| Research Article | ||
Open Vet. J.. 2025; 15(10): 5128-5135 Open Veterinary Journal, (2025), Vol. 15(10): 5128-5135 Research Article Assessment of proinflammatory cytokines and physiological parameters in male rats with L-arginine-induced enteritisSumayah Faruq Kasim1*, Muna H. AL-Saeed2 and Arwa H. M. AL-Saeed31College of Health and Medical Techniques, Middle Technical University, Baghdad, Iraq 2Department of Physiology, Pharmacology and Biochemistry, College of Veterinary Medicine, University of Basrah, Basrah, Iraq 3Department of Chemistry, College of Science, University of Basrah, Basrah, Iraq *Corresponding Author: Sumayah Faruq Kasim. College of Health and Medical Techniques, Middle Technical University, Baghdad, Iraq. Email: sumayah.faruq [at] mtu.edu.iq Submitted: 08/07/2025 Revised: 12/09/2025 Accepted: 21/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
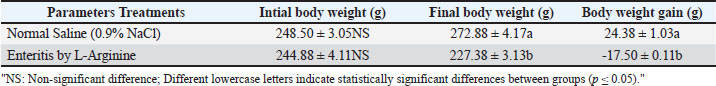
AbstractBackground: Enteritis is an inflammatory disorder of the intestine characterised by complex immunological responses and subsequent tissue destruction. L-arginine, a semi-essential amino acid involved in nitric oxide generation, demonstrates both protective and pro-inflammatory actions in varying settings. Nonetheless, its involvement in enteritis production and related inflammatory responses is not yet fully understood. Aim: This research investigated the long-term impacts of L-arginine administration in rats with enteritis, focussing on proinflammatory cytokines, serum calprotectin, haematological parameters, physiological changes, and histological alterations. Methods: Male rats received an oral administration of 500 mg/kg of L-arginine daily for a duration of 30 days to induce enteritis. The control group of rats was administered saline. Body weight, blood tests, and serum levels of tumour necrosis factor-alpha (TNF-α), Interleukin-6 (IL-6), calprotectin, and C-reactive protein (CRP) were evaluated. The small intestine and spleen were histologically examined to see how much tissue inflammation there was. Results: The administration of L-arginine to rats led to a notable decrease in body weight and haematological indices, such as red blood cell count, haemoglobin concentration, and packed cell volume. Levels of inflammatory markers TNF-α, IL-6, CRP, and calprotectin were significantly higher in comparison to control groups. Histological analysis demonstrated vascular congestion and perivascular lymphocytic infiltration in the gastrointestinal tract, as well as lymphoid hyperplasia and blood vessel congestion in the spleen, which indicates active inflammation. Conclusion: Extended administration of L-arginine in rats induces enteritis, an inflammatory condition affecting both systemic and localised areas. Proinflammatory cytokines and calprotectin correlate with biochemical and histological markers of inflammation, indicating their utility as biomarkers for the diagnosis and monitoring of enteritis. The findings indicate that L-arginine supplementation should be avoided in contexts associated with intestinal inflammation and underscore potential avenues for therapeutic intervention. Keywords: Calprotectin, Cytokines, Enteritis, Rat Model, L-Arginine. IntroductionIntestinal inflammation represents a multifaceted pathological process associated with various gastrointestinal diseases, frequently influenced by dysregulated immune responses and excessive production of proinflammatory cytokines. Interleukin-6 (IL-6) is a key mediator cytokine of the immune response and a regulator of many physiological and pathological processes. Pro-inflammatory cytokines are generally upregulated in inflammatory bowel disease (IBD) including tumour necrosis factor-alpha (TNFα) and IL-6 (Alhendi and Naser, 2023). IL-6 was first discovered as a soluble factor secreted by helper T cells to induce production of antibody and release by B cells. It was originally termed B-cell stimulatory factor-2 or B-cell differentiation factor (Hirano et al., 1986). The cytokine tumor necrosis factor- alpha is an crucial molecule involved in intestinal inflammation and barrier dysfunction (Suenaert, 2002). TNF-a has showed to increase permeability of intestine in patients with IBD by increased expression of epithelial myosin light chain kinase (Su et al., 2013). The blockers targeting TNF-a have become an invaluable part of the therapeutic armamentarium in IBD (El Mourabet et al., 2010). Inflammation constitutes a fundamental pathogenic mechanism linked to various diseases. The body activates the immune system to initiate inflammatory responses upon encountering harmful agents, involving viruses, bacteria, toxic chemicals, or tissue injury. The responses entail intricate interactions between inflammatory cells and endothelial cells, which constitute the monolayer lining the interface between blood and