| Research Article | ||
Open Vet. J.. 2025; 15(10): 5009-5017 Open Veterinary Journal, (2025), Vol. 15(10): 5009-5017 Research Article Pharmacokinetic evaluation of a long-acting, tasteless oral formulation of enrofloxacin in dogsLilia Gutierrez1, Mayte Hernández1, Jorge Luna del Villar2, Zacnite Sánchez1 and Hector Sumano1*1Departamento de Fisiología y Farmacología, Facultad de Medicina Veterinaria y Zootecnia, Universidad Nacional Autónoma de México, Mexico City, Mexico 2Departamento de Medicina, Cirugía y Zootecnia para Pequeñas Especies, Facultad de Medicina Veterinaria y Zootecnia, Universidad Nacional Autónoma de México, Mexico City, Mexico *Corresponding Author: Hector Sumano. Departamento de Fisiología y Farmacología, Universidad Nacional Autónoma de México, Ciudad de México, Coyoacan, México. Email: sumano [at] unam.mx Submitted: 26/06/2025 Revised: 30/08/2025 Accepted: 11/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
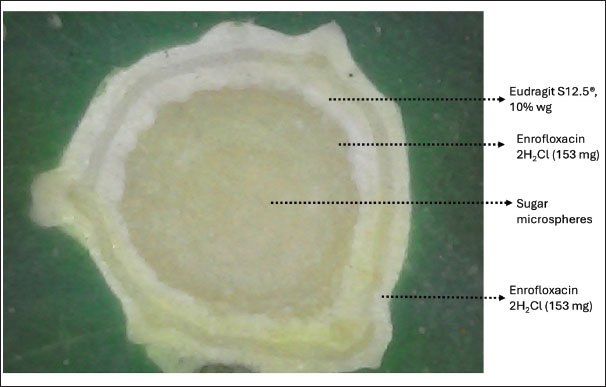
AbstractBackground: Owner medication compliance with the clinician’s prescription can influence the patient’s outcome, among other factors. Extending the dosing interval (DI) may improve medication compliance. Therefore, an oral long-acting (LA) preparation of enrofloxacin as minispheres was developed. Aim: To develop a tasteless LA preparation of enrofloxacin as minispheres that can achieve Cmax and AUC values equal to or greater than those of the reference flavored tablet. Additionally, the dose was concealed in either pate or cream cheese morsels to facilitate dosing of the experimental minispheres. Methods: The pharmacokinetics (PK) of the reference preparation and the experimental minispheres were defined by dosing them as two 10 mg/kg/day or 15 mg/kg once, respectively. The HCl·2H2O solvate of enrofloxacin was used together with pH-sensitive multilayer polymers to manufacture the minispheres. To facilitate dosing, the minispheres were concealed in pate or cream cheese morsels. HPLC analysis allowed the determination of the reference enrofloxacin plasma concentrations for both preparations. Results: The Cmax and AUC0–48 were highest for the minispheres concealed in pate, followed by cream cheese morsels. When assessing the key PK/PD ratios for enrofloxacin and considering the breakpoint of 0.5 µg/ml, i.e., Cmax/MIC0.5 ≥ 10–12 and AUC0–24/MIC0.5 ≥ 125, only the experimental minispheres concealed in cream cheese or pâté morsels exceeded the referred ratios. Predictably, minispheres showed a longer mean residence time. Therefore, a DI of 48 hours is proposed for the experimental minispheres. Conclusion: Given the PK/PD relationships obtained with the experimental minispheres, a 15 mg/kg dose every 48 hours (15 mg/kg) is recommended. This prototype will be presented to the veterinary clinical community for further evaluation to assess clinical efficacy and medication compliance performance. Keywords: Medication compliance, Dogs, Enrofloxacin, Long-acting, PK/PD. IntroductionSeveral factors modify the success of a given treatment with antibacterial drugs in veterinary medicine. Even with a correct diagnosis and an appropriate treatment, the therapeutic success may be suboptimal as the owner could fail to administer the medication as indicated on time or in an inadequate morsel (Eisen et al, 1990; Casey and Bradshaw, 2008; Maddison et al., 2021). Care for these peculiarities in each case is particularly relevant in medium- and long-term treatment. In addition, dogs may often reject oral medications, forcing the situation (Eisen et al., 1990; Maddison et al., 2021). If medication compliance is defective, the emergence of resistant bacterial organisms is also facilitated, and the veterinarian and owner’s perception could be that the drug is ineffective, doses were too low, or the diagnosis was incorrect (Barter et al., 1996). It must be easily administered to encourage medication compliance with the clinician’s instructions using a given drug preparation, and the experience must not be unpleasant to either the owner or the dog. It should not interfere with the owner’s schedule because the risk of non-medication compliance will increase. For example, as the number of drug administration increases, owner non-medication compliance also increases (Barter et al., 1996; Maddison et al., 2021). When a treatment is designed and includes a follow-up at home by the owner, it is best not to recommend a parenteral-based drug preparation, thus avoiding ethical misconduct. If additional parenteral treatment