| Case Report | ||
Open Vet. J.. 2026; 16(1): 758-762
Open Veterinary Journal, (2026), Vol. 16(1): 758-762 Case Report Iatrogenic goitrous hypothyroidism induced by long-term trimethoprim–sulfamethoxazole therapy in a dog: A case reportAntea Ljubez*, Dajna Preldžić and Denis ČamoDepartment of Clinical Sciences, University of Sarajevo – Veterinary Faculty, Sarajevo, Bosnia and Herzegovina *Corresponding Author: Antea Ljubez. Department of Clinical Sciences, University of Sarajevo – Veterinary Faculty, Sarajevo, Bosnia and Herzegovina. Emails: antea.ljubez [at] vfs.unsa.ba Submitted: 24/07/2025 Revised: 02/12/2025 Accepted: 17/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
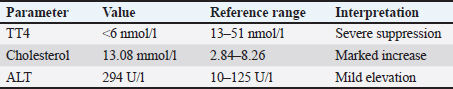
ABSTRACTBackground: Hypothyroidism is a common endocrine disorder in dogs; however, drug-induced hypothyroidism is uncommon in humans and veterinary medicine. Sulfonamides are among the few antibiotics known to interfere with thyroid function. However, reports in dogs are rare and often underrecognized. Case Description: A 12-year-old male Bichon Frise presented with alopecia and a history of slowness and weakness, weight gain despite unchanged food intake, and poor coat quality. A palpable bilateral symmetrical mass was observed in the neck area on examination. The patient had been receiving trimethoprim–sulfamethoxazole therapy for several months. Laboratory testing revealed reduced total thyroxine (TT4), consistent with iatrogenic hypothyroidism secondary to prolonged sulfonamide administration. Following drug discontinuation, TT4 values normalized within 7 days, and the dog showed marked clinical improvement, which confirmed the suspected diagnosis. Conclusion: This case highlights the potential of prolonged sulfonamide therapy to induce reversible hypothyroidism in dogs. Clinicians should be aware of this adverse effect because early recognition and drug withdrawal can lead to rapid clinical and biochemical recovery. Keywords: Dog, Hypothyroidism, Trimethoprim–sulfamethoxazole. IntroductionHypothyroidism is a common endocrine disorder in dogs that occurs when the thyroid gland produces lower amounts of tri-iodothyronine (T3) and thyroxine (T4) hormones (Dixon et al., 1999; Mooney, 2017). The most common form of primary hypothyroidism is idiopathic atrophy of the thyroid gland or lymphocytic thyroiditis (Nelson, 2014), which usually occurs in medium- to large-sized, middle-aged dogs (Dixon et al., 1999). The common clinical signs of this condition include: weight gain despite a controlled diet, lethargy, and skin and coat problems. Less commonly, clinical signs of hypothyroidism involve neurologic, cardiovascular, ophthalmic, and reproductive abnormalities (Dixon et al., 1999; Scott-Moncrieff, 2007; Mooney, 2017). The diagnosis is based on a combination of clinical signs, blood analysis (complete blood count, serum biochemistry panel) in conjunction with a decreased serum T4 and free T4 (fT4), and an increased serum thyroid-stimulating hormone (TSH) concentration (Scott-Moncrieff et al., 2015). Although hypothyroidism is one of the most common endocrine disorders in dogs, it is also often over-diagnosed (Oberbauer et al., 2025). The overdiagnosis of hypothyroidism represents a significantly greater concern than its underdiagnosis (Dixon et al., 1999). Many nonthyroidal factors, including breed, age, gender, concurrent illness, and certain drugs, can significantly affect the results of hormone measurements (Travail et al., 2024). Some medications, such as sulfonamides, can cause clinical hypothyroidism, and microscopic or even macroscopic goiter can occur (Bolton and Panciera, 2023). This case report describes a dog with sulfonamide-induced goitrous hypothyroidism. Case DetailsA 12-year-old male Bichon Frise was examined in a private veterinary clinic 6 months ago due to suspicion of urinary infection. The dog urinated frequently, sometimes uncontrollably, and blood traces were observed several times. Microscopic examination of the urine confirmed a higher number of bacteria. An antibiogram for amoxicillin and clavulanic acid was performed, and after it showed sensitivity to the aforementioned