intervention. These comparisons support the conclusion that the observed molecular effects are primarily driven by the CA-Lip treatment. ConclusionThese findings support the potential application of CA-Lip as a promising nanotherapeutic strategy for the prevention and treatment of obesity and its related metabolic disorders. CA-Lip treatment significantly reduced body weight, improved insulin sensitivity, normalized lipid profiles, modulated adipokines, and suppressed systemic inflammation. It also downregulated key genes in the Hh signaling pathway and ameliorated adipocyte hypertrophy. Further research is needed to optimize the use of CA-Lip as an anti-obesity agent. Future studies are required to establish a full dose–response profile to determine the minimal effective dose, evaluate the potential for greater therapeutic benefit at higher doses, and assess any associated toxicity. Furthermore, the efficacy and safety of CA-Lip need to be confirmed through preclinical safety assessments and pharmacokinetic analyses, with well-designed human trials required to validate its potential for obesity treatment. AcknowledgmentsNot applicable. Conflict of interestThe authors declare no conflict of interest. FundingNot applicable. Authors' contributionsEach author contributed to the study's conception; W.S.A., S.I.K., and T.K. contributed to the design, performed the experiments, and analyzed the data. O.M.Y., H. S. S., M. M. L., and E. M. M. Y. performed the experiments, data acquisition, and data analysis. M.M.A. and A.M.F. performed statistical analysis, data analysis, wrote, reviewed, and edited the manuscript. The final paper was read and approved by all authors. Data availabilityThe corresponding author can provide the datasets used and/or analyzed during the current investigation upon reasonable request. ReferencesAbd El-emam, M.M., Mostafa, M., Farag, A.A., Youssef, H.S., El-Demerdash, A.S., Bayoumi, H., Gebba, M.A., El-Halawani, S.M., Saleh, A.M., Badr, A.M. and El Sayed, S. 2023. The potential effects of quercetin-loaded nanoliposomes on amoxicillin/clavulanate-induced hepatic damage: targeting the sirt1/nrf2/nf-κb signaling pathway and microbiota modulation. Antioxidants 12, 1487; doi:10.3390/ANTIOX12081487 Alaaeldin, E., Mostafa, M., Mansour, H.F. and Soliman, G.M. 2021. Spanlastics as an efficient delivery system for the enhancement of thymoquinone anticancer efficacy: fabrication and cytotoxic studies against breast cancer cell lines. J. Drug Deliv. Sci. Technol. 65, 102725; doi:10.1016/J.JDDST.2021.102725 Aron-Wisnewsky, J., Warmbrunn, M.V., Nieuwdorp, M. and Clément, K. 2021. Metabolism and metabolic disorders and the microbiome: the intestinal microbiota associated with obesity, lipid metabolism, and metabolic health—pathophysiology and therapeutic strategies. Gastroenterology 160, 573–599; doi:10.1053/J.GASTRO.2020.10.057 Aslam, A., Sheikh, N., Shahzad, M., Saeed, G., Fatima, N. and Akhtar, T. 2022. Quercetin ameliorates thioacetamide-induced hepatic fibrosis and oxidative stress by antagonizing the Hedgehog signaling pathway. J. Cell. Biochem. 123, 1356–1365; doi:10.1002/JCB.30296 Bancroft, J.D. and Layton, C. 2013. The Hematoxylin and Eosin. 