| Research Article | ||
Open Vet. J.. 2025; 15(10): 4904-4911 Open Veterinary Journal, (2025), Vol. 15(10): 4904-4911 Research Article Determination of spermatological characteristics of Carrier pigeons (Columba livia domestica) raised by local breedersTutku Can Acısu1*, Emine Yıldırım2, Burak Altundal2, Serkan Ali Akarsu3, Aslıhan Çakır Cihangiroğlu4, Nida Badıllı1, İbrahim Halil Güngör1, Şeyma Özer Kaya1, Yasin Baykalır5, Gaffari Türk1, Mustafa Sönmez1 and Seyfettin Gür11Department of Reproduction and Artificial Insemination, Faculty of Veterinary Medicine, Fırat University, Elazığ, Turkey 2Faculty of Veterinary Medicine, Fırat University, Elazığ, Turkey 3Department of Reproduction and Artificial Insemination, Faculty of Veterinary Medicine, Atatürk University, Erzurum, Turkey 4Department of Reproduction and Artificial Insemination, Faculty of Veterinary Medicine, Siirt University, Siirt, Turkey 5Department of Biostatistics, Faculty of Veterinary Medicine, Balıkesir University, Balıkesir, Turkey *Corresponding Author: Tutku Can Acısu, Department of Reproduction and Artificial Insemination, Faculty of Veterinary Medicine, Fırat University, Elazığ, Turkey. Email: tcacisu [at] firat.edu.tr Submitted: 12/08/2025 Revised: 01/10/2025 Accepted: 08/10/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
AbstractBackground: Pigeons are among the earliest domesticated bird species and are classified into groups based on breeding purposes, such as tumblers, rollers, and Carrier pigeons. Historically used for communication, they are now primarily used in various countries for hobbies, sports, and meat production. The public commonly raises Carrier pigeons (Columba livia domestica), valued for their speed and endurance. Understanding their reproductive traits is crucial for sustainable breeding and local genetic resource preservation. Aim: This study aimed to collect semen from male Carrier pigeons raised by local breeders, assess their spermatological characteristics, and examine the correlations among these traits. To our knowledge, this represents one of the first studies to report detailed semen parameters in Carrier pigeons under controlled conditions. Methods: In the research, six male Carrier pigeons brought to the animal hospital between May and October 2024 were used. Semen was obtained by abdominal massage once per month. Samples were visually inspected for contaminants such as blood, feces, or uric acid, and only clean ejaculates were included. Ejaculates were evaluated individually. Semen volume and concentration were determined. Motility was subjectively evaluated. Morphological evaluations and assessments of sperm membrane integrity were also conducted. Ringer’s solution was used as the semen extender. All analyses were conducted immediately after semen collection for evaluation purposes. Results: The average semen volume collected from Carrier pigeons was 9.58 ± 1.62 µl, the average total motility value was 69.67% ± 1.89%, and the average semen concentration was 3630.25 ± 388.26 × 106 sperm ml−1. The average membrane integrity ratio was 73.58% ± 5.61%. Head and tail abnormalities were evaluated separately, with the average head abnormality was 6.74% ± 1.00%, the average tail abnormality was 8.72% ± 1.34%, respectively. The average abnormal sperm rate was determined to be 15.47% ± 1.19%. Conclusion: This study determined the spermatological parameters in Carrier pigeons raised by local breeders, providing baseline semen data, insights into relationships among spermatological parameters, and seasonal effects. Limitations include a small sample size and a limited semen collection period due to the number of pigeons brought to the animal hospital. Keywords: Carrier pigeons, pigeon breeding, sperm, spermatological parameters. IntroductionPigeons (Columbidae) comprise over 300 species and are known for their fast, long-distance flight, dense feathers, compact bodies, and grain-based diet (Öncel et al., 2001; Biala et al., 2015). They are globally distributed except in polar regions, some oceanic islands, the Sahara Desert interior, and the coldest temperate zones (Flannery, 2009). Domesticated over 6,000 years ago, pigeons have long been raised for racing, hobbies, and meat production, especially in Europe, the Middle East, China, and the United States (Jerolmack, 2007; Yenilmez, 2020). Historically, Carrier pigeons (Columba livia domestica) were widely used for communication (Podbielska and Radko, 2022) and are still bred as a hobby and sport. They can return home after long-distance release. Although they vary in color and body structure, they are favored for their