| Research Article | ||
Open Vet. J.. 2026; 16(2): 999-1011 Open Veterinary Journal, (2026), Vol. 16(2): 999-1011 Research Article Co-occurrence of Francisella-like endosymbionts and Candidatus Midichloria mitochondrii with Theileria annulata in Al-Diwaniyah, Iraq, Hyalomma ticksAzhar Alrikabi1*and Alaa Mohammed Abdulrazaq Al-khafaji21Polytechnic College, Al-Qadisiyah, Al-Furat Al-Awsat Technical University, Diwanyiah, Iraq 2College of Veterinary Medicine, University of Al-Qadisiyah, Diwaniyah, Iraq *Corresponding Author: Azhar Alrikabi. Polytechnic College, Al-Qadisiyah, Al-Furat Al-Awsat Technical University, Diwanyiah, Iraq. Email: Azhar.bahidh [at] atu.edu.iq Submitted: 05/07/2025 Revised: 17/12/2025 Accepted: 05/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
AbstractBackground: Ticks are significant vectors of pathogens, and their nonpathogenic intracellular bacteria, known as endosymbionts, influence tick physiology and may interact with tick-borne pathogens. Such relationships can affect transmission dynamics, and understanding them is essential for designing strategies that control tick-borne diseases and reduce their impact on human and animal health. Aim: This study aimed to determine the prevalence and density of two key endosymbiotic bacteria, Francisella-like endosymbiont (FLE) and Candidatus Midichloria mitochondrii (Ca. M. mitochondrii), and to investigate possible correlations between these symbionts and the protozoan parasite Theileria annulata in Hyalomma anatolicum ticks. Methods: Between January and October 2024, 116 ticks were collected from cattle in the Diwaniyah province, Iraq. The morphological examination was followed by molecular confirmation using mitochondrial 16S rRNA sequencing. Quantitative polymerase chain reaction was applied to detect and quantify FLE, Ca. M. mitochondrii, and T. annulata in H. anatolicum. Results: Hyalomma anatolicum was the predominant species, representing 86% of ticks, whereas H. excavatum accounted for 13%. Mitochondrial 16S rRNA sequences confirmed species identity and showed 99.48%–100% similarity to reference isolates from China, Pakistan, Turkey, India, and Russia. FLE was detected in 100% of H. anatolicum, with mean loads of 7.9 × 103 copies per tick. Ca. M. mitochondrii was detected in 99% of specimens, with mean loads of 3.7×102 copies per tick. FLE densities tended to be higher in females compared to males (9.7 × 10³ copies vs. 1.8 × 10³ copies), but the difference was not statistically significant. Candidatus Midichloria mitochondrii loads also differed significantly (4.8 × 10² copies vs. 1.3 × 10¹ copies). Theileria annulata was present in all ticks, with the highest copy numbers in semi-engorged females (1.9 × 104 copies), followed by males (2.8 × 10³ copies). Our findings revealed a statistically significant association (p < 0.05) between T. annulata and Ca. M. mitochondrii, whereas no significant correlation was observed between T. annulata and FLE. Conclusion: The presence of FLE and Ca. M. mitochondrii in H. anatolicum from the Diwaniyah province was documented. Evidence indicated associations between symbiont density and T. annulata infection, and the results point toward a possible role of endosymbiotic bacteria in pathogen persistence and the epidemiology of tick-borne diseases. Keywords: 16S rRNA, Endosymbionts, Hyalomma, Francisella, Ticks. IntroductionTicks are obligate blood-feeding ectoparasites of animals and humans and rank just behind mosquitoes as vectors of pathogens with major implications for human and veterinary health (de la Fuente et al., 2008). Their long feeding periods and multi-host life cycles enable them to transmit diverse protozoan, bacterial, and viral pathogens. Infestations in livestock cause direct harm through blood loss, skin damage, and reduced productivity, whereas tick-borne pathogens reduce milk yield, impair growth, and increase mortality (Gomes et al., 2018). Global losses due to ticks are estimated at US$22–30 billion per year. In South Asia alone, the annual economic impact of ticks and tick-borne diseases is approximately US$800 million (Nandi et al.,2023). Among the ixodid ticks, Hyalomma anatolicum is recognized as one of the most significant species. Its wide distribution across the Middle East, North Africa, and South Asia, coupled with its adaptability to varied environments, explains its prominence in tick-borne disease transmission (Shyma et al., 2012; Biglari et al., 2018). The species transmits several pathogens of veterinary and zoonotic concern, including Theileria annulata, T. equi, T. lestoquardi, Anaplasma spp., Rickettsia spp., and Crimean-Congo hemorrhagic fever virus (Celina et al., 2025). Along with Rhipicephalus microplus, it is one of the most economically damaging tick species for cattle in endemic regions (Gomes et al., 2018). Climate change and land use shifts are expanding the range and seasonality of Hyalomma ticks, which increases the risk of pathogen transmission in previously unaffected areas (Nasiri et al., 2025). Theileria annulata-induced tropical theileriosis is a major constraint on cattle production in endemic areas. The parasite infects leukocytes and erythrocytes, resulting in fever, lymph node enlargement, anemia, and potentially fatal outcomes. Even when animals survive, production losses through reduced milk yield, poor growth, and infertility are severe (Gomes et al., 2018). An estimated 250 million cattle are at risk worldwide (Afshari et al., 2021). In Iraq, T. annulata remains endemic in the central and southern provinces, with a prevalence ranging from 40% to more than 80%, depending on the region and diagnostic method (Mohammad Al-Saeed et al., 2010; Al-Fatlawi et al., 2018; Kawan, 2019; Al-Wasmee et al., 2024). Neighboring countries report comparable rates, underscoring the transboundary threat (Ali et al., 2013; Köseoğlu et al., 2021; Omer et al., 2021). These burdens highlight the need for enhanced control strategies beyond traditional acaricides, especially as resistance to chemical treatments is increasingly reported (Shyma et al., 2012). Ticks harbor diverse microbial communities that influence their physiology and association with pathogens. Obligate symbionts such as Francisella-like endosymbiont (FLE) and facultative symbionts such as Candidatus Midichloria mitochondrii are frequently detected in Hyalomma ticks. These bacteria supplement essential nutrients absent from vertebrate blood, contribute to reproduction, and may affect survival (Sassera et al., 2008; Bonnet and Pollet, 2021). FLE prevalence has been reported to exceed 90% in Hyalomma populations (Ghafar et al., 2020; Perveen et al., 2020; Soliman et al., 2025). Candidatus Midichloria mitochondrii has strong vertical transmission and the mitochondrial localization, indicating a fundamental role in ovarian developmental processes, as supported by previous studies (Olivieri et al., 2019; Luo et al., 2024). Endosymbionts can alter vector competence. In Dermacentor ticks, Rickettsia peacockii is associated with reduced infection by the pathogenic R. rickettsii (Wójcik-Fatla et al., 2015). Conversely, in Hyalomma species, FLEs have been associated with a higher prevalence of T. annulata and Rickettsia, suggesting synergistic effects rather than protection (Hussain et al., 2022; Sesmero-García et al., 2023). Some studies report co-infection rates close to 