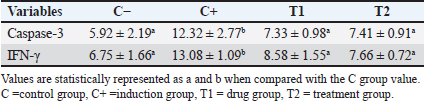| Research Article | ||
Open Vet. J.. 2026; 16(1): 337-345 Open Veterinary Journal, (2026), Vol. 16(1): 337-345 Research Article Beneficial immunomodulatory and hematological effects of bifidobacterium and lactobacillus probiotics in canine coronavirus vaccine induction in a murine modelIwan Sahrial Hamid1, Faisal Fikri1, Suryo Kuncorojakti1, Muhammad Thohawi Elziyad Purnama2, Salipudin Tasil Maslamama3, Saifur Rehman4 and Rondius Solfaine5*1Department of Veterinary Science, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 2Department of Biology, Graduate School of Natural and Applied Sciences, Eskişehir Osmangazi Üniversitesi, Eskişehir, Turkey 3Department of Agricultural Biotechnology, Faculty of Agriculture, Eskişehir Osmangazi Üniversitesi, Eskişehir, Turkey 4Department of Pathobiology, Faculty of Veterinary Sciences, University of Veterinary and Animal Sciences, Swat, Khyber Pakhtunkhwa, Pakistan 5Department of Pathology, Faculty of Veterinary Medicine, Universitas Wijaya Kusuma Surabaya, Surabaya, Indonesia *Corresponding Author: Rondius Solfaine. Department of Pathology, Faculty of Veterinary Medicine, Universitas Wijaya Kusuma Surabaya, Surabaya, Indonesia.. Email: rondius [at] uwks.ac.id Submitted: 15/08/2024 Revised: 12/12/2025 Accepted: 24/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
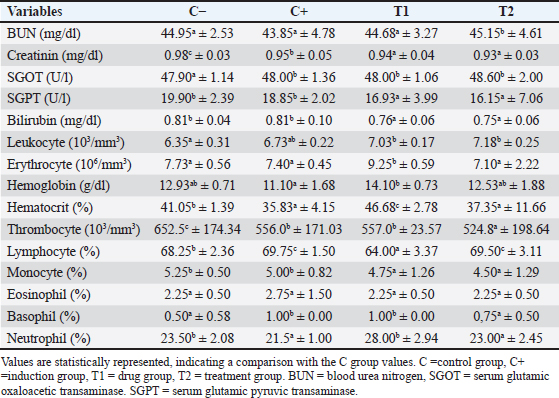
AbstractBackground: Recent coronavirus (CoV) outbreaks have raised global concerns regarding their impact on public health and animal populations. Probiotics, particularly Bifidobacterium spp. and Lactobacillus spp., are known to exert immunomodulatory and hematological benefits that may enhance vaccine responses. Aim: This study aimed to evaluate the immunomodulatory and hematological effects of Bifidobacterium and Lactobacillus probiotics in a canine CoV (CCoV) vaccine-inducing murine model. Methods: Twenty-four mice were randomly divided into the following four groups: control, CCoV induction, CCoV induction + isoprinosine, and CCoV induction + probiotics. The CCoV vaccine (60 mg/kg body weight) was administered subcutaneously for 7 days, while probiotics were administered orally for 14 days. On day 15, we collected blood samples for hematological and immunological assessments, including leukocyte count, hemoglobin, lymphocytes, neutrophils, caspase-3 (Casp-3), and interferon (IFN)-gamma (IFN-γ) levels. Lung and duodenum tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, and sectioned for hematoxylin–eosin staining. Immunohistochemistry staining for Casp-3 and IFN-γ was performed using specific primary antibodies (Abcam, UK), followed by HRP-conjugated secondary antibodies and 3,3-diaminobenzidine chromogen development. Sections were counterstained with hematoxylin and examined under a light microscope at 40× magnification. Results: The probiotic-treated group showed significant improvements in hematological parameters (leukocytes, hemoglobin, lymphocytes, and neutrophils) as well as reduced Casp-3 levels and increased IFN-γ expression compared with the other groups (p < 0.05). The T2 group exhibited reduced staining intensity in the mucosal and submucosal regions of the duodenum and alveolar structures of the lung, indicating the downregulation of apoptosis and inflammation compared with the C+ and T1 groups. Conclusion: Probiotic supplementation with Bifidobacterium and Lactobacillus demonstrated beneficial immunomodulatory and hematological effects in a murine CCoV vaccine model, supporting their potential role as adjuncts in CoV vaccination strategies. Keywords: Bifidobacterium, Canine coronavirus, Hematology, Isoprinosin, Probiotics. IntroductionCoronaviruses (CoVs) cause various diseases in animals, including respiratory and enteric infections, with canine CoV (CCoV) being a major pathogen in dogs. Although vaccination remains the primary preventive strategy, its effectiveness can vary due to strain diversity and host immune variability. Probiotics such as Lactobacillus spp. and Bifidobacterium spp. can exert immunomodulatory effects by enhancing mucosal immunity, regulating cytokine production, and reducing inflammation