| Research Article | ||
Open Vet. J.. 2026; 16(1): 669-678 Open Veterinary Journal, (2026), Vol. 16(1): 669-678 Research Article Histological distribution of endocrine cells in the pancreas of Javan Mongoose (Urva javanica)Sekar Arum Krisna Putri1, Teguh Budipitojo2*, Yuda Heru Fibrianto3 , Woro Danur Wendo2 and Vista Budiariati21Sains Veterinary Graduate School, Universitas Gadjah Mada, Mada, Indonesia 2Department of Anatomy, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Mada, Indonesia 3Department of Physiology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Mada, Indonesia *Corresponding Author: Teguh Budipitojo, Department of Anatomy, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Mada, Indonesia. Email: udipitojo [at] ugm.ac.id Submitted: 15/07/2025 Revised: 11/11/2025 Accepted: 01/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: The Javan mongoose (Urva javanica) is an endemic wild animal in Southeast Asia, including Indonesia, and is currently classified as an unprotected species. However, intensive hunting may lead to a population decline in its natural habitat, potentially altering its conservation status and posing a threat soon. Despite this concern, the anatomical data of the species remain limited. The digestive system of the Javan mongoose, an opportunistic carnivore, has not been previously documented. While a comprehensive examination of the entire digestive tract would offer broader insights, this study prioritizes the pancreas due to its central endocrine role in nutrient metabolism and adaptive feeding. This study provides the first baseline data on endocrine cell localization in Javan mongooses, laying the groundwork for future research on their digestive physiology. Aim: This study aimed to investigate the pancreatic structure of the Javan mongoose, with a specific focus on the distribution of pancreatic endocrine cells, including β, α, δ, and PP cells. Methods: Six Javan mongooses were obtained from a local hunter in Central Java, Indonesia. The anatomical and histological characteristics of the pancreas were examined, along with the expression of immunoreactive (IR) endocrine cells. Routine histological staining and immunohistochemistry (IHC) were performed. The data were analysed descriptively, semi-quantitatively, and quantitatively. Results: The pancreas of the Javan mongoose is located adjacent to the dorsal duodenum and the liver’s caudal segment. The exocrine component comprises acini and a ductal system, with ducts lined by low-to-medium cuboidal epithelium. Within the islets of Langerhans, β cells are predominantly concentrated in the central zone, while α cells are primarily located in the periphery. In contrast, δ and PP cells were uniformly distributed across the central, intermediate, and peripheral zones. Endocrine cells were also observed in the pancreatic ducts, highlighting a distinctive distribution pattern in the pancreas of Javan mongoose. Conclusion: The results reveal structural similarities to other mammalian species and highlight distinctive patterns in endocrine cell distribution, most notably, the presence of endocrine cells within the pancreatic ducts. Keywords: Anatomy, Immunohistochemistry, Pancreas, Javan mongoose (Urva javanica), Endocrine cell types. IntroductionThe Javan mongoose (Urva javanica), previously classified as Herpestes javanicus, is endemic to Southeast Asia, including Indonesia (Chutipong et al., 2016). According to the International Union for Conservation of Nature (IUCN), the species is currently listed as “Least Concern”. However, it is also included in Appendix III of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES), indicating that certain countries have requested international cooperation to regulate its trade. Although the Javan mongoose is subject to domestic legal protection, it remains involved in documented or suspected international trade, highlighting the need for global collaboration to strengthen monitoring and enforcement efforts (Cites, 2015; Susdiyanti et al., 2023; Chekunov et al., 2024). Given these concerns, further exploration and characterization of the species are both necessary and urgent to prevent its potential extinction. The Javan mongoose, known for its opportunistic carnivorous habits, feeds on a diverse array of prey, such as small mammals, birds, reptiles, and invertebrates (Mahmood and Adil, 2017; Susdiyanti et al., 2023). This varied diet necessitates a well-adapted pancreas to its metabolic demands. The exocrine portion of the pancreas produces digestive enzymes that breakdown animal tissues, whereas the endocrine portion regulates energy metabolism. This dual functionality is essential for understanding the structure and role of the pancreas (Mescher, 2018). However, no scientific studies have investigated the pancreatic endocrine cells of the Javan mongoose, including β cells (insulin), α-cells (glucagon), δ-cells (somatostatin), and PP cells (pancreatic polypeptide) in the pancreas. Various studies on the anatomical structure and IHC of the pancreas have been conducted in other animal species, including mice, dogs, monkeys, minipigs, and the Sunda porcupine (Budipitojo et al., 2016; Tsuchitani et al., 2016). Research on the pancreas of the Javan mongoose has thus far been limited to anatomical and histological descriptions, with no immunohistochemical data available regarding the distribution of endocrine cells (Shil et al., 2012; Hussein et al., 2018; Mahmood and Kadhim, 2018; Mahmood et al., 2018). The pancreas is a soft, elongated organ located in the abdominal cavity’s dorsal region. It performs two primary functions: exocrine, which produces digestive enzymes composed of acinar cells and a duct system, and endocrine, which