is necessary, it should be administered only by a certified nurse or veterinarian. When enrofloxacin is orally administered, it provides therapeutic concentrations that allow a DI of 24 hour. Treatment duration varies according to the different infections that affect dogs (López-Cardenas et al., 2013; Sumano et al., 2018; Maris et al., 2021). Enrofloxacin has a very unpleasant taste. Hence, various pharmaceutical designs have been proposed (Wu et al., 2024), and a better flavored commercial preparation (BaytrilTM Flavor-Elanco, Mexico) is commercially available. However, there is no long-acting commercial preparation of enrofloxacin for parenteral administration in small species. However, the problem is not entirely solved with the commercially available preparation because the taste is not acceptable to all dogs. The acceptance of enrofloxacin can be solved by including it in alginates, as it has already been shown in dogs and cats. (Gutierrez et al., 2021a, 2021b). However, this presentation of enrofloxacin could be further improved by extending the DI, using biocompatible pharmaceutical vehicles that can generate a modified release—long-acting (LA) preparation (Martinez et al., 2010; McDowell and Rathbone, 2014), yet, achieving a maximum serum concentration (Cmax) as high as possible with respect to the MIC (Cmax/MIC ≥ 10–12 ratio) and, at the same time, an extension of its body residence time, to achieve a large relationship of the AUC divided by MIC (AUC/MIC ≥ 125) (Canut et al., 2015; Grabowski et al., 2022). These ratios have been linked to the clinical efficacy of enrofloxacin and must be considered when designing a new tasteless preparation of enrofloxacin for dogs (Trouchon and Lefevre, 2016; Sumano et al., 2018). Additionally, given the diversity of breeds and, therefore, the sizes of dogs, it was decided for this trial to manufacture small spheres to achieve more accurate dosing and with several layers of pH-dependent Eudragit® in searching for both a high Cmax and an LA pharmacokinetics. Thus, the pharmacokinetics of both the prototype of enrofloxacin HCl 2H2O-C (enro-C: Patent-PCT/MX2014/000192) in small spheres for LA and that of the original product (BaytrilTM Flavor-Elanco, Mexico) in dogs were defined. A sparse sampling approach model was implemented for this purpose (Mahmood and Duan, 2009; Mahmood, 2014). Materials and MethodsPreparation of the small spheresEnro-C was incorporated into 500 g of sugar minispheres with a diameter of 3.8 mm using the pan coating process. The pharmaceutical design consists of multilayers of enro-C incorporated with Opadry® YS-1-7006 clear (Colorcon, Mexico) in an aqueous dispersion and the pH-dependent polymer Eudragit® S12.5 (Evonik, Helm de Mexico) in an organic dispersion, which has enteric protection properties. The total amount of enro-C incorporated was determined in triplicate by grinding 1 g of small spheres and dissolving them in phosphate buffer (pH 7.5) with constant stirring for 30 minutes. The samples were analyzed using the microbiological technique implemented by Balouri et al. (2016), using Escherichia coli ATCC 10536 as an indicator microorganism. A conventional film-coating system, which consists of a double-release system on the sugar core separated by a pH-dependent polymer layer, was used to obtain the multilayer design. Thus, the system has an immediate-release enro-C layer (outer layer), and the enteric polymer Eudragit® S12.5 subsequently releases the active ingredient in alkaline pH, as shown in Figure 1.
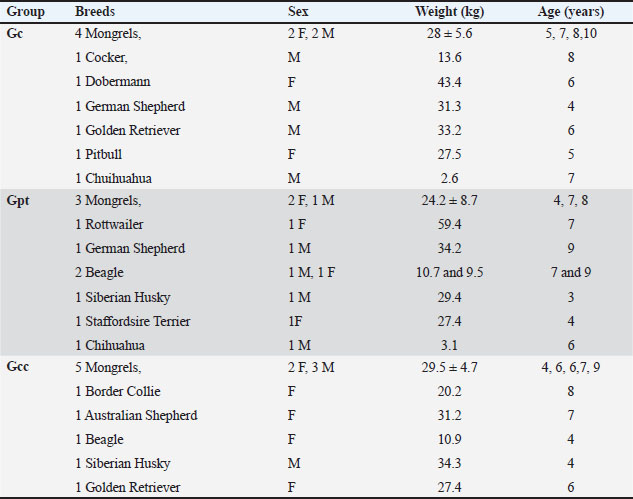
Fig. 1. Section of an enrofloxacin minisphere showing the different layers that allow for modified-release long-acting action. The first layer was formed by incorporating an aqueous dispersion of enro-C with 2% w/v Opadry® clear onto the small spheres of sugar, which were stirred for 45 minutes. The dispersion was then circulated with a peristaltic pump (Watson Marlow) at 5 rpm in conventional film coating equipment at 100 rpm and a drying temperature of 35 ± 2ºC. The second layer formed by Eudragit® S12.5 was obtained with an organic dispersion prepared at 10% weight gain of polymer in a batch of 300 g of small spheres, 10% of triethyl citrate, and 30% of talc was also added, both concerning the weight of the dry polymer (30 g). To ensure fluidity, the dispersion was prepared in isopropanol at 8% of total solids. The film was then circulated with a peristaltic pump (Watson Marlow) at a speed of 15 rpm and incorporated in conventional film