endurance and homing ability (Yılmaz and Boz, 2012). In Turkey, pigeon breeding has a long history and holds significant cultural and recreational value. Carrier pigeons are traditionally bred for racing, communication, and ornamental purposes, particularly in Central and Southeastern Anatolia regions (Yılmaz and Boz, 2012). Over time, pigeon breeding as a hobby has evolved into a well-established practice, with breeders developing local breeds and organizing informal competitions (Karaketir, 2023). In avian species, evaluating semen quality is essential for selecting breeding males and routinely monitoring their reproductive performance. Due to limitations in semen quality assessment, the diagnosis of fertility disorders in pigeons was not possible for a long time (Cheng et al., 2002). Semen is typically collected via abdominal massage under hygienic conditions. It generally has a high concentration but low volume due to the anatomical absence of accessory glands (Uçar and Uslu, 2022). The application of semen collection, storage, and artificial insemination techniques in pigeon breeding can lead to significant genetic progress. Moreover, the frequency of semen collection has been shown to affect both the qualitative and quantitative characteristics of semen in pigeons (Klimowicz et al., 2005). Additionally, a study demonstrated that various factors, especially air temperature and seasonal changes, can influence pigeon semen quality (Wannaratana et al., 2021). Although countries such as Brazil, China, Egypt, India, Nigeria, and Turkey possess rich genetic resources in pigeon species, breeders often lack sufficient knowledge about pigeon reproduction and breeding. Breeders are generally inadequate in conserving these species, and the genetic resources of many local breeds are diminishing year by year. Furthermore, a review of the current scientific literature suggests that research on this subject remains limited. Therefore, this study aimed to investigate the spermatological characteristics of Carrier pigeons raised by local breeders in Elazığ Province, Turkey, with a focus on semen quality parameters under regular and controlled collection intervals. The importance of this research lies in its potential to contribute to the limited body of knowledge on pigeon reproduction, support the development of more effective breeding strategies, and aid in the conservation of local genetic resources. Illustrated in Figure 1, these pigeons are evaluated to identify the factors affecting semen quality, providing a foundation for improving reproductive performance and genetic management in pigeons, particularly in regions where traditional breeding practices dominate and scientific input remains minimal.
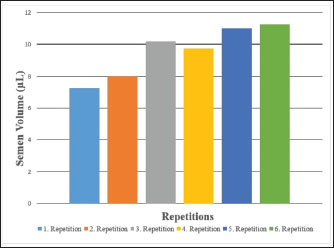
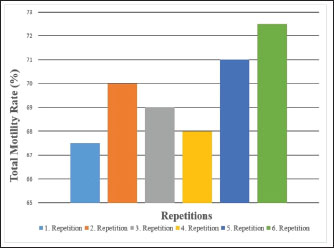
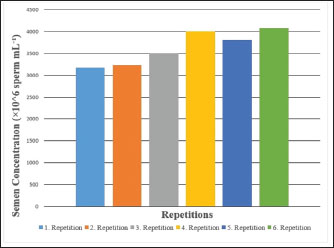
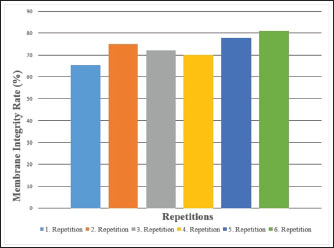
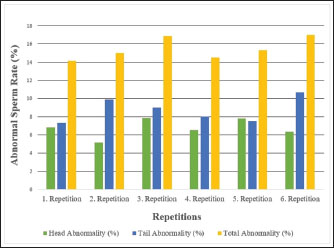
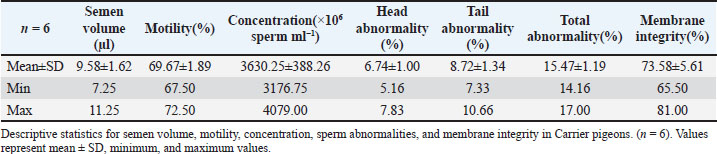
Fig. 1. A view of the Carrier pigeons used in the study. Materials and MethodsResearch area and experimental designSix clinically healthy male Carrier pigeons, 1.5 years old and weighing 350–400 g, with no pathological findings upon genital examination, were used in this study. Animals were brought to the Fırat University Animal Hospital between May and October 2024. Animal welfare principles were strictly observed throughout the study. Pigeons were housed in individual cages and fed with high-quality commercial feed. Water was provided ad libitum. The pigeons were maintained under standard environmental conditions, including a controlled temperature of 22°C ± 2°C and a 12:12 hours light/dark cycle. For ethical reasons, only pigeons whose owners consented to participate were included in the study. All owners provided signed informed consent forms before the experiment. We conducted a retrospective power analysis using G*Power (version 3.1) to assess the statistical robustness of our dataset. For two-tailed Pearson correlation tests (α=0.05, n=6), the achieved power was approximately 0.15 for a large effect size (r=0.50) and 0.08 for a medium effect size (r=0.30). For two-tailed paired t-tests (α=0.05, n=6), the achieved power was approximately 0.36 for a large effect size (d=0.80) and 0.17 for a medium effect size (d=0.50). Semen collection and analysisThe abdominal massage technique was used to collect semen from male pigeons (Cheng et al., 2002; Rakha et al., 2025). Six male Carrier pigeons were used in this study. Between May and October, semen was collected once a month from each pigeon brought to the animal hospital. The ejaculates were