| Research Article | ||
Open Vet. J.. 2025; 15(10): 5116-5127 Open Veterinary Journal, (2025), Vol. 15(10): 5116-5127 Research Article Protective effect of epigallocatechin-gallate in cryopreserved goat sperm on post-thawed qualityYudit Oktanella1*, Ayunda A. Cahyani1, Juhi S. Caula1, Sabrina N. Fathiyananda1, Fahrunnisak A. R. An-Haru1, Imam Mustofa2, Suherni Susilowati2, Nurhusien Y. Degu3, Tatik Hernawati2 and Yayuk Kholifah41Department of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia 2Department of Veterinary Reproduction, Faculty of Veterinary Medicine, Airlangga University, Surabaya, Indonesia 3Department of Veterinary Sciences, School of Medicine, IMU University, Kuala Lumpur, Malaysia 4Singosari Center for Artificial Insemination (BBIB), Malang, Indonesia *Corresponding Author: Yudit Oktanella. Department of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia. Email: yudito [at] ub.ac.id Submitted: 21/07/2025 Revised: 10/09/2025 Accepted: 18/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
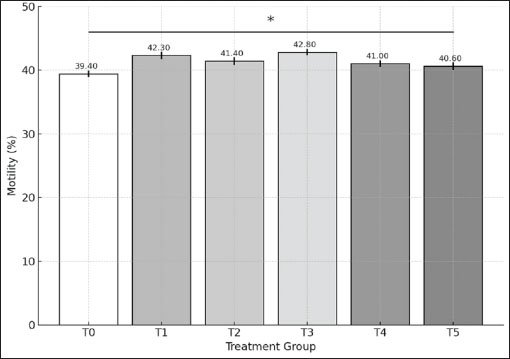
AbstractBackground: Cryopreservation is a useful technique for preserving goat sperm, but it often causes damage due to oxidative stress triggered by increased reactive oxygen species. One promising solution is the use of antioxidants, such as epigallocatechin gallate (EGCG), to reduce these harmful effects. EGCG has been recognised for its ability to safeguard sperm cells by supporting the normal function of mitochondria, regulating cell death mechanisms, and supporting genes involved in energy metabolism. Aim: This study aimed to evaluate the effects of different concentrations of EGCG added to semen extenders on the quality of goat sperm after thawing, including motility, viability, and the expression of genes related to mitochondrial activity and apoptosis. Methods: Several concentrations of EGCG (ranging from 50–800 mg/dl) were tested. Post-thaw sperm motility and viability were analysed using Open CASA®. B-cell lymphoma-2 (BCL-2) expression was observed using immunocytochemistry, while CPT2 gene expression was assessed by Reverse Transcription Quantitative Polymerase Chain Reaction. Statistical analyses were conducted using Duncan’s and Kruskal-Wallis tests, followed by pairwise comparisons. Results: The findings showed that 200 mg/dl of EGCG significantly improved sperm motility, while the 100 mg/dl dose helped maintain better viability after thawing. Interestingly, BCL-2 expression increased most notably at 500 mg/dl, indicating anti-apoptotic protection. Additionally, CPT2 gene expression was significantly upregulated at EGCG concentrations of 50, 100, 400, and 800 mg/dl compared to the control group. Conclusion: EGCG supplementation in goat semen extenders has a protective effect on post-thawed sperm by improving motility and viability and supporting mitochondrial and apoptotic pathways. These results suggest that EGCG can be a valuable additive for enhancing artificial insemination outcomes in goats. Keywords: Cryopreservation, EGCG, Food security, Gene expressions, Sperm quality. IntroductionIndonesia’s national program, “Makan Bergizi Gratis” (Free Nutritious Meals), was officially launched in 2025 and represents a strategic effort to address long-standing issues of child malnutrition. It aims to provide daily nutritious meals to nearly 90 million children and pregnant women in response to persistent challenges such as stunting, which continues to affect 21.5% of children under the age of five (Lestari et al., 2024). In this context, goats serve as a particularly significant in smallholder livestock systems, offering high-quality animal protein through both meat and milk. Consequently, upgrading the yield capacity and genetic attributes of the national goat population plays a crucial role in supporting sustainable practices of the national program. Furthermore, by enhancing reproductive efficiency in goats, a significant role in supporting sustainable livestock production systems may be realised. Accordingly, this corresponds to the United Nations Sustainable Development Goal 2: Zero Hunger, which calls for the promotion of resilient agricultural practices and the availability of safe, nutritious food for global need (United Nations, 2015). A major limitation of using cryopreserved semen for artificial insemination in goats is the reduced sperm quality observed after thawing (Kusumawati et al., 2019). This deterioration is predominantly linked to temperature-induced damage occurring throughout the cryopreservation process — an unavoidable outcome that disrupts sperm function and results in reduced motility, viability, and potential DNA damage (Rokana et al., 2022; Oktanella et al., 2024). Several underlying elements influence this decrease, including semen collection methods, extender quality, dilution techniques, freezing procedures, and thawing conditions (Kusumawati et al., 2019). During cryopreservation, spermatozoa undergo structural and functional impairments. Exposure to extreme cold may lead to damage or instability of the cell membrane, reduced viability and motility, DNA fragmentation, and even apoptosis in up to 40% of spermatozoa (Samik et al., 2014). Apoptosis, or programmed cell death, is initiated in response to diverse intracellular cues (Asadi et al., 2021). Key proteins regulate this process: pro-apoptotic proteins (e.g., BAD, BID, BIK, BIM, and BAX) trigger apoptotic pathways, while anti-apoptotic proteins [e.g., B-cell lymphoma-2 (BCL-2), BCL-XL, and BCLW] contribute to its prevention (Asadi et al., 2021). Among these, BCL-2 serves an important function in sustaining cell viability by apoptosis inhibition. BCL-2, a membrane-associated protein of the mitochondria, suppresses caspase activation by maintaining mitochondrial integrity (Qian et al., 2022). In sperm cells, a proper balance in the expression of pro- and anti-apoptotic proteins is fundamentally required for cellular stability and survival for maintaining viability and motility post-thawing (Marleen et al., 2008). A significant concern is sperm membrane integrity. Goat spermatozoa possess a thicker lipid bilayer, particularly rich in polyunsaturated fatty acids (PUFAs), making them highly susceptible to damage (Mustofa et al., 2021 b). Structural damage to the sperm membrane impairs motility and lowers fertilization success after artificial insemination (Umami et al., 2015). Therefore, maintaining sperm energy production and membrane stability is critical for post-thaw survival. One potential strategy to reduce cryopreservation-induced sperm damage is the use of Epigallocatechin gallate (EGCG), the main bioactive compound in green tea extract. For instance, a study using Kacang goat (Capra hircus) semen showed that supplementation with 0.1 mg of green tea extract per 100 ml of extender resulted in the highest post-thaw semen quality (Mustofa et al., 2021 a). Likewise, Balasubramanian and Palanisamy (2014) reported that 0.4 mg/ml EGCG improved sperm motility, viability, and membrane integrity in Murrah buffalo bulls. These findings support the