regulates metabolism through hormone secretion (Köning and Liebich, 2020). The endocrine cells of the pancreas are organized into clusters called the islets of Langerhans, which consist of β cells that produce insulin, α cells that produce glucagon, δ cells that produce somatostatin, and pancreatic polypeptide (PP) cells (that produce pancreatic polypeptide) (Mirontoneng et al., 2019). This study presents a novel contribution by examining not only the morphology and anatomy of the pancreas but also the distribution patterns of pancreatic endocrine cells in Javan mongoose using IHC methods. The findings of this study are expected to enrich scientific documentation and support conservation efforts of the Javan mongoose in Indonesia. Materials and MethodsSample collectionThis study was conducted at the Gross Anatomy and Histology Laboratory of the Faculty of Veterinary Medicine, Universitas Gadjah Mada, Indonesia. With ethical approval, six Javan mongooses were obtained following poaching incidents in Central Java, Indonesia. The pancreas of each animal was collected and examined macroscopically, with detailed documentation captured using a Nikon D3500 digital camera (Nikon Corporation, Tokyo, Japan). Each pancreas sample was fixed in 10% neutral-buffered formalin for 24 hours, followed by dehydration through a series of graded ethanol concentrations and clearing with xylene. Tissues were then infiltrated and embedded in paraffin to ensure optimal preservation (Budipitojo et al., 2016; Suvarna et al., 2019; Aqiilah et al., 2024). Examination of histological featuresThe paraffin blocks were then sectioned at 4 µm thickness, deparaffinized, and stained using routine histological methods (hematoxylin-eosin) as described previously (Suvarna et al., 2019). Immunohistochemistry stainingImmunohistochemistry was performed using validated primary antibodies, with each staining batch incorporating both positive and negative controls to ensure immunolabeling specificity. This technique was employed to identify the distribution pattern of pancreatic endocrine cells, including β cells (insulin), α cells (glucagon), δ cells (somatostatin), and PP cells (pancreas polypeptide) (Budipitojo et al., 2016). The sections were exposed to a hydrogen peroxide (H2O2) solution in methanol for 30 minutes to suppress endogenous peroxidase activity. As detailed in Table 1, primary antibodies were applied overnight at 4oC. The sections were pre-incubated with normal goat serum for 1 hour prior to the application of primary antibodies to minimize nonspecific staining. Subsequently, the secondary antibodies were applied for 1 hour at room temperature, and the immunoreactive sites were visualized using a diaminobenzidine (DAB) solution. The sections were counterstained with Mayer’s hematoxylin to enhance contrast. Following counterstaining, the samples were rinsed with running water, dehydrated through graded alcohols, cleared with xylene, and mounted using Entellan mounting medium to preserve sample integrity (Budipitojo et al., 2016). Table 1. The primary antibodies used in this study.
Data analysisHistological and IHC staining were meticulously examined using a light microscope (BX60, Olympus, Tokyo, Japan) and a stereo binocular microscope (SZ51, Olympus, Tokyo, Japan). Findings were documented using an OptiLab Advance Plus binocular digital camera at various magnifications. The distribution of pancreatic endocrine cells was analysed descriptively, semi-quantitatively, and quantitatively. Semi-quantitative scoring of endocrine IR cells was performed subjectively and categorized into three levels: few (+), moderate (++), and numerous (+++) in 400x magnification. Positive endocrine cells were identified in three pancreatic compartments: (1) islets of Langerhans, (2) exocrine acini, and (3) pancreatic ducts. Cell distribution within the islets of Langerhans was further classified into three zones: central, intermediate, and peripheral (Budipitojo et al., 2016). For a more detailed evaluation, the number of endocrine cells in each compartment was counted using ImageJ software (version 1.53 t; Wayne Rasband and contributors, National Institutes of Health, Bethesda, Maryland, USA, 2022), and their distribution was scored according to the criteria: 0=negative (−); 1–3 cells=few (+); 4–10 cells=moderate (++); 11–20 cells=numerous (+++); and >20 cells=very numerous (++++). The mean score for each zone was calculated following the methods of Fedchenko and Reifenrath (2014) and Budipitojo et al. (2016), after which a second-level scoring was performed to categorize the overall distribution as: 1–3 cells=few (+), 4–10 cells=moderate (++), and >10 cells=numerous (+++). The presence and abundance of endocrine cells in the pancreatic ducts were evaluated semi-quantitatively based on their localization. Additionally, the diameters of endocrine β, α, δ, and PP cells were measured using ImageJ software. Ethical approvalThe Preclinical Research Ethics Committee of Universitas Gadjah Mada, Indonesia, approved this study (Approval No. 18/EC-FKH/int./2025) . Date: 21 February 2025. ResultsAnatomical featuresThe pancreas of the Javan mongoose (Fig. 1A, B) is located in the abdominal cavity, dorsal to the duodenum (DU), and caudal to the liver (L). It appears as a solid, soft, ivory-yellow mass with a slight reddish hue, consisting of a corpus and two lobes (dexter and sinister) (Fig. 2A, B). The proximal dexter lobe (LD) is closely associated with the descending DU, and its ventral end curves cranially to form a hook-like structure. The corpus (C) extends from the LD toward the gastric (G) and splenic (S) regions, while the sinister lobe (LS) lies caudodorsally to the gastric (G), tapering and extending to the spleen hilum.
