| Research Article | ||
Open Vet. J.. 2026; 16(1): 653-668 Open Veterinary Journal, (2026), Vol. 16(1): 653-668 Review Article Vitamin-supplemented medical-grade honey for burn wound management in cats and dogs: a retrospective case seriesAlexandra Peteoacă1, Stathis A. Symeonidis2, Vasiliki Tachmatzidou3, Noëlle A. C. J. M. Paping4, Carlos C. F. Pleeging5, Linsey J. F. Peters6 and Niels A. J. Cremers6,7*1University of Agronomic Sciences and Veterinary Medicine of Bucharest, Faculty of Veterinary Medicine, Bucharest, Romania 2Small Animal Clinic—Stathis Symeonidis and Maria Kosmidou, Kavala, Greece 3Veterinary Clinic Vasiliki Tachmatzidou, Polygyros, Greece 4Dierenartsenpraktijk Paping, Maastricht, The Netherlands 5Dierenkliniek Bingelrade, Bingelrade, The Netherlands 6Triticum Exploitatie BV, Maastricht, The Netherlands 7Department of Gynecology and Obstetrics, Maastricht University Medical Centre, Maastricht, The Netherlands *Corresponding Author: Niels A. J. Cremers. Triticum Exploitatie BV, Maastricht, The Netherlands. Submitted: 18/07/2025 Revised: 21/11/2025 Accepted: 08/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: Burn wounds in companion animals are uncommon but can lead to serious complications and be life-threatening, requiring prompt and effective wound care. Conventional treatment methods often involve silver- or antibiotic-based products, which carry disadvantages such as cytotoxicity and risk of antimicrobial resistance. Medical-grade honey (MGH), especially when supplemented with vitamins C and E, offers a potential alternative due to its antimicrobial, anti-inflammatory, and wound-healing properties. Aim: This study aims to evaluate the clinical outcomes of vitamin-supplemented MGH in the management of burn wounds in cats and dogs. Methods: A retrospective, multi-center case series was conducted across three veterinary clinics in Romania, Greece, and the Netherlands. Nine cases involving cats (n=4) and dogs (n=5) with thermal, chemical, or iatrogenic burn wounds were included. All wounds were treated with MGH-based products (L-Mesitran range), and wound progression was documented photographically. Descriptive statistics (mean ± SD) were calculated for key parameters, including time to visible inflammation reduction, debridement, granulation tissue formation, re-epithelialization, and complete healing. Results: All patients achieved complete wound healing without complications. On average, signs of inflammation reduced within 5.2 ± 2.0 days (mean ± SD), debridement occurred in 6.7 ± 2.2 days, granulation tissue appeared after 11.7 ± 4.1 days, re-epithelialization was shown in 23.6 ± 5.9 days, and full wound closure was achieved in 64.7 ± 20.7 days. These clinical outcomes varied between individual cases, depending on wound age, size, severity, location, and patient characteristics. Conclusion: Vitamin-supplemented MGH showed promising outcomes in promoting wound healing in feline and canine burn patients. However, the small sample size, retrospective design, and lack of a control group limit the generalizability of these findings. Keywords: Burns, Cats, Dogs, Medical-grade honey, Wound healing. IntroductionBurn injuries are a leading cause of morbidity and mortality worldwide, accounting for approximately 180,000 deaths per year (Organisation, 2018). Burn wounds in domestic animals are relatively uncommon (Volk and Bohling, 2013). Nonetheless, these wounds represent a great challenge to veterinarians as they can cause serious pulmonary, metabolic, and cardiovascular aberrations and, in the most serious cases, death (Vaughn and Beckel, 2012). Most burn injuries in animals are associated with domestic accidents or the use of supplemental heat at veterinary practices to prevent hypothermia in the patient during or after anesthesia (Pavletic and Trout, 2006; Lagutchik and Ford, 2012). The severity of a burn is determined by the percentage of total body area affected, the depth of the tissue damage, and the etiology. Burn injuries are primarily classified as partial-thickness and full-thickness burns (Żwierełło et al., 2023). A partial-thickness burn involves limited damage to the epidermis and outer dermis. These burns result in quick recovery within 2 weeks and a low risk of scarring. Full-thickness burns cause extensive skin damage, typically requiring 3–6 weeks to heal and posing ahigher risk of scarring (Żwierełło et al., 2023). Severe burn wounds are associated with complex immunological and inflammatory responses, as well as systemic shock, all of which can be difficult to manage and may lead to multiple organ failure (Jeschke et al., 2020; Żwierełło et al., 2023). Delayed wound healing due to complications such as infection remains a major challenge when treating burn injuries. Conventional treatment options typically include silver-based products, such as silver sulphadiazine (SSD), and topical antibiotic ointments. However, these products have notable drawbacks, including cytotoxicity and the potential to promote antimicrobial resistance (Innes et al., 2001; Kędziora et al., 2018; Khansa et al., 2019). Consequently, there is a growing need to explore alternative and adjunctive approaches for burn management. Honey represents one such promising alternative. Although it has been used for centuries as a natural remedy for wound care, it has only regained increasing interest over the past few years in response to the rise of antimicrobial resistance. Honey exhibits broad-spectrum antimicrobial effects, which are based on its osmotic activity, the formation of hydrogen peroxide, its acidity, and the presence of antimicrobial molecules (Mandal and Mandal, 2011; Kwakman and Zaat, 2012; Almasaudi, 2021) . In addition to reducing the bacterial load, honey also promotes wound healing in a multifaceted manner, including creating a moist wound environment and stimulating autolytic debridement (Smaropoulos and Cremers, 2019; Pleeging et al., 2020). For clinical application, honey must meet specific standards and is referred to as medical-grade honey (MGH). MGH is produced and processed according to rigorous conditions to ensure safety and efficacy (Hermanns et al., 2020; Peters et al., 2025). These criteria include, but are not limited to, the honey being of organic origin and having undergone gamma-irradiation to inactivate bacterial spores. Currently, many MGH-based wound care products are on the market. Multiple studies have demonstrated the superior effects of MGH supplemented with additional compounds such as vitamins C and E (Subrahmanyam, 1998; Majtan et al., 2020; Pleeging et al., 2020). The use of supplemented MGH for the treatment of burns has been shown in several clinical studies in both animals and humans (De Rooster and Declercq, 2008; Maravelis et al., 2015; Kokkinos et al., 2017; Andrei Zbuchea, 2017; Smaropoulos and Cremers, 2020; Lukanc and Erjavec, 2022; Boekema et al., 2023). For instance, Lukanc and Erjavec (2022) showed the successful use of supplemented MGH in chemical, thermal, and iatrogenic burns in domestic animals (Lukanc and Erjavec, 2022). Moreover, ex vivo studies comparing SSD and supplemented MGH have shown that SSD may exert detrimental effects during prolonged use, whereas MGH enhances wound healing (Boekema et al., 2013; Boekema et al., 2023). Despite these promising results, literature on the use of MGH in veterinary settings remains limited (Vogt et al., 2020). To our knowledge, this is one of the few veterinary case series documenting the systematic use of MGH across multiple centers for burn treatment in companion animals. Although case series are considered to be of lower-level evidence, they are of high importance to showcase the clinical application of MGH and form the backbone for future studies. The application of the different dressings based on MGH for burns in veterinary patients has been scarcely reported. Therefore, the current retrospective case series aims to demonstrate that MGH is a safe, effective, and practical alternative to conventional wound care products in feline and canine patients with burn wounds of various causes. Materials and MethodsStudy design and case selectionA retrospective, multi-center case series was conducted to evaluate the clinical outcomes of burn wound treatment in companion animals using MGH. Nine cases were identified from three veterinary clinics located in Romania, Greece, and the Netherlands between 2022 and 2024 (Table 1). Cases were included if they met the following criteria: 1). The patient was a cat or dog presenting with burn wounds (thermal, chemical, or iatrogenic); 2). The burn was treated with medical-grade honey (L-Mesitran products), either as monotherapy or in combination with standard wound care measures; 3). Complete clinical records and photographic documentation were available throughout the treatment period; and 4). Informed owner consent was obtained for both treatment and case publication. All participating veterinarians were familiar with the use of MGH-based wound products, ensuring comparability across cases. Table 1. Overview of the included cases and treatment regimen.