antioxidant role of EGCG in mitigating oxidative stress by scavenging reactive oxygen species (ROS), thereby preserving sperm DNA and cellular function during cryopreservation. Before evaluating the quality of thawed spermatozoa, it is also crucial to assess their DNA integrity and molecular characteristics, as the cryopreservation process triggers cellular stress that may lead to DNA fragmentation and alterations in gene expression. In goat spermatozoa, oxidative stress significantly impairs fertilisation potential and embryonic development (Peris-Frau et al., 2020). One of the key genes involved in regulating sperm function is CPT2, which encodes Carnitine O-palmitoyltransferase 2—an essential mitochondrial enzyme that facilitates the beta-oxidation of long-chain fatty acids. This metabolic process requires fatty acids to bind with carnitine before entering the mitochondria, a step mediated by CPT2 (Wieser et al., 2014). Increased CPT2 gene activity is indicative of augmented energy metabolism processes, thereby enhancing sperm motility and viability. However, excessive lipid metabolism can increase ROS production, resulting in oxidative damage to membranes and DNA (Gürler et al., 2016). Conversely, low CPT2 expression limits Adenosine Triphosphate (ATP) synthesis, ultimately compromising post-thaw sperm motility and function (Susilowati et al., 2021). Assessing DNA damage and mitochondrial membrane integrity provides reliable insight into post-thaw sperm quality. Sperm that maintain stable DNA and healthy mitochondria are more likely to show good motility and viability, highlighting why molecular-level evaluations are so important in cryopreservation (Amor et al., 2018). In addition, the regulation of key genes, such as BCL-2 for apoptosis and CPT2 for energy metabolism, further supports sperm survival after thawing (Ranjan et al., 2024). In this study, we propose that pure EGCG may help protect goat sperm from oxidative stress during cryopreservation by supporting cell survival and energy balance. While EGCG is already recognized for its antioxidant properties, its role in regulating key genes—such as BCL-2 (involved in apoptosis) and CPT2 (linked to energy metabolism)—has not yet been explored in goats. To address this gap, we set out to test different doses of EGCG and evaluate their impact on post-thaw sperm quality, focusing on motility, viability, and the expression of BCL-2 and CPT2 at both cellular and molecular levels. Materials and MethodsMethodology and experimental proceduresThis study used semen samples from a healthy 2-year-old Kacang goat, a native Indonesian breed valued for its adaptability and resilience in tropical climates. Before collection, the animal underwent routine physical and health examinations to ensure it met the required standards. Semen was collected from two ejaculates obtained at the same time, with the fresh samples showing a total volume of 1.6 ml and a concentration of 4.8 billion spermatozoa per cc. The ejaculates were then processed into frozen semen, with each straw filled with 0.25 ml containing at least 50 million spermatozoa. The production of frozen semen at the Teaching Farm, Airlangga University, follows the Regulation of the Minister of Agriculture of the Republic of Indonesia No. 10/Permentan/PK.210/3/2016, which sets the requirement for maintaining a stable temperature within the semen processing facility of 18°C–22°C with a humidity level not exceeding 55%. The quality of semen was meticulously evaluated through both macroscopic and microscopic examinations. Fresh semen samples were required to have a pH of 6.4–7, a thick consistency typical caprine odour, and a milky white or cream colour. Microscopically, only samples with at least 80% progressive motility, a mass movement score of two, and a maximum sperm abnormality rate of 5% were used. In order to assess the potential protective role of EGCG on frozen-thawed goat spermatozoa, various concentrations of EGCG were incorporated into the semen diluent. The selection of EGCG dosages was supported by earlier investigations highlighting its potential in maintaining sperm quality through optimisation of motility, survival rate, and membrane structural integrity while reducing oxidative stress (Balasubramanian and Palanisamy, 2014; Mustofa et al., 2021 a). Six different treatment groups were arranged as part of the experimental design, with semen samples supplemented with EGCG at concentrations of 0 mg/dl (T0) as control, 50 (T1), 100 (T2), 200 (T3), 400 (T4), and 800 (T5) mg/dl. Such dose variations provided an in-depth analysis of how EGCG affects sperm performance after cryopreservation and molecular responses. Preparation of semen extenderExtender ATo formulate 100 ml of Extender A, the following components are incorporated: 10% skim milk powder (10 g; Merck 115338), 0.75% fructose (Merck 105323) as an accessible energy substrate, 5% egg yolk functioning as an extracellular cryoprotective agent, 0.1% penicillin (equivalent to 100,000 IU; Meiji Seika Pharma, Tokyo, Japan), 0.1 g streptomycin (Thermo Fisher Scientific, Singapore), and sterile distilled water to complete the volume. Fresh Extender A should be prepared on the same day as semen collection. Extender BExtender B was prepared by enriching Extender A with 2% glucose (2 g/100 ml; Merck 105323), 12% glycerol (Merck 104094), and EGCG at different concentrations [0 (control/T0), 50 (T1), 100 (T2), 200 (T3), 400 (T4), and 800 (T5) mg/dL]. After glucose and glycerol were mixed into Extender A and homogenised, EGCG was added to produce the treatment extenders. Semen samples were then diluted with Extender B during the equilibrium stage, allowing spermatozoa to receive the protective effect of EGCG before freezing. Semen collectionThe semen collection procedure was carried out using an artificial vagina on healthy Kacang bucks. In order to enhance sexual arousal, a teaser male was employed to facilitate the semen collection procedure. Following ejaculation, the semen samples were immediately evaluated for volume, colour, odour, and consistency, with all findings recorded as part of the macroscopic semen analysis. Semen samples exhibiting a minimum of 80% sperm motility were considered eligible for subsequent procedures. The samples are then loaded into straws, gradually cooled, and finally plunged into liquid nitrogen for long-term storage in −196°C (Bodu et al., 2025). The thawing process was carried out by immersing the frozen semen straw in water at 37°C for 20 seconds. Post-thawing motility and viability assessment of goat spermatozoa Post-thaw sperm motility was evaluated across six different treatment groups employing a Computer-Assisted Sperm Analysis (CASA) system (Open CASA®; WI-LI New Century Technical Development) available at the Teaching Farm for sperm’s progressive motility. The CASA-based motility analysis required a minimum of 20 µl of diluted semen, and the video recordings were captured in Audio video interleave format with a 60-fps frame rate, with each sample recorded over a duration of 1 second. A 60-fps