| Research Article | ||
Open Vet. J.. 2025; 15(10): 5048-5057 Open Veterinary Journal, (2025), Vol. 15(10): 5048-5057 Research Article Spatial patterns and prediction of the temporal spread of cattle theileriosis in Jambi Province, IndonesiaMorsid Andityas1, Dian Meididewi Nuraini2, Dimas Chaerul Ekty Saputra3,4, Yudhi Ratna Nugraheni5, Aan Awaludin6, Yanita Mutiaraning Viastika7, Herlina Yuliani8, Joko Prastowo5 and Sarwo Edy Wibowo7*1Department of Bioresources Technology and Veterinary, Vocational College, Universitas Gadjah Mada, Yogyakarta, Indonesia 2Department of Animal Science, Faculty of Animal Science, Universitas Sebelas Maret, Surakarta, Indonesia 3Informatics Study Program, Telkom University, Surabaya, Indonesia 4Center of Excellence for Motion Technology for Safety Health and Wellness, Research Institute of Sustainable Society, Telkom University, Surabaya, Indonesia 5Department of Parasitology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia 6Animal Science Department, Livestock Production Study Program, Politeknik Negeri Jember, Indonesia 7Department of Animal Health, Faculty of Animal Science, Universitas Jambi, Jambi, Indonesia 8Laboratory of Animal Health and Veterinary Public Health, Jambi, Indonesia *Corresponding Author: Sarwo Edy Wibowo. Department of Animal Health, Faculty of Animal Science, Universitas Jambi, Jambi, Indonesia. Email: sarwoedywibowo [at] unja.ac.id Submitted: 29/06/2025 Revised: 02/09/2025 Accepted: 12/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
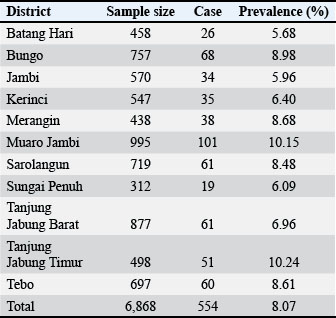
AbstractBackground: Bovine theileriosis is a tick-borne disease that can cause economic losses worldwide, including in tropical countries such as Indonesia. In some regions of Indonesia, cattle are one of the most important commodities that play a role in sustaining economic flow, including in Jambi Province. Aim: This study aimed to determine the prevalence, spatial distribution, and prediction trends of the disease up to 2027 in Jambi Province, Indonesia. Methods: Retrospective data were obtained from active and passive surveillance in Jambi Province and used for calculating the prevalence, with 6,868 cases recorded between 2018 and 2023. Spatial analysis was conducted using the R software (version 4.3.1), while time series forecasting was performed using Python on the Google Colab platform. Results: The overall prevalence during the study period was 8.07% (554/6868, 95% CI, 74.2%–8.71%). Spatial autocorrelation analysis using Moran’s I statistic resulted in a value of 0.17, with a standard deviation of 1.17 and a p-value of 0.12, indicating that the distribution of cases was random across the province. The forecasting analysis suggests that the prevalence is likely to remain stable through 2027, within a projected range of 0%–10%. Conclusion: The current prevalence of bovine theileriosis in Jambi Province is relatively low; however, the potential risk of disease introduction and spread from other regions remains. Preventive and control strategies are essential to safeguard the health of cattle and limit the transmission of bovine theileriosis in Jambi Province. Keywords: Bovine, Theileria, Tick-borne disease, Spatial analysis, Time series forecasting. IntroductionTheileriosis is a tick-borne infectious disease caused by rickettsial agents that is more prevalent in tropical, subtropical, and certain temperate regions. Theileriosis was initially characterized by light microscopy following Koch’s identification of Theileria parva (Koch 1898; Mans et al., 2015). Theileria spp. are obligate intracellular protozoan parasites classified into the order Piroplasmida and phylum Apicomplexa (Riaz et al., 2023). In mammals, theileriosis can cause hematozoa disease (Naila Gul et al., 2015). In addition, the disease is associated with several clinical symptoms, such as fever, swollen lymph nodes, anemia, and dyspnea, and in some cases, caused a high mortality rate (Mohsin et al., 2022). Theileriosis tends to be more dangerous in pregnant cows, especially in late pregnancy, leading to abortion and fetal death (Perera et al., 2014; Chen et al., 2022). Hosts with subclinical infections can be carriers of piroplasms and sources of infection for susceptible animals (Dumanli N Aktas et al., 2005). Thus, theileriosis has been associated with economic losses across various countries, including Indonesia (Ayadi et al., 2016). Several studies have reported that theileriosis in dairy cows can reduce milk volume, milk fat, milk protein, morbidity, mortality, and tick control costs compared to healthy cows (Perera et al., 2014; Rashid et al., 2018). Additionally, the economic impact of theileriosis was reported in Pakistan with an estimated total farm loss of $18,743.76, with a loss of $74.98 per animal on Holstein Friesian dairy farm (Rashid et al., 2018). The distribution of the prevalence of cattle theileriosis worldwide varies widely across regions. The prevalence of cattle theileriosis was reported in Egypt at 16.49% (94/570, 95% Confidence Interval (CI): 13.5%–19.8%) (Theileria annulata) (Selim et al., 2022), Oman at 72.3% (323/447, 95% CI: 68.11%–76.41%) (T. annulata) (Al-Fahdi et al., 2017), Sri Lanka at 55% (33/60, 95% CI: 42.42%–67.58%) (T. orientalis, T. annulata) (Subhasinghe et al., 2024), Pakistan (Multan) at 11.3% (115/1020, 95% CI: 9.33%–13.22%) (T. annulata) (Asif et al., 2022), China at 32.4% (95% CI: 9.33%–13.22%) (Chen et al., 2022), and Saudi Arabia at 1.9% (2/362, 95% CI: 0.00%–1.31%) (T. annulata) (Alanazi et al., 2021). In Jambi Province, Indonesia, cattle theileriosis is an important disease that hinders cattle development. The prevalence of theileriosis in Muara Bulian District, Batang Hari Regency, Jambi Province, in 2018 was reported to be 34% (32/94, 95% CI: 24.5%–43.6%), indicating a high prevalence of theileriosis in the area (Aziz et al., 2019). Similarly, a high prevalence of