| Research Article | ||
Open Vet. J.. 2026; 16(1): 604-615 Open Veterinary Journal, (2026), Vol. 16(1): 604-615 Research Article Influence of platelet-rich plasma and Osteon II graft on bone fracture regeneration in a rabbit modelBrwa Mohamed Ali1, Othman Jalal Ali1*, Goran Mohamad Raouf2, Salam Haji Ibrahim3 and Hana H. Mustafa11Department of Surgery and Theriogenology, College of Veterinary Medicine, University of Sulaimani, Sulaymaniyah, Iraq 2Department of Pathology and Forensic Medicine, College of Medicine, University of Sulaimani, Sulaymaniyah, Iraq 3Department of Basic Sciences, College of Veterinary Medicine, University of Sulaimani, Sulaymaniyah, Iraq *Corresponding Author: Othman Jalal Ali. Department of Surgery and Theriogenology, College of Veterinary Medicine, University of Sulaimani, Sulaimani City, Iraq. Email: othman.ali [at] univsul.edu.iq Submitted: 11/08/2025 Revised: 27/11/2025 Accepted: 09/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
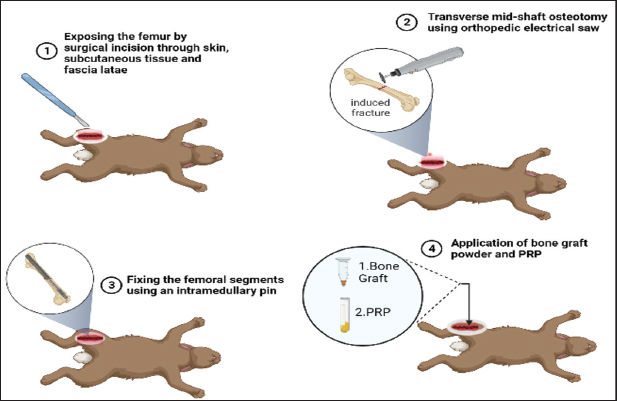
AbstractBackground: There are various cellular functions and mineralization that play a vital role in the bone healing process by ultimately restoration of the defect site to recover the original structure. Aim: This study aimed to assess the effects of autologous platelet-rich plasma (PRP) alone and with synthetic bone graft on fracture healing in experimentally induced mid-shaft transverse fracture of the femur bone in a rabbit model. Methods: A total of 60 local breed rabbits were randomly divided into three groups. Group A was treated with 0.5 ml of an autologous PRP at the fractured bone site, group B was treated with 0.025 mg synthetic bone graft and 0.5 ml PRP, and group C was treated as the control group. Samples from the fractured bone were collected at different postoperative periods for radiological and histological evaluation. Results: Radiological results showed that the rate of callus formation in rabbits treated with PRP alone and with bone graft was faster than that in the control group. Similarly, histological findings revealed that the stages of fracture healing progressed more in groups A and B than in the control group. In addition, the results of computed tomography scan imaging reinforced the results of radiological examination because the rate of callus formation was clearly enhanced in treated rabbits (Groups A and B) compared with the control group. Conclusion: Autologous PRP treatment alone or combined with synthetic bone graft, enhances the bone healing process in the rabbit model. Keywords: PRP, Fracture, Bone graft. IntroductionThere are various cellular functions and mineralization that play a vital role in the bone healing process by ultimately restoration of the defect site to recover the original structure. Femoral fracture is one of the most frequent cases in pet animals due to roadside accidents and high-velocity trauma. Intramedullary nailing is the gold standard treatment modality for diaphyseal femur fractures in pet animals (Bahney et al., 2019; Muhamad et al., 2020). Since femoral nailing provides rapid healing, functional use of the limb and predictable realignment of the bone can be achieved sooner. However, orthopedic surgeons face a major clinical challenge during the treatment of large bone fractures. To overcome these challenges and further improve the treatment success rate, several bone substitutive materials have been developed and applied clinically on their own or in co-adjuvant, which enhance the restoration of bone tissue function and osteointegration (Wang 2011; Conta et al., 2021). Activated platelets can release more than 300 molecules, which play an important role in coordination numerous cell-cell and cell-extracellular matrix integration (Filardo et al., 2015). platelet rich plasma (PRP) is a product derived from the blood, containing a high concentration of platelets that has a major impact on the healing and regeneration process of bone due to the presence of a large number of growth factors (Hall et al., 2009). A wide range of important growth factors that are involved in the process of bone healing, such as platelet-derived growth factor, transforming growth factor beta TGF-β, insulin-like growth factor-1, and epidermal growth factor (Intini, 2009). The clinical application of PRP has been reported since the beginning of the 1990s, and the first clinical benefits were initially reported in oral and maxillofacial surgeries (Redler et al., 2011). PRP is prepared by centrifugation of the blood, which results in a large concentration of platelets in a small volume of plasma. There are different methods for obtaining PRP, each one with scientific properties as to the capacity of concentration platelets and release of certain growth factors (Marx, 2001). Previous results have shown that bone grafts and PRP have a synergistic influence in acceleration of bone regeneration in animal models (Griffin et al., 2009). Although it has been shown that bone grafts play a major role in the bone healing process, this substance has its own limitations in the bone regeneration process regarding donor site morbidity and chronic pain, resulting in not always providing optimal bone healing results (Nicksic et al., 2022). Thus, this study aimed to evaluate the impact of PRP alone and in combination with an Osteon II bone graft on the healing process of experimentally induced mid-shaft femoral fractures in a rabbit model. Materials and MethodsAnimalsSixty local breed of female rabbits (Oryctolagus cuniculus), 5–7 months old, and weighing 2–2.5 kg were used in this study. The animals were housed in standard cages and maintained in a hygienic environment with free access to standard chow and drinking water in the veterinary teaching hospital belonging to the College of Veterinary Medicine, University of Sulaimani, Iraq. Statement of human and animal rightsAll the procedures and approaches of the study were conducted according to the ethics principles approved by the College of the Veterinary Medicine Research Committee, University of Sulaimani, Kurdistan Regional Government, Kurdistan, Iraq. Experimentation designThe animals were divided randomly into 3 groups, with 20 rabbits assigned to each group (n=20). 