frame rate offered the most reliable outcomes for evaluating motility (Ratnawati et al., 2019). Additionally, specific CASA settings were adjusted for goat semen analysis (Xu et al., 2022). Staining-based techniques were utilised to evaluate the viability and sperm morphology based on shape and colour retention. A 10 µl drop of post-thawing semen was placed on a microscope slide and mixed with an equal volume of eosin 1% – nigrosine 10% solution (1:1 ratio) (Merck, Germany). On a separate slide, the suspension was gently smeared and allowed to air-dry for approximately 1–2 minutes. Subsequently, the stained preparations were observed under a light microscope at 1000× magnification. Spermatozoa were considered viable if they resisted eosin-nigrosine staining, reflecting membrane integrity and the absence of dye penetration. In contrast, non-viable spermatozoa absorbed the stain, showing colouration in the head and neck regions. For each experimental unit, at least 200 spermatozoa were counted to determine the percentage of viable cells. Immunocytochemistry preparationThe immunocytochemical procedure was initiated by fixing the cells using chilled methanol (Merck, Germany) (20°C) for 3–5 minutes, followed by drying over a Bunsen burner. The slides were then washed with phosphate-buffered saline (PBS) for 5 minutes and immersed in hydrogen peroxide (Merck, Germany) at room temperature for 10 minutes to block endogenous peroxidase activity, followed by another PBS wash for 5 minutes. A primary antibody (100 µl) (Abcam®) was applied to each slide, ensuring complete coverage, and incubated at 4°C for 24 hours. After incubation, the slides were washed again with PBS for 5 minutes. A biotinylated universal secondary antibody (100 µL) (Cell Signaling Technology®No.Cat.7074) was then added and incubated at 25°C for 10 minutes, followed by another PBS wash for 5 minutes. Next, Streptavidin-Horseradish Peroxidase (Abcam®) reagent was dropped onto the slides and left to stand for 10 minutes in a refrigerator, then washed again with PBS for 5 minutes. The slides were incubated with 3,3-diaminobenzidine tetrahydrochloride chromogen (100 µl) (Abcam®) for 2–10 minutes, followed by another PBS wash for 5 minutes. For counterstaining, 100 µl of Mayer's hematoxylin (Sigma-Aldrich®) was applied and allowed to react at ambient temperature for 1–3 minutes, then rinsed thoroughly with PBS for 5 minutes to remove excess reagent (Burry, 2011). Subsequently, the slides underwent alcohol dehydration, followed by clearing and mounting procedures in preparation for microscopic observation. The immunocytochemistry slides were observed using a binocular light microscope (Olympus® CX-21; 1000x) connected to a laptop running Optilab Viewer 3.0 software. BCL-2 expression was quantified as the percentage of positively stained spermatozoa (brown colour) regardless of staining intensity or location. For each experimental unit, at least 200 spermatozoa were evaluated across 10 randomly selected microscopic fields to ensure representative sampling for statistical analysis. Image analysis was performed using ImageJ software with the ImmunoRatio plugin. BCL-2 expression was identified by the presence of brown staining in the mid-piece and head regions of the spermatozoa (Kalashnyk-Vakulenko et al., 2022). Detection of CPT2 gene expressionRNA isolationTotal RNA was extracted from thawed frozen goat semen samples using the Zymo Research® Direct-zol™ RNA Extraction Kit protocol. The extraction started by adding 100 µl of DNA/RNA Shield™ into a polymerase chain reaction (PCR) microtube at room temperature (20°C–30°C). The frozen semen was thawed in a 37°C incubator for 10 seconds, then about 100–115 µl (half a straw) was transferred into the tube. The mixture was vortexed to homogenize, and the supernatant was then removed. Next, 100 µl of RNA Lysis Buffer was added to the sediment at the bottom of the microtube, followed by centrifugation at 15,000 rpm at 4°C for 30 seconds. The supernatant was then transferred to a Spin-Away Filter (yellow) inside a collection tube and centrifuged again under the same conditions to remove genomic DNA and facilitate rapid RNA filtration into the collection tube. Subsequently, 100 µl of absolute ethanol (95%–100%) was added at a 1:1 ratio, followed by another centrifugation step. The resulting filtrate was transferred into a Zymo-Spin™ IIICG Column1 (green filter) inside a new collection tube and centrifuged to retain RNA on the green filter, while the collection tube filtrate was discarded. Two hundred sixty-fi µl of RNA Wash Buffer was then added to the green filter (without touching the filter surface) and centrifuged to remove remaining debris. To further purify the RNA, a mixture of 2.5 µl of DNase I and 37.5 µl of DNA Digestion Buffer was carefully applied onto the green filter, ensuring that the surface was not touched. The sample was then incubated at room temperature (20°C–30°C) for 15 minutes to allow efficient DNA digestion. Following incubation, 265 µl of RNA Prep Buffer was added to the sample, which was then centrifuged to remove non-RNA cellular residues, with the resulting filtrate discarded. The purification process continued with a two-step RNA washing procedure. First, 300 µl of RNA Wash Buffer was added, followed by centrifugation, after which the filtrate was discarded. Next, a second wash was performed using 265 µl of RNA Wash Buffer, with the sample centrifuged at 15,000 rpm at 4°C for 1 minute to ensure the complete removal of any remaining non-RNA residues. Finally, the green filter was transferred to a new microtube, and 100 µl of DNase/RNase-Free Water was added directly (without touching the filter surface) and centrifuged to elute the RNA into the microtube. The green filter was discarded, and the purified RNA solution was stored in a freezer for further analysis. cDNA amplificationThe cDNA amplification process was conducted using the Bio-Rad® iScript cDNA Synthesis Kit. The procedure commenced with thawing the extracted RNA. Once thawed, the RNA sample was maintained at a temperature not exceeding 4°C. A total of 12 microtubes were prepared, each containing 2 µl of 5× gDNA Buffer. Then, 3 µl of RNA template and 5 µl of RNase-Free ddH2O were added to each microtube, mixed manually at room temperature, and incubated at 42°C for 3 minutes to remove DNA. While the sample underwent incubation, the reverse transcriptase master mix was formulated by combining 24 µl of 10× King RT Mix, 12 µl of Fast King RT Enzyme Mix, 24 µl of FQ-RT Primer Mix, and 60 µl of RNase-Free Water in a new microtube. The solution was subsequently blended to achieve a consistent mixture. After preparation, 10 µl of the master mix was dispensed into each pre-incubated reaction tube and mixed thoroughly to ensure homogeneity. The amplification of cDNA was carried out using a Bio-Rad® T100 Thermal Cycler (PCR-based Gene Amplification System), following a predefined thermal protocol. The procedure commenced with an initial incubation at 42°C for 15 minutes to support pre-denaturation, followed by a