| Research Article | ||
Open Vet. J.. 2025; 15(10): 4962-4971 Open Veterinary Journal, (2025), Vol. 15(10): 4962-4971 Research Article Hyperthermia-induced sudden death: Morphological insights from the lung and heart in an experimental study of Wistar ratsEmina Dervišević1, Ines Šunjić2, Muamer Dervišević3, Francesco Sessa4, Hajrudin Spahović5, Aida Bešić6*, Ferid Krupić7 and Temima Ajanović81Department of Forensic Medicine, Faculty of Medicine, University of Sarajevo , Sarajevo, Bosnia and Herzegovina 2Department of Pathology, Cytology and Forensic Medicine, University Hospital Mostar, Mostar, Bosnia and Herzegovina 3Clinic for Lung Diseases “Podhrastovi”, University Clinical Center of Sarajevo, Sarajevo, Bosnia and Herzegovina 4Department of Medical, Surgical and Advanced Technologies “G.F. Ingrassia”, University of Catania, Catania, Italy 5Urology Clinic, Clinical Center University of Sarajevo, Sarajevo, Bosnia and Herzegovina 6Department of Clinical Sciences Veterinary Medicine, University of Sarajevo-Veterinary Faculty, Sarajevo, Bosnia and Herzegovina 7Department of Anesthesiology, Institute of Clinical Sciences, Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden 8Department of Analytical Chemistry, Faculty of Science, University of Sarajevo, Sarajevo, Bosnia and Herzegovina *Corresponding Author: Aida Bešić. Department of Clinical Sciences, Veterinary Medicine, University of Sarajevo-Veterinary Faculty, Sarajevo, Bosnia and Herzegovina, Republic of Bosnia and Herzegovina. Email: katicaaida1 [at] gmail.com Submitted: 26/06/2025 Revised: 03/09/2025 Accepted: 11/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
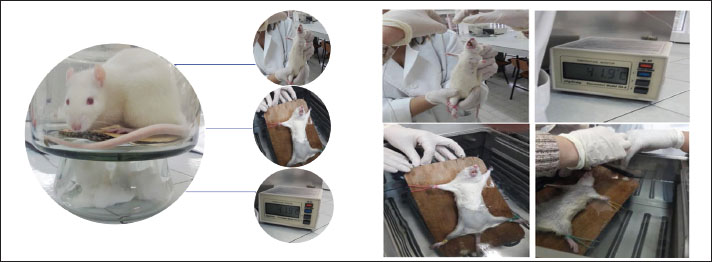
AbstractBackground: Many deaths during heat waves stem not only from body overheating but also from heat stress, which can intensify pre-existing medical conditions, leading to fatal outcomes. Aim: This study aimed to investigate whether the intensity of pathological changes in the heart muscle and lung tissue of albino rats exposed to hyperthermia correlates with different water temperatures (WT) and to determine whether the histological structure of the myocardium and lungs varies. Methods: A sample of 21 albino rats was exposed to water temperatures of 37°C, 41°C, and 44°C. Temperature readings were recorded before immersion, immediately after immersion, at the point of reaching hyperthermia, at 20 minutes, and at the time of death. Tissue samples were collected from the dissected rats, fixed in 10% buffered formalin at room temperature, embedded in paraffin, sectioned into 4–5 μm slices, and stained using the hematoxylin-eosin method. Results: The severity of myocardial histopathological alterations increased with both higher temperatures and longer exposure durations WT. However, the progression of morphological alterations in cardiomyocytes was not markedly significant, likely due to the brief exposure time, which limited the visualization of subcellular alterations in Hematoxylin and Eosin staining-stained tissue. All lung samples from the seven rats exposed to the highest temperatures displayed bronchiolitis and acute bronchitis, along with early bronchopneumonia. Conclusion: While some organs exhibit greater tolerance to heat stroke than other organs, most organs show similar alterations characterized by capillary dilation, vascular pathway disruption, and extravasation. The extent of pathological changes in myocardial and lung tissue intensified with higher temperatures and longer exposure durations to elevated WT. However, the progression of morphological alterations in cardiomyocytes did not demonstrate marked significance, likely because of the brief exposure period. This short duration may limit the detection of subcellular changes when using hematoxylin-eosin staining. Keywords: Sudden death, Forensic, Microscopic, Organs, Post-mortem. IntroductionSudden cardiac death (SCD) is defined as an unexpected death resulting from sudden circulatory arrest, often occurring within 1 hour of symptom onset or within 24 hours of the individual being seen alive and symptom-free (van den Tweel and Wittekind, 2016). SCD represents a significant public health concern, with annual incidence rates ranging from 40 to 100 per 100,000 individuals, depending on surveillance methods, definitions used, and autopsy practices across countries (Fukuda et al., 2015; Zhao et al., 2016; Illing et al., 2020). Although the overall incidence of SCD increases markedly with age, it can also affect younger populations, particularly when it is linked to genetic or structural cardiac abnormalities (Luterbacher et al., 2004; Sessa et al., 2024). Although coronary artery disease and structural heart conditions remain the most frequent causes of sudden unexplained deaths (SUDs), approximately 5%–15% of cases remain unexplained even after a thorough postmortem examination and are referred to as SUDs (Ferron et al., 2006; Sessa et al., 2021). The diagnostic complexity of SCD stems from overlapping histological features—such as myocardial hypertrophy and fibrosis—which are commonly found in both pathological and physiologically compensated conditions (Thommen, 2005). Moreover, interobserver variability in the interpretation of histopathological findings poses