| Research Article | ||
Open Vet. J.. 2025; 15(10): 4912-4927 Open Veterinary Journal, (2025), Vol. 15(10): 4912-4927 Research Article Infrared thermography assessment of body surface temperature in Komodo dragons (Varanus komodoensis) during the Komodo island dry seasonMahfud Mahfud1,2, Agik Suprayogi3, Koekoeh Santoso3, and Nyoto Santoso4 and Srihadi Agungpriyono5*1Department of Biology Education, University of Muhammadiyah Kupang, Kupang, Indonesia 2Postgraduate Student of School of Veterinary Medicine and Biomedical Sciences, Bogor Agricultural University (IPB), Bogor, Indonesia 3Division of Physiology, School of Veterinary Medicine and Biomedical Science, Bogor Agricultural University (IPB), Bogor, Indonesia 4Department of Forest Resources Conservation and Ecotourism, Faculty of Forestry, Bogor Agricultural University (IPB), Bogor, Indonesia 5Division of Anatomy, Histology and Embryology, Global Health Agromaritim One Health Collaborating Center, School of Veterinary Medicine and Biomedical Science, Bogor Agricultural University (IPB), Bogor, Indonesia *Corresponding Author: Srihadi Agungrinyono. Division of Anatomy, Histology and Embryology, Global Health Agromaritim One Health Collaborating Center, School of Veterinary Medicine and Biomedical Science, Bogor Agricultural University (IPB), Bogor, Indonesia. Email: ysrihadi [at] apps.ipb.ac.id Submitted: 18/06/2025 Revised: 15/09/2025 Accepted: 29/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: Accurate assessment of the relationship between an animal’s physiology and its surrounding environment is essential for understanding ecological adaptability, particularly with respect to body temperature regulation. The large body size and aggressive nature of Komodo dragons (Varanus komodoensis) pose challenges to direct physiological monitoring, highlighting the importance of non-invasive techniques. In this study, infrared thermography (IRT) was employed as a non-invasive tool to estimate surface body temperature, serving as a proxy for internal temperature regulation. Aim: This study aimed to evaluate variations in the surface body temperature of Komodo dragons in response to environmental temperature dynamics on Komodo Island, within Komodo National Park, using IRT. Methods: Fieldwork was conducted in June 2024 at the Loh Liang tourist site, Komodo Island, East Nusa Tenggara, Indonesia. The surface body temperatures of nine adult Komodo dragons were recorded alongside ambient temperature measurements using a FLIR ONE IRT camera (compatible with iOS and Android) at a distance of approximately 2 m. Temperature data were collected from the thoracic (costal and pectoral) and dorsal regions at 15 to 30-minute intervals between 10:00 AM and 4:00 PM daily. The environmental and body temperature data were analyzed using multiple linear regression models. Results: The ambient temperature ranged from 19.80°C to 56.50°C, reflecting the high variability associated with solar exposure and shade. In contrast, the surface body temperatures of the Komodo dragon remained relatively stable, ranging from 27.40°C to 35.40°C. Statistical analysis indicated that the minimum ambient temperature had a significant effect on the surface body temperature, whereas the maximum temperature had a minimal influence. These findings indicate that Komodo dragons primarily rely on behavioral and physiological thermoregulatory mechanisms to maintain thermal homeostasis under fluctuating environmental conditions. Conclusion: Komodo dragons maintain thermal stability through adaptive behaviors, such as basking, shade-seeking, retreating to sheltered areas, and adjusting activity levels. These thermoregulatory strategies are essential for survival in dynamic thermal environments. Further research integrating additional environmental and physiological parameters is recommended to enhance ecological understanding and inform conservation strategies. Keywords: Varanus komodoensis, Temperature, Infrared thermography, Thermoregulation. IntroductionThe Komodo dragon (Varanus komodoensis), a species endemic to Indonesia, belongs to the Varanidae family (Böhme, 2003). This species is found on the island of Flores, including the isolated Longos and Ontoloe Islands, as well as within the Komodo National Park, which consists of Komodo Island, Rinca Island, Padar Island, Nusa Kode, and Gili Motang (Jessop et al., 2021). The species is currently classified as vulnerable by the International Union for Conservation of Nature (IUCN) Red List and is listed in Appendix I of CITES (Jessop et al., 2021; Shelley and Metz, 2023). A combination of ecological and anthropogenic factors primarily threatens the conservation of the Komodo dragon. Human activities, including infrastructure development and tourism, have led to habitat fragmentation, thereby reducing the natural roaming areas of these reptiles (Jessop et al., 2018; Purwandana et al., 2021). Additionally, global climate change poses a risk by potentially increasing extreme temperatures and reducing prey availability, which could adversely affect the survival of wild Komodo dragon populations (Shine and Somaweera, 2019; Jones et al., 2020). Another critical threat is the hunting of their main prey, such as deer (Cervus timorensis), which diminishes the food resources available to the Komodo dragons (Ariefiandy et al., 2016; Purwandana et al., 2021). Negative interactions with humans, such as conflicts in tourist areas (Jessop et al., 2018; Permatasari et al., 2020) and risks associated with diseases and pathogens (Bull et al., 2010), further disrupt their natural behavior. Therefore, a comprehensive conservation strategy that incorporates habitat protection, the regulation of tourism activities, and measures to mitigate the impacts of climate change is essential. The Indonesian government has implemented a range of conservation strategies, including the establishment of Komodo National Park as a central protected zone (Government of the Republic of Indonesia, 1990). Measures to curtail the hunting of Komodo prey have been implemented (Ariefiandy et al., 2013a; Jessop et al., 2020), complemented by the integration of technology-driven monitoring systems, such as camera traps (Ariefiandy et al., 2013b), and the SMART patrol application for tracking population dynamics (Directorate General of Natural Resources and Ecosystem Conservation, 2024). Local community engagement and tourism management reforms have also been introduced to balance conservation with economic interests (Walpole and Goodwin, 2001; Jessop et al., 2018; Government of the Republic of Indonesia, 2024). However, a sharp 416% increase in tourist visits between 2021 and 2024 (Antara News, 2025) has shifted the perception of the Komodo dragon from an ecological treasure to an economic commodity. Tourism-driven exploitation can overlook key aspects of animal welfare, defined as the fulfillment of biological needs (Bracke et al., 2001). Unmet needs can lead to physiological stress, particularly when environmental conditions are suboptimal (Çırak and Esendal, 2006; Hilmer, 2010). Among environmental stressors, thermal stress is particularly relevant for ectothermic species such as the Komodo dragon. It is critical to understand how environmental factors influence Komodo dragon physiology, particularly body temperature. However, the large size of the species and its predatory behavior present safety challenges for researchers (Laver et al., 2012; Boyd et al., 2021). Therefore, a non-invasive, accurate, and safe method for body temperature measurement is essential. Infrared thermography (IRT) offers a promising alternative, enabling remote surface temperature assessment without physical or chemical restraint (McCafferty, 2007; Narayan et al., 2019; Mota-Rojas et al., 2022). Surface temperature measurements taken via IRT have been shown to correlate with core temperatures, such as rectal readings, in various animal species (Švejdová et al., 2013). IRT is cost-effective, allows data collection from a distance of up to 1 km, and minimizes stress and risk to both animals and observers (Barroso et al., 2020; Williams, 2023; Afonso et al., 2024). This study aims to investigate the relationship between the ambient environmental temperature and the surface body temperature of Komodo dragons using IRT in their natural habitat. We hypothesize that the minimum ambient temperature has a stronger effect on the body surface temperature of Komodo dragons than the maximum ambient temperature due to their behavioral thermoregulation. The findings of this study are expected to enhance the understanding of thermoregulation in Komodo dragons and support non-invasive health monitoring strategies essential for their conservation. Materials and MethodsStudy areaThe study was conducted in June 2024 at the Loh Liang tourist site, located on Komodo Island, Flores, Manggarai Regency, East Nusa Tenggara, Indonesia (Fig. 1). Data collection was carried out during the dry season in Komodo’s natural habitat. Stable climatic conditions with minimal rainfall marked this period, reducing environmental variability and facilitating the collection of more reliable data on body-surface temperature patterns. The research permit period approved by the relevant authorities determined the timing of the fieldwork, which also fell in June. Loh Liang is renowned for facilitating direct observation of Komodo dragons and is characterized by its dry tropical vegetation. This ecotourism destination attracts visitors from early morning until late afternoon. A dry climate, hilly terrain, and relatively nutrient-poor soil characterize the vegetation in this area, resulting in a sparse and scattered pattern.
