| Research Article | ||
Open Vet. J.. 2025; 15(10): 4885-4893 Open Veterinary Journal, (2025), Vol. 15(10): 4885-4893 Research Article Enhancing growth performance and innate immunity in broiler chickens through combined probiotic and herbal supplementationPavlina Hristakieva1*, Stayka Laleva1, Tsvetoslav Koynarski2, Magdalena Oblakova1, Ivan Slavov1 and Nikolay Ivanov11Agricultural Academy, Agricultural Institute, Stara Zagora, Bulgaria 2Department of Animal Husbandry, Faculty of Veterinary Medicine, Trakia University, Stara Zagora, Bulgaria *Corresponding Author: Pavlina Hristakieva. Agricultural Academy, Agricultural Institute, Stara Zagora, Bulgaria. Email: poly_31 [at] abv.bg Submitted: 07/08/2025 Revised: 03/09/2025 Accepted: 09/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: In recent years, the use of probiotics and phytogenic supplements in animal husbandry has grown significantly to improve animal health and productivity. Aim: This study aimed to evaluate the effects of dietary supplementation with a probiotic alone and in combination with three medicinal herbs—milk thistle (Silybum marianum), nettle (Urtica dioica), and dandelion (Taraxacum officinale) on broiler chicken growth performance, biochemical blood, and key innate immune parameters. Methods: An experimental, completely randomized design was conducted with 180 1-day-old Ross 308 broilers, allocated into three groups of 60 birds each with 3 replications. The feeding trial lasted from day 1 to 42 days of age. The groups were: basal diet (control), basal diet + probiotic, and basal diet + probiotic + herbs. The key parameters included body weight, blood biochemical indicators, and immune markers. Results: Probiotic and herbal supplementation significantly improved broiler growth during days 1–14 and 29–42 (p < 0.05), with the highest values observed in the group receiving the combined supplementation. The biochemical parameters, including glucose, total protein, and lipids, showed favorable changes in the supplemented groups. Immune indicators (Alternative Pathway of Complement Activation, lysozyme, and beta-lysins) were increased in the third group compared with the control. Conclusion: The combined supplementation of probiotics and herbs positively affects broiler growth and the immune system. The results support the potential of this combination as a natural alternative to antibiotics for improving poultry health and productivity. Future studies should assess the effects in larger flocks, with different additive doses, and include a comparative antibiotic group to more precisely determine their efficacy and practical applicability. Keywords: Blood biochemistry, Herbal supplementation, Immune response, Natural alternatives. IntroductionIn recent decades, the poultry industry has faced increasing pressure to reduce the use of antibiotics due to global concerns about antimicrobial resistance (AMR). This issue became particularly urgent after the European Union enforced a full ban on the use of antibiotic growth promoters in animal feed in 2006 (European Commission, 2006). Consequently, researchers have intensively explored natural alternatives capable of maintaining poultry health and productivity under intensive farming conditions. Probiotics and plant-based feed additives are among the most promising candidates. Live microorganisms that confer health benefits to the host have attracted considerable attention in poultry production. Numerous studies have demonstrated their ability to promote gut health, modulate the intestinal microbiota, enhance immune responses, and improve overall growth performance (Gadde et al., 2017; Abd El-Hack et al., 2020; Mohsin et al., 2021; Shini and Bryden, 2022; Ayana and Kamutambuko, 2024). They are increasingly viewed as a cornerstone of antibiotic-free production systems, contributing to more sustainable and ethically sound practices. Alongside probiotics, various medicinal herbs are gaining recognition for their immunomodulatory, antioxidant, and antimicrobial properties. Plants such as milk thistle (Silybum marianum L.), nettle (Urtica dioica L.), and dandelion (Taraxacum officinale L.) contain bioactive compounds that may positively influence poultry health and immune function. Silymarin, the active compound in milk thistle, is known for its hepatoprotective and antioxidant effects, which can mitigate stress-induced immunosuppression (Din et al., 2012). Nettle is rich in vitamins, flavonoids, and polyphenols that stimulate immune activity (Zare et al., 2023), whereas dandelion exhibits both anti-inflammatory and antimicrobial properties that may support gut integrity and pathogen defense (Shittu et al., 2025). Key components of the innate immune system of avians, such as the Alternative Pathway of Complement Activation (APCA), lysozyme, and beta-lysines, represent the first line of defense against invading pathogens. The complement system is activated rapidly upon infection, leading to pathogen opsonization, immune cell recruitment, and direct microbial lysis. In birds, APCA plays a crucial role against a broad spectrum of targets, including Gram-negative bacteria and virus-infected cells (Tirado et al., 2021). Lysozyme contributes to humoral