an additional challenge to accurate diagnosis (et al., 2023). From a forensic perspective, such diagnostic ambiguity has important epidemiological, preventive, and legal implications. Environmental stressors, such as hyperthermia, have recently emerged as possible triggers for sudden death, especially in indoor settings, such as hot baths and saunas, where thermoregulation is compromised (Sacco et al., 2023). Although research has elucidated many of the systemic effects of heatstroke—ranging from mitochondrial dysfunction to systemic inflammatory responses—the role of hyperthermia in precipitating fatal cardiac events remains poorly understood (Iba et al., 2025 a, 2025b; van Rhoon et al., 2020 a). Heat stress can disrupt cellular homeostasis by impairing mitochondrial electron transport chains, particularly complex I, leading to reduced adenosine triphosphate production and a shift from apoptosis to necrosis (Pereira Gomes et al., 2019). This can result in widespread inflammation and dysfunction of multiple organs, including the heart. Pathophysiologically, hyperthermia may contribute to arrhythmogenesis, myocardial ischemia, and structural cellular damage through mechanisms such as oxidative stress, increased membrane permeability, and activation of the inflammatory cascade (Pereira Gomes et al., 2019; van Rhoon et al., 2020 b; Robayo-Amortegui et al., 2024). The effects of hyperthermia appear to be modulated by the body’s water balance, vascular responses, and ambient environmental factors—all of which can influence cardiac output and thermoregulation (Richmond et al., 2015). Despite these insights, the causal relationship between hyperthermia and SCD remains speculative in forensic settings due to the lack of pathognomonic findings and reliable post-mortem biochemical markers (Marui et al., 2017). The absence of standardized criteria for diagnosing heat-related deaths further complicates the situation, especially in individuals with predisposing cardiac or genetic conditions (Mørch et al., 2017). Bathing-related deaths in hot water environments are increasingly reported, yet little is known about their underlying mechanisms (Lepock, 2003; Heled et al., 2013; Quinn et al., 2014; Natarajan et al., 2015; Walter and Carraretto, 2016; Yang et al., 2017; Lassche et al., 2019). This study aimed to investigate whether the intensity of pathological changes in the heart muscle and lung tissue of albino rats exposed to hyperthermia correlates with different water temperatures and to determine whether the histological structure of the myocardium and lungs varies. Materials and MethodsThe study was designed as a prospective, randomized experimental investigation, with rigorous monitoring, using a rodent model of hyperthermia. This study was conducted at the Medical and Veterinary Faculty of the University of Sarajevo, adhering to ethical guidelines for biomedical research involving animals. Experimental animalsA total of 21 adult Wistar rats of both sexes, weighing between 240 and 310 g, were used. The animals were housed under identical laboratory conditions and underwent a 7-day acclimatization period in a vivarium with a 12-hour light/dark cycle and ambient temperature of 20°C ± 2°C (Natarajan et al., 2015). Throughout the experiment, the rats had ad libitum access to commercial laboratory chow and water. All procedures conformed to the international guidelines (CIOMS and ICLAS) IGPBRIA, (2012) (27. Council for International Organizations and Medical Sciences; World Health Organization. International Ethical Guidelines for Biomedical Research Involving Human Subjects. Geneva, Switzerland: World Health Organization; 2002; Accessed on August 2020, n.d.; 28. Demers G. “Guidelines for Laboratory Animal Care” — ICLAS President Presentation at the Workshop on Development and Science Based Guideines for Laboratory Animal Care, Washington D.C; 2003. , n.d.). Group allocation and the hyperthermia modelThe 21 rats were randomly assigned into three experimental groups: Control-37, Hyperthermia-41, and Heat stroke-44 (n=7 per group) based on water temperature exposure: 37°C (control), 41°C, and 44°C. A water bath was preheated to the target temperatures, which were continuously monitored using a digital display and a submerged temperature probe. Before immersion, each rat was anesthetized with ketamine (10 mg/ml) at a dosage of 1.2 ml/kg body weight, administered intramuscularly. An esophageal probe was inserted just before immersion for continuous core body temperature monitoring. Temperature readings were recorded before immersion, immediately after immersion, at the point of reaching hyperthermia, at 20 minutes, and at the time of death. Experimental protocolTwenty-four hours before the experiment, the body weight of each rat was measured to calculate the anesthetic dose. Anesthesia was administered to the thigh muscle, and the rats were fixed to a wooden board. After anesthesia, the rats were immersed in water at the appropriate temperature. Following immersion, the anterior abdominal wall was opened to collect tissue samples, and the heart and lungs were dissected. Tissues were fixed in 10% buffered formalin, and sections were prepared and stained using standard hematoxylin–eosin (H&E) staining (Fig. 1).