coating equipment at 100 rpm and a drying temperature of 30ºC ± 2ºC. From the batch of spheres protected with Eudragit® S12.5, 25 g were coated with a final layer of enro-C with 34 g following the methodology used for the first enro-C incorporation. A surface layer with a 5 mg/kg concentration of enro-C without Opadry® clear was added to achieve a rapid initial release. The in vitro test was performed using a Loderan RC-8DS dissolution apparatus 1, with the basket method, as follows: temperature of 38ºC ± 0.5° C, 50 rpm, and dissolution medium of 900 ml of acetate buffer (pH 3.0), taking 1 ml of sample with medium recovery, every 15 minutes for 60 minutes, and finally, the pH was changed to 7.6 using 900 ml of phosphate buffer, sampling 1 ml with medium recovery, every 15 minutes for 2 hours. The dissolution media were selected based on the pH of the compartments where enro-C needs to be protected or dosed, and the samples were quantified using the agar well diffusion method described by Balouri et al. (2016). Animals and their groupsHealthy animals aged >2 years who attended the clinics for routine non-infectious issues or any pathology were included in this trial. The weight, age, and breeds of the dogs that participated in the study are shown in Table 1. All owners have explained the procedure to be carried out and have given their signed consent to sample their dog 3 to 4 times maximum. A total of 30 dogs were included in this study. The dogs were randomly divided into three groups with 10 dogs each, as follows: two groups were administered the minispheres LA prototype of enro-C, receiving a single dose of 15 mg/kg, and the minispheres were concealed in either cream cheese (Philadelphia®) (Gcc) or liver pâté (Zwan®) (Gpt). A larger dose was studied to compensate for a longer dosing interval. The reference group received a dose of approximately 10 mg/kg every 24 hours twice (BaytrilTM Flavor-Elanco, Mexico). Table. 1. The weight, age, and breeds of the dogs that participated in the research.
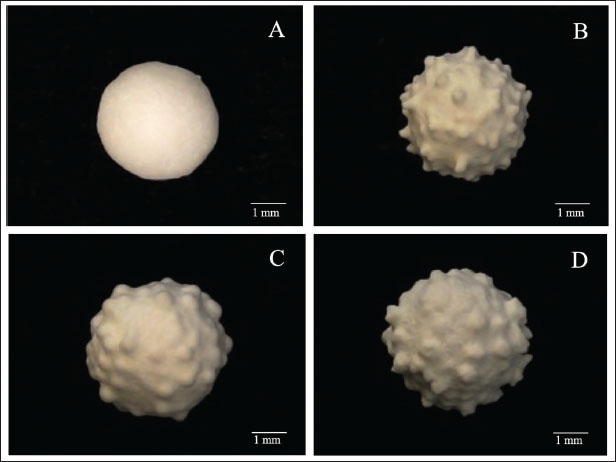
The pharmacokinetic study used a sparse sampling approach to minimize stress on dogs while maintaining statistical validity (Mahmood and Duan, 2009; Mahmood, 2014). The participant dogs were ensured that they did not receive any medication 21 days before the study. They all remained at home and continued to eat their usual diet. Each animal was dosed with the necessary number of small spheres and mixed in a piece of pâté Gpt or cream cheese Gcc to ensure a dose of 15 mg/kg of the prototype. The Baytril® Flavor-Elanco (Gc), Mexico reference group was administered without a morsel. We obtained four samples of 3 ml of blood from each participating dog each time by direct cephalic vein puncturing using heparin-Vacutainer tubes and pediatric butterfly needles. The sampling times chosen were 0.5, 1, 1.5, 2, 2.5, 3, 3.5, 4, 12, 24, 28, 36, and 48 hours after administration of each preparation. Enrofloxacin analysisThe technique for determining plasma enrofloxacin concentrations was based on HPLC according to the methodology originally described by Idowu and Perkins (2004), which was validated in our laboratory (Gutierrez et al., 2020). The method produced a linear result from 0.01 to 20.47 μg/ml (r2=0.984; y=500030x−107,045). The recovery of enrofloxacin was calculated using linear regression analysis. The samples had r2=0.977 (y=0.072322308x + 0.1233375). Precision was demonstrated by the inter-day coefficient of variance (3.1) and inter-assay error value (< 3.8). The lower quantification limit for enrofloxacin in plasma was 0.01 μg/ml with a detection limit of 0.008 μg/ml, and linearity was established up to 10 μg/ml. An absolute difference between 1.77 and a coefficient of variance of 2.2% (< 3.0%) was obtained for robustness and tolerance. Only enrofloxacin, but not its metabolite, ciprofloxacin, was evaluated. Pharmacokinetics and statistical analysisResults obtained by group and by bleeding time were processed using the software OriginPro 2025 (©OriginLab Corporation, One Roundhouse Plaza, Northampton, MA 01060 USA) for graphing, and pharmacokinetic parameters were obtained using PKMP® (APL- Software and Pharmacokinetic Services, 9671 Laforet Drive, Eden Prairie, MN 55347, USA). PKPM® performs pharmacokinetic calculations and curve adjustments using mathematical algorithms that model the behavior of the studied drug, incorporating linear and nonlinear regression analysis, as well as Bayesian methods to estimate pharmacokinetic