immunity by breaking down bacterial cell walls, primarily targeting Gram-positive agents but also affecting some Gram-negative organisms and enveloped viruses (Abdel-Latif et al., 2017). Beta-lysines are heat-stable proteins that exert bactericidal action independent of the complement system and contribute to innate defense mechanisms and immunological homeostasis (Weinert et al., 2013). Understanding the combined effects of probiotics and phytogenic additives on innate immunity is of practical importance given the growing interest in natural alternatives to antibiotics. Although the individual effects of these substances have been investigated, few studies have examined their synergistic potential. Therefore, the present study aimed to assess the impact of a probiotic-based diet alone and in combination with three commonly used medicinal herbs, namely, milk thistle, nettle, and dandelion, on growth performance, biochemical blood, and on key innate immune parameters in broiler chickens. Specifically, we evaluated the APCA activity, serum lysozyme, and beta-lysin levels after 42 days of dietary supplementation. Materials and MethodsAnimals, feeding, and experimental designThe experiment was conducted with 180 one-day-old male broiler chickens of the ROSS hybrid raised on the floor for 42 days. Birds were divided into three groups, each consisting of three replicates of 20 chickens. Treatment was performed according to the following scheme: Group I (Control) – group fed with basal feed for broiler chickens without any supplementation. Group II (Probiotic) – group fed with basal feed and supplemented with the probiotic Zoovit LL. Group III (Probiotic and Herbs) – group fed with basal feed supplemented with probiotic Zoovit LL and a mix of dry herbs. All animals received balanced, isocaloric, and isoenergetic combined feed, according to the hybrid requirements, while ensuring optimal microclimatic parameters (Table 1). Food and water were provided ad libitum to the birds. Body weight at 1 and 42 days of age, feed consumption, and feed conversion were monitored. The body weight of broilers was determined by individual weighing at 1, 14, 28, and 42 days of age. The absolute weight gain for the period from 1 to 42 days of age was calculated based on this. Feed consumption was controlled from 1 day to 42 days of age. The feed conversion was determined based on the feed consumed and the weight gain obtained. Table 1. Composition and nutritional value of compound feeds.
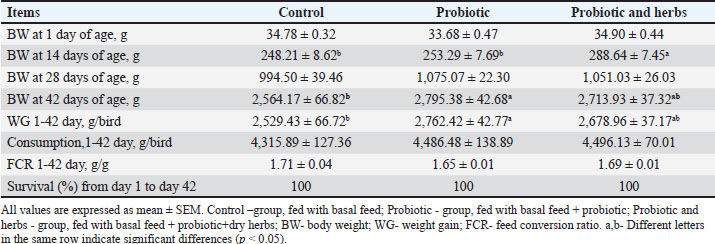
Ethical approvalThe Ethics Committee for Animal Experimentation in Sofia, Bulgaria, approved all experimental procedures. Probiotc therapyThe probiotic used in this study is called Zoovit LL and is manufactured by the “LB Lact BAS” Laboratory. The strains included in its composition are as follows: Lactobacillus delbrueckii subsp. Bulgaricus—3.5 × 10^7 CFU/ml Lactobacillus rhamnosus—2.1 × 10^7 CFU/ml Lactobacillus casei—1.8 × 10^7 CFU/ml Streptococcus thermophilus—2.9 × 10^7 CFU/ml Probiotic yeast: Kluyveromyces lactis—3.0 × 10^5 CFU/ml *CFU, colony-forming unit (viable microbes per 1 ml of solution) The probiotic product was added to the feed of the Probiotic-only group (Group II) as well as to the feed of the probiotic and herbs group (Group III) from the first day to 42nd day in a dose of 250 g/100 kg feed (0.25%) according to the manufacturer’s recommendation for broilers. Animals from all groups did not receive any antibiotics or other preparations during the experimental period. Herbal therapyEqual parts of nettle leaf (Urtica dioica L.), dandelion stem (Taraxacum officinale), and milk thistle seed (Silybum marianum) are used to represent the dry herbal mixture. This mixture of herbs is crushed in a feed mill to a particle size of 0.5 cm and is included and mixed into the combined feed of broilers of group III (probiotic and herbs) from 10 days of age in a dosage of 1 kg/100 kg feed (1%). Slaughter performanceAt the end of the experiment, after determining the final body weight of the broilers in the individual groups, 12 birds from each group were separated with an average weight for slaughter analysis. Grill weight and slaughter yield were determined. Blood collectionOn day 42 of the experiment, blood samples were obtained from the wing vein (vena cutanea ulnaris) of broiler chickens. A volume of approximately 3 ml per bird was collected using sterile Vacutainer tubes without anticoagulant. To preserve sample quality during transport, all collected tubes were immediately placed in insulated cool bags. Upon arrival at the laboratory, the samples were centrifuged at 3,000 rpm to separate the serum from the cellular fraction. The isolated serum was then stored under appropriate conditions until the initiation of analysis of innate immune parameters. Biochemical parameters of bloodSerum samples were analyzed for glucose (GLU), total protein (TP), creatinine (CREA), uric acid (UA), cholesterol (TC), triglycerides (TG), aspartate aminotransferase (AST), alkaline aminotransferase (ALT), and gamma-GT (GGT) using an Automatic Biochemical Analyzer SMT-120VP (Chengdu Seamaty Technology Co., Ltd., China) and commercial reagent. Evaluation of the alternative