blood parasite infestation was also reported in neighboring regions, where the Balai Veteriner (BVet) Bukittinggi recorded 98% positive cases from 4,832 cattle blood smears in 2018 (Susanti et al., 2019). Therefore, prevention and control strategies are necessary to minimize the dissemination risk from neighboring regions so that the economic loss will be reduced. However, information related to the epidemiological trends in Jambi Province is limited, which can hinder effective prevention and control measures. This study aimed to analyze the overall prevalence and spatial analysis of theileriosis in cattle from 2018 to 2023. Additionally, this study predicts the prevalence of cattle theileriosis up to 2027. Materials and MethodsStudy location and data sourceThe study was conducted using data from Jambi Province, Indonesia, which is located on Sumatra Island between the latitudes of 0°45′ and 2°45′ N, and longitudes of 101°10′ and 104°55′ E. The data were compiled from retrospective records of bovine theileriosis, collected through active and passive epidemiologic surveys conducted on cattle from January 2018 to December 2023, by the Laboratory of Animal Health and Veterinary Public Health, Jambi Province, Indonesia. A total of 5,612 cattle blood samples collected from 11 districts were tested using blood smear to identify Theileria spp. Theileriosis in cattle can be detected by preparing a thin blood smear from capillary blood, fixing it with methanol, and staining it with 10% Giemsa for approximately 30–45 minutes. Microscopic examination under oil immersion was then performed to identify intraerythrocytic piroplasms characteristic of Theileria spp. These districts include Batang Hari, Bungo, KerinCI: Jambi City, Merangin, Muaro Jambi, Sarolangun, Sungai Penuh, Tanjung Jabung Barat, Tanjung Jabung Timur, and Tebo. Information related to the month and year of sample collection, geographical location, number of samples collected, and number of positive cases identified were compiled into a predesigned Microsoft Excel sheet (Microsoft Corp, Redmond, WA, USA). A base map of Jambi Province from the Indonesian Geospatial portal (https://tanahair.indonesia.go.id/portal-web) was used to visualize the distribution to enhance the analysis of this epidemiological data. Spatial analysisThe choropleth maps and spatial autocorrelation analyses were conducted using the R software, version 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria). Choropleth maps were generated to illustrate the temporal changes in the prevalence rates of theileriosis in different districts in Jambi Province. Historical data spanning several years were collected and analyzed to identify trends and fluctuations in Jambi Province. For the spatial autocorrelation analysis, the prevalence of cattle theileriosis was aggregated at the district level. The aggregation process involved summing the total number of cases of theileriosis and calculating the prevalence rates for each district. These aggregated datasets formed the basis for subsequent statistical analyses of spatial data. Global Moran’s I was calculated to assess the overall spatial autocorrelation of theileriosis prevalence across the study area. This index measures the degree to which similar or dissimilar values spatially cluster. The calculation of Global Moran’s I involved using a spatial weights matrix, which defined the spatial relationships between districts. In global autocorrelation analysis, global Moran’s I was performed to assess the spatial correlation of the whole study area using the following method: (9) where I represents the Global Moran’s Index, n refers to locations, represents the value at location i, represents the value at location j, and represents the mean value of the study area. Additionally, denotes the components within the standardized weight matrix between regions i and j. The Global Moran’s I index value of 0 indicates that the spatial distribution of bovine theileriosis is random. Perfect clustering was indicated by a value of +1, whereas a value of −1 indicates a completely dispersed distribution. The significance of the Global Moran’s I was assessed by comparing the z-score to the standard normal distribution or by using Monte Carlo randomization. It was considered statistically significant (p < 0.05) if the Z-score was greater than 1.96 or less than −1.96 (Wibowo et al., 2024). To calculate the local autocorrelation of specific areas based on theileriosis prevalence in different districts in Jambi Province, the Local Moran’s I was employed with the following equation: (10) where denotes the local Moran I Index was denoted by Ii while Zi and Zj representing the deviations from the mean values. Local Moran’s I results can be interpreted as high-high (HH), low-low (LL), high-low (HL), and low-high (LH). HH clusters indicated areas where high-prevalence districts clustered together, suggesting a significant hot spot. LL clusters represent cold spots where low-prevalence districts are adjacent to other low-prevalence districts. HL and LH outliers indicate districts where high-prevalence areas are adjacent to low-prevalence areas and vice versa (Wibowo et al., 2024). Long short-term memoryThe long short-term memory (LSTM) networks were parts of specific recurrent neural networks that were used to process sequences and time series data efficiently. Therefore, LSTM networks are valuable for predicting and forecasting bovine theileriosis in Jambi Province. LSTMs address the vanishing gradient issue and effectively capture long-term relationships in data. There were three primary gates: the forget, input, and output gates. The forget gate determines which components of the previous cell state should be eliminated, considering both the input and the prior concealed state. The formula for LSTMs was as follows: (1) where ft is the forget gate activation function, σ was the