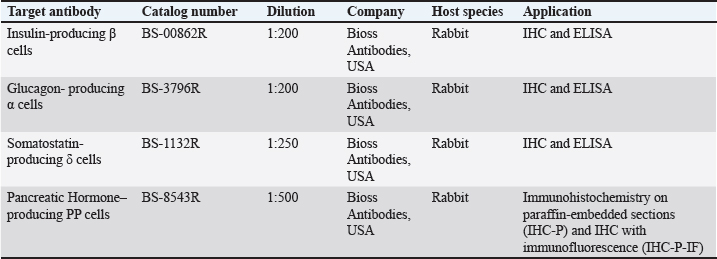
Fig. 1. Pancreatic topography in the Javan mongoose (A) Macroscopic topography of the pancreas. (B) Schematic of the pancreas in the same anatomical orientation. The dexter lobe (LD) is attached to the descending duodenum (DU), forming a hook-shaped ventral curvature. The pancreatic body (corpus, C) extends from the dexter lobe (LD) (red arrow) toward the stomach (G) and spleen (S). The sinister lobe (LS) lies caudodorsal to the stomach (G) and terminates at the splenic hilus. Abbreviations: DU: duodenum; G: stomach; GB: gallbladder; L: liver; LD: dexter lobe; LS: sinister lobe; S: spleen.
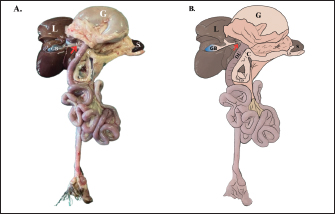
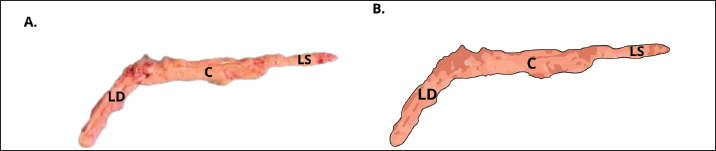
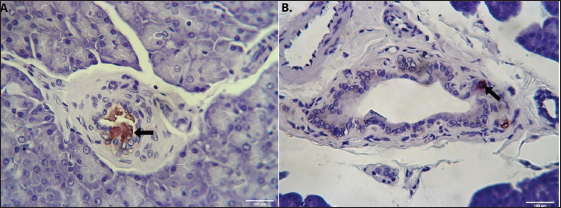
Fig. 2. Gross morphology of the pancreas of the Javan mongoose The pancreas consists of corpus (C) and two lobes: the dexter lobe (LD) and the sinister lobe (LS). The organ appears as a dense mass with a beige color and a slight reddish hue. Histological featuresHistological examination revealed that the pancreas of the Javan mongoose was encapsulated by a loose connective tissue layer, which partitioned it into distinct lobules (Fig. 3A). Each lobule contains numerous acini characterized by a single layer of pyramidal cells surrounding a narrow luminal space. The acinar cells exhibited basophilic cytoplasm, apical zymogen granules with well-defined boundaries, round to oval nuclei, and dense chromatin. Smaller centroacinar cells, which lack cytoplasmic granules, are located within the lumen and connect to a ductal system comprising intercalated, intralobular, interlobular, accessory, and main pancreatic ducts (Fig. 3B–E). These ducts are lined with cuboidal to low-cuboidal epithelium and facilitate the progressive drainage of secretions from the acini to the PD. The endocrine component comprises the islets of Langerhans (Fig. 3F), which appear as solid, pale pink, round to oval masses interspersed among the acinar clusters. These islets vary in size, contain endocrine cells associated with blood vessels and capillaries, and are more prevalent in the pancreatic sinister lobe than in the dexter lobe.
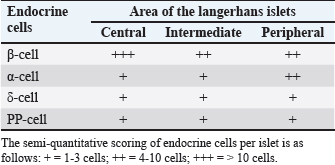
Fig. 3. Histological features of the pancreas of the Javan mongoose (A) The pancreatic capsule (black arrow) invaginates into the parenchyma, forming septa that subdivide the gland into lobules. (B) The exocrine portion shows acini (A), centroacinar cells (white arrow), intercalated duct (ID), intralobular duct (IND), connective tissue (CT), and blood vessels (BV). ID is lined by low cuboidal epithelium, whereas IND has simple cuboidal epithelium. (C) The interlobular duct (ITD) with simple cuboidal lining, along with the IND and surrounding CT (D) Accessory duct (AD) in the dexter lobe, lined by simple cuboidal epithelium and surrounded by CT, lobules (L), and BV. (E) Main pancreatic duct (PD) with associated CT, L, arterioles (AR), and venules (V). (F) Langerhans islets (LI), capillaries (CAP), and BV. Hematoxylin and eosin staining; magnification 100x and 400x; scale bar: 100 µm; 2000 µm. Endocrine cells (β, α, δ, and PP cells) characterization and localizationImmunohistochemistry analysis revealed that the pancreatic islets of Javan mongoose Langerhans contained four different types of endocrine cells: β, α, δ, and PP cells. The four endocrine cell types were heterogeneously distributed in the islets of Langerhans, with varying semi-quantitative values (Tables 2 and 3). The morphology of endocrine cells in the pancreatic tissue of the Javan mongoose was generally round to oval in shape. Table 2. Semi-quantitative number of endocrine cells in the pancreatic islets of Javan mongoose Langerhans.
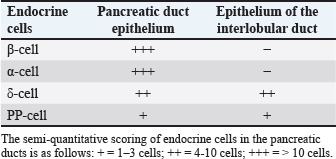
Table 3. Semi-quantitative number of endocrine cells in the pancreatic ducts of Javan mongoose.