tissues. Endothelial cells serve as primary targets for pathological stimuli and exhibit rapid responses to local alterations caused by inflammatory factors (Medzhitov, 2010). L-arginine (Arg) is a semi-essential amino acid produced from glutamine, glutamate, as well as proline through the cycle of urea, particularly via the intestinal-renal axis in mammals including rats. It plays a critical role in synthesis of protein and serves as nitric oxide synthase substrate to generate nitric oxide (NO), a key vascular protective molecule released by the endothelial cells (Wu et al., 2009). Because L-arginine has both good and bad effects on the body, its effects on intestinal inflammation are still not well understood. The goal of this study is to find out if serum calprotectin can be used as a reliable marker of intestinal inflammation in a rat model of L-arginine-induced enteritis. We also compare serum calprotectin levels to other known inflammatory markers, such as C-reactive protein (CRP), (TNF-α), and interleukin-6 (IL-6), as well as check other important physiological parameters. Materials and MethodsInduction of experimental enteritisAdult Wistar male rats, 2 months age, weighing 200–250 g, were obtained from animal house of College of Veterinary Medicine, University of Basrah, and housed under controlled laboratory conditions with a 12-hour light/dark cycle, with free access to food and water. The animals were allowed to acclimate for 15 days prior to the experiment. Rats were randomly divided into two groups: the experimental group (n=10), which received L-arginine, and the control group (n=10), which received normal saline. Enteritis was induced in the experimental group by oral administration of L-arginine dissolved in normal saline at a dose of 500 mg/kg body weight, given orally once daily for 30 consecutive days. The control group received an equivalent volume (1 ml) of physiological saline orally for the same duration. Assessment physiological parametersMeasurement of body weight and body weight gainThe body weight of each rat was recorded at baseline (prior to the start of the experiment) and at the end of the treatment period, following the method described by Al-Saeed (2012). Body weight gain was calculated as the difference between the final and initial body weights. Hematological testesAt the end of the experiment period, blood samples were collected via cardiac puncture and analyzed using a Count 60 hematology analyzer (Genex Laboratories, Germany). The following parameters were measured: red blood cell (RBC) count, white blood cell (WBC) count, hemoglobin concentration (Hb), packed cell volume (PCV), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), and differential white blood cell count (D WBC). Assessment of inflammatory markersBlood samples for the measurement of inflammatory and activity markers, including TNF-α, IL-6, serum calprotectin, and CRP, were collected on day 30. Following sample collection, the rats were sacrificed on the same day. Serum concentrations of IL-6, TNF-α, and calprotectin were determined using commercially available rat-specific ELISA kits (Immundiagnostik, Germany) according to the manufacturers’ instructions. Serum CRP levels were also measured using an ELISA kit standardized for rat CRP. All assays were performed in duplicate to ensure accuracy and reliability of the results. Histological assessmentAt the end of the study (day 30), rats were sacrificed following serum sample collection. Organs including the small intestine and spleen were harvested, opened, and immediately fixed in 10% neutral buffered formalin. After fixation, tissues were embedded in paraffin, sectioned into 4–5 µm thick slices, deparaffinized in xylene, rehydrated through graded alcohols, and stained with hematoxylin and eosin (H&E) stain for microscopic examination. Histological slides were then examined to assess morphological changes associated with L-arginine-induced enteritis and any pathological alterations in the spleen. Statistical analysisData were initially tested for normality using the Anderson-Darling test (Anderson and Darling, 1954). Subsequent statistical comparisons between the experimental and control groups were performed using the student’s t-test. All analyses were conducted using SPSS software (version 25). A p-value of less than 0.05 was considered statistically significant. Ethical approvalAll procedures adhered to ethical guidelines for the care and use of laboratory animals and received approval from the Faculty of Veterinary Medicine, University of Basrah, No. (84/37/2025). ResultsBody weight and body weight gainTable 1 shows that the enteritis group exhibited a significant decrease (p ≤ 0.05) in both final body weight and overall body weight gain compared to the controls. Table 1. Mean of body weight and body weight gain between studied groups.