activation function, Wf was the weight matrix for the forget gate, ht−1 was the hidden state from the previous cell, xt was the input at the current time step, and bf was the bias for the forget gate. The input gate determines the specific information stored in the cell state. The calculation was as follows: (2) where it was the input gate activation vector, Wi was the weight matrix for the input gate, and bi was the bias for the input gate. Simultaneously, a candidate cell state was created using the following equation: (3) where tanh was the hyperbolic tangent function, was the candidate cell state, WC was the weight matrix for the candidate cell state, and bC was the bias for the candidate cell state. The updated cell state was obtained by merging the previous cell state, the output of the forget gate, the input gate output, and the candidate cell state according to the following equation: (4) where Ct was the new cell state and Ct−1 was the previous cell state. The output gate determines the subsequent concealed state, which is a refined version of the updated cell state. The calculation was performed as follows: (5) where ot was the output gate activation vector, Wo was the weight matrix for the output gate, and bo was the bias for the output gate. The new hidden state was performed as follows: (6) where ht was the hidden state of the current time step. To predict future values, an LSTM network was trained using previous time series data to understand the patterns and relationships. The process starts with data preparation, where the input data are standardized and organized into sequences. The LSTM model was constructed with many layers of LSTM units, followed by a thick layer. The model training was performed by maximizing a loss function, such as mean squared error (MSE), using backpropagation over time. Mean squared error and root mean squared errorThe Mean Squared Error and Root Mean Squared Error (RMSE) were metrics for assessing the effectiveness of regression models. Measure the discrepancy between observed and projected values and provide valuable information on the forecast precision and dependability of a model. The MSE calculates the average of the squared discrepancies between the actual and anticipated values. The term “quality” refers to a metric that assesses an estimator’s performance and is often used in regression analysis. The MSE was performed as follows: (7) where n was the number of observations, yi was the actual value for the i-th observation, and was the predicted value for the i-th observation. The MSE is a measure that quantifies the average of the squared differences between the actual and anticipated values. By squaring the mistakes, the correctness of the model could be assessed more effectively because bigger errors are given more weightage than smaller ones. The RMSE is the square root of the MSE. The measure offers an indication of the average size of the mistakes using the same units as the target variable. This makes it easier to understand compared with MSE. The RMSE was calculated as follows: (8) The RMSE is the mathematical measure that calculates the square root of the average of the squared discrepancies between the actual and anticipated values. The straightforward interpretation of the average error size in target variable units facilitates comprehension and comparison across various datasets or models. All analysis for the time series forecast model was performed in Python on the Google Colab cloud. Ethical approvalThis study did not directly involve animals in the process and used annual surveillance data from the Laboratory of Animal Health and Veterinary Public Health Jambi Province. Therefore, ethical approval was not required. ResultsThe results revealed that the prevalence of cattle theileriosis in Jambi Province from 2018 to 2023 was 8.07% (554/6868, 95% CI, 74.2%–8.71%) (Fig. 1). Variation in the prevalence was found across districts in Jambi with East Tanjung Jabung showing the highest prevalence rate of 10.24% (51/498, 95% CI: 7.58%–12.90%), followed by Muaro Jambi District 10.15% (101/995, 95% CI: 8.27%–12.03%), Bungo District 8.98% (68/757, 95% CI: 6.95%–11.02%), Merangin District 8.68% (38/438, 95% CI: 6.04%–11.31%), Tebo District 8.61% (60/697, 95% CI: 6.53%–10.69%), Sarolangun District 8.48% (61/719, 95% CI, 6.45%–10.52%), West Tanjung Jabung District 6.96% (61/877, 95% CI, 5.27%–8.64%), Kerinci District 6.40% (35/547, 95% CI, 4.35%–8.45%), Sungai Penuh District 6.09% (19/312, 95% CI, 3.44%–8.74%), Jambi City District 5.96% (34/570, 95% CI, 4.02%–7.91%), and Batang Hari District 5.68% (26/458, 95% CI, 3.56%–7.80%) (Table 1). Between 2018 and 2019, a high prevalence of cattle theileriosis was seen in Tebo District between 2018 and 2019, with rates of 22.90% (30/131, 95% CI: 15.6%–30.2%) and 18.96% (11/58, 95% CI: 8.98%–28.96%), respectively. In 2020, although Jambi City District showed the highest apparent prevalence (25%, 1/4), the small sample size resulted in a wide 95% CI (0.63%–80.59%), making it less reliable; therefore, with adequate sample size, Merangin District 10.26% (4/39, 95% CI: 2.87%–24.23%) was considered the highest prevalence. In 2021, Sungai Penuh District recorded the highest prevalence at 18% (9/50, 95% CI: 8.52%–31.37%), while in 2022, Muaro Jambi District had the highest prevalence at 8.72% (15/172, 95% CI: 4.50%–12.94%). Meanwhile, in 2023, Jambi City District had the highest prevalence at 20.90% (14/67, 95% CI: 11.17%–30.62%). Figure 2 shows the choropleth map illustrating the annual prevalence rates of cattle theileriosis across different districts in Jambi Province.
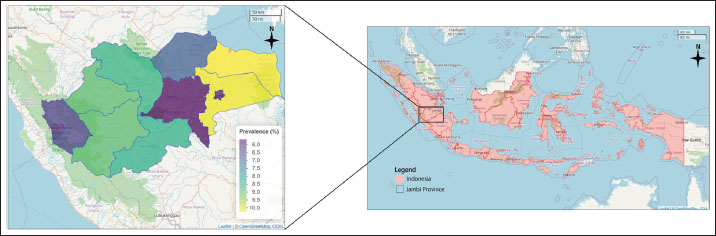
Fig. 1. Choropleth map of the total prevalence of cattle theileriosis from 2018 to 2023 in Jambi Province, Indonesia, from 2018 to 2023. Table 1. Prevalence of cattle theileriosis in each district of Jambi Province (2018–2023).