50%, implying that FLEs may enhance rather than limit pathogen persistence. Co-occurrence may involve nutrient competition, immune modulation, or changes to tissue barrier permeability (Kolo and Raghavan, 2023; Zhong et al., 2024). Understanding these mechanisms is essential for interpreting pathogen dynamics in field populations. The role of symbionts in tick biology has piqued interest in microbiome-based control strategies. Inspired by the success of Wolbachia in mosquitoes, research is exploring ways to disrupt essential tick symbionts or introduce beneficial symbionts that antagonize pathogens. Although still experimental, such approaches could complement existing methods by targeting microbial pathways essential for tick survival or pathogen transmission Adegoke et al., 2020; Bonnet and Pollet, 2021). Hyalomma ticks remain underexplored in this context, despite evidence of their heavy reliance on FLEs and Ca. M. mitochondrii. Despite the endemicity of tropical theileriosis in Iraq, there remains limited knowledge regarding the symbiont composition of H. anatolicum. An assessment of the prevalence and densities of FLE and Ca. M. mitochondrii in field-collected ticks, along with their correlation to T. annulata infection, will clarify their epidemiological roles. Such information is vital for improving molecular diagnostics, understanding co-infection dynamics, and guiding innovative vector management strategies. Al-Diwaniyah is one of Iraq’s major cattle-producing provinces where tropical theileriosis remains endemic. Its humid subtropical conditions and year-round vector activity make it a suitable region for assessing endosymbiont–pathogen relationships. This study investigated the prevalence and density of FLEs and Ca. M. mitochondrii in H. anatolicum ticks collected from cattle in Diwaniyah. It also examines the occurrence of T. annulata and explores potential associations between the pathogen and endosymbionts. This study seeks to provide insights into microbial–pathogen associations that may inform more effective approaches to the control of tropical theileriosis. Materials and MethodsTick collectionBetween January and October 2024, 116 ticks were collected from naturally infested cattle in Iraq’s Diwaniyah province. Cattle were randomly selected from different farms. To avoid damage to the specimens, ticks were removed manually using sterile forceps and immediately placed individually in sterile plastic containers containing 70% ethanol. Each container was labeled with the date of collection, host information, and locality. Samples were transported in an icebox to the Department of Parasitology, College of Veterinary Medicine, University of Al-Qadisiyah, for laboratory processing. Morphological identificationAll ticks were initially identified at the species level using morphological characteristics observed under a stereomicroscope. Identification keys described by Walker (2003) were applied to present the diagnostic features of Hyalomma species, including scutum shape, capitulum shape, coxal spurs, festoon arrangement, and genital apertures (Fig. 1). Morphological identification was the first step before molecular confirmation. DNA extraction from ticksPrior to extraction, the ticks were rinsed three times with distilled water and air-dried on sterile filter paper to remove residual ethanol. Each tick was bisected using sterile scalpels under a stereo microscope. Semi-engorged females were divided into four sections, whereas smaller males were divided into two sections. Each fragment was placed in a sterile 1.5 ml microcentrifuge tube labeled with a unique code. All 100 adult H. anatolicum ticks (76 females and 24 males) were individually processed following identical extraction procedures. DNA was extracted using the ammonium hydroxide boiling method (Ammazzalorso et al., 2015; Al-Khafaji, 2018). A total of 150 µl of 12 M ammonium hydroxide was added to each tube. Samples were incubated in a heating block at 100°C for 20 minutes. Tube caps were opened, and samples were maintained at 100°C for an additional 18 minutes under a fume hood until volumes were reduced to approximately 70–100 µl. The extracts were centrifuged at 10,000 × g for 10 minutes, and the supernatants were collected. The DNA concentration and purity were measured using a Nanodrop spectrophotometer (Thermo Scientific, USA). DNA was stored at −20°C until use.
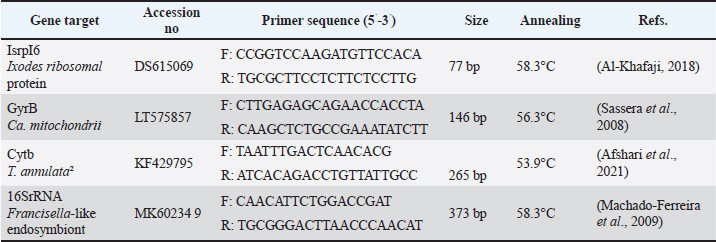
Fig. 1.Morphological detection of H. excavatum (A, B) and H. anatolicum (C, D) adult males by microscopy. (A) Dorsal view of H. excavatum showing the anterior fusion paraparmeral festoons (arch) and the bulge arrows marking the caudal ridges. (B) Ventral view showing the genital pore, ventral plates, and coxae I and IV. (C) Dorsal view showing the fetoons (absence of the anterior arch) of H. anatolicum. (D) Ventral view showing the genital pore, ventral plates (adnal and subanal plate), and coxae I and IV. Molecular identification of ticks (16S rRNA PCR)Tick species were confirmed using PCR amplification of the mitochondrial 16S rRNA gene using Ixodidae-specific primers (Han et al., 2019). The primer sequences were as follows: forward 5′-CTGCTCAATGATTTTTTAAATTGCTGTGG-3′ and reverse 5′-CCGGTCTGAACTCAGATCAAGT-3′. PCR reactions were prepared in 20 µl volumes containing 10 µl PCR Master Mix, 2 µl (0.5 pmol) of each primer, 2 µl of DNA template, and 4 µl of nuclease-free water. Cycling conditions included initial denaturation at 95°C for 3 minutes, followed by 39 cycles of denaturation at 95°C for 35 seconds, annealing at 53.8°C for 35 seconds, and extension at 72°C for 35 seconds, with a final extension at 72°C for 5 minutes. PCR products (~450 bp) were separated by electrophoresis on 1.5% agarose gels stained with ethidium bromide and visualized under UV illumination. DNA sequencing and phylogenetic analysisPositive amplicons were purified and submitted for Sanger DNA sequencing to Macrogen Inc. (South Korea). Raw sequences were trimmed and edited using Chromas software and compared with reference sequences in the National Center for Biotechnology Information GenBank database using BLAST. Phylogenetic analysis was conducted using MEGA 11 software to compare the relationships between tick sequences from Iraq and global reference strains. qPCRReal-time polymerase chain reaction was employed to determine the prevalence and density of FLE, Ca. M. mitochondrii, and T. annulata. Macrogen Inc. designed synthetic plasmids containing specific target genes (Table 1) to generate duplicate standard curves. Plasmids were serially diluted from 107 to 10¹ copies to prepare reference standards. qPCR reactions were performed in 20 µl volumes containing: 10 µl SYBR Green Master Mix (AddBio, Korea), 1 µl of each primer, 3 µl of template DNA, and 5 µl of nuclease-free water. The amplification conditions included an initial denaturation at 95°C for 10 minutes, followed by 40 cycles at 95°C for 20 seconds, annealing at optimized temperatures between 50°C and 60°C for 30 seconds, and extension at 72°C for 30 seconds. A melt curve analysis was conducted from 60°C to 95°C to confirm the specificity of the product. Copy numbers were normalized against the tick single-copy ribosomal protein L6 (rpl6) gene of Ixodes scapularis, previously validated for tick normalization (Al-Khafaji, 2018). Table 1. Gene targets for amplification and sequencing.
