| Review Article | ||
Open Vet. J.. 2025; 15(10): 4797-4813 Open Veterinary Journal, (2025), Vol. 15(10): 4797-4813 Research Article Marek’s disease: A global challenge to poultry health and productivitySri Mulyati1*, Imam Mustofa1, Aswin Rafif Khairullah2, Muhammad Khaliim Jati Kusala2, Ima Fauziah2, Riza Zainuddin Ahmad2, Fitrine Ekawasti2, Sri Suryatmiati Prihandani2, Abdul Hadi Furqoni3, Bantari Wisynu Kusuma Wardhani4, Dea Anita Ariani Kurniasih5, Andi Thafida Khalisa6, Ikechukwu Benjamin Moses7, Adeyinka Oye Akintunde8, Bima Putra Pratama9, Prima Puji Raharjo10, Kartika Afrida Fauzia11 and Syahputra Wibowo121Division of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 2Research Center for Veterinary Science, National Research and Innovation Agency (BRIN), Bogor, Indonesia 3Center for Biomedical Research, National Research and Innovation Agency (BRIN), Bogor, Indonesia 4Research Center for Pharmaceutical Ingredients and Traditional Medicine, National Research and Innovation Agency (BRIN), Bogor, Indonesia 5Research Center for Public Health and Nutrition, National Research and Innovation Agency (BRIN), Bogor, Indonesia 6Faculty of Military Pharmacy, Universitas Pertahanan, Bogor, Indonesia 7Department of Applied Microbiology, Faculty of Science, Ebonyi State University, Abakaliki, Nigeria 8Department of Agriculture and Industrial Technology, Babcock University, Ilishan Remo, Nigeria 9Research Center for Agroindustry, National Research and Innovation Agency (BRIN), South Tangerang, Indonesia 10Balai Besar Pelatihan Peternakan (BBPP) Batu, Kementerian Pertanian, Malang, Indonesia 11Research Center for Preclinical and Clinical Medicine, National Research and Innovation Agency (BRIN), Bogor, Indonesia 12Eijkman Research Center for Molecular Biology, National Research and Innovation Agency (BRIN), Bogor, Indonesia *Corresponding Author: Sri Mulyati. Division of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: sri-m [at] fkh.unair.ac.id Submitted: 30/06/2025 Revised: 01/09/2025 Accepted: 10/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
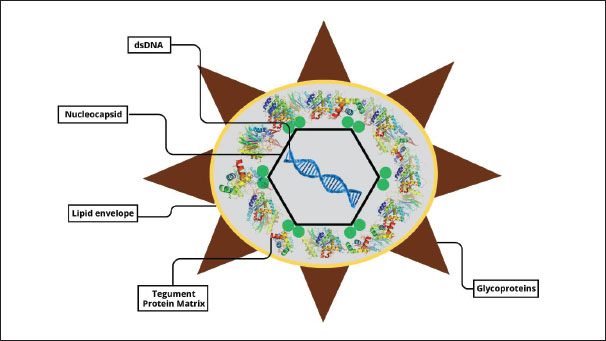
AbstractMarek’s disease is a neuropathic and lymphoproliferative viral condition that primarily affects chickens and occasionally affects quail and turkeys. It is caused by the Marek’s disease virus (MDV), a member of the family Herpesviridae, subfamily Alphaherpesvirinae, genus Mardivirus, which comprises three distinct species: MDV-1, MDV-2, and MDV-3 (herpesvirus of turkeys). Marek’s disease remains a significant global challenge despite the availability of vaccines, with outbreaks still occurring in many poultry-producing regions due to the evolving virulence of the virus and incomplete protection. The current literature provides abundant information on the etiology, pathogenesis, and control of Marek’s Disease; however, an up-to-date, comprehensive synthesis that integrates recent molecular insights, epidemiological patterns, and advanced control strategies is lacking. This review addresses this gap by systematically compiling and analyzing recent studies published in peer-reviewed journals, selected through database searches in PubMed, Scopus, and Web of Science using predefined inclusion and exclusion criteria. Key findings highlight the complexity of MDV pathogenesis, virus persistence in the poultry environment, and limitations of existing vaccines in achieving sterile immunity. The review also underscores the importance of combining vaccination with genetic selection and strict biosecurity to mitigate economic losses. Understanding these interconnected factors is crucial for guiding future research and improving disease management strategies in the poultry industry. Keywords: Marek’s disease Virus, biosecurity measures, Poultry health, Viral pathogenesis. IntroductionMarek’s disease is a neuropathic and lymphoproliferative condition that is frequently found in chickens and sporadically observed in quail and turkeys (Boodhoo et al., 2016). It is caused by the Marek’s disease virus (MDV), a member of the family Herpesviridae, subfamily Alphaherpesvirinae, genus Mardivirus (Žlabravec et al., 2024), comprising three distinct species: MDV-1 (Gallid alphaherpesvirus 2, serotype 1), MDV-2 (Gallid alphaherpesvirus 3, serotype 2), and MDV-3 (Meleagrid alphaherpesvirus 1, herpesvirus of turkeys, serotype 3) (Abayli et al., 2021). MDV-1 is the most virulent and infects chickens, whereas MDV-2 and MDV-3 are typically nonpathogenic and are used as vaccine strains (Ravikumar et al., 2022). MDV is cell-associated, unable to survive outside host cells except in feather follicle epithelial cells, where it can remain unattached (Chakraborty et al., 2022). The disease was first described in 1907 by Hungarian veterinarian József Marek, who identified it as polyneuritis causing paralysis in chickens (McPherson and Delany, 2016). With the advancement of virus isolation and analytical techniques in 1967, MDV was confirmed as the causative agent (Zhang et al., 2015). Before vaccination campaigns, Marek’s disease caused significant global economic losses—estimated at $150 million annually in the United States alone (Kennedy et al., 2017). The World Organization for Animal Health (WOAH) recently added it to the list of animal diseases of concern (WOAH, 2023). Pathogenesis begins with respiratory tract epithelial cell infection, followed by viremia, enabling the virus to spread to lymphoid cells in multiple organs (Trapp-Fragnet et al., 2021). Clinical signs include peripheral nerve hypertrophy, visceral tumors, and asymmetric paralysis (Abreu et al., 2016). The diagnosis combines clinical observation, postmortem examination, and laboratory confirmation (Singh et al., 2012). Transmission occurs via airborne particles, contaminated dust, and indirect contact through equipment, clothing, or vehicles (Woźniakowski and Samorek-Salamonowicz, 2014). Marek’s disease has major economic consequences for poultry operations, including reduced meat quality, poor feed conversion, decreased egg production, and increased susceptibility to secondary infections (Kennedy et al., 2018). The disease increases mortality, morbidity, and overall flock health management costs (Mpenda et al., 2019). The objective of this review is to provide a comprehensive, up-to-date synthesis of Marek’s Disease, including its etiology, molecular pathogenesis, epidemiology, diagnosis, control measures, and future research directions. The novelty lies in integrating recent advances in molecular biology, viral evolution, and vaccine efficacy data—particularly focusing on the emergence of very virulent plus (vv+) MDV strains and their implications for global poultry health. This review also bridges basic pathogenesis insights with applied disease control strategies, which is lacking in previous reviews. Relevant literature was identified through systematic searches in PubMed, Scopus, and Web of Science databases using combinations of keywords such as “Marek’s disease,” “Marek’s disease virus,” “pathogenesis,” “vaccine,” “epidemiology,” and “control strategies.” The inclusion criteria were peer-reviewed articles, reviews, and reports published in English between 2000 and 2025, focusing on avian species, particularly chickens. Reference lists of selected papers were also screened to identify additional relevant studies. EtiologyAccording to the most recent classification by the International Committee on Taxonomy of Viruses, MDV, the causative agent of MDV, is classified as Gallid alphaherpesvirus 2, belonging to the genus Mardivirus, family Herpesviridae, subfamily Alphaherpesvirinae, and order Herpesvirales (Davison, 2010). MDV possesses a large, double-stranded Deoxyribonucleic Acid (DNA) genome organized into a unique long (UL) region and a unique short (US) region, each flanked by terminal repeat (TR) and internal repeat (IR) sequences. The genome encodes more than 100 proteins involved in viral replication, latency, and pathogenesis. Infectious particles comprise a central icosahedral capsid enclosing the viral DNA, a proteinaceous tegument containing more than 15 proteins, and a lipid envelope embedded with approximately 10 glycoproteins that play critical roles in host cell attachment and entry (Rixon, 1993; Emad et al., 2024). Figure 1 illustrates the structural complexity of MDV, which schematically represents its key morphological components.
