| Research Article | ||
Open Vet. J.. 2025; 15(10): 5146-5156 Open Veterinary Journal, (2025), Vol. 15(10): 5146-5156 Research Article Cysteine reduces oxidative damage induced by heat stress in dromedary camel oocytes during in vitro maturationNasser Ghanem1, Mohamed Eleam2, Gamal Shawki2, Marwa El-Sheikh3, Omar A. Farid4, Beshoy S.F. Khalil5, Fatma Badawy Aboelwafa5, Najmi Mariol6 and Ayman Mesalam2*1Animal Production Department, Faculty of Agriculture, Cairo University, Giza, Egypt 2Department of Theriogenology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt 3Department of Microbial Biotechnology, Biotechnology Research Institute, National Research Centre (NRC), Dokki, Cairo, Egypt 4National Organization for Drug Control and Research, Giza, Egypt 5Genetics Department, Faculty of Agriculture, Cairo University, Giza, Egypt 6Department of Surgery and Theriogenology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya *Corresponding Author: Ayman Mesalam. Department of Theriogenology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt. Email: aymanmesalam [at] gmail.com Submitted: 07/07/2025 Revised: 10/09/2025 Accepted: 20/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: Assisted reproductive technologies (ARTs), including in vitro maturation (IVM), hold promise for improving reproductive efficiency in dromedary camels, especially under challenging environmental conditions. Aim: This study aimed to evaluate the potential effects of cysteine supplementation in IVM medium on nuclear maturation, mitochondrial activity, antioxidant status, oocyte ultrastructure, and heat stress marker genes expression using a dromedary camel oocyte model. Methods: Two experiments were conducted. In Experiment 1, cumulus-oocyte complexes (COCs) were collected and matured in vitro using varying cysteine concentrations (0.0, 0.8, 1.6, and 2.4 mM) under standard conditions (38.5°C, 5% CO2, 40 hours) to identify the optimal concentration. Experiment 2 evaluated the protective effect of the selected concentration (1.6 mM) during heat stress, where COCs were exposed to 40.5°C for the first 24 hours of maturation, followed by exposure to 38.5°C for 16 hours. Three groups were compared: control, heat-stressed without cysteine, and heat-stressed with cysteine. We evaluated oocyte nuclear maturation, mitochondrial intensity, adenosine triphosphate (ATP) content, the activity of antioxidant markers, and oocyte ultrastructure. We used RT-quantitative real-time PCR to assess the mRNA expression of cumulus expansion, oxidative stress, and mitochondrial health-related genes. Results: Results showed that heat stress conditions significantly reduced nuclear maturation, mitochondrial activity, ATP and antioxidant levels, and the mRNA expression levels of antioxidant, anti-apoptotic, cumulus expansion, and oocyte quality markers, while increasing pro-apoptotic CASP3 transcription level (p < 0.05). Conversely, cysteine administration markedly reversed heat stress-induced detrimental effects by restoring oocyte maturation and ultrastructure, mitochondrial intensity, ATP content, antioxidant balance, and gene expression profile modulation. Conclusion: Supplementation with 1.6 mM cysteine during IVM of camel oocytes effectively improves oocyte quality and mitigates heat stress-related damage. This research supports the practical application of cysteine as a supplement to IVM media, specifically to enhance oocyte developmental competence and improve ART success rates in camels exposed to thermal stress. Keywords: Cysteine, Heat stress, IVM, Dromedary camel, Antioxidant activity. IntroductionGlobal climate change poses a significant threat to animal productivity, particularly in arid and semiarid regions where livestock are frequently exposed to elevated ambient temperatures and drought conditions (Kumar, 2021). In this context, the dromedary camel (Camelus dromedarius) exhibits unique physiological and anatomical adaptations that enable it to survive and reproduce under extreme environmental stressors (Hoter et al., 2019). Despite their resilience, the reproductive performance of female camels remains suboptimal due to natural limitations, such as induced ovulation, seasonal breeding, extended calving intervals, and weak estrous expression (Bello and Bodinga, 2020). Assisted reproductive technologies, including in vitro maturation (IVM) and embryo production, have emerged as promising strategies to enhance reproductive efficiency and genetic progress in dromedary camels (Tukur et al., 2020). IVM is a critical step in the production of developmentally competent oocytes, involving the completion of meiosis and progression to the metaphase II (MII) stage (Hashimoto, 2009; Zhang et al., 2018). However, camel oocytes are particularly sensitive to thermal stress, which compromises cytoplasmic maturation, disrupts spindle integrity, and increases chromosomal abnormalities and apoptotic oocyte percentages (Saadeldin et al., 2018; Camargo et al., 2019 b). Heat stress (HS) induces oxidative stress in oocytes and embryos, leading to DNA damage, apoptosis, and enzyme inactivation, eventually resulting in cell death and subsequently impairing their development, which has a detrimental effect on reproductive performance (Azad et al., 2010; Abdelkhalek et al., 2017; Khan et al., 2023). These adverse effects result from the imbalance between reactive oxygen species (ROS) and endogenous antioxidant defenses (Lampiao, 2012). Previous studies have demonstrated that supplementing the IVM medium with exogenous antioxidants, such as caffeine, L-carnitine, or N-acetylcysteine (NAC), can alleviate heat-induced oxidative damage and enhance oocyte quality in various species, including camels (Moawad et al., 2020) and porcine (Hu et al., 2020). Cysteine, a thiol-containing amino acid and intracellular