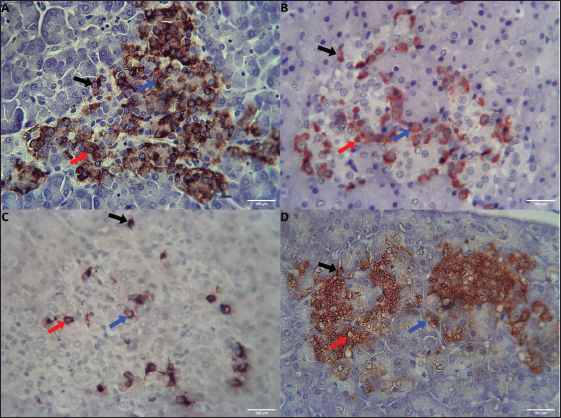
The Javan mongoose exhibits round to oval morphology of insulin-producing β cells within the islets of Langerhans. These cells, characterized by brown cytoplasmic staining in IHC, are typically round to oval in shape, with an average diameter of 6.14 µm ± 1.4 µm. They are predominantly located in the central zone, with moderate presence in both the intermediate and peripheral zones (Fig. 4A). In addition, occasional β cells were identified in the epithelium of the main pancreatic duct, where they displayed similar round and oval morphologies (Fig. 5A).
Fig. 4. Distribution of β, α, δ, and PP cells in the islets of Javan mongoose Langerhans. (A) β cells that produce insulin, (B) α cells that produce glucagon, (C) δ cells that produce somatostatin, and (D) PP cells that produce pancreatic polypeptide in the central, intermediate, and peripheral zones of the islets. The black, red, and blue arrows indicate the peripheral, intermediate, and central localizations, respectively. Immunohistochemical staining provided the images captured at 400x magnification; scale bar=100 µm.
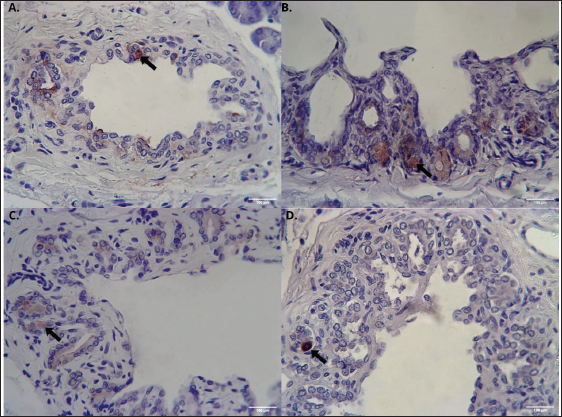
Fig. 5. Distribution of β, α, δ, and PP cells in the pancreatic duct epithelium of Javan mongoose (A) β cells that produce insulin, (B) α cells that produce glucagon, (C) δ cells that produce somatostatin, and (D) PP cells that produce pancreatic polypeptide in the pancreatic duct epithelium, exhibiting a round to oval morphology (black arrow). Immunohistochemical staining provided the images captured at 400x magnification; scale bar=100 µm. Glucagon-secreting α cells also exhibited distinct brown IHC staining and a typically round to oval morphology, with an average diameter of 7.42 µm ± 1.3 µm. Their distribution contrasts with that of β cells, α cells are moderately present at the periphery and sparsely located in the intermediate and central zones (Fig. 4B). In addition to their presence in the islets, α cells were identified within the pancreatic duct epithelium (Fig. 5B). Somatostatin-producing δ cells, which also exhibited positive brown staining in immunohistochemistry, were characterized by round to oval morphology, with an average diameter of 8.06 µm ± 2.4 µm. These cells were sparsely distributed in low numbers within the islets across the central, intermediate, and peripheral zones (Fig. 4C). In addition, δ cells were identified in the epithelium of the pancreatic (Fig. 5C) and interlobular (Fig. 6A) ducts.
Fig. 6. Distribution of δ and PP cells in the interlobular duct epithelium of the Javan mongoose (A) δ cells that produce somatostatin, (B) PP cells that produce pancreatic polypeptide, predominantly exhibiting a round morphology in the epithelium of the interlobular duct (black arrow). Immunohistochemical staining provided the images captured at 400x magnification; scale bar=100 µm. PP cells exhibited distinct brown cytoplasmic staining and were generally round to oval in morphology, with an average diameter of 6.0 µm ± 1.1 µm. These cells were sparsely distributed across the central, intermediate, and peripheral zones (Fig. 4D). PP cells were also observed in the epithelium of the pancreatic ducts (Fig. 5D) and interlobular duct (Fig. 6B) beyond the islets. DiscussionAnatomical and histological characterizationThe pancreas of the Javan mongoose is located in the abdominal region, dorsal to the DU and caudal to the liver. It consists of a corpus and two lobes, dexter and sinister. This anatomical division is similar to that observed in domestic carnivorous mammals such as dogs (Canis lupus familiaris) and cats (Felis catus domesticus) (König and Liebich, 2020; Maher et al., 2020). In dogs and cats, the pancreas forms an inverted “V” configuration with the dexter lobe positioned along the DU and the sinister lobe extending toward the spleen. However, notable interspecies differences exist. For example, the rat pancreas is divided into duodenal, gastric, and splenic lobes, whereas the Sunda porcupine exhibits a more complex structure with four distinct pancreatic lobes (Budipitojo et al., 2016; Tsuchitani et al., 2016). Compared with these species, the lobe division in the Javan mongoose reflects a simpler morphology, yet retains the basic pancreatic lobulation pattern typical of carnivorous mammals (Budipitojo et al., 2016; Tsuchitani et al., 2016). The exocrine and endocrine structures of the pancreas in the Javan mongoose show notable similarities to those of the canine pancreas, characterized by the presence of acini, centroacinar cells, intercalated ducts, intralobular ducts, interlobular ducts, main pancreatic ducts, and accessory ducts (Shunmugam and Veterinary, 2024). However, differences in the exocrine structure were observed between cats and the Javan mongoose. The main pancreatic duct is typically dominant in cats, whereas the accessory pancreatic duct is generally absent (Maher et al., 2020). Additional differences are evident in the epithelial lining of the interlobular and pancreatic ducts between dogs and