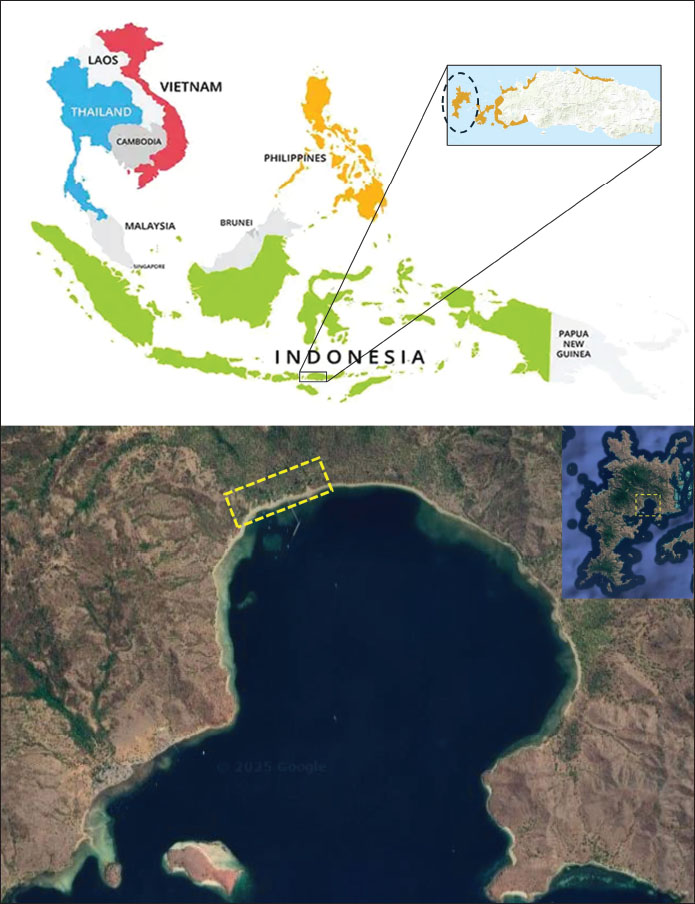
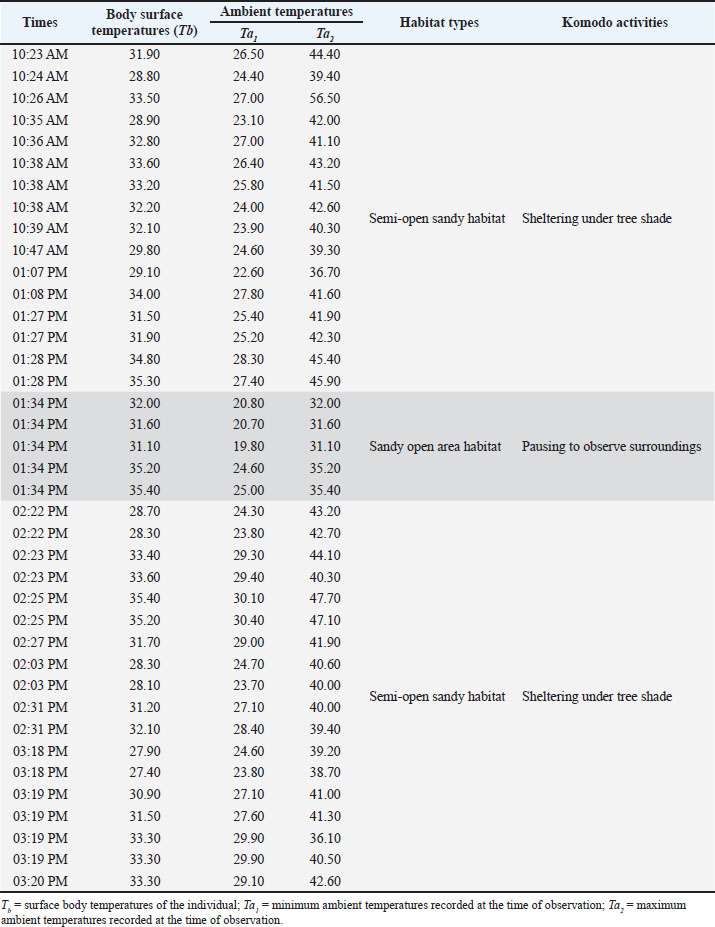
Fig. 1. The research location is on Komodo Island, Indonesia. The top panel shows the position of Komodo Island in Southeast Asia and East Nusa Tenggara Province. The bottom panel displays satellite images of the coastal area of Loh Liang tourism on Komodo Island (yellow rectangle), where infrared thermography data was collected on the surface temperature of the Komodo dragon. Data collectionDuring the data collection period, Komodo dragons were observed traversing and taking refuge along the coastline of Loh Liang Beach, which is situated at elevations between 5 and 20 m above sea level. The study focused on recording the surface body temperature of the Komodo dragons, as well as the ambient environmental temperature, which included both minimum and maximum values, from 10:00 AM to 04:00 PM. The sample consisted of nine adult Komodo dragons identified at the study site. Data were collected over nine consecutive days in June 2024. Each observation session lasted approximately 15–30 minute per individual, depending on factors such as visibility, animal behavior, and the presence of tourists interacting with or photographing the Komodo dragons. On average, each Komodo dragon was observed 7–11 times daily, resulting in 63–99 individual observation sessions. To minimize the likelihood of repeated sampling of the same individual on consecutive days, we documented distinctive features such as body size and scarring. Nonetheless, repeated sampling could occur if the identifying marks were not easily discernible. The surface body temperatures of the Komodo dragons and the surrounding environmental temperatures were assessed using a FLIR ONE for IOS/Android IRT camera, specifically the ThermaCAM Research 2.10 Pro (FLIR Systems, Danderyd, Sweden). This method is noninvasive, ensuring no physical contact with animals (McCafferty, 2007; Narayan et al., 2019; Santoso et al., 2023). Measurements were conducted in real time (Church et al., 2009) from a distance of approximately 2 m to reduce stress on the animals (Mccafferty, 2007; Afonso et al., 2024), improve measurement accuracy, and minimize potential risks to both the animals and researchers (Barroso et al., 2020; Williams, 2023). Thermal images were captured from adult Komodo dragons encountered at the site without regard to sex. The success of thermal imaging is largely dependent on the selection of the correct body part for measurement, as this choice affects the thermal window data. The thermal window is defined as the range of environmental temperatures within which an organism can maintain optimal physiological functions (Pörtner, 2010; Pörtner et al., 2017). For reptiles, specifically Komodo dragons, the thermal window has not been clearly established due to differences in skin properties, fur, or body coloration compared to mammals (Mota-Rojas et al., 2022). Despite this, the eyes, as well as the snout, are often considered the most accurate indicator of internal temperature in reptiles (In Den Bosch, 1983; Sannolo, 2019; Barroso et al., 2020). However, for the safety of researchers, the body parts that were accessible for photography included the sides of the body (thoracic region), specifically the costal, pectoral, and back (dorsal) regions. These regions can be utilized as the thermal window to evaluate physiological parameters, such as body temperature and respiration rate (Lowe et al., 2019; Farrar et al., 2020; Mota-Rojas et al., 2021; Wang et al., 2021; Mota-Rojas et al., 2022), and they correlate with internal temperatures, such as rectal temperature (Švejdová et al., 2013). To enhance the measurement accuracy, the ambient temperature was input into the camera. Data analysisTo explore the correlation between the lowest and highest environmental temperatures and Komodo dragon body temperature, data were analyzed using a multiple linear regression equation with a 95% confidence interval. This analysis was performed using the XLSTAT software, Version 2016.02.28451 (License ID: 34463000). Ethical approvalThis study involved the use of protected wildlife species, which required special permits and ethical approval. Ethical approval was granted by the Animal Care and Use Ethics Committee of the National Research and Innovation Agency (Number: 016/KE.02/SK/01/2024), and observer behavior was standardized to minimize animal stress. ResultsIRT depiction of the Komodo dragon’s surface body temperature alongside its thermal physiological responseIRT was used to assess the surface skin temperature of the Komodo dragons along with the ambient temperature of their environment (Table 1, Fig. 2). Komodo dragons are known to be active from midday to late afternoon; thus, data collection was conducted between 10:23 AM and 3:20 PM, with most observations noting dragon activity in shaded areas. Sample thermal images depicting the body surface and surroundings of the Komodo dragon during the observation period are shown below (Fig. 2). Komodo dragons were observed resting under tree shade, in sandy regions, and in semi-open spaces (transitional areas between the open and shaded zones) at 10:23 AM (Fig. 2A). The thermal image recorded a surface body temperature of 28.9°C, with the ambient temperature being lower (24.6°C) in shaded areas and higher (39.3°C) in sunlit areas. At midday (1:28 PM), a Komodo dragon was found resting at the same location, with its surface body temperature rising to 35.3°C (Fig. 2B). At 1:34 PM, a Komodo dragon covered in wet sand was observed resting in an open area near the beach. The thermal image indicated a surface body temperature of 31.1°C, with a lower ambient temperature of 19.8°C (Fig. 2C), attributed to the temporary cloudy weather. In the afternoon (2:22 PM), a Komodo dragon was found resting in a semi-open shaded area under a tree lying on sandy soil, with a body temperature of 28.7°C. The lowest temperature recorded in the shaded environment was approximately 24.3°C, whereas the temperature in the sunlit areas reached approximately 43.2°C (Fig. 2D). In the late afternoon (3:20 PM), another Komodo dragon was found lying on sandy ground in a semi-open area under tree shade, with a surface body temperature of 33.3°C. At this time, the lowest recorded ambient temperature was approximately 23.1°C, while the highest reached approximately 42.6°C (Fig. 2E). Thermal imaging studies have revealed that fluctuations in the body temperature of the Komodo dragon are influenced by their activity and location (shaded/open). As ectothermic reptiles, environmental conditions significantly affect the body temperature of Komodo dragons. In shaded areas (Figs. 1D and 2A), their surface body temperature was lower (28.7°C–29.8°C). Conversely, in open (Fig. 2C) and semi-open areas (Figs. 1E and 2B), body temperature was higher (31.1°C–35.3°C), particularly at midday (Fig. 2C, 35.3°C). Komodo dragons adjust their location to regulate body temperature, such as basking to increase body temperature (Fig. 2C) and seeking shade when temperatures become excessively high (Figs. 1B, D, E and 2A). This highlights the critical role of shade in preventing overheating and the importance of sun exposure in elevating body temperature when necessary.