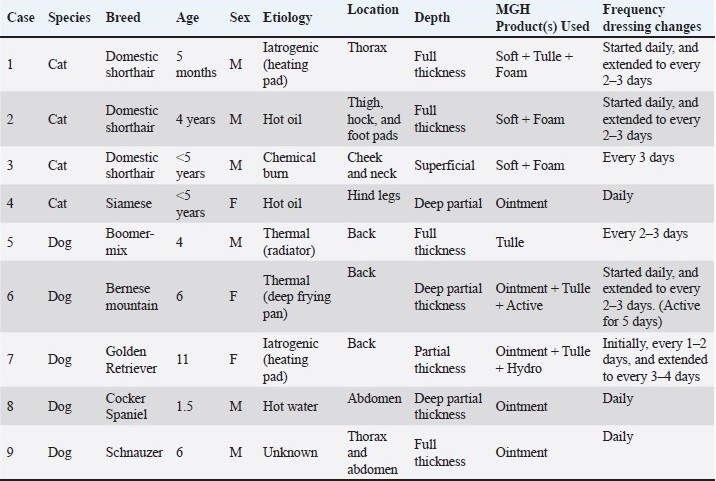
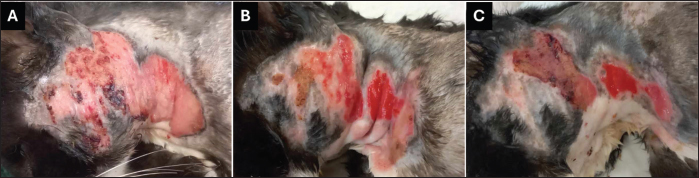
Data collectionPatient data were extracted from medical records and included species, breed, age, sex, cause, and classification of the burn, treatment protocol, healing time, complications, and outcome. Burn severity and depth were categorized according to veterinary burn classification systems (Vaughn and Beckel, 2012; Żwierełło et al., 2023), using visual cues such as color, moisture, and tissue texture to distinguish between partial- and full-thickness burns. Wound assessment data were derived from daily or periodic clinical notes and photographic evidence. Each photograph was dated and evaluated for the presence of key wound healing characteristics. When available, serial wound photographs were compared to track visual progression through inflammation, debridement, granulation, and epithelialization phases. Treatment approachAll animals were treated with MGH-based products (L-Mesitran Soft, Tulle, Ointment, Foam, or Hydro). Product selection was based on the wound’s anatomical location, exudate level, burn depth, and healing phase, as determined by the veterinarian. MGH was applied directly to the wound bed in a 2–3 mm thick layer and covered with an appropriate sterile secondary dressing. Wound cleansing prior to MGH application involved irrigation with saline or Ringer’s lactate solution to remove debris and exudate. In selected cases, mechanical or surgical debridement of necrotic tissue was performed before initiating MGH therapy. Dressing changes were initially performed daily to monitor early wound response closely and to prevent excessive exudate accumulation. Once infection and inflammation were controlled, the frequency of dressing changes was extended to every 2–3 days, depending on wound condition and exudate level. Systemic treatments (antibiotics, non-steroidal anti-inflammatory drugs, or corticosteroids) were administered when clinically indicated. Treatment duration and progression were individualized based on wound response. Assessment of wound healing parametersThe following five clinical parameters were systemically assessed for each case. 1). Reduction of inflammation: determined by a decrease in any sign of inflammation, such as erythema, edema, or exudate, or a decrease in superficial/deep infection signs as recognized by NERDS/STONEES (Woo and Sibbald, 2009). 2). Debridement: defined as the time to a clear decrease in necrotic or devitalized tissue, either spontaneously (autolytic) or assisted (mechanical/surgical). 3). Granulation tissue formation: identified by the appearance of bright red, moist, and finely granular tissue covering the wound bed. 4). Re-epithelialization: determined by the visible migration of epithelial cells from wound edges or islands, giving a glossy pink surface. 5). Complete wound healing: defined as full closure of the wound, with no open areas, infection, or necrotic tissue. The mean +/- standard deviation (SD) of the different outcome parameters was presented to provide indicative guidance regarding healing times. However, due to the relatively small sample size and the descriptive nature of the study, no statistical analyses were performed. Each parameter was recorded in days from the start of MGH treatment. However, some of the wounds were not treated directly with MGH and may have been there for a while; therefore, outcome parameters may be affected, explaining why only descriptive statistics were possible. Ethical approvalThis study did not require evaluation by an ethics committee, as the research was retrospective in nature and the subjects were not physically involved in the study. This work involved the use of non-experimental animals only, and the treatment was part of routine clinical care (non-interventional). Patients were treated according to local wound care protocols, and the MGH formulation used in this study is a registered (CE-certified and FDA-approved) wound care product intended for topical use in both humans and animals. Furthermore, owner consent was obtained for the use of images and data for publication. Patients are not recognizable from the images or associated data, and all efforts were madeto maintain the highest standards of scientific and ethical integrity. ResultsClinical cases showing the use of supplemented MGHIn the following section, we will present nine clinical cases demonstrating the application of supplemented MGH in veterinary patients. Case 1: Iatrogenic thermal burn injury in a catA 5-month-old male domestic shorthair cat was admitted to the hospital after being involved in a road traffic accident, which resulted in a diaphragmatic hernia and acetabular fracture. The patient underwent surgical intervention, including diaphragmatic hernia repair and femoral head and neck resection. Following surgery, systemic antibiotics (Amoxicillin and Clavulanic acid) were administered for 7 days. The patient had an adequate postoperative recovery, with stabilization achieved 3 days after the initial consultation. However, 3–4 days post-surgery, a non-delimited escharotic lesion was observed on the left thoracic wall. This indicated a moderate iatrogenic full-thickness thermal burn, most likely caused by a faulty heating pad used during surgery. Partial escharotomy was performed on day 12 post-surgery, marking the initiation of wound care. Blood analysis showed minor changes, including neutrophilia, monocytosis, and hematocrit values at the lower end of the reference range. The wound showed a high level of exudate and slough (Fig. 1A), and a wound swab confirmed infection with beta-hemolytic Streptococcus spp and Escherichia coli, both sensitive to doxycycline. Wound care products based on MGH supplemented with vitamins were used for wound management. For initial exudate control, an MGH-based foam dressing (L-Mesitran Foam) was applied, with dressings changed daily. Analgesia (Buprenorphine) was administered every 12 hours for the first 10 days.