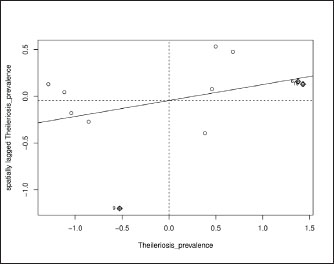
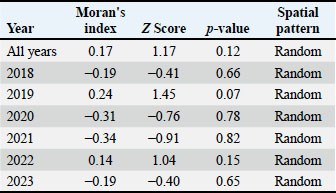
The analysis of overall years showed a Moran’s I statistic of 0.17 with a standard deviation of 1.17 and a p-value of 0.12. This indicated a random distribution in the spatial pattern of bovine theileriosis across the year (2018–2023) as seen in the scatter plot (Fig. 3). Based on the analysis of the individual year, the same patterns of random distribution were found in the spatial pattern of bovine theileriosis (Table 2). The proximity of Moran’s I values to zero further indicated that there was no substantial spatial clustering or dispersion of the disease prevalence across the districts.
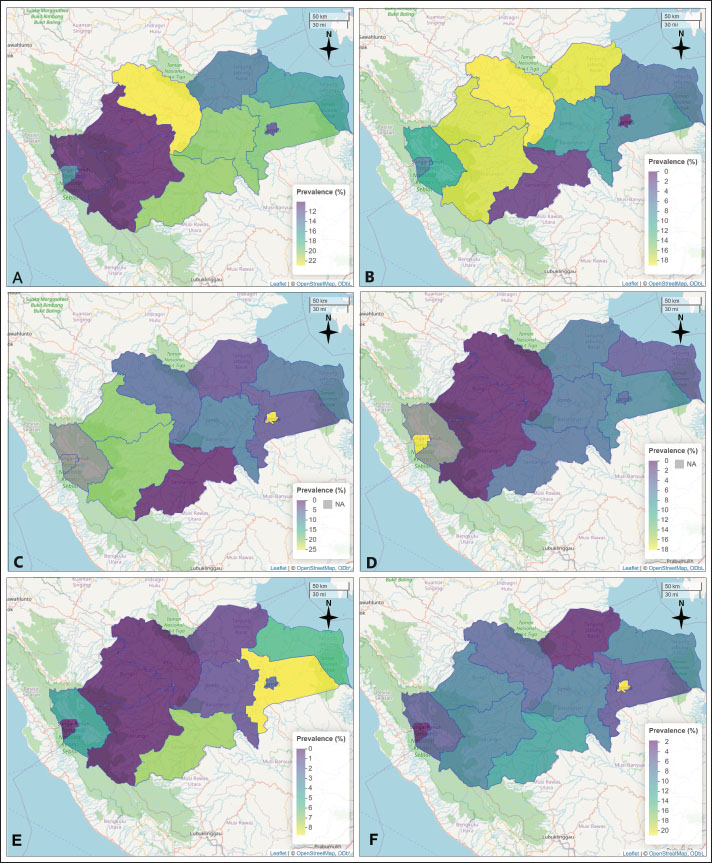
Fig. 2. Choropleth map illustrating the annual prevalence rates of cattle theileriosis across different districts in Jambi Province. The map includes the following: Prevalence for 2018; B. Prevalence for 2019; C. Prevalence for 2020; D. Prevalence for 2021; E. Prevalence for 2022.
Fig. 3. Global Moran’s I scatterplot for the prevalence of cattle theileriosis in Jambi Province, Indonesia. Table 2. Global Moran’s I result for cattle theileriosis prevalence in Jambi Province (2018–2023).
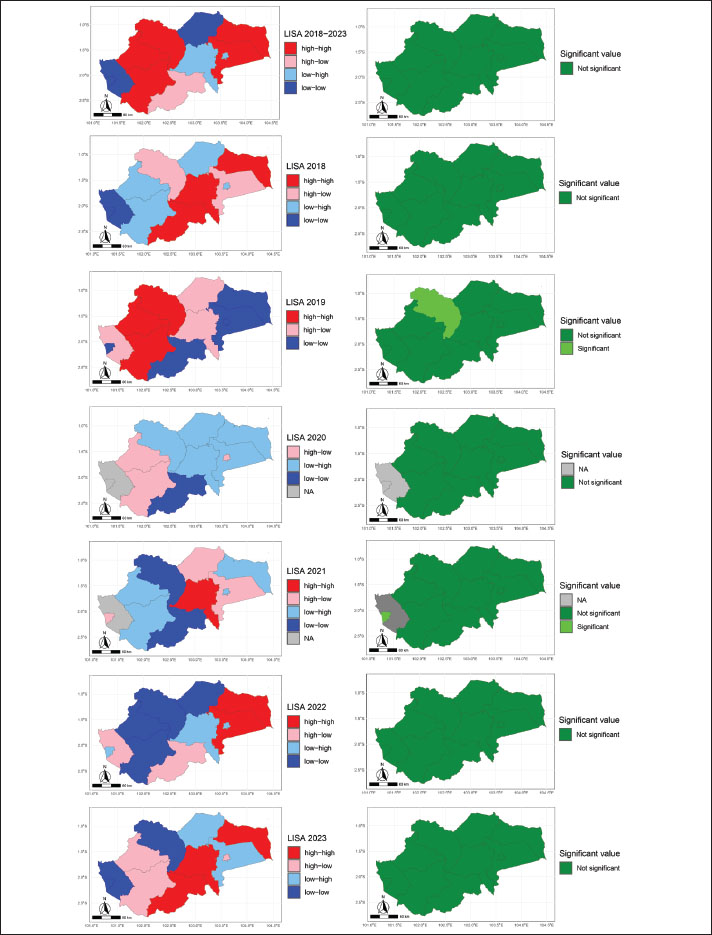
Based on the results of the Local Moran I analysis, there was a variation in the spatial distribution of data, as indicated by the quadrants “high-high,” “high-low,” “low-high,” and “low-low” (Fig. 4). In 2018, the distribution of quadrants was as follows: “high-high” occurred three times, “high-low” two times, “low-high” three times, and “low-low” three times. In 2019, a variation with “high-high” occurred two times, “high-low” four times, “low-high” once, and “low-low” seven times. In 2020, the distribution showed “high-high” and “high-low”, two and three times, respectively, while “low-high” occurred five times and “low-low” two times. 2021 recorded “high-high” once, “high-low” three times, “low-high” four times, and “low-low” twice. In 2022, there were four occurrences of “high-high,” three occurrences of “high-low,” three occurrences of “low-high,” and three occurrences of “low-low.” Finally, in 2023, the distribution of the “high-high” quadrant appeared five times, “high-low” three times, “low-high” two times, and “low-low” three times. These results reflect the variation in the spatial relationships among the observation units each year.