Fig. 2. This study investigated the body temperature distribution in Komodo dragons along with the environmental temperatures in their natural habitat. The thermal images obtained using a FLIR ONE thermal camera illustrate different observation periods. Each panel features a pair of images: a thermal image on the left and a visual photograph on the right. The highest recorded environmental temperature is highlighted with a red circle, the lowest recorded temperature at the time of measurement is marked with a blue circle, and the body temperature of the Komodo dragon at the time of measurement is indicated by a transparent circle. Table 1. Surface body temperature of Komodo dragons (Varanus komodoensis) in relation to ambient temperature, habitat type, and behavioral activities across different times of the day in Komodo National Park.
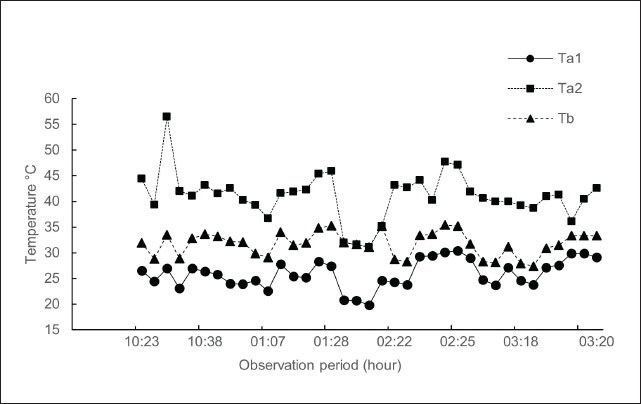
Association between the body surface temperature of the Komodo dragon and the temperature of the surrounding environmentThe ambient temperature, encompassing both its minimum and maximum values, demonstrates significant variability, ranging from 19.80°C to 56.50°C (Fig. 3). This range indicates that temperatures under natural shade conditions can experience rapid and substantial changes. Despite these pronounced fluctuations in ambient temperature, particularly with the maximum reaching 56.50°C, the body temperature of Komodo dragons remained relatively stable, confined to a range of 27.40°C–35.40°C (Fig. 3). The minimum ambient temperature varies between 19.80°C and 30.40°C. This finding illustrates the ability of Komodo dragons to sustain a stable body temperature, despite drastic changes in ambient temperature. The multiple linear regression model, which evaluates the effects of the lowest ambient temperature (Ta1) and highest ambient temperature (Ta2) on the body surface temperature (Tb) of the Komodo dragons, yielded the following equation:
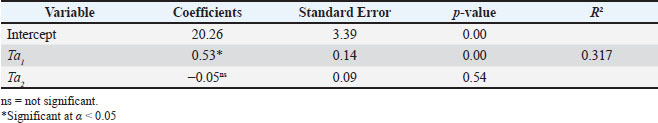
Fig. 3. During the observation period, variations were observed in the minimum environmental temperature (Ta1), maximum environmental temperature (Ta2), and body temperature of the Komodo dragons (Tb). The data revealed that Tb remained relatively constant, ranging from 27.40°C to 35.40°C, despite substantial fluctuations in environmental temperatures, particularly Ta2, which peaked at 56.50°C. Ta1 varied between 19.80°C and 30.40°C. This pattern indicates that Komodo dragons possess physiological and behavioral thermoregulatory mechanisms that allow them to maintain their body temperature near their physiological setpoints, even under changing environmental conditions. Tb=20.26 + 0.53 Ta1 ̶ 0.05 Ta2 (1) The analysis results obtained using the multiple linear regression equation are detailed in Table 2. Teble 2. Findings from the multiple linear regression analysis assessing the influence of the minimum ambient temperature (Ta1) and maximum ambient temperature (Ta2) on the body temperature of Komodo dragons (Tb).
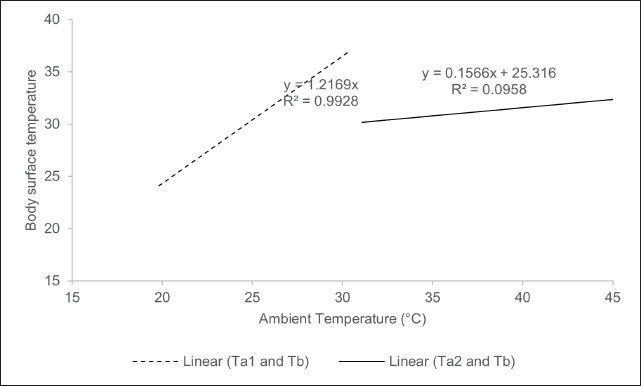
Multiple linear regression analysis indicated that the lowest ambient temperature exerted a significant influence on the body surface temperature of the Komodo dragon, with a coefficient of 0.53 (p=0.00), as shown in Table 2. This indicates that an increase of 1°C in the lowest ambient temperature corresponds to a body temperature increase of 0.53°C. Fig. 3 supports this finding by demonstrating that the body surface temperature of the Komodo dragon tends to increase more noticeably with an increase in the lowest ambient temperature. Fig. 4 illustrates the relationships between the variables. The model’s coefficient of determination (R²) was 0.317, indicating that the two environmental temperature variables can explain 31.7% of the variation in the body temperature of the Komodo dragon. The remaining variation was likely due to other factors, such as humidity, daily activity, or direct sunlight exposure, which were not included in this model. Additionally, the relationship between the highest ambient temperature and changes in the body surface temperature of the Komodo dragon was explored (Table 2, Fig. 4). The highest ambient temperature did not significantly affect the body surface temperature, with a coefficient of −0.05 (p=0.54). This indicates that variations in the highest ambient temperature do not have a substantial impact on the body surface temperature of the Komodo dragon, as a 1°C increase in the highest ambient temperature results in a 0.05°C decrease in body temperature.