the Javan mongoose. The interlobular ducts in dogs are lined by tall cuboidal epithelium, whereas the pancreatic ducts are lined by columnar epithelium that ranges from simple to pseudostratified (Shunmugam and Veterinary, 2024). Mahmood and Kadhim (2018) reported that the interlobular and main pancreatic ducts in the Indian grey mongoose (Herpestes edwardsii) are lined by simple cuboidal epithelium, a finding consistent with observations in Javan mongoose specimens from Indonesia. Distribution of endocrine cells in the pancreas of Javan mongooseThe pancreas of the Javan mongoose contains four principal types of endocrine cells: β, α, δ, and PP cells. These cells are unevenly distributed within the islets of Langerhans. β-cells predominantly occupy the central zone, α cells are primarily distributed in the peripheral zone of the islets. δ and PP cells are present in smaller numbers and are dispersed across the central, intermediate, and peripheral zones, respectively. Although species-specific variations may occur, this distribution pattern aligns with general mammalian characteristics. Tsuchitani et al. (2016) stated that many physiological processes depend on the differentiation of endocrine cells, and that these cell distribution patterns vary between species. In mice, for example, β cells dominate the central zone of the islets of Langerhans, while α and δ cells are more commonly located at the periphery. In dogs, notable variations were observed: β cells were distributed throughout the islets, α cells were localized both centrally and peripherally, and δ cells exhibited distinct distribution patterns between the sinister and dexter lobes. Interestingly, PP cells were absent in the islets of dogs (Tsuchitani et al., 2016). Similarly, in non-human primates, β cells were predominantly localized in the periphery, forming a ring-like structure; α and δ cells were irregularly distributed, and PP cells were present in greater abundance compared to other laboratory animals (Wieczorek et al., 1998; Tsuchitani et al., 2016). The spatial distribution of endocrine cells within the islets of Langerhans is not arbitrary; rather, it reflects the evolutionary adaptations to each species’ metabolic and digestive demands. Variations in islet architecture and cell ratios exemplify strategies for regulating glucose and nutrient metabolism in accordance with feeding patterns and dietary composition among carnivores, herbivores, and omnivores (Arntfield and van der Kooy, 2011; Teh et al., 2023; Contini et al., 2024). For instance, omnivores with a high demand for rapid glucose uptake typically exhibit a central core of β cells, which optimizes insulin secretion. In contrast, species adapted to intermittent feeding or fasting often display a higher proportion of α cells to enhance glucagon-mediated glucose production. From an evolutionary standpoint, islets are believed to have envolved from simpler endocrine clusters that sensed blood glucose and gradually acquired more complex cellular specialization and spatial organization to enhance hormonal secretion and paracrine interaction (Arntfield and van der Kooy, 2011). Consequently, variations in islet size, cell ratios, and localization represent distinct evolutionary strategies that ensure glucose homeostasis, aligned with ecological niches and dietary adaptation (Arntfield and van der Kooy, 2011). In most species, β cells occupy the central core of the islets, granting them direct access to the blood supply of the islets and enabling efficient insulin secretion in response to glucose. This central arrangement also enhances intercellular communication among β cells, which is crucial for synchronized insulin release (Wieland et al., 2021; Asuaje Pfeifer et al., 2023). In contrast, α cells- responsible for glucagon secretion, are generally located in the peripheral mantle surrounding the β cell core. This peripheral distribution enables α cells to respond to paracrine signals from β cells and circulating blood, ensuring glucagon’s balanced regulation as an insulin antagonist. Such spatial separation facilitates precise paracrine signaling, which is essential for maintaining glucose homeostasis (Wieland et al., 2021; Asuaje Pfeifer et al., 2023). δ cells, which produce somatostatin, and PP cells are less numerous but are also mainly positioned at the islet periphery, similar to α cells. This peripheral localization supports their paracrine regulatory roles: somatostatin inhibits both insulin and glucagon secretion to fine-tune the overall islet output, while PP cells contribute to maintaining islet architecture and cellular organization (Hauge-Evans et al., 2009; Shuai et al., 2016; Wieland et al., 2021; Tegehall et al., 2025). In addition to their organization within the islets, endocrine cells are also found in the exocrine regions of the pancreas. Interestingly, this study identified endocrine cells, specifically β, α, δ, and PP cells, within the epithelium of the pancreatic ducts, as well as δ and PP cells in the interlobular ducts. These ectopic