7th Edition. In: Suvarna, S.K., Layton, C. and Bancroft, J.D., Eds., Theory & Practice of Histological Techniques, Oxford, UK: Churchill Livingstone Elsevier. Bastías-Pérez, M., Serra, D. and Herrero, L. 2020. Dietary Options for Rodents in the Study of Obesity. Nutrients 12, 1–18; doi:10.3390/NU12113234 Bounegru, A.V. and Apetrei, C. 2020. Voltamperometric sensors and biosensors based on carbon nanomaterials used for detecting caffeic acid—a review. Int. J. Mol. Sci. 21, 9275; doi:10.3390/IJMS21239275 Castro, M.F.V., Stefanello, N., Assmann, C.E., Baldissarelli, J., Bagatini, M.D., Da Silva, A.D., Da Costa, P., Borba, L., Da Cruz, I.B.M., Morsch, V.M. and Schetinger, M.R.C. 2021. Modulatory effects of caffeic acid on purinergic and cholinergic systems and oxi-inflammatory parameters of streptozotocin-induced diabetic rats. Life Sci. 277, 119421; doi: 10.1016/J.LFS.2021.119421 Chelliah, R., Rubab, M., Vijayalakshmi, S., Karuvelan, M., Barathikannan, K. and Oh, D.H. 2025. Liposomes for drug delivery: classification, therapeutic applications, and limitations. Next Nanotechnol. 8, 100209; doi:10.1016/J.NXNANO.2025.100209 Chen, Q., Yang, Z., Liu, H., Man, J., Oladejo, A.O., Ibrahim, S., Wang, S. and Hao, B. 2024. Novel Drug Delivery Systems: an Important Direction for Drug Innovation Research and Development. Pharmaceutics 16, 674. Chen, T.W., Rajaji, U., Chen, S.M., Govindasamy, M., Paul Selvin, S.S., Manavalan, S. and Arumugam, R. 2019. Sonochemical synthesis of graphene oxide sheets supported Cu2S nanodots for high sensitive electrochemical determination of caffeic acid in red wine and soft drinks. Composites Part B. Eng. 158, 419–427; doi:10.1016/J.COMPOSITESB.2018.09.099 Chou, H.W., Hung, H.C., Lin, C.H., Lin, A.C., Du, Y.F., Cheng, K.P., Li, C.H., Chang, C.J., Wu, H.T. and Ou, H.Y. 2021. The serum concentrations of hedgehog-interacting protein, a novel biomarker, were decreased in overweight or obese subjects. J. Clin. Med. 10, 742; doi:10.3390/JCM10040742 Choudhary, S., Mourya, A., Ahuja, S., Sah, S.P. and Kumar, A. 2016. Plausible anti-inflammatory mechanism of resveratrol and caffeic acid against chronic stress-induced insulin resistance in mice. Inflammopharmacology 24, 347–361; doi:10.1007/S10787-016-0287-Y/FIGURES/5 Cioffi, F., Giacco, A., Petito, G., de Matteis, R., Senese, R., Lombardi, A., de Lange, P., Moreno, M., Goglia, F., Lanni, A. and Silvestri, E. 2022. Altered mitochondrial quality control in rats with metabolic dysfunction-associated fatty liver disease (MAFLD) induced by high-fat feeding. Genes (Basel). 13(2), 315; doi:10.3390/genes13020315 Clemente-Suárez, V.J., Redondo-Flórez, L., Beltrán-Velasco, A.I., Martín-Rodríguez, A., Martínez-Guardado, I., Navarro-Jiménez, E., Laborde-Cárdenas, C.C. and Tornero-Aguilera, J.F. 2023. The role of adipokines in health and disease. Biomedicines 11, 1290; doi:10.3390/BIOMEDICINES11051290 Erfan, O. 2017. Effect of garlic on rat diabetic renal cortex. A histological and Immunohistochemical Study. Egypt. J. Anatomy 40, 253–264; doi:10.21608/EJANA.2018.16904 Expert Panel Report: Guidelines (2013) for the management of overweight and obesity in adults, 2014. Obesity 22 Suppl 2, S41–S410. https://doi.org/10.1002/OBY.20660 Eynde, V., Dine Tariq Bouhlali, E., Hmidani, A., Bourkhis, B., Moussafir, Z., Filali-Zegzouti, Y. and Alem, C. 2023. Date (Phoenix dactylifera L.) Fruits as a potential lipid-lowering therapy: effect