individually evaluated to eliminate the uncertainty caused by pooling and to accurately determine which bird had better sperm quality. After checking for contamination with blood, feces, or uric acid, the samples were evaluated. To minimize stress and potential negative effects during semen collection, particular attention was paid to factors such as the timing of collection and the personnel involved in the procedure. The individual evaluation was performed for each ejaculate. Semen volume was determined by aspirating it from the cloaca using an automatic pipette (Brand, USA). For motility analysis, a clean microscope slide was first placed on a heating stage (Reichert, Austria) maintained at 37°C. Motility was evaluated subjectively, and to standardize the assessment, a 5 µl semen sample was diluted with 95 µl of Ringer’s solution (9 g NaCl, 0.3 g KCl, 0.33 g CaCl2, and 0.2 g NaHCO3 dissolved in 1 l of distilled water), which served as a semen extender, then covered with a coverslip and examined using a digital microscope with an LCD screen (Celestron, USA) at 10 × 40 magnification (Blesbois, 2007; Wannaratana et al., 2021). Semen concentration was calculated using the hemocytometric method with a hemacytometer (Marienfeld Superior, Germany) (Aksu et al., 2016). The rate of membrane integrity was determined by examining smears prepared from semen samples stained with eosin-nigrosin stain, which was prepared using 1.67 g eosin Y, 10 g nigrosin, and 2.9 g sodium citrate in 100 ml distilled water (Pabuççuoğlu, 1994; Sharma and Agarwal, 2021; Shetty and Kanna, 2023). The proportion of abnormal spermatozoa in semen was also determined using the same smears prepared by the eosin-nigrosin staining method (Hafez, 1987; Bearden and Fuquay, 1992). Statistical analysisAll statistical analyses were performed using the statistical software R version 4.1.2 (The R Foundation for Statistical Computing, Vienna, Austria). The study was designed as an observational experiment with repeated semen collections from six adult male pigeons once per month between May and October (six repeated measures per individual). The statistical analysis aimed not to compare groups but to evaluate the relationships among semen quality traits. Therefore, correlation analyses were conducted. Before analysis, data were checked for normality using the Shapiro–Wilk test. Pearson’s correlation coefficients (r) were calculated for normally distributed variables, while Spearman’s rank correlation was used otherwise. Numerical variables were presented as means, and categorical variables were expressed as percentages (%). The volume and concentration of semen obtained from pigeons were analyzed as mean values. Motility values, as well as abnormal sperm and membrane integrity ratios, were analyzed as percentages (%). Pearson’s correlation analysis was used to evaluate the relationships between spermatological parameters. Correlation coefficients (r) were calculated, and statistical significance was set at p < 0.05, p < 0.01, and p < 0.001 (Petrie and Watson, 2013). Ethical approvalThis study was approved by the Local Ethics Committee of Fırat University Animal Experiments with the session number 2024/07-06 dated 04.04.2024, with the decision that an ethics committee certificate is not required. ResultsThe volume of semen obtained from Carrier pigeons ranged from 7.25 to 11.25 µl, with an average of 9.58 ± 1.62 µl, as presented in Figure 2. Figure 3 shows the total motility values of the semen obtained from carrier pigeons, ranging from 67.50% to 72.50%, with a mean value of 69.67% ± 1.89%. Semen concentration, as demonstrated in Figure 4, ranged from 3176.75 × 106 to 4079.00 × 106 sperm ml−1, and the average concentration was calculated as 3630.25 ± 388.26 × 106 sperm ml−1. The membrane integrity results (Fig. 5) varied between 65.50% and 81.00% with a mean value of 73.58 ± 5.61. Abnormal spermatozoa (Fig. 6) were evaluated in two categories: head and tail abnormalities. Head abnormalities ranged from 5.16% to 7.83%, with an average of 6.74% ± 1.00%, whereas tail abnormalities ranged from 7.33% to 10.66%, with a mean of 8.72% ± 1.34%. The overall abnormal sperm rate varied between 14.16% and 17.00%, and the average abnormality rate was determined to be 15.47% ± 1.19%. Table 1 presents all findings obtained from the Carrier pigeons.
Fig. 2. Semen volume values of Carrier pigeons for each repetition between May and October.
Fig. 3. Total motility percentages of Carrier pigeons for each repetition between May and October.
Fig. 4. Semen concentration values of Carrier pigeons for each repetition between May and October.