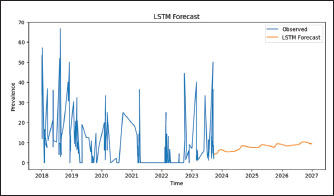
Fig. 4. LISA Moran’s I results of the prevalence of cattle theileriosis from 2018 to 2023, with the left side showing prevalence hotspots and the right side indicating the significance of these hotspots. The Long Short-Term Memory model showed a prediction of cattle Theileriosis in Jambi Province up to 2027 (Fig. 5). The blue line illustrates the observed prevalence data, which displays fluctuations with several peaks and troughs. This variability reflects the unpredictable pattern of the recorded prevalence levels across years. The orange line represents the projection value generated by the LSTM model. The analysis results showed a smoother and more consistent trend over the period (2024–2027) with an estimated bovine theileriosis prevalence forecast ranging from 0% to 10% in Jambi Province. The performance of the LSTM model was measured using two primary metrics: MSE and RMSE. The MSE, with a value of 164.34, quantifies the average of the squared discrepancies between the observed and predicted values. The RMSE, with a value of 12.82, represents the square root of the MSE and provides prediction accuracy in the same units as the original data.
Fig. 5. Prevalence of theileriosis in cattle in Jambi Province forecasts with an LSTM model. DiscussionThis study identified an 8.07% (554/6868, 95% CI, 7.42%–8.71%) prevalence of bovine theileriosis in Jambi Province, Indonesia, from 2018 to 2023, which is lower than the prevalence rates reported in previous studies in Jambi Province and other regions of Indonesia. In the Batang Hari district (Jambi Province), a prevalence of 34% (32/94, 95% CI: 24.5%–43.6%) had been reported from farms (Aziz et al., 2019). Meanwhile, higher prevalence rates were documented in South Kalimantan (30.8%) (Siswansyah, 1990), Tamangapa landfill in Makassar (27.91%, 12/43, 95% CI: 14.49%–41.32%) (Sarah et al., 2020), Kutalimbaru Seb-district, and North Sumatra (33%) (Ritonga et al., 2020). Although the prevalence of bovine theileriosis in Jambi Province is lower than that in other regions, data from BVet Bukittinggi in 2018 showed a high incidence of blood parasite infestation in cattle (98% of 4,832 blood smears) (Susanti et al., 2019), which, despite generally being chronic or mild, can cause anemia and mortality in severe cases, thus requiring comprehensive preventive measures such as vector control, improved nutrition and hygiene, and routine hematological monitoring to guide appropriate treatment. Compared to global reports, the prevalence in Jambi is lower than rates observed in countries such as China, where a pooled prevalence of 32.4% (95% CI: 9.33%–13.22%) has been reported (Chen et al., 2022), and Portugal, where 36.68% (95% CI: 34.3%–39.4%) of piroplasma-positive animals were infected with Theileria spp., including 21.3% (95% CI: 19.3%–23.5%) with T. annulata (Gomes et al., 2013). Additionally, studies in Sri Lanka, China, Sudan, and Iraq reported prevalence rates ranging from 21.13% to 55% (Ahmed et al., 2021; Madkour et al., 2023; Kebzai et al., 2024; Subhasinghe et al., 2024). However, the prevalence in Jambi is higher than that in some regions, such as India, where the prevalence of T. orientalis was reported to be 4.8% (41/862, 95% CI: 3.34%–6.18%) in Telangana and Andhra Pradesh (George et al., 2015). Geographical differences in theileriosis prevalence can be related to several factors, including local environmental factors and tick population density (Al-Fahdi et al., 2017; Selim et al., 2022; Ullah et al., 2022; Hassan et al., 2024). Environmental factors, such as temperature and humidity, directly impact the life cycle and transmission rates of tick vectors, which play a major role in disease dissemination (Nuttall, 2022). In this study, the results of global Moran’s I analysis showed that the spatial distribution of bovine theileriosis prevalence in Jambi Province from 2018 to 2023 was randomly dispersed, suggesting no clear geographic pattern of the disease. However, local Moran’s I analysis indicated significant spatial variation in several areas, including the presence of stable clusters with HH, LL, and HL or LH patterns. These findings highlight that although the overall distribution appears random, local dynamics must be considered when planning disease control interventions. Therefore, if local spatial complexities are overlooked, geographically limited strategies targeting certain regions may be insufficient, and a broader population-based approach may be more effective. Additionally, several factors influence spatial differences in the prevalence of blood parasite disease in animals, including land cover and cattle dispersal patterns, cattle density and landscape features, environmental conditions and anthropogenic factors, and host-related factors such as body condition and species-specific traits (Valkiūnas et al., 2016; Becker et al., 2020; Enslow et al., 2020; Podmokła et al., 2024). In developing countries like Indonesia, limitations in disease monitoring and reporting systems can influence the underreporting