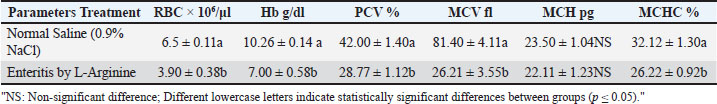
RBC counts and indicesAs presented in Table 2, the enteritis group showed a significant decrease (p ≤ 0.05) in red blood cell count, Hb, PCV, MCV, and MCHC relative to controls. Table 2. Mean of RBC counts and RBC indices between studied groups.
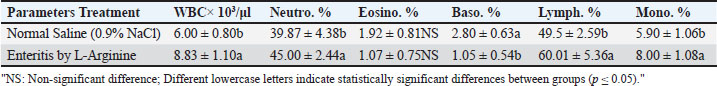
WBC counts and differentialsTable 3 indicates a significant increase (p ≤ 0.05) in total white blood cell count, neutrophils, monocytes, and lymphocytes in the enteritis group compared to the control group, whereas basophil percentages significantly decreased (p ≤ 0.05). Eosinophil levels showed no significant change (p > 0.05). Table 3. Mean of WBC counts and percentage of differential count of WBC between studied groups.
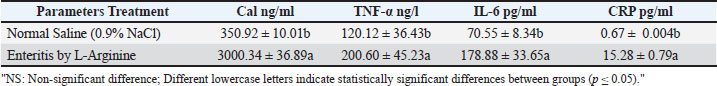
Serum inflammatory markersSerum levels of calprotectin, IL-6, TNF-α, and CRP were significantly elevated (p ≤ 0.05) in the enteritis group compared to controls (Table 4). Table 4. Mean of Calprotectin, IL-6, TNF-α and CRP between studied groups.
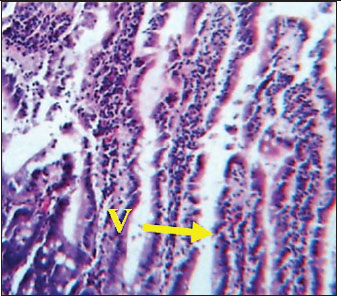
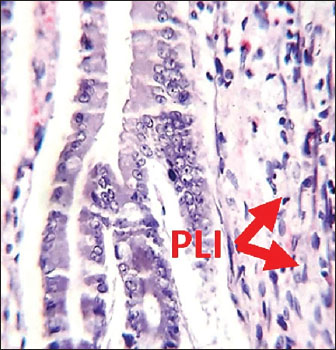
Histological analysis of intestineSections from rats treated with L-arginine showed clear histopathological alterations. Notably, perivascular lymphocytic infiltration was observed, along with the presence of plasma cells (Fig. 2), indicating an inflammatory response. The integrity of the mucosal structure appeared disrupted compared to the control, indicating tissue injury and immune cell recruitment as a result of L-arginine administration. In contrast, the intestinal section from male rats in the control group demonstrated normal histoarchitecture. The villi (V) in (Fig. 1) appeared long and slender, extending deeply into the tunica mucosa. The epithelial lining and lamina propria appeared intact, with no evident signs of cellular infiltration or tissue disruption.
Fig. 1. Histological section of male rat small intestine of control group, stained with Hematoxylin and Eosin (H&E), showing villus structure at 40x magnification using light microscopy.
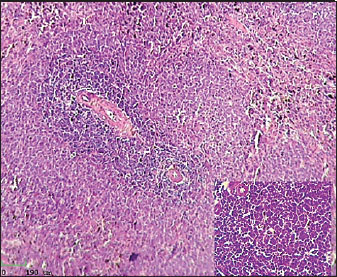
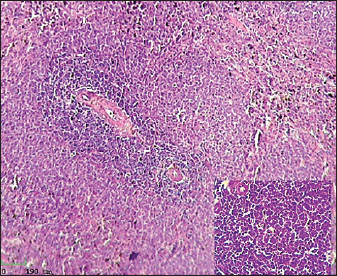
Fig. 2. Histological section of male rat small intestine treated with L-arginine stained with Hematoxylin and Eosin (H&E), showing villus structure at 40x magnification using light microscopy. Histological analysis of spleenHistological examination of the splenic tissue of rats treated with L-arginine showed notable pathological alterations. These included: Congestion of blood vessels within both the red and white pulp and lymphoid hyperplasia in the white pulp, indicating increased proliferation of lymphoid cells, suggestive of immune stimulation or chronic inflammation (Fig. 4). In contrast, the spleen from control male rats exhibited normal histological architecture. The white pulp and red pulp regions were clearly distinguishable. Central arteries and blood vessels appeared unremarkable, and no signs of congestion, inflammation, or hyperplasia were observed. The lymphoid follicles within the white pulp were of normal size and density (Fig. 3)
Fig. 3. Histological section of male rat spleen of control group, stained with Hematoxylin and Eosin (H&E), showing villus structure at 10x magnification using light microscopy.