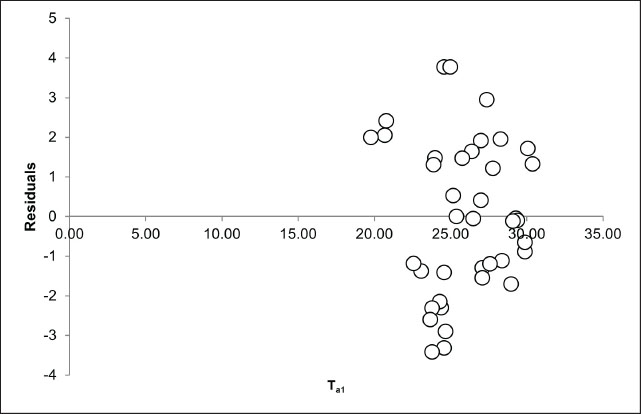
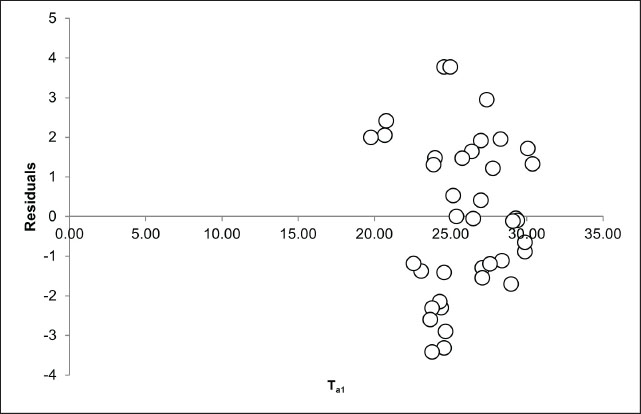
Fig. 4. The analysis of the relationship between ambient temperature (°C) and body surface temperature (°C) focused on the extremes of ambient temperature, specifically the lowest (Ta1) and highest (Ta2) values. The dashed line (---) illustrates the linear correlation between the lowest ambient temperature and body surface temperature, as described by the equation y=1.2663x. The coefficient of determination (R²) was 0.993, indicating a robust correlation between the minimum ambient temperature and body surface temperature. In contrast, the solid line (____) represents the linear relationship between the highest ambient temperature and body surface temperature, with the regression equation y=0.1566x + 25.316. The coefficient of determination (R²) was 0.096, indicating a weak correlation between the maximum ambient temperature and body surface temperature. The subsequent variable examined was the association between peak ambient temperature and body surface temperature variation in the Komodo dragon (Table 2, Fig. 4). The peak environmental temperature did not significantly influence the body surface temperature of the Komodo dragon, as indicated by a coefficient of −0.05 (p=0.54). This result implies that variations in the peak environmental temperature do not substantially affect the BST of Komodo dragons. For each 1°C rise in the peak ambient temperature, the body temperature of the Komodo dragon decreased by 0.05°C. The residual plot was examined to assess the feasibility of the linear regression model employed in this study, particularly regarding the assumptions of linearity and homoscedasticity. The residuals, as depicted in the Ta1 residual plot, are randomly distributed around the zero line without any discernible pattern, with values ranging from −4 to +4 (Fig. 5). This observation indicates that the linear regression model between the minimum environmental temperature and body temperature is valid and satisfies the fundamental assumptions of linear regression. The R² value of 0.993 (Fig. 4) supports this conclusion, indicating a strong linear relationship between the two variables. We analyzed the residual plot of the relationship between peak environmental temperature (Ta2) and body surface temperature of the Komodo dragon to evaluate the feasibility of the linear regression model. The residuals showed no clear pattern, further confirming that the linear regression model was appropriate for describing this relationship. Figure 6 (Ta2 residual plot) shows that the residuals are randomly distributed around the zero line without forming any specific pattern. Most residual values ranged from –4 to +4, with a concentration of data points in the Ta2 range of 35°C–50°C. This random distribution of residuals indicates that the linearity and homoscedasticity assumptions were acceptable. No systematic patterns, such as upward trends, downward trends, curved patterns, or funnel shapes, were observed, confirming that the linear regression assumptions were not violated. Consequently, the linear regression model remained statistically viable. However, despite the absence of a pattern in the residuals, the regression results indicated a weak relationship between the maximum environmental temperature and the body temperature of the Komodo dragon, as reflected in the coefficient of determination value (R²=0.096) (Fig. 4).
Fig. 5. Residual plot depicting the correlation between the minimum environmental temperature (Ta1) and the body surface temperature of the Komodo dragons. This plot illustrates the relationship between the minimum environmental temperature and the body temperature of Komodo dragons within a specific subset or category. The residuals were randomly distributed around the zero line, indicating no systematic pattern. This observation confirms the suitability of the linear regression model for this dataset, with residual values ranging from −4 to +4.
Fig. 6. Residual plot of the relationship between the maximum environmental temperature (Ta2) and the body surface temperature of Komodo dragons. This residual plot depicts the variation between the predicted and actual body temperatures of the Komodo dragon concerning the maximum environmental temperature. The residuals were randomly distributed and showed no specific pattern, which aligned with the weak association between the variables. The range of residual values was primarily between −4 and +4, indicating no evident breaches of the linearity assumption. DiscussionsIRT captures the surface body temperature of Komodo dragons along with their thermal physiological responsesEnvironmental conditions intricately influence animal behavior. Thermoregulation plays a crucial role in maintaining physiological stability within this influence. Typically, animal activity assessment involves behavioral observations that reflect thermal comfort and welfare. Thermoregulation enables animals to maintain internal body temperature within optimal limits, a process that is especially dependent on ambient temperature in ectothermic species (Rozen-Rechels et al., 2021). As ectotherms, Komodo dragons rely heavily on external heat sources and exhibit thermoregulatory behaviors, such as piloerection and shade-seeking, to sustain thermal equilibrium. These behaviors can be monitored using IRT (Mota-Rojas et al., 2022). Surface temperatures detected by IRT in Komodo dragons are largely attributed to peripheral vasodilation in the thoracic musculature, which facilitates heat dissipation (Vainionpää, 2014). This vasodilation is mediated via autonomic pathways involving the sympathoadrenergic system, particularly neurons located in the rostral medullary raphe and intermediolateral nucleus of the spinal cord. IRT captures these thermophysiological changes and generates radiometric images that reflect heat loss (Mota-Rojas et al., 2022). The central nervous system, particularly the brain, is highly responsive to thermal fluctuations in reptiles (Hammel et al., 1967), with the hypothalamus serving as the core regulatory center for thermal homeostasis (Dimicco and Zaretsky, 2007). Cutaneous sensory input is integrated and processed within the brain and compared with a thermoregulatory set point located in the preoptic area of the hypothalamus (Egan et al., 2005; Boulant, 2006; Lutterschmidt et al., 2011). Additionally, the pineal gland modulates thermoregulatory functions by translating photic signals into physiological and behavioral outputs via melatonin secretion (Axelrod, 1974; Rivkees et al., 1989). Melatonin fluctuations have been shown to affect body temperature in certain reptilian species (Lutterschmidt et al., 1997; Tosini, 1997), but this relationship is not universally observed (Lutterschmidt et al., 2002). Besides behavioral adjustments and cardiovascular adaptations (Dimicco and Zaretsky, 2007), reptiles’ thermal regulation also involves sympathetic-mediated cellular mechanisms. To stabilize internal temperature, these animals respond to environmental temperature shifts by modulating muscle function and reconfiguring metabolic pathways, including both anaerobic and oxidative processes (Glanville and Seebacher, 2006). Heat input across various body regions activates thermoregulatory responses, and thermal sensitivity can differ among tissues (Satinoff, 1978; Glanville and Seebacher, 2006). Correlation between the surface body temperature of the Komodo dragon and ambient environmental temperatureKomodo dragons are most active from midday to late afternoon; however, they often retreat to shaded areas when ambient temperatures rise excessively (Ballance, 2003; Fauzia et al., 2024). Minimum ambient temperatures significantly affect the surface body temperature of Komodo dragons. This observation is consistent with the ecophysiological principles of reptilian thermoregulation, in which low temperatures during the night and