| Research Article | ||
Open Vet. J.. 2026; 16(1): 635-645 Open Veterinary Journal, (2026), Vol. 16(1): 635-645 Research Article Chlorella vulgaris improves fertility in malnourished male rats by regulating the oxidative state, spermatogenesis, and inflammatory cytokinesYasser H. A. Saber1*, Ghada H. Elsayed2,3, Naglaa A. Ali2, Emtenan M. Hanafi1, Mohamed Ali1, Walid S. El-Nattat1, Heba Hozyne1, Omima H. Ezzo1, and Safaa H. Mohamed21Animal Reproduction and AI, National Research Centre, Giza, Egypt 2Hormones Department, Medical and Clinical Research Institute, National Research Centre, Giza, Egypt 3Stem Cells Lab, Centre of Excellence for Advanced Sciences, National Research Centre, Giza, Egypt *Corresponding Author: Yasser H. A. Saber. Department of Animal Reproduction and A.I, Veterinary Research Institute, National Research Centre, Dokki, Giza, Egypt. Email: Yasserhussein_2011 [at] yahoo.com Submitted: 21/07/2025 Revised: 22/11/2025 Accepted: 12/11/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
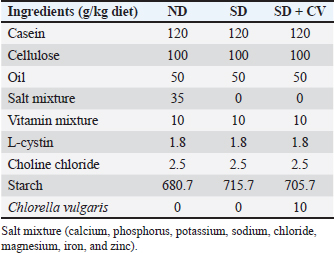
AbstractBackground: Malnutrition is one of the predisposing factors of disturbance in some physiological functions, including reproductive health. Currently, many people and animals are globally facing micronutrient deficiency, especially in lands with poor soils in organic matter and nutrients. Egyptian soil and, consequently, crops were found to be deficient in micronutrients to varying degrees. Aim: This study aimed to investigate the potential fertility-enhancing effects of the microalgae Chlorella vulgaris (CV) on male rats fed a salt-deficient diet. Methods: Forty-five adult rats (15 rats each) were divided into three groups (G1, G2, and G3). G1 received a normal balanced diet, G2 received a salt-deprived diet (SD), and G3 was supplemented with SD plus Chlorella 1% of the diet (SD + CV). After eight successive weeks of treatment, the animals were fasted overnight, anesthetized, and sacrificed. The collected plasma was analyzed for organ function-related constituents, oxidative biomarkers, and testosterone levels. Epididymal sperms were also examined. Samples of testis were analyzed for quantitative gene expression of GPX4, SOD1, and SPATA6 genes. Additionally, histopathological examination was performed for testis, liver, and kidney samples. Results: The results obtained from the current study revealed that the plasma of the SD group had higher levels of oxidative biomarkers and inflammatory cytokines, elevated urea, creatinine, and uric acid levels, and liver enzymes, and lower testosterone levels than the normal control group. In addition, lower sperm motility and higher abnormality and downregulation of SPATA6 genes and degenerative changes were found in the testes, liver, and kidneys. The administration of (SD + CV) restored these alterations. Conclusion: CV could be used as a supplement to compensate for nutrient or salt deficiency and correct the resultant disorders, especially reproductive affections, in males. Keywords: Male fertility, Zinc and copper deficiency, Chlorella vulgaris, SPATA6, Oxidative stress. IntroductionEssential metals, particularly zinc (Zn), selenium (Se), manganese (Mn), iron (Fe), copper (Cu), molybdenum (Mo), and cobalt (Co), are generally thought to be advantageous or even protective for male reproductive activities, although excess of these elements can have negative consequences as well (Ribeiro et al., 2021). Fe, Zn, and Se are examples of trace metals that are classified as components with antioxidant action (Nenkova et al., 2007). The soil in the Middle East region has low organic matter and is deficient in micronutrients. Egyptian soil and cultivated crops are deficient in Zn, Cu, Fe, and Mn to varying degrees (El-Fouly et al., 2010). This results in health problems in humans and animals fed on such crops. Currently, two billion people globally are facing micronutrient deficiency (Velu et al., 2014). Male reproduction depends on trace elements, and their lack may result in poor spermatogenesis, decreased libido, and decreased male fertility (Tvrdá et al., 2013). According to Zaman et al. (2017), maize is the most prone crop to zinc deficiencies; rice and wheat are also very sensitive. Zinc and copper must be included in the diet because they are required for many different biochemical processes in the body (El-Said et al., 2019). Chlorella vulgaris (CV) is a highly nutritious unicellular freshwater microalgae that is rich in many important components, including proteins, carbohydrates, vitamins, fatty acids, and trace elements such as zinc, copper, and magnesium. These components are essential for the activity of antioxidant metalloenzymes and male reproduction (Osama et al., 2019; Hanafi et al., 2022). Moreover, CV is rich in polyunsaturated fatty acids (PUFAs), which have been found to improve male fertility as they could increase ejaculate volume, sperm cell concentration, viability, and motility of fresh and post-thawed sperm cells (Khoshvaght et al., 2016; Hanafi et al., 2022). Furthermore, CV has strong antioxidant characteristics that are attributed to bioactive substances such as saponins, alkaloids, flavonoids, and phenols. These substances act as peroxyl radical scavengers, successfully preventing free radical production within living cells. Additionally, CV prevents lipid peroxidation and inhibits PUFA oxidation (Alshafei et al., 2022; Makipour et al., 2024). In this instance, CV, which is frequently used as a dietary supplement, might be a good option to improve the function of the male genital system under normal or oxidative stress conditions. The current study aimed to clarify the efficiency of CV in compensating for the negative effect of mineral deficiency on male fertility. Gene expression analysis and biological and histopathological changes were studied. Materials and MethodsThe microalga and CV were provided by the Food Industry and Nutrition Research Institute, National Research Center, Cairo, Egypt. Nutrient analysis of CV(100 gm powder)The CV powder was analyzed (Table 1) for minerals, carbohydrates, proteins, fats, and ash according to the method of Canelli et al. (2020). Minerals and trace elements were estimated by atomic absorption. Table 1. Types of diets prepared for animal feeding: ND, normal balanced diet, SD: Salt deprived diet; SD + CV: SD enriched with CV at a concentration of 1%.