Fig. 4. Histological section of male rat spleen treated with L-arginine, stained with Hematoxylin and Eosin (H&E), showing villus structure at 10x magnification using light microscopy. DiscussionThis study investigated the effects of chronic L-arginine administration on inflammatory responses, hematological indices, physiological parameters, and histological changes of intestine and spleen in a rat model of enteritis. The results demonstrate that L-arginine induces significant intestinal inflammation, characterized by alterations in body weight, blood parameters, inflammatory biomarkers, and tissue morphology. The significant reduction in body weight and weight gain observed in the enteritis group aligns with previous findings that chronic inflammation and intestinal damage impair nutrient absorption and metabolism, leading to weight loss (Ebohon et al., 2020). Weight fluctuations act as a precise sign of overall health and the intensity of the inflammatory response. Haematological study indicated anemia-like characteristics in L-arginine-treated rats, evidenced by reduced RBC count, haemoglobin concentration, packed cell volume, and RBC indices. These alterations are likely attributable to inflammatory enteritis impacting erythropoiesis and perhaps leading to increased red blood cell destruction or loss (Weiss et al., 2019). IL-6 also controls iron and zinc levels by inducing hepcidin and zinc transporters, leading to anemia and hypozincemia in chronic inflammation (Nemeth et al., 2004; Liuzzi et al., 2005). In bone marrow, IL-6 promotes platelet production (Ishibashi et al., 1989). Neutrophils and monocytes play an important role in intestinal inflammation as they are actively recruited to inflamed tissue, where they help the immune system as well as the inflammatory response. Neutrophils maintain gut health by discarding bacteria that penetrate the epithelial barrier. Nevertheless, they perhaps exacerbate inflammation by the release of cytokines as well as reactive oxygen species (Kang et al., 2022). Monocytes differentiate to macrophages and dendritic cells, changing immune system physiology in mucous membranes and exacerbating chronic inflammation in circumstances such as inflammatory bowel disease (Liao et al., 2024; Lu et al., 2024; Long et al., 2024). Basophils show distinct function in the context of allergic inflammation that result in increased therapeutic potency, basophils can be recruited to sites of inflammation where they release histamine, cytokines, and other mediators that amplify immune responses (Siracusa et al., 2013). A significant decrease in basophil percentages, as observed in our enteritis model, might reflect their migration from the bloodstream into inflamed tissues, leading to peripheral blood basopenia. The observed non-significant changes in eosinophil counts, despite notable alterations in other white blood cell populations, likely reflect the specific immunopathological features of L-arginine-induced enteritis. This enteritis model may engage distinct immune regulatory pathways that predominantly recruit neutrophils and monocytes, unlike other chronic inflammatory models where basophils and eosinophils are actively involved. This differential recruitment could be due to the particular cytokine milieu or tissue microenvironment induced by L-arginine, which favours neutrophil- and monocyte-mediated responses over eosinophil or basophil activation. Further mechanistic studies would be needed to elucidate these immunoregulatory distinctions fully. Markers of serum inflammatory such as TNF-α, IL-6, and CRP were significantly increased, consistent with their established roles as mediators and biomarkers of intestinal inflammation (Neurath, 2014). The key role of cytokines is also highlighted by the fact that blockade of tumour necrosis factor is commonly used as a standard therapy for intestine bowel disease in the clinic (Danese and Fiocchi, 2011). In pathological conditions such as IBD, elevated TNF-α contributes to epithelial barrier dysfunction and tissue injury, making it a prime therapeutic target (Neurath, 2014). Thus, TNF-α is a key pro-inflammatory cytokine produced primarily by activated macrophages and plays a central role in initiating and sustaining the inflammatory