early morning are particularly important in determining the body temperature of poikilothermic reptiles (Domínguez-Godoy et al., 2024). Komodo dragons actively regulate their body temperature in response to environmental conditions, particularly by adjusting to cooler periods during the night and early daylight hours, which are crucial for establishing their initial thermal state for daily activity (Auffenberg, 1981). In open habitats, minimum temperatures have a more significant effect on reptilian body temperature than maximum temperatures, as reptiles can increase their body temperature through morning basking (Christian and Bedford, 1995). Field observations have demonstrated that the surface body temperature of these animals varies based on microclimatic conditions, especially in naturally shaded areas. The minimum ambient temperature (Ta1) indicates the temperature within these shaded zones, whereas the maximum ambient temperature (Ta2) represents the environmental temperature recorded in the surrounding areas. Both body and ambient temperatures fluctuated throughout the measurement period, highlighting the dynamic thermal environment experienced by Komodo dragons. In cooler conditions, reptiles are particularly reliant on absorbing environmental heat to initiate and maintain their thermoregulation (Giacometti et al., 2021; Díaz et al., 2022). Contrary to expectations, the maximum ambient temperature appeared to have a minimal direct impact on the surface body temperature of the Komodo dragon. This finding implies that to cope with peak environmental heat, the species relies more on behavioral thermoregulation than on physiological temperature changes (Auffenberg, 1981; Harlow et al., 2010a). Komodo dragons typically mitigate thermal stress by seeking shelter under vegetation or within burrows when faced with high temperatures (Auffenberg, 1981; Fauzia et al., 2024). The observed range of ambient temperatures demonstrated significant variability. Even shaded environments can undergo rapid thermal shifts because of factors like solar radiation and canopy cover (Sannolo, 2019). Vegetation cover, which serves as a crucial thermal refuge, is affected by environmental factors, such as proximity to the coastline and local temperature patterns (Auffenberg, 1981). Komodo dragons exhibit surface body temperatures ranging from 27.4 °C to 35.4 °C, which aligns with the thermal range of 34°C–35.6°C reported by Harlow et al. (2010b). This thermoregulatory range is comparable to those observed in other varanid species, such as the water monitor lizard (Varanus salvator), which ranges from 35.0°C to 36.2°C (Traeholt, 1995), and the lace monitor (Varanus varius), which ranges from 34°C to 36°C (Seebacher and Grigg, 2001). Similar thermoregulatory behaviors have been documented in reptilian species inhabiting desert or tropical climates, including Przewalski’s toadhead agama (Phrynocephalus przewalskii) (Zhang et al., 2023), the large-scale spiny lizard (Sceloporus megalepidurus) (Domínguez-Godoy et al., 2024), and various Asian grass lizards (Takydromus) species (Mi et al., 2022). Komodo dragons, like other varanid, are adept at maintaining stable internal temperatures despite environmental variations, likely through a combination of behavioral and physiological thermoregulatory strategies (Seebacher and Murray, 2007). Komodo dragons regulate their body temperature in a range of habitats, including savannas, forests, and mangrove ecosystems, with forests offering the most optimal thermal conditions (Harlow et al., 2010a). Reptiles utilize both behavioral and physiological adaptations to cope with temperature fluctuations. Their behavioral strategies include basking in the morning sun (Fauzia et al., 2024), seeking shade during peak heat periods (Harlow et al., 2010a; Fauzia et al., 2024), engaging in gular pumping (Fauzia et al., 2024), and employing evaporative cooling through mouth gaping (Harlow et al., 2010b). At night, they remain inactive to conserve heat, resulting in a body temperature drop to 25°C–30°C (Harlow et al., 2010a,b), thereby reducing the need for prolonged morning basking (Pianka and Vitt, 2003). Additionally, postprandial basking is a common behavior that aids digestion (Darling, 1997), and thermoregulation is further achieved by moving between sunlit and shaded areas (Christian and Weavers, 2016; Díaz et al., 2022). On a physiological level, the Komodo dragon demonstrates several adaptations crucial for regulating its body temperature. These adaptations include fat storage in tail adipose tissue (Tomańska et al., 2024), thermoreception, a set-point body temperature of approximately 34°C–35.6°C (Harlow et al., 2010a), and cardiovascular modulation (Seebacher and Franklin, 2005). Heart rate variability is instrumental in thermoregulation, with increased heart rate enhancing peripheral blood flow and facilitating heat dissipation during warmer periods, whereas decreased heart rate at night helps conserve heat (Seebacher and Grigg, 2001). Furthermore, body size plays a significant role in thermoregulatory efficiency, as larger individuals exhibit slower thermal conductivity, leading to more gradual temperature changes than smaller dragons (Harlow et al., 2010a). Collectively, these physiological and behavioral adaptations enable Komodo dragons to maintain their internal temperature within optimal limits, thereby ensuring proper metabolic function, digestion, and overall fitness in their natural habitats. Despite their adaptive thermoregulatory capacity, Komodo dragons remain highly vulnerable to numerous threats, including habitat degradation, overexploitation, invasive species, pollution, disease, and climate change (WWF, 2018). Future climate projections show that rising temperatures and sea levels will have a significant impact on the population and distribution of these species (Jones et al., 2020). Climate change poses severe ecological consequences, endangering biodiversity, disrupting ecosystems, and threatening species persistence (IPCC, 2014). The IUCN has estimated that Komodo dragon populations may decline by more than 30% between 2010 and 2050, primarily due to climate-related pressures such as sea-level rise (Jirik, 2019). According to Jones et al. (2020), climate models forecast that by 2050, Komodo dragons could face an 87% reduction in available habitat, a 97% decline in habitat patch occupancy, and a 99% drop in total population size. Low-lying coastal habitats are especially at risk, with up to 71% of them potentially being submerged in the coming decades (Ashworth, 2021). Furthermore, projected increases in nighttime minimum temperatures may elevate baseline body temperatures, disrupting metabolic rates, activity patterns, and predator-prey interactions. Given that both minimum and maximum ambient temperatures affect the physiology and behavior of Komodo dragons, future research should incorporate additional environmental and physiological parameters, such as humidity, solar radiation, wind speed, activity budgets, behavioral thermoregulation indices, thermal thresholds, and thermal inertia, using comprehensive, multi-instrumental approaches. ConclusionThe body temperature of Komodo dragons exhibits temporal variation along with ambient environmental temperatures. Among these factors, the minimum ambient temperature plays a pivotal role in determining the species’ surface body temperature, whereas the maximum temperature exerts only an indirect influence. Lower environmental temperatures appear to initiate physiological thermoregulation through cardiovascular and neuroendocrine mechanisms, thereby affecting muscle function and modulating both anaerobic and oxidative metabolic pathways. In response to rising temperatures, Komodo dragons primarily rely on behavioral thermoregulation, such as shade-seeking and activity adjustment, to maintain thermal balance. This dynamic interaction between environmental