| Research Article | ||
Open Vet. J.. 2025; 15(10): 5136-5145 Open Veterinary Journal, (2025), Vol. 15(10): 5136-5145 Research Article The promoting role of aluminum oxide nanoparticles in enhancing Pseudomonas aeruginosa antigen immunogenicityHassan H. K. AL-Bayati1*, Muthanna A. Sultan2, Sanaa S. Ahmed2, Bashar S. Noomi2 and Thamer J. Shihab11Department of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Tikrit, Tikrit, Iraq 2Department of Microbiology, College of Veterinary Medicine, Tikrit University, Tikrit, Iraq *Corresponding Author: Hassan H. K. Al-Bayati. Department of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Tikrit, Tikrit, Iraq. Email: dr_patho80 [at] tu.edu.iq Submitted: 13/07/2025 Revised: 11/09/2025 Accepted: 18/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
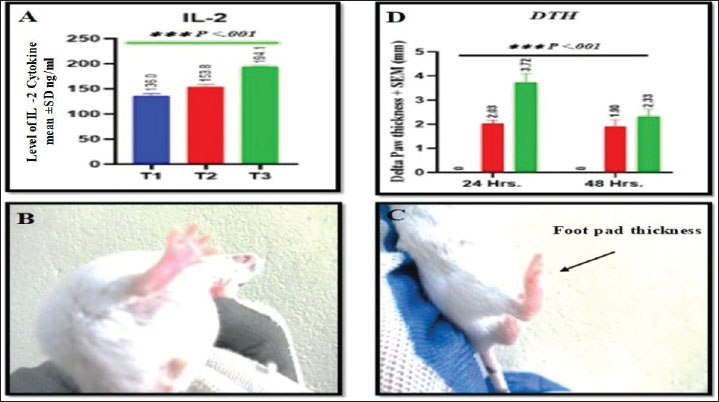
AbstractBackground: Pseudomonas aeruginosa is considered one of the major opportunistic bacteria that cause infections in both animals and humans. It is a significant problem in global health care due to its high inherent resistance to numerous antibiotics. Pathologic microorganisms are resistant to the number of available treatments, highlighting the pressing need for efficient immunization and immunotherapeutic approaches. Aim: Data regarding the use of nanoparticles as delivery are limited; therefore, we sought to develop an adjuvant vaccine to lessen the chance of developing a P. aeruginosa infection. Methods: Thirty mice were randomly assigned to three groups: T1 (Naïve control), T2 (non-nave control), and T3 (non-nave control) received two intraperitoneal (IP) doses (2-week interval) of 0.5 mg/ml of killed whole sonicated P. aeruginosa antigens (KWSPAgs) and two IP doses (2-week interval) of 1 mg/ml of KWSPAgs and aluminum oxide nanoparticles. At 14 days post-immunization, cell-mediated immunity was evaluated by delayed-type hypersensitivity, and at day 28, humoral immunity was assessed via Enzyme-Linked Immune Sorbent Assay to measure serum levels of Immunoglobulins (IgG), toll-like receptor 2 (TLR2), and IL-2. All groups were challenged IP with P. aeruginosa (0.2 × 107 CFU/ml). After 14 days post-immunization, mice were sacrificed for bacterial isolation, and internal organs were collected for histopathological and immunohistochemical analysis. Results: Pseudomonas aeruginosa antigens were sufficient to elicit immunogenicity. Moreover, the use of Nanoparticles (NPs) markedly enhanced immunogenicity through the delivery of KWSPAgs. Our results have shown that group with NP with KWSPAgs were able to induce inflammation and cellular immunity by increasing interleukin 2 and foot pad thickness, both of which are indicators of T lymphocyte proliferation. In addition, our data indicated that group with NP were able to induce humoral immunity by increasing the IgG and TLR2 levels in the immunized groups compared with Naïve control. The histopathological lesions and immune-positive cells in the infected and non-infected groups were different. Lesions were significantly higher in the infected group than in the noninfected control group at the observed time points. Conclusion: Overall, these findings highlight that NPs are a good activator for inducing cellular and humoral immunity, enhancing the delivery of future vaccine designs and/or target therapies against P. aeruginosa. Keywords: Aluminum oxide, Bacterial vaccine, Immune responses, Immunohistochemistry, Phonological changes. IntroductionSince the onset of the AMR crisis, modern medicine has turned to innovative technologies to avert the looming threat of a post-antimicrobial era. Among these advances, nanotechnology has emerged as a promising frontier, with researchers investigating the uses of nanoparticles as an approach showing remarkable potential in the development of next-generation antimicrobial agents. Pseudomonas aeruginosa is an opportunistic pathogen that causes both acute and chronic diseases (Thi et al., 2020). The Gram-negative bacterium can cause nosocomial infections and deadly infections in immunocompromised people, such as patients recovering from surgery, and HIV. (Gomila et al., 2018, Laking et al., 2025). Pseudomonas aeruginosa is frequently found in a variety of environments, including indoor settings, such as hospitals, and outdoor settings, such as parks. Because of its natural tendency to thrive in moist environments, this bacterium produces a large number of enzymes that increase its infection potential as a primary and secondary cause (Gaur et al., 2025). Over the past 10 years, P. aeruginosa has been easily grown and identified in microbiology labs, and the World Health Organization has reported that tens of millions of dollars have been spent to better understand the relationship between virulence factors and the human pathogen (Reynolds and Kollef, 2021). Consequently, there has been a recent push to improve therapeutic strategies and develop a vaccine (Crousilles et al., 2015). Nanoparticles (NPs) have been applied in medicine, and drug delivery is set to spread rapidly. NPs are small particles with dimensions between 1.0 and 100.0 nm (Adamu et al., 2023). NPs have many uses in industries, including medical, environmental research, and material engineering, and have recently attracted lots of attention. NP delivery systems are attracting interest due to their straightforward synthesis and ability to trigger strong immune responses (Zeltins, 2012). For almost a century, alums, also known as insoluble aluminum salts, have been used extensively as a traditional adjuvant to increase the effectiveness of various inactivated or subunit vaccines with unclear mechanisms (Shah et al., 2017). Furthermore, based on the phosphophilicity of aluminum, aluminum nanoparticles are a unique type that can induce robust humoral and even cellular immune responses against Ags (Wang et al., 2015). Antigens can be conjugated with group with NP to present the immunogen to the immune system in a manner similar to that of