Fig. 5. Sperm membrane integrity percentages of Carrier pigeons for each repetition between May and October.
Fig. 6. Abnormal sperm rates of Carrier pigeons for each repetition between May and October. Table 1. Spermatological characteristics and descriptive statistics of Carrier pigeons.
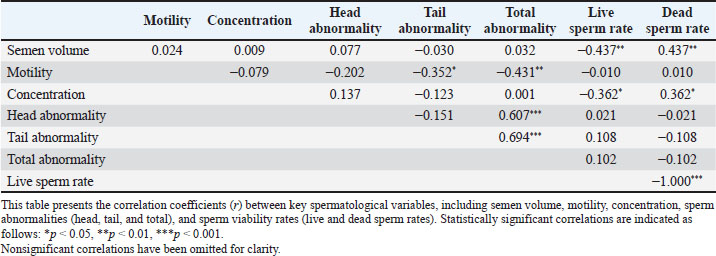
Correlation analyses among spermatological parameters revealed several statistically significant associations (Table 2). Semen volume was significantly negatively correlated with live sperm rate (r=−0.437, p < 0.01) and positively correlated with dead sperm rate (r=0.437, p < 0.01). Motility was negatively correlated with total abnormality (r=−0.352, p < 0.05) and live sperm rate (r=−0.431, p < 0.01), indicating that higher motility is associated with lower abnormality and a higher proportion of live sperm. In addition, motility demonstrated a weak, non-significant negative correlation with tail and head abnormalities. Semen concentration was significantly negatively correlated with dead sperm rate (r=−0.362, p < 0.05) and positively correlated with live sperm rate (r=0.362, p < 0.05). A strong positive correlation was observed between head and tail abnormalities (r=0.694, p < 0.001) and between head and total abnormalities (r=0.607, p < 0.001). Live sperm rate was perfectly and negatively correlated with dead sperm rate (r=−1.000, p < 0.001). Table 2. Pearson correlation coefficients among spermatological parameters in Carrier pigeons.
DiscussionThe present study is unique in elucidating the spermatological characteristics of the Carrier pigeon breed, which is the most populous and widely bred by pigeon breeders in Turkey. The experimental phase of the research spanned different seasons, so it is important to elucidate the effects of varying environmental conditions, particularly temperature and humidity, on the spermatological characteristics of pigeons and the relationships among these characteristics. The selection of male pigeons for use in artificial insemination with fresh or chilled semen necessitates a comprehensive laboratory evaluation of ejaculate quality (Gee et al., 2004; Klimowicz-Bodys et al., 2012). In this context, the present research focused on identifying key parameters that serve as indicators of semen quality in privately kept Carrier pigeons. The average volume of semen obtained from Carrier pigeons was 9.58 ± 1.62 µl. Cheng et al. (2002) reported that the average semen volume collected from different pigeon breeds was 11.00 ± 0.90 μl. Sontakke (2004) reported a semen volume of 10.50 ± 2.60 μl in their study on blue rock pigeons. Similarly, Wannaratana et al. (2021) reported that the semen volume ranged between 10.00 and 13.00 μl in a study conducted on adult pigeons. The findings of all these studies are consistent with the results of this study. Contrary to these findings, Klimowicz et al. (2005) observed higher semen volumes in adult pigeons. Factors such as study design, semen collection season, and racial and individual differences are thought to affect the semen volume obtained from pigeons. Sperm motility is an important parameter of semen quality (Klimowicz-Bodys et al., 2012; Xu et al., 2020). Sperm motility depends on the structural and functional integrity of these cells and determines their fertilization capability (Wishart and Palmer, 1986; Matsuzaki and Sasanami, 2022). In the present study, total motility values of semen obtained from Carrier pigeons varied between 67.50% and 72.50%, and the average total motility value was 69.67% ± 1.89%. In a study, the sperm motility rate in collected semen samples was measured as a minimum of 48.00% ± 3.00% and a maximum of 82.00% ± 2.00% (Cheng et al., 2002). A study conducted in Thailand reported that motility values varied from 66.35 ± 3.40 to 85.45 ± 2.91 (Wannaratana et al., 2021). On the other hand, another study documented total motility values within the range of 57.50% to 71.50% (Klimowicz et al., 2005). In another study conducted by Klimowicz et al. (2008) assessed sperm motility both subjectively using a light microscope and objectively using a computer-assisted semen analyzer (CASA). Subjective evaluations indicated that total motility values ranged from 84.21% ± 5.78% to 87.32% ± 6.84%. The motility values determined by CASA were lower than those obtained through subjective assessment. Davis and Katz (1992) reported that the subjective evaluation of the total motile sperm rate tends to overestimate sperm motility. The analyses