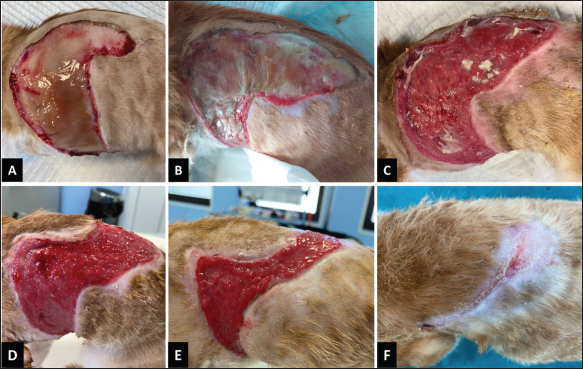
Fig. 1. Wound healing progression of the iatrogenic thermal burn injury in a feline patient. A) The wound at the start of the MGH therapy, immediately following the escharotomy (day 0). B) Exudate was significantly reduced 6 days after the start of the treatment, and a dry, yellow scab remained. C) The wound was almost fully debrided on day 11. D) On day 16 of the MGH therapy, the wound was fully debrided, and a healthy wound bed was shown. E) Re-epithelialization became evident after 27 days of MGH treatment. F) The wound was almost fully healed on day 62. By day 6 of the MGH therapy, the exudate had reduced, but yellow slough remained and formed a scab (Fig. 1B). After 11 days of treatment, the yellow scab loosened and could be cut off, revealing an almost fully debrided wound bed (Fig. 1C). Because of the marked wound improvements, dressing changes were reduced from daily to every 2–3 days from day 14 onwards. On day 16 of MGH therapy, the wound showed healthy granulation tissue, and 11 days later, re-epithelialization became evident (Fig. 1D and E). Due to the exudate control, wound care products was transitioned from the MGH-based foam to an MGH-impregnated non-adherent gauze (L-Mesitran Tulle) combined with wound gel (L-Mesitran Soft). MGH therapy achieved almost full wound closure after 62 days, and only a small area still required treatment with the MGH-based wound gel (Fig. 1F). The wounds were fully healed without further complications after 67 days of treatment. Case 2: Hot oil burn in a catA 4-year-old intact male domestic shorthair cat presented to the emergency clinic following a burn injury resulting from contact with hot oil after jumping into a frying pan on the stove. The initial assessment revealed second- and third-degree burns, primarily affecting the plantar and digital pads of the hind limbs (Fig. 2A). A treatment plan was promptly initiated, involving systemic antibiotic therapy (Amoxicillin and Clavulanic acid) and analgesia (Buprenorphine). Due to the severity of the burns, the patient was admitted for further surgical reevaluation. Three days later, a detailed examination revealed a linear third-degree burn on the inner surface of the thigh extending to the knee. Additionally, third-degree burns were observed on the cranial and lateral aspects of the hock (Fig. 3A and E), along with similar lesions on the plantar pads and the posterior aspect of the right hock. Notably, a clear demarcation between healthy tissue and the burn sites was evident.
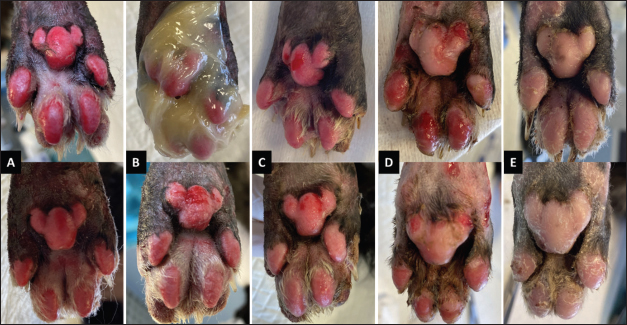
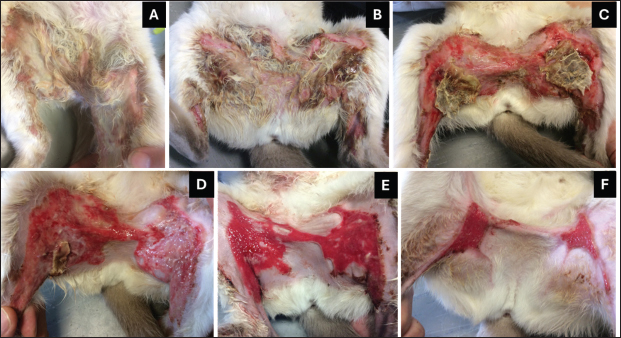
Fig. 2. Wound healing progression of the foot pads of the patient. The top row represents the right hind foot pads, and the bottom the left hind foot pads. A) The day of admission into the hospital. B) The foot pads at the start of the MGH therapy (day 0). The top image shows the application of the MGH-based wound gel. C) On day 2 of the MGH therapy, the foot pad showed a notable reduction in erythema. D) One week later, after 9 days of MGH treatment, debridement became evident, and healthy granulation tissue was shown. E) The wounds were fully healed after 20 days of MGH therapy without further complications.
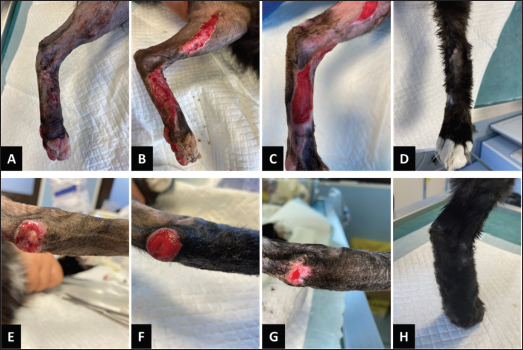
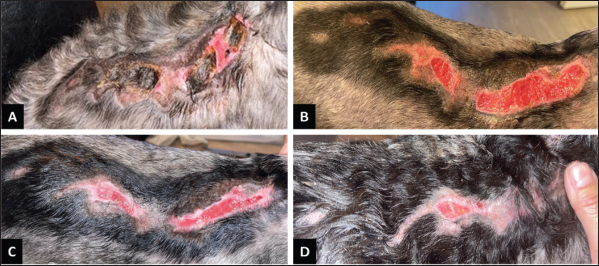
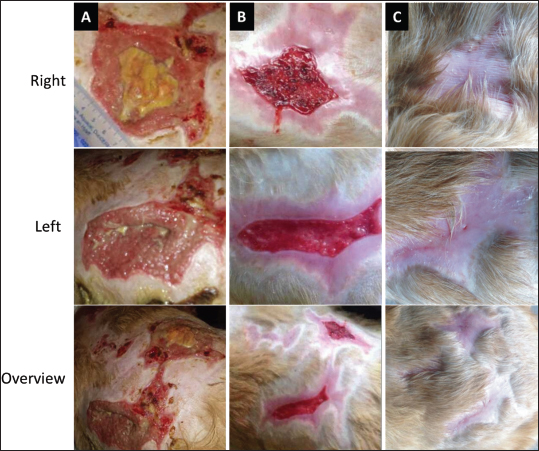
Fig. 3. Wound healing progression of the hock. The top row (A–D) visualizes the wounds on the lateral side of the hock, while the bottom row (E–H) visualizes the point of the hock. A) The wounds at the start of the MGH therapy. B) After 7 days of MGH therapy, debridement became evident. C) On day 20, the wound size significantly decreased by reepithelialization. D) The hock 84 days after the start of the MGH therapy, showing minimal scarring. E) On day 14, the severity of the wound at the point became evident. F) After 29 days of MGH therapy, the wound fully granulated. G) The wound size had decreased significantly after 45 days. H) A follow-up on day 71 showed minimal scarring on the point of the hock. Wound lavage, followed by mechanical and surgical debridement of devitalized tissue, was performed. Daily wound care, involving meticulous dressing changes with the MGH-based wound gel (L-Mesitran Soft), was administered (Fig. 2B). Progressive debridement of non-viable tissue continued until the entire affected zone was clearly delineated. After 10 days of MGH therapy, the patient was discharged from the hospital. Following discharge, bandage changes were performed every 2–3 days. As granulation tissue formed, the treatment strategy was adjusted accordingly: the MGH-based foam dressing (L-Mesitran Foam) was introduced for proximal lesions, while the MGH wound gel continued to be used for the foot pads. This personalized approach optimized healing and addressed the distinct characteristics of the various wound sites. The burns on the foot pads improved rapidly, showing reduced erythema and enhanced debridement (Fig. 2C and D). Complete healing was achieved after 20 days of MGH therapy, without further complications (Fig. 2E). The healing process of the wounds on the hock was longer due to the anatomical location. The burns on the lateral side of the hock were fully debrided after 7 days of MGH therapy (Fig. 3B). By day 10, healthy granulation tissue had formed, and epithelialization was clear at day 20 (Fig. 3C). Full healing was achieved after 71 days of MGH therapy, and a follow-up image 84 days after the start of treatment revealed minimal scarring (Fig. 3D). The severity of the wound at the point of the hock became evident after 14 days (Fig. 3E). Treatment with MGH was continued, and over time the wound showed granulation (day 29, Fig. 3F) and re-epithelialization (day 35), achieving complete healing after 48 days of MGH therapy (Fig. 3G). A follow-up on day 71 revealed minimal scarring of the affected area (Fig. 3H). Case 3: Chemical burn on the neck and cheek of a catA male domestic shorthair cat was found with superficial burn wounds on his left cheek and neck (Fig. 4A). The age of the cat was unknown since it was a stray animal, but it was estimated to be under 5 years old. The burns were most likely chemical in origin. A wound swab was taken, and bacterial culture results were negative. Nonetheless, a prophylactic antibiotic therapy with amoxicillin-clavulanic acid for 10 days was initiated, and the veterinarian also started MGH therapy.