glutathione (GSH) precursor, has also shown beneficial effects on bovine oocyte maturation by scavenging ROS and enhancing antioxidant capacity (Ali et al., 2003; Khattab et al., 2020). Furthermore, supplementation of honey bees with cysteine-proteinogenic amino acid mixtures has been reported to improve their antioxidant capacities and resilience against oxidative stress (Lee et al., 2024). Moreover, cysteine, as well as other essential IVM medium ingredients, have been linked to increased oocyte GSH content, decreased ROS, and improved maturation rates and developmental competence across multiple species (De Matos and Furnus, 2000; Barakat et al., 2020; El Sheikh et al., 2020; El-Sheikh et al., 2025). The impact of cysteine supplementation on heat-stressed dromedary camel oocytes during IVM has not been thoroughly investigated despite these promising findings. Therefore, this study aimed to determine the optimal cysteine concentration for improving oocyte maturation and evaluate its beneficial effects against heat stress. Specifically, we assessed the nuclear maturation rate, mitochondrial activity, adenosine triphosphate (ATP) content, antioxidant biomarkers, oocyte ultrastructure, and genes expression profiles in camel oocytes and cumulus cells subjected to thermal stress and treated with cysteine during IVM. Materials and methodsChemicals and mediumAll chemicals and reagents were obtained from Sigma-Aldrich (St. Louis, MO, USA), unless otherwise stated. Experimental designThis study was conducted in two experiments. Experiment 1 aimed to determine the optimal cysteine concentration that affects oocyte maturation. Cumulus-oocyte complexes (COCs) were cultured in IVM medium supplemented with 0.0 (control), 0.8, 1.6, and 2.4 mM cysteine. Oocytes were incubated at 38.5°C under 5% CO2 for 40 hours. The second experiment was conducted to evaluate the protective effect of 1.6 mM cysteine (optimal concentration of experiment 1) on heat-stressed COCs. The oocytes were exposed to 40.5°C during the first 24 hours of IVM, followed by 38.5°C for the remaining 16 hours (Ashour et al., 2020). Three groups were compared: the control and heat-stressed COCs with and without cysteine. Ovarian collection and oocyte retrievalA total of 300 ovaries were collected from healthy, nonpregnant dromedary camels at a local abattoir during the breeding season. Ovaries were transported to the laboratory within 2 hours post-slaughter in pre-warmed sterile saline (containing 100 IU/ml penicillin and 100 µg/ml streptomycin). COCs were obtained by slicing ovaries in warm TCM-199 (HEPES-buffered) supplemented with 2% heat-inactivated fetal bovine serum (FBS), 0.3 mg/ml glutamine, and 50 µg/ml gentamicin. Only COCs with homogeneous ooplasm and multilayered cumulus were selected under a stereomicroscope (Leica, Germany). In vitro maturation of oocytesSelected COCs (n=850) were washed thrice in maturation medium (TCM-199 supplemented with 10% FBS, 0.15 mg/ml glutamine, 22 µg/ml sodium pyruvate, 1 µg/ml estradiol-17β, 5 µg/ml FSH, and 50 µg/ml gentamicin) and groups of 50 COCs were cultured in 500 µl of IVM medium and incubated at 5% CO2. Control COCs were cultured at 38.5°C for 40 hours and heat-stressed oocytes were incubated at 40.5°C for the first 24 hours, followed by incubation at 38.5°C for the remainder of the IVM. Assessment of oocyte maturationCumulus expansion was scored morphologically under a stereomicroscope, and the proportion of expanded COCs (n=40 per group/four biological replicates were performed, with ten oocytes per replicate) was calculated. COCs were denuded by repeated pipetting in 0.25% hyaluronidase and examined for the extrusion of the first polar body. Denuded oocytes were fixed and stained with 0.01 mg/ml Hoechst 33342 in 0.3% PBS for 10 minutes in the dark. After washing, oocytes were mounted on glass slides and classified according to nuclear stage as immature at germinal vesicle, germinal vesicle breakdown, and metaphase I, or matured at MII stage. Assessment of mitochondrial activityMitochondrial activity was assessed using MitoTracker™ Green FM (Invitrogen, Carlsbad, CA, USA) according to Ghanem et al. (2020). After washing, oocytes (n=20 per group/four biological replicates were performed, with five oocytes per replicate) were incubated with 200 nM of the dye for 30 minutes, rinsed, mounted, and visualized under an epifluorescence microscope. Fluorescence intensity was quantified using ImageJ software (https://imagej.net/ij/download.html) and was expressed as mean gray value per oocyte after subtracting the background intensity. Data were normalized to the number of oocytes analyzed in each replicate to ensure comparability across groups. Biochemical analysis of spent IVM mediumSpent media were analyzed for ATP content, antioxidant activity [glutathione (GSH), superoxide dismutase (SOD), and catalyze (CAT)], and oxidative stress markers (MDA) using high-performance liquid chromatography (HPLC; Agilent, Santa Clara, CA, USA). HPLC was performed using an Agilent system with a Synergi RP Max column and UV detection at 210 nm. The mobile phase consisted of potassium phosphate buffer and acetonitrile (pH 2.7) at a flow rate of 2 ml/minute. For HPLC validation, the calibration curves for ATP, GSH, MDA, SOD, and CAT showed good linearity (r² > 0.99), and the limit of detection (LOD) and limit of quantitation (LOQ) values were within the standard ranges. Transmission electron microscopy analysis of mature oocytesFollowing IVM, the cumulus cells were removed by repeated pipetting in 0.25% hyaluronidase, and the oocytes (n=20 per group/four biological replicates were performed, with five oocytes per replicate) were immediately fixed in 2.5% glutaraldehyde in 0.1 M sodium phosphate buffer (pH 7.4) at room temperature for 15 minutes. After three washes