9 rabbits were selected for X-ray, and 8 rabbits for histopathological study. Furthermore, for the computed tomography (CT) scan, the remaining 9 rabbits were scanned at 30 days, 3 rabbits for each group. In group A (PRP group) (n=20): 0.5 ml of autologous PRP was applied to the site of the experimentally induced transverse fracture, followed by closure of the fascia and skin with (0/2) non-absorbable silk. In group B (PRP and bone graft group) (n=20): A synthetic prepared bone graft (0.025 mg) was applied to the site of the experimentally induced transverse fracture, followed by the application of 0.5 ml PRP. The surgical incisions were closed using non-absorbable silk (0/2). In group C (control group) (n=20): in this group, the fractured site of the experimentally induced transverse fracture was subjected to injection of 0.5 ml of normal saline, followed by closure of the fascia and skin with (0/2) non-absorbable silk. For the CT scan, three rabbits from each group (A, B, and C) were prepared for CT scanning at 30th days post the operation. PRP preparationPRP was prepared from rabbit autologous blood samples taken from the ear vein under general anesthesia. The ear of the rabbits was shaved and disinfected 3 times with ethanol (70%), and then the ear vein was punctured with a scalpel blade. From each rabbit, 1 ml of the ear-dropped blood was collected in a tube containing an anticoagulant. The collected blood was directly transferred to the laboratory for PRP preparation. PRP was prepared by two round methods. First, the blood was centrifuged at 6,000 rpm for 10 minutes. Then, the buffy coat with the plasma was aspirated into a separate tube, which was then centrifuged at 6,000 rpm for 10 minutes. Finally, the plasma was separated from the buffy coat layer and stored in a separate tube in a refrigerator for approximately 24 hours, after which it was applied to the fractured bone during the operation (Marx, 2001). Preparation of the bone graftA synthetic prepared bone graft (Osteon II) (Dentium Australia) was used, which is composed of Bi-calcium phosphate 30% coated with β-Tri-calcium phosphate 70% which increases absorption by the host cells. It is available in the form of small particles with their size of about 250 micrometers (µm). A sterilized mortar and pestle were used to grind the granules into powder. A sensitive balance was used to weight 0.025 mg for each rabbit in Group B. Surgical procedureAnesthesia was induced by an intramuscular injection of a mixture of Xylazine 2% (Interchemie werken) and Ketamine 10% (Alfasan) at doses of 0.5 and 10 mg/kg, respectively (Satheshkumar, 2005). The animals were placed on the lateral recumbency to proximally localize the opposite right hind limb proximally. The lateral aspect over the right femur was prepared for surgical operation by clipping, shaving, and surgical scrubbing with 7.5% povidone iodine (Dermosept Company). The surgical area was draped, and a surgical window was left over the femur bone for the operation. A longitudinal incision of 2–3 cm was made through the skin and subcutaneous tissue, and the fascia latae were dissected. Using a Mayo scissor, biceps femoris muscle and the vastus lateralis muscle were separated to expose the femoral shaft. A complete transverse mid-shaft osteotomy was performed using an orthopedic electrical saw (KUGEL Medical Co., Ltd.). The fractured femoral segments were routinely fixed using an intramedullary pin (Fig. 1). Finally, the muscles and fascia were approximated and sutured with simple continuous using polyglycolic acid (0/3), and the skin was sutured with simple interrupted suture using non-absorbable silk size (0/2). Postoperative care was undertaken, and enrofloxacin was injected for 5 days at a dose of 20 mg/kg body weight.
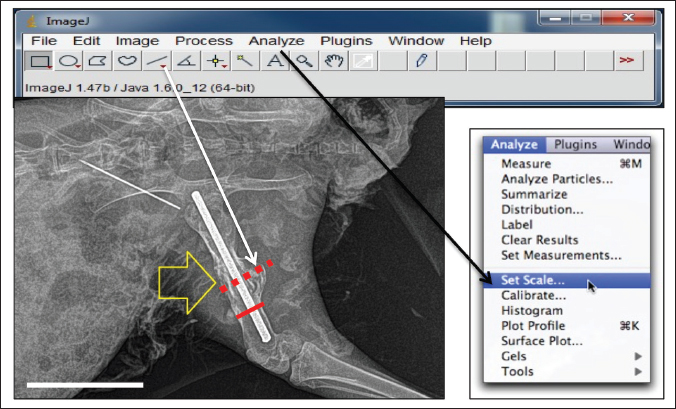
Fig. 1. The surgical steps of inducing experimental mid-shaft femoral fracture, fixing the bone fragments with an intramedullary pin, and applying PRP alone or in combination with bone graft into the fractured site (BioRender online tools). Radiographic imagingRadiographs of osteotomies were taken using a digital X-ray machine on the 10th, 20th, and 30th post-operation days. This imaging was performed to evaluate the healing process in the three groups and to compare the size of callus formation in each group. The thickness of the callus on radiographic images at 10th, 20th, and 30th days in each group. For further evaluation of the healing process, a CT scan was taken for three rabbits in each group at 30th post operation days. Three rabbits in each group were sacrificed at 30th days post-operation, and the entire femur bone of the rabbits was detached from the skin and soft tissue, such as muscle, tendons, and ligaments, and fixed in 10% Phosphate Buffered Saline-buffered formalin. These samples were transferred to Shar Hospital for CT imaging using an electron beam-type CT scan device, which assists in the complete detection of callus formation in fractured bones. ImageJ software (Version 1.49) was used to measure the degree of postoperative callus formation. Initially, the image was opened or dragged into the ImageJ software. Then, the straight-line selection tool was selected to drag a line across the calibrated ruler on the image to specify a distance (e.g., 1,000 µm). Then, the scale was set by clicking the Analyze tool, and the set scale window was opened to show the length of the dragged line in pixels (e.g., 20,000 pixels). The specified distance in pixels (20,000 pixels) determined by the freehand tool on the calibrated ruler was converted into a meter distance on the image (e.g. 1,000 µm). The callus size was measured using the straight-line tool, dragging a line over the transverse axis of the callus and the bone separately, and then calculating the callus thickness by subtracting the callus length beyond the cortex from the bone diameter at the fracture site (Fig. 2).