during viral infections. Because of their genetic uniformity, well-characterized immune responses, and extensive availability of immunological assays, murine models provide a well-established platform for evaluating immune mechanisms, making them suitable for studying viral pathogenesis and vaccine responses (Davis et al., 2017; Jin et al., 2020). CoVs are recognized as significant pathogens affecting both humans and animals, causing a range of diseases, particularly respiratory and gastrointestinal tract diseases (Yan et al., 219). Several zoonotic CoVs, such as severe acute respiratory syndrome CoV (SARS-CoV), Middle East respiratory syndrome (MERS), CoV (MERS-CoV), and SARS-CoV-2, have demonstrated the ability to cross species barriers and cause severe respiratory diseases, including SARS, MERS, and COVID-19, posing serious public health threats worldwide (Zhou et al., 2021). In addition to human infections, animal CoVs, such as the canine CoV (CCoV), also present substantial risks, particularly in domestic and livestock populations (Decaro and Buonavoglia, 2011). Although vaccination remains the primary strategy for combating CCoV, the use of probiotics to enhance host immune responses has emerged as a promising complementary approach (El-Shenawy et al., 2019; Kim et al., 2019). Probiotics, particularly Bifidobacterium and Lactobacillus strains, have been shown to enhance mucosal immunity, modulate gut microbiota, and reduce systemic inflammation (Zommiti et al., 2020). These microorganisms can strengthen the intestinal barrier, stimulate cytokine production, and regulate immune responses (Mathur et al., 2020). Furthermore, probiotics have been shown to suppress viral replication by activating pattern recognition receptors, leading to increased IFN-mediated antiviral responses (Singh and Rao, 2021). However, their specific role in modulating immune responses during CCoV infection or vaccination is still underexplored. Probiotics modulate host immunity and improve resistance against viral infections. Isoprinosin, an immunomodulatory and antiviral agent, was included in this study as a positive control because of its ability to enhance cellular immune responses and inhibit viral replication in infected hosts. To date, seven CoVs have been identified as capable of infecting humans, including SARS-CoV (2003) HCoV-NL63 (2004), HCoV-HKU1 (2005), MERS-CoV (2012), and SARS-CoV-2 (2019), along with HCoV-229E and HCoV-OC43 identified in the 1960s. SARS-CoV, MERS-CoV, and SARS-CoV-2 are classified as highly pathogenic (Decaro and Buonavoglia, 2011). Frequent outbreaks of CoV-related diseases in recent decades have heightened global awareness of their potential to cause pandemics. Besides human CoVs, numerous animal CoVs, such as TGEV, PEDV, and BCoV, are major pathogens in livestock (Costa et al., 2014; Gioti et al., 2021; Kong et al., 2024). Canine CoV (CCoV), feline CoV, and avian CoVs, such as infectious bronchitis virus, further underscore the cross-species risks and veterinary importance of this virus family. Antiviral responses are initiated by the production of IFNs, cytokines, and chemokines in infected hosts, leading to the activation of immune effector pathways such as apoptosis. The activation of caspase-mediated cell death pathways is a key mechanism (Galluzzi et al., 2018). For instance, CCoV type II has been shown to induce apoptosis in A-72 (a canine fibrosarcoma cell line frequently used for studying viral replication and cytopathic effects of canine viruses, including CCoV) cells through both intrinsic and extrinsic caspase pathways, including caspase-3 (Casp-3), -6, -8, and -9 activation and PARP cleavage (Gioti et al., 2021). The immunomodulatory potential of probiotics has gained considerable attention. Lactic acid bacteria, such as Streptococcus thermophilus, exhibit antimicrobial activities and support the development of functional foods and biopharmaceuticals (Mathur et al., 2020). Probiotics have shown protective effects against pathogenic bacteria and viruses in animal models (Villena and Kitazawa, 2017). Similarly, oral administration of L. plantarum significantly enhanced innate cytokine responses