| Research Article | ||
Open Vet. J.. 2026; 16(2): 1060-1071 Open Veterinary Journal, (2026), Vol. 16(2): 1060-1071 Research Article The effect of a pumpkin flesh flour (Cucurbita pepo) diet on the health of Cyprinus carpio against Vibrio parahaemolyticusNadia Ali Al-Shammari1*, Hanan Hussain Shtewi2, Fatima Abulhussien Sultan3and Raad Muhammed Sayed-Lafi41Department of Natural Marine Science, College of Marine Sciences, Basrah University, Basrah, Iraq 2Zoology Department, Faculty of Science, Tripoli University, Tripoli, Libya 3Department of Fisheries and Marine Resources, Collage of Agriculture, University of Basrah. Basrah, Iraq 4National University of Science and Technology, Thi-Qar, Iraq *Corresponding Author: Nadia Ali Hussain Al-Shammari. Department of Natural Marine Science, College of Marine Sciences, University of Basrah, Basrah, Iraq. Email: nadia.ali.alshmeri [at] gmail.com Submitted: 13/08/2025 Revised: 25/12/2025 Accepted: 12/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
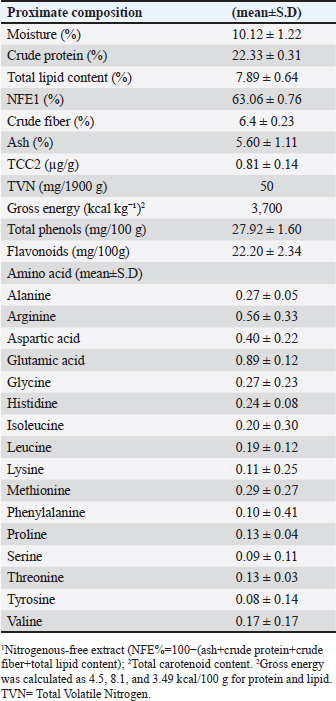
AbstractBackground: The pumpkin (Cucurbita pepo) is a promising medicinal plant that has garnered considerable scientific attention. It is a widely cultivated plant with high nutritional value and has potential as an alternative feed additive. Aim: This study aims to examine the effects of a pumpkin flesh flour (PFF) diet on common carp (Cyprinus carpio) infected with Vibrio parahaemolyticus. The research focuses on growth performance, biochemical and immunological parameters, antioxidant capacity, and the functions of the liver and kidneys. Methods: Vibrio parahaemolyticus was identified in infected common carp through molecular and biochemical analysis. PFF was added to the diet of infected fish at various percentages (0%, 0.5%, 1.0%, 1.5%, and 2.0%) over 70 days. Growth, haematological, and biochemical parameters, feed efficiency, immune responses, and antioxidant indices were measured. Results: After 70 days, the growth performance indicators (final body weight, weight gain, and specific growth rate) showed significant differences (p < 0.05) in fish fed the PFF diet. The results indicate that PFF groups are an effective and efficient dietary supplement for promoting the growth of common carp. The highest concentrations yielded the best results, with improvements also seen in blood profiles, biochemical analysis, immune responses, and antioxidant indices. Based on these findings, 1.0%, 1.5%, and 2.0% PFF are promising therapeutic plants that could be used in medical research and to protect carp aquaculture from bacterial diseases. Conclusion: This study highlights the significant potential of PFF as a sustainable and effective feed ingredient in aquaculture. It demonstrates positive effects on fish growth, feed efficiency, immune health, and disease resistance. These findings support the idea that pumpkin can play a vital role in reducing reliance on costly and unsustainable traditional protein sources, thereby contributing to the development of a more efficient and environmentally friendly aquaculture industry. Keywords: Cucurbita pepo, Cyprinus carpio, Pumpkin flesh flour, V. parahaemolyticus. IntroductionFish are a vital and valuable source of protein, fat, and energy. Aquaculture has seen significant growth in recent years, driven by increasing global demand for aquatic species (Costello et al., 2020; Vakili et al., 2024). Pathogens spread rapidly in aquaculture, resulting in significant production losses. The primary challenge for this crucial sector is that fish raised at high densities in aquaculture facilities are particularly susceptible to disease outbreaks (FAO, 2022; Irshath et al., 2023). Over the past 30 years, there has been a significant increase in experiments and reports focused on maintaining the health and boosting the immunity of aquatic organisms. However, the rapid expansion of the aquaculture industry has outpaced these efforts. Bacteria are among the most dangerous infectious agents that can harm aquaculture species. This group of pathogens receives less attention because they rarely cause primary infections and typically act as opportunistic pathogens in hosts that are already injured or severely immunocompromised (FAO, 2022). Cyprinus carpio has significant potential as a protein source because it generally grows quickly and has high fecundity. Although carp farming has advanced rapidly, there are still several restrictions on this fish. Disease control is one of the factors that limit the growth of the aquaculture industry, similar to other fish species (Al-Shammari, 2024). However, current methods for treating aquatic bacterial pathogens, such as synthetic medicines and antibiotics, are expensive, inefficient, complex, and harmful to the ecosystem (Lulijwa et al., 2020). While these treatments can be effective, their drawbacks—such as safety concerns, high costs, residual issues, and the development of multidrug-resistant bacteria—make their widespread use undesirable (Thirumalaikumar et al., 2021). Consequently, phytotherapy is a branch of medicine that uses traditional methods, often preferred over modern medicines, and emphasizes the use and understanding of medicinal herbalism (WHO, 2013 ). Although the aquaculture industry has only recently begun to adopt it, phytotherapy is increasingly seen as an alternative to manufactured medications (Citarasu, 2010). Experts in zoonotic diseases and the global microbiology community have highlighted Vibrio species as pathogens of public health concern (Austin and Austin, 2016). Vibrio parahaemolyticus, a Vibrio species in the Vibrionaceae family, is a notable example. Human gastrointestinal illnesses can result from consuming V. parahaemolyticus, a gram-negative that is broadly present in estuarine, marine, and coastal environments (Su and Liu, 2007; Nelapati et al., 2012; Ceccarelli et al., 2013; Zhang and Orth, 2013; Wang et al., 2015). Vibrio parahaemolyticus has garnered significant attention in the field of aquaculture due to its role as a prominent pathogen affecting marine and estuarine organisms. Its impact on aquaculture can lead to substantial economic losses through disease outbreaks in cultured species such as shrimp and fish. Subhashini et al. (2011) detected V. parahaemolyticus in 24 samples of prawn, 33 shrimp, and 27 crab samples by polymerase chain reaction (PCR) method. In Bangladesh, 60.2% of samples from aquaculture farms tested positive for V. parahaemolyticus, with 323 strains identified (Siddique et al., 2021) isolated from Oreochromis niloticus, Labeo rohita, and Penaeus monodon. In Malaysia, 17.71% of samples from shrimp farms were positive, with sediment samples showing the highest prevalence (Elexson et al., 2023). Nguyen et al. (2024) reported a 24.67% prevalence in fish mariculture environments in Vietnam. The pumpkin, or Cucurbita pepo, belongs to the Cucurbitaceae family and is recognized for its potential health benefits due to its rich nutritional content and bioactive compounds (Hussain et al., 2022). Cucurbita pepo extracts have been studied for their possible effects on boosting the immune system, enhancing disease resistance, and promoting growth across various industries, including aquaculture (Musthafa et al., 2017; Vakili et al., 2024). Khattab et al. (2023) observed that partially replacing fishmeal with pumpkin meal had a more positive impact on growth, nutritional efficiency, survival rates, and body composition of Oreochromis niloticus compared to the control diet. Furthermore, Mounes et al. (2024) reported that substituting soybean meal protein with pumpkin seed cake improved growth, antioxidant capacity, and immune status in Nile tilapia. Dietary pumpkin seed has been shown to boost growth, immune response, and feed efficiency in common carp, where Al-Hasson et al. (2025) observed higher survival rates following a pathogen challenge in fish receiving this supplement. In addition, Sezgin and Aydın (2021) demonstrated that soybean meal