the pathogen, eliciting a similar response. Additionally, group with NP composed of certain composites allow for both site-directed antigen delivery and prolonged antigen release to optimize immune system exposure (Gregory et al., 2013). Currently, many elements are under investigation for drug delivery and more specifically for cancer therapy and vaccine design (Al-Mousawe, 2024; Păduraru et al., 2025). Recently, some microorganisms, including P. aeruginosa, have been causing such a challenging infection to global public health, so a truly effective vaccine is urgently needed (Mao et al., 2021). To develop an effective nanovaccine candidate, this study aimed to investigate the promoting role of aluminum oxide nanoparticles in enhancing the immunogenicity of P. aeruginosa antigens by assessing their capacity to enhance antigen delivery, stimulate immune cell activation, and potentiate both humoral and cellular immune responses. Materials and MethodsExperimental animalsThis study involved 30 male BALB/C mice with ages of 10–12 weeks with a median weight of (20–25 g), were housed in the animal resource facility at the college of veterinary medicine in Tikrit University. The animals were kept under pathogen-free conditions with a maximum of four animals per cage under a 12 hours light/dark cycle at 18°C–23°C and 40%–60% humidity. The Institutional Animal Care and Use Committee’s authorized policy was followed for all experiments. Pseudomonas aeruginosa strainThe target bacteria P. aeruginosa was isolated from the target organs of the affected animals at the hospital of veterinary medicine in Tikrit city, while the source of isolation was human infected wounds (liver, spleen, kidney, and lung) in Tikrit hospital. Then, the bacteria were incubated and grown on special culture media at the laboratories of the College of Veterinary Medicine, Tikrit University for further identification by VITEK to be used in the experiment. Pseudomonas aeruginosa challenge doseNutrient agar was used to cultivate P. aeruginosa, which was subsequently incubated for 24 or at 37°C for 24 hours. Four mice in each target group received 0.2 ml of bacterial suspension grown intraperitoneally (IP). Autopsy sections of the spleen, liver, kidney, and lung (50 mm) were cultured on blood agar for 24 hours at 37°C before all animals were sacrificed 48 hours after the injection (El-Aziz et al., 2018). This practice was repeated until the injected animals passed away within 48 hours. A 0.2 ml bacterial suspension containing 1 × 106, 1 × 107, and 1 × 108 CFU/ml of virulent P. aeruginosa challenge dosage was used to inoculate the target groups. We tallied the number of deceased animals 24–48 hours after the vaccine. A volume of 0.2 ml of 1 × 107 CFU/ml was considered as the virulent P. aeruginosa challenge dose for use in the current trial (Priebe and Goldberg, 2014). Preparation of P. aeruginosa antigensPseudomonas aeruginosa was prepared using a modified protocol from Mitov et al. (1992). After being cultivated on nutrient agar for 24 hours at 37°C, the bacteria were extracted in phosphate buffered saline (PBS) (pH 7.2) and centrifuged at 3,000 rpm for 30 minutes at 4°C. After a 30 minutes chilling period on ice, the samples were resuspended in PBS and ultrasonically sonicated in 12 peaks at 2-minute intervals (Karl Klob, Germany). After centrifuging the resultant suspension for 30 minutes at 23,000 rpm, the supernatant was filtered through a 0.22 µm filter and kept at −20°C. Gram staining and culture on nutrient, eosin-methylene blue, and blood agar were used to verify sterility. The Biuret method was used to assess the antigen’s total protein concentration, which was adjusted to 0.5 mg/ml to reflect the killed whole sonicated P. aeruginosa antigens (KWSPAgs). Synthesis of Al2O3: Ag nanoparticlesAl2O3: Ag NPs were synthesized via a modified chemical reduction method (Fu et al., 2014) using aluminum nitrate, silver nitrate, and trisodium citrate as precursors. A 100 ml solution of 0.001 M Al (NO3)3·9H2O and AgNO3 was boiled, and 10 ml of 1% trisodium citrate was added dropwise with vigorous stirring until a visible color change occurred, indicating nanoparticle formation. The resulting colloid exhibited a uniform, particle-like morphology (≈20 ± 10 nm). SEM image analysis was performed using the MBF_ ImageJ software. Additionally, 45 mg of the same materials were dissolved in 100 ml deionized water for 24 hours to serve as an adjuvant. Furthermore, nanoparticles were coated with KWSPAgs following the method of Borges et al. (2007). Study designThe experimental animals with total of 30 mice were randomly mixed, and a total of 10 mice per group including Naïve (control- T1), which received 0.2 ml of orally normal saline. The second group was the KWSPAgs alone (T2) group, which was immunized with 0.5 ml of KWSPAgs only with a protein concentration of 0.5 mg/ml IP in two doses at 2-week intervals. The KWSPAgs + NPs (T3) group was immunized with 1 ml of a mix consisting of 0.5 mg/ml of KWSPAgs and 0.5 mg/ml of nanoparticle adjuvant IP in two doses at 2-week intervals. At 14 days post-immunization, cell-mediated immunity was determined by measuring the thickness of the skin test Delayed-type hypersensitivity (DTH) in the study groups to determine cellular immune responses. At 28 days after the first immunization, blood samples were taken from all groups of Naïve, KWSPAgs, and KWSPAgs+NPs to determine humoral immune responses. Immunoglobulins (IgG), Toll like Receptor2 (TLR2) and interleukin 2 (IL2) were both determined by Enzyme-Linked Immune Sorbent Assay (ELISA). All immunized groups were inoculated IP with an effective dose (0.2 1 × 107 CFU/ml) of virulent P. aeruginosa. All animals were anesthetized and then sacrificed at 14 days post-infection with P. aeruginosa. Small autopsy pieces were taken for bacterial isolation, and other organs were fixed in 10% of neutral buffer formalin for histopathological and immunohistochemical examination. Delayed-type