and natural killer cell activity, while reducing IL-6 levels in humans, suggesting its potential as a preventive strategy against viral infections, including COVID-19 (Kageyama et al., 2022). Despite these promising findings, little is known about the modulation of immune responses by probiotics in the context of CCoV vaccination. Therefore, this study aimed to evaluate the effects of probiotic supplementation, specifically Bifidobacterium spp. and Lactobacillus spp., on hematological parameters, serum biochemistry, Casp-3 expression, and IFN-gamma (IFN-γ) levels in mice immunized with the CCoV vaccine. The findings of this study may provide a foundation for the use of probiotics as immunomodulatory agents in both veterinary and human medicine. However, information regarding the role of probiotics in modulating immune responses and apoptosis-related pathways during CCoV infection or vaccination is limited. Therefore, this study aimed to evaluate the immunomodulatory and hematological effects of Bifidobacterium spp. and Lactobacillus spp. in a CCoV murine model. The administration of these probiotics could modulate hematological and immune responses, mitigate inflammation, and downregulate Casp-3 and IFN-γ expression in CCoV-induced mice. Materials and MethodsExperimental animals and study designTwenty-four healthy male BALB/c mice (Mus musculus) (6–8 weeks old, 25–30 g) were acclimatized for 1 week under standard laboratory conditions (22°C ± 2°C temperature, 55% ± 10% relative humidity, and a 12 hours light/dark cycle) with free access to feed and water. The mice were randomly assigned to four groups (n=6 per group): C (Control): received sterile distilled water (placebo), C+: induced with canine CoV (CCoV) vaccine, T1: CCoV + isoprinosin (100 mg/kg BW, orally for 14 days), T2: CCoV + probiotics (Lactobacillus acidophilus and Bifidobacterium bifidum, 1.05 × 1014 CFU/kg BW, orally for 14 days). The combination of Bifidobacterium spp. and Lactobacillus spp. was selected based on their synergistic effects in improving intestinal health and immune regulation. CCoV induction was performed using subcutaneous injection of an inactivated CCoV vaccine (Nobivac® CCoV, Merck, USA) containing ≥106.o TCID50 per dose for seven consecutive days (Decaro and Buonavoglia, 2011; Buonavoglia, 2022). Isoprinosin was administered orally once daily for 14 consecutive days, following the same schedule as the probiotic treatment, to allow direct comparison of their immunomodulatory effects. On day 15, mice were euthanized, and samples of blood, lung, and duodenum were collected for hematological and immunohistochemical evaluation. Isolation and maintenance of probiotic strainsTwo probiotic strains, Lactobacillus bulgaricus FNCC 0041 and B. bifidum BRL130, were obtained from the Food and Nutrition Study Center, Universitas Gadjah Mada (FNCC-UGM, Yogyakarta, Indonesia; Certificates No. FNCC-PSPG/06/I/2022 and FNCC-PSPG/46/II/2022). Both strains were cultured and maintained according to the FNCC-UGM microbiological procedures to ensure viability and purity. Lactobacillus bulgaricus FNCC 0041 was grown on De Man, Rogosa, and Sharpe (MRS) agar (Oxoid, UK) and incubated at 37°C for 48 hours under facultative anaerobic conditions. The strain was confirmed to be Gram-positive, nonmotile, catalase-negative, and lactic acid–producing from lactose, glucose, fructose, and mannose. The active cultures were preserved in MRS broth with 20% glycerol at −20°C and reactivated before use. Bifidobacterium bifidum BRL130 was maintained on MRS agar slants and subcultured into fresh MRS broth by sterile stab inoculation. Cultures were anaerobically incubated at 37°C for 48 hours and stored at 4 °C for up to 2 weeks. Before use, cells were subcultured twice to ensure metabolic activity and purity, as confirmed by Gram staining and colony morphology. Both isolates were verified as nonpathogenic and metabolically active, following the FNCC-UGM biosafety and identification criteria. Preparation of probiotic suspensionFor administration, each strain was cultured in MRS broth at 37°C for 24 hours. Cells were harvested by centrifugation (4,000 rpm, 10 minutes, 4°C), washed