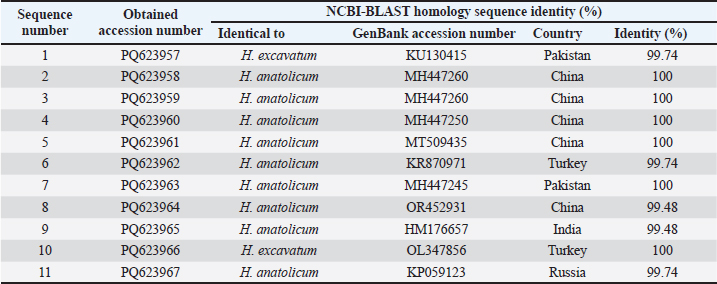
Statistical analysisStatistical analyses were performed using GraphPad Prism (GraphPad Software, version 8.4.3, USA). For data that did not follow a normal distribution, the Kruskal–Wallis test with Dunn’s correction was used to compare the genomic copy number of endosymbionts (FLE, Ca M. mitochondrii) and pathogenic protozoa (T. annulata) in tick samples (semi-engorged female and male). Differences were considered statistically significant at p < 0.05 or p < 0.01. Ethical approvalNot needed for this study. ResultsMorphological and molecular identification of ticksA total of 116 ticks were collected from cattle in the Al-Diwaniyah province and examined under a stereomicroscope. All were identified as Hyalomma ixodid ticks. Among them, 100 (86%) specimens were confirmed as H. anatolicum, while 16 (13%) specimens were identified as H. excavatum. Morphological characteristics of H. anatolicum included smaller body size, thinner form, and paler coloration than other Hyalomma species. Females displayed a shallow cervical field with nearly parallel margins. Males exhibited a less depressed posterior scutum and paracentral festoons that did not converge to form an arch, unlike the pronounced arch in H. excavatum (Fig. 1). The morphological diagnosis was supported by molecular confirmation. PCR amplification of the mitochondrial 16S rRNA gene produced a 450-bp fragment in all ticks. Electrophoresis revealed distinct bands, indicating successful amplification (Fig. 2). Overall, H. anatolicum was the predominant species in the study population and accounted for a significantly higher proportion than H. excavatum (p < 0.01). Sequence similarity and evolutionary relationshipsSequencing of the 16S rRNA fragments produced 11 high-quality sequences, which were deposited in GenBank under accession numbers PQ623957–PQ623967. Sequence alignment showed identities ranging from 99.48% to 100% with reference strains of H. anatolicum. Several Iraqi isolates, including PQ623958, PQ623959, PQ623960, PQ623961, and PQ623963, showed 100% identity with isolates from China and Pakistan. Other sequences closely matched strains reported from Turkey, India, and Russia, with identities above 99.4%. Phylogenetic analysis revealed that Iraqi isolates clustered with global H. anatolicum populations with bootstrap support above 95% (Fig. 3). No significant divergence was observed between the local and foreign sequences. Table 2 provides the details of the accession numbers, sequence identities, and country of origin. The clustering pattern in the phylogenetic tree confirmed that Iraqi H. anatolicum shares a nearly identical genetic profile with populations reported from neighboring and distant countries. Multiple sequence alignment further illustrated the strong conservation of nucleotide positions, highlighting close similarity with Chinese and Pakistani strains and divergence from H. excavatum (Fig. 4).
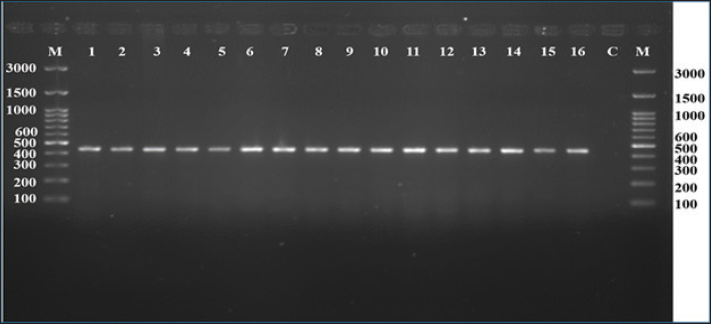
Fig. 2. Agarose gel electrophoresis of mitochondrial 16S rRNA polymerase chain reaction products from Hyalomma ticks. A 450-bp fragment was amplified in all samples (lanes 1–16). Lane C was used as a negative control with no amplification. Lane M contained a 100 bp molecular size marker (GeneDirex, South Korea). Amplification confirmed the DNA quality and molecular identification.
Fig. 3. Phylogenetic tree of H. anatolicum based on partial mitochondrial 16S rRNA sequences. The tree was constructed using the maximum likelihood method (Tamura–Nei model, 100 bootstrap replicates) in MEGA 11. Local sequences (PQ623957–PQ623967) were closely clustered with references from China, Turkey, Pakistan, India, and Russia, showing 99.48%–100% identity. The branch lengths represent the genetic distances. Table 2. Homology sequence identity (%) between local hard ticks and global NCBI-BLAST H. anaolicum and H. excavetum.