in this study were performed in the Andrology Laboratory of Fırat University Animal Hospital. As CASA (ISASv1, Proiser, Paterna, Spain) could not be used to analyze pigeon sperm, motility was assessed subjectively. An experienced team was assembled to minimize the margin of error in motility evaluations. Previous studies have indicated that variations in motility values can be attributed to factors such as pigeon breed and age, environmental conditions, timing of semen collection, season, and evaluation methods employed. Semen concentration, defined as the number of sperm per milliliter of semen, is a critical parameter in assessing semen quality and, consequently, reproductive success (Li et al., 2025). In this study, semen concentrations varied between 3176.75 × 106 sperm ml−1 and 4079.00 × 106 sperm ml−1 and the average concentration was calculated as 3630.25 ± 388.26 × 106 sperm ml−1. When comparing the semen concentration values obtained from blue rock pigeons and Carrier pigeons, it was observed that they were similar (Sontakke, 2004). The semen concentration findings reported by Cheng et al. (2002) are also consistent with the results observed in the present study. In their comparative study, Klimowicz-Bodys et al. (2012) observed that meat-type breeds exhibited lower semen concentrations than fancy pigeons. The semen concentration results reported by Klimowicz-Bodys et al. (2012) were similarly low. When all these results are evaluated, it becomes clear that variations in semen concentration are influenced by factors such as age, semen collection frequency, season, and ambient air temperature. In the present study, head- and tail-related abnormalities in the spermatozoa of Carrier pigeons were evaluated separately. Head-related abnormalities ranged from 5.16% to 7.83%, whereas tail-related abnormalities ranged from 7.33% to 10.66%. The total abnormal sperm rate ranged from 14.16% to 17.00%, with a mean value of 15.47% ± 1.19%. In a previous study on pigeon semen, the mean percentage of abnormal spermatozoa was determined to be 15.44% (Ducci et al., 2002). Comparable rates of abnormal spermatozoa have also been reported in other avian species, suggesting that a certain level of morphological abnormalities is a common feature in bird semen (Penfold, 2001; Gee et al., 2004). The findings of this study indicate that the sperm membrane integrity rate in Carrier pigeons ranged between 65.50% and 81.00% with a mean value of 73.58% ± 5.61%. This rate was measured as 88.71% ± 5.42% in meat-type pigeons and 90.87% ± 6.01% in fancy pigeons (Klimowicz-Bodys et al., 2012), indicating relatively high sperm viability across different pigeon breeds. In a study conducted on blue rock pigeons, sperm membrane integrity rates were reported to be comparable to those observed in semen samples collected during the same seasonal periods in the present study (Cheng et al., 2002). In the present study, Ringer’s solution was used as a semen extender during the evaluation of pigeon sperm. This choice was based on its physiological compatibility and its long-standing application in avian semen analysis (Owen, 1941; Klimowicz et al., 2005). Although the comparative analysis of extenders was not within the scope of this study, previous research has demonstrated that extenders commonly used for other poultry species, such as turkeys and chickens, result in a rapid decline in pigeon sperm motility (Blanco et al., 2000). This decline is likely due to species-specific differences in the composition of seminal plasma and sperm membrane physiology. Sontakke (2004) reported that TALP medium may offer improved sperm preservation in pigeons; however, further controlled studies are required to evaluate the effectiveness of TALP. Although the current findings do not directly assess extender performance, the consistent motility and viability observed in semen processed with Ringer’s solution suggest that it remains a viable option for short-term analysis in this species. Although qualitative assessments, such as biochemical evaluations, were not conducted in this study, these analyses could provide valuable insights into the molecular and metabolic factors influencing sperm quality. The absence of such evaluations represents a limitation, mainly due to the limited amount of semen obtained from the pigeons, which restricted the scope of possible analyses. Future research incorporating biochemical parameters would strengthen the understanding of spermatological