can be completely replaced by pumpkin seed cake in common carp diets without compromising growth performance or health status. As a result, pumpkin is increasingly gaining attention due to its therapeutic uses, nutritional value, and health-protective properties (Montesano et al., 2018). Consequently, this study aimed to evaluate the effects of incorporating pumpkin flesh flour (PFF) into the diet of common carp on their growth, metabolic and haematological parameters, immune response, and antioxidant activity. Material and MethodsPreparation of the pumpkin flesh flourPumpkins were purchased from local markets in Basrah Province, Iraq. To prepare the powder, the seeds and peels were manually separated and sun-dried until a constant weight was achieved. The dried components were then ground using a rotary mill and sieved through a 28-mesh screen to ensure a uniform particle size (George, 2020). Analyses of the proximate composition were performed. The levels of moisture, ash, crude protein, crude fiber, and total fat are presented in Table 1; these values were estimated according to AOAC (1990) methods. Additionally, the carotenoid and total polyphenol contents were analyzed according to Zhishen et al. (1999) and de De Carvalho et al. (2012). Isolation of bacteriaNaturally infected common carp (11.5 ± 1.12 g) were obtained from earthen tanks at the University of Basrah’s Marine Science Center. To isolate bacterial samples, 1.0 g of liver and kidney tissue was harvested from three individuals; the remaining fish were transferred to experimental glass tanks. After manual homogenization, the tissues were placed in sterile test tubes with 9 ml of distilled water. Following two minutes of vortexing, the tubes were allowed to settle for 5–10 minutes before being streaked onto culture media. The media were then incubated for 24–48 hours at 37°C to promote the growth of microorganisms on thiosulfate-citrate-bile salt-sucrose (TCBS) agar. Molecular detectionThe deoxyribonucleic acid (DNA) purification kit from the Korean company Macrogen is designed to isolate DNA from bacteria and uses universal primers. The 16S rRNA gene was amplified using (27F, 5′ AGAGTTTGATCCTGGCTCAG-3′) and (1492R, 5′-GGTTACCTTGTTACGACTT-3′), following the methods of Al-Shammari et al. (2023) and the company’s instructions. Table 1. Proximate composition analysis and total amino acids of PFF on a dry matter basis.
Biochemical characterizationUsing 2% NaCl as the suspension solution and API 20E diagnostic strips (bioMérieux, Durham, NC), partial biochemical profiles of each V. parahaemolyticus isolate were obtained. Dry slides were used for oxidase testing (BBL, Difco, Sparks, MD). Urease and Voges–Proskauer (VP) reactions were confirmed using urea (pH 6.7) and methyl red, respectively, because these reactions were consistently unclear on the API strips. Following the procedure described by Jones et al. (2012), the VP test was performed using the VP reagent (API, Durham, NC). Table 2. Composition analysis of the experimental basal diet.
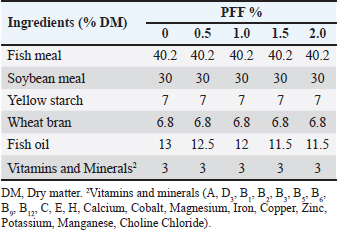
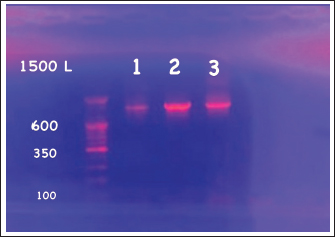
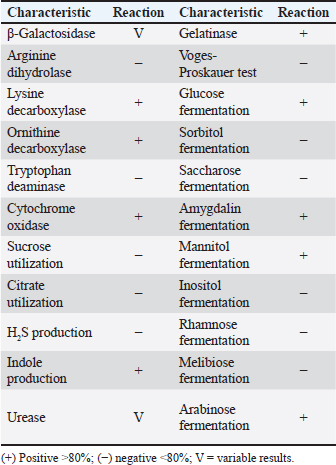
Experimental fish and dietsExperimental diets were formulated to be isonitrogenous, containing 35% crude protein and 6% lipid. PFF was added at levels of 0% (control), 0.5%, 1.0%, 1.5%, and 2.0%. Table 2 illustrates the composition and nutritional characteristics of the experimental diets. The meat grinder produced pellets, which were then dried for 24 hours at 60°C in an air-recirculation oven (Marconi, MA 035) and stored in a freezer at −18°C. After obtaining 75 infected common carp and allowing them to acclimate for a week to a commercial diet, the fish were divided into five dietary treatments using a completely randomized design, with each treatment replicated three times. There were fifteen fish per treatment, i.e., five individuals per replicate. Fish were fed twice daily at a rate of 3% (at 7:30 am and 5:00 pm). Throughout the 70-day experiment, the fish were kept under controlled conditions. Growth performance parametersFish were weighed every 2 weeks to assess proper feed intake (FI), and calculations were performed using the following equations: Weight gain (WG)=W2 − W1 (g) Specific growth rate (SGR%)=100(ln W2 − ln W1)/T Relative growth rate (RGR%)=W2 − W1 (g)/W1 (g) × 100 Feed conversion ratio (FCR)=FI /BWG (g) Total feed intake (TFI g/fish)=(cumulative feed distributed – feed refusals) / N where W1 is the initial body weight (g), W2 is the final body weight (g), T is the number of days in the feeding period, and N is the number of fish. Haematological and biochemical analysesBlood samples were collected from the caudal vein into specific tubes for each analysis. Serum was separated by centrifugation for 15 minutes at 4,000 rpm. The obtained serum was stored at –20°C for chemical and immunohistochemical analysis. Serum bactericidal and lysosomal activities were then evaluated. Additionally, aspartate aminotransferase (AST) and serum alanine aminotransferase (ALT) activities were measured using colorimetric kits from Bio-Diagnostics (Cairo, Egypt). An automatic hematology analyzer (Beckman Coulter Ac-T, Germany) was promptly used to determine the complete blood count, which was used to measure the hemoglobin concentration, red blood cells (RBC), white blood cells (WBC), mean corpuscular volume (MCV), and mean corpuscular hemoglobin (MCH). Meanwhile, colorimetric methods measured the concentrations of albumin (Alb) and total protein (TP), following the procedures by Wootton and Freeman (1982) and Tietz (1990) respectively. ALB levels were subtracted from TP levels to estimate globulin content. Additionally, creatinine and uric acid levels were measured calorimetrically with commercial kits (Bio-Diagnostics, Cairo, Egypt). Immunological and antioxidant analysisThe immunological assessments encompassed the activities of lysozyme and immunoglobulin M (IgM), which were quantified utilizing the methodologies delineated by Sanchooli et al. (2012). Concurrently, commercial assay kits procured from BioSource Inc., located in San Diego, CA, USA, were employed to ascertain the enzymatic activities of catalase (CAT), superoxide dismutase (SOD), and glutathione peroxidase (GPX) as part of the antioxidant evaluation conducted at the end of the experimental procedures. Statistical analysisSPSS version 26 (SPSS, Inc., Chicago, USA) was used for all statistical analyses. A one-way ANOVA test (p < 0.05) was performed to evaluate differences among groups, followed by a post-hoc test (LSD’s multiple range test) to compare means. Data are presented as mean ± standard deviation. Ethical approvalNot needed for this study. ResultsThe 16S rRNA gene was used to molecularly identify bacterial isolates from common carp infected with a bacterial disease. This was confirmed by the presence of 1,500 base pair sequences (Fig. 1), which indicates the sequence of the isolated V. parahaemolyticus bacteria. Figure 2 shows the electrophoresis process using 2% agarose of the bacterial isolation product. Based on Table 3, the biochemical features of the bacterial isolate from the infected common carp are summarized as follows: all isolates of V. parahaemolyticus tested positive for oxidase, indole, and glucose fermentation. The bacterium is characterized by its ability to produce gelatinase, cytochrome oxidase, and indole. It can also break down specific amino acids, as shown by positive tests for lysine and ornithine decarboxylase. Clinical isolates of V. parahaemolyticus are typically identified and exhibit negative results for H2S production, melibiose fermentation, citrate utilization, and the Voges-Proskauer test, among other characteristics. Lastly, the results for β-galactosidase and urease varied, indicating that this bacterium exhibits consistent traits. This distinctive set of biochemical reactions provides a specific fingerprint crucial for its identification and classification.
