| Case Report | ||
Open Vet. J.. 2026; 16(3): 1941-1946 Open Veterinary Journal, (2026), Vol. 16(3): 1941-1946 Case Report A rare case of concurrent renal agenesis and Ectopia in a feline systemYury Carantino Costa Andrade1*, Cicera Paloma de Sousa2, Anne Karoline Mendes da Silva1, Lara Vilela Soares1, Maria Eduarda Raffaini de Oliveira Cunha1, Suellen Rodrigues Maia3, Marian Acácia Fornazier Magalhães1, Patrícia Alves dos Reis1 and Leandro Zuccolotto Crivellenti11Veterinary Hospital of the Federal University of Uberlândia, Brazil 2HD Veterinary Clinic, Fortaleza, Minas Gerais, Brazil 3Department of Veterinary Medicine, Federal Institute of South Minas, Minas Gerais, Brazil *Corresponding Author: Yury Carantino Costa Andrade. Veterinary Hospital of the Federal University of Uberlândia, Minas Gerais, Brazil. Email: yurycarantinocosta [at] gmail.com Submitted: 17/07/2025 Revised: 22/01/2026 bAccepted: 05/02/2026 Published: 31/03/2026 © 2025 Open Veterinary Journal
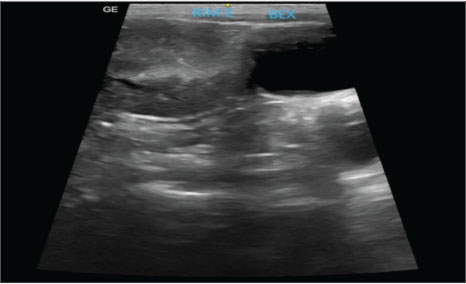
AbstractBackground: This report describes the first documented case of renal agenesis associated with ectopia of the contralateral kidney in a feline in veterinary medicine. Unilateral renal agenesis is a rare congenital anomaly characterized by the complete absence of one kidney. Renal ectopia, in turn, refers to the anomalous location of the kidney and is a rare condition. The simultaneous occurrence of these anomalies is extremely uncommon. Case Description: A 2-year-old spayed female mixed-breed cat presented with apathy, hyporexia, and vomiting. Laboratory tests revealed azotemia, and imaging tests confirmed the absence of the right kidney and the ectopic position of the left kidney, as well as alterations compatible with chronic kidney disease. Abdominal ultrasonography and excretory urography confirmed the diagnosis. Conclusion: Coexisting renal agenesis and ectopia are rare conditions in humans, and there are no reports of them in felines. Despite its rarity, the condition is compatible with life, as long as it is diagnosed early and continuously monitored. Keywords: Anomaly, Excretory urography, Rareness. IntroductionEmbryological development is a highly complex process that requires the precise and synchronized activation of molecular signals at specific moments to ensure adequate organ and system generation (Cohen et al., 2004). When these signs do not occur at the correct time, development can follow an atypical course, resulting in anomalies, such as agenesis, characterized by the complete absence of an organ (Cuckow et al., 2001). Unilateral renal agenesis is a rare congenital anomaly characterized by the complete absence of the kidney. It has been reported in different species, including dogs (Morita et al., 2005), cats (Dykeman, 2020), birds (Oliveira et al., 2020), mice, and humans (González Mediero et al., 1997; De Tomasi et al., 2017). It is even more rarely found when associated with other anomalies, such as ectopia of the contralateral kidney (Oliveira et al., 2020; Plutecki et al., 2023). Renal ectopia, defined by the anomalous location of one or both kidneys, is a rare condition that is usually an incidental finding in humans (Babu et al., 2015) and animals (Allworth and Hoffmann, 1999). The simultaneous occurrence of renal agenesis and ectopia is rarely described in the medical literature (Maachi et al., 2024), and to date, there are no documented reports in the veterinary literature. The