in the same buffer (30 minutes each), the samples were post-fixed in 1% osmium tetroxide and then dehydrated through a graded ethanol series, ending with two changes in absolute ethanol for 15 minutes each. The dehydrated oocytes were embedded in Spurr’s epoxy resin. Ultrathin sections (90 nm) were cut using an ultramicrotome (Leica EM-UC6, Japan) and mounted on 400-mesh copper grids. Sections were stained with 8% uranyl acetate and 1% lead citrate for 5 minutes before being examined under a transmission electron microscope (JEOL JEM-1400, Japan). RNA extraction from oocytes and cumulus cellsTotal RNA was extracted from oocytes and cumulus cells (n=15 per group/three biological replicates were performed, with five oocytes per replicate) using the Arcturus™ PicoPure™ RNA Isolation Kit (Thermo Fisher Scientific, USA) according to the manufacturer’s protocol. DNase treatment was performed to remove genomic DNA contamination. The RNA concentration and purity were assessed by spectrophotometry (NanoDrop 2000C, Thermo Fisher Scientific), and the samples were stored at −80°C until use. cDNA synthesis and quantitative real-time PCR (RT-qPCR)For cDNA synthesis, 1 µg of RNA was reverse-transcribed using the RevertAid First Strand cDNA Synthesis Kit with oligo (dT) primers (Thermo Fisher Scientific, Waltham, MA, USA). The reaction was performed at 42°C for 60 minutes, followed by enzyme inactivation at 70°C for 5 minutes. Gene expression analysis was performed using the Maxima SYBR Green qPCR Master Mix (Thermo Fisher Scientific). Each reaction contained 12 µl of master mix, 0.5 µl of each primer (10 µM), 2 µl of cDNA, and 5 µl of nuclease-free water. Amplification was performed using a real-time PCR thermocycler under the following conditions: 95°C for 10 minutes, followed by 40 cycles of 95°C for 15 seconds, 60°C for 1 minute, and 72°C for 30 seconds. GAPDH and ACTB were used as reference genes. Relative gene expression was calculated using the 2-ΔΔCt method. The primer sequences are listed in Table 1. The genes analyzed in this study include superoxide dismutase 2 (SOD2), glutathione peroxidase (GPX1), B-cell lymphoma apoptosis regulator 2 (BCL2), caspase3 (CASP3), hyaluronan synthase 2 (HAS2), tumor necrosis factor α-induced protein 6 (TNFAIP6), bone morphogenic protein 15 (BMP15), glucose transporter 1 (GLUT1), cyclin B1 (CCNB1), mitochondrial transcription factor A (TFAM), beta actin (ACTB), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Table 1. List of primers used for analyses the expression of target and reference genes in dromedary camel cumulus cells and oocytes under control, heat stress (HS), and HS + cysteine conditions.
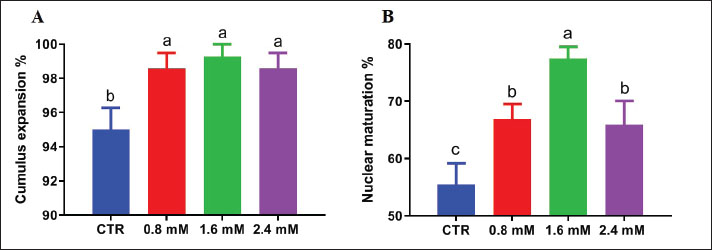
Statistical analysisData were analyzed using one-way analysis of variance in SPSS (version 22), and Duncan’s multiple range test was used to compare the treatment groups. Results are presented as mean ± SEM. Differences were considered statistically significant at p ≤ 0.05. Experiments were performed in six replicates using oocytes matured on the same day. Ethical approvalThe Zagazig University Institutional Animal Care and Use Committee reviewed and approved the experimental procedures (Approval Number ZU-IACUC/2/F/255/2024). Approval date: 27/10/2024. ResultsOptimal cysteine concentration for oocyte maturationSupplementation of the IVM medium with cysteine significantly improved cumulus expansion (Fig. 1A). Among the tested concentrations (0.8, 1.6, and 2.4 mM), 1.6 mM resulted in the highest (p < 0.05) proportion of oocytes reaching the MII stage (Fig. 1B). Although the higher concentration of cysteine (2.4 mM) improved oocyte quality compared with the control, it was less effective than 1.6 mM (Fig. 1B).
Fig. 1. Effect of varying cysteine concentrations (0.0, 0.8, 1.6, and 2.4 mM) on (A) dromedary camel cumulus expansion and (B) oocytes reaching the MII stage during IVM. Data are presented as mean ± SEM. Different superscripts indicate significant differences (p < 0.05). Cysteine effect on oocyte maturation and mitochondrial activity under heat stressExposure to heat stress (40.5°C for the first 24 hours of IVM) significantly reduced the MII rate compared with that in the control group (38.5°C). However, the inclusion of 1.6 mM cysteine in the IVM medium during heat stress restored nuclear maturation rates to levels statistically comparable to those observed in the non-stressed control (Fig. 2A). Mitochondrial intensity was significantly reduced in oocytes exposed to heat stress, indicating impaired mitochondrial function. Under heat stress, supplementation with 1.6 mM cysteine significantly improved mitochondrial fluorescence intensity, suggesting a protective effect on mitochondrial function (Fig. 2B).
Fig. 2. Effect of cysteine on oocyte maturation and mitochondrial activity under heat stress. (A) Nuclear maturation rate (% MII stage) of oocytes cultured under normal and heat-stress conditions with or without 1.6 mM cysteine supplementation. Cysteine alleviated the harmful effects of heat stress. (B) Mitochondrial activity assessed by MitoTracker fluorescence intensity in oocytes from different treatment groups: control, heat stress (HS), and HS + cysteine. Cysteine supplementation significantly restored mitochondrial function under heat stress conditions. Data are presented as mean ± SEM. Different superscripts indicate significant differences (p < 0.05). Effect of cysteine on antioxidant status and oxidative stress biomarkers under heat stressBiochemical analysis of the spent IVM media revealed that heat stress significantly reduced the concentrations of ATP, GSH, SOD, and CAT, indicating increased oxidative stress. Conversely, under heat stress, cysteine supplementation significantly restored ATP and SOD antioxidant levels while reducing MDA concentration (Fig. 3).