Fig. 1. PCR product sequencing of the 16S rRNA gene of isolated V. parahaemolyticus.
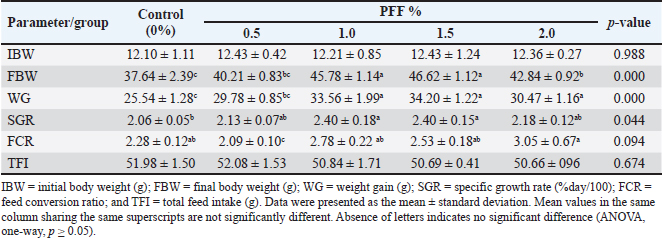
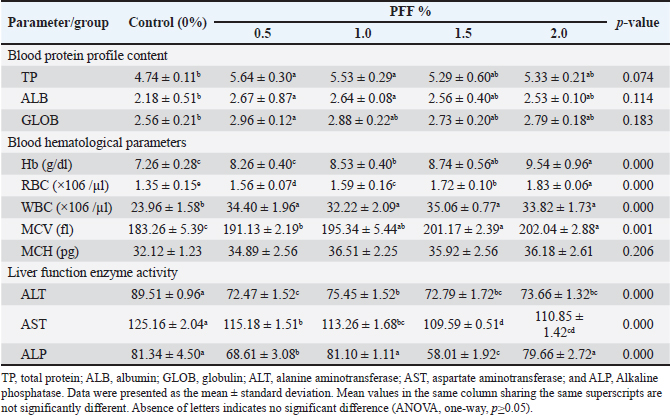
Fig. 2. Agarose gel electrophoresis for the PCR product of V. parahaemolyticus (1,500 bp) from three naturally infected fish. Supplementing the common carp’s diet with PFF has a significant positive impact on their growth performance (Table 4). All groups started with a very similar initial body weight (IBW), providing a reliable baseline for comparison (p=0.988). Following a duration of 70 days of cultivation, the findings demonstrated a noteworthy enhancement in final body weight (FBW), weight gain (WG), and specific growth rate (SGR) among all PFF groups, particularly at concentrations of 1 and 1.5% (p < 0.05). The 1.5% concentration yielded the most elevated FBW (46.62 g) in contrast to the control cohort (37.64 g). 1.0% and 1.5% of PFF diets significantly improved both weight gain (WG, p=0.000) and specific growth rate (SGR, p=0.044). Although there was no significant difference in FCR between the control and treatment groups (p=0.094), the post hoc test revealed a modest variation among some groups. At 2.0% PFF, the FCR dropped (indicating a high value), and this group exhibited the highest FCR of 3.05 when compared to the control group (p=0.041) and the 0.5% group (p=0.016). Carp fed PFF diets showed no significant difference in TFI compared to the control group (p=0.674). Table 5 illustrates the haematological and biochemical parameters associated with common carp feeding supplementation. PFF diets somewhat increase the fish’s blood protein profile. However, significant differences were seen only at 0.5% and 1.0% PFF levels for TP (p=0.010 and 0.020, respectively) and ALB (p=0.020 and 0.027, respectively) when compared to the control group. Furthermore, 0.5% demonstrated a significant difference in GLOB value compared to the control group (2.96, p=0.028). However, there is no significant difference in concentrations of 1.5% and 2.0% for TP and ALB, respectively, and 0.5% for GLOB when compared to other groups (p=0.05). Haematological parameters also showed clear improvements (Table 5), except for MCH. All the PFF groups experienced significant increases in haemoglobin (Hb) and RBC counts (p < 0.05), with the 2.0% PFF diet having the highest values (9.54 and 1.83, respectively; p=0.000). Additionally, WBC counts and MCV were significantly higher in all PFF groups, especially at 1.5% and 2.0% (p < 0.05). The addition of PFF improved liver function, as shown by the activity of liver function enzymes. The PFF groups showed a significant decrease in ALT and AST compared to the control group (p < 0.05). Regarding ALP values, the 0.5% and 1.5% PFF diets exhibited a reduction compared to the control group (p=0.000), but the 1.0% and 2.0% diets demonstrated an increase with no significant difference from the control group (p=0.530). Table 3. Biochemical properties of bacterial isolates from the infected common carp.
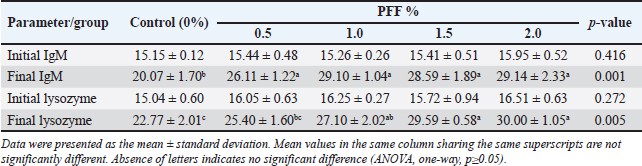
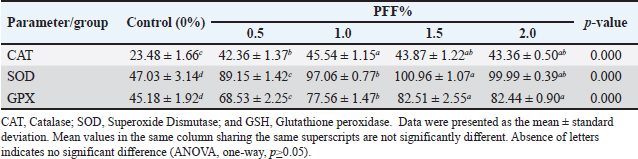
Table 6 shows the positive effect of PFF diets on immune markers (IgM and lysozyme) after 70 days. All PFF groups exhibited an increase in these markers compared to the control group (p < 0.05). There was no significant difference in final IgM levels among the PFF groups (p ≥ 0.05). During the same period, the 0.5% PFF groups did not show a significant difference in lysozyme levels compared to the control group (p=0.123) and 1.0% PFF (p=0.302). The 1.5% and 2.0% groups had the highest final lysozyme levels compared to the control group (29.59 and 30.00, respectively; p=0.001). Table 7 showed the effect of dietary pumpkin supplementation on the antioxidant indices of common carp. The results reveal significant increases in all antioxidant enzyme activity (p < 0.05). The activity of CAT and SOD was significantly higher in all RP groups compared to the control group. While the SOD value peaked at 1.5% (100.96), the CAT value peaked at 1.0% (45.54). The concentration of GPX progressively rose in the PFF groups, exhibiting significant differences among groups, except between the 1.5% and 2.0% groups (p =0.967). The highest GPx values for PFF diets were significantly higher than the control group (p < 0.05), especially at 1.5% (82.51) and 2.0% (82.44). Table 4. Growth performance indices and efficiency of common carp fed PFF diets.