Fig. 4. Wound healing progression of the burn on the left cheek and neck of the patient. A. Start of the MGH therapy (day 0). B. One week after the start of the treatment with MGH, the wound showed healthy granulation tissue. C. On day 14, the wound size decreased significantly on the neck area. The wound on the cheek showed slower improvement due to challenges with keeping the dressing in place. Since the cat was a stray and unaccustomed to handling, sedation was required during each dressing change to minimize stress and facilitate safe wound care. MGH-based wound gel and MGH-based foam dressings were used to treat the wound, and a collar was placed on the cat to prevent self-trauma. Dressings were changed every 3 days under sedation, and the cat was kept in a cage throughout the treatment period. After one week of MGH therapy, the wounds on the cheek and neck showed viable granulation tissue (Fig. 4B). One week later, the wound on the neck was almost fully healed, while the cheek wound showed a slight reduction in size (Fig. 4C). Healing of the cheek progressed significantly slower due to difficulty maintaining dressings in place in that area. Because the cat became stressed and irritated when confined for prolonged periods while wearing the cone and dressings, the team decided, after 2 weeks of treatment, to remove the cone and discontinue dressings to reduce stress. The cat remained in a cage before release for two additional weeks, at which point the wounds were nearly completely healed prior to release. Case 4: Hot oil burn in a Siamese catA young (<5 years) female Siamese cat presented to the clinic with deep partial-thickness burn wounds (Fig. 5A). The burns occurred when the cat jumped into a bucket containing a mixture of hot water and oil. The owners provided first aid by washing the area with shampoo and water. Treatment with MGH was initiated the following day.
Fig. 5. Wound healing progression of the burn on the legs of the cat. A. Three days after the start of MGH therapy. B. Five days after the start of the treatment with MGH, the wound showed initiation of the osmotic and autolytic debridement processes. C, D. Debridement and granulation became evident on day 7 of the MGH therapy (C) and were almost fully completed on day 10 (D). E. The wound showed significant re-epithelialization after 18 days of MGH. F. Full healing was almost achieved on day 54 of the MGH treatment. Upon presentation, the veterinarian administered a subcutaneous cefovecin and prescribed non-steroidal anti-inflammatory drug. Topical therapy with an MGH-based ointment (L-Mesitran Ointment) was commenced. The ointment was applied directly to the wound once daily, and the cat received a cone to prevent self-licking. By day three of the MGH therapy, the ventral-cranial border of the wound showed black necrotic tissue (Fig. 5A). MGH therapy was continued, and over the next 10 days, the wound became fully debrided revealing healthy granulation tissue (Fig. 5B–D). The cat also tolerated treatment better and displayed normal, playful behaviour. Re-epithelialization was evident after 18 days of MGH therapy, and near-complete healing was achieved after 54 days of treatment (Fig. 5E and F). Case 5: Thermal burn on the back of a dogA 4-year-old male Boomer-mix dog sustained a deep partial-thickness burn on his back after falling asleep against a radiator while still drowsy from anesthesia received for a surgical procedure. In the following days, the patient became aggressive toward the owners when touched. One week after the incident, a large burn wound was discovered on the back. The veterinarian commenced treatment with various wound care products, but no improvements were observed after 2 weeks of therapy. Consequently, the owners consulted another veterinarian familiar with MGH products for advice. At the start of MGH therapy, the wound contained necrotic tissue (Fig. 6A), which was partially removed mechanically by the owners. The wound was then cleaned with sterile saline-soaked gauze, and the surrounding fur was clipped. Treatment with MGH-impregnated non-adherent gauze (L-Mesitran Tulle), covered with secondary absorbent dressings, was initiated 3 weeks post-injury. Dressings were changed every 2–3 days by the owners at home. After 4 days of MGH therapy, the product had completely debrided residual necrotic tissue, revealing a healthy granulation bed (Fig. 6B). MGH promoted wound healing, resulting in rapid wound contraction and closure after 39 days of treatment (Fig. 6C and D).
Fig. 6. Thermal burn injury on the back of a canine patient. A) The wound at the start of the MGH treatment, when the wound was present for 3 weeks. B) The wound was fully debrided after 4 days of MGH treatment, and healthy granulation tissue was visible. C) After 15 days of MGH therapy, the wound markedly reduced in size. D) The wound was closed after 39 days of MGH treatment. Case 6: Hot oil burn on a dogA 6-year-old female Bernese Mountain dog presented to the clinic with a large, deep partial-thickness burn wound measuring approximately 30 cm (Fig. 7A). The burn occured when the dog knocked a deep-frying pan from the kitchen counter with her tail, causing the hot oil to spill onto her back. The dog received daily doses of 1.5 mg prednisone per kg body weight to reduce the inflammatory response. Given the well-documented success of supplemented MGH in burn management, treatment with these products was initiated.
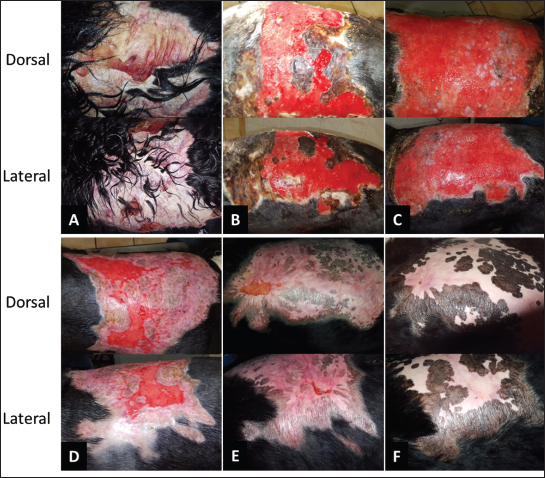
Fig. 7. Progression of the hot oil burn on the back of the patient. A) The wound at the start of the MGH therapy (day 0). B) After 1 week, the owners could start manual debridement of the wound, which was enhanced by the autolytic debridement capacities of MGH. C) A fully debrided wound with granulation tissue was present after 2 weeks of MGH therapy. D) 3 weeks after the start of the MGH therapy, the wound showed significant re-epithelialization. E) The wound was almost fully healed after 7 weeks of MGH treatment, with the exception of two small areas. F) The small wounds were also fully healed 3 months after the start of MGH therapy. The hair surrounding the wound was clipped, and the area was cleansed with 0.45% saline and gently dried with a sterile towel. A supplemented MGH-based ointment was applied daily to the wound bed, covered with MGH-impregnated non-adherent gauze and a secondary absorbent dressing, which was secured with tubular elastic net bandage. One week after starting MGH therapy, necrotic tissue was manually removed, revealing healthy granulation tissue underneath (Fig. 7B). Dressing changes were extended to every 2–3 days, and over the following week, the debridement process continued until the wound was fully debrided two weeks after initiation of therapy (Fig. 7C). Remarkably, 1 week later, the wound showed a marked reduction in size (Fig. 7D). After 7 weeks of treatment, the wound was almost fully re-epithelialized, except for a few small areas (Fig. 7E). On these remaining areas, MGH-based hydrogel plasters (L-Mesitran Active) were applied and left in place for 5 days. Complete wound healing was achieved after 3 months of MGH treatment in total (Fig. 7F). Case 7: Iatrogenic burn in a dogAn 11-year-old female Golden Retriever underwent an ovariohysterectomy for pyometra. Following surgery, the patient received antibiotics and initially recovered without complications. However, the dog later developed panting and vomiting, which were unrelated to the initial condition but affected overall health. The veterinarian prescribed antiemetics against the vomiting and, upon examination, noticed scabs and small wounds on the dog’s back. After trimming the fur, several partial-thickness burn wounds were discovered (Fig. 8A). These were suspected to have been caused by an incorrectly set heating pad during surgery. Approximately 1–2 weeks post-surgery, the owners sought help from another veterinarian experienced in MGH-based therapy.