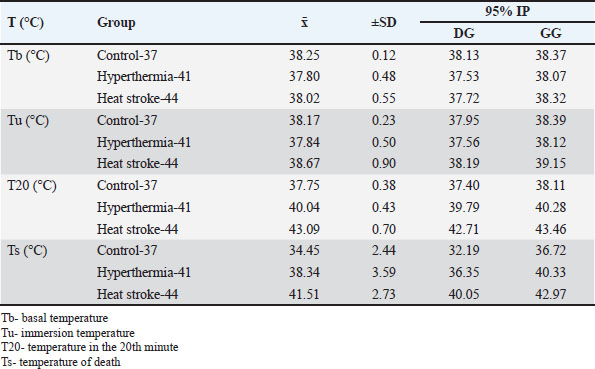
Fig. 1. Hyperthermia experimental protocol. Pathohistological analysis of myocardium and lung tissue samplesTissue analysis of the myocardium and lungs was performed using light microscopy for morphological and descriptive evaluation. Two independent pathologists blinded to group assignments performed all histological assessments. The observed histological changes were analyzed in relation to the different temperature exposures. The key pathological features included degenerative changes, such as sarcoplasmic disintegration and nuclear alterations. Blood vessels showed endothelial lining with prominent nuclei, indicating hyperthermia-induced vascular responses. Statistical analysisStatistical analysis was conducted using the Statistical Package for the Social Sciences, version 25.0. The Shapiro-Wilk test was used to assess whether the data followed a normal distribution. Analysis of variance was employed to compare parameters across multiple groups when normality assumptions were met, while the Kruskal-Wallis test was used for non-normally distributed variables. In cases where significant differences were identified among three or more groups with non-normal distribution, pairwise comparisons were carried out using the Mann–Whitney U test. A p-value of 0.05 was considered statistically significant. Ethical approvalThe Ethical Committee of the Faculty of Medicine, University of Sarajevo, approved the study (reference 02-3-4-1253/20). ResultsThe baseline core temperature was lowest in group Heat stroke-44, whereas the baseline temperature was highest in group Control-37. After 20 minutes of exposure, the lowest temperature was recorded in Control-37 (37.75°C), whereas the highest was 43.09°C ± 0.79°C in Heat stroke-44. At the time of death, Control-37 showed the lowest temperature at 34.45°C ± 2.44°C, compared to Heat stroke-44, which showed the highest temperature (Table 1). Table 1. Mean body temperature values.
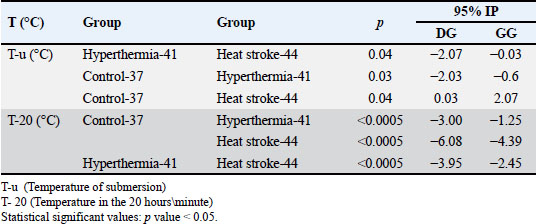
Table 2 shows only significant differences in the body temperatures of rats in the observed groups, and significant temperature differences occurred mostly after 20 minutes of exposure and death. Table 2. Differences in the body temperature values of the rats of the experimental groups at the time points.
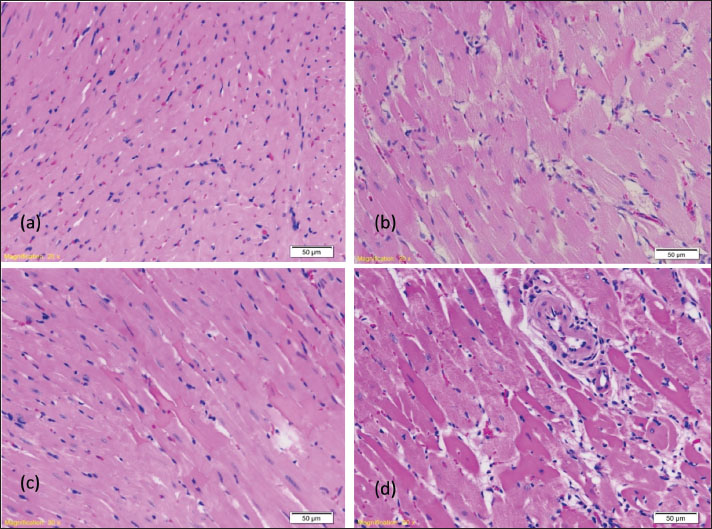
Morphological and descriptive analysis of rat myocardiumFigure 2a shows the left ventricular myocardium of the rat at low and high magnification. The large eosinophilic cytoplasm of cardiomyocytes occupies most of the surface area. Most cardiomyocytes are oriented longitudinally in these photomicrographs, and faint transverse striae are evident at high magnification. The cardiomyocyte nuclei are large, oval to rectangular, and contain granular chromatin with one or two nucleoli. A delicate interstitium containing fibroblasts and capillaries lined with endothelial cells is located between the cardiomyocytes. Fibroblasts and endothelial cells appear to be similar: both have sparse cytoplasm and small oval to flat clusters of basophilic nuclei. Erythrocytes are present inside the capillaries. The left ventricle myocardium of the control group of rats that were exposed to the temperature of 37˚C. Most cardiomyocytes are longitudinally oriented with large oval to rectangular nuclei. A delicate interstitium containing fibroblasts and capillaries lined with endothelial cells is located between the cardiomyocytes.
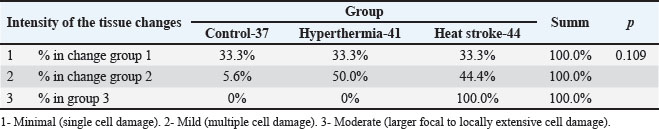
Fig. 2. (a) Control group: myocardium of the left ventricle of the rat; HE, 200×; (b) hyperthermia-41 group: rat left ventricular myocardium: second degree with multiple myocardial cell damage (in the central part of the photomicrograph), HE, 20×; (c) and (d) heat stroke-44 group: rat left ventricular myocardium: second degree with greater focal myocardial cell damage, HE, 20×. In a semiquantitative analysis of myocardial lesions caused by exposure to water temperatures of 41˚C and 44˚C, we analyzed the type and extent of myocardial changes in rats in experimental groups depending on the target water temperature and exposure duration. No signs of damage to individual cardiomyocytes were observed in rats, aligning with the first degree in the severity classification of myocardial alteration. More pronounced injury, ranging from focal to locally extensive cardiomyocyte damage consistent with third-degree changes, was detected in 2 rats from the heat stroke-44 group. No myocardial samples from any experimental group exhibited pathological features corresponding to fourth-degree changes (Table 3). Table 3. Frequency of rats in experimental groups according to the intensity of tissue changes.