Table 5. Hematological and biochemical parameters of common carp fed PFF diets.
Table 6. Immunity responses of common carp fed PFF diets.
Table 7. Antioxidant indices of common carp fed PFF diets.
DiscussionRecent research on Vibrio genetics has led to significant progress in understanding these infections. According to Al-Taee et al. (2017) vibriosis typically presents as hemorrhagic septicemia, characterized by localized necrosis of the liver, spleen, kidney, and other tissues, accompanied by widespread skin lesions. Aquatic animals are the primary reservoirs for V. parahaemolyticus infections in freshwater environments (Quintoil et al., 2007; Abdellrazeq and Khaliel, 2014). However, some studies have shown that these bacteria also live in marine environments, estuaries, and coastal areas (Nelapati et al., 2012; Velazquez-Roman et al., 2012; Ceccarelli et al., 2013; Zhang and Orth, 2013). Therefore, salinity changes in the aquatic habitats of Basra indicate high salinity levels in rivers with low freshwater flows and the intrusion of the salt front from the Arabian Gulf into the Shatt Al-Arab River (Abdullah and Lafta, 2024; Al-Silawy et al., 2025). When isolated from seafood and aquatic environments, V. parahaemolyticus strains complicate standard phenotyping and biochemical identification methods. Therefore, a common molecular technique for identifying and detecting V. parahaemolyticus is PCR-based assays (Drake et al., 2007; Subhashini et al., 2011). Mamdouh et al. (2023) observed that the biochemical features of V. parahaemolyticus samples indicate that the isolated strains are rod-shaped, motile, and produce catalase and oxidase. Additionally, Ghenem and Elhadi (2018) and Patel et al. (2018) found that the genus exhibits hemolytic activity, causing colonies to appear green on TCBS agar. The incorporation of pumpkin into the diet for development is seldom documented; it is predominantly employed to enhance color vibrancy in ornamental fish (Aulia et al., 2025). The current results indicate that supplementing with PFF improved the growth performance of common carp, especially at 1.0%, 1.5%, and 2.0%. This effect may be due to compounds present in PFF, such as carotenoids, zeaxanthin, and phenolic acids, which are known to promote growth (Eissa et al., 2025). Pumpkin flour contains high β-carotene, which plays an important role in fish colouring and improve the growth. Therefore, Teimouri et al. (2013) noticed that using carotenoids to increase the brightness of colour could also increase growth in rainbow fish. Yanti et al. (2022) found that 5% pumpkin flour significantly increased the growth of L. calcarifer. But Sausan and Safrida (2017) did not notice a significant effect on the growth of Carassius auratus fish when adding pumpkin flour. As well, Aulia et al. (2025) found that while adding pumpkin peel powder to Labidochromis caeruleus diet had no effect on growth, it did have an impact on colour enhancement. The optimal dose of yellow pumpkin peel flour for enhancing the color of lemon fish is P4 (15%), with a 30.7% increase in fish color. Additionally, it might influence the overall structure of the gut, as Al-Hasson et al. (2025) and Khattab et al. (2024) found that pumpkin encourages the development of intestinal villi, thereby increasing the surface area for nutrient absorption and enhancing digestive enzyme activity. Meanwhile, beyond serving as a protein source, raw pumpkin also has the potential to act as a nutraceutical. It contains health-promoting bioactive compounds, including squalene, phytosterols, and tocopherols. Additionally, these medicinal plants have been utilized for various pharmacological and therapeutic purposes, including anti-inflammatory, anti-cancer, antioxidant, antibacterial, and antiparasitic effects (Bardaa et al., 2016; Musthafa et al., 2017; Salehi et al., 2019; Abdelhiee et al., 2021). Serum and haematological biochemical markers are essential tools for assessing fish health, offering insights into their immune, pathological, and physiological conditions (Ammar et al., 2020). According to the study’s results, PFF showed no significant effect on TP, ALB, and GLOB compared to the control group, except at the lower concentrations (0%, 0.5%, 1.0%, and 1.0%). Therefore, the treatments did not impact these serum biochemical indicators. These findings align with those of Sezgin and Aydın (2021) who also studied common carp but used pumpkin seed cake as a substitute for dietary soybean meal. A higher RBC count and Hb concentration in fish fed PFF suggest improved oxygen-carrying capacity, potentially leading to increased metabolic activity and energy production. The effect of pumpkin on haematological parameters may be due to its high vitamin C content, which enhances iron absorption from the intestines of the fish. Other vitamins and minerals may also help improve haematopoiesis (Lim et al., 2000). Research consistently shows that pumpkin is rich in minerals such as iron and zinc, as well as vitamins like folic acid, which are vital for hemoglobin synthesis and red blood cell formation (Elinge et al., 2012; Negm, 2020). The current findings align with those of Vakili et al. (2024) who investigated the effects of pumpkin seed extract on growth factors, serum immune factors, and resistance to Yersinia ruckeri in O. mykiss. Conversely, Jalili et al. (2013) reported that replacing fish meal with plant sources (wheat gluten, corn gluten, and soybean meal) did not significantly impact blood parameters. The restoration of the common carp’s livers after being fed PFF shows that pumpkin works through a mechanism that improves ALT and ALP levels, thereby reducing liver toxicity. This suggests that PFF helps prevent liver diseases and disorders by enhancing liver function and reducing cell damage (Uribe et al., 2011). Dietary pumpkin offers hepatoprotective and nephroprotective benefits due to its antioxidant activity (Latif et al., 2020). Carotenoids are pigmented compounds produced by plants and microorganisms, including β-carotene, lycopene, and lutein (Shehu and Saidu, 2012). Furthermore, Wang et al. (2022) demonstrated that β-carotene provides significant protection to the liver. Pumpkin