hypersensitivity responseThe following protocol was used for this test, which was conducted 14 days after the first immunization and was adapted from (Chung et al., 2020). Five mice from each immunized group and the Naive group had their right footpad injected intradermally with 0.01 ml of KWSPAg (0.5 mg/ml protein concentration), and the same animals left footpad was injected intradermally with 0.01 ml of PBS inoculation. The thickness of the skin on both sides was measured after 24 and 48 hours using a conventional vernier. Determination of the ELISA KitSamples of serum IgG, TLR2, and IL2 were diluted by a factor of two in the diluent that came with the kit. ELISA was employed to evaluate the serum concentrations of IgG, TLR2, and IL2 using commercial kits that were bought from Abcam pla with the catalog numbers (ab 229388), (ab 224880), and (ab 221834), Abcam pla, Waltham, MA, U S, respectively. TLR2, IL2, and inflammatory IgG cytokines were measured using the Sandwich ELISA method specified in the tissue sample methodology. The BioTek EL X 800 ELISA reader was used to detect the intensity of color formation at 450 nm. Additional charting on the standard curve was used to determine the sample concentration (Mushtaq et al., 2022). Pathological studyWe collected 1 cm internal organ specimens, including the kidney, liver, lung, and spleen. The specimens were immediately washed with tap water after being fixed in a 10% buffer formaldehyde solution for 72 hours. Processing was then carried out regularly using a series of upgrading alcoholic concentrations from 70% to 100% for 2 hours in each concentration to remove water from the tissues, followed by xylol clearance, then two phases of semi-liquid paraffin wax infiltration at 58°C, blocks of the specimens were generated using paraffin wax, and each tissue was sectioned at 5 μm using a rotary microtome. All tissues were stained with hematoxylin and eosin, and the histopathological lesions were studied using light microscopy according to (Bancroft and Gamble, 2008). Indirect immunohistochemistry method for detecting P. aeruginosa Ag in tissue sectionsImmunohistochemistry was performed according to the procedure described by Oumarou Hama et al. (2022) with minor modifications. Immunohistochemistry can reveal intracellular or membrane-bound antigens by staining cells and tissues with an antigen-specific antibody. Sigma Aldrich’s DAB kit was used for observing tissue sections. The present study used an immunohistochemical approach to detect P. aeruginosa Ags in tissue using a particular polyclonal antibody (anti-Pseudomonas aeruginosa antibody) (ab68538) (Abcam pla, Waltham, MA, USA). Statistical analysisThe GraphPad Prism software (GraphPad Software version 9) was used to conduct the statistical analysis. Data in groups of five mice are presented as mean ± S.E.M. Three or more groups were compared using a one-way analysis of variance test. Data from groups were compared using Student’s t-test. The following figures show statistically significant differences: p < 0.05, p < 0.01, p < 0.001, and p < 0.0001.3. Ethical approvalThe Animal House at the University of Tikrit College of Veterinary Medicine provided ethical approval for this study on July 14, 2022, under Approval No. (Tu. Vet.112). ResultsWe used three groups of mice which include Naïve, KWSPAgs alone, and KWSPAgs + group with NP to study the role of NPs-AL2O3 in improving the immunogenicity of KWSPAgs. The results showed that immunizing the mice twice with KWSPAgs at 1 and 14 days, respectively, the level of IL2 was significantly increased in KWSPAgs compared with Naïve control at 28 days, which suggests the successful way of the KWSPAgs inducing the infection. In addition, the results of our study showed that the level of IL2 was significantly increased in KWSPAgs + NPs compared with KWSPAgs alone, suggesting the successful delivery of NPs increasing the immunogenicity through the mediation immune inflammatory role of IL2 (Fig. 1A). Clinically, we have also investigated the foot pad thickness as an indicator sign for inflammation after exposure to KWSPAgs and KWSPAgs+ NPs. The results of our study have shown a successful way for increasing the delivery and immunogenicity of KWPAgs. In addition, the results indicated a significant increase in the foot pad thickness in KWSPAGgs+Nps compared with KWSPAgs alone, suggesting a successful delivery method mediated in this study by using NPs (Fig. 1B–D).
Fig. 1. Nanoparticles enhancing KWSPAgs- immunization would be more fit than using infection. Mice were exposed to a double dose of 0.5 ml KWSPAgs (IP) at 2-week intervals. On day 1, DDW as a VEH was administered to these mice (IP) in the control group and KWSPAgs alone and KWSPAgs +NPs on day 14. The mice were euthanized on day 28 for various studies. (A) IL2 cytokine level from serum obtained on day 28. (B and C) Compression between the clinical macroscopic changes between the naive and immunized groups. Five mice in each group were used, and the data were confirmed in three independent experiments. (D) Comparison of the foot pad thickness between the groups at 24 and 48 hours *p ≤ 0.05, p** ≤ 0.01, ***p ≤ 0.001. NPs increased the delivery efficacy of the group with NP, which increased the immunogenicity of KWSPAgs through the upregulation of both IgG and TLR2In this study, we further investigated the humoral immunity biomarkers after we noticed that there was an immune response after exposure of mice with IP injections of KWSPAgs and KWSPAgs + NPs. The results showed that both IgG and TLR2 were significantly increased after KWSPAgs compared with Naïve group suggesting the successful preparation of KWSPAgs, which was used in this study exclusively for inducing the immune response. At the same time, there was a further upregulation of both IgG and Toll-like receptors (TLR) in KWSPAg +NPs compared with KWSPAgs alone, suggesting the successful role of NPs in promoting the immunogenicity of KWSPAgs (Fig. 2A and B).