Analyzing the type of changes in the experimental groups, similar types of changes were observed depending on the experimental groups. Analyzing the extent of the changes, damage to individual cardiomyocytes and gentle damage to individual cardiomyocytes in the Hyperthermia-41 and Heat stroke-44 groups were observed, with sometimes greater focal damage to cardiomyocytes in the Heat stroke-44 group changes, while the other groups (Hyperthermia-41, Heat stroke-44) developed minimal and multifocal degenerative changes, sometimes with mineralization (Table 4). Table 4. Pathological type of changes.
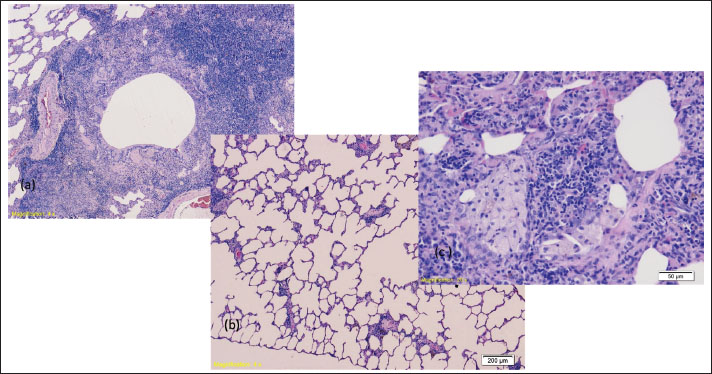
Figure 2b shows the second degree of types of changes, which is manifested through minimal and multifocal changes with minimal damage to individual cardiomyocytes. The usual structuring of the sarcoplasm, in which the network connection of individual cardiomyocytes is presented, is present. Blood vessels are lined with endothelial cells whose nuclei are large and darker in color. Figure 2c and d) shows the myocardium of rats from the experimental group that was exposed to a water temperature of 44˚C until death (heat stroke-44), where the development of extensive changes through greater focal extensive damage to cardiomyocytes is also analyzed, which is also the largest extensive change that occurred in our research. We monitored the degenerative changes, including the loss of the usual structure of the sarcoplasm, which does not show the individual cardiomyocytes’ network connection. Areas of myofibrillar degeneration are presented as brighter zones of the myocardium in which cardiomyocytes have a lower affinity for colors. Nuclei are of altered shape and position, pyknotic, and nucleoli stand out only in certain cardiomyocytes. Blood vessels are lined with endothelial cells whose nuclei are large and darker in color. Morphological-descriptive analysis of rat lung tissueThe lungs of all examined rats showed signs of acute bronchitis and bronchiolitis along with initial bronchopneumonia (Fig. 3 a). Focal acute emphysema (Fig. 3 b) and focal clusters of foamy macrophages (Fig. 3 c) were also observed.
Fig. 3. (a) Signs of bronchiolitis and consolidation of the lung parenchyma with congestion of blood vessels (HE, 4×); (b) hyperthermia-41 group: Initial signs of emphysema of the lung parenchyma (HE, 4×); heat stroke-44 group: consolidated lung parenchyma, with infiltration of inflammatory cells and the presence of small groups of foamy macrophages, the remains of aerated alveoli are observed (HE, 20×). DiscussionThe annual occurrence of SCD increases with age, being 100 times less common in people aged 30 years (0.001%) than in those aged >35 years (4.7%) (Luterbacher et al., 2004; Zhao et al., 2016; Illing et al., 2020). Globally, there has been a rise in indoor deaths, which are often associated with increased room temperatures. Autopsy rates are generally low and vary significantly by country, and protocols for conducting autopsies in cases of suspected SCD can differ substantially, even within the same country. These discrepancies in autopsy rates and methods likely contribute to variations in the reported incidence of SCD and its underlying causes. Scientific evidence regarding the role of hyperthermia as a cause of SCD is limited, highlighting the need for further research in this area. The number of sudden deaths resulting from hyperthermia is steadily increasing. Recent scientific findings indicate a causal link between hyperthermia and cardiac response based on the pathophysiological sequence of events during hyperthermia, but the exact mechanism remains unknown (Burhans et al., 2022). Diagnosing heat-related deaths at autopsy can be particularly challenging, as nonspecific features, such as skin slippage, pleural petechiae, and pulmonary and cerebral edema, are commonly observed. Core body temperature measurements taken near the time of death are rarely available, and elevated temperatures accelerate decomposition, further complicating postmortem assessment (Cioffi et al., 2024). Our aim was to develop a hyperthermia model that leads to death after exhausting the body’s thermoregulatory capacity (Morentin et al., 2019). Sugishita et al. examined causes of death in autopsies of individuals who died suddenly while engaging in sports and recreation to investigate SCD during exertion (sports and light daily activities) (Sugishita et al., 1983). High ambient temperatures trigger certain processes in the body, leading to the failure of compensatory mechanisms. Environmental factors, such as high external temperatures and excessive physical exertion, may contribute to sudden death. In autopsies of sudden death cases occurring after exertion, coronary artery sclerosis and valvular heart disease were identified in 67.0% and 7.1% of the instances, respectively (Toukola et al., 2015). SCD during exertion may also depend on predisposing heart conditions. A statistically