effectively reduces liver and kidney enzyme levels because of its antioxidant and anti-inflammatory properties, which protect these organs from damage (Negm, 2020). The current study demonstrated that the use of pumpkin flour during the experiment improved the phenotypic condition of the liver, as it contains β-cryptoxanthin (Ninčević Grassino et al., 2023). In this context, active components of medicinal herbs contribute to improving fish health (Sayed-Lafi et al., 2024a and b). Similarly, Mounes et al. (2024) observed that replacing soybean meal with pumpkin seed cake in Nile tilapia diets decreased serum enzyme levels such as AST and ALT. However, there has been limited research on the effects of natural carotenoids from vegetable sources on fish growth and liver enzyme activity. The use of pumpkin flour may also somewhat reduce the need for antibiotic treatment, such as with florfenicol, enrofloxacin, and oxytetracycline. These antibiotics improved the liver function parameters of Nile tilapia infected with V. alginolyticus (El-Gohary et al., 2020). Teleost fish primarily rely on non-specific immune responses, including antibacterial compounds such as lysozyme and IgM. For fish, the innate immune system is a more critical first line of defence against invasive infections than it is for mammals (Magnadóttir, 2006). Ekawati (2008) mentioned that the carotenoid in the diet of fish not only function as colour formation but also play a role in their reproduction and immune system. In this study, IgM and lysozyme levels increased with high percentages of PFF diet (1.0%, 1.5%, 2.0%). After breaking down bacteria, these antimicrobial substances act as opsonins, stimulating phagocytes and the complement system (Magnadóttir, 2006; Uribe et al., 2011). Furthermore, Eissa et al. (2025) indicated that pumpkin seed oil-loaded chitosan nanoparticles enhance the immune response in Litopenaeus vannamei against V. parahaemolyticus. Therefore, the PFF may enhance the fish’s resistance to V. parahaemolyticus, potentially improving health management in farmed fish and increasing their disease resistance, which is vital for successful aquaculture. These findings agree with those of Musthafa et al. (2017) who studied Oreochromis mossambicus fed diets containing pumpkin mixed seed meal. They observed improved immune responses against gram-negative bacteria and Aeromonas hydrophila. Similarly, their results align with studies on Nile tilapia (Khattab et al., 2023; Naiel et al., 2024), rainbow trout (Vakili et al., 2024), Caspian whitefish (Adel et al., 2015), and Victoria Labeo (Ngugi et al., 2015). Antioxidant activity in enzymes like CAT, SOD, and GPX serves as a key biomarker for monitoring fish health, reflecting how fish respond to external stimuli. This activity also assesses the antioxidant capacity of the fish (El-Houseiny et al., 2023). Pumpkin may have stimulated antioxidant defenses in all treated groups because it contains active ingredients such as proteins, minerals, phytosterols, triterpenes, carotenoids, lignans, polyunsaturated fatty acids, tocopherol, and phenolic and antioxidant compounds (Caili et al., 2006; Nkosi et al., 2006; Salehi et al., 2019; Abdelhiee et al., 2021). In this study, PFF was particularly effective, significantly boosting the activities of CAT, SOD, and GPX at high concentrations (1.0%, 1.5%, and 2.0%). Overall, carp fed with pumpkin demonstrated a strong and active antioxidant defense system. Similar results were observed in other studies, including those by El-Houseiny et al. (2023) and Mounes et al. (2024) on Nile tilapia, as well as Al-Hasson et al. (2025) and Sayed-Lafi et al. (2024a) on common carp. ConclusionPFF contains active components that help improve the health of fish infected by V. parahaemolyticus. It can be considered a valuable tool for enhancing growth performance, haematological and biochemical parameters, immunological responses, and antioxidative activity in common carp. Further research is needed to determine the optimal dosage for promoting growth and health in common carp, as well as to obtain more detailed findings. AcknowledgmentsThe authors thank the Department of Natural Marine Science, College of Marine Sciences, Basrah University, Iraq, for their logistical support in completing this work. Conflict of interestThe authors declare that there is no conflict of interest. FundingThe authors declare that they have not received any funding, whether institutional, private, or corporate, for the work reported in their manuscript. Authors’ contributionsNAHA supervised the experience, wrote the original draft manuscript, and isolated and identified the bacteria. HHS contributed to the writing, reviewed the data analysis, tables, and figures, conducted the literature review, and authored the final manuscript. FAMS prepared the first draft of the manuscript in addition to the methodology. RMS-L designed and implemented the research, entered the raw data, and performed statistical analyses using SPSS. All authors read, approved, and agreed with the results and conclusions. Data availabilityThis manuscript contains all the data that support the study’s findings. ReferencesA.O.A.C. 1990. Official Methods of Analysis. 15th ed., Washington DC: Association of Official Analytical Chemist. Abdelhiee, E.Y., Elbialy, Z.I., Saad, A.H., Dawood, M.A., Aboubakr, M., El-Nagar, S.H., El-Diasty, E.M., Salah, A.S., Saad, H.M. and Fadl, S.E. 2021. The impact of Moringa oleifera on the health status of Nile tilapia exposed to aflatoxicosis. Aquacult. 533, 736110. Abdellrazeq, G.S. and Khaliel, S.A. 2014. Molecular characterization and antimicrobial susceptibility of vibrios isolated from healthy and diseased aquacultured freshwater fishes. Glob. Vet. 13(3), 397–407. Abdullah, S.S. and Lafta, A.A. 2024. Geographic variation of the physical characteristics of water and air of the Shatt al-Arab in Basrah. ThiQar. Art. J. 9(47), 117–139. Adel, M., Abedian Amiri, A., Zorriehzahra, J., Nematolahi, A. and Esteban, M.A. 2015. Effects of dietary peppermint (Mentha piperita) on growth performance, chemical body composition and hematological and immune parameters of fry Caspian white fish (Rutilus frisii kutum). Fish. Shellfish. Immunol. 