parameters from individual and population data. The area under the plasma concentration vs. time curve (AUC), the maximum plasma concentration (Cmax) values, and the terminal phase half-life (T½β) were obtained from the PKMP®; the apparent volume of distribution (Vd/F) was calculated as Dose/(AUC * Kel), and the relative bioavailability was determined by comparing AUC values. The Dunn t-test was used to assess statistical differences in pharmacokinetic parameters between groups. Ethical approvalThe Institutional Committee of the National Autonomous University of Mexico approved all procedures for the Research, Care, and Use of Experimental Animals (UNAM- CICUA No. 730), and they were conducted according to Mexican Official Regulations (NOM-062-ZOO-2001). Each pet owner provided a letter of consent for their pet’s participation in the experiment. ResultsSmall sphere prototypeThe enro-C LA minisphere prototype has a yield above 99.6% in both the inner and outer layers. The increase in weight due to the EP polymer was 9.9%, with a yield of 99.76%. Although the supplier’s data sheet suggests that the weight increase for Eudragit polymers depends on the pharmaceutical form, it has been shown that an increase between 5% and 10% is sufficient to provide stability of the enclosed active principle and that it exhibits enteric release (Dvorackova et al., 2010; Jablan and Jug, 2015; Evonik Industries). The outer layer of enro-C was incorporated under the conditions established in the first process using a 225 g batch of small spheres, and the yield was 98.85%. The characterization of the enro-C LA prototype considering weight gain was as follows: the inner layer contained 153 mg enro-C/g and the outer layer 151 mg enro-C/g, giving a total enro-C content of 304 mg in each gram of small spheres. The agar well diffusion method (Balouri et al., 2016) confirmed these values. Figure 1 shows the uniformity of the coating layers of both the drug and the enteric polymer. Figure 2 shows that the initial and final diameters of the minispheres were 3.8 and 5.2 mm, respectively.
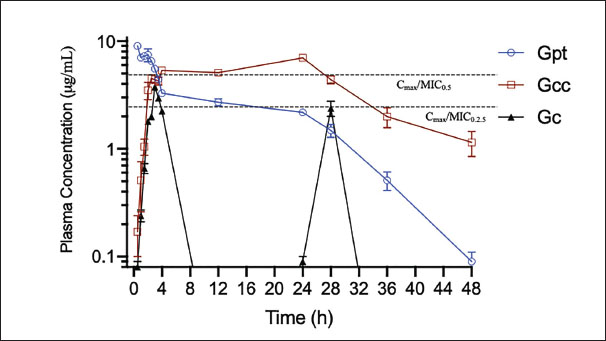
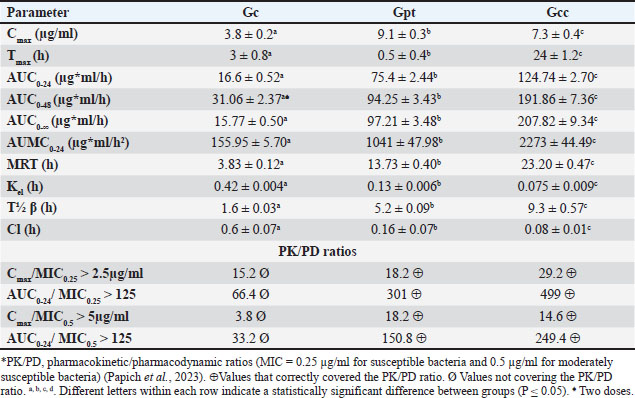
Fig. 2. Images of the layer progression in the prototype of enrofloxacin minispheres, prepared for modified release - long action. The in vitro release test showed an immediate release of enro-C from the outer layer, with 95.66% drug liberation within 15 minutes in acidic pH, similar to that in the stomach. The entire amount of enro-C of the inner layer was released within 130 minutes under alkaline conditions. The release pattern of enro-C from the inner layer of the prototype diffuses through the hydrated polymer matrix after dissolution at intestinal pH. In preparations for specific release of drugs in the colon and sustained release preparations, it was observed that a constant and slow release of the drug occurs when the medium’s pH reaches the polymer’s solubility threshold, thus improving its bioavailability (Sanchez et al., 2014; Li et al., 2014; Patra et al., 2017). First, the polymer swells when its pH is alkaline. Pores are formed through which the drug diffuses outside into the media. At the end of the dissolution test, the residues in empty spheres with almost the same weight as the incorporated polymer were recovered. PharmacokineticsFigure 3 shows the pharmacokinetics of the prototype, either concealed as Gcc or Gpt, obtained plasma concentration vs. time curves compatible with an DI of 48 hours. From this figure, it becomes apparent that if the experimental prototype studied is administered as Gcc, plasma concentrations of enrofloxacin will be prolonged as compared to the same prototype but concealed in patè (Gpt) (T½β 9.3 hours and 5.2 hours, respectively). Consequently, MRT and Kel are also extended (13.7 hours vs. 23.2 hours and 0.075 hours vs. 0.13 hours, respectively, for Gpt and Gcc). Vd values were statistically comparable for the three groups, i.e., 1.4, 1.3, and 1.25 L/kg for the Gc, Gpt, and Gcc groups, respectively (p < 0.05). Table 2 presents the mean pharmacokinetic values ± 1 SD for the evaluated pharmaceutical forms of enrofloxacin and enro-C. The statistically significant values are highlighted, and the PK/PD ratios, Cmax/MIC, and AUC/MIC are presented, considering MIC values retrieved from formal references (Papich et al., 2023). The enro-C LA minispheres prototype achieves unique PK with a DI of almost 48 hours (Gpt) and 48 hours (Gcc), and favorable PK/PD ratios are achieved in both groups.