significant correlation was noted between heat exhaustion in subjects and cardiac tissue alterations compared with the control group. Heat stroke is a life-threatening disorder characterized by a complete loss of thermoregulation, with an internal temperature exceeding 40°C and multiple organ dysfunction, including the lungs and muscles. Multiorgan failure leads to death, and the mortality associated with high temperatures ranges from 33% to 80%. Terminal events can include shock, arrhythmias, myocardial infarction, renal and liver failure, and a range of neurological deficits (Toukola et al., 2015). The body relies on two primary mechanisms to dissipate heat under thermal stress: sweat evaporation and increased blood flow to the skin. Sweating cools the body as moisture evaporates from the skin, while increased blood flow to the skin helps release heat by bringing warm blood to the surface. However, when these mechanisms are overwhelmed, the body’s core temperature rises, which can lead to heatstroke—a life-threatening condition characterized by severe hyperthermia and central nervous system dysfunction (Toukola et al., 2015). The ability to effectively dissipate heat depends on a properly functioning cardiovascular system. To ensure adequate heat loss, the heart must increase cardiac output and expand the blood vessels in the skin (cutaneous vasodilation). If the cardiovascular system is impaired, the body may struggle to enhance skin blood flow, stroke volume, and overall cardiac output, limiting its ability to regulate temperature. This can significantly increase the risk of heatstroke, particularly in individuals with pre-existing cardiovascular issues. Significant pathological changes were observed in the lung tissue samples. Notable pathological alterations, including inflammation, necrosis, degeneration, hyperplasia, and fibrosis, were documented, and representative characteristics were selected for the overall assessment. Lung injury assessments were based on alveolar wall thickening, congestion, and inflammation. The lungs appeared heavy, with cut sections revealing severe pulmonary edema and congestion, as well as subpleural hemorrhages. All hyperthermic rats exhibited pronounced lung congestion and hemorrhage, with tissue samples indicating acute bronchitis, along with symptoms of initial pneumonia. We also noted instances of focal acute emphysema and foamy macrophage accumulation. Apart from the liver, brain, and kidneys, the lungs are the organ most significantly affected during heatstroke, i.e., hyperthermia (Marchand and Gin, 2022). Pulmonary edema with pulmonary congestion is a common complication that develops in patients after a heat stroke, especially if they were not adequately hydrated. The reasons for the occurrence of the resulting pulmonary edema are multiple and include excessive and inadequate fluid replacement, endothelial injury, acute kidney injury, and transient heart muscle dysfunction. A high incidence of acute respiratory distress syndrome with as much as 75% mortality is observed in patients with heatstroke (Wallace et al., 2007). In fact, lung involvement is part of a systemic response. Pathological changes in the lungs of rats occurred upon exposure to a temperature of 39°C in the form of blood vessel dilation, congestion, and lung matrix thickening. The congestion became more pronounced with the increase in temperature, with the presence of bleeding in the alveolar space, desquamation of epithelial cells, and damage to the structure of the pulmonary alveoli (Wallace et al., 2007). Human studies indicate the development of pulmonary edema as a complication of heat stroke, with the development of multiorgan damage in the form of excessive fluid accumulation, endothelial damage with fluid leakage from vascular spaces, acute kidney injury, and transient heart muscle dysfunction. The postmortem diagnosis of death caused by heat stroke presents specific challenges. Typically, measuring the deceased’s temperature is not a standard practice. Moreover, macroscopic and microscopic findings may lack specificity, particularly if the survival time was brief. Investigative measures, the surrounding circumstances, and the careful exclusion of other potential causes of death are essential for diagnosing hyperthermia. Although the essence of forensic medicine is to determine whether a death was violent, identifying the cause of sudden death and markers that may indicate the most likely cause post-mortem is highly beneficial (WHO, 2002). The postmortem diagnosis of death resulting from hyperthermia and heat stroke poses specific challenges. The autopsy findings are complicated if the body has been in a hot environment for an extended period and rapidly enters the decomposition stage. Investigative actions are crucial for diagnosing hyperthermia-related deaths (Bustinza et al., 2013). The absence of standardized diagnostic criteria for hyperthermia-related deaths is a major