45(2), 841–847. Al-Shammari, N.A., AL-Niaeem, K.S. and Al-Hawash, A.B. 2023. Molecular identification of some zoonotic bacteria isolated from fishes Cyprinus carpio L. and Oreochromis niloticus (L.). Egypt. J. Aquat. Biol. Fish. 27(4), 1047–1062. Al-Hasson, H.A.H., Al-Niaeem, K.S. and Elbour, M. 2025. Effects of dietary pumpkin seed flour (Cucurbita pepo) supplementation on growth, physiological condition, and resistance to pathogens challenge in Cyprinus carpio. Open Vet. J. 15(9), 4418–4431. Al-Shammari, N.A.H. 2024. Isolation and identification of some pathogenic bacteria and the effect of Aeromonas hydrophila on antioxidant enzymes in common carp (Cyprinus carpio L.) and Nile tilapia (Oreochromis niloticus L.) in Basrah Governorate, Iraq. PhD thesis, unpublished, University of Basrah. College of Agriculture. 77p. Al-Silawy DA Al., -Abbawy HKH Al. and -Mahmood. 2025. Assessment of Seasonal and Spatial Variation of Heavy Metals Concentrations in Shatt Al-Arab River, Basrah, Iraq. Environ. Asia 18(2), 131–143. Al-Taee, A.M.R., Khamees, N.R. and Al-Shammari, N.A.H. 2017. Vibrio species isolated from farmed fish in Basra city in Iraq. J. Aquac. Res. Dev. 8(2), 1–8. American Public Health Association “APHA”. 2017. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA. Ammar, A.I., Afifi, A.F., Essa, A., Galal-Khallaf, A., Mokhtar, M.M., Shehab-Eldeen, S. and Rady, A.A. 2020. Cucurbita pepo seed oil induces microsatellite instability and tegumental damage to Schistosoma mansoni immature and adult worms in vitro. Infec. Drug. Resist. 3469, 3469–3484. Aulia, Y.C., Tartila, S.S.Q. and Armando, E. 2025. Evaluation of pumpkin peel powder (Cucurbita moschata durch) dietary supplementation on growth and color improvement in lemon fish (Labidochromis caeruleus). Jurnal Akuakultura Universitas Teuku Umar 9(2), 89–94. Austin, B. and Austin, D.A. 2007. Bacterial fish pathogens: diseases of farmed and wild fish. Dordrecht, The Netherlands: Springer Netherlands, p: 723. Bardaa, S., Ben Halima, N., Aloui, F., Ben Mansour, R., Jabeur, H., Bouaziz, M. and Sahnoun, Z. 2016. Oil from pumpkin (Cucurbita pepo L.) seeds: evaluation of its functional properties on wound healing in rats. Lipids Health Dis. 15, 73. Caili, F., Huan, S. and Quanhong, L. 2006. A review on pharmacological activities and utilization technologies of pumpkin. Plant Foods. Hum. Nutr. 61, 70–77. Ceccarelli, D., Hasan, N.A., Huq, A. and Colwell, R.R. 2013. Distribution, and dynamics of epidemic, and pandemic Vibrio (parahaemolyticus) virulence factors. Front. Cell. Infect. Microbiol. 3, 97. Citarasu, T. 2010. Herbal biomedicines: a new opportunity for aquaculture industry. Aquac. Int. 18(3), 403–414. Costello, C., Cao, L., Gelcich, S., Cisneros-Mata, M.Á., Free, C.M., Froehlich, H.E Golden, C.D., Ishimura, G., Maier, J., Macadam-Somer, I., Mangin, T., Melnychuk, M.C., Miyahara, M., de Moor, C.L., Naylor, R., Nøstbakken, L., Ojea, E., O’Reilly, E., Parma, A.M., Plantinga, A.J., Thilsted, S.H. and Lubchenco, J. 2020. The future of food from the sea. Nature 588(7836), 95–100. de Carvalho, L.M.J., Gomes, P.B., de Oliveira Godoy, R.L., Pacheco, S., do Monte, P.H.F., de Carvalho, J.L.V., Nutti, M.R., Neves, A.C.L., Vieira, A.C.R.A. and Ramos, S.R.R. 2012. Total carotenoid content, α-carotene and β-carotene, of landrace pumpkins (Cucurbita moschata Duch): a preliminary study. Food Res. Int. 47(2), 337–340. Drake, S.L., De Paola, A. and Jaykus, L.A. 2007. An overview of Vibrio vulnificus and Vibrio parahaemolyticus. Compr. Rev. Food Sci. Food Saf. 6(4), 120–144. Eissa, E.S.H., Salama, W.M., Elbahnaswy, S., El-Son, M.A., Eldin, Z.E., Amer, S., Almutairi, L.A., Khalil, F.M.A., Bahshwan, S.M., AL-Qurashi, M.M. and Abd El-Aziz, Y.M. 2025. Pumpkin seed oil-loaded chitosan nanoparticles enhance growth, digestive enzymes, and immune response in whiteleg shrimp (Litopenaeus vannamei): impacts on histopathology and survival against Vibrio parahaemolyticus. Aquaculture Rep. 40, p.102599. Ekawati, S. R. 2008. Peningkatan Sintasan Dan Pertumbuhan Kepiting Bakau (Scylla olivacea) Stadia Zoea Melalui Aplikasi Pakan Alami Hasil Bioenkapsulasi Karotenoid Cangkang Kepiting Non Ekonomis (Doctoral dissertation, Universitas Hasanuddin). PhD diss. Elexson, N., Eddy, M.B., Wong, J.C.J., Lesen, D., Ransangan, J. and Thung, T.Y. 2023. Prevalence of multi-antibiotics resistant (MAR) Vibrio parahaemolyticus in shrimp farms in Sarawak, Malaysia. Food Res. 7(4), 127–136. El-Gohary, M.S., El Gamal, A.M., Atia, A.A. and El-Dakroury, M.F. 2020. Treatment Trial of Nile Tilapia (Oreochromis niloticus) Experimentally Infected with Vibrio alginolyticus Isolated from Sea bass (Dicentrarchus labrax). PJBS 23(12), 1591–1600. El-Houseiny, W., Arisha, A.H., Metwally, M.M.M., Abdel-Warith, A.W.A., Younis, E.M., Davies, S.J., Hassan, B.A. and Abd-Elhakim, Y.M. 2023. Alpha-lipoic acid suppresses gibberellic acid nephrotoxicity in Nile tilapia (Oreochromis niloticus) via modulating oxidative stress, inflammation, cytokine production, and apoptosis. Pesticide Biochem. Physiol. 196, 105598. Elinge, C.M., Itodo, A.U., Peni, I.J., Sani, U. and Elinge, C.M. 2012. Proximate, mineral and anti-nutrient composition of pumpkin (Cucurbita pepo L) seeds extract. Int. J. Plant. Res. 2(5), 146–150. FAO “ Food and Agriculture Organization”. 2022. The State of World Fisheries and Aquaculture. Rome, Italy, 266 p. George, S.A.S.S.S. 2020. Preparation of pumpkin pulp and peel flour and study their impact in the biscuit industry. J. Biol. Agric. Healthc. 10(6), 25–33. Ghenem, L. and Elhadi, N. 2018. Isolation, molecular characterization, and antibiotic resistance patterns of Vibrio parahaemolyticus isolated from coastal water in the Eastern Province of Saudi Arabia. J. Water. Health. 