Fig. 3. Effect of cysteine on antioxidant status and oxidative stress biomarkers under heat stress. Concentrations of (A) Adenosine triphosphate (ATP), (B) glutathione (GSH), (C) superoxide dismutase (SOD), (D) catalyze (CAT), and (E) malondialdehyde (MDA) in spent IVM medium under control, heat stress (HS), and HS + cysteine conditions. Cysteine supplementation reversed the oxidative stress-induced changes. Bars represent mean ± SEM; different superscripts indicate significant differences (p < 0.05). Ultrastructural observations using transmission electron microscopyUltrastructural evaluation of oocytes using transmission electron microscopy revealed marked differences among treatment groups. Control oocytes displayed typical cytoplasmic organization, with round mitochondria exhibiting dense matrices and intact cristae, sparse lipid droplets, and cortical granules beneath the oolemma (Fig. 4A). In contrast, heat-stressed oocytes exhibited ultrastructural degeneration, including swollen and vacuolated mitochondria, a marked increase in the number and size of lipid droplets, and disorganized or mislocalized cortical granules (Fig. 4B). These findings indicate impaired mitochondrial function and compromised cytoplasmic maturation under thermal stress. Oocytes from the heat stress + cysteine group showed less pronounced ultrastructural alterations compared with those from the heat-stressed group. Mitochondria generally appeared more intact, with less swelling and visibility of partial cristae. Lipid droplets were fewer and more dispersed than those in the heat-stressed group, and cortical granules displayed partial realignment near the oolemma (Fig. 4C). These findings suggest that cysteine supplementation mitigated heat stress-induced subcellular damage.
Fig. 4. Transmission electron microscopy of camel oocytes from different treatment groups (n=20 oocytes per group; four biological replicates with five oocytes per replicate). (A) Control: Numerous intact mitochondria and aligned cortical granules beneath the oolemma. (B) Heat stress: Swollen and vacuolated mitochondria and dispersed cortical granules. (C) Heat stress + cysteine: Moderate mitochondrial preservation and cortical granule alignment partial restoration. Effect of cysteine on gene expression in oocytes and cumulus cells under heat stressRT-qPCR analysis showed that heat stress significantly downregulated the expression of the antioxidant gene (GPX1), anti-apoptotic gene (BCL2), cumulus expansion gene (TNFAIP6), and oocyte quality markers (BMP15, GLUT1, CCNB1). In contrast, CASP3, a proapoptotic gene, was significantly upregulated. Cysteine supplementation during heat stress significantly upregulated all beneficial genes and suppressed CASP3 expression, highlighting the capacity of cysteine to alleviate heat-induced molecular damage (Fig. 5).
Fig. 5. Relative mRNA expression of key genes related to antioxidant defense (SOD2 and GPX1), apoptosis (BCL2 and CASP3), cumulus expansion (HAS2 and TNFAIP6), oocyte competence (BMP15, GLUT1, and CCNB1), and mitochondrial function (TFAM) in (A) oocytes and (B) cumulus cells (n=15 oocytes per group; three biological replicates with five oocytes per replicate). Cysteine counteracted heat-induced dysregulation. Expression values normalized to GAPDH and ACTB levels. Bars represent mean ± SEM; different letters denote significant differences (p < 0.05). DiscussionCytoplasmic and nuclear maturation of oocytes is a critical prerequisite for successful embryonic development (Lonergan and Fair, 2016). HS impairs oocyte maturation by disrupting cytoskeletal organization, spindle formation, and mitochondrial function (Ju et al., 2005; Andreu-Vazquez et al., 2010; Nabenishi et al., 2012b). Here, we reported that HS significantly reduced nuclear maturation rates and mitochondrial activity in camel oocytes. Our findings align with previous studies reported that the nuclear maturation rate of oocytes was significantly reduced when COCs were exposed to heat stress in camel (Ashour et al., 2020) and bubaline (Khalil et al., 2023). Mitochondria play an essential role in ATP generation, redox balance regulation, and proper meiotic progression (Schatten et al., 2014; Jiang et al., 2023). A reduction in mitochondrial activity under HS suggests compromised cytoplasmic maturation and metabolic dysfunction. It has been reported that heat stress is associated with decreased mitochondrial function, distribution, and membrane potential in COCs (Abdelnour et al., 2020). Cysteine supplementation during IVM significantly improved the maturation rate and cytoplasmic competency of dromedary camel oocytes. This effect is consistent with previous reports demonstrating enhanced cumulus expansion and nuclear maturation of camel oocytes following cysteine supplementation (Khattab et al., 2020) and bovines under normal in vitro maturation conditions (Rahim et al., 2011; Elgebaly et al., 2022). In our study, the higher concentration of cysteine (2.4 mM) improved oocyte quality compared with the control but was less effective than the optimal dose (1.6 mM). This may reflect the phenomenon whereby excessive antioxidant supplementation can disrupt redox homeostasis and exert cytotoxic effects (Bouayed and Bohn, 2010). The beneficial role of cysteine may be attributed to its role in GSH synthesis, a major intracellular antioxidant that supports redox balance and oocyte competence (Rahim et al., 2011). Importantly, cysteine supplementation during HS restored both nuclear maturation and mitochondrial