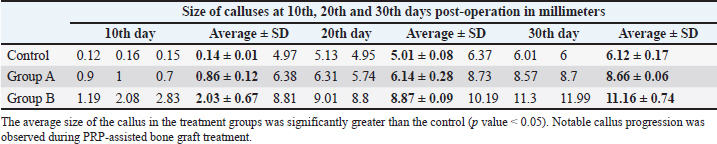
Fig. 2. Illustration of the steps required for the measurement of the callus size at the fractured site using ImageJ. After setting the scale, the straight selection tool was selected to drag a line over the bone and callus (solid red and dotted red lines, respectively). By selecting the measure tool from the Analyze tool bar, the lengths of the two lines were determined. Actual size of the callus=Diameter of the callus (at the fracture line) – Diameter of the normal bone. CT scan procedureThree rabbits from each group were euthanized on the 30th day post-operation. Then, the femur bone of the operated limb was carefully detached using a scalpel blade. The detached femurs were immersed in 10% formalin for approximately 24 hours and then transferred to the hospital for CT-scan imaging. The three femur bones of each group were arranged alongside each other within a CT scan tube, and the images were taken using an electron beam-type CT scan device (Optima company). Finally, the images were taken, and a three-dimensional format was used to assist in the observation of callus formation. Histological examinationThe femur bones were dissected from the surrounding soft tissue and fixed in 10% formalin for microscopic evaluation. Histological examination was carried out on samples of each group at 10th, 14th, 20th, and 30th post operation days for assessment of bone healing and consolidation. The samples were routinely prepared for Hematoxylin and Eosin stain (H&E). Statistical analysisGraphPad Prism (version 9.3.1) software was used for data presentation and statistical analysis. Normally distributed data (Shapiro-Wilk test) were analyzed by one-way or two-way analysis of variance (ANOVA) with Tukey’s post hoc test. Data failing the Shapiro–Wilk normality test were analyzed using Kruskal–Wallis one-way analysis of variance on ranks with a post hoc Tukey test. All results are presented as mean ± S.D, and p values of < 0.05 were considered statistically significant. Ethical approvalAll the procedures and approaches of this study were conducted and approved by the College of Veterinary Medicine Research Committee according to the principles of ethics. The data were collected and studied in accordance with other recorded retrospective clinical studies in agreement with an authorized institution. ResultsRadiological examinationObservation of radiographic assessment images at 10th, 20th, and 30th days post-operation has shown that the processes of bone healing and calluses formation were not indistinguishable between the groups, and various amounts of bone calluses were found at the fracture site. Although the calluses were less evident 10 days postoperatively, small areas of radiolucent or soft calluses were observed around the fractured lines, particularly in group B. The tissue around the fracture lines became hypertrophied and slightly radiopaque, possibly due to reorganization of the implanted bone graft. The fractured lines started to almost disappear at 20 days of the operation in the treatment groups, and the radiopacity and the number of calluses were greater than those in the control group. In addition, there was a complete formation of the hard callus around the fractured site at 30th day post the operation. However, the callus was completely radio-opaque and uniform in the treatment groups (A and B) compared with the control group. In the control group, the callus developed, but the amount and radio-opacity were lesser than in groups A and B, and the fractured line was still present. Nevertheless, the number of calluses apparently differed between groups A and B. In group B, the amount of callus and the degree of radio-opacity were slightly greater than in group A. To validate these differences, these specimens were scanned using a more precise machine (computed tomography) for complete clarification of the process of bone healing, as mentioned in the next section. ImageJ and statistical analysisAccording to our ImageJ criteria based on measuring the width and size of the calluses at the 10th, 20th, and 30th days of the operations on the radiographic images, it was found that the sizes of the calluses increased progressively in each group from 10th day to the 30th day after the operation. The average size of the callus was statistically analyzed using two-way ANOVA, with the p value < 0.05 considered statistically significant (Tables 1 and 2). Table 1. Sizes of the calluses (millimeters) in the control, PRP group) and Group B (PRP and Bone graft group) at 10th, 20th and 30th days post-operation on radiographic images using ImageJ.
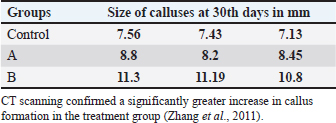
Table 2. Sizes of the calluses (mm) in the control, PRP group, and bone graft and PRP group) at 30th days post-operation on computed tomographic (CT) images.
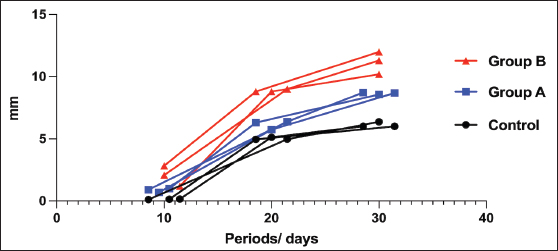
The results of this study have shown that there was a significant increase in the production of calluses in all groups except the control group. In the control group, the average degree of the callus thickness with their standard deviation on the 10th, 20th, and 30th days were 0.143 mm (±0.021 SD), 5.017 mm (±0.099 SD), and 6.127 mm (±0.211 SD), respectively. In this group, there was only a significant increase in the callus size between the 10th and 20th days of the operation, but this increase was not found from 20th to 30th day of the operation. Interestingly, a large number of calluses were produced in the treatment groups with significant differences between the different periods of the operation in groups A and B. There was a significant difference in the degree of postoperative callus formation between the treatment groups (A and B) and the control group. In group A, the average degree of the callus thickness with their standard deviation on the 10th, 20th, and 30th days were 0.867 mm (±0.153 SD), 6.143 mm (±0.351 SD), and 8.667 mm (±0.085 SD), respectively. In group B, the thickness of the callus with their standard deviation at the fractured site on the 10th, 20th, and 30th day, were 2.033 (±0.821 SD), 8.873 (±0.118 SD), and 11.16 mm (±0.908 SD), respectively. Statistically, using one-way analysis of variance, Kruskal–Wallis and Tukey’s multiple comparison tests with a p value (p < 0.05), we found a significant difference in the callus size in group B at the 10th, 20th, and 30th days compared with group A (Fig. 3).