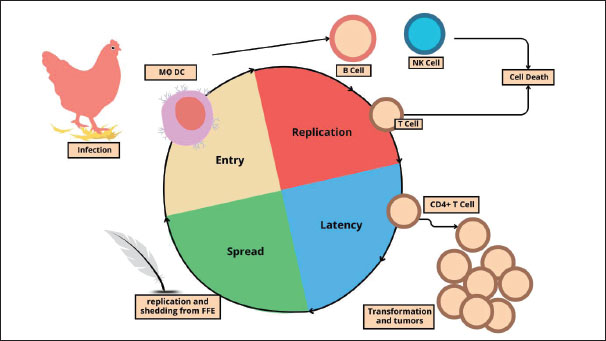
Fig. 1. Schematic representation of MDV morphology. The genus Mardivirus comprises three recognized species: MDV-1 (Gallid herpesvirus 2, serotype 1), MDV-2 (Gallid herpesvirus 3, serotype 2), and MDV-3 (Meleagrid herpesvirus 1, commonly known as Turkey herpesvirus or HVT, serotype 3). Although these species are serologically related, they differ markedly in virulence, host range, and vaccine application. MDV-1 includes all virulent and oncogenic strains, ranging from mild (mMDV) to virulent (vMDV), highly virulent (vvMDV), and very virulent plus (vv+MDV) pathotypes, and is the primary causative agent of Marek’s disease in chickens (Ongor et al., 2022; Liu et al., 2023). MDV-2 consists of naturally occurring, non-oncogenic strains that do not cause disease but are used in bivalent vaccines, often combined with HVT, to enhance protection (Bell et al., 2019). MDV-3 or HVT, a non-oncogenic virus naturally found in turkeys, is widely used as a heterologous vaccine against MDV-1 and is known for its safety and effectiveness in poultry immunization (McPherson and Delany, 2016). Over the past five decades, MDV-1 has shown a clear evolutionary trend toward increasing virulence, with shifts from mMDV to vMDV in the late 1950s, vMDV to vvMDV in the late 1970s, and vv+MDV emergence in the early 1990s (Davison and Nair, 2005). These changes have necessitated the continuous development of more potent vaccines to maintain effective disease control in poultry populations. Virus life cycleCalnek (2001) proposed a widely referenced model of the MDV life cycle, outlining the key phases of viral infection and transmission. This model describes the progression from initial infection to the development of an infectious virus in the epithelial cells of feather follicles. The four main interconnected stages of the MDV life cycle are as follows: (i) entry, (ii) replication, (iii) latency, and (iv) dissemination. Inhalation of dust containing infectious virus particles is the first step in contracting MDV (Kennedy et al., 2017). Early viral replication has been observed in the lungs of B cells and macrophages of infected animals (Bertzbach et al., 2020). Phagocytic cells, including macrophages, transport the virus to the spleen, bursa of Fabricius, and regional lymphatic tissue, where it infects additional immune cells (Davison and Nair, 2005). Both in vitro studies and infections in chickens have demonstrated that macrophages and DCs facilitate MDV replication and cell-to-cell transmission (Chakraborty et al., 2017). MDV secretes a viral CXC chemokine that was formerly known as vIL-8 but was more recently renamed vCXCL13 due to its biological characteristics (Engel et al., 2012). These chemokines are crucial for the normal infection establishment pathway because they attract B cells and some CD4+ T cells (Hughes and Nibbs, 2018). It is only found between days 4 and 10 post-infection, and the overall percentage of lytically infected cells in the primary lymphoid organs of infected hens is often rather low (1.2%) (Berthault et al., 2018). Virus replication occurs in multiple cell types during chicken infection, with B cells accounting for most infected cells (Bertzbach et al., 2020). Infected B cells are the cells most vulnerable to lytic replication and are easily identified in infected chickens (Schat, 2022). MDV produces a viral CXC chemokine, originally designated vIL-8 and more recently renamed vCXCL13 based on its biological properties (Engel et al., 2012). This chemokine plays a pivotal role in the early stages of infection by recruiting B lymphocytes and subsets of CD4+ T cells to sites of viral activity (Hughes and Nibbs, 2018). vCXCL13 expression is transient, typically detectable only between 4 and 10 days post-infection, during which the proportion of lytically infected cells in primary lymphoid organs rarely exceeds 1.2% (Berthault et al., 2018). Viral replication occurs in various cell types within the host, but B lymphocytes are the predominant and most susceptible population for lytic replication, making them readily identifiable in infected chickens (Bertzbach et al., 2020; Schat, 2022). Although B cells are important for early stages of MDV replication, recent studies indicate that they are not essential for subsequent tumor formation or later stages of pathogenesis (Yang et al., 2020). MDV can still efficiently replicate in CD4+ and CD8+ T cells in the absence of B cells (Bertzbach et al., 2018a). Emerging evidence suggests that the virus may replicate in other lymphocyte subsets, such as natural killer (NK) cells, which are part of the innate immune system and are known to produce IFN as an antiviral response (Bertzbach et al., 2019). However, the role of NK cells in MDV pathogenesis remains under investigation, and current data should be interpreted cautiously. Interferon production by NK cells and other immune cells inhibits MDV replication and reduces disease progression (Kamble et al., 2023). Primary chicken endothelial cells have also been identified as potential targets for MDV infection (Hagag et al., 2020). Recent findings challenge the earlier view that MDV infection follows a strictly sequential process, significantly expanding our understanding of the cell tropism of the virus (Vychodil et al., 2022). In addition to replicating, MDV causes T cells to go into latency (Arumugaswami et al., 2009). Lethal lymphoma results from the conversion of only a small number of latently infected cells (Mallet et al., 2022). MDV incorporates its viral genome into the telomeres of host chromosomes in tumor cells and latently infected cells (You et al., 2021a). This integration is necessary for T-cell transformation and guarantees the preservation of the viral genome and its oncogenes (Tien et al., 2023). Telomere integration is facilitated by the placement of telomeric repeats at the ends of the viral genome, most likely via the homologous recombination route (Kheimar et al., 2017). The quick development of T-cell lymphomas is a hallmark of MDV infection (Osterrieder et al., 2006). T cells (>60%) make up the majority of MDV-induced malignancies, with the majority of these cells being transformed and clonally amplified CD4+ T cells (Zhou et al., 2019). MDV can reactivate tumor cells and latently infected cells, enabling ongoing virus shedding and transmission to healthy individuals (McPherson and Delany, 2016). The epithelial cells of the hair follicles shed hair, releasing dust and tiny hairs into the surrounding air that might spread illness (Couteaudier and Denesvre, 2014). The virus starts to spread 12–14 days after infection; however, viral DNA can be found 5–7 days earlier (Liao et al., 2021). The infectious virus is believed to be encased in keratin or exocytosed, and it spreads horizontally to uninfected chickens for 16–28 weeks. Figure 2 depicts the life cycle of MDV, showing how the virus enters the host, replicates in lymphoid organs, establishes latency in T cells, and is eventually shed through the feather follicles. This process highlights the virus’s ability to target the immune system, persist within the host, and efficiently spread among chickens, contributing to disease progression and transmission within flocks.
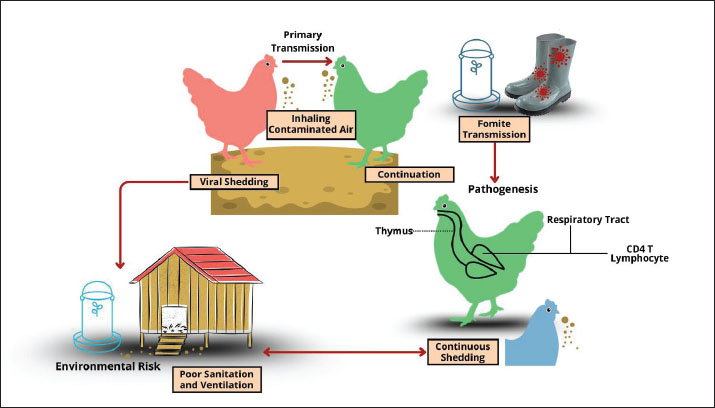
Fig. 2. The pathogenesis cycle of MDV. Virulence factorsThe MDV double-stranded DNA genome is approximately 180 kb long and is made up of internal repeats (Internal Repeat Long and Internal Repeat Short) and terminal repeats (Terminal Repeat Long and Terminal Repeat Short) on either side of the short and long unique regions (US and UL) (Fukuchi et al., 1985). MDV encodes approximately 100 genes involved in different stages of the viral life cycle (You et al., 2021b). These include several virulence factors that promote pathogenesis and tumor development either alone or in combination (Conradie et al., 2020). According to a recent analysis, these variables include the oncoprotein Meq, the viral chemokines vIL-8/vCXCL13, RLORF4, RLORF5a, pp14, and pp38 (Bertzbach et al., 2018b). The majority of earlier studies have concentrated on the function of the virus’s genes that code for proteins (Jarosinski and Osterrieder, 2012; Tai et al., 2017; Trimpert et al., 2017). However, MDV also encodes a rich repertoire of non-coding RNAs (ncRNAs), including miRNAs and viral telomerase RNA (vTR) (Chbab et al., 2010). vTR and its cellular counterpart in chicken have a conserved stem-loop structure and 88% sequence similarity (Fragnet et al., 2003). Furthermore, effective virus-induced tumor growth depends on vTR (Chbab et al., 2010). Inserting chicken TR into the viral genome can restore tumorigenesis in vTR-deficient viruses (Liao et al., 2021). These findings demonstrate that the virus most likely obtained the gene from its host and that cellular TR overexpression can promote tumor development. Two Epstein–Barr virus ncRNAs that share highly conserved cellular contact partners with humans and chickens can likewise partially or fully encode vTR (Kheimar and Kaufer, 2018). Future studies should clarify how vTR and its cellular counterparts change in natural models of this virus-induced tumor development. Additionally, MDV encodes miRNAs that are crucial for latency and carcinogenesis (Liao et al., 2020). To date, 26 mature miRNAs have been identified from 14 miRNA precursors. The MDV repeat region has three clusters of these miRNAs, which are referred to as the Meq-, Mid-, and LAT-clusters (Zhao et al., 2015). They play a role in controlling target genes in cells and viruses. The development and maintenance of latency are facilitated by immediate early gene suppression (Morgan et al., 2008). MDV-miR-M4 is one of the most highly expressed MDV miRNAs and shares the same seed region as the human miRNA miR-155 (Zhang et al., 2019). Like human miR-155, MDV-miR-M4 contributes significantly to tumor development but is not necessary to preserve the altered phenotype or sustain tumor cell growth (Ding et al., 2020). More coding and noncoding sequences that may be crucial for pathogenesis, infection, replication, and dissemination have been identified (Bertzbach et al., 2020). For instance, it identified numerous novel splice variants, including those of known genes such as pp38 (MDV073), UL15 (MDV027), UL49 (MDV062), and pp24 (MDV008) (Volkening et al., 2023). Moreover, polycistronic transcripts, polyadenylation signals, and new poly(A) cleavage sites have been discovered by RNA sequencing (Neve et al., 2017). However, its precise role and significance to the virus life cycle remain unknown. Host rangeBecause chicken breeds vary greatly in their genetic vulnerability, they are by far the most significant natural host (Trapp-Fragnet et al., 2021). MD outbreaks in Japanese quail occur naturally (Adedeji et al., 2019). Turkeys develop tumors on occasion, but a serious outbreak was recently documented in France (Coudert et al., 1995). Pheasants have also been reported to have MD (Chacón et al., 2024). Chronological history of MDThe first documented description of MD was published in 1907 by Dr. József Marek, a renowned pathologist and veterinarian who served as head of the veterinary medicine department at the Royal Hungarian Veterinary School in Budapest. He described four adult male chickens exhibiting paralysis of the legs and wings, a condition he termed “Multiple Nerve Disease (Polyneuritis) in the Wings” (Marek 1907). A postmortem examination revealed thickening of the sacral plexus and spinal pathways with mononuclear cell infiltration. Marek referred to the illness as “interstitial neuritis” or “polyneuritis,” which was later recognized as the first recorded MD case. In the early 20th century, Van der Walle and Winkler-Junius (1924) in the Kaupp (1921) in the USA reported similar conditions. These early accounts suggested that pathological changes were confined to the Central Nervous System and Peripheral Nervous System. At that time, poultry producers referred to the disease as “range paralysis” or “fowl paralysis.” In 1967, Churchill and Biggs (1967) successfully isolated and identified the causative agent—a DNA herpesvirus belonging to the subfamily Alphaherpesvirinae—which was subsequently named MDV. This discovery shifted the understanding of MD from a purely neurological condition to lymphoid neoplasia associated with viral infection. Subsequent immunological and molecular studies identified three serotypes: Serotype 1 (pathogenic and oncogenic), Serotype 2 (non-oncogenic, used for vaccines), and Serotype 3 (turkey herpesvirus, also used as a heterologous vaccine) (Yang et al., 2020). Following the introduction of the first vaccine in the 1970s, the pathogenesis pattern of MDV evolved from a relatively mild classical form to more virulent forms, including very virulent (vvMDV) and very virulent plus (vv+MDV) strains (Zeghdoudi et al., 2023). EpidemiologyMultiple outbreaks of MD have been reported globally in recent years, indicating that the virus continues to circulate and evolve despite vaccination efforts. For example, between 2018 and 2024, sporadic cases and localized outbreaks were recorded in Asia, Africa, and parts of Europe, with several reports describing the emergence of more virulent MDV pathotypes (vvMDV and vv+MDV). Recent surveillance data suggest that MDV circulation in Southeast Asia, parts of the Middle East, and sub-Saharan Africa remains a hotspot, often linked to suboptimal vaccine coverage and biosecurity measures. MD is a disease with a worldwide spread, occurring in practically every nation with a large chicken population (Atkins et al., 2013). MDV infection and transmission are more likely to occur in areas with high poultry populations and intensive rearing practices (Kennedy et al., 2018). This is because the virus can linger in the environment for weeks or even months, especially when it comes to dust particles from skin epithelium and hair follicles, which can spread the infection (Couteaudier and Denesvre, 2014). MD has been reported in several geographical locations, including temperate, tropical, and subtropical climates (Dunn and Gimeno, 2013). MD is intimately linked to the evolution of contemporary poultry technology, the movement of chickens and their products, and the pattern of globalization and intensification of the poultry sector, which facilitates the efficient and quick spread of viruses from one area to another (Nair and V, 2005). Currently, MD is classified as an endemic disease that is present in nations in Asia (Song et al., 2022), Africa (Adedeji et al., 2019), Latin America (Buscaglia et al., 2004), Europe (Biggs and Nair, 2012), North America (Dunn and Gimeno, 2013), and Australia (Renz et al., 2012). Currently, MD is prevalent in the majority of Southeast Asian nations, including Thailand (Wannaratana et al., 2022), Malaysia (Othman and Aklilu, 2019), Indonesia (Hartawan and Dharmayanti, 2016), and Vietnam (Viet Thu et al., 2022). Clinical cases are still being reported, particularly on farms with subpar vaccination programs. There have also been reports of the disease in Middle Eastern countries such as Saudi Arabia (Mohamed et al., 2016), Iran (Ghalyanchilangeroudi et al., 2022), and Turkey (Hauck et al., 2020), along with difficulties in managing increasingly severe strains. In Africa, the disease is still widespread in nations such as Ethiopia (Birhan et al., 2023), Tanzania (Chengula et al., 2025), Nigeria (Adedeji et al., 2019), and Egypt (Gamal et al., 2025), and it is frequently worsened by a lack of vaccinations. MD mortality rates can be effectively managed in nations such as Western Europe, the United States, and Japan that have developed vaccine technologies and intensive poultry production systems (Reddy et al., 2017). However, MDV, the disease’s causal agent, can continue to exist and develop in the chicken farming environment, particularly in the form of subclinical infections, even if the mortality rate is decreased (Lopez et al., 2019). This enables the virus to survive in the population and may eventually lead to the emergence of variants with increased virulence. According to a number of data from regions with high intensification rates, vaccination can lower morbidity and mortality but cannot completely stop illness or CC transfer (Atkins et al., 2013; Zhang et al., 2020). PathogenesisThe pathogenesis of MD is complex, beginning with infection of the respiratory system through inhalation of chicken house dust containing virus particles and progressing to the development of infectious viruses in the epithelial cells of feather follicles (McPherson and Delany, 2016). The four main interconnected stages of MDV pathogenesis are entry, replication, latency, and spread. Early viral replication has been observed in the lungs of B cells and macrophages of infected animals (Baaten et al., 2009). Phagocytic cells, such as macrophages and dendritic cells, transport the virus to the spleen, bursa of Fabricius, and regional lymphatic tissue, infect additional immune cells (Jin et al., 2020). Experimental studies have demonstrated that both phagocytic macrophages and dendritic cells support MDV replication and facilitate cell-to-cell transmission in vitro (Zhu et al., 2024). MDV secretes a viral chemokine that was formerly known as vIL-8 but was more recently renamed vCXCL13 due to its biological characteristics (You et al., 2021c). These chemokines are crucial for the normal establishment of infection because they attract B cells and a minor percentage of CD4+ T cells (Yang et al., 2020). Virus replication occurs in multiple cell types during infection in chickens, with B cells accounting for the majority of infected cells (Thanthrige-Don et al., 2009). Infected B cells are the cells most vulnerable to lytic replication and are easily identified in infected chickens (Trapp-Fragnet et al., 2021). The switch from cytolytic to latent infection is not entirely understood; however, latency starts approximately 1 week after infection, mostly in CD4+ T cells (Torres et al., 2019). Latently infected CD4+ T cells undergo transformation, develop tumors, and spread MDV systemically through the feather follicle epithelium in genetically vulnerable, unvaccinated chickens, where productive replication can resume (Davison and Nair, 2005). MDV spreads throughout the environment and lives in desquamated epithelial cells. CD4+ T lymphocytes limit productive (lytic) infection and inhibit the production of cellular death during latency (Zhu et al., 2024). PathologyMacroscopic lesionsThe classic form of MD is characterized by the swelling of one or more peripheral nerves (Boodhoo et al., 2016). The brachial and sciatic plexuses and nerve trunks, celiac plexus, abdominal vagus, and intercostal nerves are the most commonly afflicted and easily visible nerves (Birhan et al., 2023). The afflicted nerve is generally two to three times thicker than usual and is greatly swollen. The nerve no longer has its typical striped, sparkling appearance (Swayne et al., 1989). The nerves are edematous and grayish or yellowish in appearance (Zeghdoudi et al., 2023). This type of lymphoma manifests as small, soft, gray tumors in the liver, kidneys, heart, ovaries, and other tissues (Burgess et al., 2004). Diffuse lymphoma is the most common lesion in the acute form, affecting many visceral organs, including the heart, ovary, liver, spleen, kidneys, and proventriculus (Kaboudi et al., 2025). Occasionally, lymphoma is observed in skeletal muscle and the skin surrounding hair follicles (Osterrieder et al., 2006). The involvement of peripheral nerves in the classic form may also be observed in affected chickens (Biswas et al., 2018). Younger chickens often have modest liver enlargement in contrast to adults, whose livers are noticeably enlarged and resemble lymphoid leukosis in physical appearance (Payne and Venugopal, 2000). The thymus and bursa of Fabricius may completely disappear due to significant atrophic alterations in the acute cytolytic type of the disease brought on by certain virulent isolates (Berthault et al., 2018). Microscopic lesionsA histological study is necessary for a correct diagnosis, even when gross lesions may indicate malignancy. For this purpose, it is crucial to gather fresh tissue into fixative from multiple cases within the impacted herd (Sadigh et al., 2020). To diagnose Marek’s disease, it is helpful to obtain tissue samples from the liver, spleen, bursa of Fabricius, thymus, heart, proventriculus, kidney, gonads, nerves, skin, and other gross tumor tissues (Mete et al., 2016). Type B lesions on nerves exhibit edema, small lymphocyte and plasma cell infiltration with Schwann cell growth, and an inflammatory appearance (Reddy et al., 2021). Type C lesions are considered regressive inflammatory lesions and are frequently observed in chickens that do not exhibit gross lesions or clinical indications. Light scattering of tiny lymphocytes and plasma cells characterizes them (Zeghdoudi et al., 2023). Demyelination, which is frequently observed in the nerves, causes paralytic symptoms (Payne and Biggs, 1967). The cerebellum, cerebrum, and optic lobes are all affected by widespread vasculitis in chickens exhibiting acute temporary paralysis (Swayne et al., 1989). The cytology of lymphoproliferation in type-A nerve lesions is comparable to that of lymphomas found in visceral organs and other tissues (Das et al., 2018). The majority of lymphoid cells are small and medium lymphocytes, which are typically mixed in type. However, big lymphocytes and lymphoblasts may occasionally predominate, particularly in mature chickens (Gimeno et al., 2001). One key characteristic that distinguishes MD lymphoma from lymphoid leukosis is the presence of polymorphic lymphoid cell populations, which can be observed in tissue slices or cast smears (Kumar et al., 2018). Chickens with acute cytolytic illness have substantial atrophic alterations in their thymus and bursa of Fabricius, where most lymphoid cells are replaced (Berthault et al., 2018). These organs may also acquire neoplastic lymphomatous lesions. MDV-related atherosclerosis has been linked to arterial lesions exhibiting proliferative alterations in the aorta, coronary, celiac, and mesenteric arteries (Lucas, 1998). Clinical symptomsBecause MDV is so common, the presence of the virus in many chicken farms and the detection of virus, viral antigen, or nucleic acid without clinical illness does not prove that MD is present. Classical formEnlargement of one or more peripheral nerves is a common finding (Gall et al., 2018). The brachial and sciatic plexuses, celiac plexus, abdominal vagus, and intercostal nerves are the most commonly impacted and readily apparent post-mortem (Mescolini et al., 2022). Progressive paralysis, usually of the legs, is the