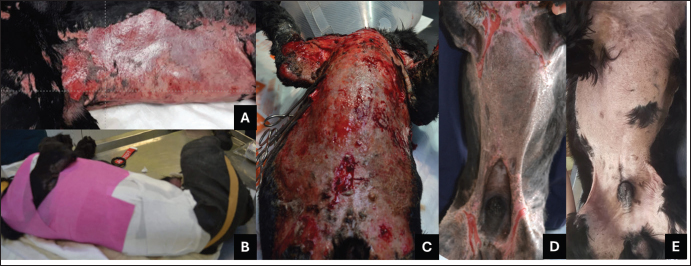
Fig. 8. Multiple iatrogenic burn wounds on the back of a canine patient. The top row shows the wound located on the right hip treated with the MGH-based hydrogel; the middle row shows the wound on the left hip treated with the MGH-impregnated non-adherent gauze; and the bottom row shows the overview of all burns on the dog’s back. A) The wound at the start of the MGH therapy. B) One month later, the wound was fully debrided and had decreased markedly in size. C) After 2 months of MGH treatment, full healing was achieved without further complications. Necrotic tissue was carefully removed with a scalpel, and a supplemented MGH-based ointment was applied, followed by MGH-impregnated non-adherent gauze and a secondary absorbent dressing. Dressings were changed every 1–2 days, dependening on the wound’s exudate level. The wounds were fully debrided of residual slough after 5 days of MGH therapy. After 1 week of treatment, each wound on either hip was treated differently: one side received MGH-impregnated non-adherent gauze, while the other side was treated with an MGH-based hydrogel (L-Mesitran Hydro). Both wounds were comparable in size and severity. After 1 month, the wounds showed marked improvement with a vibrant wound bed and reduced size (Fig. 8B). The dressing changes were reduced to every 3–4 days, allowed by reduced exudate. Full wound healing was achieved after 2 months of MGH therapy (Fig. 8C). According to the veterinarian, the wound treated with the MGH-based hydrogel demonstrated faster improvement than the wound treated with the MGH-impregnated gauze, suggesting the hydrogel might be more suitable for burn wounds. Case 8: Thermal burn on the abdomen of a dogA 1.5-year-old male Cocker Spaniel presented to the clinic with extensive thermal burns of varying depths, primarily affecting the abdominal region (Fig. 9A). The burns were sustained when the dog jumped into a shallow pool of hot water while chasing a cat. The owners provided basic first aid before bringing the dog to the clinic. Clinical examination revealed burns covering approximately 45% of the total body surface area, with deep partial-thickness burns being predominating. Wound care was initiated immediately.
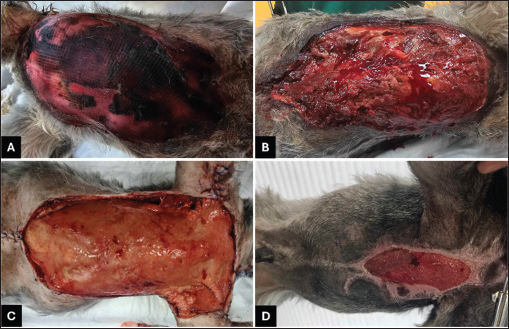
Fig. 9. The progression of the burn wound on the dog. A. The burn wound at the start of the MGH therapy. B. The daily bandaging of the patient. C. Increased signs of viable, red tissue indicating MGH is stimulating wound healing and formation of granulation tissue after 8 days of MGH therapy. D. After 2 months of MGH treatment, the wounds were almost fully closed. E. An 11-month follow-up revealed little to no scarring. The MGH-based ointment was combined with hydrofiber dressings (Aquacell Extra), with daily dressing changes (Fig. 9B). Surgical debridement was performed daily. Moreover, crystalloids and painkillers were administered daily, and blood tests (complete blood count and biochemical profile) were performed regularly, due to some initial deviations (packed cell volume,, renal markers). Furthermore, the dog was given a special diet after calculating his nutritional requirements. After 8 days of MGH therapy, the wound bed showed clear signs of revitalization, and granulation tissue began to form (Fig. 9C). Over the next 2 months, wound healing progressed steadily, with marked re-epithelialization stimulated by MGH (Fig. 9D). The wound was fully healed after 3 months of treatment, and a follow-up 11 months later revealed minimal scarring (Fig. 9E). Case 9: Large abdominal burn in a dogA 5-year-old castrated male Schnauzer presented to the clinic with extensive full-thickness burns affecting the thoracic and abdominal regions (Fig. 10A). The patient came from an animal shelter, and the cause of the burn was unknown. Wound care was initiated immediately upon admission to the clinic.
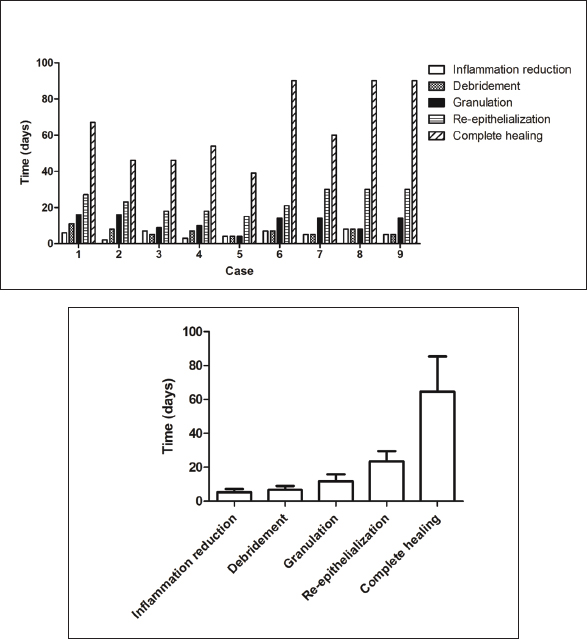
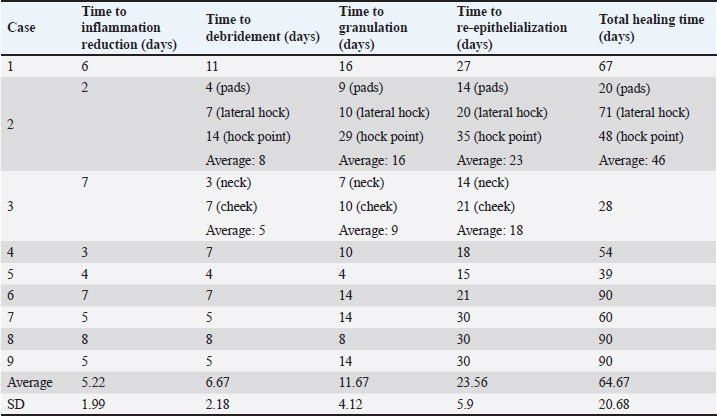
Fig. 10. The progression of the burn wound on the dog. A. The burn wound at the start of the MGH therapy. B. MGH therapy was combined with surgical debridement, which was first performed after 2 days of MGH therapy. C. After 2 weeks of MGH therapy, the wound showed healthy granulation tissue. The edges of the wound were surgically closed on the arms and lower abdomen. D. After 2 months of MGH treatment, the wounds were almost fully closed. One month later, the patient was fully healed (not pictured). At presentation, the wound was largely necrotic. Prophylactic antibiotic therapy was initiated, and treatment with an MGH-based ointment was combined with a hydrofiber dressing. Daily dressing changes were performed by the veterinarian. On the second day of MGH therapy, the wound was surgically debrided (Fig. 10B), and a second debridement was carried out on the fifth day. After 2 weeks, healthy granulation tissue had formed (Fig. 10C). In addition, the edges of the wound on the forlimbs and lower abdomen were closed primarily. The wound was almost fully healed after 2 months of MGH therapy (Fig. 10D). One month later, complete epithelialization was achieved, and the burn wound had healed without complications. Summary of findingsAcross the nine cases, supplemented MGH demonstrated consistent progression through the key phases of wound healing (Table 2, Fig. 11A). On average, any sign(s) of inflammation subsided within 5.2 ± 2.0 days, debridement occurred within 6.7 ± 2.2 days, granulation tissue appeared after 11.7 ± 4.1 days, and re-epithelialization became evident by 23.6 ± 5.9 days. Complete wound closure was achieved in 64.7 ± 20.7 days (Fig. 11B). The outcome of these parameters depends on wound age, dimensions, anatomical location, previous treatment, and patient characteristics. For example, superficial and partial-thickness burns (e.g., chemical and hot oil burns in cats) healed within 3–8 weeks, while extensive full-thickness or abdominal burns (e.g., in dogs) required up to 3 months. Despite this variability, all patients reached full wound healing without major complications, suggesting that vitamin-supplemented MGH can support consistent clinical outcomes across diverse burn etiologies and severities.