on high-fat diet and Triton-WR-1339-induced hyperlipidemic rats. Drugs and Drug Candidates 2, 422–432; doi: 10.3390/DDC2020021 Huang, C.H., Hsu, H.S. and Chiang, M.T. 2024. Influence of varied dietary cholesterol levels on lipid metabolism in hamsters. Nutrients 16, 2472; doi:10.3390/NU16152472 Huang, K.C., Chuang, P.Y., Yang, T.Y., Tsai, Y.H., Li, Y.Y. and Chang, S.F. 2024. Diabetic rats induced using a high-fat diet and low-dose streptozotocin treatment exhibit gut microbiota dysbiosis and osteoporotic bone pathologies. Nutrients 16, 1220; doi: 10.3390/NU16081220 Huang, Y., Ying, N., Zhao, Q., Chen, J., Teow, S.Y., Dong, W., Lin, M., Jiang, L. and Zheng, H. 2023. Amelioration of obesity-related disorders in high-fat diet-fed mice following fecal microbiota transplantation from inulin-dosed mice. Molecules 28, doi: 3997; 10.3390/MOLECULES28103997 Kawai, T., Autieri, M.V. and Scalia, R. 2021. Adipose tissue inflammation and metabolic dysfunction in obesity. Am. J. Physiol. Cell. Physiol. 320, C375–C391; doi:10.1152/AJPCELL.00379.2020 Khater, S., Shalabi, M., Alammash, B., Alrais, A., Ahmadi, D., Alqahtani, L., Khameis, T., Abdelaziz, S., Elkelish, A. and Dawy, K. 2023. Evaluation of flaxseed lignan-enriched extract targeting autophagy, apoptosis, and hedgehog pathways against experimentally induced obesity. J. Adv. Vet. Anim. Res. 10, 321–335; doi:10.5455/JAVAR.2023.J684 Kim, H.M., Kim, Y., Lee, E.S., Huh, J.H. and Chung, C.H. 2018. Caffeic acid ameliorates hepatic steatosis and reduces ER stress in high fat diet-induced obese mice by regulating autophagy. Nutrition 55–56(56), 63–70; doi:10.1016/J.NUT.2018.03.010 Lasker, S., Rahman, M.M., Parvez, F., Zamila, M., Miah, P., Nahar, K., Kabir, F., Sharmin, S.B., Subhan, N., Ahsan, G.U. and Alam, M.A. 2019. High-fat diet-induced metabolic syndrome and oxidative stress in obese rats are ameliorated by yogurt supplementation. Scientific Rep. 9(1), 1–15; doi:10.1038/s41598-019-56538-0 Liao, C.C., Ou, T.T., Huang, H.P. and Wang, C.J. 2014. The inhibition of oleic acid induced hepatic lipogenesis and the promotion of lipolysis by caffeic acid via up-regulation of AMP-activated kinase. J. Sci. Food Agric. 94, 1154–1162; doi:10.1002/JSFA.6386 Livak, K.J. and Schmittgen, T.D. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408; doi:10.1006/METH.2001.1262 Mansour, M.F., Behairy, A., Mostafa, M., Khamis, T., Alsemeh, A.E., Ahmed, N.M.Q. and El-Emam, M.M.A. 2025. Quercetin-loaded PEGylated liposomes alleviate testicular dysfunction in alloxan-induced diabetic rats: the role of Kisspeptin/Neurokinin B/Dynorphin pathway. Toxicol. Appl. Pharmacol. 499, 117337; doi:10.1016/J.TAAP.2025.117337 Mariana, B.D., Tiago, L.S., Ramon, R.P.P.B.D.M., Jamile, M.F., Tiago, S.M., Richard, R.C.M., Hector, G.R., Dânya, B.L., Alice, M.C.M. and Maria, G.R.D.Q. 2018. Caffeic acid reduces lipid accumulation and reactive oxygen species production in adipocytes. Afr. J. Pharm. Pharmacol. 12, 263–268; doi:10.5897/AJPP2018.4937 Masuyama, H., Mitsui, T., Eguchi, T., Tamada, S. and Hiramatsu, Y. 2016. The effects of paternal high-fat diet exposure on offspring metabolism with epigenetic changes in the mouse adiponectin and leptin gene promoters. Am. J. Physiol. Endocrinol. Metab. 