hallmark of this condition; affected chickens usually bend one leg under and extend one leg forward (Swayne et al., 1989). Lymphocyte infiltration into the sciatic nerve is the etiology of this disorder (Lopez et al., 2019). Death may result from weight loss, dyspnea, diarrhea, or malnutrition brought on by a lack of access to food and water (Boodhoo et al., 2016). Visceral formIn the acute form, peripheral nerve swelling and tumors are seen in internal organs such as the heart, ovary, spleen, liver, kidneys, and lungs (Alkubaisy and Hameed, 2023). Affected chickens typically show peripheral nerve edema, as in the classic form. The liver may be significantly enlarged in adult chickens, and its visual appearance is identical to that of lymphoid leukosis, from which this condition must be distinguished (Kumar et al., 2018). Liver swelling is typically modest in younger chickens (Abreu et al., 2016). Adult chickens with MD frequently have no nerve lesions. Cutaneous formNodular lesions develop in the feather follicles of young chicks and are immediately identifiable after plucking (Couteaudier and Denesvre, 2014). MD is also referred to as “red leg syndrome” because the hairless parts of the legs might appear vivid red (Szeleszczuk et al., 2006). Ocular formThis uncommon syndrome results in irregular and eccentric pupils, partial or complete blindness, and graying of the iris of the eye due to the invasion of altered lymphocytes (Al-Zebeeby et al., 2024). Death is uncommon. DiagnosisThere are different etiologic agents that might induce similar tumors, making it difficult to diagnose MD in chickens. Both tumor and virus diagnoses must be considered because it is not unusual for a single chicken to have multiple avian tumor viruses. A methodical procedure that incorporates history, epidemiology, gross clinical and necropsy findings, tumor cell characteristics, and virological characteristics has been suggested for the diagnosis of MD. Field diagnosisMD can be tentatively diagnosed by clinically examining infected hens and looking for clinical indicators such as depression, weight loss, flaccid neck, swollen and rough skin around feather follicles, and paralysis of the wings and legs (Biggs and Payne, 1967). Enlargement of the liver and spleen, lymphomas in the liver, kidneys, ovaries, proventriculus, lungs, nerves, heart, and skin, and atrophy of the bursa of Fabricius and thymus are all characteristics of gross lesions of MD (Abreu et al., 2016). Laboratory diagnosisThe histopathology of the damaged organ reveals significant cellular polymorphism, along with the presence of diffuse or well-defined tumor cell infiltration, lymphocytes, lymphoblasts, and fibroblasts (Murthy and Calnek, 1979). These lesions are associated with vacuolization, hepatic duct atrophy, and degeneration and necrosis of liver parenchymal cells (Abreu et al., 2016). Fabricius thymus and bursa exhibit necrosis and damage to lymphoid cells (Schat et al., 1981). As part of the virus culture process, a virus sample is inserted into several cell lines that are susceptible to infection by the virus under examination (Tien et al., 2023). The culture is considered successful if the cells exhibit alterations, also referred to as a cytopathic effect. Primary chicken embryonic kidney cells made from 18-day-old embryonated particular pathogen-free eggs can be utilized for MDV cell culture (Abd El-Ghany, 2025). Additionally, 10–11-day-old chicken embryonated specific pathogen-free eggs were used to create primary fibroblast cells for MDV isolation (Kamble et al., 2021). The virus was isolated from suspected tissue samples to detect MDV. Therefore, a 10% w/v tissue suspension was prepared and inoculated into kidney monolayer cultures of chicken embryos grown in plastic cell culture tubes for four consecutive passages using a sterile phosphate buffer solution (Ewies et al., 2021). Then, the inoculation was repeated in four consecutive passages of chicken embryo fibroblast cells. A humidified incubator with 5% CO2 was used to incubate both inoculated and uninoculated control cultures at 37°C. The culture media was changed once every 2 days. The cytopathic impact region became apparent 6–8 days after incubation (Imai et al., 1990). Serological testsSerological diagnostic techniques for identifying MDV antibodies generated in response to MDV infection include agar gel immunodiffusion (AGID), indirect fluorescent antibody, and enzyme-linked immunosorbent assay (ELISA) (Tuli and Fayisa, 2023). AGID uses glass slides coated with 1% agar in a phosphate buffer solution with 8% sodium chloride to identify MDV antibodies generated in response to MDV infection (Boodhoo et al., 2019). To permit diffusion, adjacent wells were loaded with serum or antigen and incubated for 24 hours at 37°C in a humidified environment. A cross-linked antigen-antibody complex that precipitates in the agar is created when antibodies to an antigen present in the serum sample bind together (Burgess and Davison, 2002). Using indirect fluorescent antibodies, antigen-specific antibodies can be found in a suspected sample. These antibodies target the primary antibody used for detection with fluorophore-conjugated secondary antibodies and unconjugated primary antibodies (Kurokawa and Yamamoto, 2022). Rather than attaching to antigens, fluorescent secondary antibodies attach to antigen-specific antibodies. Indirect immunofluorescence provides several benefits over direct immunofluorescence, including being inexpensive because secondary antibodies can be used for a variety of primary antibodies (Purchase, 1969). Antibodies in serum and other bodily fluids can be quantitatively estimated using indirect enzyme-linked immunosorbent assay (Zelnik et al., 2004). The binding mechanism of primary antibodies and enzyme-labeled secondary antibodies is crucial for the indirect ELISA concept (Sakamoto et al., 2018). This technique involves adding the specimen to the well of a microtiter plate coated with the antigen that certain antibodies detect. The wells were cleaned after the incubation period. An antigen-antibody combination will form in the sample if antibodies are present, and it will not wash off. However, no complex will form if the specimen has no particular antibodies. After that, enzyme-conjugated anti-isotype antibodies are added and incubated. The enzyme substrate is added following the subsequent washing step (Cheng et al., 1984). Indirect ELISA has a higher sensitivity because it requires fewer labeled antibodies and binds several labeled antibodies to each antigen molecule (Scholten et al., 1990). Molecular-based techniquesSeveral MDV genes can be amplified and quantified using a molecular technique called real-time polymerase chain reaction (RT-PCR) (Angamuthu et al., 2012). Real-time measurements of amplification are performed during the reaction. The detector measures the amount of fluorescence released during amplification. As a diagnostic tool for MD, this tool provides a quicker and more accurate test (Gall et al., 2018). Samples of frozen muscle, liver, spleen, kidney, and feather tissue were homogenized using a designated homogenizer (Blume et al., 2016). RT-PCR was performed using forward and reverse primers to target the amplification of the MDV meq, Gb, and ICP4 genes (Zhang et al., 2018). Differential diagnosisThe two most prevalent illnesses in chickens that may be distinguished from MD are lymphoid leukosis and reticuloendotheliosis. In contrast to reticuloendotheliosis and lymphoid leukosis, MD can afflict chickens of any age, including those aged 16 weeks (Woźniakowski et al., 2018). In contrast to lymphoid leukosis and reticuloendotheliosis, MD frequently causes paralysis of the wings and legs, nerve expansion, and central nervous system involvement (Payne and Venugopal, 2000). TransmissionThe main horizontal route of MD transmission is from infected to healthy chickens (Boodhoo et al., 2016). MDV-infected chickens expel the virus from their body through dander, which is made up of dust and epithelial flakes from feather follicles (Jarosinski, 2012a,b). These tiny particles can linger in the atmosphere for extended periods, especially in spaces with inadequate ventilation or low sanitary standards (Ding et al., 2024). Air tainted by these dust particles can expose chickens that lack immunity to MDV (Heidari et al., 2023). MDV initially replicates in the respiratory tract epithelium and alveolar macrophages when it enters the body through the respiratory system (Akbar et al., 2023). The thymus and bursa of Fabricius are among the key lymphoid organs where MDV subsequently expands before infecting CD4+ T cells (Schat, 2022). The body’s neurological system, skin, and viscera are only a few places where infected lymphocytes might develop into cancerous cells (Couteaudier and Denesvre, 2014). As a result of this process, infected chickens continue to lose MDV from their feather follicles, which permits the development of clinical disease and transmission within a flock (Jarosinski, 2012a,b). Fomites, which are items or equipment tainted with MDV particles from fur and dust follicles, such as food or drink containers, clothing, shoes, or automobiles, can also act as a medium for the spread of MD (Woźniakowski and Samorek-Salamonowicz, 2014). MDV does not travel vertically from mother to egg like some other poultry viruses do, but it does spread quickly from one area to another and from one chicken to another because of its effective horizontal transmission pattern (Jarosinski et al., 2007). The transmission of MDV involves a complex interplay between infected birds, the environment, and susceptible hosts, as illustrated in Figure 3. The primary routes of horizontal transmission, including inhalation of contaminated feather dander and indirect spread via fomites, as well as the virus’s persistence in the environment under poor sanitation conditions, contribute to ongoing infection cycles within poultry flocks.