Fig. 11. Healing timelines of key clinical parameters in burn wound treatment of companion animals with supplemented MGH. A. Key outcomes (reduction of inflammation, debridement, appearance of granulation tissue, re-epithelialization, and complete wound closure) are presented for each individual case, with each bar representing the timeline in days. Variation among cases reflects differences in wound duration, etiology, size, anatomical location, and individual patient characteristics. B. The average timelines of the five clinical healing parameters across all nine cases are presented as mean ± standard deviation (SD). Table 2. Summary of clinical outcomes across cases.
DiscussionBurn wounds in domestic animals are frequently caused by accidents at home or iatrogenically by heating pads used to keep the animal warm during or after anesthesia (Pavletic and Trout, 2006; Lagutchik and Ford, 2012). Often, the first choice of treatment for burns is silver-based products or antibiotic creams, which have disadvantages compared to MGH-based products (Innes et al., 2001; Kędziora et al., 2018; Khansa et al., 2019). In the current study, the use of supplemented MGH in veterinary patients was showcased in clinical cases. MGH is not only known for its antimicrobial effects but also for its wound healing effects. Its wound healing properties include creating a moist wound environment, which has been shown to result in faster healing than a dry or wet environment (Winter, 1962). This is due to its osmotic action, which withdraws excessive fluids from the wound and surrounding skin while facilitating the replenishment of lost fluids by drawing lymph fluid from the underlying circulation (Molan and Rhodes, 2015). This also results in the stimulation of autolytic debridement, as lymph is rich in matrix metalloproteases that assist in the breakdown of necrotic tissue (Molan, 2002; Scepankova et al., 2021). Moreover, MGH provides nutrition and oxygen to the wound bed and exerts anti-inflammatory and antioxidant effects, thereby aiding the wound healing process (Smaropoulos and Cremers, 2019; Pleeging et al., 2020). Supplemented MGH has been shown to have superior effects compared to non-supplemented MGH in previous studies. For example, the application of MGH supplemented with vitamins C and E, along with PEG 4000, resulted in faster healing of partial-thickness burns compared to using honey alone (Subrahmanyam, 1998). In vitro and ex vivo studies have also underlined synergistic effects of vitamins C and E with MGH (Majtan et al., 2020; Pleeging et al., 2020; Boekema et al., 2023). Raw honey and MGH have been used to treat burns in animals previously (Rooster and Declercq, 2008; Peteoaca et al., 2021). For example, one dog presented to the clinic after sustaining severe chemical burns to all four paws from caustic soda-based toilet cleanser (Rooster and Declercq, 2008). The injuries were so severe that tendons and bones were exposed, and necrosis was present. However, treatment with MGH resulted in full healing within 5 weeks. The advantage of supplemented MGH is that it contains a lower percentage of MGH, thereby allowing easier application and less sensitivity to temperature changes. Additionally, its supplements contribute to the antimicrobial function of the MGH as the ingredients work in synergy (Majtan et al., 2020; Pleeging et al., 2020). MGH products supplemented with vitamins C and E have been applied in several cats and dogs with burn injuries of various etiologies. For example, a dog presented to the clinic with full- and partial-thickness thermal burns covering 45% of its body (Maravelis et al., 2015). The patient was treated with supplemented MGH-based wound gel, which resulted in full healing within 35 days. Another case report involved a domestic shorthair cat with a large, necrotic thermal burn (Kokkinos et al., 2017). The wound bed was prepared for flap surgery by surgical debridement of the necrotic tissue and daily application of the supplemented MGH-based wound gel. Only 1 week after the start of the treatment, healthy granulation tissue had appeared, and reconstructive surgery could be performed. A case series by Lukanc and Erjavec (2022) included two domestic cats and one dog with burn injuries (Lukanc and Erjavec, 2022). Both feline patients had infected burn wounds, one of chemical origin and the other thermal. The canine patient sustained an iatrogenic thermal burn during surgery. All patients were treated with supplemented MGH products, which resulted in full healing after 1–3.5 months, depending on the severity of the wounds. The cases presented in the current study showed full healing in a range of 2 weeks to 3 months, depending on the severity and location of the wound. For example, in case number 2, despite the more severe lesions in the left hind limb, the localization of the wound over the right hock contributed to a similar timeframe for the healing process. This observation underscores the importance of considering both the severity of injuries and their specific anatomical locations in predicting and managing the healing trajectory. Another factor that significantly impacts the healing trajectory is the temperature of the burn. Burn injuries in animals vary greatly according to the temperature of the causative substance. High-temperature burns are extremely painful and destructive, frequently resulting in significant tissue damage that requires prompt surgical intervention, such as early excision and wound closure, to shorten recovery time and improve survival rates in severe cases (Gray et al., 1982; Choi et al., 2015; Puri et al., 2016). Low-temperature burns, on the other hand, initially seem less severe and cause less pain, which delays diagnosis and treatment (Choi et al., 2015). Despite their apparent mildness, low-temperature burns can cause extensive tissue damage, including second- or third-degree lesions, and often become fully apparent only after 1–2 weeks with eschar formation. The healing process may take longer as a result of these delays in manifestation and treatment (Papp et al., 2004; Choi et al., 2015). The use of certain treatments during the crucial interval between eschar development and escharotomy can be controversial, since inappropriate management may delay healing or complicate wound care. As a result, both low- and high-temperature burns require swift and specialized care to minimize wound deterioration and optimize recovery outcomes. Additionally, almost all cases presented in the current study were fully healed by secondary intention, driven by owner requests, veterinary choices, financial constraints, or other reasons. It could be argued that total healing time could be reduced by combining MGH therapy with reconstructive surgery. Healthy granulation tissue appeared in a range of 4–30 days, also dependent on the location and severity of the wound. The ability to perform reconstructive surgery quickly, facilitated by good granulation tissue, is a significant advantage. This was partially demonstrated in case 10, in which the edges of the wound bed were closed primarily