16(1), 57–69. Hussain, A., Kausar, T., Sehar, S., Sarwar, A., Ashraf, A.H., Jamil, M.A., Noreen, S., Rafique, A., Iftikhar, K., Quddoos, M.Y., Aslam, J. and Majeed, M.A. 2022. A Comprehensive review of functional ingredients, especially bioactive compounds present in pumpkin peel, flesh and seeds, and their health benefits. Food Chem. Adv. 1, 100067. Irshath, A.A., Rajan, A.P., Vimal, S., Prabhakaran, V.S. and Ganesan, R. 2023. Bacterial pathogenesis in various fish diseases: recent advances and specific challenges in vaccine development. Vaccines 11(2), 470. Jalili, R.B., Tukmechi, A., Agh, N., Noori, F. and Ghasemi, A. 2013. Replacement of dietary fish meal with plant sources in rainbow trout (Oncorhynchus mykiss); effect on growth performance, immune responses, blood indices and disease resistance. Iran. J. Fish. Sci. 12(3), 577–591. Jones, J.L., Lüdeke, C.H., Bowers, J.C., Garrett, N., Fischer, M., Parsons, M.B., Bopp, C.A. and DePaola, A. 2012. Biochemical, serological, and virulence characterization of clinical and oyster Vibrio parahaemolyticus isolates. J. Clin. Microbiol. 50(7), 2343–2352. Khattab, H.M., Singer A.M., Abdelhakeem, F. and Desoky, A.M. and Ahmed, R.A. 2024. Effect of replacing fishmeal with dietary pumpkin meal on the intestinal histology of the Nile Tilapia (Oreochromis niloticus). EJABF. 28(5), 1771–1779. Latif, M., Faheem, M., Asmatullah., Hoseinifar, S.H. and Van Doan, H. 2020. Dietary black seed effects on growth performance, proximate composition, antioxidant and histo-biochemical parameters of a culturable fish, rohu (Labeo rohita). Animals 11(1), 48. Lim, C., Klesius, P.H., Li, M.H. and Robinson, E. H. 2000. Interaction between dietary levels of iron and vitamin C on growth, hematology, immune response and resistance of channel catfish (Ictalurus punctatus) to Edwardsiella ictaluri challenge. Aquac 185(3-4), 313–327. Lulijwa, R., Rupia, E. J. and Alfaro, A. C. 2020. Antibiotic use in aquaculture, policies and regulation, health and environmental risks: a review of the top 15 major producers. Rev. Aquacult. 12(2), 640-663. Magnadóttir, B. 2006. Innate immunity of fish (overview). Fish. Shellfish. Immunol. 20(2), 137–151. Mamdouh, N., Khalil, S.A. and El-Naggar, M.M. 2023. Phenotypic and molecular characterization of vibrio species isolated from cultured marine Dicentrarchus labrax. Alex. J. Vet. Sci. 76(2), 49–57. Montesano, D., Blasi, F., Simonetti, M., Santini, A. and Cossignani, L. 2018. Chemical and nutritional characterization of seed oil from Cucurbita maxima L. (var. Berrettina) pumpkin. Foods 7(3), 30. Mounes, H.A.M., Abd-el Azeem, Z.M.A., Abd El-bary, D.A., Al-Sagheer, A.A., Abd-Elhakim, Y.M., Hassan, B.A., Sadek, S.S. and Ahmed, K.M. 2024. Effect of substituting soybean meal in Oreochromis niloticus diets with pumpkin (Cucurbita maxima) seed cake on water quality, growth, antioxidant capacity, immunity, and carcass composition. Animals 14(2), 195. Musthafa, M.S., Jawahar Ali, A.R., Arun Kumar, M.S., Paray, B.A., Al-Sadoon, M.K., Balasundaram, C. and Harikrishnan, R. 2017. Effect of Cucurbita mixta (L.) seed meal enrichment diet on growth, immune response and disease resistance in Oreochromis mossambicus. Fish. Shellfish. Immunol. 68, 509–515. Naiel, M.A., Fawzy, R.M., Zaki, E.M., Abo-Elmatty, A.M., El-Tarabily, K.A. and Negm, S.S. 2024. Pumpkin seed extract attenuates toxic effects of ochratoxin-contaminated diets in terms of growth performance, antioxidant and immunological indices of Nile tilapia. Aquac. Rep. 38, 102300. Negm, S.H. 2020. Effect of pumpkin (Cucurbita pepo L.) on immune system and liver functions of rats induced with liver cirrhosis. Int. J. Dairy. Food &. Environ. Sci. 3(1), 16–23. Nelapati, S., Nelapati, K. and Chinnam, B.K. 2012. Vibrio parahaemolyticus-An emerging foodborne pathogen-A Review. Vet. World 5, 48–62. Ngugi, C.C., Oyoo-Okoth, E., Mugo-Bundi, J., Orina, P.S., Chemoiwa, E.J. and Aloo, P.A. 2015. Effects of dietary administration of stinging nettle (Urtica dioica) on the growth performance, biochemical, hematological and immunological parameters in juvenile and adult Victoria Labeo (Labeo victorianus) challenged with Aeromonas hydrophila. Fish. Shellfish. Immunol. 44(2), 533–541. Nguyen, K.C.T., Truong, P.H., Truong, H.T., Ho, X.T. and Nguyen, P.V. 2024. Prevalence, multidrug resistance, and biofilm formation of Vibrio parahaemolyticus isolated from fish mariculture environments in Cat Ba Island, Vietnam. PHRP 15(1), 56–67. Ninčević Grassino, A., Rimac Brnčić, S., Badanjak Sabolović, M., Šic Žlabur, J., Marović, R. and Brnčić, M. 2023. Carotenoid Content and Profiles of Pumpkin Products and By-Products. Molecules 28(2), 858. Nkosi, C.Z., Opoku, A.R. and Terblanche, S.E. 2006. In vitro antioxidative activity of pumpkin seed (Cucurbita pepo) protein isolate and its in vivo effect on alanine transaminase and aspartate transaminase in acetaminophen-induced liver injury in low protein fed rats. Phytotherapy Res. 20, 780–783. Patel, R.K., Savalia, C.V., Kumar, R., Suthar, A.P., Patel, S.A., Patel, N.G., Kalyani, I.H. and Gupta, S. 2018. Determination of Vibrio parahaemolyticus from retail shrimp of South Gujarat of Navsari District and their drug resistance pattern. Int. J. Curr. Microbiol. Appl. Sci. 7(2), 2704–2710. Quintoil, M.N., Porteen, K. and Pramanik, A.K. 2007. Studies on occurrence of Vibrio parahaemolyticus in fin fishes and shellfishes from different ecosystem of West Bengal. Livest. Res. Rural Dev. 19(1), 1–6. Salehi, B., Capanoglu, E., Adrar, N., Catalkaya, G., Shaheen, S., Jaffer, M., Giri, L., Suyal, R., Jugran, A.K., Calina, D. and Docea, A.O. 2019. Cucurbits plants: A key emphasis to its pharmacological potential. Molecules, 24(10), 1854. Sanchooli, O., Moradloo, A.H. and Ghorbani, R. 2012. Measurement of alkaline phosphatase and lysozyme enzymes in epidermal mucus of different weights of Cyprinus carpio. World. J. Fish. &. Mar. Sci. 4(5), 521–524. Sausan, N. and Safrida MA Sarong. 2017. Pengaruh penambahan tepung labu kuning (Cucurbita moschata) dalam pakan buatan terhadap kualitas warna ikan maskoki (Carassius auratus). Jurnal Ilmiah Mahasiswa 2(2), 1–14. Sayed-Lafi, R.M., Shtewi, H.H., Sultan, F.A. and Al-Shammari, N.A.H. 2024. Pomegranate peel extract diet enhances health and immunity of common carp (Cyprinus carpio) against Aeromonas veronii. Open. Vet. J. 14(11), 2762. Sayed-Lafi, R.M., Sultan, F.A.M. and Al-Tameemi, R.A. 2024. Effect of diet with plant-derived bioactive compounds on physiological and tissular responses in the common carp (Cyprinus carpio). La. Técnica. 