Fig. 3. MDV transmission in chickens. Risk factorsMD risk factors are directly linked to the chicken’s age, immune condition, maintenance system, and virus virulence degree. The most vulnerable are very young chicks, particularly those in the first few weeks following hatching, as their immune systems are still developing and cannot mount a sufficient defense (Zhu et al., 2024). Given that dust particles and epithelial debris from virus-carrying feather follicles can survive in these conditions for weeks or even months, the danger is also increased when hens are housed in high-density, unsanitary, and inadequately ventilated situations (Woźniakowski and Samorek-Salamonowicz, 2014). Furthermore, the virulent level of circulating MDV strains significantly influences the pattern of illness incidence and severity; even in vaccinated chickens, very virulent (vvMDV), and very virulent plus (vv+MDV) strains can cause large levels of mortality (Liu et al., 2023). Another significant risk factor is the chicken’s immune condition, particularly if it has been exposed to other immunosuppressive substances, such as Newcastle disease and infectious bursal disease viruses, which can weaken the body’s defenses against MDV infection (Schat and Skinner, 2014). Genetic influences cannot be overlooked because chickens from lines with poor genetic resistance are more prone to MD than those from lines with higher genetic resistance (Chang et al., 2014). The danger of MD emergence and development in a population depends heavily on the intricate interplay between the genetic background of the chickens being reared, their age, management and biosecurity practices, immunological status, and the degree of viral virulence (Chu et al., 2019). Economic impactPrior to the development of vaccinations, MD posed a significant financial risk to the chicken industry, leading to mortality rates of up to 60% and 10% in laying and broiler flocks, respectively (Dejyong et al., 2023). Although sporadic losses still occur because vaccinations are not 100% effective, they are no longer a significant concern (Atkins et al., 2013). The projected cost of fatalities and mortality in the United States because of MD in 1984 was approximately $12 million. When broken down by category, the financial burden of MD includes the following: (i) decreased egg production, which directly reduces revenue from laying hens; (ii) decreased feed conversion rate, leading to higher feed costs for the same weight gain; and (iii) increased vaccination and biosecurity expenses, which significantly add to production costs. The total economic impact is approximately $169 million in the US and $943 million globally when these losses are included (Dunn and Gimeno, 2013). Recent global estimates suggest that current losses, combining mortality, reduced productivity, and prevention measures, are between $1 and $2 billion annually, although these numbers cannot be confirmed (Zeghdoudi et al., 2023). The poultry industry continues to be very concerned about MDV due to its unpredictable outbreaks and the potential for vaccines to eventually fail due to the emergence of more virulent MDV strains (Yu et al., 2023). In Ethiopia, MD was initially identified in 1983, and between 1983 and 1986, a 3% incidence rate was noted in industrial chicken farms (Getahun, 2023). MD may be important for contemporary chicken production in Ethiopia, as evidenced by the high mortality rate (46%) in commercial poultry farms during an outbreak in central Ethiopia (Demeke et al., 2017). During this outbreak, mortality alone resulted in an estimated financial loss of 76,000 birr over 3.5 months, without accounting for production losses or prevention costs. Ethiopian chickens are not currently vaccinated against MD, despite its economic importance in Ethiopian chicken production (Duguma et al., 2005). TreatmentThere is no proven specific treatment for MD, particularly in hens that already have neoplastic tumors or clinical signs. MDV is a DNA herpes virus that can live and conceal itself in lymphoid cells, making it challenging for antiviral treatment to reach these cells (Lopez et al., 2019). Consequently, prevention rather than therapy, specifically through vaccination and the application of stringent biosecurity protocols, is the best control strategy (Reddy et al., 2017). VaccinationThe vaccination works quite well; in commercial settings, it frequently provides more than 90% protection (Baigent et al., 2007). Turkey strain FC-126 herpesvirus is commonly used and works very well against aggressive MDV (Calderón et al., 2022). Bivalent vaccination that combines serotype-2 strain SB-1 with HVT works in concert to offer superior defense against virulent MDV (Neerukonda et al., 2019). Since immunity develops in 2 weeks and handling of the chicks is not necessary, in-ovo vaccination is the preferred method (Abd El-Ghany, 2025). MDV has developed greater virulence and resistance to these vaccinations, as they do not stop viral infection. Therefore, the current vaccines are combination vaccinations that employ either an attenuated strain of MDV, CVI988/Rispens, or HVT and Gallidherpesvirus type 3 (Sadigh et al., 2018). Widespread vaccination significantly reduced illness losses by more than 99% and provided hosts with another crucial tool against the virus (Atkins et al., 2013). Many nations have implemented the attenuated form of MDV, CVI988, which is considered the best protective vaccine currently available (Ralapanawe et al., 2016). The MDV vaccination is given in the ED in vivo. The vaccine inoculum is automatically injected into the amniotic fluid of most eggs (Williams and Hopkins, 2011). However, this illness presents a significant threat to the chicken industry and complicates management measures due to rising vaccination failure rates and the appearance of increasingly aggressive pathogens. MD vaccination has numerous intrinsic flaws despite its effectiveness rate of over 95% (Atkins et al., 2013). The most significant finding is that it cannot cause “sterile immunity” in hosts that have received vaccinations (Yu et al., 2023). This makes it possible for virulent virus strains to proliferate and spread across the environment regardless of the vaccination level of the host. HVT does not provide protection against MD in turkeys, but CVI988/Rispens vaccination does (Ralapanawe et al., 2016). ControlThe poultry sector faces a challenge in controlling MD because the virus can persist in the environment for a long period and keeps evolving. The first step in control efforts is vaccination, which is currently the most effective way to reduce the severity and death rate from MDV infection (Atkins et al., 2013). Marek’s vaccine is typically administered to day-old chicks via subcutaneous injection or the in-ovo technique to provide them with a foundational degree of protection prior to their exposure to environmental infection (Peebles et al., 2017). Vaccination can lessen disease severity, decrease tumor formation, and lower death rates, allowing chickens to grow more ideally even though it cannot completely stop the virus from entering the body (Fazel et al., 2024). In addition to vaccination, rigorous biosecurity measures are crucial for controlling MD. These measures include managing litter and dust from chicken feather follicles, keeping an eye on vehicle and human movement between farm sites, thoroughly cleaning cages and equipment, and regulating cage density and ventilation levels (Bavananthasivam et al., 2022). The danger of MD spreading can be considerably reduced by reducing environmental contamination and severing the transmission chain from affected flocks to healthy flocks (Kennedy et al., 2017). Genetic selection can also aid in the control of MD by preserving and breeding chicken breeds with greater innate resistance to MDV (Hearn and Cheng, 2023). This procedure can be used in conjunction with current vaccination and biosecurity measures because certain chicken strains are known to be more resistant than others. ConclusionMD is one of the most important neuropathic and lymphoproliferative illnesses in the poultry sector and is found in practically every nation with a high degree of intensified chicken farming. Its distribution pattern has expanded worldwide. The disease’s history, transmission patterns, epidemiology, and control are only a few of the factors that highlight MD’s status as a complicated threat to poultry productivity and health. Even with current vaccine technology, MD is challenging to fully control due to risk variables such as chicken age, MDV strain virulence level, immune state, and degree of biosecurity and husbandry management. Successful MD control is primarily dependent on the application of comprehensive preventive measures, such as vaccination with suitable strains, stringent biosecurity procedures, genetic selection for increased resistance, and continuous monitoring and surveillance, even though there is no known cure for the disease. More flexible and effective control strategies will be developed with a better understanding of the epidemiological features, transmission patterns, and distribution of MDV to reduce the negative health and economic effects of MD on the global poultry industry. AcknowledgmentsThe authors thank Universitas Airlangga and Badan Riset dan Inovasi Nasional. Author’s contributionsSM, ARK, IM, PPR, and BWKW drafted the manuscript. BPP, MKJK, DAAK, IBM, and SW revised and edited the manuscript. FE, ATK, AOA, KAF, and BPP prepared and critically checked the manuscript. IF, SSP, RZA, and AHF edited the references. All authors have read and approved the final version of the manuscript. Conflict of interestThe authors declare no conflict of interest. FundingData availabilityAll references are open access, so data can be obtained from the internet. ReferencesAbayli, H., Karabulut, B., Ozbek, R., Ongor, H., Timurkaan, N. and Tonbak, S. 2021. Detection and molecular characterization of a highly oncogenic Marek’s disease virus from vaccinated hens in Turkey. Acta Virol. 65(2), 212–220. Abd El-ghany, W.A. 2025. In ovo vaccination technology: an alternative approach to post-hatch vaccination in modern poultry operations. Microbiol. Res. 16(1), 7. Abreu, D., Santos, F., José, D., Tortelly, R., Nascimento, E. and Pereira, V. 2016. Pathological aspects of a subclinical Marek’s disease case in free-range chickens. Rev. Bras. Cienc. Avic. 18(1), 197–199. Adedeji, A.J., Akanbi, O.B., Luka, P.D. and Abdu, P. 2019. Natural outbreak of Marek’s disease in indigenous chicken and Japanese quail (Coturnix coturnix japonica) in Jos, Plateau State, Nigeria. Open Vet. J. 9(2), 151–156. Akbar, H., Fasick, J.J., Ponnuraj, N. and Jarosinski, K.W. 2023. Purinergic signaling during Marek’s disease in chickens. Sci. Rep. 13(1), 2044. Alkubaisy, M.A. and Hameed, S.S. 2023. Detection of pathotype of Marek’s Disease in layer chicken in Iraq. University of Thi-Qar J Agri. Res. 12(2), 25–34. Al-Zebeeby, A., Abbas, A.H., Alsaegh, H.A. and Alaraji, F.S. 2024. The first record of an aggressive form of ocular tumour enhanced by Marek’s disease virus infection in layer flock in Al-Najaf, Iraq. Vet. Med. Int. 1(1), 1793189. Angamuthu, R., Baskaran, S., Gopal, D.R., Devarajan, J. and Kathaperumal, K. 2012. Rapid detection of the Marek’s disease viral genome in chicken feathers by loop-mediated isothermal amplification. J. Clin. Microbiol. 50(3), 961–965. Arumugaswami, V., Kumar, P.M., Konjufca, V., Dienglewicz, R.L., Reddy, S.M. and Parcells, M.S. 2009. Latency of Marek’s disease virus (MDV) in a reticuloendotheliosis virus-transformed T-cell line. I: uptake and structure of the latent MDV genome. Avian Dis. 53(2), 149–155. Atkins, K.E., Read, A.F., Walkden-Brown, S.W., Savill, N.J. and Woolhouse, M.E.J. 2013. The effectiveness of mass vaccination on Marek’s disease virus (MDV) outbreaks and detection within a broiler barn: a modeling study. Epidemics 5(4), 208–217. Baaten, B.J.G., Staines, K.A., Smith, L.P., Skinner, H., Davison, T.F. and Butter, C. 2009. Early replication in pulmonary B cells after infection with Marek’s disease herpesvirus by the respiratory route. Viral. Immunol. 22(6), 431–444. Baigent, S.J., Smith, L.P., Currie, R.J.W. and Nair, V.K. 2007. Correlation of Marek’s disease herpesvirus vaccine virus genome load in feather tips with protection, using an experimental challenge model. Avian Pathol. 