twice with sterile phosphate-buffered saline (pH 7.2), and resuspended in sterile distilled water. Cell density was adjusted spectrophotometrically (OD600=1.0 ≈ 1 × 109 CFU/ml). For the combined treatment, equal volumes (1:1, v/v) of L. bulgaricus and B. bifidum suspensions were mixed immediately before oral administration. Fresh suspensions were prepared daily and maintained on ice to preserve viability. Viable counts were confirmed by serial dilution and plating on MRS agar before dosing (Zommiti et al., 2020; Singh and Rao, 2021). Hematological and biochemical analyses were performedBlood samples (3 ml) were collected via cardiac puncture using ethylenediaminetetraacetic acid-coated and plain tubes (BD®, USA). After clotting for 10–20 minutes, the serum was separated by centrifugation at 4,000 rpm for 15 minutes (Hettich EBA 200®, Germany). Serum biochemical parameters were determined using an automated analyzer (Hitachi 902®, Roche Diagnostics, USA). Hematological parameters, including erythrocytes, leukocytes, hemoglobin (Hb), hematocrit (PCV), lymphocytes, neutrophils, and platelets, were evaluated according to standard hematological protocols (Purnama et al., 2021). Histopathology and immunohistochemistryLung and duodenum tissues were fixed in 10% neutral buffered formalin for 24–48 hours, dehydrated using graded ethanol (70%, 80%, and 96%), cleared with xylene, and embedded in paraffin at 60°C. Paraffin sections (4–5 µm) were cut with a microtome, floated on a 40–45°C water bath, and mounted on glass slides. Routine hematoxylin and eosin (H&E) staining was performed to evaluate histopathological changes. Sections were deparaffinized, rehydrated, and subjected to antigen retrieval in citrate buffer (pH 6.0) at 95°C for 15 minutes. Endogenous peroxidase activity was blocked using hydrogen peroxide (3%). Slides were incubated overnight at 4°C with primary antibodies against Casp-3 and IFN-γ (Abcam, UK), followed by HRP-conjugated secondary antibodies and visualization with 3,3-diaminobenzidine chromogen. The sections were counterstained with hematoxylin and observed under a light microscope at 40× magnification (Salazar et al., 2016; Hamid et al., 2018). The expression of Casp-3 and IFN-γ was semi-quantitatively evaluated using the H-score method, in which staining intensity (0=none, 1=weak, 2=moderate, 3=strong) was multiplied by the percentage of positive cells, yielding a total score from 0 to 300 for each sample. Statistical analysisAll quantitative data are expressed as mean ± SD. Statistical analysis was conducted using one-way analysis of variance, followed by Tukey’s post hoc test to assess intergroup differences. Differences were considered statistically significant at p < 0.05. Data analyses were performed using the Statistical Package for the Social Sciences version 25 (IBM Corp., Armonk, NY) (Mathur et al., 2020; Kosmerl et al., 2021). Ethical approvalAll experimental procedures were conducted in accordance with institutional and national regulations for animal research. The Animal Ethics and Care Committee, Faculty of Veterinary Medicine, Universitas Wijaya Kusuma Surabaya, Indonesia, approved this study (Approval No. 158-KKE). Animal welfare was prioritized throughout the study, and all procedures adhered to the principles of replacement, reduction, and refinement. ResultsHematological profileHematological analysis revealed significant improvements (p < 0.05) in leukocytes, hemoglobin, lymphocytes, and neutrophils among the treatment groups. Notably, the T2 group (administered Bifidobacterium spp. and Lactobacillus spp.) demonstrated significant differences (p < 0.05) in leukocyte, hemoglobin, and neutrophil levels compared with the T1 and C− groups. Leukocyte and lymphocyte counts significantly decreased (p < 0.05) in the T2 group compared with the positive control (C+), whereas hemoglobin levels significantly increased in the T2 group compared with the T1 and C+ groups. These findings suggest a beneficial modulation of hematological parameters following the administration of probiotics Bifidobacterium spp. and Lactobacillus spp. (Table 1). Table 1. Hematological and serum biochemical assessment.