temperature and the thermoregulatory strategies of the Komodo dragon enables the species to remain within a safe physiological range, ensuring survival through basking, sheltering, and managing activity periods. Despite their adaptability to fluctuating thermal environments, Komodo dragons face increasing threats from climate change, which may significantly compromise their long-term survival. Therefore, future research should integrate a broader set of variables, including additional environmental, physiological, and behavioral metrics, to inform effective ecological understanding and conservation planning. AcknowledgmentThe authors wish to express their gratitude to the Indonesian Education Scholarship (BPI) through the Center for Higher Education Funding and Assessment (PPAT), Ministry of Higher Education, Science, and Technology of the Republic of Indonesia, in collaboration with the Indonesia Endowment Fund for Education (LPDP) and the Ministry of Finance of the Republic of Indonesia, for their generous financial support. We also extend our sincere appreciation to the Directorate of Species and Genetic Biodiversity Conservation, Directorate General of Natural Resources and Ecosystem Conservation, and Ministry of Environment and Forestry of the Republic of Indonesia for granting the necessary research permits. Furthermore, we are grateful to the Komodo National Park Authority for providing access to the research site and for their invaluable support and assistance during the fieldwork. Conflict of interestThe authors declare no conflicts of interest. FundingThis research was funded by the Indonesian Education Scholarship (BPI) through the Center for Higher Education Funding and Assessment (PPAT), Ministry of Higher Education, Science, and Technology of the Republic of Indonesia, Indonesia Endowment Fund for Education (LPDP), Ministry of Finance of the Republic of Indonesia, under contract number 00540/J5.2.3./BPI.06/9/2022, and the Directorate of Research, Technology, and Community Service, which operates under the Directorate General of Higher Education, Research, and Technology of the Ministry of Education, Culture, Research, and Technology of the Republic of Indonesia, for their financial support of this research, as outlined in contract number 027/E5/PG.02.00.PL/2024. Authors’ contributionsMM contributed to the conceptualization, methodology, software development, validation, formal analysis, investigation, preparation of the original draft, review and editing, and visualization. AS was involved in the methodology, software development, validation, formal analysis, preparation of the original draft, review and editing, visualization, and supervision. KS contributed to the formal analysis, resources, data curation, original draft preparation, and visualization. NS was responsible for the methodology, resources, data curation, preparation of the original draft, visualization, and supervision. SA was involved in project administration and funding acquisition, as well as conceptualization, methodology, resources, data curation, preparation of the original draft, review and editing, visualization, and supervision. Data availabilityThis manuscript contains all raw data related to the measurements of body surface temperature and ambient temperature. The Supplementary Materials include the processed dataset used for multiple linear regression analysis. ReferencesAfonso, A.L., Lopes, G. and Ribeiro, A.F. 2024. Lizard body temperature acquisition and lizard recognition using artificial intelligence. Sensors. 2024. 24(13), 4135; doi:10.3390/S24134135 Antara News. 2025. Taman Nasional Komodo catat 334.206 orang lakukan kunjungan wisata pada 2024. ANTARA News Megapolitan. Available via https://megapolitan.antaranews.com/berita/334614/taman-nasional-komodo-catat-334206-orang-lakukan-kunjungan-wisata-pada-2024 Ariefiandy, A., Forsyth, D.M., Purwandana, D., Imansyah, J., Ciofi, C., Rudiharto, H., Seno, A.A. and Jessop, T.T. 2016. Temporal and spatial dynamics of insular Rusa deer and wild pig populations in Komodo National Park. J. Mammal. 97(6), 1652–1662; doi:10.1093/JMAMMAL/GYW131 Ariefiandy, A., Purwandana, D., Coulson, G., Forsyth, D.M. and Jessop, T.S. 2013a. Monitoring the ungulate prey of the Komodo Dragon Varanus komodoensis: distance sampling or faecal counts?. Wildlife Biol. 19(2), 126–137; doi: 10.2981/11-098 Ariefiandy, A., Purwandana, D., Seno, A., Ciofi, C. and Jessop, T.S. 2013b. Can camera traps monitor Komodo Dragons a large ectothermic predator?. PLos One. 8(3), 58800; doi: 10.1371/JOURNAL.PONE.0058800 Ashworth, J. 2021. Komodo dragon is now listed as endangered as rising sea levels threaten its survival. Natural History Museum. Available via https://www.nhm.ac.uk/discover/news/2021/september/komodo-dragon-is-now-listed-as-endangered-from-rising-sea-levels.html? Auffenberg, W. 1981. The behavioral ecology of the Komodo monitor. University Presses of Florida, Gainesville, FL. Autumn, K., Jindrich, D., Denardo, D. and Mueller, R. 1999. Locomotor performance at low temperature and the evolution of nocturnality in geckos. Evolution 53(2), 580–599; doi:10.1111/j.1558-5646.1999.tb03793.x Axelrod, J. 1974. The pineal gland: a neurochemical transducer. Science 184(4144), 1341–1348; doi:10.1126/science.184.4144.1341 Ballance A. 2003. South Sea Islands: a natural history. Firefly Books. Barroso, F.M., Riaño, G., Sannolo, M., Carretero, M.A. and Rato, C. 2020. Evidence from Tarentola mauritanica (Gekkota: phyllodactylidae) helps validate thermography as a tool to infer internal body temperatures of lizards. J. Therm. Biol. 93, 102700; doi:10.1016/J.jtherbio.2020.102700 Böhme. 2003. Checklist of the living monitor lizards of the world (family Varanidae). Zoologische. Verhandelingen. 341, 4–43. Boulant, J.A. 2006. Neuronal basis of Hammel’s model for set-point thermoregulation. J. Appl. Physiol. 100(4), 1347–1354; doi:10.1152/japplphysiol.01064.2005 Boyd, B.S., Colon, F., Doty, J.F. and Sanders, K.C. 2021. Beware of the Dragon: a case report of a Komodo Dragon attack. Foot. Ankle. Orthop. 6(2), 1–6; doi:10.1177/24730114211015623 Bracke, M.B.M., Metz, J.H.M., Dijkhuizen, A.A. and Spruijt, B.M. 2001. Development of a decision support system for assessing farm animal welfare in relation to husbandry systems: strategy and prototype. J. Agric. Environ. Ethics 14(3), 321–337; doi:10.1023/A:1012282207323 Bull, J.J., Jessop, T.S. and Whiteley, M. 2010. Deathly Drool: evolutionary and ecological basis of septic bacteria in Komodo Dragon mouths. PLos One. 5(6), e11097; doi:10.1371/journal.pone.0011097 Christian, K.A. and Bedford, G.S. 1995. Seasonal changes in thermoregulation by the frillneck lizard, Chlamydosaurus kingii, in tropical Australia. Ecology 76(1), 124–132; doi:10.2307/1940636 Christian, K.A. and Weavers, B.W. 2016. Thermoregulation of monitor lizards in Australia: an evaluation of methods in thermal biology. Ecol. Monogr. 66(2), 139–157. Church, J.S., Cook, N.J. and Schaefer, A.L. 2009. Recent applications of infrared thermography for animal welfare and veterinary research: everything from chicks to Elephants. InfraMation 2009 Proceedings, InfraMation: North Billerica 10, pp 215–224. Çırak, C. and Esendal, E. 2006. Drought stress of soybean. J. Agric. Fac. Ondokuz. Mayıs. Univ. 21, 231–237. Darling, K. 1997. Komodo Dragon. Lothrop, Lee and Shepard Books. Lothrop, Lee and Shepard Books. Díaz, J.A., Izquierdo-Santiago, R. and Llanos-Garrido, A. 2022. Lizard thermoregulation revisited after two decades of global warming. Funct. Ecol. 36(12), 3022–3035; doi:10.1111/1365-2435.14192 Dimicco, J.A. and Zaretsky, D.V. 2007. The dorsomedial hypothalamus: a new player in thermoregulation. Am. J. Physiol. Regul. Integr. Comp. Physiol. 292(1), 47–63; doi:10.1152/ajpregu.00498.2006 Directorate General of Natural Resources and Ecosystem Conservation. 