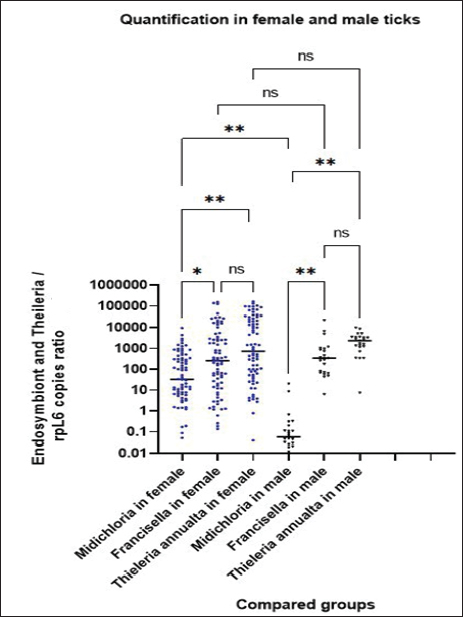
Detection and quantification of endosymbiont bacteriaqPCR assays confirmed the presence of FLE and Ca. M. mitochondrii in H. anatolicum. The standard curves showed amplification efficiencies of 94%–98% and R² values between 0.944 and 0.988, which validated the method. FLE was detected in all examined ticks (100%; n=100) with a mean bacterial load of 7.9 × 10³ copies per tick, whereas Ca. M. mitochondrii was identified in 99% of samples (n=99/100) with a mean copy number of 3.7 × 10². However, the FLE densities were significantly higher than those of Ca. M. mitochondrii (p < 0.05). Female ticks carried higher bacterial loads than males, with mean FLE values of 9.7 × 10³ and 1.8 × 10³, respectively. The mean Ca. M. mitochondrii loads were significantly higher (p < 0.05) in females (4.8 × 10²) than in males (1.3 × 10¹). The highest FLE concentrations were observed in semi-engorged females and males, which exceeded those of Ca. M. mitochondrii (p < 0.05; Fig. 5). Detection and quantification of the protozoan pathogen T. annulataTheileria annulata DNA was detected by qPCR in all H. anatolicum ticks (100%; n=100/100). The standard curves showed amplification efficiencies between 96% and 97%, with R² values above 0.970. The mean copy number of T. annulate was 1.5 × 104. Semi-engorged females carried the highest parasite loads, with mean copy numbers of 1.9 × 103, whereas males carried 2.8 × 103. Although higher values were recorded in semi-engorged females, statistical analysis confirmed that the differences between males and females were not significant (p > 0.01, Fig. 5). Comparative abundance of microorganismsComparative quantitative analysis of endosymbiont bacteria and protozoan copy numbers showed distinct patterns of relative abundance for FLE, T. annulata, and Ca. M. mitochondrii. Theileria annulata exhibited markedly higher density values in both male and female ticks, a trend similarly observed for FLE. In contrast, the lowest levels were observed for Ca. M. mitochondrii (Fig. 5). Statistical analysis revealed a significant positive correlation between T. annulata and Ca. M. mitochondrii (r=0.785; p < 0.01), which may indicate an ecological association. No significant correlation was detected between T. annulata and FLE. DiscussionOur study demonstrated that H. anatolicum was the predominant tick species infesting cattle in AlDiwaniyah province, Iraq, with 86% (100/116) of the collected specimens identified as H. anatolicum and only 13% (16/116) as H. excavatum. This is consistent with the results of previous studies in Iraq (Hasson, 2012; Al-Fatlawi et al., 2018; Al-Abedi and Al-Amery, 2021; Al-Shabbani and Al-Fatlawi, 2023; Al-Zubaidei et al., 2023), Iran (Biglari et al., 2018; Nasiri et al., 2025), India (Shyma et al., 2012), Pakistan (Khan and Ashraf, 2017), and China (Luo et al., 2024). The Hyalomma genus exhibits strong adaptability across diverse climates, including hot, humid, and cold environments, enabling its widespread distribution (Sajid et al., 2018). Notably, the morphologically identified species in the present study were consistent with diagnostic criteria described in Iraqi surveys (Ali et al., 2021; Aziz, 2022; Al-Shabbani and Al-Fatlawi, 2023). However, the accurate morphological identification of Hyalomma ticks is hindered by considerable intraspecific variation and morphological similarities among closely related species, leading to frequent misidentification (Chegeni et al., 2019; Tavakolian et al., 2025). In recent years, molecular techniques utilizing genetic markers have become essential tools for precise species identification and taxonomic resolution. Therefore, our results, including molecular identification, provided essential confirmation. Amplification of the mitochondrial 16S rRNA gene was detected in a specific band (450-bp fragments) in all tested samples, indicating reliable amplification and species-level confirmation. The 11 sequenced isolates (PQ623957–PQ623967) clustered with reference sequences from India, China, Russia, Turkey, and Pakistan, with identities ranging between 99.48% and 100%, according to phylogenetic analysis. The bootstrap values exceeded 1,000%, confirming strong support for clustering. Intraspecific divergence in ticks typically ranges between 0.005 and 0.010 among H. anatolicum populations from different countries, indicating limited regional differentiation. Interspecific comparisons between H. anatolicum and H. excavatum showed higher divergence values (0.015–0.025), confirming their distinct taxonomic status within the genus. Comparable conservation of mitochondrial sequences has been described in related studies from Asia and Europe, where H. anatolicum populations exhibited minimal divergence across regions (Boyer et al., 2019; Chikufenji et al., 2024; Ngnindji-Youdje et al., 2025). Understanding the identification and distribution of tick species is essential for managing the diseases they transmit, as different species carry specific pathogens. This knowledge is essential for formulating successful disease prevention, diagnosis, and control strategies (Boyer et al., 2019; Chikufenji et al., 2024; Ngnindji-Youdje et al., 2025). The Hyalomma genus, comprising more than 20 species, exemplifies vector specificity, predominantly inhabiting Asia, Europe, and Africa. These ticks are recognized as significant vectors for various pathogens, such as Theileria spp. Babesia spp. Anaplasma spp., Ehrlichia spp., Rickettsia spp., Francisella tularensis, Coxiella burnetii, and Borrelia turcica (Olivieri et al., 2019; Adegoke et al., 2020; Mohammed et al., 2023; Tukmechi et al., 2024; Celina et al., 2025; Tavakolian et al., 2025). Recent research has highlighted the complex microbial communities within ticks, including endosymbionts, that can influence tick biology and pathogen transmission dynamics. The FLE endosymbiont is frequently found in Hyalomma ticks (Soliman et al., 2025). Real-time PCR assays in this study revealed a universal FLE infection. All 100 H. anatolicum specimens carried FLE, indicating 100% prevalence, with mean copy numbers of 7.9 × 103. The universal presence of FLE is consistent with earlier reports where the prevalence of FLE approached 100% in Hyalomma populations from Pakistan, North