diagnosis of renal ectopia and agenesis is obtained through imaging tests, such as radiography, ultrasound, and excretory urography; they allow the identification of the anomalous location of the kidney, associated structural alterations, and the absence of the organ in the case of agenesis (Allworth and Hoffmann, 1999; Rajabioun et al., 2017). This report describes the first documented case of renal agenesis associated with ectopia of the contralateral kidney in a feline, highlighting the diagnostic methods used, therapeutic approach, and subsequent clinical management. Case DetailsA 2-year-old spayed female mixed-breed adult cat weighing 2.15 kg presented with apathy, hyporexia, and acute-onset emesis episodes. The patient’s medical history included previous exams performed at 6 months of age, with negative results for FIV and FeLV (SNAP FIV/FeLV Combo-Idexx® Test), while living with four healthy felines in the same environment. On physical examination, moderate dehydration (7%) was identified, with systolic blood pressure (SBP) of 155 mmHg, measured using the Doppler method, and no other relevant clinical changes. Laboratory tests showed azotemia: creatinine serum: 2.2 mg/dl [reference interval: 0,6–1,6 mg/dl]; urea: 117 mg/dl [reference interval: 10–56 mg/dl], and no relevant changes in the blood count. Urinalysis and urine protein-to-creatinine ratio (UPC) were also performed, revealing a low urine specific gravity (USG: 1.015) (reference interval: USG >1,035) and a UPC of 0.7 (reference interval: UPC < 0,5). The patient was admitted for dehydration correction with intravenous fluid therapy using lactated Ringer’s and supportive therapy for vomiting and hyporexia, which included maropitant citrate (0.1 ml/kg, s.c., SID) and mirtazapine (3.75 mg/animal, PO, every 48 hours). Abdominal ultrasound examination revealed the left kidney in an ectopic position in the left caudal region of the abdomen, adjacent to the bladder. The organ had irregular contours, measuring approximately 3.42 cm in length along the longitudinal axis, with moderate loss of internal structure and reduced corticomedullary differentiation, consistent with chronic nephropathy (Fig. 1). The right kidney was not identified, and the other abdominal organs were within normal limits. Ultrasound findings were consistent with left renal ectopia.
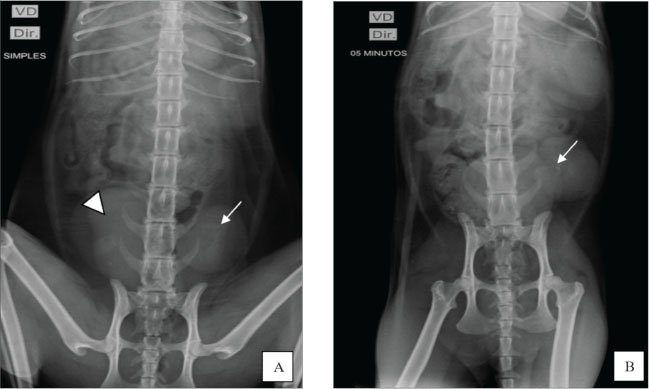
Fig. 1. Ultrasound image showing the left kidney in an ectopic position in the left caudal region of the abdomen, close to the bladder. The organ presented with irregular contours, moderate loss of internal structure, and reduced CM differentiation. Once the patient was stabilized and after considering the absence of the right kidney and atypical location of the left, excretory urography was performed with intravenous administration of iodinated contrast (Omnipaque ®) at a dose of 600 mg/kg via access to the cephalic vein in a rapid bolus to better elucidate the condition. Before contrast media administration, plain abdominal radiographs were performed in ventrodorsal (VD), oblique (the X-ray entered the body through the right ventrolateral side and exited through ventrolateral on the left side), and right lateral projections. All projections showed the absence of the right renal silhouette, while the left kidney was located in the caudal portion of the abdominal cavity, presenting slightly lobulated contours and measuring 3.96 cm in length (Fig. 2A).