311, E236–E245; doi:10.1152/AJPENDO.00095.2016 Mccafferty, B.J., Hill, J.O. and Gunn, A.J. 2020. Obesity: scope, lifestyle interventions, and medical management. Tech. Vasc. Interv. Radiol. 23, 100653; doi: 10.1016/J.TVIR.2020.100653 Nosavanh, L., Yu, D.H., Jaehnig, E.J., Tong, Q., Shen, L. and Chen, M.H. 2015. Cell-autonomous activation of Hedgehog signaling inhibits brown adipose tissue development. Proc. Natl. Acad. Sci. U. S. A. 112, 5069–5074; doi:10.1073/PNAS.1420978112 Payab, M., Hasani‐Ranjbar, S., Shahbal, N., Qorbani, M., Aletaha, A., Haghi‐Aminjan, H., Soltani, A., Khatami, F., Nikfar, S., Hassani, S., Abdollahi, M. and Larijani, B. 2020. Effect of the herbal medicines in obesity and metabolic syndrome: a systematic review and meta-analysis of clinical trials. Phytotherapy Res. 34, 526–545; doi:10.1002/PTR.6547 Ramalho, M.J., Coelho, M.A.N. and Pereira, M.C. 2018. Nanocarriers for the delivery of temozolomide in the treatment of glioblastoma: a review. Design Dev. New Nanocarriers 511, 687–722; doi: 10.1016/B978-0-12-813627-0.00018-1 Rohm, T.V., Meier, D.T., Olefsky, J.M. and Donath, M.Y. 2022. Inflammation in obesity, diabetes and related disorders. Immunity 55, 31; doi:10.1016/J.IMMUNI.2021.12.013 Salem, G.A., Mohamed, A.A.R., Khater, S.I., Noreldin, A.E., Alosaimi, M., Alansari, W.S., Shamlan, G., Eskandrani, A.A., Awad, M.M., El-Shaer, R.A.A., Nassan, M.A., Mostafa, M. and Khamis, T. 2023. Enhancement of biochemical and genomic pathways through lycopene-loaded nano-liposomes: alleviating insulin resistance, hepatic steatosis, and autophagy in obese rats with non-alcoholic fatty liver disease: Involvement of SMO, GLI-1, and PTCH-1 genes. Gene 883, 147670; doi: 10.1016/J.GENE.2023.147670 Saravanan, G. and Ponmurugan, P. 2012. Ameliorative potential of S-allylcysteine: effect on lipid profile and changes in tissue fatty acid composition in experimental diabetes. EToxP 64, 639–644; doi:10.1016/J.ETP.2010.12.007 Shahin, N.N., Shamma, R.N. and Ahmed, I.S. 2022. A Nano-Liposomal Formulation of Caffeic Acid Phenethyl Ester Modulates Nrf2 and NF-κβ Signaling and Alleviates Experimentally Induced Acute Pancreatitis in a Rat Model. Antioxidants (Basel). 11; doi: 10.3390/ANTIOX11081536 Shi, Y. and Long, F. 2017. Hedgehog signaling via Gli2 prevents obesity induced by high-fat diet in adult mice. Elife 6, e31649; doi: 10.7554/ELIFE.31649 Sigafoos, A.N., Paradise, B.D. and Fernandez-Zapico, M.E. 2021. Hedgehog/GLI signaling pathway: transduction, regulation, and implications for disease. Cancers 13, 3410; doi:10.3390/CANCERS13143410 Stasiłowicz-Krzemień, A., Rosiak, N., Miklaszewski, A. and Cielecka-Piontek, J. 2023. Screening of the anti-neurodegenerative activity of caffeic acid after introduction into inorganic metal delivery systems to increase its solubility as the result of a mechanosynthetic approach. Int. J. Mol. Sci. 24, 9218; doi:10.3390/IJMS24119218/S1 Tak, Y.J. and Lee, S.Y. 2021. Long-term efficacy and safety of anti-obesity treatment: where do we stand?. Curr. Obes. Rep. 10, 14–30; doi:10.1007/S13679-020-00422-W/TABLES/2 Tam, B.T., Morais, J.A. and Santosa, S. 2020. Obesity and ageing: two sides of the same coin. Obes. Rev. 21, e12991; doi: 10.1111/OBR.12991 Tarbiah, N.I., Khalifa, F.K., Alkhattabi, N.A., Banjabi, A.A., Ghazali, R.F., Alzahri, R.Y., Almerabi, N.A. and Mansour, A.A. 2024. Synergistic effect of ferulic acid