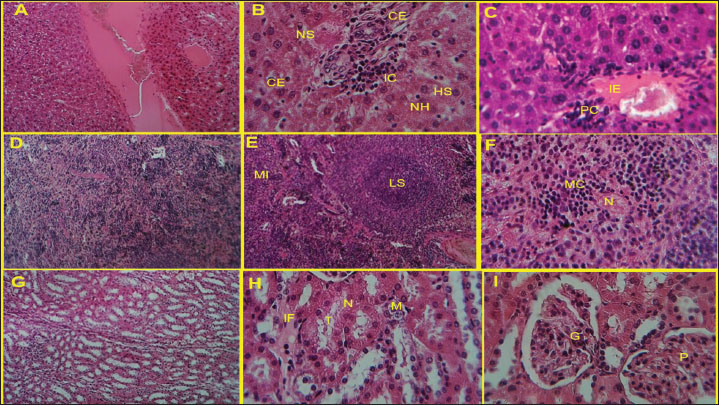
Fig. 2. (A and B) We further investigated humoral immunity after we noticed a cellular immune response after exposure of mice. Injections of KWSPAgs and KWSPAgs + NPs, both IgG and TLR2 were significantly increased after KWSPAgs compared with Naïve group suggesting the successful preparation of KWSPAgs for inducing the immune response. At the same time, there was a further upregulation of both IgG and TLR in KWSPAg +NPs compared with KWSPAgs alone, suggesting the successful role of NPs in promoting the immunogenicity of KWSPAgs compared with KWSPAgs alone. *p ≤ 0.05, p** ≤ 0.01. Nanoparticles increased the delivery efficacy of KWSPAgs, leading to significant histopathological changes in liver, spleen, and kidney tissuesThe results of our study have shown that there is a severe inflammatory reaction in the KWSPAgs group compared with normal histological sections in the naïve group, suggesting the successful and active role of KWSPAgs in inducing immunogenicity. In detail, the liver of the KWPAG and KWSPAg+ NP groups showed a severe inflammatory reaction with infiltration of inflammatory cells, necrosis of hepatocytes, narrowing of sinuses, and hepatocyte swelling. Cellular edema was also severe, and central vein distension with few mononuclear cells was observed. In the immunized group, the liver showed perivascular cuffing followed by inflammatory edema (Fig. 3A–C). The section in the spleen of the infected and non-infected groups at 14 days post-infection with P. aeruginosa showed that the spleen of naïve group with normal histological sections, whereas the diffuse lesion consisted of a large necrotic area with mononuclear cells infiltration in the white and red pulp, with reactive lymphoid follicle, white pulp two foci of CFU, and prominent necrotic area region of lymphoid tissue suppuration (Fig. 3D–F). The last organ that was investigated was the kidney, and the results of our study showed that the kidney of the control group had no lesions, whereas the kidney-related tissue of the KWSPAgs and KWSPAgs+NPs groups appeared to be edematous fluid between tubules with mononuclear cell infiltration and narrowing of distal and proximal tubules and some with coagulative necrosis. The cortex of the immunized group showed hypertrophy in glomeruli with polymorphic cell infiltration. (Fig. 3G–I)
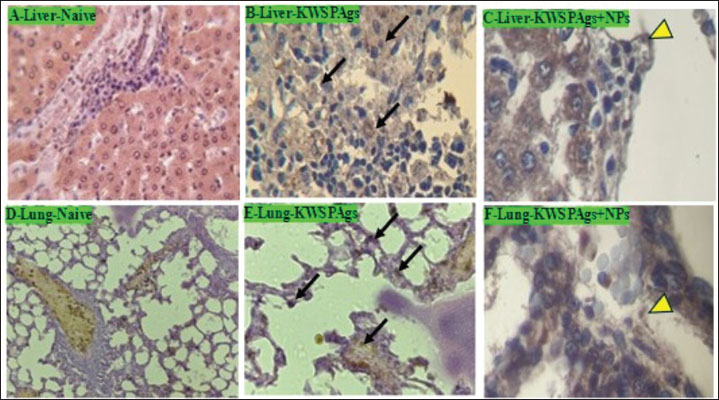
Fig. 3. Microscopic assessment of control, immunized, and non-immunized groups at 28 days post-infection with P. aeruginosa H and E stain (400, 100X). (A) The control group had no liver lesions. (B) The liver of the KWSPAgs immunized group showed severe inflammatory reaction with infiltration of inflammatory cells (IC), necrosis of hepatocytes (NH), narrowing of sinusoidal (NS), and hepatocyte swelling (HS). Cellular edema (CE) was severe and there was a slight vacuolar fatty change. (C) The liver of the immunized group had perivascular cuffing (PC) followed by inflammatory edema (IE), central vein distension, and few mononuclear cells. (D) Control group with no lesion in spleen tissue. (E) Infected group with a reactive lymphoid follicle, white pulp with a prominent necrotic area of lymphoid tissue suppuration (LS). The white and red pulp has mononuclear cells infiltrating it (MI). (F) Mononuclear cell infiltration with two foci of CFU (MC) in spleen with small area of necrotic lesion (N). (G) Kidney of control group with no lesion. (H) Kidney tissue of infected group with edematous fluid between tubules (IF) with mononuclear cells infiltration (M) and narrowing of distal and proximal tubules (T) and some of it with coagulative necrosis (N). (I) Kidney of immunized group, the cortex showed hypertrophy in glomeruli (G) with polymorphic cells infiltration (P). Nanoparticles increased the efficacy of KWSPAg delivery, leading to significant changes in immune histochemistry biomarkersThe results of our study have shown that immune histochemistry staining of the liver and lung of naive and immunized groups at 28 days post-infection with P. aeruginosa. Naive immunohistochemistry sections showed negative staining cells, whereas the immunized group showed potent positive staining represented by pink-red-brown cells (black arrow) and scattered pink-red-brown cells (yellow head arrow). Immunohistochemical staining of the naive, KWSPAgs and KWSPAgs+NPs Figure 4A–F.