intensity to levels comparable to those in the control group. This observation aligns with studies in bovine (Nabenishi et al., 2012a) and porcine oocytes (Hu et al., 2020), where cysteine or NAC supplementation alleviated mitochondrial dysfunction under thermal stress. This mitochondrial protection may result from increased GSH biosynthesis, ROS scavenging, and mitochondrial membrane stabilization. Oxidative stress markers in spent IVM media further confirmed the protective role of cysteine. HS decreased SOD, CAT, GSH, and ATP levels, indicating enhanced lipid peroxidation and compromised antioxidant defense. These findings support previous reports that HS leads to excessive ROS generation and antioxidant depletion in bovine (Sakatani et al., 2008) and bubaline oocytes (Waiz et al., 2016). Cysteine supplementation significantly improved antioxidant levels and reduced MDA concentrations, suggesting redox balance restoration and reduced oxidative damage. In agreement with our findings, Hu et al. (2020) reported that the addition of cysteine to IVM protected porcine oocytes against heat stress-induced oxidative stress and apoptosis. Similarly, Whitaker and Knight (2008) found that NAC-amide supplementation raised SOD and catalase activity in porcine oocytes. Ultrastructural evaluation of oocytes by transmission electron microscopy provided compelling evidence of cytoplasmic integrity and damage across treatment groups. Heat-stressed oocytes showed substantial subcellular disorganization, characterized by swollen or vacuolated mitochondria, excessive lipid droplet accumulation, and mis-localized cortical granules, indicating hallmarks of compromised oocyte quality and early signs of apoptosis (Andreu-Vazquez et al., 2010; Gustina et al., 2017; Camargo et al., 2019 a). These findings align with previous reports in heat-exposed oocytes of other species, such as cattle and pigs (Nabenishi et al., 2012b; Hu et al., 2020), highlighting the thermal insult sensitivity of camel oocytes. The results showed that oocytes treated with cysteine during heat stress exhibited partial restoration of ultrastructure. Mitochondria appeared less swollen, often clustered around the ooplasm’s periphery, a pattern previously associated with metabolically competent oocytes in mice and cattle (Bavister and Squirrell, 2000; Wang et al., 2009). The improved mitochondrial distribution may reflect enhanced ATP production (Torner et al., 2004). In addition, cysteine-treated oocytes showed electron-dense vesicles of variable size and distribution throughout the cytoplasm, which may act as nutrient reserves or participate in cortical granule formation, as suggested in earlier studies (Russe, 1983; Fair et al., 1997). Cortical granules in the cysteine group were often found aligned beneath the oolemma, either isolated or forming short linear clusters, a pattern indicative of cytoplasmic maturation and readiness for zona hardening at fertilization (Zhang et al., 2011; Pavani et al., 2020). Collectively, these findings reinforce the role of cysteine in maintaining mitochondrial and cortical granule integrity and suggest its contribution to oocyte quality through improved subcellular organization and metabolic resilience. At the molecular level, HS downregulated key genes involved in antioxidant defense (SOD2 and GPX1), apoptosis inhibition (BCL2), oocyte competence (BMP15, GLUT1, and CCNB1), cumulus expansion (HAS2 and TNFAIP6), and mitochondrial function (TFAM), while upregulating the pro-apoptotic gene CASP3. These alterations reflect impaired developmental potential and increased apoptotic signaling under thermal stress. BCL2 is upregulated in camel oocytes and cumulus cells exposed to severe HS (Saadeldin et al., 2018). Similarly, under heat stress, bubaline oocytes exhibit downregulation of the antiapoptotic gene BCL2 (Khalil et al., 2023). Additionally, BCL2 was downregulated in bubaline 4- and 8-16 cell embryos that were developed from oocytes retrieved during the hot period (Sadeesh et al., 2016). CCNB1 regulation was altered in COCs that were thermally stressed, and their developmental competence was negatively affected (Stamperna et al., 2020). Studies on camels have also shown that high-quality camel oocytes express more BMP15 (Fathi et al., 2017). Different mitochondrion-related genes were considerably downregulated during summer in contrast to winter (Gendelman and Roth, 2012). Bovine oocytes recovered during the summer showed a significant decrease in TFAM, indicating a decline in fertility (Ferreira et al., 2016). Consistently, oxidative stress-induced CASP3 overexpression in rabbit oocytes has been reported (Arias-Alvarez et al., 2018). Cysteine supplementation reversed these effects, upregulating protective and functional genes and suppressing CASP3. These gene expression changes are consistent with the findings of previous studies that reported that the addition of cysteine and cystine to the culture medium significantly reduced DNA damage and caspase-3 activity in mature goat oocytes (Zhou et al., 2016). In addition, L-cysteine-treated bovine oocytes had significantly higher levels of BMP-15 and GDF-9 (Elgebaly et al., 2022), reinforcing the capacity of cysteine to preserve oocyte molecular integrity. Collectively, these findings confirm that cysteine acts not only as an antioxidant but also as a modulator of gene expression related to maturation, metabolism, and apoptosis. Its inclusion in IVM media significantly protects against heat-induced dysfunctions in dromedary camel