denaturation step at 95°C for 3 minutes. The reaction was then held at 4°C to stabilise the synthesised cDNA prior to the RT-PCR process. Following amplification, the samples were stored appropriately, and a portion was assessed for cDNA concentration using UV absorbance measurement via a NanoDrop® ND-1000 spectrophotometer. Detection of CPT2 gene expression via RT-PCRCPT2 gene expression was assessed using RT-PCR (Bio-Rad CFX96), which is integrated with computer software for graphical data visualisation of CPT2 gene expression in post-thawed frozen goat semen samples. The RT-PCR process was conducted over 60 minutes using the following primers for C. hircus CPT2: CPT2 forward (22 bases, 5’-AGTCAGTAGGCACTATTCTGTG-3’) and CPT2 reverse (22 bases, 5’-CCTGAAAATGTCCTTAAAACCG-3’), designed using primer template from Oktanella et al. (2024). The amplicon length generated by the CPT2 primers was 165 base pairs (bp). The GC content of the forward and reverse primers was 45% and 40.9%, respectively, as determined based on the product information of the Humanizing Genomics Macrogen® primer kit and pre-optimisation trials prior to RT-PCR analysis. An optimal GC content for RT-PCR ranges between 40% and 60% (Sasmito et al., 2014). The melting temperature (Tm) of the primers was also determined according to the Humanizing Genomics Macrogen® primer kit. Based on the optimisation outcomes and manufacturer’s guidelines, the Tm for the CPT2 forward and reverse primers were identified as 51.4°C and 50.2°C, respectively. Tm values depend on the guanine and cytosine base content within a primer. A low Tm may result in inefficient primer annealing, whereas an excessively high Tm can lead to inadequate hybridisation, complicating the separation of hydrogen bonds between bases (Yuenleni, 2019). To maximise primer-template binding accuracy, the Tm difference between the two primers should not exceed 5°C (Sasmito et al., 2014). Besides using specific primers, RT-PCR also necessitates a housekeeping gene, which maintains stable expression, to function as an internal control or reference standard (Ho and Patrizi, 2021). Housekeeping genes are stable and enable normalisation of mRNA expression levels in RT-PCR samples (Irie et al., 2023). This study employed hydroxymethyl-bilane synthase (HMBS) as the housekeeping gene. The primers for HMBS were HMBS forward (20 bases, 5’-CTTGCCAGAGAAGAGTGTGG-3’) and HMBS reverse (20 bases, 5’-CAGCCGTGTGTTGAGGTTTC-3’), producing an amplicon length of 116 bp. The RT-PCR procedure involves several key steps. It begins with a pre-denaturation stage at 95°C for 3 minutes, followed by denaturation at the same temperature for 10 seconds. This is then continued with 35 cycles of annealing at 34.5°C for 30 seconds to enable primer attachment to the specific DNA target. This is followed by an elongation step at 72°C for 30 seconds to synthesise the complementary DNA strand. A final extension step is carried out at 65°C for 5 seconds to complete DNA synthesis. This is followed by a brief finalisation at 95°C for 5 seconds to ensure the amplification process is complete. Data analysisThe data were processed using several statistical tools. First, normality and homogeneity were checked using the Shapiro-Wilk and Levene’s tests. The results showed that the data were not normally distributed and lacked uniformity. Therefore, non-parametric methods were chosen, including the Duncan test, Kruskal-Wallis test, and Pairwise Comparison. All analyses were performed using SPSS® version 23.0 for Windows, with a 95% confidence level (α=0.05). Ethical approvalThis study was carried out in accordance with established ethical principles set by the Research Ethics Commission of Brawijaya University, Malang, Indonesia. Permission was obtained from the Chair of the Research Ethics Commission of Universitas Brawijaya, as documented in the Ethical Clearance Certificate numbered 055-KEP-UB-2023. This research adhered strictly to recognised ethical guidelines at all stages of its implementation. ResultsEvaluation of goat’s fresh semen qualityThe post-thaw sperm qualityThe Shapiro–Wilk normality test and Levene’s test for homogeneity indicated that the data were not normally distributed and not homogeneous (p < 0.05). Post-thaw sperm motility differed significantly among treatment groups (p < 0.05). The mean motility in the control group (T0) was 39.40%, while treatment groups T1, T2, T3, T4, and T5 showed mean values of 42.30%, 41.40%, 42.80%, 41.00%, and 40.60%, respectively (Fig. 1). Post hoc comparisons revealed that motility in T3 was significantly higher than in T0 (p < 0.05), whereas no significant differences were detected between T0 and T1, T2, T4, or T5 (p > 0.05).
Fig. 1. The average of post-thawing spermatozoa motility in each of treatment group as determined using the ANOVA test which shows *p < 0.05 among T1, T2, T3, T4, and T5 to T0. Notes: Treatment groups: T0=0 mg/dL EGCG (control), T1=50 mg/dL, T2=100 mg/dL, T3=200 mg/dL, T4=400 mg/dL, and T5=800 mg/dL. Data are presented as mean values with significance indicated ***p < 0.001. Sperm’s viabilitySpermatozoa staining is done to help with morphological and morphometric observations of spermatozoa, as shown in Figure 2.
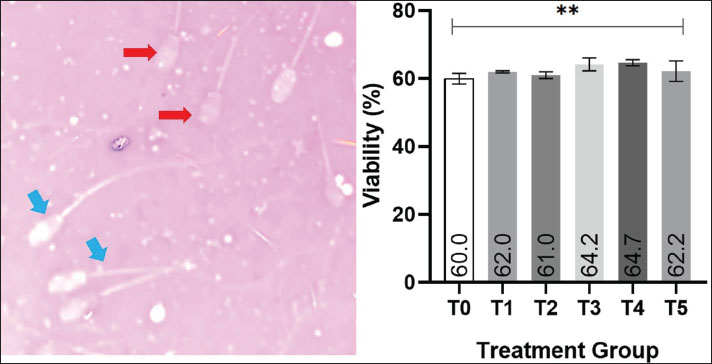
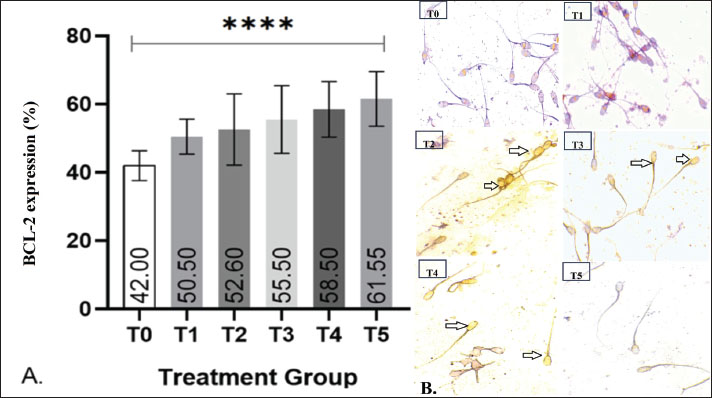
Fig. 2. Effect of EGCG supplementation on post-thaw sperm viability in goats. (A) Image of (blue arrow) live and (red arrow) dead sperm on eosin-nigrosine staining (Merck, Germany) with 400× magnification, Olympus CX-21®, Tokyo, Japan. (B) The average of post-thawing spermatozoa viability in each of treatment group as determined using the ANOVA test which shows *p < 0.05 between T3 and T4 to T0, T1, T2, and T5. Notes: Treatment groups: T0=0 mg/dL EGCG (control), T1=50 mg/dL, T2=100 mg/dL, T3=200 mg/dL, T4=400 mg/dL, and T5=800 mg/dL. Data are presented as mean values with significance indicated ***p < 0.001. The Shapiro–Wilk test for normality and Levene’s test for homogeneity indicated that the data were normally distributed and homogeneous (p > 0.05). Therefore, the analysis was continued with a one-way analysis of variance (ANOVA). The results showed that EGCG supplementation had a significant effect on post-thaw sperm viability (p < 0.05). The mean viability values for T0, T1, T2, T3, T4, and T5 were 60.0%, 62.0%, 61.0%, 64.2%, 64.7%, and 62.2%, respectively. Statistical comparison revealed that T4 exhibited the highest viability, which was significantly different from T0, T1, T2, and T5 (p < 0.05), but not significantly different from T3 (p > 0.05). BCL-2 expressionThe post-thaw expression of BCL-2 in Kacang goat spermatozoa is shown in Fig. 3. Positive BCL-2 expression was identified by brownish staining localized in the head and midpiece regions of the spermatozoa, reflecting its anti-apoptotic role in enhancing sperm survival after cryopreservation. Representative microscopic images (Fig. 3B) further confirm these findings, showing more intense brown staining in the head and midpiece of spermatozoa in treatment groups compared with the control.