Fig. 2. Excretory urography. A) Ventrodorsal plain radiography revealed no visualization of the right renal silhouette, whereas the left kidney (arrow) was identified in the caudal portion of the abdominal cavity, next to the bladder (arrowhead). B) Contrast-enhanced radiograph in the VD projection shows the left ureter filled with contrast (arrow) in the renal pelvis, indicating the beginning of the pyelogram. Greater opacification is noted in the left kidney, which shows signs of slight pelvic dilation. After contrast administration, radiographs were taken in the ventrodorsal, right lateral, and oblique projections at 5, 15, and 30 minutes. At 5 minutes, the image showed the initial phase of the left nephrogram, confirming the absence of the right kidney. At the end of the evaluation, the remaining kidney showed signs of pyelectasis and renal cortical thinning (Fig. 2B). After 24 hours of hospitalization and clinical stabilization, the animal was discharged from the hospital and monitored for 1 month. Four weeks after the absence of clinical signs, the complementary examinations showed stability compared with the examinations at admission. At that time, laboratory findings indicated azotemia: creatinine: 2.6 mg/Dl [reference interval: 0,6–1,6 mg/dl]); urea: 120 mg/dl [reference interval: 10–56 mg/dl]; phosphorus: 5,0 mg/dl [reference interval: 3,5–4,5 mg/dl]) and significant proteinuria (UPC: 0.91). Treatment was instituted with a specific renal diet (Royal Canin Veterinary Diet Renal para Gatos®), enalapril (0.25 mg/kg, BID), and omega-3 supplementation (40 mg EPA + 25 mg DHA/kg). The patient remained clinically stable and was monitored with routine quarterly exams, aiming to monitor the stage II of chronic kidney disease (CKD) according to the International Renal Interest Society (IRIS) classification. Evolution of ureter and renal pelvis dilation in the last evaluation, approximately 3 months after the initial consultation, no changes in renal pelvis dilation were observed compared with the examination previously described in this study. The patient is stable and is being monitored periodically every 2 months. DiscussionTo date, no records have been found describing the simultaneous occurrence of renal agenesis and ectopia in cats, making this report the first to document the condition in cats. It is important to highlight related differential diagnoses, such as solitary crossed renal ectopia, a condition in which one of the kidneys is absent and the ureter crosses the midline. This anomaly has been described in 35 cases in the medical literature and, more rarely, in veterinary literature, including the variant with renal fusion (Allworth and Hoffmann, 1999; Hebel et al., 2020; Maachi et al., 2024). This possibility was ruled out in our case because it was possible to identify the path of the ureter and the position of the remaining kidney through excretory urography. Generally, congenital anomalies of the urinary tract are rarely reported in veterinary medicine and usually manifest through nonspecific clinical signs when they occur (Choi et al., 2012). In the presented case, the nonspecific clinical signs led to laboratory and imaging tests, which were essential to confirm the diagnosis. Renal ectopia is often an incidental finding, with no evident functional impairment (Rajabioun et al., 2017); however, associated alterations, such as hydronephrosis and ectopic ureters, should be investigated. Our report showed alterations compatible with chronicity in the remaining kidney, which may be related to functional renal mass and nephron number reduction. This condition triggers compensatory mechanisms, such as hypertrophy and hyperfiltration, which, over time, can lead to progressive renal damage, culminating in CKD (Brenner et al., 1988). In medicine, the term “solitary kidney” has been used to