antibiotic, therapy was initiated [Amoxicillin/Clavulanic acid (12.5 mg/kg, PO, q 12 hours)]. The antibiotic was changed due to poor tolerance. Trimethoprim-sulfamethoxazole was initiated without performing a urine culture [sulfamethoxazole/trimethoprim (30 mg/kg, PO, q 12 hours)]. Treatment has been continued for the next few months due to the lack of improvement in the patient’s condition. Apart from the abovementioned symptoms, the dog showed additional signs of illness in the form of slowness and weakness, its coat became thinner, and weight gain was also observed despite consuming the same amount of food for the next few months. At reevaluation, the dog was alert and responsive but showed signs of lethargy and thinner fur. Due to the suspicion of Cushing’s disease, an adrenocorticotropic hormone stimulation test was performed. The test was negative. Abdominal ultrasound examination revealed small uroliths and a cyst on the left kidney and several cystoliths. The right adrenal gland was enlarged, which is why pheochromocytoma was suspected, and the planned surgical procedure to remove uroliths was postponed. The dog was referred to the Clinic for Internal Diseases, Oncology, and Emergency Medicine of the University of Sarajevo – Veterinary Faculty for further investigation 6 months after the first examination due to the lack of visible improvement. At clinical examination, the patient was found to be responsive. According to the WSAVA recommendations (2013), the body condition score was 6/9. The body temperature, heart rate, and respiratory rate were within the reference ranges. The fur was diffusely thinned, and a bilateral symmetrical subcutaneous mass was palpated in the region of the larynx. The remaining physical examination was unremarkable. The history and clinical examination were followed by blood sampling from the cephalic vein into an IDEXX EDTA KE/1.3 microtube for complete blood count and an L-heparin LH/1.3 microtube for heparinized plasma biochemistry panel (Chem 17 CLIP), and electrolytes (Lyte 4 CLIP). A ProCyte® Hematology Analyzer (Idexx Laboratories Inc.) was used to perform a complete blood count, and a Catalyst One® Chemistry Analyzer (Idexx Laboratories Inc.) was used to analyze the biochemistry panel and electrolytes. The complete blood count showed microcytosis, low mean corpuscular hemoglobin and reticulocyte hemoglobin concentration, and mildly elevated plateletcrit values. The biochemistry profile revealed mildly elevated alanine transaminase and gamma-glutamyl transferase levels and significantly elevated cholesterol levels [13.08 mmol/l (reference ranges: 2.84–8.26 mmol/l)] (Table 1). The abdominal ultrasonography was repeated. The presence of previously found uroliths and a cyst on the left kidney, as well as the presence of several cystoliths, was confirmed. No previously recorded changes in the adrenal gland were observed. The exact number of renal uroliths and cystoliths was confirmed by abdominal X-ray. Based on characteristic clinical signs, palpable neck mass, blood tests with a focus on cholesterol, and a history of prolonged antibiotic use, trimethoprim–sulfamethoxazole-induced goitrous hypothyroidism was suspected. A total T4 (TT4) IDEXX Catalyst Test was performed. TT4 measures both forms of thyroxine hormone, protein-bound and fT4, in a blood sample. The test showed undetectable TT4 hormone values [<6 nmol/l (reference ranges: 13–51 nmol/l)] (Table 2). The antibiotic was immediately discontinued. Urine was examined microscopically. The presence of a large number of coccoid bacteria and inflammatory cells was confirmed, and microbiological analysis was conducted. While the antibiotic-induced hypothyroidism was suspected, levothyroxine treatment was not ordinated. At reevaluation, after only 7 days, the dog was bright, alert, and responsive. Physical examination revealed that the neck masses had significantly decreased. The TT4 test was repeated, and the results were within the reference values (24 nmol/l) (Table 2). Streptococcus sp. resistant to trimethoprim–sulfamethoxazole was isolated by urine microbiological analysis. Table 1. Laboratory data table.