locations are thought to reflect the developmental and structural processes that allow endocrine cells to persist outside the islets (Pauerstein et al., 2017). Endocrine progenitor cells undergo delamination and migration before aggregating into the islets of Langerhans during pancreatic organogenesis, a process regulated by signalling pathways such as semaphorin–neuropilin, which govern radial dispersion and islet morphogenesis (Pauerstein et al., 2017). As both endocrine and exocrine compartments originate from common pancreatic progenitors, a subset of endocrine cells may remain outside the islets, appearing as solitary cells or small clusters within the acinar and ductal regions. Anatomically, these extra-insular endocrine cells are often localized near ducts or connective tissue, facilitating paracrine interaction with exocrine cells (Hu et al., 2025). This proximity enables functional crosstalk: endocrine cells modulate exocrine secretions, while exocrine-derived factors influence local endocrine activity (Hu et al., 2025; Overton and Mastracci, 2022). Furthermore, pancreatic cellular plasticity allows for the possibility of trans-differentiation between exocrine and endocrine phenotypes under specific physiological or pathological conditions, thereby contributing to the persistence of endocrine cells within exocrine compartments and highlighting their potential role in regeneration and repair (Hu et al., 2025). Previous research highlights the importance of the spatial arrangement of endocrine cells within the islets, evolutionary and functional adaptations to dietary and metabolic needs. Our findings on Javan mongoose partially support these studies, showing a central concentration of β cells and a peripheral placement of α cells. However, unlike the typical peripheral distribution observed in other species, δ and PP cells in Javan mongoose display a more uniform distribution throughout the islets. Endocrine cells were also found within the ductal epithelium, reinforcing the hypothesis that this distribution reflects the underlying developmental and structural characteristics of the pancreas. Overall, this study provides both confirmatory and novel insights into the distribution of endocrine cells in Javan mongoose, thereby enhancing comparative understanding across mammalian species. ConclusionThe pancreas of the Javan mongoose (U. javanica) has a soft consistency and is located dorsally to the duodenal mass. It is ivory yellow with a slight reddish hue and consists of a corpus, dexter lobe, and sinister lobe. Histologically, the Javan mongoose pancreas comprises both exocrine and endocrine components. The exocrine portion includes acini and a duct network. The pancreatic ducts are categorized into five types: intercalated, intralobular, interlobular, accessory, and main pancreatic ducts, all of which are lined by low cuboidal to cuboidal epithelium. The endocrine component consists of the islets of Langerhans, which in the Javan mongoose contain four distinct types of endocrine cells-β, α, δ, and PP. These cells exhibit a heterogeneous distribution, with variations in quantity, depending on the specific islet region. Notably, δ and PP cells are more uniformly distributed across the islets in this species, contrasting with the predominantly peripheral localization observed in other mammals. Furthermore, the presence of endocrine cells within the pancreatic ducts underscores the unique characteristics of the Javan mongoose pancreas, a combination of conserved and species-specific patterns that may reflect evolutionary and metabolic adaptations. AcknowledgmentsThe authors would like to thank Direktorat Jenderal Pendidikan Tinggi, Riset, dan Teknologi (Ditjen Diktiristek) of the Ministry of Education, Culture, Research, and Technology (Kemendikbudristek), for providing research funding through the PMDSU program (Contract No. 067/C3/DT.05.00/PL/2025). The authors also express their gratitude to Universitas Gadjah Mada for its support through the research grant “Program Asistensi Riset-Afirmasi Universitas Gadjah Mada Tahun Anggaran 2025” (Grant No. 1031/UN1.P/KPT/HUKOR/2025). FundingThe author declares that the Direktorat Jenderal Pendidikan Tinggi, Riset, dan Teknologi (Ditjen Diktiristek) of the Ministry of Education, Culture, Research, and Technology (Kemendikbudristek) provided financial support for this research and publication through the PMDSU program under contract number No. 067/C3/DT.05.00/PL/2025, and that additional financial support was provided by Universitas Gadjah Mada through the research grant “Program Asistensi Riset-Afirmasi Universitas Gadjah Mada Tahun Anggaran 2025” (Grant no. 1031/UN1.P/KPT/HUKOR/2025), which provided funding for this research. Authors’ contributionsSAKP contributed to the research implementation, including sample and data collection, data analysis, and the preparation and writing of the manuscript. TB, YHF, and WDW were involved in the study design, supervision of the research process, data analysis, and manuscript development. All authors have read and approved the final version of the manuscript before submission for publication. Conflict of interestThe authors declare that no financial or commercial relationships existed during the course of this research that could be understood as a potential conflict of interest. Data availabilityThe original data and contributions from this study are available in the article or supplementary materials. Further inquiries should be directed to the corresponding author. ReferencesAqiilah, K., Budipitojo, T., Wijayanto, H., Budiariati, V., Pangestiningsih, T.W., Saragih, A., Kustiati, U. and Wihadmadyatami, H. 2024. Morphological Structure of the Tongue of Gekko gecko in Yogyakarta, Indonesia. J. Trop. Biodiversity Biotechnol. 