Fig. 4. Immunohistochemical staining of the liver of naive, KWSPAgs, and KWSPAgs+NPs at 14 days post-infection with P. aeruginosa. Naïve immunohistochemistry showed negative staining cells (Fig. 4A), KWSPAgs showed potent positive staining represented by pink-red-brown cells (black arrow) (Fig. 4B), and KWSPAgs+NPs showed scattered pink-red-brown cells (yellow head arrow) (Fig. 4C). Immunohistochemical staining, the lung of Naïve, KWSPAgs, and KWSPAgs+NPs at 14 days post-infection with P. aeruginosa (Fig. 4D–F). DiscussionPseudomonas aeruginosa is a dominant pathogen that contributes to morbidity and mortality (Hudson and Hay, 1980). Pseudomonas aeruginosa is considered one of the major nosocomial infections and is associated with antimicrobial resistance. As the treatment against this bacteria has been costing millions of dollars annually, a deep investigation and understanding of the immune response by the host against infection is needed as a primary way for future therapeutic targets as well as for future vaccine design if possible, which both of these options have been a challenge among increasing the cases of infection annually. The use of nanoparticle delivery systems as carriers for molecules of various sizes to enhance the pharmacokinetic and pharmacodynamic characteristics of different kinds of therapeutic molecules has recently drawn attention to nanoparticle research in the fields of vaccination and medication delivery, so nanoparticles have been utilized to limit the availability of drugs to specific locations, safeguard the drug entity in the systemic circulation, and transport the medication to the site of action at a steady and regulated rate (Jurado-Martín et al., 2021; Al-Juhaishi et al., 2025). On the other hand, designing a vaccine against P. aeruginosa has been a major challenge in reducing infections (Najahi-Missaoui et al., 2020; Sendra et al., 2024) thus, there is a critical need to establish a primary method for that and establish a first step that could be considered as a start-up for a major project of vaccine design. Interleukins are active glycoproteins that are mostly produced by macrophages and activated lymphocytes (Johansen and Gøtzsche, 2015). IL-2 was one of the earliest cytokines to be identified and first promote lymphocyte proliferation. Since then, it has emerged that activated T cells are the main source of this cytokine, which also functions as an autocrine growth factor for T cells and a paracrine growth factor for NK cells. IL-2 plays a vital role in the development of immunity. IL-2 plays a role during the inflammatory response and is considered a potent inducer of T-cell proliferation, including T-helper 1 and T-helper 2 effector T-cell differentiation (Elmslie et al., 1991). These studies agree with the results of our study, where IL2 was significantly increased in KWSPAgs compared with Naïve group suggesting the successful immunogenicity of KWSPAgs. At the same time, the level of IL2 was significantly increased in KWSPAgs +NPs compared with KWSPAgs alone, suggesting the successful delivery of NPs, increasing the immunogenicity and the chances of establishing start-up first-step vaccine design. IgGs are crucial mediators of immunological protection against invading pathogens. They exert their biological effects through several effector systems (Hoyer et al., 2008). The protein IgG is among the most prevalent immunoglobulins and can contribute up to 20% of plasma protein. It is the major class of immunoglobulins (Hoyer et al., 2008; Li et al., 2025). Recent studies have shown such a link between cases of P. aeruginosa and IgG elevation in both experimental and clinical studies (Kapur et al., 2014). These results agreed with the results of our study, which showed that KWSPAs were significantly increased compared with Naïve group suggesting the successful role of KWSPAgs, while NPs improved the delivery of KWSPAgs compared with KWSPAgs alone, suggesting the successful role of NPs reaching out to more space area compared with KWSPAgs alone without NPs. TLRs are considered a crucial activator player in innate immunity by recognizing specific patterns of microbial components. TLR signaling pathways arise from intracytoplasmic TLR domains, which are conserved among all TLRs and have demonstrated their ability to identify particular microbial component patterns that are shared by all infections (Salauddin et al. (2025). The results of our study have revealed that TLRs were significantly increased in KWSPAgs compared with Naïve group while group with NP were successfully approving their successful role in activating the delivery of KWSPAgs to induce TLR2. Cellular immunity is considered an indicator after P. aeruginosa infection (Katoh et al., 2016). Cytokines are considered one of the major immune markers for cellular immunity and an indicator of immune response. Our results have shown that KWSPAGs were significantly increased in T3 (KWSPAgs + NPs) compared with T2 (KWSPAgs alone), whereas IL2 was significantly increased in KWSPAgs compared with Naïve. The results of our study have indicated that P. aeruginosa could induce the immune response compared with