Table 2. Treatment timeline.
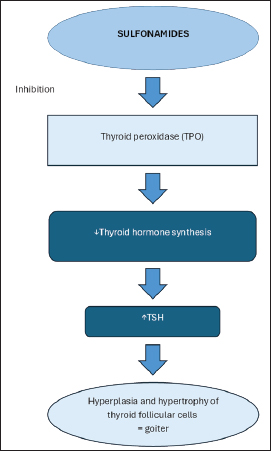
DiscussionMany drugs, including sulfonamides, can affect thyroid hormone synthesis, secretion, distribution, and metabolism (Bolton and Panciera, 2023). Sulfonamides inhibit gram-positive and gram-negative bacteria and some protozoa. Resistance and toxicity associated with sulfonamides led to their potentiation (Papich, 2018). Trimethoprim–sulfamethoxazole is a frequently used potentiated sulfonamide, which is also associated with hypothyroidism in dogs. According to Cohen et al. (1981), exclusively the sulfonamide component of the drug is responsible for the lowering of thyroid hormone levels, and the effects of trimethoprim alone are small and unlikely to cause hypothyroidism. Hypothyroidism is thought to be caused by the ability of sulfonamides to inhibit thyroid peroxidase, and as a result of this inhibition, decreased serum thyroid hormone secretion occurs (Gulikers and Panciera, 2002; Papich, 2018) (Fig. 1). The effects of sulfonamides on thyroid function vary between species and are dose- and time-dependent (Gulikers and Panciera, 2002; Anyogu et al., 2017). In contrast to the severe thyroid dysfunction induced in rats and dogs, humans show just mild decreases in thyroid function during antibiotic treatment. It was believed that the effects on the thyroid gland occur at high doses (exceeding 30 mg/kg/day) when given for a month or more (Gulikers and Panciera, 2002), which corroborates with our case report in which the dog was taking 30 mg/kg twice a day for several months. Frank et al. (2005) proved that trimethroprim–sulfamethoxazole, even in much lower doses, affects the thyroid gland function similarly to that described when given at 30 mg/kg, PO, every 12 hours after only 3 weeks of administration. Thyroid function tests in dogs treated with trimethoprim–sulfamethoxazole are indistinguishable from those in dogs with spontaneous hypothyroidism. These tests show decreased TT4 and Ft4 values in conjunction with an increase in serum TSH concentration. Recent reports have described sulfonamide-associated hypothyroidism in cats, with clinical signs and findings comparable to those in dogs. (Halman et al., 2024; Alexander et al., 2025). Normalization of thyroid function tests may occur in as few as 7 days after sulfonamide discontinuation, as was the case with the dog described here, and levothyroxine supplementation is usually not necessary during this period. Elevated values may also remain for up to 12 weeks (Gulikers and Panciera, 2002). Because of increased serum TSH values, hypertrophy and hyperplasia of thyroid follicular cells in the form of microscopic goiter can occur, and if these processes are severe, gross enlargement in the form of macroscopic goiter can develop (Gulikers and Panciera, 2002; Brenner et al., 2009; Bolton and Panciera, 2023) (Fig. 1), as in this case. Clinical signs, as well as hematological and biochemical blood parameters, may correspond to those occurring in primary hypothyroidism. 