Fig. 3. Mean ± 1 SD plasma profiles for enrofloxacin minispheres prepared for long action and concealed in cream cheese (Philadelphia®) (Gcc), pâté (Zwan®) (Gpt) administered at a dose of 15 mg/kg. The reference values were obtained from Elanco’s Baytril™ Flavor (Gc), administered twice with a 24 hours interval at 10 mg/kg and without morsel. Table. 2. Mean ± 1 SD pharmacokinetic parameters for the enro-C LA small spheres prototype concealed in cream cheese (Philadelphia®) (Gcc), pâté (Zwan®) (Gpt) administered at a dose of 15 mg/kg. The reference values achieved by Elanco’s Baytril™ Flavor (Gc) administered twice with a 24 hours interval at 10 mg/kg are also presented.
DiscussionThe compliance of pet owners’ medication with veterinary instructions is crucial to ensure optimal results. Several factors influence medication compliance. For example, 47% of dog owners did not fully adhere to prescribed medication regimens. Older dog owners were more compliant, as were those who used morsels to conceal the medication. One-third of the owners reported that their pets were reluctant to take their medication, and it has been determined that owners comply better when the medication is given only once a day (Odom et al., 2024). Accordingly, the objective of this research was to develop a preparation that could both extend the DI and be administered concealed in a morsel without undermining its bioavailability, as it is known to be affected when administered with cheese or sausages (Bushra et al., 2001). With the tasteless prototype achieved in this trial and using pâté or cheese as a morsel, plus clear and specific verbal and/or written instructions as it has been shown that doing so enhances medication compliance (Barter et al., 1996; Humpries, 2008; Gerrard, 2015), under or overdosing may be avoided in this case, given the unusual DI of 48 hours. Hence, this pharmaceutical preparation may merit subsequent medication compliance and efficacy studies, particularly considering that it is almost tasteless and can be administered with either a meaty or cream-cheese morsel (Humpries, 2008; Gerrard, 2015). Hence, there were no rejections or difficulties in dosing dogs with the enro-C LA prototype. Enrofloxacin can display its optimal clinical efficacy when high doses are employed, meeting the PK/PD ratios of a concentration-dependent antibacterial drug (Papich et al., 2023; Trouchon and Lefebvre, 2016). Ideally, plasma concentrations should reach the ratios of Cmax/MIC=10–12 and/or AUC0–24/MIC ≥ 125. Several authors consider the first proportion to be the most important factor in obtaining maximum clinical efficacy (Blondeau and Fitch, 2019; Gutiérrez et al., 2025). Moreover, it has been postulated that the ratio of Cmax/MIC=16 can be considered adequate in its bactericidal effect and it could impede the selection of mutant bacteria (Blondeau & Fitch, 2019). Enro-C LA was designed with an immediate-release coating. The pharmacokinetic values of enro-C are explained in terms of facilitating its higher water solubility compared with that of the parental enrofloxacin, which has a very low water solubility rate (Miranda-Calderón et al., 2014). This feature generated a high Cmax (9.1 ± 0.3 and 7.3 ± 0.4 for pâté and cream cheese, respectively). The other layers permitted a sustained release of enro-C and, consequently, an LA effect. Competitive PK/PD indexes were obtained for Gpt and Gcc, respectively, while AUC0–24/MIC0.25, were 301 and 499 for Gpt and 499 for Gcc. The former ratios can be regarded as mutant preventive ones (Papich et al., 2023), and further studies are required to rule out potential side effects. If a moderately resistant bacterium is considered (0.5 µg/ml), the Cmax/MIC ≥ 10 ratios obtained are as follows: 18.2 and 14.6 for Gpt and Gcc, respectively, and can still be considered adequate. As far as the AUC0–24/MIC0.5 ≥ 125 ratio is concerned, and for a MIC value of 0.5 µg/ml, the ratios are 150 and 249 for Gpt and Gcc, respectively. The reference preparation had an AUC0–24/MIC0.5 ratio of 33. From the pharmacological viewpoint, high clinical efficacy should follow the administration of the minispheres LA of enro-C, delivered with the referred morsels (Ambrose et al., 2003; Gebru et al., 2012). With the experimental data obtained in this trial, it is not possible