pathway of complement activationThe function of the alternative complement pathway was assessed using the technique described by Sotirov et al. (2021). Briefly, 100 μl of each serum sample was diluted with 350 μl of veronal-veronal sodium buffer containing 146 mM NaCl, 1.8 mM sodium salt of 5,5-diethylbarbituric acid, 3.2 mM 5,5-diethylbarbituric acid, 1 mM EGTA, and 0.8 mM MgCl2. Serial dilutions were prepared in 96-well U-bottom microplates (Flow Laboratories, UK) with ratios ranging from 8/45 to 2/45. To each well, 50 μl of buffer and 100 μl of a 1% rabbit erythrocyte suspension were added. Plates were incubated for 60 minutes at 37°C and then centrifuged at 150 g for 3 minutes at 23°C. Following centrifugation, 150 μl of the supernatant was carefully transferred to flat-bottom wells, and hemolysis was quantified by measuring absorbance at 540 nm using a “Sumal-PE2” ELISA reader (Carl Zeiss, Germany). Blood serum lysozyme activity determinationLysozyme concentration was determined based on the method of Sotirov et al. (2011), incorporating enhancements for improved sensitivity. A 2% agarose gel was prepared in phosphate buffer (0.07 M NaHPO4/NaH2PO2), and combined with a suspension of Micrococcus lутеус (24-hour culture) was added at 67°C. This mixture was poured into 14-cm Petri dishes and left to solidify at room temperature. A precision borer was then used to cut uniform 5-mm wells into the gel. Each well received 50 μl of undiluted serum. To construct a calibration curve, standard lysozyme solutions ranging from 0.025 to 3.125 μg/ml were also applied. After incubating the plates at 37°C for 20 hours, the diameters of the clear zones (indicative of lysozyme activity) were measured, and serum lysozyme concentrations were calculated using Trakia University software. Assessment of beta-lysin activityBeta-lysins were measured spectrophotometrically following the protocol by Karakolev and Nikolov (2015). The assay was conducted in 96-well plates with flat bottoms. Each reaction well received 80-μl serum and 80 μl of a Bacillus subtilis ATCC 6633 spore suspension. For negative controls, 80 μl of saline and 80 μl of the spore suspension were used. The plates were gently mixed on a microplate shaker, and the initial optical density (OD) was measured at 630 nm. Plates were incubated for 2 hours at 37°C, after which a second OD measurement was performed. Beta-lysin activity was calculated using the following formula:% lysis=[(OD1—OD2) / OD1] × 100 where OD1 represents absorbance before incubation and OD2 after incubation. As the control OD values remained unchanged, lysis was solely attributed to the presence of beta-lysins in the serum samples. Statistical analysisStatistical evaluation was performed using IBM SPSS Statistics, version 19. For the innate immune parameters, a one-way analysis of variance was conducted using the General Linear Model procedure to examine the effects of treatment groups. Results were considered statistically significant at p < 0.05. Data are presented as means ± standard error of the mean (SEM). ResultsProduction parametersAt 14 days of age, the highest average body weight was observed in the birds that received a multi-strain probiotic at a dose of 250 g/kg feed and a herbal mixture at a dose of 1 kg/kg feed, followed by the group of birds that received feed with the addition of only a probiotic at a dose of 250 g/kg feed, and the lowest body weight was observed in the birds fed a basal diet (p < 0.05). This trend was also observed at 28 days of age, and the differences between the weights of the studied groups were not proven. The addition of probiotics alone or in combination with the dry herb mixture did not significantly affect the remaining parameters of feed consumption and feed conversion for the entire experimental period. The beneficial effect of the product on the survival rate was not statistically significant because no mortality was recorded among all groups (Table 2). Table 2. Growth performance of broiler chickens.
The live weight of broilers from the groups receiving probiotic and probiotic with herbs was higher compared to the control group at p < 0.05. The carcass weight of the group receiving only probiotics in the feed (Probiotic) had the highest values compared to the control and the group receiving the dual supplementation (Probiotic and herbs). No significance was fund for the parameter comparing the live weight of the birds with the carcass percentage (Table 3). Table 3. Carcass weight and carcass rate of broilers (42days of age).
Biochemical parameters of bloodTable 4 presents the results for the serum biochemical indicators at the end of the experimental period. Except for creatinine, uric acid, aspartate aminotransferase, and gammaglutamyltransferase, statistically significant differences were found in the values of all remaining indicators. Regarding glucose, we witnessed higher levels of the indicator in the control group (14.84 mmol/l), followed by the group receiving a probiotic + herbs supplement (14.37 mmol/l) and the probiotic only group (13.62 mmol/l with (p < 0.01). In total protein, we observed a significant difference between the three groups (p < 0.01). Table 4. Biochemical blood parameters.