Africa, and the Middle East (Azagi et al., 2017; Ghafar et al., 2020; Benyedem et al., 2022; Soliman et al., 2025). In comparison, FLE prevalence in Ixodes and Dermacentor rarely exceeds 30% (Scoles, 2004; de Carvalho et al., 2011; Wójcik-Fatla et al., 2015). The variations in the prevalence of FLE observed in the present study compared with earlier research can be attributed to several factors, including geographic influences, tick species diversity, sampling techniques, and environmental changes (Zając et al., 2023; Soliman et al., 2025). In this investigation, Ca. M. mitochondrii was the second most prevalent endosymbiotic bacteria in 99 % of samples, with mean copy numbers of 3.7 × 102. The detection of Ca. M. mitochondrii in H. anatolicum agrees with findings from China, where Luo et al. (2024) reported similar prevalence in Xinjiang populations, and with results of Hoffman et al. (2022), who documented a prevalence surpassing 90% in other Hyalomma ticks. Previous work on Ixodes ricinus confirmed the nearly universal detection of Ca. M. mitochondrii in female ovaries, highlighting its strong association with reproductive tissues and vertical transmission (Sassera et al., 2008; Buysse et al., 2021; Uzum et al., 2023). Our findings demonstrated a significantly higher density of FLE in tick samples than in Ca. M mitochondrii. Previous studies have indicated that the presence and role of Ca. M. mitochondrii in Hyalomma ticks exhibit notable variability compared with the more consistent presence of FLE. Although Ca. M. mitochondrii is recognized as an endosymbiont in various tick species, its association with Hyalomma appears less stable and potentially secondary, influenced by evolutionary dynamics and ecological factors (Buysse and Duron, 2018; Buysse et al., 2021). Despite the presence of FLE and Ca. M mitochondrii in males and females of H. anatolicum, but different in density and mean copy numbers of FLE in females (semi-engorged) and males (9.7 × 103 vs. 1.8 × 103), respectively. While Ca. M. mitochondrii mean copy numbers showed (4.8 × 102 vs. 1.3 × 10). The variation in the abundance of endosymbiotic bacteria between male and female ticks indicates that these microbes play sex-specific roles and influence reproduction, development, and disease transmission. The gut microbiomes of male and female ticks exhibit significant taxonomic and functional differences, which may reflect their distinct lifestyles and reproductive strategies (Floriano et al., 2023; Shamoon-Pour et al., 2025). The identification of H. anatolicum as a prevalent tick species infesting cattle in Iraq raises significant concerns regarding its potential role in transmitting Theileria species (Al-Mayah and Abdul-Karim, 2020). Based on the current study’s findings, the T. annulata infection had the highest prevalence (n=100/100) in H. anatolicum, which is consistent with previous studies in Iraq, including 62.2% in Al-Diwaniyah (Albayati, et al., 2023), 40% in Babylon (Al-Wasmee et al., 2024), and 88% in Baghdad (Kawan, 2019). Neighboring countries reported 39.9% in Iran (Afshari et al., 2021), 16% in Turkey (Köseoğlu et al., 2021), 40% in Saudi Arabia (Omer et al., 2021), and 50% in Pakistan (Ali et al., 2013). Mean parasite loads in semi-engorged females (1.9 × 104 copies), followed by males (2.8 × 103 copies). The difference was not statistically significant. Ticks are significant vectors of various pathogens that transmit diseases to both humans and animals. Their ability to transmit pathogens, known as vector competence, is a complex trait influenced by multiple factors, including the presence of symbiotic microorganisms within their tissues. These endosymbionts can establish diverse relationships with their tick hosts, ranging from mutualistic to parasitic, and can profoundly impact tick physiology, immunity, and co-infecting pathogen development (Kolo and Raghavan, 2023; Zhong et al., 2024). The result indicated a significant relationship between T. annulata and Ca. M. mitochondrii. The parallel increase in their densities indicates potential biological cooperation within the tick host. Such correlation may reflect metabolic or immunological associations that enable both microorganisms to persist more efficiently and refer to a complex symbiotic balance that influences H. anatolicum pathogen dynamics. To identify the potential interaction between FLE and increased prevalence and density of T. annulata, Sesmero-García et al. (2023) supported the hypothesis that FLE may facilitate pathogen colonization. Hussain et al. (2022) demonstrated a similar pattern with increased Rickettsia prevalence in FLE-positive ticks. Mechanisms may involve metabolic support, host immune defense suppression, or gut environment stabilization. High humidity and mixed farming systems in Diwaniyah may facilitate tick survival and enhance endosymbiont prevalence, contributing to higher transmission of Theileria. In mosquitoes, the associations between Wolbachia and pathogens have shown both inhibitory and facilitative effects depending on the strain. Comparable complexity may exist in ticks, where FLE could enhance rather than inhibit T. annulata growth. These findings have substantial epidemiological implications for cattle production in Iraq. T. annulata-induced tropical theileriosis reduces milk yield, weight gain, and fertility. Economic losses in South Asia are estimated at US$800 million annually, with untreated infection mortality rates reaching up to 40% in susceptible herds (Sajid et al., 2018). In Iraq, Al-Wasmee et al. (2024) documented reductions in milk yield of approximately 20% in infected herds, whereas Albayati et al. (2023) reported mortality exceeding 30% during outbreaks. The high prevalence of tick infection demonstrated in the present study supports the conclusion that cattle in Al-Diwaniyah remain under constant exposure pressure and require sustained surveillance and vector management.
Fig. 4. Multiple sequence alignment of mitochondrial 16S rRNA partial sequences of H. anatolicum with global references. Local isolates PQ623957–PQ623967 showed 99.48%–100% sequence identity with sequences from China and Pakistan, confirming close similarity. H. excavatum reference sequences displayed clear differences that separated them from H. anatolicum.