Naïve group. The results obtained from the current study were consistent with those of previous studies, where IL2 was successfully induced after experimental inoculation of P. aeruginosa (Takeda and Akira, 2004). Inflammation can be created by several different causes, including immune system activation, and the infiltration of immune cells is well known to be correlated with immune cells (Zuercher et al., 2005; Roe, 2021). The results of our study have indicated a significant infiltration of inflammatory cells as well as in immune histochemistry in the KWSPAgs group compared with Naïve alone, whereas this infiltration has been significantly increased in KWSPAgs+NPs compared with Naive and KWSPAgs alone. These results were consistent with those of previous studies that have shown that P. aeruginosa plays a critical role in enhancing the immune pathological changes in different organs compared with non-immunized groups (Berube et al., 2016; Al-Bayati et al., 2024). An opportunistic infection, P. aeruginosa infects people with a variety of illnesses, including cancer, immunodeficiency, burn wounds, and chronic obstructive pulmonary disease (Goncalves et al., 2016). Pseudomonas aeruginosa has the ability to adapt to the environment’s hosts by secreting a variety of virulence factors, which contribute to successful infection and disease (Qin et al., 2022). These results agree with those of Vidaillac and Chotirmall (2021), who showed that dual doses of protein-related P. aeruginosa resulted in an immune response and that the response was seen in different organs, including the liver. Another study correlated P. aeruginosa interaction with histopathological changes in the liver (Daugherty et al., 2000), showing an immune response in hepatocytes challenged with P. aeruginosa. On the other hand, the changes in the spleen, which were shown in these results, agreed with those of Cheluvappa et al. (2009), who showed that the tissue of the spleen is significantly affected after infection with P. aeruginosa. Immunohistochemistry technique has been used to investigate the role of P. aeruginosa after infection (Alhazmi, 2018). Mice immunized with protein-related P. aeruginosa showed an immune response in the hepatocytes compared with naïve mice (Becerra et al., 2016). Our study agrees with a previous study, which showed that rats immunized with P. aeruginosa-related proteins showed a significant immune response, while a vaccination against pulmonary P. aeruginosa showed a significant change in the immune histochemistry biomarkers of immunized mice compared with Naïve group (Scarff and Goldberg, 2008). ConclusionThe dual-dose KWSPAgs effectively induced immunogenicity, which was significantly enhanced by nanoparticles. KWSPAgs promoted T-cell proliferation (shown by increased IL-2 and footpad thickness), boosted antibody responses (IgG and TLR2), and increased inflammatory cell infiltration and immune biomarkers against P. aeruginosa infection, with potential to improve immune activation and decrease immune pathological changes. AcknowledgmentsA special thank you to the College of Veterinary Medicine of Tikrit University Deanship. We would also like to extend our thanks and gratitude to the Animal House and its laboratories for providing the necessary materials to finish the study. Conflict of interestThe authors have no potential conflicts of interest to disclose concerning the research, writing, or publication of this paper. FundingThe authors did not receive any funding from funding agencies, industry, or other sources for this study. Authors’ contributionThe research was designed and assisted by Hassan H. K. Al-Bayati, Muthanna Sultan carried out the study and wrote the manuscript, Bashar Sadeq Noomi and Thamer Jaddoa Shihab assisted with the analysis of the findings, and Sanaa Sauod Ahmed critically reviewed the manuscript. Data availabilityThe manuscript contains all of the relevant information, and the corresponding author can be contacted with any additional questions. ReferencesAdamu, S.S., Umaru, H.A., Albert, H.O. and Muhammad, A.L. 2023. Effect of green synthesized zinc oxide nanoparticles using Allium cepa extracts on triton X-100-induced hyperlipidemia in rats. Int. J. Nutr. Sci. 8(1), 36–46. Al-Bayati, H.H., Abdullah, S.A., Shihab, T.J., Sultan, M. and Jumaa, Q.S. 2024. Immunogenicity of culture filtered proteins and whole-cell killed formalin of Listeria monocytogenes to induced cellular immune response in vivo. Ope. Vet. J. 14(12), 3581. Alhazmi, A. 2018. Spleen tyrosine kinase as a target therapy for pseudomonas aeruginosa infection. Immun 10(4), 255–263. Bancroft, J.D. and Gamble, M. (Eds.). 2008. The theory and practice of histological techniques. Berlin: Elsevier Health Sciences. Becerra, S.C., Roy, D.C., Sanchez, C.J., Christy, R.J. and Burmeister, D.M. 2016. An optimized staining technique for the detection of Gram-positive and Gram-negative bacteria within tissue. BMC. Res. Notes 4(4), 1–10. Berube, B.J., Rangel, S.M. and Hauser, A.R. 2016. Pseudomonas aeruginosa: breaking down barriers. Curr. Genet. 