and caffeic acid on metabolic syndrome and immune response in rats. Ann. Clin. Anal. Med. 15, doi: 42–46; 10.4328/ACAM.22005 Teperino, R., Amann, S., Bayer, M., Mcgee, S.L., Loipetzberger, A., Connor, T., Jaeger, C., Kammerer, B., Winter, L., Wiche, G., Dalgaard, K., Selvaraj, M., Gaster, M., Lee-Young, R.S., Febbraio, M.A., Knauf, C., Cani, P.D., Aberger, F., Penninger, J.M., Pospisilik, J.A. and Esterbauer, H. 2012. Hedgehog partial agonism drives Warburg-like metabolism in muscle and brown fat. Cell 151, 414–426; doi:10.1016/J.CELL.2012.09.021 Tian, W., Gonzales, G.B., Wang, H., Yang, Y., Tang, C., Zhao, Q., Zhang, J., Zhang, H. and Qin, Y. 2025. Caffeic acid and chlorogenic acid mediate the ADPN-AMPK-PPARα pathway to improve fatty liver and production performance in laying hens. J. Anim. Sci. Biotechnol. 16, 49; doi:10.1186/S40104-025-01175-Z Vekic, J., Zeljkovic, A., Stefanovic, A., Jelic-Ivanovic, Z. and Spasojevic-Kalimanovska, V. 2019. Obesity and dyslipidemia. Metabolism 92, 71–81; doi:10.1016/J.METABOL.2018.11.005 Wang, L., Yan, Y., Wu, L. and Peng, J. 2023. Natural products in non-alcoholic fatty liver disease (NAFLD): novel lead discovery for drug development. Pharmacol. Res. 196, 106925; doi:10.1016/J.PHRS.2023.106925 Wei, H., Li, J., Shi, S., Zhang, L., Xiang, A., Shi, X., Yang, G. and Chu, G. 2019. Hhip inhibits proliferation and promotes differentiation of adipocytes through suppressing hedgehog signaling pathway. Biochem. Biophys. Res. Commun. 514, 148–156; doi:10.1016/J.BBRC.2019.04.047 WHO. 2023. Obesity and overweight. Geneva, Switzerland: WHO. Available via https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (Accessed 16 September 2025) Xu, J., Ge, J., He, X., Sheng, Y., Zheng, S., Zhang, C., Xu, W. and Huang, K. 2020. Caffeic acid reduces body weight by regulating gut microbiota in diet-induced-obese mice. J. Funct. Foods 74, 104061; doi:10.1016/J.JFF.2020.104061 Yan, J., Bak, J., Go, Y., Park, J., Park, M., Lee, H.J. and Kim, H. 2023. Scytosiphon lomentaria extract ameliorates obesity and modulates gut microbiota in high-fat-diet-fed mice. Nutrients 15, 58; doi: 10.3390/NU15040815/S1 Yeh, C.W., Liu, H.K., Lin, L.C., Liou, K.T., Huang, Y.C., Lin, C.H., Tzeng, T.T., Shie, F.S., Tsay, H.J. and Shiao, Y.J. 2017. Xuefu Zhuyu decoction ameliorates obesity, hepatic steatosis, neuroinflammation, amyloid deposition and cognition impairment in metabolically stressed APPswe/PS1dE9 mice. J. Ethnopharmacol. 209, 50–61; doi:10.1016/J.JEP.2017.07.036 Zhang, Z., Zhang, X.X., Liu, Z.F., Guo, X.R., Cui, X.W., Ji, C.B., Zhong, H. and Chi, X. 2020. Inhibition of hedgehog signaling promotes white adipose tissue browning. Mol. Cell. Endocrinol. 518, 110970; doi:10.1016/J.MCE.2020.110970 | ||
| How to Cite this Article |
| Pubmed Style Alansari WS, El-emam MMA, Altalhi R, Khater SI, Khamis T, Sherkawy HS, Fares AM, Lotfy MM, Youssef OM, Yassin EMM. Therapeutic efficacy of liposomal caffeic acid in high-fat diet-induced obesity in rats: Targeting hedgehog signalling pathway, adipokines, and insulin resistance. Open Vet. J.. 2025; 15(10): 4936-4948. doi:10.5455/OVJ.2025.v15.i10.13 Web Style Alansari WS, El-emam MMA, Altalhi R, Khater SI, Khamis T, Sherkawy HS, Fares AM, Lotfy MM, Youssef OM, Yassin EMM. Therapeutic efficacy of liposomal caffeic acid in high-fat