Fig. 3. Effect of EGCG supplementation on BCL-2 protein expression in post-thawed goat sperm. (A) The graph presents the mean percentage of BCL-2 expression in post-thaw Kacang goat spermatozoa across EGCG treatment groups. One-way ANOVA analysis revealed a highly significant difference (****p < 0.0001) between the control (T0) and treatment groups (T1–T5). (B) Representative microscopic images of BCL-2 expression detected in post-thaw spermatozoa, visualized using the Avidin–Biotin Complex staining technique at 400× magnification. Positive BCL-2 expression is indicated by black arrows. Notes: Treatment groups: T0=0 mg/dL EGCG (control), T1=50 mg/dL, T2=100 mg/dL, T3=200 mg/dL, T4=400 mg/dL, and T5=800 mg/dL. Data are presented as mean values with significance indicated ***p < 0.001. Quantitative analysis revealed that the control group (T0) exhibited the lowest BCL-2 expression, with a mean value of 42%. In contrast, all EGCG-supplemented groups (T1–T5) demonstrated higher BCL-2 expression, with mean values of 50.5%, 52.6%, 55.5%, 58.5%, and 61.55%, respectively. Statistical analysis using ANOVA indicated a highly significant difference among groups (p < 0.0001). The highest expression was observed in T5, suggesting that EGCG supplementation effectively upregulated BCL-2 in a dose-dependent manner. CPT2 gene expressionThe nonparametric Kruskal–Wallis test revealed a significant difference in CPT2 expression among the six treatment groups (p < 0.05). As shown in Figure 4, the control group (T0) exhibited the highest mean expression (43.39%), whereas all EGCG-supplemented groups (T1–T5) showed lower values, ranging from 34.50% (T1) to 40.03% (T4). Post hoc comparisons indicated that CPT2 expression was significantly reduced in the treatment groups relative to T0 (p < 0.0005). This downregulation of CPT2 suggests that EGCG supplementation effectively attenuates oxidative stress during cryopreservation.
Fig. 4. Graphic of CPT2 gene expression. Notes: Treatment groups: T0=0 mg/dL EGCG (control), T1=50 mg/dL, T2=100 mg/dL, T3=200 mg/dL, T4=400 mg/dL, and T5=800 mg/dL. Data are presented as mean values with significance indicated (***p < 0.001). Notes: Treatment groups: T0=0 mg/dL EGCG (control), T1=50 mg/dL, T2=100 mg/dL, T3=200 mg/dL, T4=400 mg/dL, and T5=800 mg/dL. Data are presented as mean values with significance indicated ***p < 0.001. DiscussionSperm’s motilityThe low post-thaw progressive motility of spermatozoa is commonly attributed to cryopreservation-induced stress. This process exposes cells to oxidative stress through the overproduction of ROS, such as hydroxyl (OH−) and superoxide (O2−), which disrupt membrane integrity and cellular function. According to Mustofa et al. (2021 b), ROS levels increase due to excessive lipid metabolism during the freezing–thawing process. Goat sperm membranes, rich in PUFAs, are highly susceptible to lipid peroxidation, especially under oxidative stress (Simorangkir, 2020). Endogenous antioxidant enzymes—such as superoxide dismutase, glutathione reductase, glutathione peroxidase, and catalase—normally scavenge ROS. However, under cryopreservation stress, the balance between ROS and these antioxidants can be disrupted, resulting in elevated lipid peroxidation and membrane damage (Mustofa et al., 2021 b). The peroxidation of docosahexaenoic acid, a major PUFA in sperm membranes, compromises structural stability and leads to DNA fragmentation (Susilowati et al., 2021). High ROS levels can also affect mRNA translation by damaging nitrogenous bases, thereby hindering protein synthesis and impairing ATP production required for sperm motility (Yan and Zaher, 2019; Ferreira-Silva et al., 2021). The observed improvement in motility in group T3 suggests an optimal effect of epigallocatechin gallate (EGCG) supplementation in semen extenders. EGCG possesses free hydroxyl groups that can donate electrons to neutralize ROS (Zhang et al., 2020). In conjunction with endogenous antioxidants, EGCG facilitates the conversion of ROS into less harmful byproducts such as water and oxygen (Simorangkir, 2020). This antioxidant mechanism likely accounts for the significant enhancement in sperm motility observed in T3 compared with the control (T0). Beyond oxidative stress, other factors may also influence post-thaw motility, including the composition of the diluent, exposure to ambient temperature, and physical or osmotic stress during freezing and thawing. Such stressors can disrupt docosahexaenoic acid bonds, compromise membrane integrity, and increase susceptibility to lysis (Rawash et al., 2018). Despite these unmeasured variables, it is noteworthy that all EGCG-treated groups (T1–T5) maintained post-thaw motility above the 40% threshold, indicating that EGCG supplementation supports the preservation of sperm motility after thawing. Sperm’s viabilityDuring cryopreservation, sperm cells undergo temporary metabolic arrest and are subjected to multiple episodes of extreme cold. The first occurs during equilibration and may induce cold shock, increase osmotic pressure, and contribute to oxidative stress (Zelpina et al., 2012). Elevated osmotic pressure can disrupt phospholipid chains and membrane proteins, reduce membrane selectivity, and impair ionic and nutrient balance (Bebas and Wayan, 2020). As ice crystals form within cells, mechanical damage occurs. Simultaneously, the rise in solute concentration can dissolve the lipoprotein layers of sperm membranes, altering membrane permeability upon thawing (Alhuur et al., 2020). This contributes to a decrease in membrane viability by as much as 50% (Zelpina et al., 2012; Khalil et al., 2018). The peroxidation of PUFA-rich membranes further worsens viability by increasing permeability and inducing apoptosis or necrosis (Fadhilah et al., 2021). EGCG, a catechin with potent antioxidant activity, works by donating hydrogen atoms or electrons to neutralize free radicals (Min and Kwon, 2014). EGCG’s antioxidant action can occur both extracellularly—by forming a protective barrier—and intracellularly, by scavenging ROS and stabilizing redox balance (Singh et al., 2011; Shukla et al., 2018; Rokana et al., 2022). EGCG also chelates metals and boosts endogenous antioxidant enzyme activity, maintaining membrane integrity (Min and Kwon, 2014). The mechanism of action of EGCG can work outside the cell (extracellular) and inside the cell (intracellular). Extracellularly, EGCG or antioxidants work by interacting with the cellular matrix, which forms an extracellular protection barrier and helps prevent oxidative damage (Singh et al., 2011). According to (Rokana et al., 2022), EGCG works in cells (intracellular), which can reduce the production of ROS by complementing unpaired electrons so as to reduce damage to proteins, lipid membranes, and nucleic acids. Also, EGCG is proven to be able to prevent oxidative stress when free radical production exceeds the antioxidant capacity of cells by maintaining redox balance and then neutralizing free radicals so that cell viability can be maintained (Shukla et al., 2018). EGCG