cascade within the intestinal mucosa. It promotes leukocyte recruitment, activates endothelial cells, and induces the production of other cytokines and chemokines, thereby amplifying the immune response. IL-6 plays a central role in inflammation and immunity. Produced early at inflammation sites, it triggers the liver to produce acute phase proteins like CRP and serum amyloid A, while reducing others such as albumin (Heinrich et al., 1990). Prolonged high levels of serum amyloid A can cause amyloidosis, damaging organs (Gillmore et al., 2001). Thus, IL-6 is a pleiotropic cytokine involved in the acute phase response and regulation of immune cell differentiation and survival. Persistent elevation of IL-6 correlates with disease severity and progression in intestinal inflammatory disorders. C-reactive protein is a homopentameric acute-phase inflammatory protein that shows elevated expression through inflammatory cases such as some cardiovascular diseases, rheumatoid arthritis, and infection. There is evidence that CRP does more than just show when there is inflammation or an infection. It also plays a big part in controlling inflammatory processes. The main parts of inflammation and the body's reaction to infection that CRP helps with are the complement pathway, apoptosis, phagocytosis, release of nitric oxide, and production of cytokines (Sproston and Ashworth, 2018). Thus, CRP not only shows that the body is inflamed, but it also helps the body's defence system by encouraging complement activation and the opsonisation of germs and damaged cells. The amounts show how bad the inflammation is and can be used to predict how well a treatment will work for intestinal inflammation. Since that we argued about these physiological aspects of TNF-α, IL-6, and CRP, it became clear how important these molecules are, not only as markers but also as active players in the development of the gut inflammation observed in the present investigation. Calprotectin levels were very high and strongly linked to histological results, suggesting that it could be used as a biomarker to check for disease activity and inflammation in the intestines (Khaki-Khatibi et al., 2020). Calprotectin binds to Toll-like receptor 4 and is released by neutrophils and monocytes the moment they are triggered. This helps make the inflammation processes stronger in enteritis. According to Wang et al. (2025) a histological study, L-arginine caused problems in the model rats' small intestine and the spleen Plasma cells in the gut lining, clogged blood vessels, and perivascular lymphocytic infiltration are all signs of continued inflammation and the recruitment of immune cells. Lymphoid hyperplasia of the spleen is when there are more lymphocytes than usual. It usually happens because of an inflammatory or immune response. This finding usually means that the immune system is working harder or longer in the spleen, which shows that the immune system is still controlling the body as a whole. This kind of hypertrophy is a sign of an active immune system or long-term inflammation (Hobbie et al., 2024). During enteritis, the spleen is an important part of the immune system because it filters the blood and is where immune cells first become active. According to Mebius and Kraal (2005). There is a white pulp in the spleen that helps activate and multiply lymphocytes. This makes it possible for adaptive immune reactions to work together. In the context of enteritis, inflammation in the intestines can cause microbial products and inflammatory chemicals to move into the bloodstream. The spleen is very important for getting rid of these substances and keeping the defense system working as a whole. This shows how important the spleen is for keeping the immune system in balance during times of inflammation in the gut. It also helps control inflammation outside of the gut and stops infections from spreading. L-arginine may make rats more prone to inflammation by changing the immune system and working as a building block for nitric oxide. Some people think that taking L-arginine supplements could improve the function of endothelial cells and improve the health of circulatory systems (Wu et al., 2009), Potentially through mechanisms associated with nitric oxide and oxidative