or overlooked of theileriosis in cattle, as it is often neglected compared with other diseases. The forecasting results of this study indicated that the prevalence of bovine theileriosis ranges from 0% to 10% until 2027 with a stable pattern. This stability can be attributed to the consistency of disease factors in Jambi province, as observed in previous periods. These factors include the environment, management, and biological aspects, which are relatively the same across the years. The most important factor is Hyalomma tick infestation, which seems to be continuously found in Jambi cattle (Selim et al., 2020; Ullah et al., 2021; Selim et al., 2022). The lack of acaricides used to manage tick populations increases cattle’s vulnerability to infection (Selim et al., 2020; Ullah et al., 2021). Additionally, cattle not treated with acaricides are particularly at risk (Selim et al., 2022; Hassan et al., 2024). The acaricides treatment is mostly important during where the highest number of thicks is found (El Damaty et al., 2022; Mohsin et al., 2022; Selim et al., 2022). Several limitations related to the data were identified in this study. This study focused on analyzing retrospective data on bovine theileriosis in Jambi Province from 2018 to 2023. The data were collected through both active and passive surveillance methods with varying sample sizes. Additionally, no information regarding potential risk factors is available from the regions. It is important to acknowledge the limitations of the lack of data on potential risk factors that could influence disease dynamics. This gap may have hindered a deeper understanding of the epidemiology of bovine theileriosis in the region. However, the results of this study highlight the prevalence, forecasting, and spatial distribution patterns of bovine theileriosis in Jambi Province during the specified timeframe. ConclusionBovine theileriosis in Jambi Province, Indonesia, in 2018–2023 revealed an 8.07% (554/6868, 95% CI, 7.42%–8.71%) prevalence. The global Moran’s I analysis indicated that the spatial autocorrelation of the prevalence of bovine theileriosis had a random pattern during this period. Forecasting for 2027 suggests that the disease is likely to remain stable, potentially influenced by factors observed in previous years that may continue to affect the prevalence. Further research is necessary to identify Theileria spp. species and model risk factors that could help predict the interrelationships influencing bovine theileriosis in Jambi Province. AcknowledgmentsThe authors would like to express their gratitude to The Laboratory of Animal Health and Veterinary Public Health Jambi Province. We also thank Mr. Zurbein for helping with data collection. Conflict of interestAll authors declare no conflicts of interest. FundingThis study received no specific grant. Author’s contributionMorsid Andityas: original draft preparation, conceptualization, methodology, data analysis, visualization, draft review, and editing. Dian Meididewi Nuraini: conceptualization, original draft preparation, review, and editing. Chaerul Ekty Saputra: conceptualization, methodology, data analysis, visualization, draft review and editing, and supervision. Yudhi Ratna Nugraheni: review and editing. Aan Awaludin: review and editing. Herlina Yuliani: sample collection, project administration, review, and editing. Yanita Mutiaraning Viastika: sample collection, project administration, review, and editing. Joko Prastowo: review and editing. Sarwo Edy Wibowo: conceptualization, methodology, project administration, sample collection, and supervision. Data availabilityAll data supporting the findings of this study are available within the manuscript. ReferencesAhmed, R.B., Mahmood, S.L., Kakrash, N.A., Zangana, I.K. and Sheikh, M.O.B. 2021. Conventional and molecular diagnosis of theileriosis (Theileria annulata) in cattle in Sulaimani Province, Northern Iraq. Passer J. Basic Appl. Sci. 3(2), 150–155. Alanazi, A.D., Alouffi, A.S., Alshahrani, M.Y., Alyousif, M.S., Abdullah, H.H.A.M., Allam, A.M., Elsawy, B.S.M., Abdel-Shafy, S., Alsulami, M.N., Khan, A. and Iqbal, F. 2021. A report on tick burden and molecular detection of tick-borne pathogens in cattle blood samples collected from four regions in Saudi Arabia. Ticks Tick. Borne. Dis. 12(3), 101652. Al-Fahdi, A., Alqamashoui, B., Al-Hamidhi, S., Kose, O., Tageldin, M.H., Bobade, P., Johnson, E.H., Hussain, A.R., Karagenc, T., Tait, A., Shiels, B., Bilgic, H.B. and Babiker, H. 2017. Molecular surveillance of Theileria parasites of livestock in Oman. Ticks Borne. Dis. 8(5), 741–748. Asif, M., Said, M.B., Parveen, A., Ejaz, A., Ikram, M., Awais, M.M., Ozubek, S., Aktas, M., Baber, M. and Iqbal, F. 2022. Seasonal survey, risk factor’s analysis and genotyping of Theileria annulata infecting cattle in Punjab province, Pakistan. Acta Trop. 234, 106587. Ayadi, O., Gharbi, M. and Benchikh Elfegoun, M.C. 2016. Milk losses due to bovine tropical theileriosis (Theileria annulata infection) in Algeria. Asian Pacific Trop. Biomed. 