62(1), 109–113. Borges, O., Tavares, J., De Sousa, A., Borchard, G., Junginger, H.E. and Cordeiro-Da-Silva, A. 2007. Evaluation of the immune response following a short oral vaccination schedule with hepatitis B antigen encapsulated into alginate-coated chitosan nanoparticles. Eur. J. Pharm. Sci. 32(4-5), 278–290. Cheluvappa, R., Denning, G.M., Lau, G.W., Grimm, M.C., Hilmer, S.N. and Le Couteur, D.G. 2009. Pseudomonas aeruginosa and sepsis hyperlipidemia. Pathology 41(7), 615–621. Chung, K.L., Convery, C., Ejikeme, I. and Ghanem, A.M. 2020. A systematic review of delayed inflammatory reactions after hyaluronic acid filler injection to estimate the incidence of delayed type hypersensitivity reaction. Aesthet. Surg. J. 40(5), P286–P300. Crousilles, A., Maunders, E., Bartlett, S., Fan, C., Ukor, E.F., Abdelhamid, Y. and Welch, M. 2015. Which microbial factors are important in Pseudomonas aeruginosa infections?. Future Microbiol. 10(11), 1825–1836. Daugherty, A.L., Mckee, M.L., Fitzgerald, D.J. and Mrsny, R.J. 2000. Epithelial application of Pseudomonas aeruginosa exotoxin A results in a selective targeting to cells in the liver, spleen and lymph node. J. Control. Release 65(1-2), 297–302. El-Aziz, G.A., El-Kholy, A.I. and El-Sokkary, R.H. 2018. Protective and curative effects of garlic extract against P. aeruginosa infection in mice. Microbial Pathogenesis 118, 177–182. Fu, Y., Zhang, Y., Chang, X., Zhang, Y., Ma, S., Sui, J., Yin, L., Pu, Y. and Liang, G. 2014. Systemic immune effects of titanium dioxide nanoparticles after repeated intratracheal instillation in rat. Int. J. Mol. Sci. 15(4), 6961–6973. Gaur, P., Polipalli, Y., Bandhu, X., Chakravarty, S., Banerjee, S., Meza, C. and Banerjee, A. 2025.Unveiling the potential of bacterial keratinase with antiacne drugs for cosmeceutical applications: an in silico evaluation. J. Pharmacol. 111, 1–24. Gomila, A., Carratalà, J., Badia, J.M., Camprubí, D., Piriz, M., Shaw, E., Diaz-Brito, V., Espejo, E., Nicolás, C., Brugués, M., Perez, R., Lérida, A., Castro, A., Biondo, S., Fraccalvieri, D., Limón, E., Gudiol, F. and Pujol, M. 2018. Preoperative oral antibiotic prophylaxis reduces Pseudomonas aeruginosa surgical site infections after elective colorectal surgery: a multicenter prospective cohort study. BMC Infect. Dis. 18, 1–9. Gonçalves-De-Albuquerque, C.F., Silva, A.R., Burth, P., Rocco, P.R.M., Castro-Faria, M.V. and Castro-Faria-Neto, H.C. 2016. Possible mechanisms of Pseudomonas aeruginosa-associated lung disease. Int. J. Med. Microbiol. 306(1), 20–28. Gregory, A.E., Titball, R. and Williamson, D. 2013. Vaccine delivery using NPs. Front. Cell. Infect. Microbiol. 3, 13. Hoyer, K.K., Dooms, H., Barron, L. and Abbas, A.K. 2008. Interleukin-2 in the development and control of inflammatory disease. Immunol. Rev. 226(1), 19–28. Hudson, L. and Hay, F.C. 1980. Practical immunology. 3rd ed. Oxford. London: Blackwell Scientific Publication, pp: 98–105. Johansen, H.K. and Gøtzsche, P.C. 2015. Vaccines for preventing Pseudomonas aeruginosa infection in cystic fibrosis. Cochrane Database Syst. Rev. (8), 1399. Jurado-Martín, I., Sainz-Mejías, M. and Mcclean, S. 2021. Pseudomonas aeruginosa: an audacious pathogen with an adaptable arsenal of virulence factors. Int. J. Mol. Sci. 22(6), 3128. Kapur, R., Einarsdottir, H.K. and Vidarsson, G. 2014. IgG-effector functions: “good, bad, and ugly”. Immunol. Lett. 160(2), 139–144. Katoh, H., Yasumoto, H., Shimizu, M., Hamaoka, S., Kinoshita, M., Akiyama, K. and Sawa, T. 2016. IV. Immunoglobulin for acute lung injury and bacteremia in Pseudomonas aeruginosa pneumonia. J. Crit. 2015, 67–79. Laking, H., Johnston, W., Butcher, J.P., Kean, R. and Ramage, G. 2025. Biofilms of medical significance in and around humans, 1st ed. In: Microbial biofilm dynamics: contemporary approaches, models and analytical tools. Eds., Kumar Shukla, A and Roberto Monteiro, D. CRC Press. Li, L., Cai, S., Lin, S., Bai, L., Yu, Z., Sun, W. and Dong, L. 2025. Potential roles of antibodies with different classes in IgG4-related diseases. Rheumatol. Autoimmun. 5, 174–185. Mao, L., Chen, Z., Wang, Y. and Chen, C. 2021. Design and application of nanoparticles as vaccine adjuvants against human coronavirus infection. J. Inorg. Biochem. 219, 111454. Mitov, I., Denchev, V. and Linde, K. 1992. Humoral and cell-mediated immunity in mice after immunization with live oral Salmonella typhimurium vaccines: auxotrophic mutants with two attenuating markers. Vaccine 10, 61–66. Mushtaq, M., Sadique, U., Said, F., Shah, M., Amanullah, H. and Anwer, F. 2022. Immunomodulatory and hepato-protective role of water based supplemented Bacillus clausii in broiler chicks. J. Biol. Chem. J. Saudi Soc. Agric. Sci. 21(2), 108–113. Najahi-Missaoui, W., Arnold, R.D. and Cummings, B.S. 2020. Safe nanoparticles: are we there yet?. Int. J. Mol. Sci. 22(1), 385. Oumarou Hama, H., Aboudharam, G., Barbieri, R., Lepidi, H. and Drancourt, M. 2022. Diagn. Immunohistochemical diagnosis of human infectious diseases: a review. Pathol 17(1), 17. Păduraru, L., Panainte, A.D., Peptu, C.A., Apostu, M., Vieriu, M., Bibire, T., Sava, A. and Bibire, N. 2025. Smart drug delivery systems based on cyclodextrins and chitosan for cancer therapy. Pharmaceuticals 18(4), 564. Priebe, G.P. and Goldberg, J.B. 2014. Vaccines for Pseudomonas aeruginosa: a long and winding road. Exp. Rev. Vacc. 13(4), 507–519. Qin, S., Xiao, W., Zhou, C., Pu, Q., Deng, X., Lan, L. and Wu, M. 2022. Pseudomonas aeruginosa: pathogenesis, virulence factors, antibiotic resistance, interaction with the host, technological advances, and emerging therapeutics. Signal Transducer Target. Ther. 7(1), 199. Reynolds, D. and Kollef, M. 2021. Epidemiology, pathogenesis, and treatment of Pseudomonas aeruginosa infections: an update. Drugs 81, 2117–2131. Roe, K. 2021. An inflammation classification system using cytokine parameters. Scand. J. Immunol. 