stress, prolonged or excessive administration appears to disrupt intestinal homeostasis (Schmitt and Melk, 2017; Donato et al., 2018). In conclusion, L-arginine plays a multifaceted role in the function of the intestines, acting as both an important regulator of the immune system and a possible cause of inflammation in the intestines. An amino acid called L-arginine is semi-essential because it is a key building block for making NO. The molecule NO is an important signaling one that helps keep the blood flowing through the mucosa, protects the integrity of the epithelium, and controls immune functions in the digestive system. Taking extra L-arginine has been shown to strengthen the gut barrier, lower oxidative stress, and speed up the healing of damaged mucosa in a number of animal models. At the same time, giving too much or too long of an injection of L-arginine can make inflammation worse by increasing the production of NO above normal amounts. This causes oxidative stress and tissue damage. The paradoxical effect has been documented in experimental models, shown that this strange effect happens when high amounts of L-arginine are given. These doses cause enteritis, which is characterised by immune cells entering the gut and damaging the mucosa. This result shows a point at which L-arginine stops being helpful and starts being harmful in the intestinal environment. The results show how important it is to carefully consider dosage and length when giving L-arginine supplements, especially in medical situations where the gut is inflamed or not working properly. ConclusionThis study revealed that prolonged administration of L-arginine to male rats induces enteritis and elevates levels of various inflammatory cytokines, involving serum calprotectin along with CRP. In addition, these markers of inflammation were also strongly correlated with the biochemical and histopathological changes that were noted in the small intestine and spleen. Furthermore, to the negative impacts in body weight, there were also hematological changes indicating that the ‘arginine-induced’ intestinal inflammation had an effect on overall physiology. Calprotectin showed the most promise as a predictor of inflammation in the intestines as it tended to remain elevated after the acute phase of inflammation subsided, paralleling its association with inflammatory bowel diseases. The described elevated congestion of blood vessels, increase in white blood cells, and increase in tissue of the lymphatic system gives support to the claim L-arginine is pro-inflammatory substances were harmful in this experimental situation. This study thus demonstrates the potential of pro-inflammatory cytokines as well as calprotectin for diagnosis and monitoring of enteritis. It also reinforces the idea that supplementation of L-arginine might need to be considered with more caution, particularly in the case of predisposing or complexing conditions with intestinal inflammation. Future study should aim to clarify the molecular underpinnings of L-arginine-induced inflammation and investigate therapeutic approaches to alleviate its negative consequences. AcknowledgmentsThe authors express their gratitude to the College of Veterinary Medicine, University of Basrah, for their significant support and for supplying the necessary facilities to conduct this research. We are especially grateful to the Department of Physiology, Pharmacology, and Biochemistry for providing access to the animal house and laboratory resources, which were essential for the effective completion of the study. FundingSelf funding. Conflict of interestThe authors want to be certain that it clear that there are no conflicts of interest to report. Authors' contributionsAll authors contributed significantly to the conception and design of the study, Muna H. AL-Saeed and Arwa H. M. AL-Saeed were performed the experimental work and collected the data, Sumayah Faruq Kasim drafted the manuscript. Muna H. AL-Saeed assisted in data analysis and interpretation. Sumayah Faruq Kasim critically revised the manuscript for important intellectual content. All authors read and approved the final version of the manuscript before submission. Data availabilityAvailable. ReferencesAL-Saeed, M.H. 2012. Hypothyroidic effects of soybean isoflavonoid, carbimazole and dexamethasone and the role of zinc sulfate in ameliorating their effects in female rabbits (Lepus cuniculus domastica). Thesis of Ph.D. in physiology in College of Vet. Med. Un. of Basrah. Iraq. Alhendi, A. and Naser, S.A. 2023. The dual role of interleukin-6 in Crohn’s disease pathophysiology. Front. Immunol. 