describe patients who have only one kidney, either due to unilateral renal agenesis, congenital hypoplasia, or surgical removal. A study conducted by Kim et al. (2019) involving patients with a single kidney due to nephrectomy or congenital causes, demonstrated that those who underwent nephrectomy had a higher risk of developing CKD. This difference may be attributed to the early onset of adaptive mechanisms in cases of congenital solitary kidney, which promotes a more efficient compensatory functional response. However, unlike what they reported, the animal in the present case had no history of nephrectomy and was a young patient, but already presented signs compatible with renal chronicity, which shows that functional adaptation may not be sufficient to prevent early development of kidney damage even in congenital cases. In this report, the definitive diagnosis of renal ectopia and agenesis was established based on excretory urography (Chang et al., 2008) and abdominal ultrasonography, without the need for surgical intervention (Heuter, 2005; Dykeman, 2020). Hebel et al. (2020) described the use of excretory urography for renal morphological and functional assessment in a case of crossed renal ectopia in a feline, reinforcing the importance of this modality as a diagnostic tool. Concomitant alterations of the urinary and reproductive systems are possible due to the common embryonic origin in the intermediate mesoderm, which justifies the frequent association between renal and uterine anomalies (Dykeman, 2020). Several cases involving this correlation have already been described in the veterinary literature, such as that of a Persian female with unilateral aplasia of the right uterine horn, ipsilateral renal aplasia, and absence of the corresponding ureter, and that of a 1-year-old cat diagnosed with renal agenesis associated with segmental aplasia of the uterus after surgical reintervention (Goo et al., 2009; Carvalho et al., 2013). However, unlike these cases, the patient in this report had already undergone ovariohysterectomy and did not present any clinical signs of remaining ovaries. The observed alterations were restricted to the urinary system, with emphasis on agenesis of the right kidney and ectopia of the left kidney. The smaller celiotomy incision for ovariohysterectomy is a minimally invasive approach that may result in less tissue trauma and faster recovery (Naiman et al., 2014). However, a potential disadvantage of this technique is its restricted field of view. Unlike wide-incision laparotomy, reduced celiotomy limits complete visual inspection or adequate digital palpation of all abdominopelvic structures (Mayhew et al., 2014). In the described case, an incision of 3 cm (1.18 inches) was performed, which significantly restricted the visualization and manipulation of intra-abdominal structures. This possibly prevented the identification of the ectopic kidney, which was located adjacent to the bladder, and the absence of the contralateral kidney during the surgical procedure. Although renal ectopia is asymptomatic in many cases, this congenital anomaly should be considered among the differential diagnoses when abdominal masses located in the caudal region are present, especially when observed on plain radiographs. Despite its limitations in detailed anatomical assessment, abdominal radiography can efficiently exhibit deviations from the expected renal topography. The identification of an ectopic kidney can be challenging in the absence of clinical signs or obvious functional changes, as demonstrated in the present report (Webb, 1974). In these cases, additional imaging tests, such as excretory urography, are essential for diagnostic confirmation, offering a less invasive alternative to exploratory laparotomy. To date, the reported case represents the first documented case of this