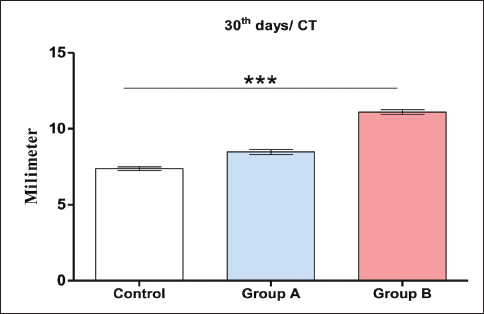
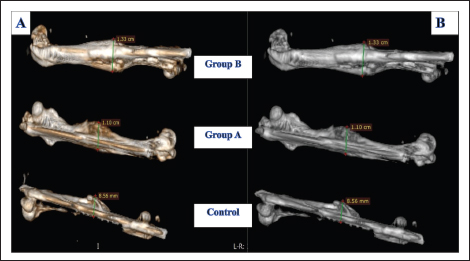
Fig. 3. Average size of calluses between the control and treatment groups (A and B) at 10th, 20th and 30th days post the operation. There were significant differences between them in which the smallest callus formation was obtained in the control group and the largest callus formation was obtained in groups A and B, using two-way RM ANOVA, Geisser-Greenhouse’s epsilon (p value < 0.05) (post-hoc Tukey test was used). Moreover, our results have clearly shown that the rate of callus formation was faster in group A than in the control group by about one-fold of the period. There were no significant differences between the callus size on the 20th day from group A (6.143 mm ± 0.351 SD) and those on 30th days in the control group (6.127 mm ± 0.211 SD). A similar situation was also observed in group B, where the callus size was larger in approximately one-fold of one period (10) days than in group A. The size of the callus in group B on the 20th day was 8.873 mm (±0.118 SD), whereas in group A and on the 30th day, it was approximately similar to that in group B on the 20th day postoperatively (8.667 mm ± 0.085 SD). Thus, in both groups, the rate of callus formation was faster than that in the control group. When groups A and B were combined, they showed much faster healing of the fractured bone than the control group and even than the individual application of these two materials (PRP and bone graft). Computed tomography (CT scan) examinationFor a complete examination of callus formation in all groups, three rabbits from each group were euthanized on the 30th day of the experiment. The CT scan images confirmed the radiographic findings. The calluses were completely developed at 30th day post the operation, but the number of calluses was greater in groups A and B than in the control group. The average widths of the calluses at 30th days post the operation in groups A, B, and the control group were 8.48, 11.09, and 7.37 mm, respectively. In groups A and B, the calluses were more uniform and extended toward the metaphysis of the bone, whereas in the control group, the callus was not uniform and did not extend toward the metaphysis of the bone as it appeared locally around the fractured line. Interestingly, the callus configuration also varied between the treatment groups. It was found that the callus is more uniform and thicker in about 20% of patients in group B (PRP and bone graft) than in group A (PRP) (Fig. 4). To confirm these data, the fractured bones were viewed using different reader’s methods, such as classic, Aligo, and thermo appearance (Fig. 5).
Fig. 4. Average size of calluses between the control and treatment groups (A and B) at 30 days postoperatively. There were significant differences between the control and B groups, with the smallest callus formation obtained in the control group and the largest callus formation obtained in groups A and B, using one-way ANOVA, Kruskal-Wallis and Tukey’s multiple comparison tests, (p value < 0.05), (Post-hoc Tukey test). (Error bars=standard deviation). GraphPad Prism (version 9.3.1).
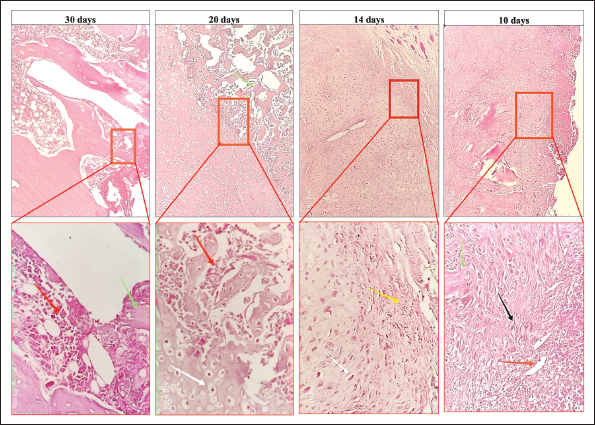
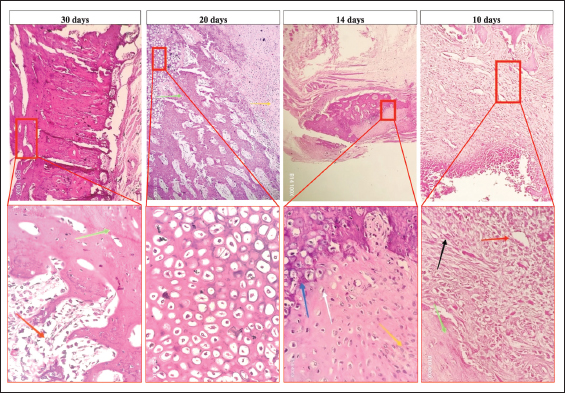
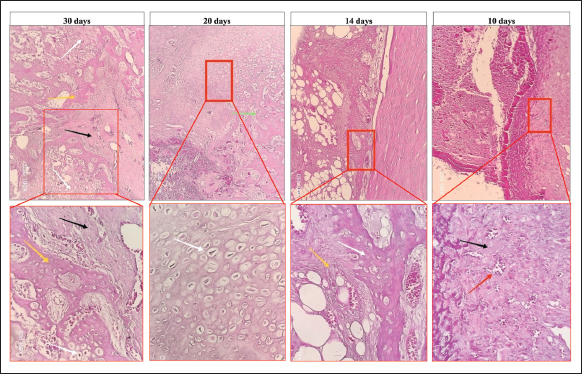
Fig. 5. CT scan images from groups A, B, and the control group 30 days after the operation. The images used by readers were Aligo (A) and the classic (B) appearance. There was variation in the amount of callus around the fractured bones in all groups, with the callus being greater in groups A and B than in the control group. Histopathological studyThe histopathological study showed that the process of fracture healing varied in groups A and B compared with the control group. Initially, on the 10th day of the operation in group A, the fractured bone showed marked diffuse dissemination of loose granulation tissue, which actively proliferated from the periosteum. Many bone trabeculae were found at the fractured site, which are considered dead bones because they are free from cells (Fig. 6). In group B, and during 10th day, it was observed a significant amount of loose granulation tissue formation at the fracture site (Fig.7), where obvious cellular bone trabeculae extended with newly formed blood vessels toward the fractured site. However, in the control group, the above-mentioned active proliferating granulomatous inflammatory process of bone healing was slower; it only revealed the proliferation of granulation tissue and the osteoblast in combination with the obvious fibrous connective tissue at the fracture site (Fig. 8).