to explain how such high PK/PD ratios are obtained, particularly when the prototype is included in cream cheese. The presence of divalent ions, such as calcium, has decreased the bioavailability of enrofloxacin (Ziółkowski et al., 2014; Imaoka et al., 2018). In contrast, it has been described that the negative or positive interaction depends on the time-sequence in which the calcium molecules become available for interaction, if gastrointestinal absorption of calcium is allowed in a 2-hour period; presumably, higher concentrations of serum calcium will enhance the antibacterial activity of enrofloxacin and metabolites, at least at the level of the central compartment (Aguilera et al., 2007). This point may be an interesting area for future research, and studies to characterize whether enro-C crystals behave similarly to enrofloxacin-base in the presence of calcium ions are still lacking. Currently, it is necessary to conduct open clinical trials to verify the clinical efficacy linked to the high PK/PD ratios achieved and to study whether the indication of 1 dose of enro-C LA concealed in a morsel every 48 hours shows better compliance indexes than dosing every 24 hours. Additionally, it is necessary to determine whether adverse effects are associated with the notable bioavailability of enro-C in the minispheres, such as erosion in joint cartilages, fragility of tendons and ligaments, and even toxicity in the CNS (Trouchon and Lefebvre, 2016). Finally, it is possible to advance the relative ease of the manufacturing process of enro-C LA to be easily upscaled. ConclusionProducing an enrofloxacin HCl-2H2O (enro-C) preparation as multiple layers of minispheres and free of unpleasant taste for dogs is feasible. Additionally, to facilitate oral dosing, the required dose of small spheres can be concealed in cream cheese or pâté, achieving outstanding key pharmacokinetic parameters, i.e., Cmax, AUC0–24, and MRT, and consequently particularly high PK/PD ratios that support a 48-h DI. In turn, this DI is likely to contribute to better compliance with treatment regimens indicated by clinicians. AcknowledgmentsProject PAPIIT- 200425. Universidad Nacional Autonoma de México FundingNone. Authors’ contributionsL.G. contributed to the study design, performed all pharmacokinetic analyses, data interpretation, statistical analyses, and drafted the manuscript. M.H. contributed to the study design and sample collection. J.L. contributed to the sample collection. Z.S. contributed to the study design, data interpretation, and drafting and revising of the manuscript. H.S contributed to the study design, sample collection, data interpretation, and drafting and revising of the manuscript. Conflict of interestThe authors declare no conflicts of interest associated with this manuscript. Data availabilityData are available upon request. ReferencesAguilera, R., Gutiérrez, O.L. and Sumano, L.H. 2007. Enhancement of enrofloxacin serum antibacterial activity by calcium primed broilers. Res. Vet. Sci. 82, 80–44. Ambrose, P.G., Bhavnani, S.M. and Owens, R.C. 2003. The clinical pharmacodynamics of quinolones. Infect. Dis. Clin. North Am. 17(3), 529–543. Balouiri, M., Sadiki, M. and Ibnsouda, S.K. 2006. Methods for in vitro evaluating antimicrobial activity: a review. J. Pharm. Anal. 6(2), 71–79. Barter, L.S., Watson, A.D. and Maddison, J.E. 1996. Owner Medication Compliance with Short-Term Antimicrobial Medication in Dogs. AVJ 74(4), 277–280. Blondeau, J.M. and Fitch, S.D. 2019. Mutant prevention and minimum inhibitory concentration drug values for enrofloxacin, ceftiofur, florfenicol, tilmicosin, and telithromycin tested against swine pathogens Actinobacillus pleuropneumoniae, Pasteurella multocida, and Streptococcus suis. PLos One 14, 1–10. Bushra, R., Aslam, N. and Khan, A.Y. 2011. Food-drug interactions. Oman Med. J. 26(2), 77–83. Canut, B.A., Aguilar, A.L., Cobo, R.J., Giménez, M.M.J. and Rodríguez-Gascón, A. 2015. Análisis farmacocinético/farmacodinámico en microbiología: una herramienta para la evaluación del tratamiento antimicrobiano. Enf. Infect. Microbiol. Clin. 33(1), 48–57; doi:10.1016/j.eimc.2013.04.023 Casey, R.A. and Bradshaw, J.W.S., 2008. Owner medication compliance and clinical outcome measures for domestic cats undergoing clinical behavior therapy. J. Vet. 58, 59–69. Dvorackova, K., Rabisková, M., Gajdziok, J., Vetchy, D., Muselík, J., Bernatoniene, J., Bajerova, M. and Drottnerova, P. 2010. Coated capsules for drug targeting the proximal and distal parts of the human intestine. Acta Pol. Pharm. 