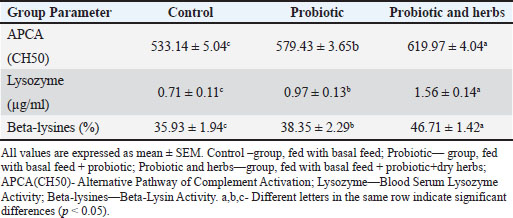
The indicator increased in the probiotic-only group by 34.70 g/l, compared with its lower concentration in the control group (33.33 g/l) and the group taking a probiotic + herbal supplement (31.48 g/l). A decrease in serum cholesterol concentration was observed in the group receiving a combination of probiotic + herbs (2.56 mmol/l) compared with the control (3.04 mmol/l) (p < 0.01). Higher serum triglyceride levels were observed in the group receiving a combination of probiotic and herbal supplement of 0.36 mmol/l compared with the probiotic-only group of 0.20 mmol/l. The differences are statistically significant (p=0.010). In our study, the transaminase enzyme alanine aminotransferase had the highest concentration in the group receiving a probiotic preparation of 11.17 U/l, followed by the control, 9.17 U/l, and the group receiving a probiotic and herbal supplement of 8.17 U/l at p < 0.01 between the probiotic group, compared to the control and probiotic + herbs. The level of aspartate aminotransferase was not significantly higher in the group receiving a combination of probiotic + herbs (260.67 U/l). The indicator has the lowest serum concentration of 219.00 U/l in the control group. A higher gammaglutamyltransferase level of 18.50 U/l was observed in the control group. The serum parameter concentration between the probiotic group and the group receiving probiotic + herbs demonstrated a minimal difference of 2.50%. Innate immune parametersTable 5 summarizes the effects of dietary supplementation on key innate immune parameters in broiler chickens. A clear enhancement of immune activity was observed in birds that received the combined supplementation of probiotic and herbal extracts. Table 5. Effects of probiotic and herbal supplementation on innate immune parameters in broiler chickens (Mean ± SEM).
The highest APCA activity was recorded in birds fed a diet enriched with both probiotics and herbs, reaching 619.97 ± 4.04 CH50. This value was highly significant (p < 0.001) compared to both the probiotic-only group (579.43 ± 3.65 CH50) and the control group (533.14 ± 5.04 CH50). While probiotic supplementation alone resulted in a statistically significant increase over the control (p < 0.001), the dual treatment elicited a much stronger immunostimulatory effect, underscoring the potential synergistic action of the herbs and probiotics. A similar trend was evident in serum lysozyme levels. The highest concentrations were again found in the group receiving both probiotic and herbal additives (1.56 ± 0.14 µg/ml), significantly surpassing those in the probiotic-only (0.97 ± 0.13 µg/ml) and control groups (0.71 ± 0.11 µg/ml). The difference between the dual-treated group and the other groups was statistically significant (p < 0.01). Notably, although probiotic supplementation slightly elevated lysozyme levels compared with the control, the difference was not statistically significant (p > 0.05), highlighting the key role of herbal components in enhancing this immune parameter. The most pronounced treatment effect was observed in the beta-lysin activity. Broilers fed the combined supplement showed significantly elevated lysine levels (46.71% ± 1.42%) compared with the probiotic-only (38.35% ± 2.29%) and control groups (35.93% ± 1.94%). The difference between the dual treatment and other groups was highly significant (p < 0.001). Interestingly, the probiotic-only group exhibited beta-lysine levels that were only marginally higher than the controls, suggesting the limited immunomodulatory capacity of the probiotic alone in this parameter. DiscussionThe results for the impact of a multi-strain probiotic-based diet alone at a dose of 250 g/kg feed and in combination with three commonly used medicinal herbs (milk thistle, nettle, and dandelion) at a dose of 1 kg/100 kg feed on broiler productivity showed statistically significant differences in broilers’ body weight. Birds from the probiotic and probiotic + herb groups had higher live weight, growth, and carcass weight at 42 days. These results are consistent with many other similar studies. Adil et al. (2025) reported that the inclusion of up to 1 g/kg of multi-strain probiotics generally yielded positive effects on several parameters in broiler breeder hens. Bai et al. (2013) reported that the application of a mixture of Limosilactobacillus fermentum and Saccharomyces cerevisiae has a positive effect on live weight and carcass yield. Many authors have reported that the addition of probiotic products has a positive effect on productive indicators in birds (Abazza et al., 