Fig. 6. Micrograph of the healing site in group A, stained with H&E, at (×10) and (× 400) magnifications: at 10 days, the bone defect was filled with mature granulation tissue, including blood vessels (red arrow), fibroblasts (black arrow), and green arrow (lamellar bone). At 14 days, fibrocartilaginous tissues that formed a soft callus containing fibrous tissue (yellow arrow) and chondrocytes (white arrow) were observed. At 20 days, the fractured area was filled with hypertrophic cartilage without calcification (white arrow) and woven bone formation with bone marrow (red arrow). Finally, at 30 days, remodeling of the woven bone to the lamellar bone associated with the bone marrow (red arrow) and development of the lamellar bone (green arrow) were observed.
Fig. 7. Micrograph of the healing site in group B, stained with hematoxylin and eosin (H&E) at (×10) and (×400) magnifications: at 10 days, the defect was filled with mature granulation tissue, involving blood vessels (red arrow) and fibroblasts (black arrow) adjacent to the lamellar bone (green arrow). At 14 days, fibrocartilaginous tissue forms a soft callus, containing fibrous tissue (yellow arrow) and chondrocytes (white arrow), indicating the beginning of calcification (blue arrow). At 20 days, the fractured area was filled with fibrocartilaginous tissue (yellow arrow) with woven bone formation (green arrow) and hypertrophic cartilage (red box). Finally, at 30 days, there was complete remodeling of woven bone to the lamellar bone, containing bone marrow (red arrow), and the lamellar bone (green arrow).
Fig. 8. Micrograph section from the healing site in the control group, stained with H&E, at (×10) and (×400) magnifications: at 10 days, the defect was filled with mature granulation tissue including blood vessels (red arrow) and fibrous connective tissue (black arrow). At 14 days, fibrocartilaginous tissue that formed a soft callus was observed. Fibrous tissue (yellow arrow) and chondrocytes (white arrow) are shown. At 20 days, the fractured area was filled with hypertrophic cartilage without calcification (white arrow). Finally, fibrocartilaginous tissue (black arrow), woven bone without lamellar bone formation (yellow arrow), and hypertrophic cartilage (white arrow) were observed at 30 days. The photomicrographic specimen of the fractured bone at 14th day post operation has showed progressive bone regeneration. In group A, the bone trabeculae were increased and became thicker compared with those on the 10th day. Moreover, an enormous proliferation of thicker pinkish granulation tissue that extended considerably among the bone trabeculae was observed. A number of large blood vessels were found engorged with blood within the granulation tissue (Fig. 6). Meanwhile, group B showed greater proliferation of thicker collagenous granulation tissue on the 14th day, with many newly formed blood vessels within the granulation, as well as newly formed trabecular bones. A considerable amount of pinkish amorphous ground substances was deposited within these newly formed tissues. The proliferation of these substances at the fracture site indicates the early development of soft calluses that bridge fracture gaps (Fig. 7). In the control group, at 14th day post operation, it was showed only diffused proliferation of granulation tissue that extended considerably within the bone trabeculae was observed (Fig. 8). The photomicrograph at 20th day post the operation in Group A showed complete formation of the hyaline cartilage at the fracture site that extended deeply and formed a woven or trabecular bone at the fractured site. This alteration of the soft tissue into cartilaginous tissue indicated the complete formation of the hard callus around the fracture site, which was assumed to have developed from the perichondrium tissue that aids in the immobilization of the fractured segments. Moreover, an excessive amount of regeneration of hematopoietic tissue was observed within the regenerated trabecular bones (Fig. 6). However, specimens in group B showed a more advanced development of the callus than those in group A. In group B, a considerable formation of clear hyaline cartilage was observed, as indicated by a larger number of chondrocytes at the fracture site. In addition, an extensive amount of extracellular matrix was deposited within the callus, apparently between the chondrocytes (Fig. 7). However, in the control group, hyaline cartilages developed around the fracture site, which extended deeply and formed a woven or trabecular bone. The developed hyaline cartilages in the control group were apparently less dense than those in groups A and B, with a smaller number of chondrocytes, developed blood vessels, and deformed bone trabeculae (Fig. 8). The histological feature at 30th days post operation was shown a development of an obvious amount of hyaline cartilage at the fractured site. In group A, a remarkable condensed hyaline cartilage was developed, indicating a continuous callus formation at the fractured site. Moreover, the cartilaginous tissue became denser and a complete trabecular bone was established, indicating the development of hard calluses responsible for complete fracture immobilization and subsequent remodeling of the hyaline cartilage at the fracture site (Fig. 6). Group B showed significant formation of hyaline cartilage at the fracture area, which was accompanied by deposition of the extracellular tissue matrices, indicating a large amount of bone matrix deposition with a subsequent hard callus formation. In addition, there was an obvious development of woven bone with hematopoietic tissue. In this group, there was apparent advanced bone healing than in group A because of the presence of more extracellular tissue matrices and more condensed cellular elements (Fig. 7). Interestingly, when group A and B compared to the control group at 30th day, there was obvious variation in the development of the cellular and tissue matrices. In the control group, a small amount of less condensed hyaline cartilage and clear woven bone with their hematopoietic tissue were developed. Bone matrices and cellular elements were less than those in the treatment groups (Fig. 8). DiscussionIn his study, an autologous PRP alone and in combination with synthetic bone graft was applied to experimentally induced femoral fracture in the rabbit model to enhance the process of fracture healing. Currently, different materials are applied to the fractured bones in different animal models, and PRP is among the