diet-induced obesity in rats: Targeting hedgehog signalling pathway, adipokines, and insulin resistance. https://www.openveterinaryjournal.com/?mno=264853 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.13 AMA (American Medical Association) Style Alansari WS, El-emam MMA, Altalhi R, Khater SI, Khamis T, Sherkawy HS, Fares AM, Lotfy MM, Youssef OM, Yassin EMM. Therapeutic efficacy of liposomal caffeic acid in high-fat diet-induced obesity in rats: Targeting hedgehog signalling pathway, adipokines, and insulin resistance. Open Vet. J.. 2025; 15(10): 4936-4948. doi:10.5455/OVJ.2025.v15.i10.13 Vancouver/ICMJE Style Alansari WS, El-emam MMA, Altalhi R, Khater SI, Khamis T, Sherkawy HS, Fares AM, Lotfy MM, Youssef OM, Yassin EMM. Therapeutic efficacy of liposomal caffeic acid in high-fat diet-induced obesity in rats: Targeting hedgehog signalling pathway, adipokines, and insulin resistance. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4936-4948. doi:10.5455/OVJ.2025.v15.i10.13 Harvard Style Alansari, W. S., El-emam, . M. M. A., Altalhi, . R., Khater, . S. I., Khamis, . T., Sherkawy, . H. S., Fares, . A. M., Lotfy, . M. M., Youssef, . O. M. & Yassin, . E. M. M. (2025) Therapeutic efficacy of liposomal caffeic acid in high-fat diet-induced obesity in rats: Targeting hedgehog signalling pathway, adipokines, and insulin resistance. Open Vet. J., 15 (10), 4936-4948. doi:10.5455/OVJ.2025.v15.i10.13 Turabian Style Alansari, Wafa S., Mahran Mohamed Abd El-emam, Rawan Altalhi, Safaa I. Khater, Tarek Khamis, Hoda S. Sherkawy, Abdelhamid M. Fares, Marwa M. Lotfy, Ola Mohammed Youssef, and Engy Mohamed Mohamed Yassin. 2025. Therapeutic efficacy of liposomal caffeic acid in high-fat diet-induced obesity in rats: Targeting hedgehog signalling pathway, adipokines, and insulin resistance. Open Veterinary Journal, 15 (10), 4936-4948. doi:10.5455/OVJ.2025.v15.i10.13 Chicago Style Alansari, Wafa S., Mahran Mohamed Abd El-emam, Rawan Altalhi, Safaa I. Khater, Tarek Khamis, Hoda S. Sherkawy, Abdelhamid M. Fares, Marwa M. Lotfy, Ola Mohammed Youssef, and Engy Mohamed Mohamed Yassin. "Therapeutic efficacy of liposomal caffeic acid in high-fat diet-induced obesity in rats: Targeting hedgehog signalling pathway, adipokines, and insulin resistance." Open Veterinary Journal 15 (2025), 4936-4948. doi:10.5455/OVJ.2025.v15.i10.13 MLA (The Modern Language Association) Style Alansari, Wafa S., Mahran Mohamed Abd El-emam, Rawan Altalhi, Safaa I. Khater, Tarek Khamis, Hoda S. Sherkawy, Abdelhamid M. Fares, Marwa M. Lotfy, Ola Mohammed Youssef, and Engy Mohamed Mohamed Yassin. "Therapeutic efficacy of liposomal caffeic acid in high-fat diet-induced obesity in rats: Targeting hedgehog signalling pathway, adipokines, and insulin resistance." Open Veterinary Journal 15.10 (2025), 4936-4948. Print. doi:10.5455/OVJ.2025.v15.i10.13 APA (American Psychological Association) Style Alansari, W. S., El-emam, . M. M. A., Altalhi, . R., Khater, . S. I., Khamis, . T., Sherkawy, . H. S., Fares, . A. M., Lotfy, . M. M., Youssef, . O. M. & Yassin, . E. M. M. (2025) Therapeutic efficacy of liposomal caffeic acid in high-fat diet-induced obesity in rats: Targeting hedgehog signalling pathway, adipokines, and insulin resistance. Open Veterinary Journal, 15 (10), 4936-4948. doi:10.5455/OVJ.2025.v15.i10.13 |