9(4), 1–16; doi:10.22146/jtbb.90995 Arntfield, M.E. and Van Der Kooy, D. 2011. Β-Cell evolution: how the pancreas borrowed from the brain: The shared toolbox of genes expressed by neural and pancreatic endocrine cells may reflect their evolutionary relationship. BioEssays 33(8), 582–587; doi:10.1002/bies.201100015 Asuaje Pfeifer, M., Langehein, H., Grupe, K., Müller, S., Seyda, J., Liebmann, M., Rustenbeck, I. and Scherneck, S. 2023. PyCreas: a tool for quantification of localization and distribution of endocrine cell types in the islets of Langerhans. Front. Endocrinol. 14, 1–13; doi:10.3389/fendo.2023.1250023 Budipitojo, T., Fibrianto, Y.H. and Mulyani, G.T. 2016. The types of endocrine cells in the pancreas of Sunda porcupine (Hystrix javanica). Vet. World 9(6), 563–567; doi:10.14202/vetworld.2016.563-567 Chekunov, S., Stringham, O., Toomes, A., Prowse, T. and Cassey, P. 2024. Scale of unregulated international trade in Australian reptiles and amphibians. Conservation. Biol. 38(5), 1–12; doi:10.1111/cobi.14355 Chutipong, W., Duckworth, J.W., Timmins, R., Willcox, D. H. A. and Ario, A. 2016. Herpestes javanicus. The IUCN Red List of Threatened Species, 2016: Available via https://www.iucnredlist.org/ja/species/70204120/70204139 Cites 2015. CITES Appendices I, II, and III. J. Minim. Access Surg. 4(3), 85–87. Available via http://www.ncbi.nlm.nih.gov/pubmed/19547689 Contini, M., Beguelini, M., Ruiz, T., Taboga, S. and Rafacho, A. 2024. Comparative study of endocrine pancreatic tissue in bats: assessing variations among frugivorous, insectivorous, and nectarivorous diets. Tissue Cell 88, 102413; doi:10.1016/j.tice.2024.102413 Fedchenko, N. and Reifenrath, J. 2014. Different Approaches for Interpretation and Reporting of Immunohistochemistry Analysis Results in The Bone Tissue . A Review. Diagnostic. Pathol. 9, 221. Hauge-Evans, A.C., King, A.J., Carmignac, D., Richardson, C.C., Robinson, I.C.A.F., Low, M.J., Christie, M.R., Persaud, S.J. and Jones, P.M. 2009. Somatostatin secreted by islet δ-cells fulfills multiple roles as a paracrine regulator of islet function. Diabetes 58(2), 403–411; doi:10.2337/db08-0792 Hu, C., Chen, Y., Yin, X., Xu, R., Yin, C., Wang, C. and Zhao, Y. 2025. Pancreatic endocrine and exocrine signaling and crosstalk in physiological and pathological status. Signal. Transduction. Targeted. Therapy. 10(1), 1–30. doi:10.1038/s41392-024-02098-3 Hussein, A.J., Hussein, H.A. and Abdulzahra, H.K. 2018. Amorphological and Histological study of the liver and pancreas of small indian mongoose (Herpestes Javanicus). Basrah. J. Vet. Res. 17(3) , 629–639. König, H.E. and Liebich, H.G. 2020. Veterinary Anatomy of Domestic Animals: textbook and Colour Atlas. 7th ed., New York , NY: Thieme, pp: 391–5. Maher, M.A., Farghali, H.A.M., Elsayed, A.H. and Reem, R.T. 2020. Gross Anatomy and Ultrasonography of Spleen and Pancreas in Rabbit (Oryctolagus cuniculus) and Cat (Felis catus domesticus). Int. J. Vet. Sci. 9(1), 58–65. Mahmood, H.B. and Kadhim, K. 2018. Histomorphology and Histochemical Study of Duodenum and Pancreas in Gray Mongoose (Herpestes edwardsii) In Iraq. Indian J. Natural Sci. 9(51), 15368–15377. Mahmood, T. and Adil, A. 2017. Diet composition of small Indian mongoose (Herpestes javanicus) varies seasonally in its native range. Anim. Biol. 67(1), 69–80; doi:10.1163/15707563-00002516 Mahmood, T., Fazal, F., Akrim, F., Fatima, H. and Nadeem, M.S. 2018. Comparative morphology and anatomy of two sympatric mongoose species (Herpestes javanicus and H. edwardsii) from potohar plateau. Pak. J. Zool. 50(1), 385–387; doi:10.17582/journal.pjz/2018.50.1.sc6 Mescher, A.L. 2018. Junqueira’s Basic Histology: text and Atlas, 15th ed., New York, NY: McGraw-Hill Education, pp: 427–9. Mirontoneng, G.S., Kairupan, C.F. and Durry, M.F. 2019. Gambaran Mikroskopik Endokrin Pankreas pada Tikus Wistar yang Diberikan Sukrosa Dosis Bertingkat. Jurnal. Biomedik. 7(2), 108–112. Overton, D.L. and Mastracci, T.L. 2022. Exocrine-Endocrine Crosstalk: the Influence of Pancreatic Cellular Communications on Organ Growth, Function and Disease. Front. Endocrinol. 13, 1–13; doi:10.3389/fendo.2022.904004 Pauerstein, P.T., Tellez, K., Willmarth, K.B., Park, K.M., Hsueh, B., Arda, H.E., Gu, X., Aghajanian, H., Deisseroth, K., Epstein, J.A. and Kim, S.K. 2017. A radial axis defined by semaphorin-to-neuropilin signalling controls pancreatic islet morphogenesis. Develop. (Cambridge). 