most applicable materials because of its safety, minimal invasiveness during application, and enhanced wound healing (Singh et al., 2017). PRP enhances bone regeneration through osteoblast proliferation owing to the presence of a high level of growth factors. A single dose of autologous PRP was applied to the fractured bone alone and combined with a synthetic bone graft after inducing experimentally induced fractures at the femoral mid shaft. During the postoperative observation, it was found that the animals of the treatment groups showed proper locomotion compared with the control group. The gross anatomical configuration of the fractured bones showed the production of tougher and uniform calluses in the treatment groups than in the control group. Similarly, in the radiological study, a significant difference in callus formation was observed between the two treatment groups and the control group. In this study, imagej was used to calculate the amount of callus formation on radiographic images at different periods. Interestingly, the size of the calluses varied between the treatment and control groups. A single dose of autologous PRP showed a great enhancement of callus formation in the first week of fracture healing. The sizes of calluses at 10th days post the operation were 80% and 57% greater in groups A and B, respectively, than in the control group. It was found that the callus is more uniform and thicker in approximately 20% of the lambs in group B (PRP and bone graft) than in group A PRP. Similarly, in a study on 20 lambs aged 4 months who were undergoing osteotomy for the femur bone, PRP was applied to the fractured site at the 1st, 10th, and 20th days postoperatively. The results showed no significant differences between the treatment PRP and control groups (Hernandez-Fernandez et al.,2013). Therefore, the application of a single dose of PRP at the beginning of the operation appeared to substantially affect the healing process. Although we noticed that this enhancement was continued and the size of the calluses was greater at 20th and 30th days postoperatively in groups A and B, they were approximately 18% and 30% at 20 days and 29% and 22% at 30 days in groups A and B, respectively. In a study on 40 New Zealand rabbits that underwent tibial reconstruction using bone autograft alone, bone allograft alone, and in combination with PRP for 24 weeks, the following results were obtained: It was found that when PRP was combined with the bone grafts, it significantly increased the process of bone resorption and acceleration of bone union (Nather et al., 2012). Similarly, in this study, all the animals were observed for about 4 weeks and were radiographed at 10th, 20th, and 30th post the operations, where they showed a significant difference in the process of bone healing in all stages of callus formation in groups A and B when compared to the control group. In another study on 20 New Zealand rabbits, both the radiological and histological effects of PRP were found to significantly enhance the processes of fracture healing in the duration of only 1 month; however, this enhancement was not continued in the second month postoperatively (Molina-Miñano et al., 2009). Therefore, different animal species showed different results, which might be due to the species of the animals, sources of PRP, and the optimized dose of PRP group (Hernandez-Fernandez et al., 2013). In this study, a rabbit model with an autologous PRP showed a remarkable process of bone healing compared with the control. Nevertheless, the effect of PRP becomes more obvious over time, contradicting the fact that growth factor release from the platelets only occurs in the first few days of their application. The initial effect of the growth factors may just make a difference between acceptance of bone graft and revascularization, or may be incomplete revascularization makes the bone graft encapsulated and lead to gradual loss of bone graft (Bahney et al., 2019). However, in some analyzed studies, the beneficial effect of PRP on bone regeneration could not be demonstrated in combination with every bone substitution material and in every animal model. For instance, in goats, using an autogenous cancellous bone with the addition of PRP did not improve bone healing of the critical size on the forehead-bone defect (Fennis et al., 2004; Mooren et al., 2007). Moreover, the addition of PRP to β-tricalcium phosphate in a pig model did not improve bone healing on anterior spinal fusion (Wiltfang et al., 2004). In addition, the addition of PRP to bovine cancellous blocks did not have a positive effect in a rat mandibular defect (Ali and Ali, 2019). However, PRP in combination with cancellous blocks showed a negative effect when applied around the titanium implants in the tibial defect (You et al., 2007) and in dental implants (De Vasconcelos Gurgel et al., 2007) in a dog model (Grand et al., 2009). To confirm these data, the fractured bones were viewed by CT scanning using different reader’s methods, such as classic, Aligo, and thermo appearance. CT scans were taken at 30th days post operation because at this period the calluses were completely developed in all groups and apparently without biases. The results showed that the callus formation rate in rabbits treated with PRP (Group A) and those treated with bone graft and PRP (Group B) was significantly greater than that in the control group. Similarly, in a study on 32 mini-pigs, the effect of PRP in combination with autologous bone marrow and grafts on the surgically induced metaphyseal bone defect was evaluated. The results of radiological and CT scans 6 weeks postoperatively showed a significant amount of bone formation in the animals treated with bone marrow mixed with calcium phosphate and PRP compared with those treated with bone marrow mixed with calcium phosphate and the pigs treated with calcium phosphate only (Malhotra et al., 2015). Interestingly, we used ImageJ to measure the size of callus formation at the fracture site on both the radiographic and CT images. On the CT images, we found that the sizes of the calluses on the 30th day postoperatively in groups A and B were approximately 13% and 33%, respectively, greater than those in the control group. Furthermore, a micro-CT scan was used to measure newly formed bone in rabbits treated with PRP and vascular endothelial growth factor (VEGF). They found that the animals treated with PRP developed a higher number of calluses than the control and even those treated with