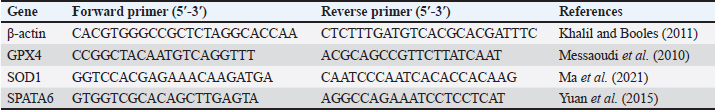
Biological experimentForty-five adult male albino Wistar rats, weighing 200–270 g were obtained from the Animal Care Unit of the National Research Centre (NRC), Egypt. The animals were fed a standard chow diet and water ad libitum. They were housed in transparent plastic cages with wood shavings in a freely ventilated room with controlled room temperature (25°C ± 1°C), humidity (50% ± 10%), and alternating 12 hours cycles of light and dark. The animals were allowed to acclimatize in their new environment for 1 week before the commencement of the experiment. Then, they were randomly divided into three groups (G1, G2, G3) with 15 rats each. G1 received a normal balanced diet (ND), G2 received a salt-deprived diet (SD), and G3 was supplemented with SD plus encapsulated Chlorella 1% of the diet. SamplingAfter eight successive weeks of treatment, the animals were fasted overnight, euthanized under anesthesia with ketamine hydrochloride Intramuscular (45 mg/kg, IM), and sacrificed by cervical dislocation. Blood samples were collected from the medial retro-orbital venous plexus of rats using micro hematocrit capillaries (Abdi-Azar and Maleki, 2014). Heparinized blood samples were collected for biochemical analysis using commercial reagent kits (Roche Diagnostics, Laval, QC, Canada). The testis and epididymis were carefully dissected out. One testis from each rat was preserved at −80°C for later evaluation of quantitative real-time mRNA gene expression in the testes, and the other was preserved in 10% formalin for histopathological examination. Epididymal semen was also collected for microscopic examination of the epididymis semen picture. Blood samples were centrifuged, and the collected plasma was kept in −20 C for analysis of possible changes in organ function-related parameters Aspartate transaminase (AST), Alanine transaminase (ALT), Gamma-glutamyl transferase (GGT), urea, uric acid, and creatinine for kidney), total protein, albumin, and some oxidative and antioxidant biomarkers Malondialdehyde (MDA and catalase), glucose, and triglyceride were performed using commercial kits from Biodiagnostic Egypt. Inflammatory cytokines Tumor Necrosis Factor α (TNFα), InterLeukin-6 (IL-6), and Toll-like receptors (TLR), ghrelin, and testosterone hormone levels were analyzed with ELISA kits (TNF-α, IL-6 kit was purchased from Bio-Source International Inc, 542, Flynn Road, Camarillo, California, USA; ghrelin kit from DRG International, Inc, USA). Evaluation of the epididymal spermatozoaThe caudal part of the epididymis was cut and squeezed to harvest the epididymal sperms and suspended in 1 ml warm phosphate-buffered saline. Sperm motility was estimated immediately on a clean warm slide. The concentration of sperms was determined after diluting the epididymal content with 2.9% sodium citrate dehydrate solution (Türk et al., 2007). Sperm cell count in 1 mm3 of the suspension was detected using a hemocytometer. Then, the total number of sperms/epididymal tail were calculated=sperm number in 1 mm3 of diluted suspension × dilution rate × 1 ml (total volume of the epididymal suspension). Sperm abnormalities were assessed via slide smearing of one drop of epididymal content, followed by staining with eosin nigrosine. A total of 200 sperm were examined by a light microscope for sperm abnormalities such as banana-shaped, straight, and other abnormalities (Okamura et al., 2005). Quantitative real-time polymerase chain reactionRNA was isolated from rat testicular tissues using RNA Easy Mini Kit (Qiagen, Germany) (Cat. No.: 74104). Then, the concentration and purity of the total extracted RNA were determined using a NanoDrop One microvolume UV spectrophotometer (Thermo Fisher Scientific, USA). The RNA of each treatment was converted to first-strand cDNA according to the manufacturer’s instructions using the Revert Aid First Strand cDNA Synthesis Kit (Cat. No.: K1621; Thermo Fisher Scientific, USA). The specific primer sequences are listed in Table 2. The expression levels of Glutathione Peroxidase 4 (GPX4), Super Oxide Dismutase 1 (SOD1), and SPATA6 genes were normalized with respect to β-actin transcript using Maxima SYBR Green qPCR Master Mix (2X) (Cat. NO.: K0221; ThermoFisher Scientific, USA) and calculated by 2−ΔΔCT method (Livak and Schmittgen, 2001). The reaction conditions were as follows: 95°C for 10 minutes, 95°C for 15 seconds, 55°C for 30 seconds, and 72°C for 30 seconds, with a total of 40 cycles of amplification. The DNA Technology Detecting Thermocycler DT Lite 4S1 (Russia) was used for gene expression quantitation. Table 2. Primers used for qRT–PCR.