14, 1295230; doi:10.3389/fimmu.2023.1295230 Anderson, T.W. and Darling, D.A. 1954. A test of goodness of fit. J. Am. Stat. Assoc. 49(268), 765–769; doi:10.1080/01621459.1954.10501232 Danese, S. and Fiocchi, C. 2011. Ulcerative colitis. New Engl. J. Med. 365, 1713–1725. Donato, A.J., Machin, D.R. and Lesniewski, L.A. 2018. Mechanisms of dysfunction in the aging vasculature and role in age-related disease. Circ. Res. 123, 825–848. Ebohon, O., Irabor, F. and Omorregie, E.S. 2020. Sub-acute toxicity Study of methanol extract of Tetrorchidium didymstemon leaves using biochemical analyses and gene expression in Wistar rats. Heliyon 6(6), 4313. El Mourabet, M., El-Hachem, S., Harrison, J.R. and Binion, D.G. 2010. Anti-TNF antibody therapy for inflammatory bowel disease during pregnancy: a clinical review, Curr. Drug Targets. 11, 234–241. Gillmore, J.D., Lovat, L.B., Persey, M.R., Pepys, M.B. and Hawkins, P.N. 2001. Amyloid load and clinical outcome in AA amyloidosis in relation to circulating concentration of serum amyloid A protein. Lancet 358, 24–29. Heinrich, P.C., Castell, J.V. and Andus, T. 1990. Interleukin-6 and the acute phase response. Biochem. J. 265, 621–636. Hirano, T., Yasukawa, K., Harada, H., Taga, T., Watanabe, Y., Matsuda, T., Kashiwamura, S.I., Nakajima, K., Koyama, K., Iwamatsu, A., Tsunasawa, S., Sakiyama, F., Matsui, H., Takahara, Y., Taniguchi, T. and Kishimoto, T. 1986. Complementary DNA for a novel human interleukin (BSF-2) that induces B lymphocytes to produce immunoglobulin. Nature 324(6092), 73–76; doi:10.1038/324073a0 Hobbie, K., Elmore, S.A. and Kolenda-Roberts, H.M. 2024. Nonneoplastic Lesion Atlas (A guide for standardizing terminology in toxicologic pathology for rodents). Immune System: Spleen - Hyperplasia, Lymphocyte. Available via https://ntp.niehs.nih.gov/atlas/nnl/immune-system/spleen/HyperplasiaLymphocyte (Accessed 10 January 2025). Ishibashi, T., Kimura, H., Shikama, Y., Uchida, T., Kariyone, S., Hirano, T., Kishimoto, T., Takatsuki, F. and Akiyama, Y. 1989. Interleukin-6 is a potent thrombopoietic factor in vivo in mice. Blood 74, 1241–1244. Kang, L., Fang, X., Song, Y.H., He, Z.X., Wang, Z.J., Wang, S.L., Li, Z.S. and Bai, Y. 2022. Neutrophil–epithelial crosstalk during intestinal inflammation. Cellular Mol. Gastroenterol. Hepatol. 14(6), 1257–1267; doi:10.1016/j.jcmgh.2022.09.002 Khaki-Khatibi, F., Qujeq, D., Kashifard, M., Moein, S., Maniati, M. and Vaghari-Tabari, M. 2020. Calprotectin in inflammatory bowel disease. Int. J. Clin. Chem. 510, 556–565; doi:10.1016/j.cca.2020.08.025 Liao, X., Liu, J., Guo, X., Meng, R., Zhang, W., Zhou, J., Xie, X. and Zhou, H. 2024. Origin and function of monocytes in inflammatory bowel disease. J. Inflammation Res. 17, 2897–2914; doi:10.2147/JIR.S450801 Liuzzi, J.P., Lichten, L.A., Rivera, S., Blanchard, R.K., Aydemir, T.B., Knutson, M.D., Ganz, T. and Cousins, R.J. 2005. Interleukin-6 regulates the zinc transporter Zip14 in liver and contributes to the hypozincemia of the acute-phase response. Proc. Natl. Acad. Sci. 102, 6843–6848. Long, D., Mao, C., Xu, Y. and Zhu, Y. 2024. The emerging role of neutrophil extracellular traps in ulcerative colitis. Front. Immunol. 15, 1425251; doi: 10.3389/fimmu.2024.1425251 Lu, H., Suo, Z., Lin, J., Cong, Y. and Liu, Z. 2024. Monocyte-macrophages modulate intestinal homeostasis in inflammatory bowel disease. Biomark. Res. 12, 76; doi:10.1186/s40364-024-00612-x Mebius, R.E. and Kraal, G. 2005. Structure and function of the spleen. Nature Rev. Immunol. 5(8), 606–616; doi:10.1038/nri1669 Medzhitov. 2010. Inflammation: new adventures of an old flame. Cell 140, 771–776. Mourabet, M., El-Hachem, S., Harrison, J. and Binion, D. 2010. Anti-TNF antibody therapy for inflammatory bowel disease during pregnancy: a clinical review. Curr. Drug Targets 11, 234–241. Nemeth, E., Rivera, S., Gabayan, V., Keller, C., Taudorf, S., Pedersen, B.K. and Ganz, T. 2004. IL-6 mediates hypoferremia of inflammation by inducing the synthesis of the iron regulatory hormone hepcidin. J. Clin. Invest. 