2024. Smart patrol application: a technological innovation for advancing natural resource conservation initiatives. Indonesian: Direktorat Jenderal Konservasi Sumber Daya Alam dan Ekosistem. Domínguez-Godoy, M.A., Arenas-Ríos, E., Uriostegui-Escoto, D., Lucio, R.A., Díaz De La Vega-pérez, A.H. and Méndez De La Cruz, F.R. 2024. Behavioural thermoregulation prevents thermal stress in lizard sperm fertility. Eur. J. Wildl. Res. 70(6), 1–14; doi:10.1007/s10344-024-01864-6 Egan, G.F., Johnson, J., Farrell, M., Mcallen, R., Zamarripa, F., Mckinley, M.J., Lancaster, J., Denton, D. and Fox, P.T. 2005. Cortical, thalamic, and hypothalamic responses to cooling and warming the skin in awake humans: a positron-emission tomography study. Proc. Nat. Acad. Sci. United States Amer. 102(14), 5262–5267; doi:10.1073/pnas.0409753102 Farrar, K.L., Field, A.E., Norris, S.L. and Jacobsen. 2020. Comparison of Rectal and Infrared Thermometry Temperatures in Anesthetized Swine (Sus scrofa). J. Am. Assoc. Lab. Anim. Sci. 59(2), 221–225; doi:10.30802/AALAS-JAALAS-19-000119 Fauzia, A.M., Kusrini, M.D., Mulyani, Y.A., Sari, F.E., Nusantara, M.G.G., Setiawan, R.A. and Ariefiandy, A. 2024. Community Perspectives in ethogram for komodo dragon (Varanus komodoensis) in the Wild. Media Konserv. 29(2), 91–100. Giacometti, D., Yagi, K.T., Abney, C.R., Jung, M.P. and Tattersall, G.J. 2021. Staying warm is not always the norm: behavioural differences in thermoregulation of two snake species. Can. J. Zool. 99(11), 974–983; doi:10.1139/cjz-2021-0135 Glanville, E.J. and Seebacher, F. 2006. Compensation for environmental change by complementary shifts of thermal sensitivity and thermoregulatory behaviour in an ectotherm. J. Exp. Biol. 209(24), 4869–4877; doi:10.1242/jeb.02585 Government of the Republic of Indonesia. 1990. Law No. 5 of 1990 on the Conservation of Biological Natural Resources and Their Ecosystems. Indonesian: Pemerintah Republik Indonesia. 1990. Undang-Undang Nomor 5 Tahun 1990 tentang Konservasi Sumber Daya Alam Hayati dan Ekosistemnya. Available via http://www.dpr.go.id/dokjdih/document/uu/602.pdf Government of the Republic of Indonesia. 2009. Law of the Republic of Indonesia Number 18 of 2009 regarding Animal Husbandry and Animal Health. Jakarta (ID): State Secretariat. In Indonesian: Pemerintah Republik Indonesia. 2009. Undang-Undang Republik Indonesia Nomor 18 Tahun 2009 tentang Peternakan dan Kesehatan Hewan. Jakarta, Indonesia: Sekretariat Negara. Government of the Republic of Indonesia. 2024. Government Regulation Number 36 of 2024 delineates the classifications and rates of non-tax state revenue pertinent to the Ministry of Environment and Forestry. Indonesian: Pemerintah Republik Indonesia. 2024. Peraturan Pemerintah Nomor 36 Tahun 2024. 236043. Hammel, H.T., Caldwell, F.T. and Abrams, R.M. 1967. Regulation of body temperature in the Blue-Tongued Lizard. Science 156(3779), 1260–1262; doi:10.1126/science.156.3779.1260 Harlow, H.J., Purwandana, D., Jessop, T.S. and Phillips, J.A. 2010a. Body temperature and thermoregulation of Komodo dragons in the field. J. Therm. Biol. 35(7), 338–347; doi:10.1016/j.jtherbio.2010.07.002 Harlow, H.J., Purwandana, D., Jessop, T.S. and Phillips, J.A. 2010b. Size-related differences in the thermoregulatory habits of free-ranging Komodo Dragons. 2010, 1–9; doi: 10.1155/2010/921371 Hilmer, S.S. 2010. Ecophysiology of feral cats (Felis catus) in Australia. Fachbereich Biowissenschaften, PhD. In Den Bosch, H.A.J. 1983. Snout temperatures of reptiles, with special reference to the changes during feeding behaviour in Python molurus bivittatus (Serpentes, Boidae): a study using infrared radiation. Amphibia-Reptilia 4(1), 49–61; doi:10.1163/156853883X00265 IPCC. 2014. Part A: Global and sectoral aspects. (Contribution of working group ii to the fifth assessment report of the intergovernmental panel on climate change). Climate Change 2014: Impacts, Adaptation, and Vulnerability. p. 1132. Available via https://www.ipcc.ch/pdf/assessment-report/ar5/wg2/WGIIAR5-FrontMatterA_FINAL.pdf Jessop, A., Purwandana, D., Ciofi, C., Jeri Imansyah, M., Panggur, M.R. and Ariefiandy, A. 2018. Effects of human activities on Komodo dragons in Komodo National Park. Biodivers. Conserv. 27(13), 3329–3347; doi: 10.1007/s10531-018-1601-3 Jessop, T.S., Ariefiandy, A., Azmi, M., Ciofi, C., Imansyah, J. and Purwandana, D. 2021. Varanus komodoensis, Komodo Dragon. The IUCN Red List of Threatened Species, Paris, 8235. T22884A123633058. Jessop, T.S., Ariefiandy, A., Forsyth, D.M., Purwandana, D., White, C.R., Benu, Y.J., Madsen, T., Harlow, H.J. and Letnic, M. 2020. Komodo dragons are not ecological analogs of apex mammalian predators. Ecology 101(4), 0–2; doi:10.1002/ecy.2970 Jirik, K. 2019. LibGuides: Komodo Dragon (Varanus komodoensis) fact sheet: population & conservation status. IUCN Red List of Threatened Species, Paris. doi: 10.2305/IUCN.UK.2021-2.RLTS.T22884A123633058.EN Jones, A.R., Jessop, T.S., Ariefiandy, A., Brook, B.W., Brown, S.C., Ciofi, C., Benu, Y.J., Purwandana, D., Sitorus, T. and Wigley, T.M.L. 2020. Identifying island safe havens to prevent the extinction of the World’s largest lizard from global warming. Ecol. Evol. 10(19), 10492–10507; doi:10.1002/ece3.6705 Laver, R.J., Purwandana, D., Ariefiandy, A., Imansyah, J., Forsyth, D., Ciofi, C. and Jessop, T.S. 2012. Life-History and spatial determinants of somatic growth dynamics in komodo dragon populations. PLos One. 7(9), 1–10; doi:10.1371/journal.pone.0045398 Lowe, G., Sutherland, M., Waas, J., Schaefer, A., Cox, N. and Stewart, M. 2019. Infrared thermography—a non-invasive method of measuring respiration rate in calves. Animals. 2019. 9(8), 535; doi:10.3390/ANI9080535 Lutterschmidt, D.I., Lutterschmidt, W.I. and Hutchison, V.H. 1997. Melatonin and chlorpromazine: thermal selection and metabolic rate in the bullsnake, Pituophis melanoleucus. Comparative. Biochem. Physiol. Part. C. Pharmacol. Toxicol. Endocrinol. 118(3), 271–277; doi:10.1016/S0742-8413(97)00106-0 Lutterschmidt, D.I., Lutterschmidt, W.I. and Hutchison, V.H. 2011. Melatonin and thermoregulation in ectothermic vertebrates: a review. Can. J. Zool. 81(1), 1–13; doi:10.1139/Z02-189 Lutterschmidt, D.I., Lutterschmidt, W.I., Ford, N.B. and Hutchison, V.H. 2002. Behavioral thermoregulation and the role of melatonin in a nocturnal snake. Hormones. Behav. 41(1), 41–50; doi:10.1006/hbeh.2001.1721 Mccafferty, D.J. 2007. The value of infrared thermography for research on mammals: previous applications and future directions. Mamm. Rev. 37(3), 207–223; doi:10.1111/J.1365-2907.2007.00111.X Mi, C., Ma, L., Wang, Y., Wu, D., Du, W. and Sun, B. 2022. Temperate and tropical lizards are vulnerable to climate warming due to increased water loss and heat stress. Proc. Biol. Sci. 289(1980), 20221074; doi: 10.1098/rspb.2022.1074 Mota-Rojas, D., Napolitano, F., Braghieri, A., Guerrero-Legarreta, I., Bertoni, A., Martínez-Burnes, J., Cruz-Monterrosa, R., Gómez, J., Ramírez-Bribiesca, E., Barrios-García, H., José, N., Álvarez, A., Mora-Medina, P. and Orihuela, A. 2021. Thermal biology in river buffalo in the humid tropics: neurophysiological and behavioral responses assessed by infrared thermography. J. Anim. Behav. Biometeorol. 