36(6), 467–474. Bavananthasivam, J., Alqazlan, N., Alizadeh, M., Matsuyama-Kato, A., Astill, J., Kulkarni, R.R. and Sharif, S. 2022. The regulatory microenvironment in feathers of chickens infected with very virulent Marek’s disease virus. Viruses 14(1), 112. Bell, A.S., Kennedy, D.A., Jones, M.J., Cairns, C.L., Pandey, U., Dunn, P.A., Szpara, M.L. and Read, A.F. 2019. Molecular epidemiology of Marek’s disease virus in central Pennsylvania, USA. Virus. Evol. 5(1), 42. Berthault, C., Larcher, T., Härtle, S., Vautherot, J.F., Trapp-Fragnet, L. and Denesvre, C. 2018. Atrophy of primary lymphoid organs induced by Marek’s disease virus during early infection is associated with increased apoptosis, inhibition of cell proliferation and a severe B-lymphopenia. Vet. Res. 49(1), 31. Bertzbach, L.D., Conradie, A.M., You, Y. and Kaufer, B.B. 2020. Latest insights into Marek’s disease virus pathogenesis and tumorigenesis. Cancers. 12(3), 647. Bertzbach, L.D., Kheimar, A., Ali, F.A.Z. and Kaufer, B.B. 2018. Viral factors involved in Marek’s disease virus (MDV) pathogenesis. Curr. Clin. Microbiol. Rep. 5(1), 238–244. Bertzbach, L.D., Laparidou, M., Härtle, S., Etches, R.J., Kaspers, B., Schusser, B. and Kaufer, B.B. 2018. Unraveling the role of B cells in the pathogenesis of an oncogenic avian herpesvirus. Proc. Natl. Acad. Sci. U. S. A. 115(45), 11603–11607. Bertzbach, L.D., Van Haarlem, D.A., Härtle, S., Kaufer, B.B. and Jansen, C.A. 2019. Marek’s disease virus infection of natural killer cells. Microorganisms 7(12), 588. Biggs, P.M. and Nair, V. 2012. The long view: 40 years of Marek’s disease research. Avian Pathol 41(1), 3–9; doi: 10.1080/03079457.2011.646237 Biggs, P.M. and Payne, L.N. 1967. Studies on Marek’s disease. I. Experimental transmission. J. Natl. Cancer Inst. 39(2), 267–280. Birhan, M., Gelaye, E., Ibrahim, S.M., Berhane, N., Abayneh, T., Getachew, B., Zemene, A., Birie, K., Deresse, G., Adamu, K., Dessalegn, B., Gessese, A.T., Kinde, M.Z. and Bitew, M. 2023. Marek’s disease in chicken farms from Northwest Ethiopia: gross pathology, virus isolation, and molecular characterization. Virol. J. 20(1), 45. Biswas, D., Ruba, T., Yasmin, A., Nazneen, M., Hossain, M. and Khan, M. 2018. Pathological investigation of Marek’s disease in a pullet farm of Bangladesh and detection of Marek’s disease virus by PCR. SAARC J. Agric. 16(1), 181–192. Blume, G.R., Cardoso, S.P., Oliveira, M.L.B., Matiolli, M.P., Gómez, S.Y.M., Reis Júnior, J.L., Sant’Ana, F.J.F. and Martins, N.R.S. 2016. Visceral Marek’s disease in white-peafowl (Pavo cristatus). Arq. Bras. Med. Vet. Zootec. 68(6), 1602–1608. Boodhoo, N., Gurung, A., Sharif, S. and Behboudi, S. 2016. Marek’s disease in chickens: a review with focus on immunology. Vet. Res. 47(1), 119. Boodhoo, N., Kamble, N., Kaufer, B.B. and Behboudi, S. 2019. Replication of Marek’s disease virus is dependent on synthesis of de novo fatty acid and prostaglandin E2. J. Virol. 93(13), e00352-19. Burgess, S.C. and Davison, T.F. 2002. Identification of the neoplastically transformed cells in Marek’s disease herpesvirus-induced lymphomas: recognition by the monoclonal antibody AV37. J. Virol. 76(14), 7276–7292. Burgess, S.C., Young, J.R., Baaten, B.J., Hunt, L., Ross, L.N., Parcells, M.S., Kumar, P.M., Tregaskes, C.A., Lee, L.F. and Davison, T.F. 2004. Marek’s disease is a natural model for lymphomas overexpressing Hodgkin’s disease antigen (CD30). Proc. Natl. Acad. Sci. U. S. A. 101(38), 13879–13884. Buscaglia, C., Nervi 4, P. and Risso, M. 2004. Characterization of four very virulent Argentinian strains of Marek’s disease virus and the influence of one of those isolates on synergism between Marek’s disease vaccine viruses. Avian Pathol. 33(2), 190–195. Calderón, K., Rojas-Neyra, A., Carbajal-Lévano, B., Luján-Valenzuela, L., Ticona, J., Isasi-Rivas, G., Montalvan, A., Criollo-Orozco, M., Huaccachi-Gonzáles, E., Tataje-Lavanda, L., Alvarez, K.L.F., Fernández-Sánchez, M., Fernández-Díaz, M., Tang, N., Yao, Y. and Nair, V. 2022. A recombinant turkey herpesvirus expressing the F protein of newcastle disease virus genotype XII Generated by NHEJ-CRISPR/Cas9 and Cre-LoxP Systems Confers Protection against Genotype XII Challenge in Chickens. Viruses 14(4), 793. Calnek, B.W. 2001. Pathogenesis of Marek’s disease virus infection. Curr. Top. Microbiol. Immunol. 255, 25–55. Chacón, R.D., Sánchez-Llatas, C.J., Astolfi-Ferreira, C.S., Raso, T.F. and Ferreira, A.J.P. 2024. Diversity of Marek’s disease virus strains in infections in backyard and ornamental birds. Animals. 14(19), 2867. Chakraborty, P., Kuo, R.I., Wu, Z., Morris, K.M., Dutia, B.M., Kaiser, P. and Smith, J. 2022. The role of dendritic cells in the host response to Marek’s Disease Virus (MDV) as shown by transcriptomic analysis of susceptible and resistant birds. Pathogens 11(11), 1340. Chakraborty, P., Vervelde, L., Dalziel, R.G., Wasson, P.S., Nair, V., Dutia, B.M. and Kaiser, P. 2017. Marek’s disease virus infection of phagocytes: a de novo in vitro infection model. J. Gen. Virol. 98(5), 1080–1088. Chang, S., Xie, Q., Dunn, J.R., Ernst, C.W., Song, J. and Zhang, H. 2014. Host genetic resistance to Marek’s disease sustains protective efficacy of herpesvirus of turkey in both experimental and commercial lines of chickens. Vaccine 32(16), 1820–1827. Chbab, N., Egerer, A., Veiga, I., Jarosinski, K.W. and Osterrieder, N. 2010. Viral control of vTR expression is critical for efficient formation and dissemination of lymphoma induced by Marek’s disease virus (MDV). Vet. Res. 41(5), 56. Cheng, Y.Q., Lee, L.F., Smith, E.J. and Witter, R.L. 1984. An enzyme-linked immunosorbent assay for the detection of antibodies to Marek’s disease virus. Avian Dis. 28(4), 900–911. Chengula, A.A., Mpete, H. and Makasali, R.J. 2025. Surveillance and Molecular Characterization of Marek’s Disease Virus (MDV) Strains Circulating in Tanzania. Viruses 17(5), 698. Chu, Q., Ding, Y., Cai, W., Liu, L., Zhang, H. and Song, J. 2019. Marek’s Disease Virus Infection Induced Mitochondria Changes in Chickens. Int. J. Mol. Sci. 20(13), 3150. Churchill, A.E. and Biggs, P.M. 1967. Agent of Marek’s disease in tissue culture. Nature 215(5100), 528–530. Conradie, A.M., Bertzbach, L.D., Trimpert, J., Patria, J.N., Murata, S., Parcells, M.S. and Kaufer, B.B. 2020. Distinct polymorphisms in a single herpesvirus gene are capable of enhancing virulence and mediating vaccinal resistance. PLos Pathog. 16(12), e1009104. Coudert, F., Vuillaume, A., Wyers, M. and Legros, F.X. 1995. 1ères Journées de la Recherche Avicole Angers. INRA Institut National de la Recherche Agronomique; Paris, France. Une Nouvelle Pathologie chez la Dinde. La Maladie de Marek; pp. 164–166. Couteaudier, M. and Denesvre, C. 2014. Marek’s disease virus and skin interactions. Vet. Res. 45(1), 36. Das, S., Das, D., Panda, S., Sagarika, S. and Jena, B. 2018. Clinico-pathological studies of Marek’s disease in chickens. Int. J. Livest. Res. 8(1), 207–217. Davison, A.J. 2010. Herpesvirus systematics. Vet. Microbiol. 143(1), 52–69. Davison, F. and Nair, V. 2005. Use of Marek’s disease vaccines: could they be driving the virus to increasing virulence?. Expert Rev. Vaccines 4(1), 77–88. Dejyong, T., Chanachai, K., Prarakamawongsa, T., Kongkaew, W., Thiptara, A., Songserm, T., Rukkwamsuk, T., Tagopacheco, D. and Phimpraphai, W. 2023. Economic and value chain analysis to support an investigation and risk mitigation efforts on Marek’s disease in layers in the southern part of Thailand. Vet. World 16(1), 35–45. Demeke, B., Jenberie, S., Tesfaye, B., Ayelet, G., Yami, M., Lamien, C.E. and Gelaye, E. 2017. Investigation of Marek’s disease virus from chickens in central Ethiopia. Trop. Anim. Health Prod. 49(2), 403–408. Ding, K., Yu, Z.H., Yu, C., Jia, Y.Y., He, L., Liao, C.S., Li, J., Zhang, C.J., Li, Y.J., Wu, T.C., Cheng, X.C., Zhou, Z.Y. and Yu, Z.L. 2020. Effect of gga-miR-155 on cell proliferation, apoptosis and invasion of Marek’s disease virus (MDV) transformed cell line MSB1 by targeting RORA. BMC. Vet. Res. 16(1), 23. Ding, T., Xiong, M., Xu, Y., Pu, X., Wang, Q.S., Xu, M.R., Shao, H.X., Qian, K., Dang, H.B. and Qin, A.J. 2024. Dynamic changes in viral loads during co-infection with a recombinant turkey herpesvirus vector vaccine and very virulent Marek’s disease virus in vivo. Viruses 16(7), 1042. Duguma, R., Yami, A., Dana, N., Hassen, F. and Esatu, W. 2005. Marek’s disease in local chicken strains of Ethiopia reared under confined management regime in central Ethiopia. Revue Méd. Vét. 156(11), 541–546. Dunn, J.R. and Gimeno, I.M. 2013. Current status of Marek’s disease in the United States and worldwide based on a questionnaire survey. Avian Dis. 57(2 Suppl), 483–490. Emad, A., El-Kenawy, A.A. and El-Tholoth, M. 2024. Molecular characterization of Marek’s Disease virus reveals reticuloendotheliosis virus-long terminal repeat integration in the genome of the field isolates in Egypt. Poult. Sci. 103(6), 103722. Engel, A.T., Selvaraj, R.K., Kamil, J.P., Osterrieder, N. and Kaufer, B.B. 2012. Marek’s disease viral interleukin-8 promotes lymphoma formation through targeted recruitment of B cells and CD4+ CD25+ T cells. J. Virol. 86(16), 8536–8545. Ewies, S., Madbouly, H., Tamam, S., Arafa, A., Mady, W. and Hamad, E. 2021. Isolation and Molecular Characterization of Marek’s Disease Virus from Layer Chickens in Egypt. J. Vet. Med. Res. 27(2), 168–176. Fazel, F., Matsuyama-Kato, A., Alizadeh, M., Zheng, J., Fletcher, C., Gupta, B., St-Denis, M., Boodhoo, N. and Sharif, S. 2024. A Marek’s Disease Virus Messenger RNA-Based Vaccine Modulates Local and Systemic Immune Responses in Chickens. Viruses 16(7), 1156. Fragnet, L., Blasco, M.A., Klapper, W. and Rasschaert, D. 2003. The RNA subunit of telomerase is encoded by Marek’s disease virus. J. Virol. 77(10), 5985–5996. Fukuchi, K., Tanaka, A., Schierman, L.W., Witter, R.L. and Nonoyama, M. 1985. The structure of Marek disease virus DNA: the presence of unique expansion in nonpathogenic viral DNA. Proc. Natl. Acad. Sci. U. S. A. 82(3), 751–754. Gall, S., Kőrösi, L., Cortes, A.L., Delvecchio, A., Prandini, F., Mitsch, P. and Gimeno, I.M. 2018. Use of real-time PCR to rule out Marek’s disease in the diagnosis of peripheral neuropathy. Avian Pathol. 47(4), 427–433. Gamal, M.A.N., El-Nagar, E.M.S., Khattab, M.S. and Salem, H.M. 2025. Molecular discernment and histopathological features of oncogenic Marek’s disease virus among different farmed avian species in Egypt. Sci. Rep. 15(1), 15409. Getahun, T.K. 2023. Marek’s disease; current status in Ethiopia and its vaccination trial. Glob. Sci. Res. J. 11(1), 1–10. Ghalyanchilangeroudi, A., Hosseini, H., Nazarpak, H.H., Molouki, A., Dezfoulian, O. and Morshed, R. 2022. Molecular Characterization and Phylogenetic Analysis of Marek’s Disease Virus in Iran. Avian Dis. 66(3), 1–5. Gimeno, I.M., Witter, R.L., Hunt, H.D., Lee, L.F., Reddy, S.M. and Neumann, U. 2001. Marek’s disease virus infection in the brain: virus replication, cellular infiltration, and major histocompatibility complex antigen expression. Vet. Pathol. 38(5), 491–503. Hagag, I.T., Wight, D.J., Bartsch, D., Sid, H., Jordan, I., Bertzbach, L.D., Schusser, B. and Kaufer, B.B. 2020. Abrogation of Marek’s disease virus replication using CRISPR/Cas9. Sci. Rep. 10(1), 10919. Hartawan, R. and Dharmayanti, N.L.P.I. 2016. The Meq Gene Molecular Profile of Marek’s Disease Virus Serotype 1 From Kampung and Arabic Chicken Farms in Sukabumi, West Java, Indonesia. HAYATI J. Biosci. 23(4), 160–167. Hauck, R., Mays, J., Dunn, J.R. and Shivaprasad, H.L. 2020. Two Cases of Marek’s Disease in Backyard Turkeys. Avian Dis. 64(3), 347–351. Hearn, C.J. and Cheng, H.H. 2023. Contribution of the TCRβ Repertoire to Marek’s Disease Genetic Resistance in the Chicken. Viruses 15(3), 607. Heidari, M., Zhang, H., Sunkara, L.T. and Ahmad, S.M. 2023. Role of T Cells in Vaccine-Mediated Immunity against Marek’s Disease. Viruses 15(3), 648. Hughes, C.E. and Nibbs, R.J.B. 2018. A guide to chemokines and their receptors. FEBS. J. 285(16), 2944–2971. Imai, K., Yuasa, N., Kobayashp, S., Nakamura, K., Tsukamoto, K. and Hihara, H. 1990. Isolation of Marek’s disease virus from Japanese quail with lymphoproliferative disease. Avian Pathol. 