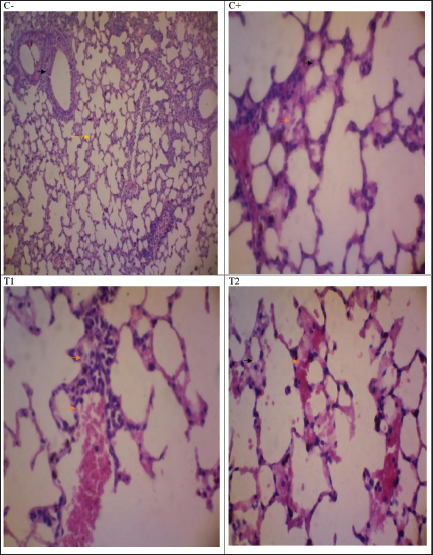
The observed increase in hemoglobin and erythrocyte counts suggests enhanced oxygen transport and erythropoietic activity, whereas leukocyte normalization reflects controlled immune activation rather than hyperstimulation. Villena and Kitazawa (2017) and Ibrahim et al. (2021) reported similar effects, demonstrating that probiotic supplementation promotes cytokine balance and supports hematopoietic recovery during viral or inflammatory challenges. In contrast, isoprinosin (T1) administration resulted in only mild immunostimulation, consistent with its known antiviral mechanism, but without significant hematological modulation. These hematological improvements corresponded with histopathological and immunohistochemical findings, which collectively demonstrated reduced inflammation and apoptosis in probiotic-treated mice. Thus, Lactobacillus and Bifidobacterium appear to exert dual actions, enhancing host defense mechanisms while maintaining hematological stability in CCoV-induced mice. Lung histopathologyThe histopathological analysis of the lung and duodenum tissues in the present study (Fig. 1) showed that the C+ group exhibited severe pathological changes, including multifocal alveolar septal destruction, inflammation, pulmonary emphysema (air space enlargement), hemorrhage, and leukocyte infiltration. Additional findings included interstitial edema, alveolar atrophy, atelectasis, and cellular debris accumulation within the alveolar lumen. In contrast, the T2 group exhibited fewer histological alterations, including inhibition of alveolar septal destruction, reduced inflammatory cell infiltration, and improved duodenal mucosal integrity, suggesting that probiotic administration has a protective effect against CCoV-induced tissue damage.
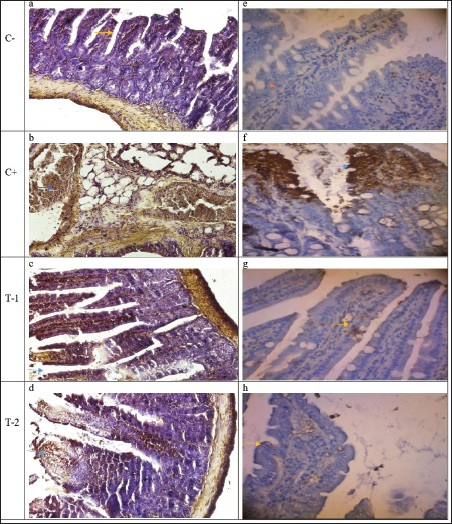
Fig. 1. Histological examination of the control group (C−) revealed typical cell structures in the lung tissue. In the C+ group, lung tissues exhibited histomorphological features such as diffuse alveolar cell damage, hyaline tissue deposition, septal alveolar hemorrhages, leukocyte cell infiltration, and alveolar septal proliferation. T1 showed mononuclear cell infiltration and congestion, along with plasma accumulation in lung tissue. The treatment group (T2) exhibited neutrophil cell infiltration and plasma fibrin accumulation in lung tissue (H&E, 40). Duodenal immunohistochemistry and cytokine expressionImmunohistochemical evaluation of duodenal tissues demonstrated significantly lower (p < 0.05) expression of Casp-3 and IFN-γ in the T2 group than in the C+ and T1 groups (Table 2; Fig. 2). The C+ group exhibited intense Casp-3 and IFN-γ staining, particularly within the mucosa and lamina propria, indicating apoptosis and heightened immune activation. In contrast, the T2 group exhibited mild Casp-3 and IFN-γ expression, maintaining normal mucosal architecture similar to that of the negative control (C−).
Fig. 2. The microscopic examination of small intestine tissue expressing Caspase-3 revealed distinct observations. The control group (C−) exhibited typical cell structures in the duodenal tissues without caspase-3 and IFN-γ expression (a,e). In contrast, the induction group (C+) displayed histomorphological features, including diffuse damage to enterocyte cells, hyaline tissue deposition, mucosal hemorrhages, leukocyte cell infiltration, and cell proliferation. Notably, the expression of Casp-3 and IFN-γ is observed prominently in the lamina propria and muscularis tunica area (b, f). Within the drug group (T1), noticeable indications include mononuclear cell infiltration, congestion, and plasma accumulation in intestinal tissues. Importantly, this reveals a moderate expression of Casp-3 and IFN-γ in the submucosa and lamina propria (c,g). Conversely, the treatment group (T2) demonstrated neutrophil cell infiltration and plasma fibrin accumulation in the duodenal tissues with mild expression of Casp-3 and IFN-γ in the mucosa and submucosa duodenal tissues (d,h) (immunohistochemistry, 40x). Table 2. Expression of Casp-3 and IFN-γ in the duodenum organs.