oocytes. ConclusionSupplementation of in vitro maturation medium with 1.6 mM cysteine effectively mitigates the adverse effects of heat stress on dromedary camel oocytes. Cysteine enhances nuclear maturation and mitochondrial activity, restores oocyte ultrastructure and antioxidant defenses, and supports oocyte viability and developmental competence by modulating gene expression. These results suggest that cysteine is a promising additive for improving oocyte quality and resilience under thermal stress, offering practical applications in camel breeding and ART programs in hot climates. Nonetheless, the study was limited to a single heat stress condition and specific cysteine concentrations in vitro. Further research under diverse conditions and in vivo settings is warranted to confirm and extend these findings. AcknowledgmentsThe authors would like to thank all staff members at the Department of Theriogenology, Faculty of Veterinary Medicine, Zagazig University, for their support and assistance throughout this study. Conflict of interestThe authors report no conflicts of interest. FundingThis research did not receive any specific grant from public, commercial, or not-for-profit funding agencies. Authors' contributionsAyman Mesalam and Nasser Ghanem designed the experiments, Mohamed Eleam performed the experiments, Gamal Shawki wrote the first draft of the manuscript, Marwa El-Sheikh, Omar A. Farid, Beshoy S.F. Khalil, Fatma Badawy Aboelwafa, and Najmi Mariol provided assistance, and Ayman Mesalam analyzed the data and revised and edited the manuscript. Data availabilityThe data supporting the findings of this study are available from the corresponding author upon reasonable request. ReferencesAbdelkhalek, A.E., Gabr, S.A., Khalil, W.A., Shamiah, S.M., Pan, L., Qin, G. and Farouk, M.H. 2017. In vitro production of Sudanese camel (Camelus dromedarius) embryos from epididymal spermatozoa and follicular oocytes of slaughtered animals. Pol. J. Vet. Sci. 20, 95–101. Abdelnour, S.A., Yang, C.Y., Swelum, A.A., Abd El-hack, M.E., Khafaga, A.F., Abdo, M., Shang, J.H. and Lu, Y.Q. 2020. Molecular, functional, and cellular alterations of oocytes and cumulus cells induced by heat stress and shock in animals. Environ. Sci. Pollut. Res. Int. 27, 38472–38490. Ali, A.A., Bilodeau, J.F. and Sirard, M.A. 2003. Antioxidant requirements for bovine oocytes varies during in vitro maturation, fertilization and development. Theriogenology 59, 939–949. Andreu-Vázquez, C., López-Gatius, F., García-Ispierto, I., Maya-Soriano, M.J., Hunter, R.H.F. and López-Béjar, M. 2010. Does heat stress provoke the loss of a continuous layer of cortical granules beneath the plasma membrane during oocyte maturation?. Zygote 18, 293–299. Arias-Alvarez, M., Garcia-Garcia, R.M., Lopez-Tello, J., Rebollar, P.G., Gutierrez-Adan, A. and Lorenzo, P.L. 2018. Alpha-Tocopherol modifies the expression of genes related to oxidative stress and apoptosis during in vitro maturation and enhances the developmental competence of rabbit oocytes. Reprod. Fertil. Dev. 30, 1728–1738. Ashour, G., El-Sayed, A., Khalifa, M. and Ghanem, N. 2020. Effect of heat stress on developmental competence of in vitro matured oocytes of Camelus dromedaries with different qualities. World Vet. J. 10, 658–664. Azad, N., Iyer, A., Vallyathan, V., Wang, L., Castranova, V., Stehlik, C. and Rojanasakul, Y. 2010. Role of oxidative/nitrosative stress-mediated Bcl-2 regulation in apoptosis and malignant transformation. Ann. N. Y. Acad. Sci. 1203, 1–6. Barakat, I.A.H., Kaabi, A.M. and Alajmi, R.A. 2020. The role of honeybee pollen as a natural source of antioxidants in the in vitro maturation medium of sheep oocytes and its effect on gene expression. Environ. Sci. Pollut. Res. Int. 27, 31350–31356. Bavister, B.D. and Squirrell, J.M. 2000. Mitochondrial distribution and function in oocytes and early embryos. Hum. Reprod. 15, 189–198. Bello, A. and Bodinga, H. 2020. Common reproductive problem associated with one humped camel (Camelus dromedarius) in West Africa. Insights Vet. Sci. 4, 1–3. Bouayed, J. and Bohn, T. 2010. Exogenous antioxidants--Double-edged swords in cellular redox state: health beneficial effects at physiologic doses versus deleterious effects at high doses. Oxidative Med. Cellular Longevity 3, 228–237. Camargo, L.S.A., Aguirre-Lavin, T., Adenot, P., Araujo, T.D., Mendes, V.R.A., Louro, I.D., Beaujean, N. and Souza, E.D. 2019. Heat shock during in vitro maturation induces chromatin modifications in the bovine embryo. Reproduction 158, 313–322. Camargo, L.S.A., Costa, F.Q., Munk, M., Wohlres-Viana, S., Serapião, R.V., Carvalho, B.C., Campos, P.H., Vieira, A.C., Nogueira, L.A.G. and Viana, J.H.M. 2019. Contrasting effects of heat shock during in vitro maturation on development of in vitro-fertilized and parthenogenetic bovine embryos. Reprod. Domest. Anim. 54, 1357–1365. De Matos, D.G. and Furnus, C.C. 2000. The importance of having high glutathione (GSH) level after bovine in vitro maturation on embryo development: effect of β-mercaptoethanol, cysteine and cystine. Theriogenology 53, 761–771. El Sheikh, M., Mesalam, A.A., Idrees, M., Sidrat, T., Mesalam, A., Lee, K.L., Kong, I.K., 2020. Nicotinamide Supplementation during the In Vitro Maturation of Oocytes Improves the Developmental Competence of Preimplantation Embryos: Potential Link to SIRT1/AKT Signaling. Cells 9, 1550. Elgebaly, M.M., Hazaa, A.B.M., Amer, H.A. and Mesalam, A. 2022. L-Cysteine improves bovine oocyte developmental competence in vitro via activation of oocyte-derived growth factors BMP-15 and GDF-9. Reprod. Domest. Anim. 57, 734–742. El-Sheikh, M., Mesalam, A.A., El-Sayed, A.F., Mesalam, A., Metwally, H.M., Lee, S.H. and Kong, I.K. 2025. Nicotinamide dual treatment enhances morula-to-blastocyst transition through binding to Zonula Occludens-1 protein. Theriogenology 237, 110–119. Fair, T., Hulshof, S.C., Hyttel, P., Greve, T. and Boland, M. 1997. Oocyte ultrastructure in bovine primordial to early tertiary follicles. Anat. Embryol. (Berl). 