characteristics in Carrier pigeons. Given the limited number of similar studies on Carrier pigeons and the variability in methodologies, seasons, and environmental conditions, direct comparisons of results remain challenging. This difficulty is further compounded by factors such as the small number of studies available in the literature, differences in sample sizes, variation in the timing or season of semen collection, and discrepancies in the breeds and ages of pigeons used and the geographical regions where the studies were conducted (Cheng et al., 2002; Sontakke, 2004; Klimowicz et al., 2005; Wannaratana et al., 2021). It is important to acknowledge these limitations to contextualize the findings appropriately and avoid overgeneralization. This study provides essential baseline data on the spermatological characteristics of Carrier pigeons, offering valuable insights for both applied reproductive practices and future scientific research. By identifying key semen quality parameters and examining the influence of factors such as season and semen extenders, the findings can contribute to the development of more effective breeding strategies, artificial insemination protocols, and genetic conservation efforts. However, certain limitations, including the small sample size and the restriction of data collection to the May to October period, should be considered when interpreting the results. Despite these constraints, this study makes a meaningful contribution to the limited body of literature on pigeon reproduction and highlights important directions for future research. ConclusionIn conclusion, this study characterized the spermatological parameters of Carrier pigeons by evaluating semen samples collected between May and October at the Reproduction and Artificial Insemination Clinic. Semen volume, total motility, semen concentration, and membrane integrity rates were systematically measured, and the overall semen quality was assessed through detailed morphological evaluations. These findings provide reference values for the spermatological profile of the Carrier pigeon breed under controlled conditions. However, to achieve a more comprehensive understanding of semen quality and reproductive capacity in this species, further research is required. Such studies should involve advanced molecular and biochemical analyses, including assessments of DNA integrity, mitochondrial function, and proteomic profiling. This study provides reference values for key spermatological parameters of Carrier pigeons based on semen samples collected between May and October. Although these findings contribute valuable baseline data, the results’ generalizability is restricted by the small sample size and limited sampling period. Future studies should include larger sample sizes, year-round sampling, and advanced molecular and biochemical analyses such as assessments of DNA integrity, mitochondrial function, and proteomic profiling to achieve a more comprehensive understanding of semen quality and reproductive capacity in this species. AcknowledgmentThe authors would like to express their sincere gratitude to the staff at Fırat University Animal Hospital and the pigeon breeders for their valuable support. Conflict of interestThe authors declare no conflict of interest. FundingThe research was supported by the project numbered 1919B012315344 accepted on 26.03.2024 within the scope of Scientific and Technological Research Council of Turkey (TÜBİTAK)-2209-A University Students Research Projects, and some of the consumables used during the study were financed by this project support. Authors’ contributionTCA and EY contributed to the project idea, design, and execution of the study. TCA, EY, BA AÇC, NB and İHG contributed to the acquisition of data. TCA, YB, and SAA analyzed the data. TCA drafted and wrote the manuscript. TCA, ŞÖK, GT, MS, and SG reviewed the manuscript critically. All authors have read and approved the final manuscript. Data availabilityAll data supporting the findings of this study are available within the manuscript. ReferencesAksu, E.H., Akman, O., Ömür, A.D., Karakuş, E., Can, I., Kandemir, F.M., Dorman, E. and Uçar, O. 2016. 