6(9), 801–802. Aziz, N., Maksudi, M. and Prakoso, Y.A. 2019. Correlation between hematological profile and theileriosis in Bali cattle from Muara Bulian, Jambi, Indonesia. Vet. World 12(9), 1358–1361. Becker, D.J., Albery, G.F., Kessler, M.K., Lunn, T.J., Falvo, C.A., Czirják, G., Martin, L.B. and Plowright, R.K. 2020. Macroimmunology: the drivers and consequences of spatial patterns in wildlife immune defense. J. Anim. Ecol. 89(4), 972–995. Chen, Y., Chen, Y.Y., Liu, G., Lyu, C., Hu, Y., An, Q., Qiu, H.Y., Zhao, Q. and Wang, C.R. 2022. Prevalence of Theileria in cattle in China: a systematic review and meta-analysis. Microb. Pathog. 162, 105369. Dumanli N Aktas., Cetinkaya, M., Cakmak, A., Koroglu, E., Saki, C.E., Erdogmus, Z., Nalbantoglu, S., Ongor, H., Simşek, S., Karahan, M. and Altay, K. 2005. Prevalence and distribution of tropical theileriosis in eastern Turkey. J. Parasitol. 127(1), 9–15. El Damaty, H.M., Yousef, S.G., El-Balkemy, F.A., Nekouei, O., Mahmmod, Y.S. and Elsohaby, I. 2022. Seroprevalence and risk factors of tropical theileriosis in smallholder asymptomatic large ruminants in Egypt. Front. Vet. Sci. 9, 1004378. Enslow, C., Vallender, R., Rondel, E. and Koper, N. 2020. Host dispersal and landscape conversion are associated with the composition of haemosporidian parasites of the golden-winged warbler. Parasitology 147(1), 96–107. George, N., Bhandari, V., Reddy, D.P. and Sharma, P. 2015. Emergence of new genotype and diversity of Theileria orientalis parasites from bovines in India. Infect. Genet. Evol. 36(1), 27–34. Gomes, J., Soares, R., Santos, M., Santos-Gomes, G., Botelho, A., Amaro, A. and Inácio, J. 2013. Detection of Theileria and Babesia infections among asymptomatic cattle in Portugal. Ticks Tick. Borne. Dis. 4(1–2), 148–151. Hassan, M.R., Abdullah, H.H.A.M., Abdel-Shafy, S., El-Maadawy, R.S., Abd El-gawad, S.M. and Khater, H. 2024. Molecular epidemiology of bovine theileriosis and genetic diversity of Theileria annulata in three governorates of Egypt. J. Parasit. Dis. 48(3), 525–536. Kebzai, F., Ashraf, K., Rehman, M.U., Akbar, H. and Avais, M. 2024. Molecular Analysis and Associated Risk Factors of Theileria annulata in Cattle from Various Zones of Balochistan, Pakistan. Kafkas Univ. Vet. Fak. Derg. 30(1), 15–21. Koch, R. 1898. Reise-Berichte über Rinderpest, Bubonenpest in Indien und Afrika, Tsetse-oder Surrakrankheit, Texasfieber, tropische Malaria, Schwarzwasserfieber. Berlin, Heidelberg, Germany: Springer Berlin Heidelberg, pp: 1–36. Madkour, B.S., Karmi, M., Youssef, M.A., Abdelraouf, A. and Abdel-Rady, A. 2023. Epidemiological and diagnostic investigation on bovine theileriosis in Aswan Governorate, Egypt. J. Parasit. Dis. 47(1), 124–130. Mans, B.J., Pienaar, R. and Latif, A.A. 2015. A review of Theileria diagnostics and epidemiology. Int. J. For Parasitology. Parasites Wildlife 4(1), 104–118. Mohsin, M., Hameed, K., Kamal, M., Ali, A., Rafiq, N., Usman, T., Khan, W., Abbasi, A.A., Khan, R.U. and Yousafzai, G.J. 2022. Prevalence and risk factors assessment of theileriosis in livestock of Malakand Division, Pakistan. Saudi Soc. Agricult. Sci. 21(4), 242–247. Naila Gul, N.G., Sultan Ayaz, S.A., Irum Gul, I.G., Mian Adnan, M.A., Sumaira Shams, S.S. and Noor-ul-Akbar, N.U.A. 2015. Tropical theileriosis and East coast fever in cattle: present, past and future perspective. Int. J. Curr. Microbiol. Appl. SCI. 4(8), 1000–1018. Nuttall, P.A. 2022. Climate change impacts on ticks and tick-borne infections. Biol 77(6), 1503–1512. Perera, P.K., Gasser, R.B., Firestone, S.M., Anderson, G.A., Malmo, J., Davis, G., Beggs, D.S. and Jabbar, A. 2014. Oriental theileriosis in dairy cows causes a significant milk production loss. Parasit. Vectors 7(1), 1–8. Podmokła, E., Dubiec, A., Pluciński, B., Zając, B. and Gustafsson, L. 2024. Haemoparasite infection risk in multi-host avian system: an integrated analysis. Parasitology 151(1), 1242–1253. Rashid, M., Akbar, H., Rashid, I., Saeed, K., Ahmad, L., Ahmad, A.S., Shehzad, W., Islam, S. and Farooqi, S. 2018. Economic Significance of Tropical Theileriosis on a Holstein Friesian Dairy Farm in Pakistan. J. Parasitol. 104(3), 310–312. Riaz, M., Nasreen, N., Khan, A. and Said, M.B. 2023. Differential diagnosis of theileriosis through blood smear examination and polymerase chain reaction in small ruminants from Pakistan. Open Vet. J. 13(6), 697–704. Ritonga, M.Z., Putra, A., Prastia, A., Nasution, F. and Br Ginting, R. 2020. Detection Of Blood Parasites In Cattle In Kutalimbaru Subdistrict, Deli Serdang Regency, North Sumatera. In E3S Web of Conferences, EDP Sciences, Les Ulis Cedex, France, 151, pp 2019–2021. Sarah, R.A., Mustakdir, Z., Ismail, I., Mursalim, F.M., Kholilullah, Z.A., Rell, F., Ris, A., Nur, R., Jamaluddin, A.W., Suharto, R.H., Wahyuda, A.A.P.J. and Yusuf, B. 2020. Detection of Theileria sp. in Grazing Cattle at Tamangapa Landfill Makassar. JRVI 7(2), 55–61. Selim, A., Weir, W. and Khater, H. 2022. Prevalence and risk factors associated with tropical theileriosis in Egyptian dairy cattle. Vet. World 15(4), 919–924. Selim, A.M., Das, M., Senapati, S.K., Jena, G.R., Mishra, C., Mohanty, B., Panda, S.K. and