Fig. 5. FLE, Ca. M. mitochondrii and T. annulata densities in female and male H. anatolicum ticks. Note that the copy number ratio has been subjected to a log10 transformation. This shows a significant difference (p ˂ 0.05) between the three different microorganisms after normalization with the internal control L6 (rpl6) gene of Ixodes scapularis. ConclusionHyalomma anatolicum is the main cattle tick in Al-Diwaniyah province, accounting for 86% of all examined specimens. Molecular analysis confirmed almost complete genetic similarity to H. anatolicum populations from other regions. FLEs and Ca. M. mitochondrii were highly prevalent, and females had higher symbiont densities than males. T. annulata was found in all samples, with the highest loads in semi-engorged females. Statistical analysis revealed a positive correlation between Ca. M. mitochondrii and T. annulata, indicating a possible biological association that may support pathogen persistence. The findings show the central role of H. anatolicum in transmitting tropical theileriosis and point to microbiome-based approaches as future strategies for controlling the disease. AcknowledgmentsWe sincerely thank the College of Veterinary Medicine, University of Al-Qadisiyah, for their support and for providing the facilities necessary to conduct this study. Conflict of interestThe authors declare no conflict of interest. FundingNo specific funding source is available for this study; however, the work was self-funded. Authors' contributionsAlaa designed the study, and Azhar performed and analyzed the result; both contributed to the manuscript writing Data availabilityThe corresponding author can provide complete data when requested. ReferencesAdegoke, A., Kumar, D., Bobo, C., Rashid, M.I., Durrani, A.Z., Sajid, M.S. and Karim, S. 2020. Tick-borne pathogens shape the native microbiome within tick vectors. Microorganisms 8, 1299. Afshari, A., Tavassoli, M., Esmaeilnejad, B., Habibi, G. and Esmaeilnia, K. 2021. Molecular characterization and phylogenetic analysis of pathogenic Theileria spp. isolated from cattle and sheep based on cytochrome b gene in Iran. Arch. Razi Inst. 76, 243. Al-Abedi, G.J.K. and Al-Amery, A.M.A. 2021. Molecular diagnosis and phylogenetic analysis of Babesia species isolated from ticks of infested cattle in Wasit Governorate, Iraq. Iraqi J. Agri. Sci. 52(1), 136–145. Albayati, H.H., Al Khafaji, A.M., Kamel, A. and Al-Karagoly, H. 2023. Microscopical and phylogenetic analysis of Theileria annulata in Iraqi local breed cattle. Vet. Integr. Sci. 21, 457–472. Al-Fatlawi, M., Ali, M. and Albayati, H. 2018. Morphological and phylogenetic study of Hyalomma anatolicum in Al-Najaf, Iraq. Iraqi. J. Vet. Sci. 32, 261–266. Al-Zubaidei, H.H., Hasson, R.H., Al-Ani, M.O., Fayyad, E.J., Abbas, S.F. and Al-Khfaji, T.H. 2023. Geographical distribution of Ixodidae (hard ticks) in all provinces Iraq. Iraqi J. Vet. Sci. 37(Supplement IV), 197–201. AL-Mayah, S.H. and Abdul-Karim, A.T. 2020. Epidemiology and seasonal variation of ixodid ticks and Piroplasmida detection in cattle of Basrah province, Iraq. Indian J. Forensic Med. Toxicol. 14(3), 1391–1398. Ali, M.J., Atiyah, W.R., Al-Fatlawi, M.A. and Khlaif, S.F. 2021. Genotypic analysis of ticks species infesting cattle in Al-Diwaniyah abattoir. Iraqi. J. Vet. Sci. 35, 673–677. Ali, Z., Maqbool, A., Muhammad, K., Khan, M.S. and Younis, M. 2013. Prevalence of Theileria annulata infected hard ticks of cattle and buffalo in Punjab, Pakistan. J. Anim. Plant Sci. 23, 20–26. Al-Khafaji, A. 2018. Interactions between pathogenic and non-pathogenic Rickettsiales and the tick host. Liverpool, UK: The University of Liverpool. Al-Shabbani, A.H. and Al-Fatlawi, M.A. 2023. Morphological and genotypical identification of Hyalomma anatolicum isolated from cattle in Al-Daghara city, Al-Qadisiyah province, Iraq. Iraqi. J. Vet. Sci. 37, 683–687. Al-Wasmee, A.K.K., Hassone, W.S., Al-Janabi, H.T. and Al-Jabory, H.A. 2024. Molecular prevalence of theileriosis in calf at Babylon, Iraq. Adv. Anim. Vet. Sci. 12, 1061–1065. Ammazzalorso, A.D., Zolnik, C.P., Daniels, T.J. and Kolokotronis, S.O. 2015. To beat or not to beat a tick: comparison of DNA extraction methods for ticks (Ixodes scapularis). PeerJ 3, e1147. Azagi, T., Klement, E., Perlman, G., Lustig, Y., Mumcuoglu, K.Y., Apanaskevich, D.A. and Gottlieb, Y. 2017. Francisella-like endosymbionts and Rickettsia species in local and imported Hyalomma ticks. Appl. Environ. Microbiol. 83, e01302–e01-17. Aziz, K. 2022. Morphological and molecular identification of Ixodid ticks that infest ruminants in Erbil province, Kurdistan Region-Iraq. Passer. J. Basic. Appl. Sci. 4, 8–13. Benyedem, H., Lekired, A., Mhadhbi, M., Dhibi, M., Romdhane, R., Chaari, S., Rekik, M., Ouzari, H.I., Hajji, T. and Darghouth, M.A. 2022. First insights into the microbiome of Tunisian Hyalomma ticks gained through next-generation sequencing with a special focus on H. scupense. PLos One. 17, 268172. Biglari, P., Bakhshi, H., Chinikar, S., Belqeiszadeh, H., Ghaffari, M., Javaherizadeh, S., Faghihi, F. and Telmadarraiy, Z. 2018. Hyalomma anatolicum as the main infesting tick in an important livestock rearing region, central area of Iran. Iran. J. Public Health 47, 742. Bonnet, S.I. and Pollet, T. 2021. Update on the intricate tango between tick microbiomes and tick‐borne pathogens. Parasite. Immunol. 43, e12813. Boyer, P.H., Almeras, L., Plantard, O., Grillon, A., Talagrand-Reboul, E., Mccoy, K., Jaulhac, B. and Boulanger, N. 2019. Identification of closely related Ixodes species by protein profiling with MALDI-TOF mass spectrometry. PLos One 14, 223735. Buysse, M. and Duron, O. 2018. Multi-locus phylogenetics of the Midichloria endosymbionts reveals variable specificity of association with ticks. Parasitology 145, 1969–1978. Buysse, M., Floriano, A.M., Gottlieb, Y., Nardi, T., Comandatore, F., Olivieri, E., Giannetto, A., Palomar, A.M., Makepeace, B.L., Bazzocchi, C., Cafiso, A., Sassera, D. and Duron, O. 2021. A dual endosymbiosis supports nutritional adaptation to hematophagy in the invasive tick Hyalomma marginatum. eLife, 10, e72747. Celina, S.S., Italiya, J., Tekkara, A.O. and Černý, J. 2025. Crimean-Congo haemorrhagic fever virus in ticks, domestic, and wild animals. Front. Vet. Sci. 11, 1513123. Chegeni, A.H., Hosseini, R., Telmadarraiy, Z. and Abdigoudarzi, M. 2019. The Iranian Hyalomma (Acari: ixodidae) with molecular evidences to understand taxonomic status of species complexes. Persian. J. Acarol. 