2008; Midilli et al., 2008; Ignatova et al., 2009; Adil et al., 2024). Nahak et al. (2021) and Al-Sagan et al. (2020) found that the combination of probiotics and herbs in compound feeds has a synergistic positive effect on live weight and growth. In this regard, dandelion is one such phytogenic agent that has shown promising results in poultry nutrition (Al-Kassie et al., 2010; Yan et al., 2012; Qureshi et al., 2015; Qureshi et al., 2017). Qureshi et al. (2016) showed that additional dandelion leaves improve broiler growth parameters during the rearing period. In the present experiment, we found significantly lower glucose levels in the group that received a probiotic supplement compared to the control, as well as a lower concentration of triglycerides in the probiotic group compared to the one that received a probiotic and herbs supplement. Higher total protein and alanine aminotransferase concentrations in the probiotic group than in the other two groups. Serum cholesterol concentration was lower in the group receiving a combination of probiotics and herbs than in the control group. Hasibuan et al. (2019) reported an increase in productive qualities and a decrease in blood serum cholesterol levels, but did not affect crude protein levels following probiotic, javanese turmeric, and breadfruit leaf herbal extract administration in broilers. The results demonstrate that the combined dietary supplementation of a probiotic and a phytogenic blend containing milk thistle, nettle, and dandelion markedly enhances innate immune responses in broiler chickens. Improvements were observed in all parameters of interest, including – APCA, serum lysozyme activity, and beta-lysine levels, all of which are key indicators of humoral, non-specific immunity in poultry. The APCA activity enhancement observed in this trial supports earlier findings that dietary interventions can modulate complement pathways in chickens. The APCA values were significantly higher in the group receiving both the probiotic and herbs than in the control and probiotic-only groups, indicating a potential synergistic effect between the microbial and botanical components. This aligns with the work of Sotirov et al. (2021) who reported that certain feed additives can boost complement activity in poultry, thereby improving the ability of birds to combat Gram-negative bacteria and viruses (Tirado et al., 2021). Interestingly, while the probiotic alone did elevate APCA activity compared with the control, the increase was modest compared with the dual treatment. This suggests that probiotics might act primarily by stabilizing the gut microbiota and modulating mucosal immunity (Gadde et al., 2017; Abd El-Hack et al., 2020), whereas the addition of phytogenics likely contributes to the production of bioactive compounds that directly stimulate systemic immune mechanisms. For instance, silymarin from milk thistle possesses potent antioxidant and hepatoprotective properties that can modulate immune signaling (Din et al., 2012), whereas flavonoids in nettle and polyphenols in dandelion enhance macrophage activity and cytokine production (Zare et al., 2023; Shittu et al., 2025). Lysozyme activity followed a similar trend, with the highest levels detected in birds receiving the combination treatment. Lysozyme plays a critical role in early-stage immune defense by degrading bacterial peptidoglycan and lysing invading microbes (Abdel-Latif et al., 2017). Although the probiotic-only group showed a slight increase in serum lysozyme compared with controls, the difference was not statistically significant. This again highlights the key role of phytogenic components in modulating innate immunity. The current results confirm earlier observations that dietary supplementation with lysozyme-enhancing compounds improves poultry resistance to infectious agents (Abdel-Latif et al., 2017; Ayana and Kamutambuko, 2024). The most notable improvement in immune parameters was observed in beta-lysine activity, where the dual-treated group significantly outperformed the probiotic-only and control groups. Beta-lysines are cationic proteins that are released by platelets and immune cells and are known for their bactericidal activity independent of the complement system (Weinert et al., 2013). The limited response in the probiotic-only group suggests that probiotics alone may not strongly influence lysine production, whereas the bioactive compounds present in milk thistle, nettle, and dandelion may stimulate platelet or leukocyte activity, leading to increased lysine secretion. These findings explain that although probiotics alone may