67(2), 191–199. Eisen, S.A., Miller, D.K., Woodward, R.S., Spitznagel, E. and Przybeck, T.R. 2015. The effect of prescribed daily dose frequency on patient medication medication compliance. Arch. Intern. Med. 150(9), 1881–1884; doi:10.1016/j.archinternmed.2015.09.010 Evonik, Industries. 2025. Leading beyond chemistry, infographic. Available via https://www.evonik.com/en/news/press-releases/2024/03/evonik-makes-five-generations-healthier-with-eudragit--coatings-.html Gebru, E., Damte, M., Choi., Lee, Y.H., Kim. and Park. 2012. Mutant prevention concentration and phenotypic and molecular basis of fluoroquinolone resistance in clinical isolates and in vitro-selected mutants of Escherichia coli from dogs. Microb 154(3-4), 384–394. Gerrard, E. 2015. Owner medication compliance—educating clients to act on pet care advice. Vet. Times , 1–8. Grabowski., Gaffke, L., Pierzynowska, K., Cyske, Z., Choszcz, M., Węgrzyn, G. and Węgrzyn, A. 2022. Enrofloxacin—the ruthless killer of eukaryotic cells or the last hope in the fight against bacterial infections?. Int. J. Mol. Sci. 23(7), 3648. Gutiérrez, L., Lechuga, T., Marcos, X., García, P., Gutiérrez, C. and Sumano, H. 2021. Comparative bioavailability of enrofloxacin in dogs when concealed in noncommercial morsels, either as tablet or as enrofloxacin-alginate dried beads. J. Vet. Pharmacol. 58, 59–69. Gutiérrez, L., Mejía-Arthur I Posadas., -Manzano, E., Rodríguez-Becerril, N.L. and ópez-Ordaz R Sumano. 2025. Evaluation of different PK/PD ratios of three enrofloxacin preparations to the clinical response of pneumonic calves. J. Vet. Sci. 26, 7; doi:10.1016/j.jvs.2013.07.016 Gutierrez, L., Tapia, G., Gutierrez, E. and Sumano, H. 2019. Evaluation of a tasteless enrofloxacin pharmaceutical preparation for cats. Naive Pooled-Sample approach to study its pharmacokinetics. Animals 11(8), 2312. Humphries, J.P. 2008. Client communications for good medication compliance. In Proceedings of the 33rd World Small Animal Veterinary Congress, pp 479–482. Imaoka, A., Abiru, K., Akiyoshi, T. and Ohtani, H. 2018. Food intake attenuates the drug interaction between new quinolones and aluminum. J. Pharm. Health Care Sci. 4, 1–5. Jablan, J. and Jug, M. 2015. Development of Eudragitu00ae S100-based pH-responsive zaleplon microspheres by spray-drying: tailoring drug release properties. Powder Technol. 283, 334–343. Li, P., Yang, Z., Wang, Y., Peng, Z., Li, S., Kong, L. and Wang, Q. 2015. Microencapsulation of coupled folate and chitosan nanoparticles for targeted delivery of combination drugs to colon. J. Microencapsul. 32, 40–45. López-Cadenas, C., Sierra-Vega, M., García-Vieitez, J., Diez-Liébana, M., Sahagún-Prieto, A. and Fernández- Martínez, N. 2014. Enrofloxacin: pharmacokinetics and metabolism in domestic animal species. Curr. Drug Metab. 14, 1042–1058. Maddison, J., Cannon, M., Davies, R., Farquhar, R., Faulkner, B., Furtado, T., Harvey, G., Leathers, F., Snowden, A. and Wright, I. 2021. Owner medication compliance in veterinary practice: recommendations from a roundtable discussion. CAH 26(6). Mahmood, I. and Duan, J. 2009. Population pharmacokinetics with a small sample size. Drug Metab. Drug Interac. 24(2-4), 259–274. Mahmood, I. 2014. Naive pooled-data approach for pharmacokinetic studies in pediatric patients with a very small sample size. Am. J. Therap. 21(4), 269–274. Maris, A.S., Mody, P., Brewer, D.J. and Humphries, R.M. 2021. Fluoroquinolones: an update for clinical microbiologists. Clin. Microbiol. Newsl. 