after just 2 weeks, thereby significantly speeding up recovery. Moderate-quality scars were obtained due to the extent of epithelialization in most of the presented cases, which could be considered a minor drawback. A key limitation of this retrospective study is the absence of standardized wound area measurements. Although photographic documentation was available for all cases, images were not consistently captured with calibration markers or modern automated applications, limiting reliable retrospective quantification of wound size reduction using image analysis tools such as ImageJ. Similarly, validated semi-quantitative wound scoring systems were not employed across clinics, which limited our ability to provide objective metrics beyond descriptive statistics of healing time. Future prospective studies should incorporate standardized photographic protocols, calibrated wound measurement techniques, and validated scoring systems to enable robust statistical evaluation and facilitate comparisons across studies. Finally, although the study’s focus is on local wound management, it is important to recognize the broader patient management methods that contribute to successful burn treatment outcomes. Ensuring proper nutritional management and performing regular blood tests to monitor for problems such as anemia or hypoalbuminemia are critical components of comprehensive care. For example, one case report described the importance of nutritional support in a kitten with a thermal burn wound (Birkbeck et al., 2020). The use of a natural product in our study resulted in overall positive responses from the owners. Additionally, MGH-based dressings have an extended wear time, which allows patients to come to the clinic less frequently. This also contributed to overall owner satisfaction. ConclusionThis multi-center case series highlights the potential of vitamins C- and E-supplemented MGH as an effective alternative treatment for the management of burn wounds in companion animals. The observed cases demonstrated favorable wound healing trajectories, characterized by effective debridement, rapid granulation tissue formation, and successful re-epithelialization. MGH appeared to support a moist, antimicrobial wound environment, even in complex or previously non-healing burns. The absence of any adverse effects and the ease of application further underscore the practical value of MGH in veterinary wound care. While the outcomes are promising, the retrospective and descriptive nature, and lack of a control group of this study limit the ability to draw definitive conclusions. Prospective controlled studies are recommended to further validate these findings and establish standardized protocols for the use of MGH in veterinary burn wound management. AcknowledgmentsWe thank the owners of the animals for their trust in our treatments and for providing their informed consent. Conflict of interestL.J.F.P. and N.A.J.C. are employed by Triticum Exploitatie BV, Maastricht, the Netherlands. However, they were not involved in the design, treatment, and presentation of the results. All other authors declare no conflict of interest. FundingThis research received no external funding. Authors’ contributionsConceptualization: A.P., C.C.F.P., L.J.F.P., and N.A.J.C.; formal analysis: A.P., C.C.F.P., L.J.F.P., and N.A.J.C.; investigation: all authors; methodology: all authors; resources: A.P., S.A.S., V.T., A.C.J.M.P., and C.C.F.P.; visualization: A.P., C.C.F.P., and L.J.F.P.; writing original draft: A.P., C.C.F.P., L.J.F.P., and N.A.J.C; writing—review and editing: all authors; supervision: N.A.J.C.; project administration: A.P., C.C.F.P., L.J.F.P., and N.A.J.C. All authors have read and agreed to the published version of the manuscript. Data availabilityThe data supporting this study’s findings are available from the corresponding author upon reasonable request. All data relevant to the study are included in the article. ReferencesAlmasaudi, S. 2021. The antibacterial activities of honey. Saudi. J. Biol. Sci. 28, 2188–2196. Andrei Zbuchea. 2017. Honey, Food and Medicine: scientific Rationale and Practical Efficiency in External Administration of Medicinal Honey for Wound Healing. J. Agricult. Sci. Technol. B. 7, 206–219. Birkbeck, R., Donaldson, R. and Chan, D.L. 2020. Nutritional management of a kitten with thermal burns and septicaemia. JFMS. Open. Rep. 6, 2055116920930486. Boekema, B.K.H.L., Chrysostomou, D., Ciprandi, G., Elgersma, A., Vlig, M., Pokorná, A., Peters, L.J.F. and Cremers, N.A.J. 2023. Comparing the antibacterial and healing properties of medical-grade honey and silver-based wound care products in burns. Burns. Boekema, B.K.H.L., Pool, L. and Ulrich, M.M.W. 2013. The effect of a honey based gel and silver sulphadiazine on bacterial infections of in vitro burn wounds. Burns 39, 754–759. Choi, M.S.S., Lee, H.J. and Lee, J.H. 2015. Early intervention for low-temperature burns: comparison between Early and Late Hospital Visit Patients. Arch. Plast. Surg. 42, 173–178. De Rooster, H. and Declercq, J. 2008. Honing in de wondzorg: mythe of wetenschap? Deel 2: klinische gevallen bij de hond. Vlaams. Diergeneeskundig. Tijdschrift. 78, 75–80. Gray, D.T., Pine, R.W., Harnar, T.J., Marvin, J.A., Engrav, L.H. and Heimbach, D.M. 1982. Early surgical excision versus conventional therapy in patients with 20 to 40 percent burns. A comparative study. Am. J. Surg. 144, 76–80. Hermanns, R., Mateescu, C., Thrasyvoulou, A., Tananaki, C., Wagener, F.A.D.T.G. and Cremers, N.A.J. 2020. Defining the standards for medical grade honey. J. Apicultural Res. 59, 125–135. Innes, M.E., Umraw, N., Fish, J.S., Gomez, M. and Cartotto, R.C. 2001. The use of silver coated dressings on donor site wounds: a prospective, controlled matched pair study. Burns 27, 621–627. Jeschke, M.G., Van Baar, M.E., Choudhry, M.A., Chung, K.K., Gibran, N.S. and Logsetty, S. 2020. Burn injury. Nat. Rev. Dis. Primers 6, 11. Kędziora, A., Speruda, M., Krzyżewska, E., Rybka, J., Łukowiak, A. and Bugla-Płoskońska, G. 2018. Similarities and differences between Silver Ions and Silver in Nanoforms as Antibacterial Agents. Int. J. Mol. Sci. 19, 444; doi:10.3390/ijms19020444. Khansa, I., Schoenbrunner, A.R., Kraft, C.T. and Janis, J.E. 2019. Silver in Wound Care-Friend or Foe?: a comprehensive review. Plast. Reconstr. Surg. Glob. Open 7, 2390. Kokkinos, P., Kouki, M., Montzolis, G., Savvas, I., Delligiani, A. and Papazoglou, L.G. 2017. Lateral caudal axial pattern flap after tail amputation for coverage of a dorsal pelvic and perineal skin defect in a cat. Austral. Vet. Practitioner. 47, 25–28. Kwakman, P.H.S. and Zaat, S.A.J. 2012. Antibacterial components of honey. IUBMB. Life 64, 48–55. Lagutchik, M.S. and Ford, A. 2012. Care of the Environmentally Injured Animal. Advanced Monitoring and Procedures for Small Animal Emergency and Critical Care. Lukanc, B. and Erjavec, V. Treating burns in cats and dogs Using Medical Honey. In Proceedings of 7th Socratic Lectures, Ljubljana, Slovenia, University of Ljubljana, Faculty of Health Sciences, 2022, 7, pp 77–81. Majtan, J., Sojka, M., Palenikova, H., Bucekova, M. and Majtan, V. 2020. Vitamin C Enhances the antibacterial activity of Honey against Planktonic and Biofilm-Embedded Bacteria. Molecules 25, 1–13. Mandal, M.D. and Mandal, S. 2011. Honey: its medicinal property and antibacterial activity. Asian. Pac. J. Trop. Biomed. 