VEGF (Kasten et al., 2012). In addition to other studies using micro CT on rabbit models, they confirmed that the application of PRP alone and in combination with bioactive borate glass (BG) resulted in better bone formation compared with the control group and the group treated with BG scaffold alone (Zhang et al., 2011). In the current study, it was found that the callus configuration also varied between the treatment groups. It was found that the callus is more uniform and thicker in about 20% of patients in group B (PRP and bone graft) than in group A PRP. Thus, we confirmed that PRP improved bone healing in the rabbit model and that PRP, when combined with bone grafts, was more effective for critical bone repair. The histological findings further confirmed the significant difference between groups A and B and the control group, where the application of PRP alone and with synthetic bone graft enhanced the process of bone healing. Similarly, other studies have also confirmed similar findings (Schneppendahl et al., 2015). In a study on 20 adult male New Zealand rabbits, the tibia was partially resected, followed by the application of PRP in combination with β-tricalcium in the first group and aspirated bone marrow in combination with β-tricalcium in the second group. After 4 weeks, the rabbits were euthanized, and the tibia were evaluated using digital radiography and histomorphometry. The results showed that the PRP provided a greater amount of bone consolidation than bone marrow at the fractured site (Batista et al., 2011). In this study, we also confirmed that the process of fracture healing was faster in the treatment groups than in the control group. In the treatment groups, the photomicrograph showed the production of the granulation tissue in advanced with their fibroplasia and cellular differentiation at the first week postoperatively. Interestingly, during the 20th and 30th days postoperatively, this histological upregulation was continued, where characterized by the production of a remarkable condensed hyaline cartilage as it indicates a continuous callus formation at the fractured site. Moreover, the cartilaginous tissue became denser, and a complete trabecular bone was completely established and formed hard calluses that were responsible for complete fracture immobilization with a subsequent remodeling of the hyaline cartilage at the fracture site. Experimental study of tibial fracture in 32 mini-pigs, they found that using bone marrow along with PRP can be used as an alternative to an autograft (Hakimi et al., 2014). However, in a few different studies, it was recorded that neither calcium phosphate alone nor with PRP produced any effect on bone formation in the dogs’ models (Rabillard et al., 2009). Nevertheless, in this study, the results were interestingly different; the stages of healing were faster in groups A and B than in the control group. ConclusionThe application of a single dose of autologous PRP alone and in combination with synthetic bone graft enhanced the process of callus formation at the site of fractured bone, and the fractured lines disappeared earlier than the normal period of fracture healing. AcknowledgmentThe authors would like to thank the veterinary teaching hospital of the College of Veterinary Medicine, Sulaimani University, for their support in housing and managing the animals during the study period. Conflict of interestThe authors have no conflicts of interest to disclose. FundingThe University of Sulaimani provided the funds for this study. Authors’ contributionEra M. Ali was involved in performing this experimental study as part of fulfillment for obtaining the degree of master of science in veterinary surgery. Ali contributed as a main supervisor of this M Sc degree, he designed, supervised, and followed the project. Goran M. R. contributed to the preparation and reading of the histopathological section. Finally, Salam H. I. contributed to the paper’s design, proofreading, and statistical analysis. Data availabilityAll data were provided in the manuscript. ReferencesAli, B.M. and Ali, O.J. 2019. The effects of autologous platelet-rich plasma on the bone fracture healing in rabbits. Al-Anbar J. Vet. Sci. 12(2), 109–121; doi: 10.37940/AJVS.2019.12.2.14 Bahney, C.S., Zondervan, R.L., Allison, P., Theologis, A., Ashley, J.W., Ahn, J. and Hankenson, K.D. 2019. The cellular biology of fracture healing. J. Orthopedic. Res. 37(1), 35–50. Batista, M.A., Leivas, T.P., Rodrigues, C.J., Arenas, G.C.F., Belitardo, D.R. and Guarniero, R. 2011. Comparison between the effects of platelet-rich plasma and bone marrow concentrate on defect consolidation in the rabbit tibia. Clinics 66, 1787–1792. Conta, G., Libanori, A., Tat, T., Chen, G. and Chen, J. 2021. Triboelectric nanogenerators for therapeutic electrical stimulation. Adv. Mater. 33(26), 2007502. De Vasconcelos Gurgel, B.C., Gonçalves, P.F., Pimentel, S.P., Ambrosano, G.M.B., Nociti Júnior, F.H., Sallum, E.A. and Casati, M.Z. 2007. Platelet‐rich plasma may not provide any additional effect when associated with guided bone regeneration around dental implants in dogs. Clin. Oral Implants. Res. 18, 649–654. Fennis, J.P.M., Stoelinga, P.J.W. and Jansen, J.A. 2004. Mandibular reconstruction: a histological and histomorphometric study on the use of autogenous scaffolds, particulate cortico-cancellous bone grafts and platelet rich plasma in goats. Int. J. Oral. Maxillofacial. Surg. 33, 48–55. Filardo, G., Matteo, B.D., Martino, A.D., Merli, M.L., Cenacchi, A., Fornasari, P., Marcacci, M. and Kon, E. 2015. Platelet-rich plasma intra-articular knee injections show no superiority versus viscosupplementation: a randomized controlled trial. Am. J. Sports. Med. 43, 1575–1582; doi:10.1016/j.ajsm.2015.07.010 Grand, J.G., Dalibert, E., Fellah, B., Gauthier, O., Niebauer, G.W. and Rabillard, M. 2009. Effects of autologous platelet rich plasma gel and calcium phosphate biomaterials on bone healing in an ulnar ostectomy model in dogs. Vet. Comparative. Orthopedics. Traumatol. 