modestly stimulate immune function, the inclusion of phytogenics significantly amplifies these effects, likely through multiple complementary mechanisms. These results support the concept of combining microbial and botanical feed additives as a viable alternative to antibiotic growth promoters in poultry production, a strategy that aligns with EU policy (European Commission, 2006) and global efforts to curb AMR (Shini and Bryden, 2022; Obianwuna et al., 2023; Obianwuna et al., 2024). Limitations and future researchAlthough the combined probiotic and herbal supplementation showed promising effects on growth and innate immunity, the study was limited by a small sample size, single dose, and focus on innate parameters only. Future research should explore different doses, larger flocks, and additional immune, microbiota, and molecular markers to fully elucidate the mechanisms and practical potential of these natural supplements. ConclusionThe addition of probiotics alone or in combination with a mixture of herbs has a positive effect on the final live weight of broilers. In this study, no differences were found between the carcass yields of broilers receiving probiotics only and probiotics and herbs compared with the control group. Lower glucose levels and lower triglyceride concentrations in the group receiving probiotic supplementation compared to the control and probiotic and combination of herbs, respectively. Higher total protein and alanine aminotransferase concentrations in the probiotic group compared with the other two groups. Serum cholesterol concentration was lower in the group receiving a combination of probiotics and herbs than in the control group. Further studies are warranted to explore the molecular mechanisms underlying these immune enhancements and the long-term effects of such dietary strategies on disease resistance and production performance in commercial poultry operations. AcknowledgmentsThe authors would like to acknowledge the support of the technical staff at the Experimental Center for Animal Husbandry at the Agricultural Institute—Stara Zagora for the implementation of the experimental part of this project. The authors would also like to thank all their colleagues at the Agricultural Institute - Stara Zagora, for their cooperation. FundingThis study was funded by the Scientific Research Fund of the Ministry of Education, project number KP-06 H76/6. Authors’ contributionsAll authors participated in the drafting of the manuscript. PH, SL, TK, and MO participated in the study design and conception. PH, SL, TK, MO, IS, and NI performed the tests and collected the required data. The collected data were analyzed by PH, SL, TK, MO, IS, and NI. All authors have revised and approved the final version of the manuscript. Conflicts of interestThe authors declare no conflicts of interest. Data availabilityAll data supporting this study’s findings are available within the manuscript. ReferencesAbaza, I.M., Shehata, M.A., Shoieb, M.S. and Hassan, I.I. 2008. Evaluation of some natural feed additive in growing chick diets. Int. J. Poult. Sci. 7(9), 872–879; doi: 10.3923/ijps.2008.872.879. Abd El-hack, M.E., El-Saadony, M.T., Shafi, M.E., Qattan, S.Y.A., Batiha, G.E., Khafaga, A.F., Abdel-Moneim, A.M.E. and Alagawany, M. 2020. Probiotics in poultry feed: a comprehensive review. J. Anim. Physiol. Anim. Nutr. 104(6), 1835–1850; doi:10.1111/jpn.13454 Abdel-Latif, H.M.R., El-Far, A.H., Elbestawy, A.R., Ghanem, R., Mousa, S.A. and Abd El-hamid, H.S. 2017. Exogenous dietary lysozyme improves growth performance and gut microbiota in broiler chickens by targeting mRNA expression of antioxidant and non-specific immunity genes. PLos One 12(10), 185153; doi:10.1371/journal.pone.0185153 Adli, D.N., Sholikin, M.M., Sitaresmi, P.I., Ani Nurgiartiningsih, V.M. and Crooijmans, R.P.M.A. 2025. Dose-dependent effects of probiotics on the reproductive performance, egg characteristics, and seminal traits of broiler breeders: a model-based meta-analysis. J. Anim. Physiol. Anim. Nutr. (Berl). 5. 