1, 154–160; doi: 10.1016/S2221-1691(11)60016-6. Maravelis, G., Voutsinou, A., Tsampa, N. and Papazoglou, L. 2015. Management of an extensive partial and full thickness skin burn in a dog with the aid of medical honey. Hellenic. J. Companion. Anim. Med. 4, 37. Molan, P. and Rhodes, T. 2015. Honey: a Biologic Wound Dressing. Wounds 27, 141–151. Molan, P.C. 2002. Re-introducing honey in the management of wounds and ulcers - theory and practice. Ostomy. Wound. Manage. 48, 28–40. Organisation, W.H. 2018. Burns - Fact Sheet [Online]. Available via https://www.who.int/news-room/fact-sheets/detail/burns [Accessed]. Papp, A., Kiraly, K., Härmä, M., Lahtinen, T., Uusaro, A. and Alhava, E. 2004. The progression of burn depth in experimental burns: a histological and methodological study. Burns 30, 684–690. Pavletic, M.M. and Trout, N.J. 2006. Bullet, bite, and burn wounds in dogs and cats. Vet. Clin. North. Am. Small. Anim. Pract. 36, 873–893. Peteoaca A., Ionascu, I., Tanase A. 2021. Iatrogenic thermal injury management in a geriatric dog - a case report. Scientific Works. Series C, Veterinary Medicine., LXVII, 102–112. Peters, L.J., Majtan, J., Mossialos, D., Szweda, P., Mateescu, C., Ozturk, F., Wagener, F.A. and Cremers, N.A. 2025. Medical-grade honey: its definition and refined standards. J. Wound. Care. 34(2), 2–12. Pleeging, C.C.F., Coenye, T., Mossialos, D., De Rooster, H., Chrysostomou, D., Wagener, F. and Cremers, N. a. J. 2020. Synergistic Antimicrobial Activity of Supplemented Medical-Grade Honey against Pseudomonas aeruginosa Biofilm Formation and Eradication. Antibiotics (Basel). 9, 1–16. Puri, V., Khare, N.A., Chandramouli, M.V., Shende, N. and Bharadwaj, S. 2016. Comparative Analysis of Early Excision and Grafting vs Delayed Grafting in Burn Patients in a Developing Country. J. Burn. Care. Res. 37, 278–282. Rooster, H. and Declercq, J. 2008. Honey in wound management: myth or science? Part 2: Clinical cases of dogs. Vlaams. Diergeneeskundig. Tijdschrift. 77, 75–80. Scepankova, H., Combarros-Fuertes, P., Fresno, J.M., Tornadijo, M.E., Dias, M.S., Pinto, C.A., Saraiva, J.A. and Estevinho, L.M. 2021. Role of Honey in Advanced Wound Care. Molecules 26, 1–19. Smaropoulos, E. and Cremers, N.A.J. 2019. The pro-healing effects of medical grade honey supported by a pediatric case series. Complement. Ther. Med. 45, 14–18. Smaropoulos, E. and Cremers, N.A.J. 2020. Treating severe wounds in pediatrics with medical grade honey: a case series. Clin. Case. Rep. 8, 469–476. Subrahmanyam, M. 1998. A prospective randomised clinical and histological study of superficial burn wound healing with honey and silver sulfadiazine. Burns 24, 157–161. Vaughn, L. and Beckel, N. 2012. Severe burn injury, burn shock, and smoke inhalation injury in small animals. Part 1: burn classification and pathophysiology. J. Vet. Emerg. Crit. Care. (San. Antonio). 22, 179–186. Vogt, N.A., Vriezen, E., Nwosu, A. and Sargeant, J.M. 2020. A Scoping Review of the Evidence for the Medicinal Use of Natural Honey in Animals. Front. Vet. Sci. 7, 618301. Volk, S.W. and Bohling, M.W. 2013. Comparative wound healing—Are the small animal veterinarian’s clinical patients an improved translational model for human wound healing research?. Wound. Repair. Regeneration. 21, 372–381. Winter, G.D. 1962. Formation of the scab and the rate of epithelization of superficial wounds in the skin of the young domestic pig. Nature 193, 293–294. Woo, K.Y. and Sibbald, R.G. 2009. A cross-sectional validation study of using NERDS and STONEES to assess bacterial burden. Ostomy. Wound. Manage. 55, 40–48. Żwierełło, W., Piorun, K., Skórka-Majewicz, M., Maruszewska, A., Antoniewski, J. and Gutowska, I. 2023. Burns: classification, Pathophysiology, and Treatment: A Review. Int. J. Mol. Sci. 24. | ||
| How to Cite this Article |
| Pubmed Style Peteoacă A, Symeonidis SA, Tachmatzidou V, , Pleeging CCF, Peters LJF, Cremers NAJ. Vitamin-supplemented medical-grade honey for burn wound management in cats and dogs: a retrospective case series. Open Vet. J.. 2026; 16(1): 653-668. doi:10.5455/OVJ.2026.v16.i1.61 Web Style Peteoacă A, Symeonidis SA, Tachmatzidou V, , Pleeging CCF, Peters LJF, Cremers NAJ. Vitamin-supplemented medical-grade honey for burn wound management in cats and dogs: a retrospective case series. https://www.openveterinaryjournal.com/?mno=271656 [Access: February 04, 2026]. doi:10.5455/OVJ.2026.v16.i1.61 AMA (American Medical Association) Style Peteoacă A, Symeonidis SA, Tachmatzidou V, , Pleeging CCF, Peters LJF, Cremers NAJ. Vitamin-supplemented medical-grade honey for burn wound management in cats and dogs: a retrospective case series. Open Vet. J.. 2026; 16(1): 653-668. doi:10.5455/OVJ.2026.v16.i1.61 Vancouver/ICMJE Style Peteoacă A, Symeonidis SA, Tachmatzidou V, , Pleeging CCF, Peters LJF, Cremers NAJ. Vitamin-supplemented medical-grade honey for burn wound management in cats and dogs: a retrospective case series. Open Vet. J.. (2026), [cited February 04, 2026]; 16(1): 653-668. doi:10.5455/OVJ.2026.v16.i1.61 Harvard Style Peteoacă, A., Symeonidis, . S. A., Tachmatzidou, . V., , Pleeging, . C. C. F., Peters, . L. J. F. & Cremers, . N. A. J. (2026) Vitamin-supplemented medical-grade honey for burn wound management in cats and dogs: a retrospective case series. Open Vet. J., 16 (1), 653-668. doi:10.5455/OVJ.2026.v16.i1.61 Turabian Style Peteoacă, Alexandra, Stathis A. Symeonidis, Vasiliki Tachmatzidou, Noëlle A. C. J. M. Paping, Carlos C. F. Pleeging, Linsey J. F. Peters, and Niels A. J. Cremers. 2026. Vitamin-supplemented medical-grade honey for burn wound management in cats and dogs: a retrospective case series. Open Veterinary Journal, 16 (1), 653-668. doi:10.5455/OVJ.2026.v16.i1.61 Chicago Style Peteoacă, Alexandra, Stathis A. Symeonidis, Vasiliki Tachmatzidou, Noëlle A. C. J. M. Paping, Carlos C. F. Pleeging, Linsey J. F. Peters, and Niels A. J. Cremers. "Vitamin-supplemented medical-grade honey for burn wound management in cats and dogs: a retrospective case series." Open Veterinary Journal 16 (2026), 653-668. doi:10.5455/OVJ.2026.v16.i1.61 MLA (The Modern Language Association) Style Peteoacă, Alexandra, Stathis A. Symeonidis, Vasiliki Tachmatzidou, Noëlle A. C. J. M. Paping, Carlos C. F. Pleeging, Linsey J. F. Peters, and Niels A. J. Cremers. "Vitamin-supplemented medical-grade honey for burn wound management in cats and dogs: a retrospective case series." Open Veterinary Journal 16.1 (2026), 653-668. Print. doi:10.5455/OVJ.2026.v16.i1.61 APA (American Psychological Association) Style Peteoacă, A., Symeonidis, . S. A., Tachmatzidou, . V., , Pleeging, . C. C. F., Peters, . L. J. F. & Cremers, . N. A. J. (2026) Vitamin-supplemented medical-grade honey for burn wound management in cats and dogs: a retrospective case series. Open Veterinary Journal, 16 (1), 653-668. doi:10.5455/OVJ.2026.v16.i1.61 |