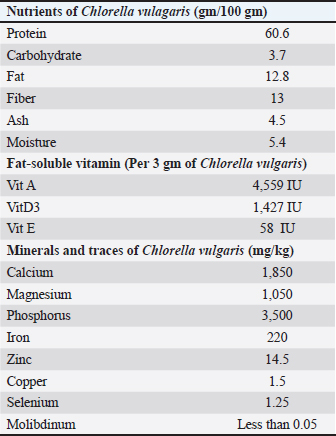
Histopathological examinationFor the light microscopic examination, tissue specimens (testis, livers, and kidneys) were fixed in 10% buffered formalin using tissue cassettes. Tissue sections were cut into 4–5 μm, and stained with Hematoxylin and Eosin (Suvarna et al., 2018), and subjected to microscopic examination by a pathologist. Tissue samples were examined using a Leica UA510CA light microscope and a Leica Q-Win Image Analysis system (Leica Micros Imaging Solutions Ltd., Cambridge, UK). Statistical analysisResults are presented as mean ± SEM. Statistically significant differences between groups were determined using one-way analysis of variance (one-way ANOVA), followed by Tukey–Kramer multiple comparison test. GraphPad Prism version 8 was used for data analysis. The significance level was set at p < 0.05 for all tests. Ethical approvalAnimal caring and treatments throughout the experiment were carried out according to the approval and guidelines given by the Institutional Ethical Committee for Medical Research of the National Research Centre, Egypt (Code No: 12-115). This study was approved by the Institutional Ethical Committee for Medical Research of the National Research Centre, Egypt. The study was reported in accordance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines (ethical approval no. 20170). ResultsAnalysis of CVnutrientsThe results of the current study (Table 3) revealed that CV was a rich source of protein and vitamins A and D. It was also rich in minerals and traces of essential minerals for different biological processes, such as calcium, magnesium, phosphorus, iron, zinc, copper, and selenium. Simultaneously, the hazardous molybdenum was found nil. Table 3. Analysis of nutrients antioxidant activity of Chlorella vulgaris.
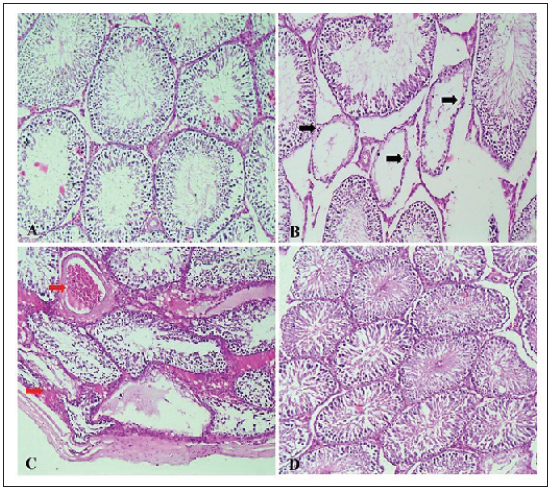
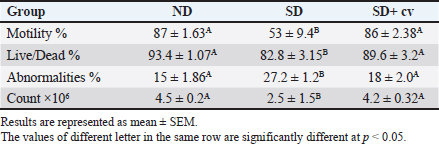
Epididymis spermatozoa profile of rats treated with CVThe current study (Table 4) showed that rats fed a SD had significantly lower sperm count, motility, and viability and an elevated percentage of sperm abnormalities compared with the control group fed a balanced diet (ND). Animals supplemented with the SD diet fortified by chlorella (SD + CV) showed all semen profile values back toward normal. These results were confirmed by the histopathological findings, where the SD group showed depletion and necrosis of spermatogonial cells (Fig. 3B) compared with the control group. The CV-supplemented group showed marked restoration of normal histoarchitecture (Fig. 3D).
Fig. 3. (A–D): Photomicrograph of testicular tissue sections stained with H&E (×100) showing normal seminiferous tubules of regular contour, spermatogenesis, and narrow intertubular spaces with Leydig cells (A) in rats of the ND group. Rats that received a SD showed severe vacuolar degeneration and necrosis of spermatogonial cells as well as desquamation of spermatogonial cells lining seminiferous tubules (black arrows) with subcapsular congestion (red arrows) (C). Testis of (SD + CV) treated rats showing mild testicular degeneration associated with marked restoration of normal histologic architecture (D). Table 4. Semen picture of rats given Chlorella vulgaris.