antioxidants are also metal chelators and inhibit the formation of ROS and fat peroxidation by increasing superoxide dismutase activity, restoring glutathione peroxidase and glutathione activity so that the integrity of the spermatozoa plasma membrane can be intact and cell viability can be maintained (Min and Kwon, 2014). The improvement observed in groups T3 and T4 suggests that EGCG at moderate concentrations enhances sperm viability by reducing oxidative stress during the cryopreservation process. EGCG acts as a potent antioxidant through its hydroxyl groups, which donate electrons to neutralize ROS, thereby protecting sperm membranes from lipid peroxidation and structural damage. However, the lack of additional benefit in the T5 group (highest EGCG concentration) indicates a dose-dependent effect. At elevated concentrations, EGCG may undergo autoxidation, producing hydroxyl radicals that trigger pro-apoptotic pathways (Husna et al., 2017; Roychoudhury et al., 2017). Environmental factors such as pH, temperature, and oxygen availability can accelerate this destabilization, diminishing the protective capacity of EGCG at high doses. Taken together, the findings highlight that moderate EGCG supplementation (T3–T4) provides optimal protection, whereas excessive doses (T5) may counteract the beneficial antioxidant effects. BCL-2 expressionBCL-2 is a key anti-apoptotic protein. In this study, its expression was lowest in the control group (T0) and increased with EGCG supplementation across T1–T5. Similar results were reported by Samik et al. (2014) in bulls and Alvarez-Rodriguez et al. (2019) in rams using different antioxidants, showing enhanced BCL-2 expression and reduced pro-apoptotic markers. In contrast, resveratrol had minimal effects on motility and viability in boar sperm despite increasing BCL-2 (Yeste et al., 2020). Melatonin supplementation also upregulated BCL-2 and preserved mitochondrial integrity in human sperm (Zhou et al., 2021). Sperm undergo both apoptosis and necrosis during cryopreservation, with apoptotic rates increasing post-thaw (Samik et al., 2014). While apoptotic cells maintain membrane integrity, they exhibit cytoplasmic shrinkage and nuclear condensation, eventually undergoing phagocytosis without inflammation (Asadi et al., 2021). Apoptosis is regulated via intrinsic (mitochondrial) and extrinsic (death receptor) pathways. The intrinsic pathway involves mitochondrial signals like cytochrome c release, regulated by BCL-2 family proteins (Marleen et al., 2008). BCL-2 inhibits cytochrome c release and thus prevents caspase-9 activation, promoting cell survival. Therefore, a high BCL-2/BAX ratio is essential for cell viability. In this study, supplementation with 300 mg/dl EGCG (T4) produced the highest post-thaw sperm viability (64.7%), while the 500 mg/dl group (T5) exhibited the greatest BCL-2 expression (61.55%). These findings highlight EGCG’s dual role: at moderate concentrations it effectively preserves sperm viability by reducing oxidative stress, and at higher concentrations it enhances anti-apoptotic signaling through BCL-2 upregulation. The localization of BCL-2 expression in the sperm head and midpiece further suggests a protective mechanism for maintaining membrane and mitochondrial integrity during cryopreservation. Nevertheless, this study did not evaluate pro-apoptotic markers such as Bax or caspase activity, which limits a full understanding of the balance between apoptotic and anti-apoptotic pathways. Future investigations should therefore assess both pro- and anti-apoptotic signals to clarify how EGCG modulates cell survival and determine the optimal dose that maximizes cryoprotection without inducing pro-oxidant effects. CPT2 gene expressionCryopreservation also affects mitochondrial metabolism. The CPT2 gene encodes carnitine palmitoyltransferase 2, an enzyme critical for β-oxidation and ATP production (Wang et al., 2021). However, excessive ROS can impair CPT2 function, lowering energy availability for sperm motility (Ferreira-Silva et al., 2021). Variability in CPT2 expression may be influenced by PUFAs availability and the integrity of DNA/mRNA, which regulate its transcription. Low CPT2 expression results in ATP depletion, while overexpression may trigger hypermotility and premature capacitation, compromising fertilisation (Sigauke et al., 2003; Zhang et al., 2021). Excessive ROS disrupts cAMP signalling and promotes premature capacitation. Nguyen et al. (2023) and Hidalgo et al. (2020) reported similar findings, showing impaired CPT2 expression and reduced sperm viability under oxidative stress. Conversely, high CPT2 expression correlated with better mitochondrial function. EGCG’s antioxidant properties may contribute to the regulation of CPT2 expression by maintaining mitochondrial membrane stability and protecting DNA integrity (Simorangkir, 2020). Zhou et al. (2021) similarly reported improved mitochondrial function following EGCG supplementation. In the present study, CPT2 expression was significantly reduced in all EGCG-treated groups (T1–T5) compared with the control (T0), with mean values ranging from 34.50% to 40.03% versus 43.39% in T0. This downregulation indicates that EGCG supplementation may suppress oxidative stress–related gene activity while promoting mitochondrial protection. Although T3 (200 mg/dl EGCG) did not show the lowest CPT2 expression, it corresponded with the highest post-thaw motility observed in previous results, suggesting that this concentration may optimally balance antioxidant defense and mitochondrial function. The reduction in CPT2 expression may also reflect decreased demand for fatty acid transport into mitochondria under conditions of lower oxidative stress. However, limitations of this study include the absence of a direct correlation analysis between CPT2 expression and sperm motility, as well as the lack of enzymatic assays to assess lipid metabolism pathways. Future studies should explore how CPT2 interacts with other oxidative stress and apoptotic markers, and evaluate whether EGCG exerts similar regulatory effects across different species. ConclusionOur study demonstrates that supplementation of Kacang goat semen extenders with epigallocatechin gallate (EGCG) exerts dose-dependent protective effects during cryopreservation. A concentration of 200 mg/dl (T3) significantly improved post-thaw sperm motility, while 300 mg/dl (T4) yielded the highest sperm viability. At the molecular level, 500 mg/dl (T5) induced the greatest upregulation of BCL-2 expression, indicating enhanced anti-apoptotic activity. Conversely, CPT2 expression was significantly reduced across all EGCG-treated groups compared with the control, suggesting a protective effect through reduced oxidative stress and improved mitochondrial stability. Taken together, these results suggest that EGCG may serve as a beneficial additive in cryopreservation protocols, with encouraging prospects for improving fertility outcomes and advancing reproductive biotechnology in small ruminant. AcknowledgementsWith sincere