challenge for forensic pathologists. To address this issue, the National Association of Medical Examiners has defined heat-related deaths as those resulting from exposure to high ambient temperatures that either caused or significantly contributed to the fatal outcome. The Association also advises that diagnosing heat-related deaths should be based on a history of high-temperature exposure, thorough investigation, and elimination of other potential causes of death (Donoghue et al., 1997). Given the increasing likelihood of heat-related deaths being processed through forensic facilities, identifying high-risk periods is crucial so that morgues can prepare for the anticipated workload increase associated with these types of deaths. To this end, a mathematical metric known as the excess heat factor has recently been developed based on long-term local meteorological data. This factor provides a more precise assessment of a heat event’s severity, offering a better alternative to relying solely on maximum daily temperatures for tracking heat stress. This calculation enables a more accurate quantification of the environmental temperature load, thereby predicting the likelihood of a significant increase in the number of heat-related fatalities. It can also be used in clinical settings to forecast increases in heat-related morbidity (Byard, 2013; Hall et al., 2021). ConclusionThe most representative changes were found in rats exposed to compensatory mechanisms for extended periods, reflecting the development and severity of tissue damage. Our findings indicate that vascular changes occur shortly after hyperthermia onset. While some organs exhibit greater tolerance to heat strokes, many demonstrate similar alterations characterized by capillary dilation and interstitial extravasation, observable after 30 minutes at 40.5°C. A notable limitation is the absence of Immunohistochemistry (IHC) investigations. Immunohistochemistry would have enabled us to explore the molecular and cellular mechanisms underlying the observed morphological changes, such as the expression of heat shock proteins, inflammatory markers, and apoptotic factors. In addition, one of the biggest achievements in bioanalytical chemistry is the creation of immunosensors. The reason for this success is that it attains the exceptional selectivity that the antibodies exhibit because a coating of immunoglobulin G on the sensor’s surface is immobilized to produce them. Living organisms produce antibodies and serve as a defense against chemicals known as antigens. Serum proteins are what they are. These two antagonists combine to create a stable complex during the antigen-antibody response, which shields the body from the foreign material. Including IHC would have provided a more comprehensive understanding of the cellular and molecular responses of tissues to hyperthermic stress. Another limitation of this study is the relatively small sample size. Although the results provide valuable preliminary data, a larger cohort of animals would have enhanced the statistical power and robustness of our findings. Future studies with a larger number of animals will be essential for confirming our observations and providing a more comprehensive understanding of HTID. AcknowledgementsNone. Conflict of interestThe authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper. FundingNo funding. Authors’ contributionsAll authors contributed equally to this research. Data availabilityAll data are provided in the manuscript. ReferencesBurhans, W.S., Rossiter Burhans, C.A. and Baumgard, L.H. 2022. Invited review: lethal heat stress: The putative pathophysiology of a deadly disorder in dairy cattle. J. Dairy Sci. 105(5), 3716–3735; doi:10.3168/jds.2021-21080 Bustinza, R., Lebel, G., Gosselin, P., Bélanger, D. and Chebana, F. 2013. Health impacts of the July 2010 heat wave in Québec, Canada. BMC. Public Health 13(1), 56; doi:10.1186/1471-2458-13-56 Byard, R.W. 2013. Heat-related deaths. Forensic Sci. Med. Pathol. 9(4), 469–470; doi:10.1007/s12024-013-9413-y Cioffi, A., Cecannecchia, C., Baldari, B., De Simone, S. and Cipolloni, L. 2024. Fatal Heat Stroke: a Case Report and Literature Review. Forensic Sci. 4(3), 417–431; doi:10.3390/forensicsci4030026 Dervišević, E., Čamdžić, N., Kuskunović-Vlahovljak, S. and Dervišević, M. 2023a. Multiorgan morphological changes caused by hyperthermia: case study on experimental model. Acta Marisiensis - Seria. Medica. 69(3), 220–225; doi:10.2478/amma-2023-0026 Dervišević, E., Dorić, M., Radović, S., Dervišević, L., Katica, M., Ajanović, Z., Salihbegović, A. and Sarajlić, N. 2023b. Morphological changes in myocardium of Wistar rat caused by hyperthermia. J. King Saud Univ. - Sci. 35(2), 102473; doi:10.1016/j.jksus.2022.102473 Donoghue, E.R., Graham, M.A., Jentzen, J.M., Lifschultz, B.D., Luke, J.L. and Mirchandani, H.G. 1997. Criteria for the Diagnosis of Heat-Related Deaths: national Association of Medical Examiners. Am. J. Forensic Med. Pathol. 