anomaly in felines, contributing to the understanding of congenital urinary tract malformations in small animals. The scarcity of similar reports makes estimating prognosis and survival rates difficult. The patient’s clinical follow-up demonstrated good general condition, with tests compatible with stage II CKD, according to the IRIS classification (2023). The patient had persistent azotemia, mild arterial hypertension (150 mmHg), and significant proteinuria (urine protein-to-creatinine ratio, UPC: 0.91), with a urine specific gravity (USG) of 1.010. Given these findings, treatment was instituted with a specific renal diet (Royal Canin Veterinary Diet Renal para Gatos®), enalapril, and omega-3 fatty acid supplementation, as recommended by IRIS (2023). The use of angiotensin-converting enzyme inhibitors contributed to the control of proteinuria and arterial hypertension, lowering the risk of organ damage. Continuous clinical and laboratory monitoring was performed in patients with renal agenesis and ectopia, focusing on the management of chronic kidney disease. Stage II indicates impaired renal function even in the absence of clinical signs, justifying the need for periodic monitoring. This protocol includes comprehensive laboratory evaluations, SBP measurement, and imaging tests to monitor disease progression and detect early complications associated with ureteral changes. Due to their abnormal location, ectopic kidneys are more susceptible to complications such as uroliths, hydronephrosis, and pyelonephritis due to inefficient urinary drainage (Rajabioun et al., 2017). In this case, the drainage was impaired, resulting in pyelectasis and subsequent CKD diagnosis. Proper clinical staging and regular follow-up are essential for effective management and to prevent worsening of the condition (Webb, 1974; Arena et al., 2007). ConclusionThe coexistence of renal agenesis and ectopia is rare in humans and has not been reported in felines in the veterinary literature. Despite its rarity, this condition is compatible with life when diagnosed early and continuously monitored. Diagnostic confirmation requires imaging tests, such as abdominal ultrasound and contrast-enhanced radiographs, to identify the absence of a kidney, the ectopic location of the remaining kidney, and any associated urinary tract changes. AcknowledgmentsThe authors express their gratitude to Dr. Lucas Facundo from Radiosonar for kindly providing the ultrasound image and to Dr. Antonia Iraina from Veter for providing the contrast radiograph image. Conflict of interestThe authors declare no conflict of interest. FundingThis study received no specific grant. Authors' contributionsYury Carantino Costa Andrade: Original draft, Investigation, Data curation, Conceptualization. Cicera Paloma de Sousa: Writing, review, and editing. Anne Karoline Mendes da Silva: Writing, reviewing, and editing. Lara Vilela Soares: Writing, reviewing, and editing. Maria Eduarda Raffaini de Oliveira Cunha: Writing, review, and editing. Suellen Rodrigues Maia: Writing, review, and editing. Marian Acácia Fornazier Magalhães: Writing, review, and editing. Patrícia Alves dos Reis: Writing, review, and editing. Leandro Zuccolotto Crivellenti: Writing, review, and editing, validation, and supervision. Data availabilityAll data generated or analyzed during this study are included in this published article. ReferencesAllworth, M.S. and Hoffmann, K.L. 1999. Crossed renal ectopia with fusion in a cat. Radiol. Ultrasound 40(4), 357–360; doi:10.1111/j.1740-8261.1999.tb02125.x Arena, F., Arena, S., Paolata, A., Campenni, A., Zuccarello, B. and Romeo, G. 2007. Is a complete urological evaluation necessary in all newborns with asymptomatic renal ectopia?. Int. J. Urol. 14(6), 491–495; doi:10.1111/j.1442-2042.2007.01764.x Babu, C.S.R., Agrawal, B., Sharma, V., Kumar, A., Jain, R. and Gupta, O.P. 2015. Crossed renal ectopia without fusion: a multidetector computed tomography study. Int. J. Anat. Res. 3(4), 1585–1592; doi:10.16965/ijar.2015.293 Brenner, B.M., Garcia, D.L. and Anderson, S. 1988. Glomeruli and blood pressure. Less of one, more of the other?. Am. J. Hypertens. 