appreciation, the authors acknowledge to the staff and the facilities provided by the Teaching Farm, Universitas Airlangga. This research was funded through the 2023 collaborative research grant (Riset Kolaboratif DPP SPP) of the Faculty of Veterinary Medicine, Universitas Brawijaya. Conflict of interestThe authors hereby declare that there are no competing interests associated with the conduct or publication of this research. FundingCollaborative research grant (Riset Kolaboratif DPP SPP) of the Faculty of Veterinary Medicine, Universitas Brawijaya. Authors' contributionsYO, IM: conceptualization, data collection, and writing of the original manuscript draft. SS, NY, TH: literature search, design, manuscript preparation and review. YK: data acquisition, manuscript review. AC, JC, SF, FA: sharing in writing the manuscript draft and editing, experimental studies, data analysis, and statistical analysis. All authors have approved the final manuscript for publication. Data availabilityAll data supporting the findings of this study are available in the manuscript, and no additional data sources are required ReferencesAlhuur, K.R., Soeparna, S. and Rd, D. 2020. The interaction effect of equilibration time and freezing rate toward the membrane plasma sperm integrity of Priangan ram sperm post-thawing. J. Ilmu. Ternak. Dan. Prod. 8, 73–78. Alvarez-Rodriguez, M., Alvarez, M., Muriel, A., Anel, L. and Paz, D. 2019. Quercetin supplementation improves post-thaw sperm quality and reduces oxidative stress in ram semen. Theriogenology 133, 50–57. Amor, H., Zeyad, A., Alkhaled, Y., Laqqan, M., Saad, A., Ben Ali, H. and Hammadeh, M.E. 2018. Relationship between nuclear DNA fragmentation, mitochondrial DNA damage and standard sperm parameters in spermatozoa of fertile and sub-fertile men before and after freeze-thawing procedure. Andrologia 50(1), 1–9. Asadi, A., Ghahremanim, R., Abdolmaleki, A. and Rajaei, F. 2021. Role of sperm apoptosis and oxidative stress in male infertility: a narrative review. IJRM 19, 493–504. Balasubramanian, D. and Palanisamy, K. 2014. Effect of epigallocatechin-3-gallate (EGCG) on cryopreserved semen quality in Murrah buffalo bulls. Int. J. Vet. Sci. Res. 9, 312–318. Bebas, W. and Wayan, G. 2020. Optimal glycerol and dimethyl sulfoxide cryoprotectant levels in astaxanthin phosphate duck egg yolk extender on the quality of frozen boar semen. J. Vet. 21, 115–123. Bodu., Sarıözkan, S., Bucak, M.N. and Ateşşahin, A. 2025. Advances in cryopreservation of goat sperm: extender formulation, antioxidants, and molecular approaches. Front. Vet. Sci. 12, 1554771. Burry, R.W. 2011. Controls for Immunocytochemistry. J. Histochem. Cytochem. 59, 6–12. Fadhilah, Z.H., Perdana, F. and Syamsudin, R.A. 2021. Review: analysis of catechin and epigallocatechin gallate (EGCG) compounds as antioxidants in various types of tea. J. Pharm. Sci. 8, 31. Ferreira-Silva, J.C., Rocha, C.C. and Mendes, C.M. 2021. The role of oxidative stress in sperm metabolism and fertilization potential. Andrology 9, 15–28. Gürler, H., Malama, E., Heppelmann, M., Calisici, O., Leiding, C., Kastelic, J.P. and Bollwein, H. 2016. Effects of cryopreservation on sperm viability, synthesis of reactive oxygen species, and DNA damage of bovine sperm. Theriogenology 86, 562–571. Hidalgo, M., Martorell, P. and Pujol, C. 2020. Oxidative stress and lipid metabolism in post-thawing spermatozoa. Theriogenology 146, 129–140. Ho, K.H. and Patrizi, A. 2021. Assessment of common housekeeping genes as reference for gene expression studies using RT-qPCR in mouse choroid plexus. Sci. Rep. 11, 3278. Husna, U., Sujuti, H. and Dalhar, M. 2017. Effect of propolis extract administration on Bcl-2 expression and apoptosis in rats' brain cells model of traumatic brain injury. JKB. 29(3), 196–201. Irie, N., Warita, K., Tashiro, J., Zhou, Y., Ishikawa, T., Oltvai, Z.N. and Warita, T. 2023. Expression of housekeeping genes varies depending on mevalonate pathway inhibition in cancer cells. Heliyon 9, 18017. Kalashnyk-Vakulenko, I., Gubina-Vakulyck, G., Onishchenko, A., Nakonechna, O., Gorbach, T., Tkachenko, V. and Tkachenko, A. 2022. Circulating levels of Bcl-2 and its expression in the nasal mucosa of patients with chronic rhinosinusitis. J. Clin. Med. Kaz. 19, 63–67. Khalil, W.A., El-Harairy, M.A., Zeidan, A.E.B., Hassan, M.A.E. and Mohey-Elsaeed, O. 2018. Evaluation of bull spermatozoa during and after cryopreservation: structural and ultrastructural insights. Int. J. Vet. Sci. Med. 6, 49–56. Kusumawati, E.D., Rahadi, S., Nurwathon, S. and Yulianti, D.L. 2019. Post-thaw quality of Etawa crossbreed (PE) goat spermatozoa at 37°C with different time intervals. J. Ilmu. Ternak. Dan. Vet. Trop. 6, 246–250. Lestari, D.L., Gusmira, Y.H., A’arif, M. and Amelia, A.Y. 2024. Free Nutritious Meal Policy as a Solution to Overcoming the Stunting Problem in Indonesia. Innov. J. Soc. Sci. Res. 4, 10021–10031. Marleen, L., Peters, A. and Koch, M. 2008. BCL-2 family proteins and their role in apoptosis regulation: a review. J. Cell. Physiol. 217, 331–338. Min, K. and Kwon, T.K. 2014. Anticancer effects and molecular mechanisms of epigallocatechin-3-gallate. Integr. Med. Res. 3, 16–24. Mustofa, A., Riyadhi, M.A. and Susilowati, S. 2021. Effects of green tea extract (EGCG) on post-thawing quality of frozen semen in Kacang goats. Vet. Med. Anim. Sci. 2, 45–53. Mustofa, A., Setiadi, M.A., Wibowo, T.B. and Riyadi, P. 2021. Effect of green tea extract supplementation on the quality of frozen-thawed Kacang goat semen. Vet. World 14, 1240–1246. Nguyen, N.D., Le, M.T., Dang, H.N.T., Van Nguyen, T., Nguyen, Q.H.V. and Cao, T.N. 2023. Impact of semen oxidative stress on sperm quality: initial results from Vietnam. J. Int. Med. Res. 51(1), 1–12. Oktanella, Y., Mustofa, I., An-Haru, F.A.F.R., Putri, D.D.M., Hendrawan, V.F., Susilowati, S., Degu, N.Y. and Hernawati, T. 2024. Conserving goat sperm post-thawed gene expression and cellular characteristics using the antioxidant coenzyme Q10 supplementation. Vet. World 17, 1637–1647. Peris-Frau, P., Soler, A.J., Iniesta-Cuerda, M., Martín-Hidalgo, D., Fernández-Santos, M.R. and Garde, J. 2020. Sperm cryopreservation: evolution from past to future in animal reproduction. Theriogenology 150, 246–258. Qian, S., Wei, Z., Yang, W., Huang, J., Yang, Y. and Wang, J. 2022. The role of BCL-2 family proteins in regulating apoptosis and cancer therapy. Front. Oncol. 12, 1–16. Ranjan, R., Sharma, K., Kumar, M., Swain, D.K., Singh, S.P., Kharche, S.D., Singh, M.K. and Chauhan, M.S. 2024. IGF-1 stabilizes goat sperm mitochondrial transmembrane potential and reduces DNA fragmentation. CryoLetters 44, 327–332. Ratnawati, D., Isnaini, N. and Susilawati, T. 2019. Factors Affecting Spermatozoa Motility Analysis using CASA. Indones. Bull. Anim. Vet. Sci. 29, 145. Rawash, Z., El-Raey, M. and Ibrahim, E. 2018. Effects of reduced glutathione on Boer goat semen freezability. Asian Pac. J. Reprod. 7, 33. Rokana, N., Singh, R. and Pande, V. 2022. Cold shock and its impact on sperm motility and viability during cryopreservation. Cryobiol. Cryotechnol. 