43(12), 97–107. Martinez, M.N., Lindquist, D. and Modric, S. 2010. Terminology challenges: defining modified release dosage forms in veterinary medicine. J. Pharm. Sci. 99(8), 3281–3290. McDowell, A. and Rathbone, M.J. 2014. Veterinary drug delivery. J. Pharm. Bioallied Sci. 6(1), 1. Miranda-Calderón, J.E., Gutiérrez, L., Flores-Alamo, M., García-Gutiérrez, P. and Sumano, H. 2014. Enrofloxacin hydrochloride dihydrate. Acta Cryst. E. 26(70, Pt. 4), 0468–0469. Odom, T.F., Riley, C.B., Benschop, J. and Hill, K.E. 2024. Factors associated with medication nonmedication compliance in dogs in New Zealand. Animals 14(17), 2557. Papich, M.G., Gunnet, L.A. and Lubbers, B.V. 2023. Revision of fluoroquinolone breakpoints used for interpretation of antimicrobial susceptibility testing of canine bacterial isolates. Am. Vet. Res. 84(11), ajvr.23.07.0159. Patra, C.N., Priya, R., Swain, S., Kumar Jena, G., Panigrahi, K.C. and Ghose, D. 2017. Pharmaceutical significance of eudragit: a review. Futur. J. Pharm. Sci. 3(1), 33–45. Sánchez, Z., Melgoza, L.M., Sandoval, H. and Caraballo, I. 2014. Elaboración de differences diseños de spheres de alta vendidad y liberación entérica de L-arginina para uso en ruminates. Tesis, Universidad Autónoma Metropolitana-Xochimilco. Sumano, L.H., Ocampo, C.L. and Gutiérrez, O.L. 2001. Non-bioequivalence of enrofloxacin and Baytril in cows. Dtsch. Tierarztl. Wochenschr. 108(7), 311–314. Sumano, L.H., Ocampo, C.L. and Gutiérrez, O.L. 2003. Bioequivalence of six generic enrofloxacin preparations in pigs. Pig J. , 51. Sumano, L.H.S., Ocampo, C.L., Tapia, G., Mendoza, C.J. and Gutiérrez, O.L. 2018. Pharmacokinetics of enrofloxacin HCl-2H2O (Enro-C) in dogs and pharmacokinetic/pharmacodynamic Monte Carlo simulations against Leptospira spp. Vet. Sci. 19(5), 600–607. Trouchon, T. and Lefebvre, S. 2016. Review of enrofloxacin for veterinary use. Open J. Vet. Med. 6(2), 40–58. Wu, H., Zhang, X., Liu, J., Ma, B., Chen, P., Qin, Q. and Deng, K. 2023. Progress in the preparation of enrofloxacin for use in veterinary medicine. Ziółkowski, H., Jaroszewski, J.J., Maślanka, T., Grabowski, T., Katolik, K., Pawęska, J., Siemianowska, M., Jasiecka, A., Markiewicz, W. and Spodniewska, A. 2015. Influence of oral co-administration of a preparation containing calcium and magnesium and food on enrofloxacin pharmacokinetics. Res. Vet. Sci. 97, 99–104. | ||
| How to Cite this Article |
| Pubmed Style Gutierrez L, Hernández M, Villar JLD, Sánchez Z, Sumano H. Pharmacokinetic evaluation of a long-acting, tasteless oral formulation of enrofloxacin in dogs. Open Vet. J.. 2025; 15(10): 5009-5017. doi:10.5455/OVJ.2025.v15.i10.19 Web Style Gutierrez L, Hernández M, Villar JLD, Sánchez Z, Sumano H. Pharmacokinetic evaluation of a long-acting, tasteless oral formulation of enrofloxacin in dogs. https://www.openveterinaryjournal.com/?mno=267141 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.19 AMA (American Medical Association) Style Gutierrez L, Hernández M, Villar JLD, Sánchez Z, Sumano H. Pharmacokinetic evaluation of a long-acting, tasteless oral formulation of enrofloxacin in dogs. Open Vet. J.. 2025; 15(10): 5009-5017. doi:10.5455/OVJ.2025.v15.i10.19 Vancouver/ICMJE Style Gutierrez L, Hernández M, Villar JLD, Sánchez Z, Sumano H. Pharmacokinetic evaluation of a long-acting, tasteless oral formulation of enrofloxacin in dogs. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5009-5017. doi:10.5455/OVJ.2025.v15.i10.19 Harvard Style Gutierrez, L., Hernández, . M., Villar, . J. L. D., Sánchez, . Z. & Sumano, . H. (2025) Pharmacokinetic evaluation of a long-acting, tasteless oral formulation of enrofloxacin in dogs. Open Vet. J., 15 (10), 5009-5017. doi:10.5455/OVJ.2025.v15.i10.19 Turabian Style Gutierrez, Lilia, Mayte Hernández, Jorge Luna Del Villar, Zacnite Sánchez, and Hector Sumano. 2025. Pharmacokinetic evaluation of a long-acting, tasteless oral formulation of enrofloxacin in dogs. Open Veterinary Journal, 15 (10), 5009-5017. doi:10.5455/OVJ.2025.v15.i10.19 Chicago Style Gutierrez, Lilia, Mayte Hernández, Jorge Luna Del Villar, Zacnite Sánchez, and Hector Sumano. "Pharmacokinetic evaluation of a long-acting, tasteless oral formulation of enrofloxacin in dogs." Open Veterinary Journal 15 (2025), 5009-5017. doi:10.5455/OVJ.2025.v15.i10.19 MLA (The Modern Language Association) Style Gutierrez, Lilia, Mayte Hernández, Jorge Luna Del Villar, Zacnite Sánchez, and Hector Sumano. "Pharmacokinetic evaluation of a long-acting, tasteless oral formulation of enrofloxacin in dogs." Open Veterinary Journal 15.10 (2025), 5009-5017. Print. doi:10.5455/OVJ.2025.v15.i10.19 APA (American Psychological Association) Style Gutierrez, L., Hernández, . M., Villar, . J. L. D., Sánchez, . Z. & Sumano, . H. (2025) Pharmacokinetic evaluation of a long-acting, tasteless oral formulation of enrofloxacin in dogs. Open Veterinary Journal, 15 (10), 5009-5017. doi:10.5455/OVJ.2025.v15.i10.19 |