1–19; doi:https://doi.org/10.1111/jpn.14123 Adli, D.N., Sholikin, M.M., Ujilestari, T., Ahmed, B., Sadiqqua, A., Harahap, M.A., Sofyan, A. and Sugiharto, S. 2024. Effect of fermentation of herbal products on growth performance, breast meat quality, and intestinal morphology of broiler chickens: a meta-analysis. Ital. J. Anim. Sci. 23, 734–750; doi: 10.1080/1828051X.2024.2351441 Al-Kassie, G.A.M., Al-Jumaa, Y.M.F. and Jameel, Y.J. 2008. Effect of probiotic (Aspergillus niger) and prebiotic (Taraxacum officinale) on blood picture and biochemical properties of broiler chicks. Int. J. Poult. Sci. 7, 1182–1184; doi: 10.3923/ijps.2008.1182.1184 Al-Sagan, A.A., Al-Yemni, A.H., Al-Abdullatif, A.A., Attia, Y.A. and Hussein, E.O.S. 2020. Effects of Different Dietary Levels of Blue Lupine (Lupinus angustifolius) seed meal with or without probiotics on the performance, carcass criteria, immune organs, and gut morphology of broiler chickens. Front. Vet. Sci. 7, 124; doi:10.3389/fvets.2020.00124 Ayana, G.U. and Kamutambuko, R. 2024. Probiotics in disease management for sustainable poultry production. Adv. Gut Microbiome Res.1, 1–11; doi: 10.1155/2024/4326438 Bai, S.P., Wu, A.M., Ding, X.M., Lei, Y., Bai, J., Zhang, K.Y. and Chio, J.S. 2013. Effects of probiotic-supplemented diets on growth performance and intestinal immune characteristics of broiler chickens. Poult. Sci. 92, 663–670; doi:10.3382/ps.2012-02813 Din, M., Chand, N., Khan, S., Sultan, A., Mushtaq, M. and Ullah, R. 2012. Hepatoprotective Role of Milk Thistle (Silybum marianum) in Meat Type Chicken Fed Aflatoxin B1 Contaminated Feed. Pak. Vet. J. 32(3), 443–446; http://www.pvj.com.pk/pdf-files/32_3/443-446.pdf European Commission. 2006. Ban on Antibiotics as Growth Promoter in Animal Feed Enters into Effect. Regulation (EC) No 1831/2003 of the European Parliament and of the Council on Additives for Use in Animal Nutrition. Brussels, Belgium Available via https://eur-lex.europa.eu/eli/reg/2003/1831/oj/eng. Gadde, U., Kim, W.H., Oh, S.T. and Lillehoj, H.S. 2017. Alternatives to antibiotics for maximizing growth performance and feed efficiency in poultry: a review. Anim. Health Res. Rev. 18(1), 26–45; doi:10.1017/S1466252316000207 Hasibuan, A.S., Silalahi, J. and Masfria, M. 2019. The Effect of Herbal Extracts and Probiotic Feeding on Productivity and Quality of Broilers. Asian J. Pharm. Res. Dev. 7(3), 05–09; doi:10.22270/ajprd.v7i3.521 Ignatova, M., Sredkova, V. and Marasheva, V. 2009. Effect of dietary inclusion of probiotic on performance and some blood indices. Biotechnol. Anim. Husband. 25, 1079–1085. Karakolev, R. and Nikolov, D. 2015. Fluctuations in serum beta-lysine activity in laying hens depending on age, hybrid affiliation and rearing. Poult. Farm. 3, 18–19. Midilli, M., Alp, M., Kocabach, N., Muglah, O., Turan, N., Yilmaz, H. and Cakir, S. 2008. Effects of dietary probiotic and prebiotic supplementation on growth performance and serum IgG concentration of broilers. S. Afr. J. Anim. Sci. 38, 21–27. Mohsin, M., Abbas, R.Z., Yin, G., Sindhu, Z.U.D., Abbas, A., Huang, Z., Aleem, M.T., Saeed, Z., Afzal, M.Z., Ejaz, A. and Shoaib, M. 2021. Probiotics as therapeutic, antioxidant and immunomodulatory agents against poultry coccidiosis. World’s. Poult. Sci. J. 77(2), 331–345; doi:10.1080/00439339.2021.1883412 Nahak, T.E.M., Wahyuni, A.E.T.H. and Tabbu, C.R. 2021. Probiotics and herbs combination in commercial feed additives as growth promoter in broiler chicken. BIO Web of Conferences, 33, 04008; doi: 10.1051/bioconf/20213304008 Obianwuna, U.E., Agbai Kalu, N., Wang, J., Zhang, H., Qi, G., Qiu, K. and Wu, S. 2023. Recent Trends on Mitigative Effect of Probiotics on Oxidative-Stress-Induced Gut Dysfunction in Broilers under Necrotic Enteritis Challenge: a Review. Antioxidants 12(4), 911; doi:10.3390/antiox12040911 Obianwuna, U.E., Chang, X., Oleforuh-Okoleh, V.U., Onu, P.N., Zhang, H., Qiu, K. and S Wu, S. 2024. Phytobiotics in poultry: revolutionizing broiler chicken nutrition with plant-derived gut health enhancers. J. Anim. Sci. Biotechnol. 15 (1), 1–33; doi: 10.1186/s40104-024-01101-9 Qureshi, S., Adil, S., Abd El-Hack, M.E., Alagawany, M. and Farag, M.R. 2017. Beneficial uses of dandelion herb (Taraxacum officinale) in poultry nutrition. World’s Poult. Sci. J. 73, 1-11; doi: 10.1017/S0043933917000459 Qureshi, S., Banday, M.T., Adil, S., Shakeel, I. and Munshi, Z.H. 2015. Effect of dandelion leaves and fenugreek seeds with or without enzyme addition on performance and blood biochemistry of broiler chicken and evaluation of their in vitro antibacterial activity. Indian J. Anim. Sci. 85, 1248–1254. Qureshi, S., Banday, M.T., Shakeel, I., Adil, S. and Khan, A.A. 2016. Effect of raw and enzyme-treated dandelion leaves and fenugreek seed supplemented diet on gut microflora of broiler chicken. Appl. Biol. Res. 18, 76–79; doi:10.5958/0974-4517.2016.00012.4 Shini, S. and Bryden, W.L. 2022. Probiotics and gut health: linking gut homeostasis and poultry productivity. Anim. Prod. Sci. 62(13), 1090–1112; doi: 10.1071/AN20701 Shittu, R.O., Ceesay, I. and Pwavodi, P.C. 2025. Antioxidant and antimicrobial activities of dandelion root extract (Taraxacum officinale) and its cytotoxic effect on MDA-MB-231 breast cancer cells. Discov. Appl. Sci. 7, 136; doi:10.1007/s42452-024-06419-7 Sotirov, L., Denev, S., Chobanova, S., Bozakova, N., Velichkova, K., Dinev, T. and Koynarski, T. 2021. Effects of dietary marine microalgae Schizochytrium limacinum on natural humoral immunity of broiler chickens. Bulg. J. Agric. Sci. 27(1), 194–199. Sotirov, L., Koynarski, T., Semerdjiev, V., Dimov, D., Laleva, S., Slavova, P., Iliev, M. and Yarkov, D. 2011. Effect of breed upon blood lysozyme and complement activity in different sheep breeds. Agric. Sci. Technol. 