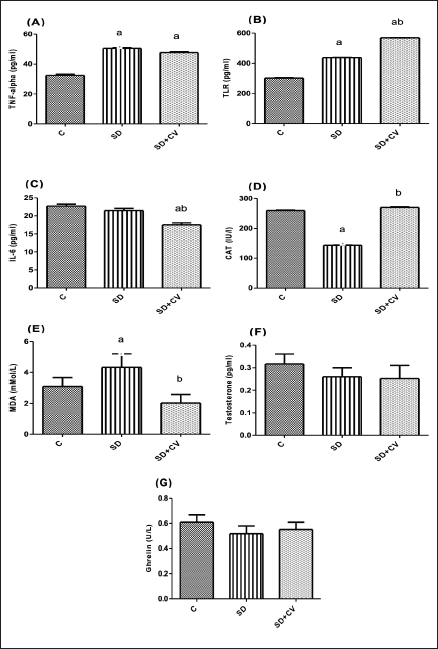
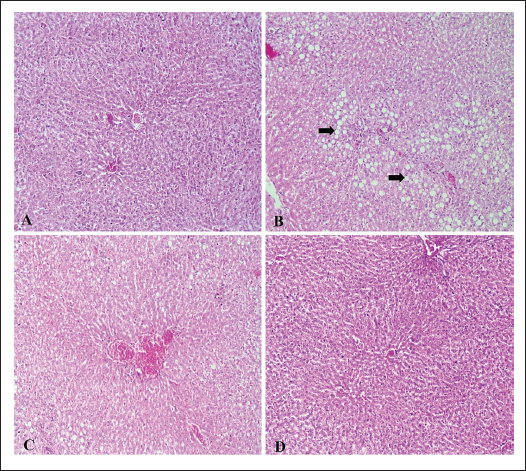
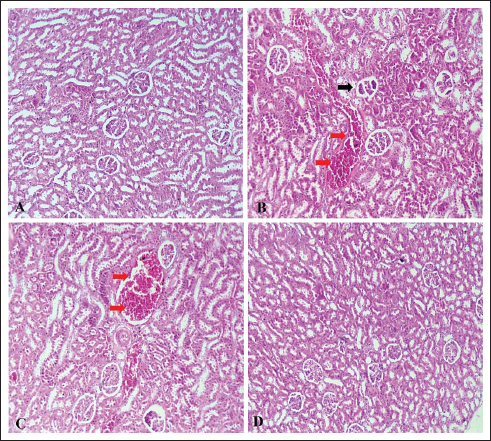
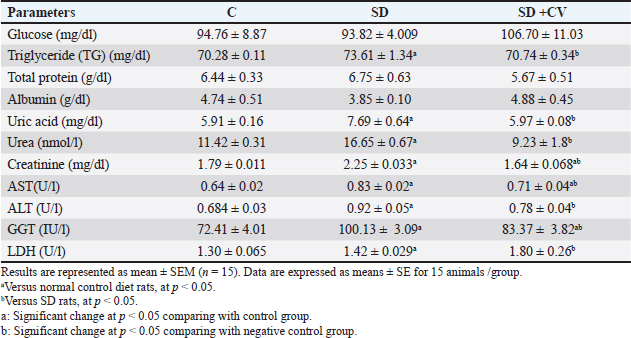
Changes in plasma chemistry in rats treated with SD and CVThe results of the current study showed nonsignificant changes in glucose, total protein, and albumin levels (Table 5). Kidney and liver function-related biomarkers (uric acid, urea, creatinine, AST, ALT, GGT), and lactate dehydrogenase (LDH) were elevated in the SD group (Table 5). This was confirmed via histopathological examination. The examined liver sections of rat (SD) showed severe vacuolar degeneration and necrosis of hepatocytes cells (Fig. 4B) (Fig. 4C). In addition, the kidney (SD group) showed severe atrophy of the glomerular tuft associated with severe congestion of blood vessels (Fig. 5B and C). The SD group showed higher serum levels of inflammatory cytokines (TNF-α, TLR, and IL-6) (Fig. 1A–C), increased serum levels of lipid peroxidation oxidative biomarker (MDA), reduced catalase activity (Fig. 1D and E), and non-significant changes in testosterone and ghrelin (Fig. 1F and G). On the other hand, CV administration recovered all normal values (Table 5 and Fig. 1).
Fig. 1. (A–G): Effect of chlorella on serum levels of TNF- (A), TLR (B), IL-6 (C), CAT (D), MDA (E), testosterone (F), and ghrelin (G). All data are presented as mean ± SEM (n=6). aVersus normal control diet rats, at p < 0.05. bVersus SD rats, at p < 0.05. One-way ANOVA was used, followed by the Tukey–Kramer multiple comparison test.
Fig. 4. (A–D): Photomicrograph of liver tissue sections stained with H&E (×100) showing no histopathological changes in the hepatic parenchyma (A) in the control and ND groups. Rats that received a SD showed severe vacuolar degeneration of hepatocytes and marked micro- and macrovasicular steatosis in all three zones (black arrows) (B), associated with severe hepatic congestion (C). Liver of (SD + CV) treated rats showing mild vacuolar degeneration associated with marked restoration of normal histologic architecture (D).
Fig. 5. (A–D): Photomicrograph of kidney tissue sections stained with H&E (×100) showing no histopathological changes in the kidney parenchyma (A) in the control and ND groups. Rats that received a SD showed severe atrophy of the glomerular tuft (black arrows) associated with severe congestion of the interstitial blood vessels (red arrows) (B, C). Kidneys of (SD + CV) treated rats showing mild vacuolar degeneration associated with marked restoration of normal histologic architecture (D). Table 5. Changes in some biochemical parameters in different treatment groups.
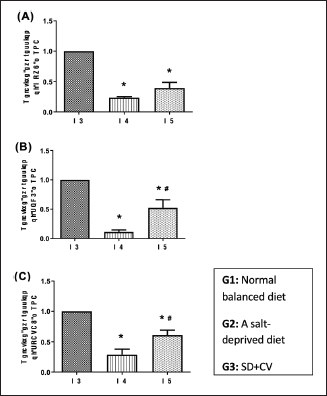
Molecular outcomesThe current study showed that the expression levels of GPX4, SOD1, and SPATA6 genes were significantly reduced in the testes of rats that received a SD compared with the ND group (p < 0.05) (Fig. 2A–C). On the other hand, the chlorella-treated group had increased levels of GPX4 and significantly increased levels of SOD1 compared with the SD group (p < 0.05), but the levels were still lower than those in the control group (Fig. 2A and B).