18(1), 11–14; doi:10.1097/00000433-199703000-00002 Ferron, C., Trewick, D., Le Conte, P., Batard, E., Girard, L. and Potel, G. 2006. Canicule de l’été 2003: étude descriptive des décès par coup de chaleur au CHU de Nantes. La Presse Médicale 35(2), 196–199; doi:10.1016/S0755-4982(06)74553-5 Gomes, I.P., Duarte, J.A., Maia, A.L.C., Rubello, D., Townsend, D.M., de Barros, A.L.B. and Leite, E.A. 2019. Thermosensitive nanosystems associated with hyperthermia for cancer treatment. Pharmaceuticals 12(4), 171; doi: 10.3390/ph12040171 Hall, C., Ha, S., Yen, I.H. and Goldman-Mellor, S. 2021. Risk factors for hyperthermia mortality among emergency department patients. Ann. Epidemiology 64, 90–95; doi:10.1016/j.annepidem.2021.09.009 Heled, Y., Fleischmann, C. and Epstein, Y. 2013. Cytokines and their role in hyperthermia and heat stroke. Jbcpp 24(2), 85–96; doi:10.1515/jbcpp-2012-0040 Iba, T., Kondo, Y., Maier, C.L., Helms, J., Ferrer, R. and Levy, J.H. 2025. Impact of hyper- and hypothermia on cellular and whole-body physiology. J. Intensive Care 13(1), 4; doi:10.1186/s40560-024-00774-8 Ieda, M. and Fukuda, K. 2015. Cardiac Innervation and Sudden Cardiac Death. Circulat. Res. 116(12), 2005–2019; doi:10.1161/CIRCRESAHA.116.304679 Illing, G., Lessig, R. and Stang, A. 2020. Analyse der Kodierungsqualität der Leichenschauscheine und Obduktionsraten in Chemnitz, 2010–2013. Das Gesundheitswesen 82(04), 354–360; doi:10.1055/a-0820-3452 International Guiding Principles for Biomedical Research Involving Animals (IGPBRIA). 2012. Available via https://media-01.imu.nl/storage/iclas.org/5196/cioms-iclas-principles-final.pdf Lassche, G., Crezee, J. and Van Herpen, C.M.L. 2019. Whole-body hyperthermia in combination with systemic therapy in advanced solid malignancies. Crit. Rev. Oncology/Hematology 139, 67–74; doi:10.1016/j.critrevonc.2019.04.023 Lepock, J.R. 2003. Cellular effects of hyperthermia: relevance to the minimum dose for thermal damage. Int. J. Hyperthermia 19(3), 252–266; doi:10.1080/0265673031000065042 Luterbacher, J., Dietrich, D., Xoplaki, E., Grosjean, M. and Wanner, H. 2004. European Seasonal and Annual Temperature Variability, Trends, and Extremes Since 1500. Science 303(5663), 1499–1503; doi:10.1126/science.1093877 Marchand, M. and Gin, K. 2022. The Cardiovascular System in Heat Stroke. CJC. Open 4(2), 158–163; doi:10.1016/j.cjco.2021.10.002 Marui, S., Misawa, A., Tanaka, Y. and Nagashima, K. 2017. Assessment of axillary temperature for the evaluation of normal body temperature of healthy young adults at rest in a thermoneutral environment. J. Physiol. Anthropol. 36(1), 18; doi:10.1186/s40101-017-0133-y Mørch, S.S., Andersen, J.D.H. and Bestle, M.H. 2017. Treatment of hyperthermia. Ugeskrift For Laeger. 179(30), 179. Morentin, B., Suárez-Mier, M.P., Monzó, A., Molina, P. and Lucena, J.S. 2019. Sports-related sudden cardiac death due to myocardial diseases on a population from 1–35 years: a multicentre forensic study in Spain. Forensic Sci. Res. 4(3), 257–266; doi:10.1080/20961790.2019.1633729 Natarajan, R., Northrop, N.A. and Yamamoto, B.K. 2015. Protracted effects of chronic stress on serotonin-dependent thermoregulation. Stress 18(6), 668–676; doi:10.3109/10253890.2015.1087502 Pereira Gomes, I., Aparecida Duarte, J., Chaves Maia, A.L., Rubello, D., Townsend, D.M., Branco De Barros, A.L. and Leite, E.A. 2019. Thermosensitive nanosystems associated with hyperthermia for cancer treatment. Pharmaceuticals 12(4), doi:10.3390/ph12040171 Quinn, C.M., Duran, R.M., Audet, G.N., Charkoudian, N. and Leon, L.R. 2014. Cardiovascular and thermoregulatory biomarkers of heat stroke severity in a conscious rat model. J. Appl. Physiol. 117(9), 971–978; doi:10.1152/japplphysiol.00365.2014 Richmond, V.L., Davey, S., Griggs, K. and Havenith, G. 2015. Prediction of Core Body Temperature from Multiple Variables. Ann. Occup. Hygiene 59(9), 1168–1178; doi:10.1093/annhyg/mev054 Robayo-Amortegui, H., Quintero-Altare, A., Florez-Navas, C., Serna-Palacios, I., Súarez-Saavedra, A., Buitrago-Bernal, R. and Casallas-Barrera, J.O. 2024. Fluid dynamics of life: exploring the physiology and importance of water in the critical illness.In Frontiers in Medicine (Vol. 11). Frontiers Media SA, 11, https://doi.org/10.3389/fmed.2024.1368502 Sacco, M.A., Gualtieri, S., Calanna, L., Ricci, P. and Aquila, I. 2023. Exploring the Potential of Proteome Analysis as a Promising Tool for Evaluation of Sudden Cardiac Death (SCD) in Forensic Settings: a Literature Review. Int. J. Mol. Sci. 24(18), 14351; doi:10.3390/ijms241814351 Sessa, F., Chisari, M., Salerno, M., Esposito, M., Zuccarello, P., Capasso, E., Scoto, E. and Cocimano, G. 2024. Congenital heart diseases (CHDs) and forensic investigations: searching for the cause of death. Exp. Mol. Pathol. 