14, 23–30. Roychoudhury, S., Agarwal, A., Virk, G. and Cho, C.L. 2017. Potential role of green tea catechins in the management of oxidative stress-associated infertility. Reprod. Biomed. Online. 34, 487–498. Samik, J., Nizanski, W., Majewska, A. and Dziekonska, A. 2014. Osteopontin supplementation improves cryopreserved bull semen quality by enhancing BCL-2 expression. Anim. Reprod. Sci. 145, 82–89. Sasmito, D.A., Susilowati, S. and Sardjono, T.W. 2014. The effect of primer design on the efficiency of RT-PCR amplification. Indonesian J. Biotechnol. 19(2), 75–82. Sigauke, E., Rakheja, D., Kitson, K. and Bennett, M. J. 2003. Carnitine palmitoyltransferase II deficiency: a clinical, biochemical, and molecular review. Lab. Invest. 83, 1543–1554. Shukla, A.S., Jha, A.K., Kumari, R., Rawat, K., Syeda, S. and Shrivastava, A. 2018. Role of catechins in chemosensitization. In: Role of nutraceuticals in cancer chemosensitization. Eds., Bharti, A.C. and Aggarwal, B.B. Cambridge, MA: Academic Press, pp: 169–198. Simorangkir, H.A.H. 2020. Microencapsulation of a combination of curcumin from turmeric (Curcuma longa) and epigallocatechin-3-gallate (EGCG) from green tea leaves (Camellia sinensis): an innovation in preventive therapy for diabetic retinopathy in type 2 diabetes mellitus patients. Scripta Score Sci. Med. J. 1, 11. Singh, B.N., Shankar, S. and Srivastava, R.K. 2011. Green tea catechin, epigallocatechin-3-gallate (EGCG): mechanisms, perspectives and clinical applications. Biochem. Pharmacol. 82, 1807–1821. Susilowati, S., Mustofa, I., Wurlina, W., Hernawati, T. and Oktanella, Y. 2021. Maintaining the quality of Kacang buck semen in chilled storage with the addition of green tea extract in extender. Trop. Anim. Sci. J. 44, 408–414. Umami, N., Mustofa, I. and Susilowati, S. 2015. The impact of cryopreservation on goat sperm motility and fertilization potential. J. Anim. Sci. Technol. 37, 456–463. United Nations. 2015. Transforming our world: The 2030 Agenda for sustainable development. United Nations. Available via https://sdgs.un.org/2030agenda Wang, M., Wang, K., Liao, X., Hu, H., Chen, L., Meng, L., Gao, W. and Li, Q. 2021. Carnitine palmitoyltransferase system: a new target for anti-inflammatory and anticancer therapy. Front. Pharmacol. 12, 760581. Wieser, M., Stadler, G. and Jennings, P. 2014. Carnitine palmitoyltransferase 2 (CPT2) and its role in lipid metabolism. BBA-Mol. Cell Res. 1841, 645–651. Xu, B., Wang, R., Wang, Z., Liu, H., Wang, Z., Zhang, W., Zhang, Y., Su, R., Liu, Z., Liu, Y., Li, J. and Zhang, J. 2022. Evaluation of lipidomic change in goat sperm after cryopreservation. Front. Vet. Sci. 9, 1–11. Yan, L.L. and Zaher, H.S. 2019. How do cells cope with RNA damage and its consequences?. J. Biol. Chem. 294, 15158–15171. Yeste, M., Sancho, S., Briz, M. and Bonet, S. 2020. The effect of resveratrol on boar sperm cryopreservation: bCL-2 expression and mitochondrial function. Reprod. Biol. 20, 71–79. Yuenleni, Y. 2019. Steps for PCR optimization. Indonesia. J. Lab. 1, 51. Zelpina, E., Rosadi, B. and Sumarsono, T. 2012. Post-thaw spermatozoa quality of frozen semen from dairy cattle. Sci. J. Anim. Sci. 15, 86–94. Zhang, X., Zhang, Z., Liu, S., Li, J., Wu, L., Lv, X., Xu, J., Chen, B., Zhao, S. and Yang, H. 2021. CPT2 down-regulation promotes tumor growth and metastasis through inducing ROS/NFκB pathway in ovarian cancer. Transl. Incol. 14, 1–10. Zhang, Y., Lin, H., Liu, C., Huang, J. and Liu, Z. 2020. A review for physiological activities of EGCG and the role in improving fertility in humans/mammals. Biomed. Pharmacother. 127, 1–10. Zhou, Y., Wang, H. and Li, P. 2021. EGCG improves sperm quality and mitochondrial function in cryopreserved human spermatozoa. Reprod. Biol. 21, 23–31./. | ||
| How to Cite this Article |
| Pubmed Style Oktanella Y, Cahyani AA, Caula JS, Fathiyananda SN, An-haru FAR, Mustofa I, Susilowati S, Degu NY, Hernawati T, Kholifah Y. Protective effect of epigallocatechin-gallate in cryopreserved goat sperm on post-thawed quality. Open Vet. J.. 2025; 15(10): 5116-5127. doi:10.5455/OVJ.2025.v15.i10.29 Web Style Oktanella Y, Cahyani AA, Caula JS, Fathiyananda SN, An-haru FAR, Mustofa I, Susilowati S, Degu NY, Hernawati T, Kholifah Y. Protective effect of epigallocatechin-gallate in cryopreserved goat sperm on post-thawed quality. https://www.openveterinaryjournal.com/?mno=272173 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.29 AMA (American Medical Association) Style Oktanella Y, Cahyani AA, Caula JS, Fathiyananda SN, An-haru FAR, Mustofa I, Susilowati S, Degu NY, Hernawati T, Kholifah Y. Protective effect of epigallocatechin-gallate in cryopreserved goat sperm on post-thawed quality. Open Vet. J.. 2025; 15(10): 5116-5127. doi:10.5455/OVJ.2025.v15.i10.29 Vancouver/ICMJE Style Oktanella Y, Cahyani AA, Caula JS, Fathiyananda SN, An-haru FAR, Mustofa I, Susilowati S, Degu NY, Hernawati T, Kholifah Y. Protective effect of epigallocatechin-gallate in cryopreserved goat sperm on post-thawed quality. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 5116-5127. doi:10.5455/OVJ.2025.v15.i10.29 Harvard Style Oktanella, Y., Cahyani, . A. A., Caula, . J. S., Fathiyananda, . S. N., An-haru, . F. A. R., Mustofa, . I., Susilowati, . S., Degu, . N. Y., Hernawati, . T. & Kholifah, . Y. (2025) Protective effect of epigallocatechin-gallate in cryopreserved goat sperm on post-thawed quality. Open Vet. J., 15 (10), 5116-5127. doi:10.5455/OVJ.2025.v15.i10.29 Turabian Style Oktanella, Yudit, Ayunda A. Cahyani, Juhi S. Caula, Sabrina N. Fathiyananda, Fahrunnisak A. R. An-haru, Imam Mustofa, Suherni Susilowati, Nurhusien Y. Degu, Tatik Hernawati, and Yayuk Kholifah. 2025. Protective effect of epigallocatechin-gallate in cryopreserved goat sperm on post-thawed quality. Open Veterinary Journal, 15 (10), 5116-5127. doi:10.5455/OVJ.2025.v15.i10.29 Chicago Style Oktanella, Yudit, Ayunda A. Cahyani, Juhi S. Caula, Sabrina N. Fathiyananda, Fahrunnisak A. R. An-haru, Imam Mustofa, Suherni Susilowati, Nurhusien Y. Degu, Tatik Hernawati, and Yayuk Kholifah. "Protective effect of epigallocatechin-gallate in cryopreserved goat sperm on post-thawed quality." Open Veterinary Journal 15 (2025), 5116-5127. doi:10.5455/OVJ.2025.v15.i10.29 MLA (The Modern Language Association) Style Oktanella, Yudit, Ayunda A. Cahyani, Juhi S. Caula, Sabrina N. Fathiyananda, Fahrunnisak A. R. An-haru, Imam Mustofa, Suherni Susilowati, Nurhusien Y. Degu, Tatik Hernawati, and Yayuk Kholifah. "Protective effect of epigallocatechin-gallate in cryopreserved goat sperm on post-thawed quality." Open Veterinary Journal 15.10 (2025), 5116-5127. Print. doi:10.5455/OVJ.2025.v15.i10.29 APA (American Psychological Association) Style Oktanella, Y., Cahyani, . A. A., Caula, . J. S., Fathiyananda, . S. N., An-haru, . F. A. R., Mustofa, . I., Susilowati, . S., Degu, . N. Y., Hernawati, . T. & Kholifah, . Y. (2025) Protective effect of epigallocatechin-gallate in cryopreserved goat sperm on post-thawed quality. Open Veterinary Journal, 15 (10), 5116-5127. doi:10.5455/OVJ.2025.v15.i10.29 |