Fig. 2. (A–C): Effect of chlorella on the expression levels of GPX4 (A), SOD1 (B), and SPATA6 (C). Data are represented as mean ± SEM (n=6). *Versus ND rats, at p < 0.05. #Versus SD rats, at p < 0.05. One-way ANOVA was used, followed by the Tukey–Kramer multiple comparison test. Histopathological investigationTesticular changesHistopathological examination of control rats (ND) revealed normal seminiferous tubules with regular contour, spermatogenesis, and narrow intertubular spaces with normal Leydig cells (Fig. 3A). The testes of rats that received a SD showed tubular degeneration/atrophy, severe vacuolar degeneration, and depletion and necrosis of spermatogonial cells (Fig. 3B), spermatocytes, and elongated spermatid, as well as desquamation of spermatogonial cells lining of seminiferous tubules with subcapsular congestion. Some examined sections also showed intertubular edema (Fig. 3C). The volume of spermatogonial cells, spermatocytes, and Sertoli cells decreased significantly, and degenerated spermatozoa increased compared to the control group. A photomicrograph of the testis (SD + CV) showed mild testicular degeneration associated with marked restoration of the normal histopathologic architecture (Fig. 3D). LiverHistopathological examination of control rats (ND) showed no changes in the hepatic parenchyma (Fig. 4A). The examined liver sections of rat (SD) showed severe vacuolar degeneration and necrosis of hepatocytes cells, marked micro and macrovasicular steatosis in all three zones (Fig. 4B) associated with severe hepatic hemorrhages (Fig. 4C). A photomicrograph of the liver (SD + CV) showed mild vacuolar degeneration of hepatic cells associated with marked restoration of the normal histopathologic hepatic architecture (Fig. 4D). KidneyThe histopathological examination of control rats (ND) showed no histopathological changes in the kidney parenchyma (Fig. 5A). Photomicrographs of the kidney (SD) showed severe atrophy of the glomerular tuft associated with severe congestion of the blood vessels (Fig. 5B and C). Photomicrograph of the kidney (SD + CV) showed mild renal degeneration associated with marked restoration of normal histopathologic architecture (Fig. 5D). DiscussionOur study showed that Chlorella sp. microalgae improve sperm count, motility, and viability. These findings are similar to those reported by El-Nady et al. (2024). Semen formation requires a variety of minerals and traces, which deficiency leads to spermatogenesis impairment, reduced libido, and impaired male fertility (Knazicka et al., 2012). Copper is essential for sperm viability and morphology (Ismail and Ahmed, 2015), blood cell formation, bone marrow activity, and the production of testosterone and thyroid hormones (Riaz and Muhammad, 2019). Copper is essential for zinc and iron activity as metalloenzymes that regulate many biochemical processes and act as antioxidants to prevent protein degeneration and DNA fragmentation (Fadl et al., 2022). Chlorella is rich in zinc, which plays crucial roles in the development of immunological and reproduction gene expression. It also plays a critical role in sperm motility and the maintenance of their natural morphology (Sathasivam and Ki, 2018). In this instance, the green unicellular algae CV, which is frequently used as a dietary supplement rich in vitamins, minerals, and antioxidant compounds, might be a good option for salt-deficient animals. Earlier studies reported that CV has a high amount of antioxidant components, such as lutein, astaxanthin, neoxanthin, SOD, GPX, and ascorbic acid, which protect against oxidative damage of proteins, lipids, and DNA. It prevents and reduces the effects of oxidative stress on the hypothalamic–pituitary–gonadal axis and stimulates the secretion of male reproductive hormones and spermatogenesis (Barati et al., 2020). Our results were confirmed by the histopathological examination of the testis, where the SD + CV group showed mild testicular degeneration associated with marked restoration of the normal histologic architecture. Copper and zinc participate in the activity of more than 20 metallo-enzymes as cofactors and metalloproteins that are associated with the destruction of free radicals (Eva et al., 2015). SD diet is a direct cause of elevated ROS and inflammatory biomarkers, and these changes may consequently result in elevated liver enzymes. Earlier studies reported that copper deficiency elevated AST, ALT, ALP, urea, and creatinine levels (Tikoo et al., 2018). On the other hand, chlorella is a rich source of antioxidants like β-carotenoids, chlorophyll, ascorbic acid, α-tocopherol, phenolic compounds, astaxanthin, flavonoids, and trace elements (zinc, copper, and magnesium), which are crucial for the function of antioxidant metallo-enzymes (Saberi-Karimian et al., 2023; El-Nady et al., 2024). One possible explanation for the lipid-lowering benefits of CV is the high fiber content of this alga, which increases fecal fat excretion and decreases intestinal absorption of dietary fats (Shibata et al., 2001). Conversely, CV can increase the rate at which cholesterol is broken down in the liver by downregulating the enzyme cholesterol seven-alpha hydroxylase. Additionally, CV contains omega-3 and niacin, which are both utilized in TG-reducing therapies (Zuliani et al., 2009). Nevertheless, animal studies suggest that CV enhances glucose absorption and lowers NEFA levels, which may lead to a possible reduction in blood glucose levels (Alshafei et al., 2022; Sanayei et al., 2022). In addition, CV may have a role in carbohydrate metabolism (Ko et al., 2012). Our findings revealed that the serum levels of TLR4, TNF-α was significantly increased compared with the normal