8, 291–308. Chikufenji, B., Chatanga, E., Galon, E.M., Mohanta, U.K., Mdzukulu, G., Ma, Y., Nkhata, M., Umemiya-Shirafuji, R. and Xuan, X. 2024. First report of dog ticks and tick-borne pathogens they are carrying in Malawi. J. Vet. Med. Sci. 86, 150–159. De Carvalho, I.L., Santos, N., Soares, T., Zé-Zé, L. and Núncio, M.S. 2011. Francisella-like endosymbiont in Dermacentor reticulatus collected in Portugal. Vector-Borne Zoonotic Dis. 11, 185–188. Floriano, A.M., Batisti Biffignandi, G., Castelli, M., Olivieri, E., Clementi, E., Comandatore, F., Rinaldi, L., Opara, M., Plantard, O., Palomar, A.M., Noël, V., Vijay, A., Lo, N., Makepeace, B.L., Duron, O., Jex, A., Guy, L. and Sassera, D. 2023. The evolution of intramitochondriality in Midichloria bacteria. Environ. Microbiol. 25, 2102–2117. de la Fuente, J., Estrada, L., -Pena, A., Venzal, J.M., Kocan, K.M. and Sonenshine, D.E. 2008. Overview: ticks as vectors of pathogens that cause disease in humans and animals. Front. Biosci. 13, 6938–6946. Ghafar, A., Cabezas-Cruz, A., Galon, C., Obregon, D., Gasser, R.B., Moutailler, S. and Jabbar, A. 2020. Bovine ticks harbour a diverse array of microorganisms in Pakistan. Parasites Vectors 13(1), 1. Gomes, J., Bilgiç, H.B. and Karagenç, T. 2018. Theileriosis and tick control management in different Mediterranean livestock production systems. Anim. Health. Livest. Mediterr. Perspect. 39, 34–40. Han, R., Yang, J.F., Mukhtar, M.U., Chen, Z., Niu, Q.L., Lin, Y.Q., Liu, G.Y., Luo, J.X., Yin, H. and Liu, Z.J. 2019. Molecular detection of Anaplasma infections in ixodid ticks from the Qinghai-Tibet Plateau. Infect. Dis. Poverty. 8, 83–90. Hasson, R.H. 2012. Tick distribution and infestation among sheep and cattle in Baghdad’s south suburb. Kufa J. Vet. Med. Sci. 3, 77–90. Hoffman, T., Sjödin, A., Öhrman, C., Karlsson, L., Mcdonough, R.F., Sahl, J.W., Birdsell, D., Wagner, D.M., Carra, L.G., Wilhelmsson, P., Pettersson, J.H.O., Barboutis, C., Figuerola, J., Onrubia, A., Kiat, Y., Piacentini, D., Jaenson, T.G.T., Lindgren, P.E., Moutailler, S., Fransson, T., Forsman, M., Nilsson, K., Lundkvist, A. and Olsen, B. 2022. Co-occurrence of Francisella, spotted fever group Rickettsia, and Midichloria in Avian-associated Hyalomma rufipes. Microorganisms 10, 1393. Hussain, S., Parveen, N., Hussain, A., Song, B., Aziz, M.U., Zeb, J., Li, J., George, D., Cabezas-Cruz, A. and Sparagano, O. 2022. The symbiotic continuum within ticks: Opportunities for disease control. Front. Microbiol. 13, 854803. Kakar, M.E., Khan, M.A., Khan, M.S., Ashraf, K., Kakar, M.A., Hamdullah, Jan, S. and Razzaq, A. 2017. Prevalence of tick infestation in different breeds of cattle in Balochistan. J. Anim. Plant Sci. 27(3), 797–802. Kawan, M.H. 2019. Molecular surveillance and phylogenetic analysis of Theileria annulata in bovine at Baghdad city/Iraq. Iraqi J. Vet. Med. 43, 93–101. Kolo, A.O. and Raghavan, R. 2023. Impact of endosymbionts on tick physiology and fitness. Parasitology 150, 859–865. Köseoğlu, A.E., Can, H., Güvendi, M., Erkunt Alak, S., Kandemir, C., Taşkın, T., Demir, S., Akgül, G., Değirmenci Döşkaya, A., Karakavuk, M., Döşkaya, M., Gürüz, A.Y. and Ün, C. 2021. Molecular investigation of bacterial and protozoal pathogens in ticks collected from different hosts in Turkey. Parasites. Vectors. 14, 270. Luo, T., Hu, E., Gan, L., Yang, D., Wu, J., Gao, S., Tuo, X., Bayin, C.G., Hu, Z. and Guo, Q. 2024. Candidatus Midichloria mitochondrii can be vertically transmitted in Hyalomma anatolicum. Exp. Parasitology. 265, 108828. Machado-Ferreira, E., Piesman, J., Zeidner, N.S. and Soares, C.A.G. 2009. Francisella-like endosymbiont DNA and Francisella tularensis virulence-related genes in Brazilian ticks (Acari: Ixodidae). J. Med. Entomol. 46, 369–374. Mohammad Al-saeed, A.T., Omer, L.T., Abdo, J., Habibi, G., Salih, D.A., Seitzer, U. and Ahmed, J. 2010. Epidemiological studies on tropical theileriosis (Theileria annulata infection of cattle) in Kurdistan Region, Iraq. Parasitol. Res. 106, 403–407. Mohammed, R.R., Enferadi, A., Sidiq, K.R., Sarani, S., Khademi, P., Jaydari, A. and Ahmed, A.K. 2023. Molecular detection of Francisella tularensis isolated from ticks of livestock in Kurdistan Region, Iraq. Vector-Borne. Zoonotic. Dis. 23, 514–519. Nandi, A., Manisha., Solanki, V., Tiwari, V., Sajjanar, B., Sankar, M., Saini, M., Shrivastava, S., Bhure, S.K. and Ghosh, S. 2023. Protective efficacy of multiple epitope-based vaccine against Hyalomma anatolicum, vector of Theileria annulata and Crimean–Congo hemorrhagic fever virus. Vaccines 11, 881. Nasiri, Z., Alipour, H., Kalantari, M., Soltani, A., Hosseini-Chegeni, A., Dabaghmanesh, S., Yousefi, S. and Azizi, K. 2025. Bionomics and ecological characteristics of hard ticks of Ixodidae in Fars province, southwestern Iran. Exp. Appl. Acarol. 94, 39. Ngnindji-Youdje, Y., Lontsi-Demano, M., Diarra, A.Z., Foyet, J., Tchuinkam, T. and Parola, P. 2025. Ticks (Acari: ixodidae) and tick-borne diseases in Cameroon: current understanding and future directions for more comprehensive surveillance. One Health 20, 100949. Olivieri, E., Epis, S., Castelli, M., Varotto Boccazzi, I., Romeo, C., Desirò, A., Bazzocchi, C., Bandi, C. and Sassera, D. 2019. Tissue tropism and metabolic pathways of Midichloria mitochondrii suggest tissue-specific functions in the symbiosis with Ixodes ricinus. Ticks Tick-borne Dis. 10(5), 1070 -1077. Omer, S.A., Alsuwaid, D.F. and Mohammed, O.B. 2021. Molecular characterization of ticks and tick-borne piroplasms from cattle and camel in Hofuf, Eastern Saudi Arabia. Saudi. J. Biol. Sci. 28, 2023–2028. Perveen, N., Bin Muzaffar, S. and Al-Deeb, M.A. 2020. Population dynamics of Hyalomma dromedarii on camels in the United Arab Emirates. Insects 11, 320. Sajid, M.S., Kausar, A., Iqbal, A., Abbas, H., Iqbal, Z. and Jones, M.K. 2018. An insight into the ecobiology, vector significance and control of Hyalomma ticks (Acari: ixodidae): A review. Acta Tropica 187, 229–239. Sassera, D., Lo, N., Bouman, E.A.P., Epis, S., Mortarino, M. and Bandi, C. 2008. “Candidatus Midichloria” endosymbionts bloom after the blood meal of the host, the hard tick Ixodes ricinus. Appl. Environ. Microbiol. 