137, 104907; doi:10.1016/j.yexmp.2024.104907 Sessa, F., Esposito, M., Messina, G., Di Mizio, G., Di Nunno, N. and Salerno, M. 2021. Sudden Death in Adults: a Practical Flow Chart for Pathologist Guidance. Healthcare 9(7), 870; doi:10.3390/healthcare9070870 Sugishita, Y., Matsuda, M., Iida, K., Koshinaga, J. and Ueno, M. 1983. Sudden cardiac death at exertion. Japanese Circulat. J. 47(5), 562–572; doi:10.1253/jcj.47.562 Thommen, O. 2005. Heat wave 2003 and mortality in Switzerland. Swiss Med. Weekly , doi:10.4414/smw.2005.11009 Toukola, T., Hookana, E., Junttila, J., Kaikkonen, K., Tikkanen, J., Perkiömäki, J., Kortelainen, M.L. and Huikuri, H.V. 2015. Sudden cardiac death during physical exercise: characteristics of victims and autopsy findings. Ann. Med. 47(3), 262–267; doi:10.3109/07853890.2015.1025824 Van Den Tweel, J.G. and Wittekind, C. 2016. The medical autopsy as quality assurance tool in clinical medicine: dreams and realities. Virchows Archiv. 468(1), 75–81; doi:10.1007/s00428-015-1833-5 Van Rhoon, G.C., Franckena, M. and Ten Hagen, T.L.M. 2020. A moderate thermal dose is sufficient for effective free and TSL based thermochemotherapy. Adv. Drug Del. Rev. 163–164, 145–156; doi:10.1016/j.addr.2020.03.006 Wallace, W.A.H., Fitch, P.M., Simpson, A.J. and Howie, S.E.M. 2007. Inflammation-associated remodelling and fibrosis in the lung – a process and an end point. Int. J. Exp. Pathol. 88(2), 103–110; doi:10.1111/j.1365-2613.2006.00515.x Walter, E.J. and Carraretto, M. 2016. The neurological and cognitive consequences of hyperthermia. Crit. Care 20(1), 199; doi:10.1186/s13054-016-1376-4 Yang, J., Weng, W., Wang, F. and Song, G. 2017. Integrating a human thermoregulatory model with a clothing model to predict core and skin temperatures. Appl. Ergonom. 61, 168–177; doi:10.1016/j.apergo.2017.01.014 Zhao, P., Wang, J.G., Gao, P., Li, X. and Brewer, R. 2016. Sudden unexpected death from natural diseases: fifteen years’ experience with 484 cases in Seychelles. J. Forensic Legal Med. 37, 33–38; doi:10.1016/j.jflm.2015.10.004 | ||
| How to Cite this Article |
| Pubmed Style Dervišević E, Šunjić I, Dervišević M, Sessa F, Spahović H, Bešić A, Krupić F, Ajanović T. Hyperthermia-induced sudden death: Morphological insights from the lung and heart in an experimental study of Wistar rats. Open Vet. J.. 2025; 15(10): 4962-4971. doi:10.5455/OVJ.2025.v15.i10.15 Web Style Dervišević E, Šunjić I, Dervišević M, Sessa F, Spahović H, Bešić A, Krupić F, Ajanović T. Hyperthermia-induced sudden death: Morphological insights from the lung and heart in an experimental study of Wistar rats. https://www.openveterinaryjournal.com/?mno=267059 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.15 AMA (American Medical Association) Style Dervišević E, Šunjić I, Dervišević M, Sessa F, Spahović H, Bešić A, Krupić F, Ajanović T. Hyperthermia-induced sudden death: Morphological insights from the lung and heart in an experimental study of Wistar rats. Open Vet. J.. 2025; 15(10): 4962-4971. doi:10.5455/OVJ.2025.v15.i10.15 Vancouver/ICMJE Style Dervišević E, Šunjić I, Dervišević M, Sessa F, Spahović H, Bešić A, Krupić F, Ajanović T. Hyperthermia-induced sudden death: Morphological insights from the lung and heart in an experimental study of Wistar rats. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4962-4971. doi:10.5455/OVJ.2025.v15.i10.15 Harvard Style Dervišević, E., Šunjić, . I., Dervišević, . M., Sessa, . F., Spahović, . H., Bešić, . A., Krupić, . F. & Ajanović, . T. (2025) Hyperthermia-induced sudden death: Morphological insights from the lung and heart in an experimental study of Wistar rats. Open Vet. J., 15 (10), 4962-4971. doi:10.5455/OVJ.2025.v15.i10.15 Turabian Style Dervišević, Emina, Ines Šunjić, Muamer Dervišević, Francesco Sessa, Hajrudin Spahović, Aida Bešić, Ferid Krupić, and Temima Ajanović. 2025. Hyperthermia-induced sudden death: Morphological insights from the lung and heart in an experimental study of Wistar rats. Open Veterinary Journal, 15 (10), 4962-4971. doi:10.5455/OVJ.2025.v15.i10.15 Chicago Style Dervišević, Emina, Ines Šunjić, Muamer Dervišević, Francesco Sessa, Hajrudin Spahović, Aida Bešić, Ferid Krupić, and Temima Ajanović. "Hyperthermia-induced sudden death: Morphological insights from the lung and heart in an experimental study of Wistar rats." Open Veterinary Journal 15 (2025), 4962-4971. doi:10.5455/OVJ.2025.v15.i10.15 MLA (The Modern Language Association) Style Dervišević, Emina, Ines Šunjić, Muamer Dervišević, Francesco Sessa, Hajrudin Spahović, Aida Bešić, Ferid Krupić, and Temima Ajanović. "Hyperthermia-induced sudden death: Morphological insights from the lung and heart in an experimental study of Wistar rats." Open Veterinary Journal 15.10 (2025), 4962-4971. Print. doi:10.5455/OVJ.2025.v15.i10.15 APA (American Psychological Association) Style Dervišević, E., Šunjić, . I., Dervišević, . M., Sessa, . F., Spahović, . H., Bešić, . A., Krupić, . F. & Ajanović, . T. (2025) Hyperthermia-induced sudden death: Morphological insights from the lung and heart in an experimental study of Wistar rats. Open Veterinary Journal, 15 (10), 4962-4971. doi:10.5455/OVJ.2025.v15.i10.15 |