30%–40% hypothyroid dogs have mild nonregenerative anemia, approximately 75% have elevated cholesterol levels, and mildly elevated liver enzymes (Panciera, 1994; Dixon et al., 1999). In contrast to the sulfonamide component, which is responsible for lowering thyroid hormone levels, the trimethoprim component is most likely responsible for the hematologic abnormalities. The most common hematologic abnormalities associated with trimethoprim administration in humans include thrombocytopenia, leukopenia, and granulocytopenia (Frank et al., 2005). According to Frank et al. (2005), 1/6 of the dogs developed significant neutropenia after 3 weeks of trimethoprim–sulfamethoxazole administration. Both clinical signs and hypercholesterolemia, as well as mildly elevated ALT values, were present in the dog, but there were no significant hematological abnormalities. To the best of the authors’ knowledge, two more cases with clinically evident macroscopic goitrous hypothyroidism secondary to treatment with a potentiated sulfonamide have been reported, except for the dog described here: a 4-month-old Boxer that had been treated for 5 weeks with trimethoprim–sulfamethoxazole and chloramphenicol because of aspiration pneumonia (Seelig et al., 2008), and a sulfonamide-induced hypothyroid crisis in a 4-year-old Labrador Retriever that had been treated for 10 days because of a lower respiratory tract bacterial infection (Brenner et al., 2009). In the first case, apart from the presence of a bilateral symmetrical mass in the neck area, the dog showed no signs of the disease, but hypercholesterolemia and an increase in creatine kinase activity, as well as decreased TT4 values and elevated TSH values were present. Neurological signs without other more common signs of hypothyroidism were present in the second case, but hypercholesterolemia and elevated TSH values were absent. To the best of the authors’ knowledge, this is the only case of trimethoprim–sulfamethoxazole-induced goitrous hypothyroidism in an older dog with both clinical signs (lethargy, weakness, thinner coat, and weight gain despite consuming the same amount of food) and blood findings (hypercholesterolemia, mildly elevated liver enzymes, and decreased TT4), which also occur in dogs with primary hypothyroidism.
Fig. 1. Effect of sulfonamides on thyroid gland function. Sulfonamides inhibit thyroid peroxidase, resulting in reduced secretion of circulating thyroid hormones. The consequent decrease of negative feedback results in the compensatory stimulation of TSH release. Ultimately, hyperplasia and hypertrophy of thyroid follicular cells are induced, clinically manifesting as goiter. ConclusionAlthough hypothyroidism is one of the most common endocrine disorders in dogs, it continues to present a diagnostic challenge, primarily due to the frequent misinterpretation of clinical and laboratory findings. The overdiagnosis of this condition is a significantly greater clinical concern than the underdiagnosis. Unjustified treatment of dogs that are not truly hypothyroid can lead to unnecessary medication, potential side effects, and delayed identification of the actual underlying condition. Thyroid function tests and clinical signs in dogs treated with trimethoprim–sulfamethoxazole may be indistinguishable from those in dogs with spontaneous hypothyroidism. Therefore, it is essential to rule out drug-induced hypothyroidism before initiating levothyroxine therapy. For patients receiving prolonged trimethoprim–sulfamethoxazole treatment, healthcare providers should perform routine monitoring of thyroid function. AcknowledgmentsNone. Conflict of interestThe authors declare no conflict of interest. FundingThis study received no specific grant. Authors’ contributionsConcept – ALJ; Supervision – DČ; Data Collection and/or Processing – ALJ, DČ, DP; Analysis and/or Interpretation – ALJ, DČ; Literature Search – ALJ, DČ, DP; Writing Manuscript – ALJ; Critical Review – DČ, DP. Data availabilityAll data supporting this study’s findings are available within the manuscript. ReferencesAlexander, D., Cobucci, G.C., Da Motta, C.C., Cid, G.C., Belizzi, L.S., De Jesus, A.C., Souza, H.J. and Peterson, M.E. 2025. Iatrogenic primary hypothyroidism associated with sulfamethoxazole-trimethoprim treatment. J. Vet. Intern. Med. 39(2), e70045; doi:10.1111/jvim.70045 Anyogu, D.C., Shoyinka, V.S. and Ihedioha, J.I. 2017. Effects of prolonged treatment with co-trimoxazole on the thyroid gland, liver, and epididymal sperm reserve in dogs. Clin. Med. Insights. Pathol. 10, 1179555717742881; doi:10.1177/1179555717742881 Bolton, T.A. and Panciera, D.L. 2023. Influence of medications on thyroid function in dogs: an update. J. Vet. Intern. Med. 37(5), 1626–1640; doi:10.1111/jvim.16823 Brenner, K., Harkin, K. and Schermerhorn, T. 2009. Iatrogenic, sulfonamide-induced hypothyroid crisis in a Labrador Retriever. Aust. Vet. J. 87(12), 503–505; doi:10.1111/j.1751-0813.2009.00521.x Cohen, H.N., Pearson, D.W.M., Thomson, J.A., Ratcliffe, W.A. and Beastall, G.H. 1981. Trimetophrim and thyroid function. Lancet 317(8221), 676–677; doi:10.1016/S0140-6736(81)91606-8 Dixon, M., Reid, S.W.J. and Mooney, C.T. 1999. Epidemiological, clinical, haematological and biochemical characteristics of canine hypothyroidism. Vet. Rec. 23(17), 481–487; doi:10.1136/vr.145.17.481 Frank, L.A., Hnilica, K.A., May, E.R., Sargent, S.J. and Davis, J.A. 2005. Effects of sulfamethoxazole-trimethoprim on thyroid function in dogs. Am. J. Vet. Res. 66(2), 256–259; doi:10.2460/ajvr.2005.66.256 Gulikers , K.P. and Panciera, D.L. 2002. Influence of various medications on canine thyroid function. Compend. Contin. Educ. Pract. Vet. 24(7), 511–523 Halman, C., Gavaghan, B. and Korman, R. 2024. Clinical hypothyroidism in a cat associated with sulfonamide administration for the management of intracranial nocardiosis. Austral. Vet. J. 102(12), 633–637; doi:10.1111/avj.13368 Mooney, C.T., Ettinger, S.J., Feldman, E.C. and Côté, E. 2017. Canine hypothyroidism. In Textbook of veterinary internal medicine: diseases of the dog and the cat. Eds., Ettinger, S.J., Feldman, E.C. and Côté, E. St. Louis, MO: Elsevier. Nelson, R.W. 2014. Disorders of the thyroid gland. In Small animal internal medicine. Eds., Nelson, R.W. and Couto, C.G. St. Louis, MP: Mosby, Elsevier. Oberbauer, A.M., Belanger, J.M., Petroff, B.K., Brown, D.E., Wolfe, C.R. and Famula, T.R. 2025. Repeated thyroid function evaluations in the dog: a retrospective study of 8,309 dogs. Front. Vet. Sci. 12, 1653398; doi:10.3389/fvets.2025.1653398 Panciera, D.L. 1994. Hypothyroidism in dogs: 66 cases (1987-1992). J. Am. Vet. Med. Assoc. 204(5), 761–767. Papich, M.G. 2018. Sulfonamides and potentiated sulfonamides. In Veterinary pharmacology and therapeutics. Eds., Papich, M.G. and Riviere, J.E. Hoboken, NJ: Wiley-Blackwell. Scott-Moncrieff, J., Feldman, E.C., Nelson, R.W., Reusch, C.E., Scott-Moncrieff, J.C., Canine, F. and Endocrinology. 2015. Hypothyroidism. In Canine and feline endocrinology. Eds., Feldman, E.C., Nelson, R.W., Reusch, C.E. and Scott-Moncrieff, J.C. St. Louis, MO: Elsevier Saunders. Scott-Moncrieff, J.C. 2007. Clinical signs and concurrent diseases of hypothyroidism in dogs and cats. Vet. Clin. North Am. Small Anim. Pract. 