74, 6138–6140. Scoles, G.A. 2004. Phylogenetic analysis of the Francisella-like endosymbionts of Dermacentor ticks. J. Med. Entomol. 41, 277–286. Sesmero-García, C., Cabanero-Navalon, M.D. and Garcia-Bustos, V. 2023. The importance and impact of Francisella-like endosymbionts in Hyalomma ticks in the era of climate change. Diversity 15, 562. Shamoon-Pour, M., Canessa, E.H., Macher, J., Fruitwala, A., Draper, E., Policriti, B., Chin, M., Nunez, M., Puccio, P., Fang, Y., Wang, X.R. and Hathout, Y. 2025. Genomic and proteomic analyses of bacterial communities of Ixodes scapularis ticks from Broome County, New York. Microorganisms 13, 258. Shyma, K.P., Kumar, S., Sharma, A.K., Ray, D.D. and Ghosh, S. 2012. Acaricide resistance status in Indian isolates of Hyalomma anatolicum. Exp. Appl. Acarol. 58, 471–481. Soliman, A.M., Mahmoud, H.Y.A.H., Amer, M.M., Mohamed, S., Hifumi, T. and Tanaka, T. 2025. Molecular prevalence and phylogenetic characterization of Francisella-like endosymbionts in ticks infesting camels and cattle from Southern Egypt. Microb. Pathog. 205, 107639. Tavakolian, Z., Tukmechi, A., Ownagh, A. and Enferadi, A. 2025. Molecular detection of Francisella spp. in blood, milk, and ticks infesting small ruminants in Urmia, Iran. Vector-Borne. Zoonotic. Dis. 25, 454–462. Tukmechi, A., Ownagh, A., Enferadi, A. and Khademi, P. 2024. First molecular detection of Francisella tularensis in turtle (Testudo graeca) and ticks (Hyalomma aegyptium) in Northwest of Iran. Int. J. Parasitol. Parasites. Wildl. 23, 100892. Uzum, Z., Ershov, D., Pavia, M.J., Mallet, A., Gorgette, O., Plantard, O., Sassera, D. and Stavru, F. 2023. Three-dimensional images reveal the impact of the endosymbiont Midichloria mitochondrii on the host mitochondria. Nat. Commun. 14, 4133. Walker, A.R., Bouattour, A., Camicas, J.L., Estrada-Peña, A., Horak, I.G., Latif, A.A., Pegram, R.G. and Preston, P.M. 2003. Ticks of domestic animals in Africa: a guide to identification of species. Edinburgh, UK: Bioscience Reports. Wójcik-Fatla, A., Zając, V., Sawczyn, A., Cisak, E., Sroka, J. and Dutkiewicz, J. 2015. Occurrence of Francisella spp. in Dermacentor reticulatus and Ixodes ricinus ticks collected in eastern Poland. Ticks. Tick-Borne. Dis. 6, 253–257. Zając, Z., Obregon, D., Foucault-Simonin, A., Wu-Chuang, A., Moutailler, S., Galon, C., Kulisz, J., Woźniak, A., Bartosik, K. and Cabezas-Cruz, A. 2023. Disparate dynamics of pathogen prevalence in Ixodes ricinus and Dermacentor reticulatus ticks occurring sympatrically in diverse habitats. Sci. Rep. 13, 10645. Zhong, Z., Wang, K. and Wang, J. 2024. Tick symbiosis. Curr. Opin. Insect. Sci. 62, 101163. | ||
| How to Cite this Article |
| Pubmed Style Alrikabi A, Al-khafaji AMA. Co-occurrence of Francisella-like endosymbionts and Candidatus Midichloria mitochondrii with Theileria annulata in Al-Diwaniyah, Iraq, Hyalomma ticks. Open Vet. J.. 2026; 16(2): 999-1011. doi:10.5455/OVJ.2026.v16.i2.21 Web Style Alrikabi A, Al-khafaji AMA. Co-occurrence of Francisella-like endosymbionts and Candidatus Midichloria mitochondrii with Theileria annulata in Al-Diwaniyah, Iraq, Hyalomma ticks. https://www.openveterinaryjournal.com/?mno=269047 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.21 AMA (American Medical Association) Style Alrikabi A, Al-khafaji AMA. Co-occurrence of Francisella-like endosymbionts and Candidatus Midichloria mitochondrii with Theileria annulata in Al-Diwaniyah, Iraq, Hyalomma ticks. Open Vet. J.. 2026; 16(2): 999-1011. doi:10.5455/OVJ.2026.v16.i2.21 Vancouver/ICMJE Style Alrikabi A, Al-khafaji AMA. Co-occurrence of Francisella-like endosymbionts and Candidatus Midichloria mitochondrii with Theileria annulata in Al-Diwaniyah, Iraq, Hyalomma ticks. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 999-1011. doi:10.5455/OVJ.2026.v16.i2.21 Harvard Style Alrikabi, A. & Al-khafaji, . A. M. A. (2026) Co-occurrence of Francisella-like endosymbionts and Candidatus Midichloria mitochondrii with Theileria annulata in Al-Diwaniyah, Iraq, Hyalomma ticks. Open Vet. J., 16 (2), 999-1011. doi:10.5455/OVJ.2026.v16.i2.21 Turabian Style Alrikabi, Azhar, and Alaa Mohammed Abdulrazaq Al-khafaji. 2026. Co-occurrence of Francisella-like endosymbionts and Candidatus Midichloria mitochondrii with Theileria annulata in Al-Diwaniyah, Iraq, Hyalomma ticks. Open Veterinary Journal, 16 (2), 999-1011. doi:10.5455/OVJ.2026.v16.i2.21 Chicago Style Alrikabi, Azhar, and Alaa Mohammed Abdulrazaq Al-khafaji. "Co-occurrence of Francisella-like endosymbionts and Candidatus Midichloria mitochondrii with Theileria annulata in Al-Diwaniyah, Iraq, Hyalomma ticks." Open Veterinary Journal 16 (2026), 999-1011. doi:10.5455/OVJ.2026.v16.i2.21 MLA (The Modern Language Association) Style Alrikabi, Azhar, and Alaa Mohammed Abdulrazaq Al-khafaji. "Co-occurrence of Francisella-like endosymbionts and Candidatus Midichloria mitochondrii with Theileria annulata in Al-Diwaniyah, Iraq, Hyalomma ticks." Open Veterinary Journal 16.2 (2026), 999-1011. Print. doi:10.5455/OVJ.2026.v16.i2.21 APA (American Psychological Association) Style Alrikabi, A. & Al-khafaji, . A. M. A. (2026) Co-occurrence of Francisella-like endosymbionts and Candidatus Midichloria mitochondrii with Theileria annulata in Al-Diwaniyah, Iraq, Hyalomma ticks. Open Veterinary Journal, 16 (2), 999-1011. doi:10.5455/OVJ.2026.v16.i2.21 |