control, while IL-6 levels were significantly lowered (p ˂ 0.01) by the co-administration of CV. A previous study showed that chlorella polyscharride stimulated cytokines like IL-1β, TNF-α secretion in macrophages via the TLR4-mediated protein kinase (Hsu et al., 2010; Pi et al., 2025). The antioxidant and anti-inflammatory properties of CV microalga have been well documented (Ahmed et al., 2014). Some of these antioxidants include chlorophyll, carotenoids, astaxanthin, lutein, and phycobili-proteins (Eissa et al., 2020; Farag et al., 2023). Moreover, Cheng et al. (2016) observed that the protective mechanisms of CV may also be related to its anti-inflammatory activity, as it has been shown to downregulate pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6, while enhancing antioxidant defenses. More recently, El-Gendy et al. (2024) reported that CV supplementation reduced inflammatory markers in vivo, thereby confirming its role in mitigating inflammation-associated tissue injury and contributing to its overall cardioprotective action. These findings are consistent with those of Cheng et al. (2016), who demonstrated that chlorella antioxidant activities were indicated by decreased MDA concentration and increased SOD and GPX activities in chlorella-treated rats. Results showed the downregulation of SPATA genes, which are present in the testes and are essential to the processes of spermatogenesis and fertilization. This was confirmed by the histological picture of the testis of the same group, which showed severe vacuolar degeneration and necrosis of spermatogonial cells. These SPATA genes are involved in acrosome formation, mitochondrial function maintenance, sperm generation, motility, germ cell growth, testicular development, and cytoplasm transport and removal during spermiogenesis (Yuan et al., 2015; Aitken et al., 2016). Therefore, the SPATA gene family is of undisputed importance in male fertility. An earlier study demonstrated that hypermethylation in the promoter regions of SPATA4, SPATA5, and SPATA6 correlated with oligozoospermia and male infertility (Sujit et al., 2020). The current study revealed that the chlorella-treated group took the values of SPATA6 gene expression levels to normal (p < 0.05). Nevertheless, it was still lower than the SPATA6 gene level of the ND grouping. These findings indicate that chlorella has a significant effect on spermatogenesis and fertilization. ConclusionNon-encapsulated CV showed high nutritive value, antioxidant power, and higher potential to stimulate spermatogenesis-stimulating SPATA6 gene expression. Therefore, it can be used as a feed supplement to humans and animals with poor nutrition status, especially in lands with mineral deficiency and fed on zinc-deficient serials and crops. The findings suggest that CV co-administration can be a promising therapeutic agent to reduce the adverse effects of mineral deficiency on male reproductive performance. AcknowledgmentsThe entire work team would like to express their gratitude to NRC Egypt, which funded this work through internal project No 13050210. Conflict of interestThe authors declare no conflict of interest. FundingNot available Authors’ contributionsYHA: animal experiment, sampling and examination of semen, editing, and reviewing. GHE: quantitative gene expression analysis and writing–review. NAA: biochemistry analysis of blood plasma and setting of the animal experiment and writing–review. EMH: methodology, data curation, writing–review, and editing. MA: histopathological investigation and revision. WS: Examination of semen quality and chemistry. H H: preparation of microalgae microcapsules and study of their properties and nutritional value. OHE: supervision of all parts of the research work and editing. SHM: responsible for biological study. All authors have read and approved the final version of the manuscript. Data availabilityAll related data are included in the revised manuscript. ReferencesAbdi-Azar, H. and Maleki, S. 2014. Comparison of the anesthesia with thiopental sodium alone and their combination with Citrus aurantium L. (Rutaceae) essential oil in male rat. Bull. Env. Pharmacol. Life. Sci. 3, 37–44. Ahmed, F., Fanning, K., Netzel, M., Turner, W., Li, Y. and Schenk, P.M. 2014. Profiling of carotenoids and antioxidant capacity of microalgae from subtropical coastal and brackish waters. Food. Chem. 165, 300–306. Aitken, R.J., Gibb, Z., Baker, M.A., Drevet, J. and Gharagozloo, P. 2016. Causes and consequences of oxidative stress in spermatozoa. Reprod. Fertil. Dev. 28, 1–10. Alshafei, M.M., Hanafi, E.M., Fouad, M.T., Kassem, S.S., k, A. and Elbakry, H. 2022. Effect of microencapsulated spirulina platensis and bifidobacterium longum spermatogonial on metabolic syndrome of female rat model Egypt. J. Chem. 65(11), 749–759. Barati, E., Nikzad, H. and Karimian, M. 2020. Oxidative stress and male infertility: current knowledge of pathophysiology and role of antioxidant therapy in disease management. Cell. Mol. Life. Sci. 7, 93–113. Canelli, G., Tarnutzer, C., Carpine, R., Neutsch, L., Bolten, C.J., Dionisi, F. and Mathys, A. 2020. Biochemical and nutritional evaluation of chlorella and auxenochlorella biomasses relevant for food application. Front. Nutr. 7, 565996. Chen, S.R., Batool, A., Wang, Y.Q., Hao, X.X., Chang, C.H., Cheng, C.Y. and Liu, Y.X. 2016. The control of male fertility by spermatid-specific factors: searching for contraceptive targets from spermatozoon’s head to tail. Cell. Death. Dis. 7, e2472. Eissa, M.M., Ahmed, M.M., Abd Eldaim, M.A., Orabi, S.H., Elbaz, H.T., Mohamed, M.A., Elweza, A.E. and Mousa, A.A. 2020. Methanolic extract of Chlorella vulgaris protects against sodium nitrite-induced reproductive toxicity in male rats. Andrologia 52(11), e13811. El-Fouly, M.M., Morshed, A.A., Hassanien, A.H.M.A. and El-Dahshouri, M.F. 2010. Depletion of Zn, Fe, Mn and Cu from cultivated Egyptian soils by field crops. J. Food Agric. Environ. 