Patra, R.C. 2020. Molecular epidemiology, risk factors and hematological evaluation of asymptomatic Theileria annulata infected cattle in Odisha, India. Iran. J. Vet. Res. 21(4), 250–256. Siswansyah, D.D. 1990. Prevalence of theileriosis, babesiosis and anaplasmosis in cattle and buffaloes in South Kalimantan. Penyakit Hewan 39, 50–54. Subhasinghe, J., Mahakapuge, T.A.N., Madusanka, K.S., Rajakaruna, R.S., Jabbar, A. and Perera, P.K. 2024. First report of oriental theileriosis in the Intermediate Zone, Sri Lanka: is Theileria orientalis type 7 always apathogenic?. Vet. Parasitol. Reg. Stud. Rep. 55, 101115. Susanti, T., Hartini, R. and Santosa, B. 2019. Parasit Darah dan Profil Hematologinya Secara Kwalitatif pada Sapi di Wilayah Reginal BVet Bukittinggi Tahun 2018. Buletin Informasi Kesehatan Hewan 21(98), 29–34. Ullah, K., Numan, M., Alouffi, A., Almutairi, M.M., Zahid, H., Khan, M., Islam, Z.U., Kamil, A., Safi, S.Z., Ahmed, H., Tanaka, T. and Ali, A. 2022. Molecular Characterization and Assessment of Risk Factors Associated with Theileria annulata Infection. Microorganisms 10(8), 1–11. Ullah, R., Shams, S., Khan, M.A., Ayaz, S., Akbar, N.U., Din, Q.U., Khan, A., Leon, R. and Zeb, J. 2021. Epidemiology and molecular characterization of Theileria annulata in cattle from central Khyber Pakhtunkhwa, Pakistan. PLos One 16(1), 1–17. Valkiūnas, G., Iezhova, T.A. and Sehgal, R.N.M. 2016. Deforestation does not affect the prevalence of a common trypanosome in African birds. Acta Trop. 162, 222–228. Wibowo, S.E., Andityas, M., Nuraini, D.M.Z., Nugraheni, Y.R., Awaludin, A., Rahayu, P. and Insulistyowati, A. 2024. Spatial analysis of bovine anaplasmosis in Jambi Province, Indonesia: 2018-2022. Vet. Integr. Sci. 22(3), 857–869. | ||
| How to Cite this Article |
| Pubmed Style Andityas M, Nuraini DM, Saputra DCE, Nugraheni YR, Awaludin A, Viastika YM, Yuliani H, Prastowo J, Wibowo SE. Spatial patterns and prediction of the temporal spread of cattle theileriosis in Jambi Province, Indonesia. Open Vet. J.. 2025; 15(10): 5048-5057. doi:10.5455/OVJ.2025.v15.i10.23 Web Style Andityas M, Nuraini DM, Saputra DCE, Nugraheni YR, Awaludin A, Viastika YM, Yuliani H, Prastowo J, Wibowo SE. Spatial patterns and prediction of the temporal spread of cattle theileriosis in Jambi Province, Indonesia. https://www.openveterinaryjournal.com/?mno=267562 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.23 AMA (American Medical Association) Style Andityas M, Nuraini DM, Saputra DCE, Nugraheni YR, Awaludin A, Viastika YM, Yuliani H, Prastowo J, Wibowo SE. Spatial patterns and prediction of the temporal spread of cattle theileriosis in Jambi Province, Indonesia. Open Vet. J.. 2025; 15(10): 5048-5057. doi:10.5455/OVJ.2025.v15.i10.23 Vancouver/ICMJE Style Andityas M, Nuraini DM, Saputra DCE, Nugraheni YR, Awaludin A, Viastika YM, Yuliani H, Prastowo J, Wibowo SE. Spatial patterns and prediction of the temporal spread of cattle theileriosis in Jambi Province, Indonesia. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5048-5057. doi:10.5455/OVJ.2025.v15.i10.23 Harvard Style Andityas, M., Nuraini, . D. M., Saputra, . D. C. E., Nugraheni, . Y. R., Awaludin, . A., Viastika, . Y. M., Yuliani, . H., Prastowo, . J. & Wibowo, . S. E. (2025) Spatial patterns and prediction of the temporal spread of cattle theileriosis in Jambi Province, Indonesia. Open Vet. J., 15 (10), 5048-5057. doi:10.5455/OVJ.2025.v15.i10.23 Turabian Style Andityas, Morsid, Dian Meididewi Nuraini, Dimas Chaerul Ekty Saputra, Yudhi Ratna Nugraheni, Aan Awaludin, Yanita Mutiaraning Viastika, Herlina Yuliani, Joko Prastowo, and Sarwo Edy Wibowo. 2025. Spatial patterns and prediction of the temporal spread of cattle theileriosis in Jambi Province, Indonesia. Open Veterinary Journal, 15 (10), 5048-5057. doi:10.5455/OVJ.2025.v15.i10.23 Chicago Style Andityas, Morsid, Dian Meididewi Nuraini, Dimas Chaerul Ekty Saputra, Yudhi Ratna Nugraheni, Aan Awaludin, Yanita Mutiaraning Viastika, Herlina Yuliani, Joko Prastowo, and Sarwo Edy Wibowo. "Spatial patterns and prediction of the temporal spread of cattle theileriosis in Jambi Province, Indonesia." Open Veterinary Journal 15 (2025), 5048-5057. doi:10.5455/OVJ.2025.v15.i10.23 MLA (The Modern Language Association) Style Andityas, Morsid, Dian Meididewi Nuraini, Dimas Chaerul Ekty Saputra, Yudhi Ratna Nugraheni, Aan Awaludin, Yanita Mutiaraning Viastika, Herlina Yuliani, Joko Prastowo, and Sarwo Edy Wibowo. "Spatial patterns and prediction of the temporal spread of cattle theileriosis in Jambi Province, Indonesia." Open Veterinary Journal 15.10 (2025), 5048-5057. Print. doi:10.5455/OVJ.2025.v15.i10.23 APA (American Psychological Association) Style Andityas, M., Nuraini, . D. M., Saputra, . D. C. E., Nugraheni, . Y. R., Awaludin, . A., Viastika, . Y. M., Yuliani, . H., Prastowo, . J. & Wibowo, . S. E. (2025) Spatial patterns and prediction of the temporal spread of cattle theileriosis in Jambi Province, Indonesia. Open Veterinary Journal, 15 (10), 5048-5057. doi:10.5455/OVJ.2025.v15.i10.23 |