195, 327–336. Fathi, M., Ashry, M., Salama, A. and Badr, M.R. 2017. Developmental competence of Dromedary camel (Camelus dromedarius) oocytes selected using brilliant cresyl blue staining. Zygote 25, 529–536. Ferreira, R.M., Chiaratti, M.R., Macabelli, C.H., Rodrigues, C.A., Ferraz, M.L., Watanabe, Y.F., Smith, L.C., Meirelles, F.V. and Baruselli, P.S. 2016. The Infertility of Repeat-Breeder Cows During Summer Is Associated with Decreased Mitochondrial DNA and Increased Expression of Mitochondrial and Apoptotic Genes in Oocytes. Biol. Reprod. 94, 66. Gendelman, M. and Roth, Z. 2012. Incorporation of coenzyme Q10 into bovine oocytes improves mitochondrial features and alleviates the effects of summer thermal stress on developmental competence. Biol. Reprod. 87, 118. Ghanem, N., Amin, A., Saeed, A.M., Abdelhamid, S.M., El-Sayed, A., Farid, O.A., Dessouki, S.M. and Faheem, M.S. 2020. Effects of curcumin supplementation on viability and antioxidant capacity of buffalo granulosa cells under in vitro culture conditions. World Vet. J. 10, 146–159. Gustina, S., Hasbi, H., Karja, N.W.K., Setiadi, M.A. and Supriatna, I. 2017. Ultrastructure changes in buffalo (Bubalus bubalis) oocytes before and after maturation in vitro with sericin. Anim. Sci. J. 88, 1911–1915. Hashimoto, S. 2009. Application of in vitro maturation to assisted reproductive technology. J. Reprod. Dev. 55, 1–10. Hoter, A., Rizk, S. and Naim, H.Y. 2019. Cellular and molecular adaptation of Arabian camel to heat stress. Front. Genet. 10, 588. Hu, X., Cheng, L., Wang, X., Luo, G., Zhao, T., Tian, J. and An, L. 2020. N-acetyl-l-cysteine protects porcine oocytes undergoing meiotic resumption from heat stress. Reprod. Toxicol. 91, 27–34. Jiang, Y., He, Y., Pan, X., Wang, P., Yuan, X. and Ma, B. 2023. Advances in Oocyte Maturation In Vivo and In Vitro in Mammals. Int. J. Mol. Sci. 24, 9059. Ju, J.C., Jiang, S., Tseng, J.K., Parks, J.E. and Yang, X. 2005. Heat shock reduces developmental competence and alters spindle configuration of bovine oocytes. Theriogenology 64, 1677–1689. Khalil, B.S.F., El-Assal, S.E.D. and Ghanem, N. 2023. Expression of heat shock and apoptosis genes in riverine buffalo (Bubalus bubalis) cumulus–oocyte complexes during in-vitro maturation under thermal stress conditions. Egypt. Pharm. J. 22, 361–371. Khan, I., Mesalam, A., Heo, Y.S., Lee, S.H., Nabi, G. and Kong, I.K. 2023. Heat Stress as a Barrier to Successful Reproduction and Potential Alleviation Strategies in Cattle. Animals (Basel). 2359, 2359. Khattab, A.K., Abu Elnaga, N.A., Shedeed, H.A., Kamel, A.M., Abd Rabu, M.A.B. and El-Bahrawy, K.A. 2020. Antioxidant supplementation effects on in vitro maturation and fertilization of dromedary camel oocytes. J. Camelid Sci. 13, 10–21. Kumar, P. 2021. Climate change and cities: challenges ahead. Front. Sustain. Cities 3, 645613. Lampiao, F. 2012. Free radicals generation in an in vitro fertilization setting and how to minimize them. World J. Obstet. Gynecol. 1, 29–34. Lee, S., Dobes, P., Marciniak, J., Mascellani Bergo, A., Kamler, M., Marsik, P., Pohl, R., Titera, D., Hyrsl, P. and Havlik, J. 2024. Phytochemical S-methyl-L-cysteine sulfoxide from Brassicaceae: a key to health or a poison for bees?. Open Biol. 14, 240219. Lonergan, P. and Fair, T. 2016. Maturation of Oocytes in Vitro. Annu. Rev. Anim. Biosci. 4, 255–268. Moawad, A.R., Ghoneim, I.M., Darwish, G.M., Badr, M.R., El-Badry, D.A. and El-Wishy, A.B.A. 2020. Factors affecting in vitro embryo production: insights into dromedary camel. J. Anim. Reprod. Biotechnol. 35, 119–141. Nabenishi, H., Ohta, H., Nishimoto, T., Morita, T., Ashizawa, K. and Tsuzuki, Y. 2012a. The effects of cysteine addition during in vitro maturation on the developmental competence, ROS, GSH and apoptosis level of bovine oocytes exposed to heat stress. Zygote 20, 249–259. Nabenishi, H., Takagi, S., Kamata, H., Nishimoto, T., Morita, T., Ashizawa, K. and Tsuzuki, Y. 2012b. The role of mitochondrial transition pores on bovine oocyte competence after heat stress, as determined by effects of cyclosporin A. Mol. Reprod. Dev. 79, 31–40. Pavani, K.C., Rocha, A., Oliveira, E., Da Silva, F.M. and Sousa, M. 2020. Novel ultrastructural findings in bovine oocytes matured in vitro. Theriogenology 143, 88–97. Rahim, B., Jalal, S. and Yosef, N., 2011. Effect of cysteine supplementation on in vitro maturation of bovine oocyte. Afr. J. Biotechnol. 10, 15830–15833 Rüsse, I. 1983. Oogenesis in cattle and sheep. Bibl. Anat. 24, 77–92. Saadeldin, I.M., Swelum, A.A.A., Elsafadi, M., Mahmood, A., Alfayez, M. and Alowaimer, A.N. 2018. Differences between the tolerance of camel oocytes and cumulus cells to acute and chronic hyperthermia. J. Therm. Biol. 74, 47–54. Sadeesh, E.M., Sikka, P., Balhara, A.K. and Balhara, S. 2016. Developmental competence and expression profile of genes in buffalo (Bubalus bubalis) oocytes and embryos collected under different environmental stress. Cytotechnology 68, 2271–2285. Sakatani, M., Yamanaka, K., Kobayashi, S. and Takahashi, M. 2008. Heat shock-derived reactive oxygen species induce embryonic mortality in in vitro early stage bovine embryos. J. Reprod. Dev. 54, 496–501. Schatten, H., Sun, Q.Y. and Prather, R. 2014. The impact of mitochondrial function/dysfunction on IVF and new treatment possibilities for infertility. Reprod. Biol. Endocrinol. 