8(2), 713–720. El-Gendy, A.E.N.G., El-Sayed, S.M., El-Desoky, M.M., El-Magd, M.A. and Abdel-Aziz, S.A. 2024. Chlorella vulgaris supplementation mitigates cardiotoxicity through modulation of oxidative stress and inflammation in rats. Life 14(10), 1289. El-Nady, I.A., ShehabEl-Din, M.I., Khalifa, W.H., Danial, E.N., Aboelmaaty, A.M., Hanafi, E.M. and Madboli, A.E. 2024. The effects of dietary supplementation of Chlorella vulgaris alga and Lactobacillus reuteri bacteria in female rabbit puberty subjected to heat stress. Egypt. JVet. Sci. 1–13. El-Said, H., El Khder, G.M., Hussein, E.M., Helmy, N.A. and Salah El Deen, M. 2019. Investigation of the effects of copper deficiency on fertility and some hematobiochemical parameters in rams with trial for treatment. Benha Vet. Med. J. 37, 210–215. Eva, T., Rohan, P., Suresh, C. and Ashok, A. 2015. Effect of iron and copper in male reproduction. J. Assist. Reprod. Genet. 32(1), 3–16. Fadl, A.M., Abdelnaby, E.A. and El–Sherbiny, H.R. 2022. Supplemental dietary zinc sulphate and folic acid combination improves testicular volume and haemodynamics, testosterone levels and semen quality in rams under heat stress conditions. Reprod. Domestic Anim. 57, 567–576. Farag, M.R., Alagawany, M., Mahdy, E.A.A., El-Hady, E., Abou-Zeid, S.M., Mawed, S.A., Azzam, M.M., Crescenzo, G. and Abo-Elmaaty, A.M.A. 2023. Benefits of Chlorella vulgaris against cadmium chloride-induced hepatic and renal toxicities via restoring the cellular redox homeostasis and modulating Nrf2 and NF-KB pathways in male rats. Biomedicines 11(9), 2414. Hanafi, E.M., Tawfeek, F.M., El Nattat, W.S., Alshafei, M.M., Kassem, S.S., Ahmed, K.A., Danial, E.N., Elbakry, H. and El-Sayed, H.S. 2022. Microencapsulated symbiotic and phytosteroids to enhance reproductive pattern in male albino rats. Adv. Anim. Vet. Sci. 10(7), 1424. Hsu, H.Y., Jeyashoke, N., Yeh, C.H., Song, Y.J., Hua, K.F. and Chao, L.K. 2010. Immunostimulatory bioactivity of algal polysaccharides from Chlorella pyrenoidosa activates macrophages via toll-like receptor 4. J. Agric. Food Chem. 58(2), 927–936. Ismail, M.H. and Ahmed, S. 2015. Evaluation of copper and zinc among hyper and hypothyroidism patients. Eur. Acad. Res. 3(5), 522–534. Khalil, W.K. and Booles, H.F. 2011. Protective role of selenium against over-expression of cancer-related apoptotic genes induced by o-cresol in rats. Arh. Hig. Rada. Toksikol. 62(2), 121–129. Khoshvaght, A., Towhidi, A., Zare-Shahneh, A., Noruozi, M., Zhandi, M., Dadashpour Davachi, N. and Karimi, R. 2016. Dietary n-3 PUFAs improve fresh and post-thaw semen quality in Holstein bulls via alteration of sperm fatty acid composition. Theriogenology 85, 807–812. Knazicka, Z., Tvrda, E. and Lukac, N. 2012. Dose and time dependent effect of copper on viability of bull spermatozoa. J. Env. Sci. Health 49, 94–100. Ko, S.C., Kim, D. and Jeon, Y.J. 2012. Protective effect of a novel antioxidative peptide purified from a marine Chlorella ellipsoidea protein against free radical-induced oxidative stress. Food. ChemToxicol. 50, 2294–2302. Livak, K.J. and Schmittgen, T.D. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4), 402–408. Ma, Y.B., Gao, M., Zhang, T.D., Chong, T., Li, H.C., Wang, Z.M. and Zhang, L.D. 2021. Oxidative stress disrupted prepubertal rat testicular development after xenotransplantation. Oxidative. Med. Cellular. Longevity. 17, 1699990. Makipour, A., Hosseinifar, S., Khazaeel, K., Tabandeh, M.R. and Jamshidian, J. 2024. Protective effect of chlorella vulgaris on testicular damage, sperm parameters, androgen production, apoptosis and oxidative stress index in male rats following doxorubicin administration. Reproductive. Toxicol. 128, 108653. Messaoudi, I., Banni, M., Saïd, L., Saïd, K. and Kerkeni, A. 2010. Involvement of selenoprotein P and GPx4 gene expression in cadmium-induced testicular pathophysiology in rat. Chem. Biol. Interact. 188(1), 94–101. Nenkova, G., Petrov, L. and Alexandrova, A. 2007. Role of trace elements for oxidative status and quality of human sperm. Balk. Med. J. 34, 343–348. Okamura, A., Kamijima, M., Shibata, E., Ohtani, K., Takagi, K., Ueyama, J., Watanabe, Y., Omura, M., Wang, H., Ichihara, G., Kondo, T. and Nakajima, T. 2005. A comprehensive evaluation of the testicular toxicity of dichlorvos in Wistar rats. Toxicology 213, 129–137. Osama, E., Galal, A.A.A., Abdalla, H. and El-Sheikh, S.M.A. 2019. Chlorella vulgaris ameliorates testicular toxicity induced by deltamethrin in male rats via modulating oxidative stress. Andrologia 51(3), e13214. Pi, Y., Yuan, Q., Qin, S., Lan, C., Nong, Q., Yun, C., Tang, H., Leng, J., Xiao, J., Zhao, L. and Zhang, L. 2025. Chlorella pyrenoidosa polysaccharide CPP-3a promotes M1 polarization of macrophages via TLR4/2-MyD88-NF-κB/p38 MAPK signaling pathways. Mar. Drugs. 23(7), 290. Riaz,, M. and Muhammad, G. 2019. Copper deficiency in ruminants in Pakistan. Matrix. Sci. Medica. 2(1), 18–20. Ribeiro, J.C., Braga, P.C., Martins, A.D., Silva, B.M., Alves, M.G. and Oliveira, P.F. 2021. Antioxidants present in reproductive tract fluids and their relevance for fertility. Antioxidants 10(9), 1441. Saberi-Karimian, M., Orooji, A., Taghizadeh, N., Ekhteraee Toosi, M.S., Ferns, G.A., Aghasizadeh, M. and Ghayour-Mobarhan, M. 2023. Curcumin’s effect on serum zinc, copper and magnesium levels in obese individuals. Avicenna. J. Phytomed. 