37(4), 709–722; doi:10.1016/j.cvsm.2007.03.003 Seelig, D.M., Whittemore, J.C., Lappin, M.R., Myers, A.M. and Avery, P.R. 2008. Goitrous hypothyroidism associated with treatment with trimethoprim-sulfamethoxazole in a young dog. J. Am. Vet. Med. Assoc. 232(8), 1181–1185; doi:10.2460/javma.232.8.1181 Travail, V., Fernandez Sanchez, C., Costo, J.M., Valentine, N., Conroy, M., Lee, V., Bouziopoulos, D., Bateman, K., Gatehouse, E., Cruzado-Perez, J., Pollard, D., Lamb, V., Juvet, F. and Kelly, D. 2024. Assessment of the likelihood of hypothyroidism in dogs diagnosed with and treated for hypothyroidism at primary care practices: 102 cases (2016-2021). J. Vet. Intern. Med. 38(2), 931–941; doi:10.1111/jvim.16993 WSAVA Global Nutrition Committee. 2020. Body condition score. Toronto, ON, Canada: World Small Animal Veterinary Association. Available via https://wsava.org/wp-content/uploads/2020/01/Body-Condition-Score-Dog.pdf (Accessed 11 June 2025). | ||
| How to Cite this Article |
| Pubmed Style Ljubez A, Preldžić D, Čamo D. Iatrogenic goitrous hypothyroidism induced by long-term trimethoprim–sulfamethoxazole therapy in a dog: A case report. Open Vet. J.. 2026; 16(1): 758-762. doi:10.5455/OVJ.2026.v16.i1.73 Web Style Ljubez A, Preldžić D, Čamo D. Iatrogenic goitrous hypothyroidism induced by long-term trimethoprim–sulfamethoxazole therapy in a dog: A case report. https://www.openveterinaryjournal.com/?mno=273051 [Access: February 04, 2026]. doi:10.5455/OVJ.2026.v16.i1.73 AMA (American Medical Association) Style Ljubez A, Preldžić D, Čamo D. Iatrogenic goitrous hypothyroidism induced by long-term trimethoprim–sulfamethoxazole therapy in a dog: A case report. Open Vet. J.. 2026; 16(1): 758-762. doi:10.5455/OVJ.2026.v16.i1.73 Vancouver/ICMJE Style Ljubez A, Preldžić D, Čamo D. Iatrogenic goitrous hypothyroidism induced by long-term trimethoprim–sulfamethoxazole therapy in a dog: A case report. Open Vet. J.. (2026), [cited February 04, 2026]; 16(1): 758-762. doi:10.5455/OVJ.2026.v16.i1.73 Harvard Style Ljubez, A., Preldžić, . D. & Čamo, . D. (2026) Iatrogenic goitrous hypothyroidism induced by long-term trimethoprim–sulfamethoxazole therapy in a dog: A case report. Open Vet. J., 16 (1), 758-762. doi:10.5455/OVJ.2026.v16.i1.73 Turabian Style Ljubez, Antea, Dajna Preldžić, and Denis Čamo. 2026. Iatrogenic goitrous hypothyroidism induced by long-term trimethoprim–sulfamethoxazole therapy in a dog: A case report. Open Veterinary Journal, 16 (1), 758-762. doi:10.5455/OVJ.2026.v16.i1.73 Chicago Style Ljubez, Antea, Dajna Preldžić, and Denis Čamo. "Iatrogenic goitrous hypothyroidism induced by long-term trimethoprim–sulfamethoxazole therapy in a dog: A case report." Open Veterinary Journal 16 (2026), 758-762. doi:10.5455/OVJ.2026.v16.i1.73 MLA (The Modern Language Association) Style Ljubez, Antea, Dajna Preldžić, and Denis Čamo. "Iatrogenic goitrous hypothyroidism induced by long-term trimethoprim–sulfamethoxazole therapy in a dog: A case report." Open Veterinary Journal 16.1 (2026), 758-762. Print. doi:10.5455/OVJ.2026.v16.i1.73 APA (American Psychological Association) Style Ljubez, A., Preldžić, . D. & Čamo, . D. (2026) Iatrogenic goitrous hypothyroidism induced by long-term trimethoprim–sulfamethoxazole therapy in a dog: A case report. Open Veterinary Journal, 16 (1), 758-762. doi:10.5455/OVJ.2026.v16.i1.73 |