19(1), 119–129. Jarosinski, K.W. 2012. Marek’s disease virus late protein expression in feather follicle epithelial cells as early as 8 days postinfection. Avian Dis. 56(4), 725–731. Jarosinski, K.W. and Osterrieder, N. 2012. Marek’s disease virus expresses multiple UL44 (gC) variants through mRNA splicing that are all required for efficient horizontal transmission. J. Virol. 86(15), 7896–7906. Jarosinski, K.W., Margulis, N.G., Kamil, J.P., Spatz, S.J., Nair, V.K. and Osterrieder, N. 2007. Horizontal transmission of Marek’s disease virus requires US2, the UL13 protein kinase, and gC. J. Virol. 81(19), 10575–10587. Jin, H., Kong, Z., Mehboob, A., Jiang, B., Xu, J., Cai, Y., Liu, W., Hong, J. and Li, Y. 2020. Transcriptional Profiles Associated with Marek’s Disease Virus in Bursa and Spleen Lymphocytes Reveal Contrasting Immune Responses during Early Cytolytic Infection. Viruses 12(3), 354. Kaboudi, K., Choura, I., Ammouna, F., Amara, A. and Ghram, A. 2025. Prevalence, gross pathology, and histopathology of Marek’s disease in backyard chickens in Northeastern Tunisia (2012-2020). J. World Poultry Res 15(1), 81–91. Kamble, N., Gurung, A., Kaufer, B.B., Pathan, A.A. and Behboudi, S. 2021. Marek’s Disease Virus Modulates T Cell Proliferation via Activation of Cyclooxygenase 2-Dependent Prostaglandin E2. Front. Immunol. 12(1), 801781. Kamble, N., Reddy, V.R.A.P., Jackson, B., Anjum, F.R., Ubachukwu, C.C., Patil, A. and Behboudi, S. 2023. Inhibition of Marek’s Disease Virus Replication and Spread by 25-hydroxycholesterol and 27-hydroxycholesterol In Vitro. Viruses 15(8), 1652. Kaupp, B. F. 1921. Poultry disease work of Europe and of the poultry congress. Poultry Sci 1(1), 30–31; doi: 10.3382/ps.0010030 Kennedy, D.A., Cairns, C., Jones, M.J., Bell,, A.S., Salathé, R.M., Baigent, S.J., Nair, V.K., Dunn, P.A. and Read, A.F. 2017. Industry-Wide Surveillance of Marek’s Disease Virus on Commercial Poultry Farms. Avian Dis. 61(2), 153–164. Kennedy, D.A., Dunn, P.A. and Read, A.F. 2018. Modeling Marek’s disease virus transmission: a framework for evaluating the impact of farming practices and evolution. Epidemics 23(1), 85–95. Kheimar, A. and Kaufer, B.B. 2018. Epstein-Barr virus-encoded RNAs (EBERs) complement the loss of Herpesvirus telomerase RNA (vTR) in virus-induced tumor formation. Sci. Rep. 8(1), 209. Kheimar, A., Previdelli, R., Wight, D. and Kaufer, B. 2017. Telomeres and Telomerase: role in Marek’s Disease Virus Pathogenesis, Integration and Tumorigenesis. Viruses 9(7), 173. Kumar, M.A., Palanivelu, M., Barathidasan, R., Kumar, D., Singh, S.D., Lateef, S.K., Singh, R. and Dhama, K. 2018. Cytological and immunocytological detection and differentiation of Marek’s disease and lymphoid leucosis in poultry. Virusdisease 29(3), 349–354. Kurokawa, A. and Yamamoto, Y. 2022. Development of monoclonal antibodies specific to Marek disease virus-EcoRI-Q (Meq) for the immunohistochemical diagnosis of Marek disease using formalin-fixed, paraffin-embedded samples. J. Vet. Diagn. Invest. 34(3), 458–464. Liao, Y., Lupiani, B., Ai-Mahmood, M. and Reddy, S.M. 2021. Marek’s disease virus US3 protein kinase phosphorylates chicken HDAC 1 and 2 and regulates viral replication and pathogenesis. PLos Pathog. 17(2), e1009307. Liao, Y., Zhuang, G., Sun, A., Khan, O.A., Lupiani, B. and Reddy, S.M. 2020. Marek’s Disease Virus Cluster 3 miRNAs Restrict Virus’ Early Cytolytic Replication and Pathogenesis. Viruses 12(11), 1317. Liu, J.L., Teng, M., Zheng, L.P., Zhu, F.X., Ma, S.X., Li, L.Y., Zhang, Z.H., Chai, S.J., Yao, Y. and Luo, J. 2023. Emerging Hypervirulent Marek’s Disease Virus Variants Significantly Overcome Protection Conferred by Commercial Vaccines. Viruses 15(7), 1434. Lopez, O.S., Villar, A.D. and Chaparro, G.J. 2019. Challenges in the diagnosis and control of Marek’s disease virus in Colombia. Rev. MVZ. Córdoba 24(1), 7157–7169. Lucas, A. 1998. Atherosclerosis in Marek’s disease virus infected hypercholesterolemic roosters is reduced by HMGCoA reductase and ACE inhibitor therapy. Cardiovasc. Res. 38(1), 237–246. Mallet, C., Cochard, J., Leclercq, S., Trapp-Fragnet, L., Chouteau, P. and Denesvre, C. 2022. Hypoxia and HIF-1 Trigger Marek’s Disease Virus Reactivation in Lymphoma-Derived Latently Infected T Lymphocytes. J. Virol. 96(5), 142721. Marek, J. 1907. Multiple Nervenentzündung (Polyneuritis) bei Hühnern. Dtsch. Tierärztl. Wschr. 15(1), 417–421. Mcpherson, M.C. and Delany, M.E. 2016. Virus and host genomic, molecular, and cellular interactions during Marek’s disease pathogenesis and oncogenesis. Poult. Sci. 95(2), 412–429. Mescolini, G., Lupini, C., Di Francesco, A., Davidson, I., Felice, V., Bellinati, L., Cecchinato, M. and Catelli, E. 2022. Marek’s disease in genetically susceptible Cochin chickens in Italy: a case report. Vet. Ital. 58(1), 117–124. Mete, A., Gharpure, R., Pitesky, M.E., Famini, D., Sverlow, K. and Dunn, J. 2016. Marek’s Disease in Backyard Chickens, A Study of Pathologic Findings and Viral Loads in Tumorous and Nontumorous Birds. Avian Dis. 60(4), 826–836. Mohamed, M.H.A., El-Sabagh, I.M., Al-Habeeb, M.A. and Al-Hammady, Y.M. 2016. Diversity of Meq gene from clinical Marek’s disease virus infection in Saudi Arabia. Vet. World 9(6), 572–578. Morgan, R., Anderson, A., Bernberg, E., Kamboj, S., Huang, E., Lagasse, G., Isaacs, G., Parcells, M., Meyers, B.C., Green, P.J. and Burnside, J. 2008. Sequence conservation and differential expression of Marek’s disease virus microRNAs. J. Virol. 82(24), 12213–12220. Mpenda, F.N., Schilling, M.A., Campbell, Z., Mngumi, E.B. and Buza, J. 2019. The genetic diversity of local african chickens: a potential for selection of chickens resistant to viral infections. J. Appl. Poult. Res. 28(1), 1–12. Murthy, K.K. and Calnek, B.W. 1979. Pathogenesis of Marek’s disease; effect of immunization with inactivated viral and tumor-associated antigens. Infect. Immun. 26(2), 547–553. Nair. and V. 2005. Evolution of Marek’s disease -- a paradigm for incessant race between the pathogen and the host. Vet. J. 170(2), 175–183. Neerukonda, S.N., Katneni, U.K., Bhandari, N. and Parcells, M.S. 2019. Transcriptional Analyses of Innate and Acquired Immune Patterning Elicited by Marek’s Disease Virus Vaccine Strains: turkey Herpesvirus (HVT), Marek’s Disease Virus 2 (strain SB1), and Bivalent Vaccines (HVT/SB1 and HVT-LT/SB1). Avian Dis. 63(4), 670–680. Neve, J., Patel, R., Wang, Z., Louey, A. and Furger, A.M. 2017. Cleavage and polyadenylation: ending the message expands gene regulation. RNA Biol. 14(7), 865–890. Ongor, H., Timurkaan, N., Abayli, H., Karabulut, B., Kalender, H., Tonbak, S., Eroksuz, H. and Çetinkaya, B. 2022. First report of Serotype-1 Marek’s disease virus (MDV-1) with oncogenic form in backyard turkeys in Turkey: a molecular analysis study. BMC Vet. Res. 18(1), 30. Osterrieder, N., Kamil, J.P., Schumacher, D., Tischer, B.K. and Trapp, S. 2006. Marek’s disease virus: from miasma to model. Nat. Rev. Microbiol. 4(4), 283–294. Othman, I. and Aklilu, E. 2019. Marek’s disease herpesvirus serotype 1 in broiler breeder and layer chickens in Malaysia. Vet. World 12(3), 472–476. Payne, L.N. and Biggs, P.M. 1967. Studies on Marek’s disease. II. Pathogenesis. J. Natl. Cancer Inst. 39(2), 281–302. Payne, L.N. and Venugopal, K. 2000. Neoplastic diseases: marek’s disease, avian leukosis and reticuloendotheliosis. Rev. Sci. Tech. 19(2), 544–564. Peebles, E.D., Barbosa, T.M., Cummings, T.S., Dickson, J., Womack, S.K. and Gerard, P.D. 2017. Comparative effects of in ovo versus subcutaneous administration of the Marek’s disease vaccine and pre-placement holding time on the processing yield of Ross 708 broilers. Poult. Sci. 96(11), 3944–3948. Purchase, H.G. 1969. Immunofluorescence in the study of Marek’s disease. I. Detection of antigen in cell culture and an antigenic comparison of eight isolates. J. Virol. 3(6), 557–565. Ralapanawe, S., Walkden-Brown, S.W., Renz, K.G. and Islam, A.F.M.F. 2016. Protection provided by Rispens CVI988 vaccine against Marek’s disease virus isolates of different pathotypes and early prediction of vaccine take and MD outcome. Avian Pathol. 45(1), 26–37. Ravikumar, R., Chan, J. and Prabakaran, M. 2022. Vaccines against Major Poultry Viral Diseases: strategies to Improve the Breadth and Protective Efficacy. Viruses 14(6), 1195. Reddy, S.M., Izumiya, Y. and Lupiani, B. 2017. Marek’s disease vaccines: current status, and strategies for improvement and development of vector vaccines. Vet. Microbiol. 206(1), 113–120. Reddy, Y.V.P., Kumar, A.A., Amaravathi, P., Rayulu, V.C. and Suryanarayana, M.V.A.N. 2021. Clinico-pathological and Immunocytochemical studies on Marek’s Disease in poultry. Pharma. Innov. J. 10(3), 610–615. Renz, K.G., Cooke, J., Clarke, N., Cheetham, B.F., Hussain, Z., Fakhrul Islam, A.F.M., Tannock, G.A. and Walkden-Brown, S.W. 2012. Pathotyping of Australian isolates of Marek’s disease virus and association of pathogenicity with meq gene polymorphism. Avian Pathol. 41(2), 161–176. Rixon. 1993. Structure and assembly of herpesviruses. Semin. Virol. 4(3), 135–144. Rong, S., Wheeler, D. and Weber, F. 2014. Efficient Marek’s disease virus (MDV) and herpesvirus of turkey infection of the QM7 cell line that does not contain latent MDV genome. Avian Pathol. 43(5), 414–419. Sadigh, Y., Powers, C., Spiro, S., Pedrera, M., Broadbent, A. and Nair, V. 2018. Gallid herpesvirus 3 SB-1 strain as a recombinant viral vector for poultry vaccination. NPJ. Vaccines 3(1), 21. Sadigh, Y., Tahiri-Alaoui, A., Spatz, S., Nair, V. and Ribeca, P. 2020. Pervasive Differential Splicing in Marek’s Disease Virus can Discriminate CVI-988 Vaccine Strain from RB-1B Very Virulent Strain in Chicken Embryonic Fibroblasts. Viruses 12(3), 329. Sakamoto, S., Putalun, W., Vimolmangkang, S., Phoolcharoen, W., Shoyama, Y., Tanaka, H. and Morimoto, S. 2018. Enzyme-linked immunosorbent assay for the quantitative/qualitative analysis of plant secondary metabolites. J. Nat. Med. 72(1), 32–42. Schat, K.A. 2022. The Importance of the Bursa of Fabricius, B Cells and T Cells for the Pathogenesis of Marek’s Disease: a Review. Viruses 14(9), 2015. Schat, K.A. and Skinner, M.A. 2014. Avian Immunosuppressive Diseases and Immunoevasion. Avian Immunol. 1(1), 275–297. Schat, K.A., Calnek, B.W. and Fabricant, J. 1981. Influence of the bursa of Fabricius on the pathogenesis of Marek’s disease. Infect. Immun. 31(1), 199–207. Scholten, R., Hilgers, L.A.T., Jeurissen, S.H.M. and Weststrate, M.W. 1990. Detection of Marek’s disease virus antigen in chickens by a novel immunoassay. J. Virol. Methods 27(2), 221–226. Singh, S.D., Barathidas, R., Kumar, A., Deb, R., Verma, A.K. and Dhama, K. 2012. Recent trends in diagnosis and control of Marek’s disease (MD) in poultry. Pak. J. Biol. Sci. 15(20), 964–970. Song, B., Zeb, J., Hussain, S., Aziz, M.U., Circella, E., Casalino, G., Camarda, A., Yang, G., Buchon, N. and Sparagano, O. 2022. A Review on the Marek’s Disease Outbreak and Its Virulence-Related meq Genovariation in Asia between 2011 and 2021. Animals (Basel). 12(5), 540. Swayne, D.E., Fletcher, O.J., Tyler, D.E. and Page, R.K. 1989. Marek’s disease virus-induced transient paralysis in chickens. 