3, 3 diindolylmethane leads to apoptosis, decreases sperm quality, affects blood estradiol 17 β and testosterone, oestrogen (α and β) and androgen receptor levels in the reproductive system in male rats. Andrologia 48(10), 1155–1165. Bearden, J. and Fuquay, W. 1992. Applied animal reproduction. Englewood Cliffs, New Jersey, USA: Prentice Hall. Biala, A., Dybus, A., Pawlina, E. and Proskura, W.S. 2015. Genetic diversity in eight pure breeds and urban form of domestic pigeon (Columba livia var. domestica) based on seven microsatellite loci. J. Anim. Plant Sci. 25(6), 1741–1745. Blanco, J.M., Gee, G., Wildt, D.E. and Donoghue, A.M. 2000. Species variation in osmotic, cryoprotectant, and cooling rate tolerance in poultry, eagle, and peregrine falcon spermatozoa. Biol. Reprod. 63(4), 1164–1171. Blesbois, E. 2007. Current status in avian semen cryopreservation. World's. Poult. Sci. J. 63(2), 213–222. Cheng, F., Guo, T., Wu, J., Lin, T., Ursem, P., Colenbrander, B. and Fung, H. 2002. Annual variation in semen characteristics of pigeons (Columba livia). Poultry. Sci. 81, 1050–1056. Davis, R.O. and Katz, D.F. 1992. Standardization and comparability of CASA instruments. J. Androl. 13(1), 81–86. Ducci, M., Gazzano, A., Tedeschi, D., Sighieri, C. and Martelli, F. 2002. Coenzyme Q10 levels in pigeon (Columba livia) spermatozoa. Asian J. Androl. 4(1), 73–76. Flannery, M.C. 2009. The value of pigeons. Am. Biol. Teach. 71(7), 430–434. Gee, G.F., Bertschinger, H., Donoghue, A.M., Blanco, J. and Soley, J. 2004. Reproduction in nondomestic birds: physiology, semen collection, artificial insemination and cryopreservation. Avian. Poult. Biol. Rev. 15(2), 47–101. Hafez, E.S.E. 1987. Physiology of reproduction. Reproduction in farm animals. Philadelphia, USA: Lea and Febiger. Jerolmack, C. 2007. Animal archaeology: domestic pigeons and the nature-culture dialectic. Qual. Sociol. Rev. 3(1), 74–95. Karaketir, E. 2023. Kuştan al haberi: yazışmadan yarışa posta güvercinleri üzerine bir inceleme. Anasay 23, 291–329. Klimowicz, M., Łukaszewicz, E. and Dubiel, A. 2005. Effect of collection frequency on quantitative and qualitative characteristics of pigeon (Columba livia) semen. Br. Poult. Sci. 46(3), 361–365. Klimowicz, M.D., Nizanski, W., Batkowski, F. and Savic, M.A. 2008. The comparison of assessment of pigeon semen motility and sperm concentration by conventional methods and the CASA system (HTM IVOS). Theriogenology 70(1), 77–82. Klimowicz-Bodys, M.D., Batkowski, F., Ochrem, A.S. and Savič, M.A. 2012. Comparison of assessment of pigeon sperm viability by contrast-phase microscope (eosin-nigrosin staining) and flow cytometry (SYBR-14/propidium iodide (PI) staining) evaluation of pigeon sperm viability. Theriogenology 77, 628–635. Li, Y., Sun, Y., Ni, A., Tesfay, H.H., Isa, A.M., Zong, Y., Ma, H., Yuan, J. and Chen, J. 2025. Factor analysis of semen quality in chicken and its impact on fertility. Animals 15(13), 1906. Matsuzaki, M. and Sasanami, T. 2022. Sperm motility regulation in male and female bird genital tracts. J. Poult. Sci. 59(1), 1–7. Öncel, K., Turgut, K. and Şenel, V. 2001. Güvercinler Dünyası. Samsun Serinofil Derneği Yayını. Owen, R.D. 1941. Artificial insemination of pigeons and doves. Poultry. Sci. 20, 428–431. Pabuççuoğlu, S. 1994. Kanatlılarda reprodüksiyon ve sun'i tohumlama. İstanbul Üniversitesi Veteriner Fakültesi Yayını. Penfold, L. 2001. Characterization of northern pintail (Anas acuta) ejaculate and the effect of sperm preservation on fertility. Reprod 121, 267–275. Petrie, A. and Watson, P. 2013. Statistics for the veterinary and animal science. Chichester, West Sussex, UK: Wiley-Blackwell. Podbielska, A. and Radko, A. 2022. Genetic structure of racing pigeons (Columba livia) kept in Poland based on microsatellite markers. Genes. (Basel). 13(7), 1175. Rakha, B.A., Zuha, S. and Akhter, S. 2025. Semen extraction and analysis in avian species.In Spermatology: methods and protocols. Álvarez-Rodríguez, M New York, NY: Springer US, pp: 81–91. Sharma, R. and Agarwal, A. 2021. Sperm vitality: eosin-nigrosin dye exclusion.In Manual of sperm function testing in human assisted reproduction. Agarwal, A., Henkel, R. and Majzoub, A United Kingdom. Cambridge, UK: Cambridge University Press, pp: 47–51. Shetty, A. and Kanna, S. 2023. Eosin nigrosin staining technique in assessment of sperm vitality in medical laboratories–A snippet from our experience on implementing the staining, interpretation and quality control procedures. Indian J. Obstetr. Gynecol. Res. 10(2), 227–229. Sontakke, S. 2004. Semen characteristics, cryopreservation, and successful artificial insemination in the blue rock pigeon (Columba livia). Theriogenology 62(1–2), 139–153. Uçar. and Uslu, B.A. 2022. Kanatlı Hekimliği: reprodüksiyon ve Suni Tohumlama. Ankara, Turkey: Güneş Tıp Kitabevleri. Wannaratana, S., Olanratmanee, E.O., Charoenmuang, K., Boriharnthanawuth, T., Tangtrongwanich, B., Jongpattana, T., Sukhor, Y., Kongthip, A. and Sananmuang, T. 2021. Seasonal effect on semen availability and quality of racing pigeon in Thailand. Vet. World 14(6), 1459–1464. Wishart, G.J. and Palmer, F.H. 1986. Correlation of the fertilising ability of semen from individual male fowls with sperm motility and ATP content. Br. Poult. Sci. 27(1), 97–102. Xu, X., Tan, Y., Mao, H., Liu, H., Dong, X. and Yin, Z. 2020. Analysis of long noncoding RNA and mRNA expression profiles of testes with high and low sperm motility in domestic pigeons (Columba livia). Genes. (Basel). 