13(3), 223–230. Sanayei, M., Kalejahi, P., Mahinkazemi, M., Fathifar, Z. and Barzegar, A. 2022. The effect of Chlorella vulgaris on obesity related metabolic disorders: a systematic review of randomized controlled trials. J. Complementary. Integr. Med. 19(4), 833–842. Sathasivam, R. and Ki, J.S. 2018. A review of the biological activities of microalgal carotenoids and their potential use in healthcare and cosmetic industries. Mar. Drugs 16(1), 26. Shibata, S., Oda, K., Onodera-Masuoka, N., Matsubara, S., Kikuchi-Hayakawa, H., Ishikawa, F., Iwabuchi, A. and Sansawa, H. 2001. Hypocholesterolemic effect of indigestible fraction of Chlorella regularis in cholesterol-fed rats. J. Nutr. Sci. Vitaminol. 47, 373–377. Sujit, K.M., Singh, V., Trivedi, S., Singh, K., Gupta, G. and Rajender, S. 2020. Increased DNA methylation in the spermatogenesis-associated (SPATA) genes correlates with infertility. Andrology 8(3), 602. Suvarna, K.S., Layton, C. and Bancroft, J.D. 2018. Bancroft’s theory and practice of histological techniques E-Book. New York: Elsevier Health Sciences. Tikoo, J., Sodan, S. and Singh, R. 2018. Hematobiochemical alterations in calves born from copper supplemented dams. Ntas. Polivet. 19(2), 222–225. Türk, G., Ateşşahin, A., Sönmez, M., Yüce, A. and Çeribaşi, A.O. 2007. Lycopene protects against cyclosporine A-induced testicular toxicity in rats. Theriogenology 67, 778–785. Tvrdá, E., Peter, S., Jana, L. and Norbert, L. 2013. Mineral nutrients and male fertility. J. Microbiol. Biotechnol. Food. Sci. 3(1), 1–14. Velu, G., Ortiz-Monasterio, I., Cakmak, I., Hao, Y. and Singh, R.P. 2014. Biofortification strategies to increase grain zinc and iron concentrations in wheat. J. Cereal Sci. 59, 365–372. Yuan, S., Stratton, C.J., Bao, J., Zheng, H., Bhetwal, B.P., Yanagimachi, R. and Yan, W. 2015. Spata6 is required for normal assembly of the sperm connecting piece and tight head-tail conjunction. Proc. Natl. Acad. Sci. 112(5), E430–E439. Zaman, Q.U., Aslam, Z., Yaseen, M., Ihsan, M.Z., Khaliq, A., Fahad, S., Bashir, S., Ramzani, P.M.A. and Naeem, M. 2017. Zinc biofortification in rice: leveraging agriculture to moderate hidden hunger in developing countries. Arch. Agronomy. Soil. Sci. 64(2), 1338–1343. Zuliani, G., Galvani, M., Leitersdorf, E., Volpato, S., Cavalieri, M. and Fellin, R. 2009. The role of polyunsaturated fatty acids (PUFA) in the treatment of dyslipidemias. Curr. Pharmaceut. Des. 15, 408–793. | ||
| How to Cite this Article |
| Pubmed Style Saber YHA, Elsayed GH, Ali NA, Hanafi EM, Ali M, El-nattat WS, Hozyne H, Ezzo OH, Mohamed SH. Chlorella vulgaris improves fertility in malnourished male rats by regulating the oxidative state, spermatogenesis, and inflammatory cytokines. Open Vet. J.. 2026; 16(1): 635-645. doi:10.5455/OVJ.2026.v16.i1.59 Web Style Saber YHA, Elsayed GH, Ali NA, Hanafi EM, Ali M, El-nattat WS, Hozyne H, Ezzo OH, Mohamed SH. Chlorella vulgaris improves fertility in malnourished male rats by regulating the oxidative state, spermatogenesis, and inflammatory cytokines. https://www.openveterinaryjournal.com/?mno=272261 [Access: February 04, 2026]. doi:10.5455/OVJ.2026.v16.i1.59 AMA (American Medical Association) Style Saber YHA, Elsayed GH, Ali NA, Hanafi EM, Ali M, El-nattat WS, Hozyne H, Ezzo OH, Mohamed SH. Chlorella vulgaris improves fertility in malnourished male rats by regulating the oxidative state, spermatogenesis, and inflammatory cytokines. Open Vet. J.. 2026; 16(1): 635-645. doi:10.5455/OVJ.2026.v16.i1.59 Vancouver/ICMJE Style Saber YHA, Elsayed GH, Ali NA, Hanafi EM, Ali M, El-nattat WS, Hozyne H, Ezzo OH, Mohamed SH. Chlorella vulgaris improves fertility in malnourished male rats by regulating the oxidative state, spermatogenesis, and inflammatory cytokines. Open Vet. J.. (2026), [cited February 04, 2026]; 16(1): 635-645. doi:10.5455/OVJ.2026.v16.i1.59 Harvard Style Saber, Y. H. A., Elsayed, . G. H., Ali, . N. A., Hanafi, . E. M., Ali, . M., El-nattat, . W. S., Hozyne, . H., Ezzo, . O. H. & Mohamed, . S. H. (2026) Chlorella vulgaris improves fertility in malnourished male rats by regulating the oxidative state, spermatogenesis, and inflammatory cytokines. Open Vet. J., 16 (1), 635-645. doi:10.5455/OVJ.2026.v16.i1.59 Turabian Style Saber, Yasser H. A., Ghada H. Elsayed, Naglaa A. Ali, Emtenan M. Hanafi, Mohamed Ali, Walid S. El-nattat, Heba Hozyne, Omima H. Ezzo, and Safaa H. Mohamed. 2026. Chlorella vulgaris improves fertility in malnourished male rats by regulating the oxidative state, spermatogenesis, and inflammatory cytokines. Open Veterinary Journal, 16 (1), 635-645. doi:10.5455/OVJ.2026.v16.i1.59 Chicago Style Saber, Yasser H. A., Ghada H. Elsayed, Naglaa A. Ali, Emtenan M. Hanafi, Mohamed Ali, Walid S. El-nattat, Heba Hozyne, Omima H. Ezzo, and Safaa H. Mohamed. "Chlorella vulgaris improves fertility in malnourished male rats by regulating the oxidative state, spermatogenesis, and inflammatory cytokines." Open Veterinary Journal 16 (2026), 635-645. doi:10.5455/OVJ.2026.v16.i1.59 MLA (The Modern Language Association) Style Saber, Yasser H. A., Ghada H. Elsayed, Naglaa A. Ali, Emtenan M. Hanafi, Mohamed Ali, Walid S. El-nattat, Heba Hozyne, Omima H. Ezzo, and Safaa H. Mohamed. "Chlorella vulgaris improves fertility in malnourished male rats by regulating the oxidative state, spermatogenesis, and inflammatory cytokines." Open Veterinary Journal 16.1 (2026), 635-645. Print. doi:10.5455/OVJ.2026.v16.i1.59 APA (American Psychological Association) Style Saber, Y. H. A., Elsayed, . G. H., Ali, . N. A., Hanafi, . E. M., Ali, . M., El-nattat, . W. S., Hozyne, . H., Ezzo, . O. H. & Mohamed, . S. H. (2026) Chlorella vulgaris improves fertility in malnourished male rats by regulating the oxidative state, spermatogenesis, and inflammatory cytokines. Open Veterinary Journal, 16 (1), 635-645. doi:10.5455/OVJ.2026.v16.i1.59 |