3. Differentiation of field cases from classical Marek’s disease by central nervous system lesions. Avian Pathol. 18(3), 413–431. Szeleszczuk, P., Samorek-Salamonowicz, E., Kozdruñ, W., Król, K., Malicka, E. and Karpiñska, E. 2006. Pierwszy przypadek syndromu Alabama Red Leg u brojlerów w Polsce. Med. Wet. 62(12), 1391–1394. Tai, S.H.S., Hearn, C., Umthong, S., Agafitei, O., Cheng, H.H., Dunn, J.R. and Niikura, M. 2017. Expression of Marek’s Disease Virus Oncoprotein Meq During Infection in the Natural Host. Virology 503(1), 103–113. Thanthrige-Don, N., Abdul-Careem, M.F., Shack, L.A., Burgess, S.C. and Sharif, S. 2009. Analyses of the spleen proteome of chickens infected with Marek’s disease virus. Virology 390(2), 356–367. Tien, Y.T., Akbar, H. and Jarosinski, K.W. 2023. Temperature-induced reactivation of Marek’s disease virus-transformed T cells ex vivo. Front. Vet. Sci. 10(1), 1145757. Torres, A., Marin, S., Costa, C. and Martins, N. 2019. An Overview on Marek’s Disease Virus Evolution and Evidence for Increased Virulence in Brazil. Braz. J. Poult. Sci. 21(1), 1–9. Trapp-Fragnet, L., Schermuly, J., Kohn, M., Bertzbach, L.D., Pfaff, F., Denesvre, C., Kaufer, B.B. and Härtle, S. 2021. Marek’s disease virus prolongs survival of primary chicken B-cells by inducing a senescence-like phenotype. PLos Pathog. 17(10), e1010006. Trimpert, J., Groenke, N., Jenckel, M., He, S., Kunec, D., Szpara, M.L., Spatz, S.J., Osterrieder, N. and Mcmahon, D.P. 2017. A phylogenomic analysis of Marek’s disease virus reveals independent paths to virulence in Eurasia and North America. Evol. Appl. 10(10), 1091–1101. Tuli, N.F. and Fayisa, W.O. 2023. Review on Virulence and Diagnostic Methods of Mareks Disease Virus in Chickens. J. Intern. Med. Res. Rep. 2(3), 1–7. Van Der Walle, N. and Winkler-Junius, E. 1924. De Neuritisepizootie bij Kippen te Barnveld in 1921. T. Vergelijk. Geneesk. Gezondhleer. 10(1), 34–50. Viet Thu, H.T., Trang, H.N., Phuoc Chien, N.T., Ngu, N.T. and Hien, N.D. 2022. Occurrence of Marek’s Disease in Backyard Chicken Flocks in Vietnam. Avian Dis. 66(2), 230–236. Volkening, J.D., Spatz, S.J., Ponnuraj, N., Akbar, H., Arrington, J.V., Vega-Rodriguez, W. and Jarosinski, K.W. 2023. Viral proteogenomic and expression profiling during productive replication of a skin-tropic herpesvirus in the natural host. PLos Pathog. 19(6), e1011204. Vychodil, T., Wight, D.J., Nascimento, M., Jolmes, F., Korte, T., Herrmann, A. and Kaufer, B.B. 2022. Visualization of Marek’s Disease Virus Genomes in Living Cells during Lytic Replication and Latency. Viruses 14(2), 287. Wannaratana, S., Tunterak, W., Prakairungnamthip, D., Sasipreeyajan, J. and Thontiravong, A. 2022. Genetic characterization of Marek’s disease virus in chickens in Thailand reveals a high genetic diversity of circulating strains. Transbound. Emerg. Dis. 69(6), 3771–3779. Williams, C.J. and Hopkins, B.A. 2011. Field evaluation of the accuracy of vaccine deposition by two different commercially available in ovo injection systems. Poult. Sci. 90(1), 223–226. WOAH. 2023. WOAH Terrestrial Manual, France. World Organisation for Animal Health, chapter 3.3.13: Marek’s Disease. pp. 1–15. Woźniakowski, E. and Samorek-Salamonowicz. 2014. Direct detection of Marek’s disease virus in poultry dust by loop-mediated isothermal amplification. Arch. Virol. 159(11), 3083–3087. Woźniakowski, G., Frant, M. and Mamczur, A. 2018. Avian Reticuloendotheliosis in Chickens - An Update on Disease Occurrence and Clinical Course. J. Vet. Res. 62(3), 257–260. Yang, Y., Dong, M., Hao, X., Qin, A. and Shang, S. 2020. Revisiting cellular immune response to oncogenic Marek’s disease virus: the rising of avian T-cell immunity. Cellular Mol. Life Sci. 77(16), 3103–3116. You, Y., Conradie, A.M., Kheimar, A., Bertzbach, L.D. and Kaufer, B.B. 2021. The Marek’s Disease Virus Unique Gene MDV082 Is Dispensable for Virus Replication but Contributes to a Rapid Disease Onset. J. Virol. 95(15), 13121. You, Y., Hagag, I.T., Kheimar, A., Bertzbach, L.D. and Kaufer, B.B. 2021. Characterization of a Novel Viral Interleukin 8 (vIL-8) Splice Variant Encoded by Marek’s Disease Virus. Microorganisms 9(7), 1475. You, Y., Vychodil, T., Aimola, G., Previdelli, R.L., Göbel, T.W., Bertzbach, L.D. and Kaufer, B.B. 2021. A Cell Culture System to Investigate Marek’s Disease Virus Integration into Host Chromosomes. Microorganisms 9(12), 2489. Yu, Z.H., Zhang, Y.P., Lan, X.G., Wang, Y.N., Guo, R.R., Li, K., Gao, L., Qi, X.L., Cui, H.Y., Wang, X.M., Gao, Y.L. and Liu, C.J. 2023. Differences in Pathogenicity and Vaccine Resistance Discovered between Two Epidemic Strains of Marek’s Disease Virus in China. Viruses 15(4), 945. Zeghdoudi, M., Latifa, M., Seloua, M., Rima, S., Mardja, T. and Aoun, L. 2023. Updating of epidemiological and pathological features of Marek’s disease in laying hens and broilers. Vet. Med-Czech. 68(11), 443–448. Zelnik, V., Harlin, O., Fehler, F., Kaspers, B., Göbel, T.W., Nair, V.K. and Osterrieder, N. 2004. An enzyme-linked immunosorbent assay (ELISA) for detection of Marek’s disease virus-specific antibodies and its application in an experimental vaccine trial. J. Vet. Med. B. Infect. Dis. Vet. Public Health 51(2), 61–67. Zhang, L., Zhu, C., Heidari, M., Dong, K., Chang, S., Xie, Q. and Zhang, H. 2020. Marek’s disease vaccines-induced differential expression of known and novel microRNAs in primary lymphoid organ bursae of White Leghorn. Vet. Res. 51(1), 19. Zhang, Y., Tang, N., Luo, J., Teng, M., Moffat, K., Shen, Z., Watson, M., Nair, V. and Yao, Y. 2019. Marek’s Disease Virus-Encoded MicroRNA 155 Ortholog Critical for the Induction of Lymphomas Is Not Essential for the Proliferation of Transformed Cell Lines. J. Virol. 93(17), e713–e719. Zhang, Y., Tang, N., Sadigh, Y., Baigent, S., Shen, Z., Nair, V. and Yao, Y. 2018. Application of CRISPR/Cas9 Gene Editing System on MDV-1 Genome for the Study of Gene Function. Viruses 10(6), 279. Zhang, Z., Liu, S., Ma, C., Zhao, P. and Cui, Z. 2015. Absolute quantification of a very virulent Marek’s disease virus dynamic quantity and distributions in different tissues. Poult. Sci. 94(6), 1150–1157. Zhao, P., Li, X.J., Teng, M., Dang, L., Yu, Z.H., Chi, J.Q., Su, J.W., Zhang, G.P. and Luo, J. 2015. In vivo expression patterns of microRNAs of Gallid herpesvirus 2 (GaHV-2) during the virus life cycle and development of Marek’s disease lymphomas. Virus Genes 50(2), 245–252. Zhou, X., Wu, S., Zhou, H., Wang, M., Wang, M., Lü, Y., Cheng, Z., Xu, J. and Ai, Y. 2019. Marek’s Disease Virus Regulates the Ubiquitylome of Chicken CD4+ T Cells to Promote Tumorigenesis. Int. J. Mol. Sci. 20(9), 2089. Zhu, Z.J., Teng, M., Liu, Y., Chen, F.J., Yao, Y., Li, E.Z. and Luo, J. 2024. Immune escape of avian oncogenic Marek’s disease herpesvirus and antagonistic host immune responses. NPJ. Vaccines. 9(1), 109. Žlabravec, Z., Slavec, B., Rožmanec, E., Koprivec, S., Dovč, A. and Zorman Rojs, O. 2024. First Report of Marek’s Disease Virus in Commercial Turkeys in Slovenia. Animals 14(2), 250. | ||
| How to Cite this Article |
| Pubmed Style Mulyati S, Mustofa I, Khairullah AR, Kusala MKJ, Fauziah I, Ahmad RZ, Ekawasti F, Prihandani SS, Furqoni AH, Wardhani BWK, Kurniasih DAA, Khalisa AT, Moses IB, Akintunde AO, Pratama BP, Raharjo PP, Fauzia KA, Wibowo S. Marek’s disease: A global challenge to poultry health and productivity. Open Vet. J.. 2025; 15(10): 4797-4813. doi:10.5455/OVJ.2025.v15.i10.2 Web Style Mulyati S, Mustofa I, Khairullah AR, Kusala MKJ, Fauziah I, Ahmad RZ, Ekawasti F, Prihandani SS, Furqoni AH, Wardhani BWK, Kurniasih DAA, Khalisa AT, Moses IB, Akintunde AO, Pratama BP, Raharjo PP, Fauzia KA, Wibowo S. Marek’s disease: A global challenge to poultry health and productivity. https://www.openveterinaryjournal.com/?mno=267956 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.2 AMA (American Medical Association) Style Mulyati S, Mustofa I, Khairullah AR, Kusala MKJ, Fauziah I, Ahmad RZ, Ekawasti F, Prihandani SS, Furqoni AH, Wardhani BWK, Kurniasih DAA, Khalisa AT, Moses IB, Akintunde AO, Pratama BP, Raharjo PP, Fauzia KA, Wibowo S. Marek’s disease: A global challenge to poultry health and productivity. Open Vet. J.. 2025; 15(10): 4797-4813. doi:10.5455/OVJ.2025.v15.i10.2 Vancouver/ICMJE Style Mulyati S, Mustofa I, Khairullah AR, Kusala MKJ, Fauziah I, Ahmad RZ, Ekawasti F, Prihandani SS, Furqoni AH, Wardhani BWK, Kurniasih DAA, Khalisa AT, Moses IB, Akintunde AO, Pratama BP, Raharjo PP, Fauzia KA, Wibowo S. Marek’s disease: A global challenge to poultry health and productivity. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4797-4813. doi:10.5455/OVJ.2025.v15.i10.2 Harvard Style Mulyati, S., Mustofa, . I., Khairullah, . A. R., Kusala, . M. K. J., Fauziah, . I., Ahmad, . R. Z., Ekawasti, . F., Prihandani, . S. S., Furqoni, . A. H., Wardhani, . B. W. K., Kurniasih, . D. A. A., Khalisa, . A. T., Moses, . I. B., Akintunde, . A. O., Pratama, . B. P., Raharjo, . P. P., Fauzia, . K. A. & Wibowo, . S. (2025) Marek’s disease: A global challenge to poultry health and productivity. Open Vet. J., 15 (10), 4797-4813. doi:10.5455/OVJ.2025.v15.i10.2 Turabian Style Mulyati, Sri, Imam Mustofa, Aswin Rafif Khairullah, Muhammad Khaliim Jati Kusala, Ima Fauziah, Riza Zainuddin Ahmad, Fitrine Ekawasti, Sri Suryatmiati Prihandani, Abdul Hadi Furqoni, Bantari Wisynu Kusuma Wardhani, Dea Anita Ariani Kurniasih, Andi Thafida Khalisa, Ikechukwu Benjamin Moses, Adeyinka Oye Akintunde, Bima Putra Pratama, Prima Puji Raharjo, Kartika Afrida Fauzia, and Syahputra Wibowo. 2025. Marek’s disease: A global challenge to poultry health and productivity. Open Veterinary Journal, 15 (10), 4797-4813. doi:10.5455/OVJ.2025.v15.i10.2 Chicago Style Mulyati, Sri, Imam Mustofa, Aswin Rafif Khairullah, Muhammad Khaliim Jati Kusala, Ima Fauziah, Riza Zainuddin Ahmad, Fitrine Ekawasti, Sri Suryatmiati Prihandani, Abdul Hadi Furqoni, Bantari Wisynu Kusuma Wardhani, Dea Anita Ariani Kurniasih, Andi Thafida Khalisa, Ikechukwu Benjamin Moses, Adeyinka Oye Akintunde, Bima Putra Pratama, Prima Puji Raharjo, Kartika Afrida Fauzia, and Syahputra Wibowo. "Marek’s disease: A global challenge to poultry health and productivity." Open Veterinary Journal 15 (2025), 4797-4813. doi:10.5455/OVJ.2025.v15.i10.2 MLA (The Modern Language Association) Style Mulyati, Sri, Imam Mustofa, Aswin Rafif Khairullah, Muhammad Khaliim Jati Kusala, Ima Fauziah, Riza Zainuddin Ahmad, Fitrine Ekawasti, Sri Suryatmiati Prihandani, Abdul Hadi Furqoni, Bantari Wisynu Kusuma Wardhani, Dea Anita Ariani Kurniasih, Andi Thafida Khalisa, Ikechukwu Benjamin Moses, Adeyinka Oye Akintunde, Bima Putra Pratama, Prima Puji Raharjo, Kartika Afrida Fauzia, and Syahputra Wibowo. "Marek’s disease: A global challenge to poultry health and productivity." Open Veterinary Journal 15.10 (2025), 4797-4813. Print. doi:10.5455/OVJ.2025.v15.i10.2 APA (American Psychological Association) Style Mulyati, S., Mustofa, . I., Khairullah, . A. R., Kusala, . M. K. J., Fauziah, . I., Ahmad, . R. Z., Ekawasti, . F., Prihandani, . S. S., Furqoni, . A. H., Wardhani, . B. W. K., Kurniasih, . D. A. A., Khalisa, . A. T., Moses, . I. B., Akintunde, . A. O., Pratama, . B. P., Raharjo, . P. P., Fauzia, . K. A. & Wibowo, . S. (2025) Marek’s disease: A global challenge to poultry health and productivity. Open Veterinary Journal, 15 (10), 4797-4813. doi:10.5455/OVJ.2025.v15.i10.2 |