1(4 Pt 1), 335–347. Carvalho, F.R., Wartluft, A.N. and Melivilu, R.M. 2013. Unilateral uterine segmentary aplasia, papillary endometrial hyperplasia, and ipsilateral renal agenesis in a cat: a case report. Feline. Med. Surg. 15(4), 349–352; doi:10.1177/1098612X12467786 Chang, J., Jung, J.H., Yoon, J., Choi, M.C., Park, J.H., Seo, K.M. and Jeong, S.M. 2008. Segmental aplasia of the uterine horn with ipsilateral renal agenesis in a cat. J. Vet. Med. Sci. 70, 641–643; doi:10.1292/jvms.70.641 Choi, J., Lee, H., Lee, Y. and Choi, H. 2012. Simple ectopic kidney in three dogs. J. Vet. Med. Sci. 74, 1373–1375. Cohen, H.L., Kravets, F., Zucconi, W., Ratani, R., Shah, S. and Dougherty, D. 2004. Congenital genitourinary system abnormalities. Semin. Roentgenol. 39(2), 282–303; doi:10.1053/j.ro.2003.12.005 Cuckow, P.M., Nyirady, P. and Winyard, P.J.D. 2001. Normal and abnormal development of the urogenital tract. Prenat. Diagn. 21(11), 908–916; doi:10.1002/pd.214 De Tomasi, L., David, P., Humbert, C., Silbermann, F., Arrondel, C., Tores, F., Fouquet, S., Desgrange, A., Niel, O., Bole-Feysot, C., Nitschké, P., Roume, J., Cordier, M.P., Pietrement, C., Isidor, B., Khau Van Kien, P., Gonzales, M., Saint-Frison, M.H., Martinovic, J., Novo, R., Piard, J., Cabrol, C., Verma, I.C., Puri, R., Journel, H., Aziza, J., Gavard, L., Said-Menthon, M.H., Heidet, L., Saunier, S. and Jeanpierre, C. 2017. Mutations in GREB1L Cause Bilateral Kidney Agenesis in Humans and Mice. Am. J. Hum. Genet. 101(5), 803–814; doi:10.1016/j.ajhg.2017.09.026 Dykeman, D. 2020. Segmental uterine aplasia and ipsilateral renal agenesis in a ragdoll cat. Can. Vet. J. 61, 424–426. González Mediero, J.A., García, J., Tinajas Saldaña, A., Santiago, D., González Vicente, A.M. and Urrutia Avisrros, M. 1997. Unilateral renal agenesis associated with pelvic renal ectopy. Arch. Esp. Urol. 50(9), 1004–1006. Goo, M.J., Williams, B.H., Hong, I.H., Park, J.K., Yang, H.J., Yuan, D.W., Lee, H.R., Hong, K.S., Ki, M.R. and Jeong, K.S. 2009. Multiple urogenital abnormalities in a Persian cat. Feline Med. Surg. 11(2), 153–155; doi:10.1016/j.jfms.2008.04.007 Hebel, M., Ruszkowski, J.J., Giza, E. and Pomorska-Mól, M. 2020. Crossed renal ectopia with fusion in a pelvic inlet area, atypical portal vein and coccygeal deformation in a young female cat. BMC Vet. Res. 16(1), 314; doi:10.1186/s12917-020-02535-9 Heuter, K.J. 2005. Clin. Tech. Excretory urography Small Anim. Pract. Small. Anim. Pract. 20(1), 39–45. International Renal Interest Society (IRIS). 2023. IRIS Staging of Chronic Kidney Disease (CKD). Available via https://www.iris-kidney.com/guidelines/staging.html Kim, S., Chang, Y., Lee, Y.R., Jung, H.S., Hyun, Y.Y., Lee, K.B., Joo, K.J., Yun, K.E., Shin, H. and Ryu, S. 2019. Solitary kidney and risk of chronic kidney disease. Eur. J. Epidemiol. 34(9), 879–888; doi:10.1007/s10654-019-00520-7 Maachi, Y., Babty, M., Fouimtizi, J., Slaoui, A., Karmouni, T., El Khader, K., Koutani, A. and Ibn Attya Al Andaloussi, A. 2024. Crossed renal ectopia: a case report and literature review. Urol. Case. Rep. 54, 102690; doi:10.1016/j.eucr.2024.102690 Mayhew, P.D., Mayhew, K.N., Shilo-Benjamini, Y., Kass, P.H. and Pascoe, P.J. 2014. Prospective evaluation of access incision position for minimally invasive surgical organ exposure in cats. J. Am. Vet. Med. Assoc. 245(10), 1129–1134; doi:10.2460/javma.245.10.1129 Morita, T., Michimae, Y., Sawada, M., Uemura, T., Araki, Y., Haruna, A. and Shimada, A. 2005. Renal Dysplasia with Unilateral Renal Agenesis in a Dog. J. Comp. Pathol. 