12, 1–11. Stamperna, K., Giannoulis, T., Nanas, I., Kalemkeridou, M., Dadouli, K., Moutou, K., Amiridis, G.S. and Dovolou, E. 2020. Short term temperature elevation during IVM affects embryo yield and alters gene expression pattern in oocytes, cumulus cells and blastocysts in cattle. Theriogenology 156, 36–45. Torner, H., Brüssow, K.P., Alm, H., Ratky, J., Pöhland, R., Tuchscherer, A. and Kanitz, W. 2004. Mitochondrial aggregation patterns and activity in porcine oocytes and apoptosis in surrounding cumulus cells depends on the stage of pre-ovulatory maturation. Theriogenology 61, 1675–1689. Tukur, H.A., Aljumaah, R.S., Swelum, A.A.A., Alowaimer, A.N. and Saadeldin, I.M. 2020. The making of a competent oocyte–a review of oocyte development and its regulation. J. Anim. Reprod. Biotechnol. 35(2), 2–11. Waiz, S.A., Raies-Ul-Haq, M., Dhanda, S., Kumar, A., Goud, T.S., Chauhan, M.S. and Upadhyay, R.C. 2016. Heat stress and antioxidant enzyme activity in bubaline (Bubalus bubalis) oocytes during in vitro maturation. Int. J. Biometeorol. 60, 1357–1366. Wang, L.Y., Wang, D.H., Zou, X.Y. and Xu, C.M. 2009. Mitochondrial functions on oocytes and preimplantation embryos. J. Zhejiang Univ. Sci. B 10, 483–492. Whitaker, B.D. and Knight, J.W. 2008. Mechanisms of oxidative stress in porcine oocytes and the role of anti-oxidants. Reprod. Fertil. Dev. 20, 694–702. Zhang, C.H., Wang, Z.B., Quan, S., Huang, X., Tong, J.S., Ma, J.Y., Guo, L., Wei, Y., Ouyang, Y.C., Hou, Y., Xing, F.Q. and Sun, Q.Y. 2011. GM130, a cis-Golgi protein, regulates meiotic spindle assembly and asymmetric division in mouse oocyte. Cell Cycle 10, 1861–1870. Zhang, T., Fan, X., Li, R., Zhang, C. and Zhang, J. 2018. Effects of pre-incubation with C-type natriuretic peptide on nuclear maturation, mitochondrial behavior, and developmental competence of sheep oocytes. Biochem. Biophys. Res. Commun. 497, 200–206. Zhou, Z., Jia, R.X., Zhang, G., Wan, Y., Zhang, Y., Fan, Y., Wang, Z., Huang, P. and Wang, F. 2016. Using cysteine/cystine to overcome oxidative stress in goat oocytes and embryos cultured in vitro. Mol. Med. Rep. 14, 1219–1226. | ||
| How to Cite this Article |
| Pubmed Style Ghanem N, Eleam M, Shawki G, El-sheikh M, Farid OA, Khalil BS, Aboelwafa FB, Mariol N, Mesalam A. Cysteine reduces oxidative damage induced by heat stress in dromedary camel oocytes during in vitro maturation. Open Vet. J.. 2025; 15(10): 5146-5156. doi:10.5455/OVJ.2025.v15.i10.32 Web Style Ghanem N, Eleam M, Shawki G, El-sheikh M, Farid OA, Khalil BS, Aboelwafa FB, Mariol N, Mesalam A. Cysteine reduces oxidative damage induced by heat stress in dromedary camel oocytes during in vitro maturation. https://www.openveterinaryjournal.com/?mno=269550 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.32 AMA (American Medical Association) Style Ghanem N, Eleam M, Shawki G, El-sheikh M, Farid OA, Khalil BS, Aboelwafa FB, Mariol N, Mesalam A. Cysteine reduces oxidative damage induced by heat stress in dromedary camel oocytes during in vitro maturation. Open Vet. J.. 2025; 15(10): 5146-5156. doi:10.5455/OVJ.2025.v15.i10.32 Vancouver/ICMJE Style Ghanem N, Eleam M, Shawki G, El-sheikh M, Farid OA, Khalil BS, Aboelwafa FB, Mariol N, Mesalam A. Cysteine reduces oxidative damage induced by heat stress in dromedary camel oocytes during in vitro maturation. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5146-5156. doi:10.5455/OVJ.2025.v15.i10.32 Harvard Style Ghanem, N., Eleam, . M., Shawki, . G., El-sheikh, . M., Farid, . O. A., Khalil, . B. S., Aboelwafa, . F. B., Mariol, . N. & Mesalam, . A. (2025) Cysteine reduces oxidative damage induced by heat stress in dromedary camel oocytes during in vitro maturation. Open Vet. J., 15 (10), 5146-5156. doi:10.5455/OVJ.2025.v15.i10.32 Turabian Style Ghanem, Nasser, Mohamed Eleam, Gamal Shawki, Marwa El-sheikh, Omar A. Farid, Beshoy S.f. Khalil, Fatma Badawy Aboelwafa, Najmi Mariol, and Ayman Mesalam. 2025. Cysteine reduces oxidative damage induced by heat stress in dromedary camel oocytes during in vitro maturation. Open Veterinary Journal, 15 (10), 5146-5156. doi:10.5455/OVJ.2025.v15.i10.32 Chicago Style Ghanem, Nasser, Mohamed Eleam, Gamal Shawki, Marwa El-sheikh, Omar A. Farid, Beshoy S.f. Khalil, Fatma Badawy Aboelwafa, Najmi Mariol, and Ayman Mesalam. "Cysteine reduces oxidative damage induced by heat stress in dromedary camel oocytes during in vitro maturation." Open Veterinary Journal 15 (2025), 5146-5156. doi:10.5455/OVJ.2025.v15.i10.32 MLA (The Modern Language Association) Style Ghanem, Nasser, Mohamed Eleam, Gamal Shawki, Marwa El-sheikh, Omar A. Farid, Beshoy S.f. Khalil, Fatma Badawy Aboelwafa, Najmi Mariol, and Ayman Mesalam. "Cysteine reduces oxidative damage induced by heat stress in dromedary camel oocytes during in vitro maturation." Open Veterinary Journal 15.10 (2025), 5146-5156. Print. doi:10.5455/OVJ.2025.v15.i10.32 APA (American Psychological Association) Style Ghanem, N., Eleam, . M., Shawki, . G., El-sheikh, . M., Farid, . O. A., Khalil, . B. S., Aboelwafa, . F. B., Mariol, . N. & Mesalam, . A. (2025) Cysteine reduces oxidative damage induced by heat stress in dromedary camel oocytes during in vitro maturation. Open Veterinary Journal, 15 (10), 5146-5156. doi:10.5455/OVJ.2025.v15.i10.32 |