11(4), 349. Yenilmez, F. 2020. Hayvancılık Alanında Akademik Araştırmalar: Güvercinler: İksad Yayınevi. Yılmaz, O. and Boz, A. 2012. Tarihten günümüze Türkiye'de güvercin (Columba livia) yetiştiriciliği. ADÜ. Ziraat. Derg. 9(1), 45–51. | ||
| How to Cite this Article |
| Pubmed Style Acısu TC, Yıldırım E, Altundal B, Akarsu SA, Cihangiroğlu AC, Badıllı N, Gungor �H, Kaya �O, Baykalır Y, Turk G, Sonmez M, Gur S. Determination of spermatological characteristics of Carrier pigeons (Columba livia domestica) raised by local breeders. Open Vet. J.. 2025; 15(10): 4904-4911. doi:10.5455/OVJ.2025.v15.i10.10 Web Style Acısu TC, Yıldırım E, Altundal B, Akarsu SA, Cihangiroğlu AC, Badıllı N, Gungor �H, Kaya �O, Baykalır Y, Turk G, Sonmez M, Gur S. Determination of spermatological characteristics of Carrier pigeons (Columba livia domestica) raised by local breeders. https://www.openveterinaryjournal.com/?mno=276863 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.10 AMA (American Medical Association) Style Acısu TC, Yıldırım E, Altundal B, Akarsu SA, Cihangiroğlu AC, Badıllı N, Gungor �H, Kaya �O, Baykalır Y, Turk G, Sonmez M, Gur S. Determination of spermatological characteristics of Carrier pigeons (Columba livia domestica) raised by local breeders. Open Vet. J.. 2025; 15(10): 4904-4911. doi:10.5455/OVJ.2025.v15.i10.10 Vancouver/ICMJE Style Acısu TC, Yıldırım E, Altundal B, Akarsu SA, Cihangiroğlu AC, Badıllı N, Gungor �H, Kaya �O, Baykalır Y, Turk G, Sonmez M, Gur S. Determination of spermatological characteristics of Carrier pigeons (Columba livia domestica) raised by local breeders. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4904-4911. doi:10.5455/OVJ.2025.v15.i10.10 Harvard Style Acısu, T. C., Yıldırım, . E., Altundal, . B., Akarsu, . S. A., Cihangiroğlu, . A. C., Badıllı, . N., Gungor, . �. H., Kaya, . �. O., Baykalır, . Y., Turk, . G., Sonmez, . M. & Gur, . S. (2025) Determination of spermatological characteristics of Carrier pigeons (Columba livia domestica) raised by local breeders. Open Vet. J., 15 (10), 4904-4911. doi:10.5455/OVJ.2025.v15.i10.10 Turabian Style Acısu, Tutku Can, Emine Yıldırım, Burak Altundal, Serkan Ali Akarsu, Aslıhan Cakır Cihangiroğlu, Nida Badıllı, İbrahim Halil Gungor, Şeyma Ozer Kaya, Yasin Baykalır, Gaffari Turk, Mustafa Sonmez, and Seyfettin Gur. 2025. Determination of spermatological characteristics of Carrier pigeons (Columba livia domestica) raised by local breeders. Open Veterinary Journal, 15 (10), 4904-4911. doi:10.5455/OVJ.2025.v15.i10.10 Chicago Style Acısu, Tutku Can, Emine Yıldırım, Burak Altundal, Serkan Ali Akarsu, Aslıhan Cakır Cihangiroğlu, Nida Badıllı, İbrahim Halil Gungor, Şeyma Ozer Kaya, Yasin Baykalır, Gaffari Turk, Mustafa Sonmez, and Seyfettin Gur. "Determination of spermatological characteristics of Carrier pigeons (Columba livia domestica) raised by local breeders." Open Veterinary Journal 15 (2025), 4904-4911. doi:10.5455/OVJ.2025.v15.i10.10 MLA (The Modern Language Association) Style Acısu, Tutku Can, Emine Yıldırım, Burak Altundal, Serkan Ali Akarsu, Aslıhan Cakır Cihangiroğlu, Nida Badıllı, İbrahim Halil Gungor, Şeyma Ozer Kaya, Yasin Baykalır, Gaffari Turk, Mustafa Sonmez, and Seyfettin Gur. "Determination of spermatological characteristics of Carrier pigeons (Columba livia domestica) raised by local breeders." Open Veterinary Journal 15.10 (2025), 4904-4911. Print. doi:10.5455/OVJ.2025.v15.i10.10 APA (American Psychological Association) Style Acısu, T. C., Yıldırım, . E., Altundal, . B., Akarsu, . S. A., Cihangiroğlu, . A. C., Badıllı, . N., Gungor, . �. H., Kaya, . �. O., Baykalır, . Y., Turk, . G., Sonmez, . M. & Gur, . S. (2025) Determination of spermatological characteristics of Carrier pigeons (Columba livia domestica) raised by local breeders. Open Veterinary Journal, 15 (10), 4904-4911. doi:10.5455/OVJ.2025.v15.i10.10 |