9(1), 2103; doi:10.31893/jabb.21003 Mota-Rojas, D., Pereira, A.M.F., Martínez-Burnes, J., Domínguez-Oliva, A., Mora-Medina, P., Casas-Alvarado, A., Rios-Sandoval, J., De Mira Geraldo, A. and Wang, D. 2022. Thermal imaging to assess the health status in wildlife animals under human care: limitations and perspectives. Animals 12(24), 3558; doi:10.3390/ani12243558 Narayan, E., Perakis, A. and Meikle, W. 2019. Using thermal imaging to monitor body temperature of Koalas (Phascolarctos cinereus) in A Zoo Setting. Animals 9(12), 1094; doi:10.3390/ani9121094 Permatasari, N.V., Kawigraha, A., Abidin, F. and Puspita, A.N.G. 2020. Komodo conservation and development permission jurassic park super premium tourism. In C. M. Hapsari and A. Hastomo (Eds.), Proceedings of the National Seminar: Green Development and Licensing: Diplomacy, Institutional Readiness, and Standardization Patterns. Postgraduate School, Diponegoro University (UNDIP), Semarang, Indonesia. pp:149–56. Pianka, E.R. and Vitt, L.J. 2003. Lizards: windows to the evolution of diversity (Vol. 5). University of California Press, Berkeley, CA. Pörtner, H.O. 2010. Oxygen- and capacity-limitation of thermal tolerance: a matrix for integrating climate-related stressor effects in marine ecosystems. J. Exp. Biol. 213(6), 881–893. Pörtner, H.O., Bock, C. and Mark, F.C. 2017. Oxygen- and capacity-limited thermal tolerance: bridging ecology and physiology. J. Exp. Biol. 220(15), 2685–2696; doi:10.1242/jeb.134585 Purwandana, D., Ariefiandy, A., Azmi, M., Nasu, S.A., Sahudin., Dos, A.A. and Jessop, T.S. 2021. Turning ghosts into dragons: improving camera monitoring outcomes for a cryptic low-density Komodo dragon population in eastern Indonesia. Wildl. Res. 49(4), 295–302; doi:10.1071/WR21057 Rivkees, S.A., Carlson, L.L. and Reppert, S.M. 1989. Guanine nucleotide-binding protein regulation of melatonin receptors in lizard brain. Proc. Nat. Acad. Sci. 86(10), 3882–3886; doi:10.1073/pnas.86.10.3882 Rozen-Rechels, D., Rutschmann, A., Dupoué, A., Blaimont, P., Chauveau, V., Miles, D.B., Guillon, M., Richard, M., Badiane, A., Meylan, S., Clobert, J. and Le Galliard, J.F. 2021. Interaction of hydric and thermal conditions drive geographic variation in thermoregulation in a widespread lizard. Ecol. Monogr. 91(2), e01440; doi:10.1002/ecm.1440 Sannolo, M. 2019. Reptiles under the sun: using lacertid lizards to study thermal and water ecology in ectotherms. Faculdade de Ciências, PhD, pp:1–233. Santoso, K., Prawida, A.E., Ridwan, Y., Suprayogi, A., Ulum, M.F. and Seminar, K.B. 2023. Mapping surface temperature pattern of Giraffa camelopardalis in Ragunan Zoo Using infrared thermal camera. In the AIP Conference Proceedings. The 4th International Conference on Life Sciences and Technology (ICoLiST) will be held in Malang, East Java Indonesia on August 31, 2021. 2634(1). doi: 10.1063/5.0111647 Satinoff, E. 1978. Neural organization and evolution of thermal regulation in mammals. Science 201(4350), 16–22; doi:10.1126/science.351802 Seebacher, F. and Franklin, C.E. 2005. Physiological mechanisms of thermoregulation in reptiles: a review. J. Comp. Physiol. B. Biochem. Syst. Environ. Physiol. 175(8), 533–541; doi:10.1007/s00360-005-0007-1 Seebacher, F. and Grigg, G.C. 2001. Changes in heart rate are important for thermoregulation in the varanid lizard Varanus varius. J. Comp. Physiol. B. Biochem. Syst. Environ. Physiol. 171(5), 395–400; doi: 10.1007/s003600100188 Seebacher, F. and Murray, S.A. 2007. Transient receptor potential ion channels control thermoregulatory behaviour in reptiles. PLos One. 2(3), e281; doi:10.1371/journal.pone.0000281 Shelley, F.M. and Metz, R. 2023. Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES). Geogr. Traffick. 287(292), 287–292; doi:10.5040/9798400656385.0078 Shine, R. and Somaweera, R. 2019. Last lizard standing: the enigmatic persistence of the Komodo dragon. Glob. Ecol. Conserv. 18, 624; doi:10.1016/j.gecco.2019.e00624 Švejdová, K., Šoch, M., Šimková, A., Zábranský, L., Novák, P., Brouček, J., Čermák, B., Pálka, V. and Šimák-Líbalová, K. 2013. Measuring the body surface temperature of animals using a thermographic camera. Acta Univ. Cibiniensis - Ser. E Food Technol. 17(2), 99–106; doi: 10.2478/aucft-2013-0017 Tomańska, A., Stawinoga, M., Szturo, K., Styczyńska, M., Klećkowska-Nawrot, J., Janeczek, M., Goździewska-Harłajczuk, K., Melnyk, O. and Gębarowski, T. 2024. Biological Significance of the Komodo Dragon’s Tail (Varanus komodoensis, Varanidae). Animals 14(15), 2142; doi:10.3390/ani14152142/S1 Tosini, G. 1997. The pineal complex of reptiles: physiological and behavioral roles. Ethol. Ecol. Evol. 9(4), 313–333; doi:10.1080/08927014.1997.9522875 Traeholt, C. 1995. A radio-telemetric study of the thermoregulation of free living water monitor lizards, Varanus S. salvator. J. Comp. Physiol. 165(2), 125–131. Vainionpää, M. 2014. Thermographic imaging in cats and dogs : Usability as a clinical method. Available via http://hdl.handle.net/10138/45311 Walpole, M.J. and Goodwin, H.J. 2001. Local attitudes towards conservation and tourism around Komodo National Park, Indonesia. Environ. Conserv. 28(2), 160–166; doi:10.1017/s0376892901000169 Wang, F.K., Shih, J.Y., Juan, P.H., Su, Y.C. and Wang, Y.C. 2021. Non-Invasive cattle body temperature measurement using infrared thermography and auxiliary sensors. Sensors 21(7), 2425; doi:10.3390/s21072425 Williams, K.F. 2023. Thermal imaging for wildlife applications. Pelagic Publishing, Europe. doi: 10.53061/iptu9371 WWF. 2018. Living planet report - 2018: aiming higher. Grooten, M. and Almond, R.E.A.(Eds). WWF, Gland, Switzerland. p.72 Zhang, Q., Han, X.Z., Burraco, P., Wang, X.F., Teng, L.W., Liu, Z.S. and Du, W.G. 2023. Oxidative stress mediates the impact of heatwaves on survival, growth and immune status in a lizard. In Proc. Royal Soc. B: Biol. Sci. 290, 1768; doi:10.1098/rspb.2023.1768 | ||
| How to Cite this Article |
| Pubmed Style Mahfud M, Suprayogi A, Santoso K, Santoso N, Agungpriyono S. Infrared thermography assessment of body surface temperature in Komodo dragons (Varanus komodoensis) during the Komodo island dry season. Open Vet. J.. 2025; 15(10): 4912-4927. doi:10.5455/OVJ.2025.v15.i10.11 Web Style Mahfud M, Suprayogi A, Santoso K, Santoso N, Agungpriyono S. Infrared thermography assessment of body surface temperature in Komodo dragons (Varanus komodoensis) during the Komodo island dry season. https://www.openveterinaryjournal.com/?mno=265184 [Access: January 24, 2026]. doi:10.5455/OVJ.2025.v15.i10.11 AMA (American Medical Association) Style Mahfud M, Suprayogi A, Santoso K, Santoso N, Agungpriyono S. Infrared thermography assessment of body surface temperature in Komodo dragons (Varanus komodoensis) during the Komodo island dry season. Open Vet. J.. 2025; 15(10): 4912-4927. doi:10.5455/OVJ.2025.v15.i10.11 Vancouver/ICMJE Style Mahfud M, Suprayogi A, Santoso K, Santoso N, Agungpriyono S. Infrared thermography assessment of body surface temperature in Komodo dragons (Varanus komodoensis) during the Komodo island dry season. Open Vet. J.. (2025), [cited January 24, 2026]; 15(10): 4912-4927. doi:10.5455/OVJ.2025.v15.i10.11 Harvard Style Mahfud, M., Suprayogi, . A., Santoso, . K., Santoso, . N. & Agungpriyono, . S. (2025) Infrared thermography assessment of body surface temperature in Komodo dragons (Varanus komodoensis) during the Komodo island dry season. Open Vet. J., 15 (10), 4912-4927. doi:10.5455/OVJ.2025.v15.i10.11 Turabian Style Mahfud, Mahfud, Agik Suprayogi, Koekoeh Santoso, Nyoto Santoso, and Srihadi Agungpriyono. 2025. Infrared thermography assessment of body surface temperature in Komodo dragons (Varanus komodoensis) during the Komodo island dry season. Open Veterinary Journal, 15 (10), 4912-4927. doi:10.5455/OVJ.2025.v15.i10.11 Chicago Style Mahfud, Mahfud, Agik Suprayogi, Koekoeh Santoso, Nyoto Santoso, and Srihadi Agungpriyono. "Infrared thermography assessment of body surface temperature in Komodo dragons (Varanus komodoensis) during the Komodo island dry season." Open Veterinary Journal 15 (2025), 4912-4927. doi:10.5455/OVJ.2025.v15.i10.11 MLA (The Modern Language Association) Style Mahfud, Mahfud, Agik Suprayogi, Koekoeh Santoso, Nyoto Santoso, and Srihadi Agungpriyono. "Infrared thermography assessment of body surface temperature in Komodo dragons (Varanus komodoensis) during the Komodo island dry season." Open Veterinary Journal 15.10 (2025), 4912-4927. Print. doi:10.5455/OVJ.2025.v15.i10.11 APA (American Psychological Association) Style Mahfud, M., Suprayogi, . A., Santoso, . K., Santoso, . N. & Agungpriyono, . S. (2025) Infrared thermography assessment of body surface temperature in Komodo dragons (Varanus komodoensis) during the Komodo island dry season. Open Veterinary Journal, 15 (10), 4912-4927. doi:10.5455/OVJ.2025.v15.i10.11 |