133(1), 64–67; doi:10.1016/j.jcpa.2005.01.002 Naiman, J.H., Mayhew, P.D., Steffey, M.A., Culp, W.T.N., Runge, J.J. and Singh, A. 2014. Laparoscopic treatment of ovarian remnant syndrome in dogs and cats: 7 cases (2010-2013). J. Am. Vet. Med. Assoc. 245, 1251–1257; doi:10.2460/javma.245.11.1251 Oliveira, E.L.R., Costa, A.L.M., Borges, M.F., Nemer, V.C., Baldissera, R.A., Vasconcelos, M., Pereira, K.H.N.P. and Schimming, B.C. 2020. Unilateral Renal Agenesis in P. chilensis in Chilean-Flamingo. Acta Sci. Vet. 48, 496; doi: 10.22456/1679-9216.100607 Plutecki, D., Kozioł, T., Bonczar, M., Ostrowski, P., Skorupa, A., Matejuk, S., Walocha, J., Pękala, J., Musiał, A., Pasternak, A. and Koziej, M. 2023. Renal agenesis: a meta‐analysis of its prevalence and clinical characteristics based on 15,641,184 patients. Nephrology 28, 525–533. Rajabioun, M., Salari Sedigh, H. and Mirshahi, A. 2017. Bilateral simple ectopic kidney in a cat: a case report. Forum 8(2), 175. Webb, A.I. 1974. Renal ectopia in a dog. Aust. Vet. J. 50, 519–521; doi:10.1111/j.1751-0813.1974.tb14059.x/
| ||
| How to Cite this Article |
| Pubmed Style Andrade YCC, Sousa CPD, Silva AKMD, Soares LV, , Maia SR, Magalhães MAF, Reis PAD, Crivellenti LZ. A rare case of concurrent renal agenesis and Ectopia in a feline system. doi:10.5455/OVJ.2026.v16.i3.49 Web Style Andrade YCC, Sousa CPD, Silva AKMD, Soares LV, , Maia SR, Magalhães MAF, Reis PAD, Crivellenti LZ. A rare case of concurrent renal agenesis and Ectopia in a feline system. https://www.openveterinaryjournal.com/?mno=271339 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.49 AMA (American Medical Association) Style Andrade YCC, Sousa CPD, Silva AKMD, Soares LV, , Maia SR, Magalhães MAF, Reis PAD, Crivellenti LZ. A rare case of concurrent renal agenesis and Ectopia in a feline system. doi:10.5455/OVJ.2026.v16.i3.49 Vancouver/ICMJE Style Andrade YCC, Sousa CPD, Silva AKMD, Soares LV, , Maia SR, Magalhães MAF, Reis PAD, Crivellenti LZ. A rare case of concurrent renal agenesis and Ectopia in a feline system. doi:10.5455/OVJ.2026.v16.i3.49 Harvard Style Andrade, Y. C. C., Sousa, . C. P. D., Silva, . A. K. M. D., Soares, . L. V., , Maia, . S. R., Magalhães, . M. A. F., Reis, . P. A. D. & Crivellenti, . L. Z. (2026) A rare case of concurrent renal agenesis and Ectopia in a feline system. doi:10.5455/OVJ.2026.v16.i3.49 Turabian Style Andrade, Yury Carantino Costa, Cicera Paloma De Sousa, Anne Karoline Mendes Da Silva, Lara Vilela Soares, Maria Eduarda Raffaini De Oliveira Cunha, Suellen Rodrigues Maia, Marian Acácia Fornazier Magalhães, Patrícia Alves Dos Reis, and Leandro Zuccolotto Crivellenti. 2026. A rare case of concurrent renal agenesis and Ectopia in a feline system. doi:10.5455/OVJ.2026.v16.i3.49 Chicago Style Andrade, Yury Carantino Costa, Cicera Paloma De Sousa, Anne Karoline Mendes Da Silva, Lara Vilela Soares, Maria Eduarda Raffaini De Oliveira Cunha, Suellen Rodrigues Maia, Marian Acácia Fornazier Magalhães, Patrícia Alves Dos Reis, and Leandro Zuccolotto Crivellenti. "A rare case of concurrent renal agenesis and Ectopia in a feline system." doi:10.5455/OVJ.2026.v16.i3.49 MLA (The Modern Language Association) Style Andrade, Yury Carantino Costa, Cicera Paloma De Sousa, Anne Karoline Mendes Da Silva, Lara Vilela Soares, Maria Eduarda Raffaini De Oliveira Cunha, Suellen Rodrigues Maia, Marian Acácia Fornazier Magalhães, Patrícia Alves Dos Reis, and Leandro Zuccolotto Crivellenti. "A rare case of concurrent renal agenesis and Ectopia in a feline system." doi:10.5455/OVJ.2026.v16.i3.49 APA (American Psychological Association) Style Andrade, Y. C. C., Sousa, . C. P. D., Silva, . A. K. M. D., Soares, . L. V., , Maia, . S. R., Magalhães, . M. A. F., Reis, . P. A. D. & Crivellenti, . L. Z. (2026) A rare case of concurrent renal agenesis and Ectopia in a feline system. doi:10.5455/OVJ.2026.v16.i3.49 |