3(4), 302–305. Tirado, T.C., Bavia, L., Ambrosio, A.R., Campos, M.P., De Almeida Santiago, M., Messias-Reason, I.J. and Figueiredo, F.B. 2021. A comparative approach on the activation of the three complement system pathways in different hosts of visceral leishmaniasis after stimulation with Leishmania infantum. Dev. Comp. Immunol. 120, 104061; doi:10.1016/j.dci.2021.104061 Weinert, B.T., Schölz, C., Wagner, S.A., Iesmantavicius, V., Su, D., Daniel, J.A. and Choudhary, C. 2013. Lysine succinylation is a frequently occurring modification in prokaryotes and eukaryotes and extensively overlaps with acetylation. Cell Rep. 4(5), 842–851; doi:10.1016/j.celrep.2013.07.024 Yan, L., Zhang, Z.F., Park, J.C. and Kim, I.H. 2012. Evaluation of Houttuynia cordata and Taraxacum officinale on growth performance, nutrient digestibility, blood characteristics, and fecal microbial shedding in diet for weaning pigs. Asian-Australasian J. Anim. Sci. 25, 1439–1444. Zare, M., Esmaeili, N., Paolacci, S. and Stejskal, V. 2023. Nettle (Urtica dioica) additive as a growth promoter and immune stimulator in fish. Aquac. Nutr. 8261473, 1–21; doi: 10.1155/2023/8261473 | ||
| How to Cite this Article |
| Pubmed Style Hristakieva P, Laleva S, Koynarski T, Oblakova M, Slavov I, Ivanov N. Enhancing growth performance and innate immunity in broiler chickens through combined probiotic and herbal supplementation. Open Vet. J.. 2025; 15(10): 4885-4893. doi:10.5455/OVJ.2025.v15.i10.8 Web Style Hristakieva P, Laleva S, Koynarski T, Oblakova M, Slavov I, Ivanov N. Enhancing growth performance and innate immunity in broiler chickens through combined probiotic and herbal supplementation. https://www.openveterinaryjournal.com/?mno=275957 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.8 AMA (American Medical Association) Style Hristakieva P, Laleva S, Koynarski T, Oblakova M, Slavov I, Ivanov N. Enhancing growth performance and innate immunity in broiler chickens through combined probiotic and herbal supplementation. Open Vet. J.. 2025; 15(10): 4885-4893. doi:10.5455/OVJ.2025.v15.i10.8 Vancouver/ICMJE Style Hristakieva P, Laleva S, Koynarski T, Oblakova M, Slavov I, Ivanov N. Enhancing growth performance and innate immunity in broiler chickens through combined probiotic and herbal supplementation. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4885-4893. doi:10.5455/OVJ.2025.v15.i10.8 Harvard Style Hristakieva, P., Laleva, . S., Koynarski, . T., Oblakova, . M., Slavov, . I. & Ivanov, . N. (2025) Enhancing growth performance and innate immunity in broiler chickens through combined probiotic and herbal supplementation. Open Vet. J., 15 (10), 4885-4893. doi:10.5455/OVJ.2025.v15.i10.8 Turabian Style Hristakieva, Pavlina, Stayka Laleva, Tsvetoslav Koynarski, Magdalena Oblakova, Ivan Slavov, and Nikolay Ivanov. 2025. Enhancing growth performance and innate immunity in broiler chickens through combined probiotic and herbal supplementation. Open Veterinary Journal, 15 (10), 4885-4893. doi:10.5455/OVJ.2025.v15.i10.8 Chicago Style Hristakieva, Pavlina, Stayka Laleva, Tsvetoslav Koynarski, Magdalena Oblakova, Ivan Slavov, and Nikolay Ivanov. "Enhancing growth performance and innate immunity in broiler chickens through combined probiotic and herbal supplementation." Open Veterinary Journal 15 (2025), 4885-4893. doi:10.5455/OVJ.2025.v15.i10.8 MLA (The Modern Language Association) Style Hristakieva, Pavlina, Stayka Laleva, Tsvetoslav Koynarski, Magdalena Oblakova, Ivan Slavov, and Nikolay Ivanov. "Enhancing growth performance and innate immunity in broiler chickens through combined probiotic and herbal supplementation." Open Veterinary Journal 15.10 (2025), 4885-4893. Print. doi:10.5455/OVJ.2025.v15.i10.8 APA (American Psychological Association) Style Hristakieva, P., Laleva, . S., Koynarski, . T., Oblakova, . M., Slavov, . I. & Ivanov, . N. (2025) Enhancing growth performance and innate immunity in broiler chickens through combined probiotic and herbal supplementation. Open Veterinary Journal, 15 (10), 4885-4893. doi:10.5455/OVJ.2025.v15.i10.8 |