14(2), 95–102. Sezgin, A. and Aydın, B. 2021. Effect of replacing dietary soybean meal with pumpkin (Cucurbita pepo) seed cake on growth, feed utilization, haematological parameters and fatty acid composition of mirror carp (Cyprinus carpio). Aquac. Res. 52(11), 5870–5881. Shehu, R.A. and Saidu, Y. 2012. Screening for total carotenoids and β-carotene in some widely consumed vegetables in Nigeria. Niger. J. Basic Appl. Sci. 20(3), 225–229. Siddique, A.B., Moniruzzaman, M., Ali, S., Dewan, M.N., Islam, M.R., Islam, M.S., Amin, M.B., Mondal, D., Parvez, A.K. and Mahmud, Z.H. 2021. Characterization of pathogenic Vibrio parahaemolyticus isolated from fish aquaculture of the southwest coastal area of Bangladesh. Front. Microbiol. 12, 635539. Su, Y.C. and Liu, C. 2007. Vibrio parahaemolyticus: a concern of seafood safety. Food Microbiol. 24(6), 549–558. Subhashini, N., Krishnaiah, N., and Kiranmayi, C. B. 2011. Detection of Vibrio parahaemolyticus in shell fish by cultural and polymerase chain reaction. B-335. Teimouri, M., Amirkolaie, A.K. and Yeganeh, S. 2013. The effects of dietary supplement of spirulina platensis on blood carotenoid concentration and fillet color stability in rainbow trout (Oncorhynchus mykiss). Aquaculture 414, 224–228. Thirumalaikumar, V.P., Wagner, M., Balazadeh, S. and Skirycz, A. 2021. Autophagy is responsible for the accumulation of proteogenic dipeptides in response to heat stress in Arabidopsis thaliana. FEBS. J. 288(1), 281–292. Tietz, N.W. 1990. Blood gases and electrolytes.In Fundamentals of Clinical Chemistry. Burtis, C.A. and Bruns, D.E Philadelphia, PA: Sounders, pp: 903–8. Uribe, C., Folch, H., Enriquez, R. and Moran, G. 2011. Innate and adaptive immunity in teleost fish: a review. Vet. Med. 56(10), 486–503. Vakili, K., Khajehrahimi, A.E., Salimi, B., Kakoolaki, S. and Ghorbanzadeh, A. 2024. Evaluation of the effects of pumpkin seed extract (Cucurbita pepo) on growth factors, serum immune factors, and resistance to Yersinia ruckeri in rainbow trout (Oncorhynchus mykiss). J. Selva Andina Res. Soc. 15(1), 3–13. Velazquez-Roman, J., León-Sicairos, N., Flores-Villaseñor, H., Villafaña-Rauda, S. and Canizalez-Roman, A. 2012. Association of pandemic Vibrio parahaemolyticus O3: k6 present in the coastal environment of Northwest Mexico with cases of recurrent diarrhea between 2004 and 2010. Appl. Environ. Microbiol. 78, 1794–1803. Wang , L., Hua, X., Shi, J., Jing, N., Ji, T., Lv, B., Liu, L. and Chen, Y. 2022. Ochratoxin A: occurrence and recent advances in detoxification. Toxicon 210, 11–18. Wang, R., Zhong, Y., Gu, X., Yuan, J., Saeed, A.F. and Wang, S. 2015. The pathogenesis, detection, and prevention of Vibrio parahaemolyticus. Front. Microbiol. 6, 144. WHO. 2013. WHO traditional medicine strategy: 2014–2023. World Health Organization. Wootton, I.D.P. and Freeman, H. 1982. Microanalysis in medical biochemistry. London, UK: Churchill Livingstone, pp 91–114. Yanti, R.Z., Muchlisin, Z.A., Muhammadar, A.A. and Fadli, N. 2022. Application of pumpkin flour in the diet of Barramundi Lates calcarifer fingerling. Depik Jurnal. Ilmu-Ilmu. Perairan. Pesisir. Dan. Perikanan. 11(2), 176–181. Zhang, L. and Orth, K. 2013. Virulence determinants for Vibrio parahaemolyticus infection. Curr. Opin. Microbiol. 16, 70–77. Zhishen, J., Mengcheng, T. and Jianming, W. 1999. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 64(4), 555–559.A.O.A.C. 1990. Official Methods of Analysis. 15th Edition, Association of Official Analytical Chemist, Washington DC. | ||
| How to Cite this Article |
| Pubmed Style Al-shammari NA, Shtewi HH, Sultan FA, Sayed-lafi RM. The effect of a pumpkin flesh flour (Cucurbita pepo) diet on the health of Cyprinus carpio against Vibrio parahaemolyticus. Open Vet. J.. 2026; 16(2): 1060-1071. doi:10.5455/OVJ.2026.v16.i2.26 Web Style Al-shammari NA, Shtewi HH, Sultan FA, Sayed-lafi RM. The effect of a pumpkin flesh flour (Cucurbita pepo) diet on the health of Cyprinus carpio against Vibrio parahaemolyticus. https://www.openveterinaryjournal.com/?mno=277194 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.26 AMA (American Medical Association) Style Al-shammari NA, Shtewi HH, Sultan FA, Sayed-lafi RM. The effect of a pumpkin flesh flour (Cucurbita pepo) diet on the health of Cyprinus carpio against Vibrio parahaemolyticus. Open Vet. J.. 2026; 16(2): 1060-1071. doi:10.5455/OVJ.2026.v16.i2.26 Vancouver/ICMJE Style Al-shammari NA, Shtewi HH, Sultan FA, Sayed-lafi RM. The effect of a pumpkin flesh flour (Cucurbita pepo) diet on the health of Cyprinus carpio against Vibrio parahaemolyticus. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1060-1071. doi:10.5455/OVJ.2026.v16.i2.26 Harvard Style Al-shammari, N. A., Shtewi, . H. H., Sultan, . F. A. & Sayed-lafi, . R. M. (2026) The effect of a pumpkin flesh flour (Cucurbita pepo) diet on the health of Cyprinus carpio against Vibrio parahaemolyticus. Open Vet. J., 16 (2), 1060-1071. doi:10.5455/OVJ.2026.v16.i2.26 Turabian Style Al-shammari, Nadia Ali, Hanan Hussain Shtewi, Fatima Abulhussien Sultan, and Raad Muhammed Sayed-lafi. 2026. The effect of a pumpkin flesh flour (Cucurbita pepo) diet on the health of Cyprinus carpio against Vibrio parahaemolyticus. Open Veterinary Journal, 16 (2), 1060-1071. doi:10.5455/OVJ.2026.v16.i2.26 Chicago Style Al-shammari, Nadia Ali, Hanan Hussain Shtewi, Fatima Abulhussien Sultan, and Raad Muhammed Sayed-lafi. "The effect of a pumpkin flesh flour (Cucurbita pepo) diet on the health of Cyprinus carpio against Vibrio parahaemolyticus." Open Veterinary Journal 16 (2026), 1060-1071. doi:10.5455/OVJ.2026.v16.i2.26 MLA (The Modern Language Association) Style Al-shammari, Nadia Ali, Hanan Hussain Shtewi, Fatima Abulhussien Sultan, and Raad Muhammed Sayed-lafi. "The effect of a pumpkin flesh flour (Cucurbita pepo) diet on the health of Cyprinus carpio against Vibrio parahaemolyticus." Open Veterinary Journal 16.2 (2026), 1060-1071. Print. doi:10.5455/OVJ.2026.v16.i2.26 APA (American Psychological Association) Style Al-shammari, N. A., Shtewi, . H. H., Sultan, . F. A. & Sayed-lafi, . R. M. (2026) The effect of a pumpkin flesh flour (Cucurbita pepo) diet on the health of Cyprinus carpio against Vibrio parahaemolyticus. Open Veterinary Journal, 16 (2), 1060-1071. doi:10.5455/OVJ.2026.v16.i2.26 |