22, 460–466. Griffin, X.L., Smith, C.M. and Costa, M.L. 2009. The clinical use of platelet-rich plasma in the promotion of bone healing: a systematic review. Injury 40, 158–162. Hakimi, M., Grassmann, J.P., Betsch, M., Schneppendahl, J., Gehrmann, S., Hakimi, A.R., Kröpil, P., Sager, M., Herten, M., Wild, M., Windolf, J. and Jungbluth, P. 2014. The composite of bone marrow concentrate and PRP as an alternative to autologous bone grafting. PLos One. 9, e100143. Hall, M.P., Band, P.A., Meislin, R.J., Jazrawi, L.M. and Cardone, D.A. 2009. Platelet-rich plasma: current concepts and application in sports medicine. Journal. Am. Acad. Orthopedic. Surgeons 17, 602–608. Hernandez-Fernandez, A., Vélez, R., Soldado, F., Saenz-Ríos, J.C., Barber, I. and Aguirre-Canyadell, M. 2013. Effect of administration of platelet-rich plasma in early phases of distraction osteogenesis: an experimental study in an ovine femur model. Injury 44, 901–907. Intini, G. 2009. The use of platelet-rich plasma in bone reconstruction therapy. Biomaterials 30, 4956–4966. Kasten, P., Beverungen, M., Lorenz, H., Wieland, J., Fehr, M. and Geiger, F. 2012. Comparison of platelet-rich plasma and VEGF-transfected mesenchymal stem cells on vascularization and bone formation in a critical-size bone defect. Cells. Tissues. Organs. 196, 523–533. Malhotra, R., Kumar, V., Garg, B., Singh, R., Jain, V., Coshic, P. and Chatterjee, K. 2015. Role of autologous platelet-rich plasma in treatment of long-bone nonunions: a prospective study. Musculoskelet. Surg. 99, 243–248. Marx, R.E. 2001. Platelet-rich plasma (PRP): what is PRP and what is not PRP?. Implant Dent. 10, 225–228. Molina-Miñano, F., López-Jornet, P., Camacho-Alonso, F. and Vicente-Ortega, V. 2009. Plasma rich in growth factors and bone formation: a radiological and histomorphometric study in New Zealand rabbits. Braz. Oral Res. 23, 275–280. Mooren, R.E.C.M., Merkx, M.A.W., Bronkhorst, E.M., Jansen, J.A. and Stoelinga, P.J.W. 2007. The effect of platelet-rich plasma on early and late bone healing: an experimental study in goats. Int. J. Oral. Maxillofacial. Surg. 36, 626–631. Muhamad, S., Ali, O., Abbas, B., Marif, H., Sleman, R., Ali, B. and Ali, G. 2020. A retrospective study of 181 fracture cases managed in a veterinary teaching hospital; 181 cases (2014–2018). Iraqi. J. Vet. Sci. 35, 23–31; doi:10.33899/ijvs.2020.126228.1266 Nather, A., Wong, K.L., David, V. and Pereira, B.P. 2012. Allografts with autogenous platelet-rich plasma for tibial defect reconstruction: a rabbit study. J. Orthopedic Surg. 20, 375–380. Nicksic, P.J., Donnelly, D.A.T., Hesse, M., Bedi, S., Verma, N., Seitz, A.J. and Poore, S.O. 2022. Electronic bone growth stimulators for osteogenesis augmentation in vitro and in vivo models: a narrative review of electrical stimulation mechanisms and device specifications. Front. Bioengineering. Biotechnol. 10, 793945. Rabillard, M., Grand, J.G., Dalibert, E., Fellah, B., Gauthier, O. and Niebauer, G.W. 2009. Effects of autologous platelet rich plasma gel and calcium phosphate biomaterials on bone healing in an ulnar ostectomy model in dogs. Vet. Comp. Orthop. Traumatol. 22, 460–466. Redler, L., Thompson, S., Hsu, S., Ahmad, C. and Levine, W. 2011. Platelet-rich plasma therapy: a systematic literature review and evidence for clinical use. Physician Sportsmedicine. 39, 42–51. Schneppendahl, J., Lögters, T.T., Sager, M., Wild, M., Hakimi, M., Windolf, J., Grassmann, J.P. and Jungbluth, P. 2015. Treatment of a diaphyseal long-bone defect with autologous bone grafts and platelet-rich plasma in a rabbit model. Vet. Comp. Orthop. Traumatol. 28, 164–171. Singh, R., Rohilla, R., Gawande, J. and Kumar Sehgal, P. 2017. To evaluate the role of platelet-rich plasma in healing of acute diaphyseal fractures of the femur. Chin. J. Traumatol. 20, 39–44. Satheshkumar, S.P. 2005. Ketamine-Xylazine anesthesia in rabbits. Indian Vet. J. 82(4), 388–389. You, T.M., Choi, B.H., Li, J., Jung, J.H., Lee, H.J., Lee, S.H. and Jeong, S.M. 2007. The effect of platelet-rich plasma on bone healing around implants placed in bone defects treated with Bio-Oss: a pilot study in the dog tibia. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 103, e8–e12. Wang, J. 2011. The End of the Revolution: china and the Limits of Modernity. Los Angeles, CA: SAGE Publications. Wiltfang, J., Kloss, F.R., Kessler, P., Nkenke, E., Schultze‐Mosgau, S., Zimmermann, R. and Schlegel, K.A. 2004. Effects of platelet‐rich plasma on bone healing in combination with autogenous bone and bone substitutes in critical‐size defects: an animal experiment. Clin. Oral Implants Res. 15, 187–193. Zhang, Y.D., Wang, G., Sun, Y. and Zhang, C.Q. 2011. Combination of platelet-rich plasma with degradable bioactive borate glass for segmental bone defect repair. Acta. Orthopaedica. Belgica. 77, 110. | ||
| How to Cite this Article |
| Pubmed Style Ali BM, Ali OJ, Raouf GM, Ibrahim SH, Mustafa HH. Influence of platelet-rich plasma and Osteon II graft on bone fracture regeneration in a rabbit model. doi:10.5455/OVJ.2026.v16.i1.56 Web Style Ali BM, Ali OJ, Raouf GM, Ibrahim SH, Mustafa HH. Influence of platelet-rich plasma and Osteon II graft on bone fracture regeneration in a rabbit model. https://www.openveterinaryjournal.com/?mno=276698 [Access: February 04, 2026]. doi:10.5455/OVJ.2026.v16.i1.56 AMA (American Medical Association) Style Ali BM, Ali OJ, Raouf GM, Ibrahim SH, Mustafa HH. Influence of platelet-rich plasma and Osteon II graft on bone fracture regeneration in a rabbit model. doi:10.5455/OVJ.2026.v16.i1.56 Vancouver/ICMJE Style Ali BM, Ali OJ, Raouf GM, Ibrahim SH, Mustafa HH. Influence of platelet-rich plasma and Osteon II graft on bone fracture regeneration in a rabbit model. doi:10.5455/OVJ.2026.v16.i1.56 Harvard Style Ali, B. M., Ali, . O. J., Raouf, . G. M., Ibrahim, . S. H. & Mustafa, . H. H. (2026) Influence of platelet-rich plasma and Osteon II graft on bone fracture regeneration in a rabbit model. doi:10.5455/OVJ.2026.v16.i1.56 Turabian Style Ali, Brwa Mohamed, Othman Jalal Ali, Goran Mohamad Raouf, Salam Haji Ibrahim, and Hana H. Mustafa. 2026. Influence of platelet-rich plasma and Osteon II graft on bone fracture regeneration in a rabbit model. doi:10.5455/OVJ.2026.v16.i1.56 Chicago Style Ali, Brwa Mohamed, Othman Jalal Ali, Goran Mohamad Raouf, Salam Haji Ibrahim, and Hana H. Mustafa. "Influence of platelet-rich plasma and Osteon II graft on bone fracture regeneration in a rabbit model." doi:10.5455/OVJ.2026.v16.i1.56 MLA (The Modern Language Association) Style Ali, Brwa Mohamed, Othman Jalal Ali, Goran Mohamad Raouf, Salam Haji Ibrahim, and Hana H. Mustafa. "Influence of platelet-rich plasma and Osteon II graft on bone fracture regeneration in a rabbit model." doi:10.5455/OVJ.2026.v16.i1.56 APA (American Psychological Association) Style Ali, B. M., Ali, . O. J., Raouf, . G. M., Ibrahim, . S. H. & Mustafa, . H. H. (2026) Influence of platelet-rich plasma and Osteon II graft on bone fracture regeneration in a rabbit model. doi:10.5455/OVJ.2026.v16.i1.56 |