144(20), 3744–3754; doi:10.1242/dev.148684 Shil, S., Das, B., Uddin, M., Rahman, M. and Quasem, M. 2012. Anatomy of digestive and respiratory system of Indian grey mongoose (Herpestes edwardsii). Univ. J. Zool. Rajshahi. Univ. 31, 83–84; doi:10.3329/ujzru.v31i0.15438 Shuai, H., Xu, Y., Yu, Q., Gylfe, E. and Tengholm, A. 2016. Fluorescent protein vectors for pancreatic islet cell identification in live-cell imaging. Pflugers. Archiv Eur. J. Physiol. 468(10), 1765–1777; doi:10.1007/s00424-016-1864-z Shunmugam, R. and Veterinary, T.N. 2024. Histo-Architecture of The Pancreas in Adult Dogs. Int. J. Livestock Res. 14(7), 7–13. https://www.researchgate.net/publication/385982641_HistoArchitecture_of_The_Pancreas_in_Adult_Dogs#fullTextFileContent Susdiyanti, T., Cita, K.D., Yuliani, N., Diantama, N. and Sasongko, D.A. 2023. Diversity of Mammals in 8 Years of Jati (Tectona grandis Linn. f) Unggul Nusantara (JUN). Jurnal Sains Natural 13(1), 21; doi:10.31938/jsn.v13i1.395 Suvarna, S.K., Layton, C. and Bancroft, J.D. 2019. Bancroft’s Theory and Practice of Histological Techniques. 8th ed., London, United Kingdom: Elsevier Ltd., pp: 381–426. Tegehall, A., Korsgren, O., Ingvast, S., Gasparyan, G., Granlund, L. and Lundberg, M. 2025. Islet delta-cell architecture is remodelled in the human pancreas during type 1 diabetes. Scientific. Rep. 15(1), 1–8; doi:10.1038/s41598-025-04471-w Teh, H. X., Phang, S. J., Looi, M. L., Kuppusamy, U. R., and Arumugam, B. (2023). Molecular pathways of NF-ĸB and NLRP3 inflammasome as potential targets in the treatment of inflammation in diabetic wounds: a review. Life Sci. 334, 122228; doi: 10.1016/j.lfs.2023.122228 Tsuchitani, M., Sato, J. and Kokoshima, H. 2016. A comparison of the anatomical structure of the pancreas in experimental animals. J. Toxicologic Pathol. 29(3), 147–154; doi:10.1293/tox.2016-0016 Wieczorek, G., Pospischil, A. and Perentes, E. 1998. A comparative immunohistochemical study of pancreatic islets in laboratory animals (rats, dogs, minipigs, nonhuman primates). Exp. Toxicologic. Pathol. 50(3), 151–172; doi:10.1016/S0940-2993(98)80078-X Wieland, F.C., Van Blitterswijk, C.A., Van Apeldoorn, A. and Lapointe, V.L.S. 2021. The functional importance of the cellular and extracellular composition of the islets of Langerhans. J. Immunol. Regenerative Med. 13, 100048; doi:10.1016/j.regen.2021.100048 | ||
| How to Cite this Article |
| Pubmed Style Putri SAK, Budipitojo T, Fibrianto YH, Wendo WD, Budiariati V. Histological distribution of endocrine cells in the pancreas of Javan Mongoose (Urva javanica). Open Vet. J.. 2026; 16(1): 669-678. doi:10.5455/OVJ.2026.v16.i1.62 Web Style Putri SAK, Budipitojo T, Fibrianto YH, Wendo WD, Budiariati V. Histological distribution of endocrine cells in the pancreas of Javan Mongoose (Urva javanica). https://www.openveterinaryjournal.com/?mno=270992 [Access: February 04, 2026]. doi:10.5455/OVJ.2026.v16.i1.62 AMA (American Medical Association) Style Putri SAK, Budipitojo T, Fibrianto YH, Wendo WD, Budiariati V. Histological distribution of endocrine cells in the pancreas of Javan Mongoose (Urva javanica). Open Vet. J.. 2026; 16(1): 669-678. doi:10.5455/OVJ.2026.v16.i1.62 Vancouver/ICMJE Style Putri SAK, Budipitojo T, Fibrianto YH, Wendo WD, Budiariati V. Histological distribution of endocrine cells in the pancreas of Javan Mongoose (Urva javanica). Open Vet. J.. (2026), [cited February 04, 2026]; 16(1): 669-678. doi:10.5455/OVJ.2026.v16.i1.62 Harvard Style Putri, S. A. K., Budipitojo, . T., Fibrianto, . Y. H., Wendo, . W. D. & Budiariati, . V. (2026) Histological distribution of endocrine cells in the pancreas of Javan Mongoose (Urva javanica). Open Vet. J., 16 (1), 669-678. doi:10.5455/OVJ.2026.v16.i1.62 Turabian Style Putri, Sekar Arum Krisna, Teguh Budipitojo, Yuda Heru Fibrianto, Woro Danur Wendo, and Vista Budiariati. 2026. Histological distribution of endocrine cells in the pancreas of Javan Mongoose (Urva javanica). Open Veterinary Journal, 16 (1), 669-678. doi:10.5455/OVJ.2026.v16.i1.62 Chicago Style Putri, Sekar Arum Krisna, Teguh Budipitojo, Yuda Heru Fibrianto, Woro Danur Wendo, and Vista Budiariati. "Histological distribution of endocrine cells in the pancreas of Javan Mongoose (Urva javanica)." Open Veterinary Journal 16 (2026), 669-678. doi:10.5455/OVJ.2026.v16.i1.62 MLA (The Modern Language Association) Style Putri, Sekar Arum Krisna, Teguh Budipitojo, Yuda Heru Fibrianto, Woro Danur Wendo, and Vista Budiariati. "Histological distribution of endocrine cells in the pancreas of Javan Mongoose (Urva javanica)." Open Veterinary Journal 16.1 (2026), 669-678. Print. doi:10.5455/OVJ.2026.v16.i1.62 APA (American Psychological Association) Style Putri, S. A. K., Budipitojo, . T., Fibrianto, . Y. H., Wendo, . W. D. & Budiariati, . V. (2026) Histological distribution of endocrine cells in the pancreas of Javan Mongoose (Urva javanica). Open Veterinary Journal, 16 (1), 669-678. doi:10.5455/OVJ.2026.v16.i1.62 |