113, 1271–1276. Neurath, M.F. 2014. Cytokines in inflammatory bowel disease. Nature Rev. Immunol. 14(5), 329–342; doi:10.1038/nri3661 Schmitt, R. and Melk, A. 2017. Molecular mechanisms of renal aging. Kidney Int. 92, 569–579. Siracusa, M.C., Kim, B.S., Spergel, J.M. and Artis, D. 2013. Basophils and allergic inflammation. J. Allergy Clin. Immunol. 132(4), 789–801; doi:10.1016/j.jaci.2013.07.046 Sproston, N.R. and Ashworth, J.J. 2018. Role of C-Reactive Protein at Sites of Inflammation and Infection. Front. Immunol. 9, 754; doi: 10.3389/fimmu.2018.00754 Su, L., Nalle, S.C., Shen, L., Turner, E.S., Singh, G., Breskin, L.A., Khramtsova, E.A., Khramtsova, G., Tsai, P.Y., Fu, Y.X., Abraham, C. and Turner, J.R. 2013. TNFR2 activates MLCK-dependent tight junction dysregulation to cause apoptosis-mediated barrier loss and experimental colitis. Gastroenterology 145, 407–415. Suenaert, P. 2002. Anti-tumor necrosis factor treatment restores the gut barrier in Crohn’s disease. Am. J. Gastroenterol. 97, 2000–2004. Wang, S.N., Wang, Y.K., Li, P., Kong, L.C., Deng, W.Y., Jiang, B. and Li, Y.Y. 2025. Immune cell crisis and excess histopathological features during the development and progression of H. pylori infection in the gastric mucosa. Int. J. Gen. Med. 18, 1345–1357; doi:10.2147/IJGM.S474925 Weiss, G., Ganz, T. and Goodnough, L.T. 2019. Anemia of inflammation. Blood 133(1), 40–50; doi:10.1182/blood-2018-06-856500 Wu, G., Bazer, F.W., Davis, T.A., Kim, S.W., Li, P., Marc Rhoads, J., Carey Satterfield, M., Smith, S.B., Spencer, T.E. and Yin, Y. 2009. Arginine metabolism and nutrition in growth, health and disease. Amino Acids 37(1), 153–168. | ||
| How to Cite this Article |
| Pubmed Style Kasim SF, Al-saeed MH, Al-saeed AHM. Assessment of proinflammatory cytokines and physiological parameters in male rats with L-arginine-induced enteritis. Open Vet. J.. 2025; 15(10): 5128-5135. doi:10.5455/OVJ.2025.v15.i10.30 Web Style Kasim SF, Al-saeed MH, Al-saeed AHM. Assessment of proinflammatory cytokines and physiological parameters in male rats with L-arginine-induced enteritis. https://www.openveterinaryjournal.com/?mno=269755 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.30 AMA (American Medical Association) Style Kasim SF, Al-saeed MH, Al-saeed AHM. Assessment of proinflammatory cytokines and physiological parameters in male rats with L-arginine-induced enteritis. Open Vet. J.. 2025; 15(10): 5128-5135. doi:10.5455/OVJ.2025.v15.i10.30 Vancouver/ICMJE Style Kasim SF, Al-saeed MH, Al-saeed AHM. Assessment of proinflammatory cytokines and physiological parameters in male rats with L-arginine-induced enteritis. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5128-5135. doi:10.5455/OVJ.2025.v15.i10.30 Harvard Style Kasim, S. F., Al-saeed, . M. H. & Al-saeed, . A. H. M. (2025) Assessment of proinflammatory cytokines and physiological parameters in male rats with L-arginine-induced enteritis. Open Vet. J., 15 (10), 5128-5135. doi:10.5455/OVJ.2025.v15.i10.30 Turabian Style Kasim, Sumayah Faruq, Muna H. Al-saeed, and Arwa H. M. Al-saeed. 2025. Assessment of proinflammatory cytokines and physiological parameters in male rats with L-arginine-induced enteritis. Open Veterinary Journal, 15 (10), 5128-5135. doi:10.5455/OVJ.2025.v15.i10.30 Chicago Style Kasim, Sumayah Faruq, Muna H. Al-saeed, and Arwa H. M. Al-saeed. "Assessment of proinflammatory cytokines and physiological parameters in male rats with L-arginine-induced enteritis." Open Veterinary Journal 15 (2025), 5128-5135. doi:10.5455/OVJ.2025.v15.i10.30 MLA (The Modern Language Association) Style Kasim, Sumayah Faruq, Muna H. Al-saeed, and Arwa H. M. Al-saeed. "Assessment of proinflammatory cytokines and physiological parameters in male rats with L-arginine-induced enteritis." Open Veterinary Journal 15.10 (2025), 5128-5135. Print. doi:10.5455/OVJ.2025.v15.i10.30 APA (American Psychological Association) Style Kasim, S. F., Al-saeed, . M. H. & Al-saeed, . A. H. M. (2025) Assessment of proinflammatory cytokines and physiological parameters in male rats with L-arginine-induced enteritis. Open Veterinary Journal, 15 (10), 5128-5135. doi:10.5455/OVJ.2025.v15.i10.30 |