93(2), 12970. Salauddin, M., Bhattacharyya, D., Samanta, I., Saha, S., Xue, M., Hossain, M.G. and Zheng, C. 2025. Role of TLRs as signaling cascades to combat infectious diseases: a review. Cell. Mol. Life Sci. 82(1), 122. Scarff, J.M. and Goldberg, J.B. 2008. Vaccination against Pseudomonas aeruginosa pneumonia in immunocompromised mice. Clin. Vaccine Immunol. 15(2), 367–375. Sendra, E., Fernández-Muñoz, A., Zamorano, L., Oliver, A., Horcajada, J.P., Juan, C. and Gómez-Zorrilla, S. 2024. Impact of multidrug resistance on Pseudomonas aeruginosa virulence and fitness: a microbiological and clinical perspective. Infection 52(4), 1235–1268. Shah, R.R., Hassett, K.J. and Brito, L.A. 2016. Overview of vaccine adjuvants: introduction, history, and current status. In: Vaccine adjuvants: methods and protocols. Methods in molecular biology, Ed., Singh, S.M. New York: Humana Press, vol. 1494, pp: 1–13. Takeda, K. and Akira, S. 2004. TLR signaling pathways. In: Seminars in immunology. London, UK: Academic Press, Vol. 16, pp: 3–9. Thi, M.T.T., Wibowo, D. and Rehm, B.H. 2020. Pseudomonas aeruginosa biofilms. J. Mol. Sci. 21(22), 8671. Vidaillac, C. and Chotirmall, S.H. 2021. Pseudomonas aeruginosa in bronchiectasis: infection, inflammation, and therapies. Expert Rev. Respir. Med. 15(5), 649–662. Wang, T., Zhen, Y., Ma, X., Wei, B. and Wang, N. 2015. Phospholipid bilayer-coated aluminum nanoparticles as an effective vaccine adjuvant delivery system. ACS. Appl. Mater. Inter. 7(12), 6391–6396. Zeltins, A. 2012. Construction and characterization of virus-like particles: a review. Mol. Biotechnol. 53, 92–107. Zuercher, A.W., Imboden, M.A., Jampen, S., Bosse, D., Ulrich, M., Chtioui, H. and Lang, A.B. 2005. Cellular immunity in healthy volunteers treated with an octavalent conjugate P. aeruginosa vaccine. Clin. Exp. Immunol. 142(2), 381–387. | ||
| How to Cite this Article |
| Pubmed Style Al-bayati HHK, Sultan MA, Ahmed SS, Noomi BS, Shihab TJ. The promoting role of aluminum oxide nanoparticles in enhancing Pseudomonas aeruginosa antigen immunogenicity. Open Vet. J.. 2025; 15(10): 5136-5145. doi:10.5455/OVJ.2025.v15.i10.31 Web Style Al-bayati HHK, Sultan MA, Ahmed SS, Noomi BS, Shihab TJ. The promoting role of aluminum oxide nanoparticles in enhancing Pseudomonas aeruginosa antigen immunogenicity. https://www.openveterinaryjournal.com/?mno=270691 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.31 AMA (American Medical Association) Style Al-bayati HHK, Sultan MA, Ahmed SS, Noomi BS, Shihab TJ. The promoting role of aluminum oxide nanoparticles in enhancing Pseudomonas aeruginosa antigen immunogenicity. Open Vet. J.. 2025; 15(10): 5136-5145. doi:10.5455/OVJ.2025.v15.i10.31 Vancouver/ICMJE Style Al-bayati HHK, Sultan MA, Ahmed SS, Noomi BS, Shihab TJ. The promoting role of aluminum oxide nanoparticles in enhancing Pseudomonas aeruginosa antigen immunogenicity. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5136-5145. doi:10.5455/OVJ.2025.v15.i10.31 Harvard Style Al-bayati, H. H. K., Sultan, . M. A., Ahmed, . S. S., Noomi, . B. S. & Shihab, . T. J. (2025) The promoting role of aluminum oxide nanoparticles in enhancing Pseudomonas aeruginosa antigen immunogenicity. Open Vet. J., 15 (10), 5136-5145. doi:10.5455/OVJ.2025.v15.i10.31 Turabian Style Al-bayati, Hassan H. K., Muthanna A. Sultan, Sanaa S. Ahmed, Bashar S. Noomi, and Thamer J. Shihab. 2025. The promoting role of aluminum oxide nanoparticles in enhancing Pseudomonas aeruginosa antigen immunogenicity. Open Veterinary Journal, 15 (10), 5136-5145. doi:10.5455/OVJ.2025.v15.i10.31 Chicago Style Al-bayati, Hassan H. K., Muthanna A. Sultan, Sanaa S. Ahmed, Bashar S. Noomi, and Thamer J. Shihab. "The promoting role of aluminum oxide nanoparticles in enhancing Pseudomonas aeruginosa antigen immunogenicity." Open Veterinary Journal 15 (2025), 5136-5145. doi:10.5455/OVJ.2025.v15.i10.31 MLA (The Modern Language Association) Style Al-bayati, Hassan H. K., Muthanna A. Sultan, Sanaa S. Ahmed, Bashar S. Noomi, and Thamer J. Shihab. "The promoting role of aluminum oxide nanoparticles in enhancing Pseudomonas aeruginosa antigen immunogenicity." Open Veterinary Journal 15.10 (2025), 5136-5145. Print. doi:10.5455/OVJ.2025.v15.i10.31 APA (American Psychological Association) Style Al-bayati, H. H. K., Sultan, . M. A., Ahmed, . S. S., Noomi, . B. S. & Shihab, . T. J. (2025) The promoting role of aluminum oxide nanoparticles in enhancing Pseudomonas aeruginosa antigen immunogenicity. Open Veterinary Journal, 15 (10), 5136-5145. doi:10.5455/OVJ.2025.v15.i10.31 |