DiscussionIn the hematological evaluation, the T1 group (CCoV + isoprinosin) exhibited a marked improvement in several blood parameters compared with the negative control (C−) group. Notably, an increase in erythrocyte count, hemoglobin concentration, and PCV percentage was observed, reflecting a positive hematopoietic response to isoprinosin administration. The slight elevation in leukocyte levels suggests a mild immune system activation without evidence of pathological inflammation. Other hematological parameters, including platelet, lymphocyte, and monocyte counts, remained within normal physiological limits. Collectively, these findings indicate that isoprinosin exerts both hematoprotective and immunostimulatory effects in mice experimentally induced with canine CoV (CCoV). Histological examination of the control group (C−) reveals typical and well-preserved cellular structures in the lung tissue, indicating normal pulmonary architecture. In contrast, the induction group (C+) displayed marked pathological alterations, including diffuse alveolar cell damage, hyaline membrane formation, alveolar septal thickening, hemorrhage, and leukocyte infiltration within the interstitial and alveolar spaces. These findings are consistent with the findings of severe inflammatory responses and tissue injury induced by CCoV infection. In the drug-treated group (T1), the lung sections showed evidence of mononuclear cell infiltration, vascular congestion, and plasma accumulation, but with less extensive tissue damage compared with the C+ group. This indicates that isoprinosin treatment contributes to the partial protection of pulmonary tissue through its immunomodulatory and antiviral properties. The probiotic-treated group (T2) exhibited only mild histopathological changes, characterized by limited neutrophil infiltration and minimal plasma fibrin accumulation. The preservation of alveolar structure and reduced inflammatory cell infiltration in this group suggest that Bifidobacterium spp. and Lactobacillus spp. have a protective effect against CCoV-induced pulmonary injury. Overall, these histopathological observations support the role of probiotics in mitigating lung tissue damage and modulating inflammatory responses during viral infection. This aligns with the anti-inflammatory roles of Lactobacillus and Bifidobacterium species, which have been shown to mitigate respiratory inflammation and enhance epithelial integrity during viral infection (Kageyama et al., 2022). The T1 group exhibited moderate congestion and leukocyte infiltration, suggesting partial protection through the immunostimulatory effects of isoprinosin. Collectively, these results indicate that probiotic supplementation reduced CCoV-associated lung damage through immunomodulatory and anti-apoptotic mechanisms, consistent with the hematological stability observed in T2. A paradoxical reduction in IFN-γ expression was noted in the probiotic-treated group despite its known immunostimulatory potential. This finding likely reflects the regulatory rather than proinflammatory role of probiotics during viral challenge. Probiotics are known to balance cytokine responses by suppressing excessive IFN-γ while enhancing anti-inflammatory mediators, such as IL-10 and TGF-β (Villena and Kitazawa, 2017; Lehtoranta et al., 2020). Such modulation prevents tissue damage caused by immune overactivation, aligning with the reduced Casp-3 expression and preserved histological structures in the T2 group. Therefore, the downregulation of both Casp-3 and IFN-γ in probiotic-treated mice suggests that apoptotic and inflammatory signaling pathways are attenuated, supporting their role in maintaining intestinal and systemic immune homeostasis. Mechanistic insights and supporting evidenceThe beneficial outcomes observed in this study are consistent with the established probiotic mechanisms. Lactobacillus and Bifidobacterium spp. enhance mucosal immunity, modulate the gut–lung immune axis, and regulate cytokine responses to viral pathogens (Singh and Rao, 2021). These effects are attributed to their ability to strengthen epithelial barriers, compete with pathogens, and influence dendritic and T-cell responses. Previous research demonstrated that L. acidophilus and B. bifidum supplementation in animal models reduced inflammatory cytokines, improved hematological indices, and increased antioxidant activity (Ranji et al., 2019; Kosmerl et al., 2021). Additionally, L. plantarum administration in viral infection models significantly decreased weight loss, viral load, and pulmonary inflammation (Kageyama et al., 2022). Together, these findings support the current study’s evidence that probiotic supplementation mitigates inflammation, suppresses apoptosis, and enhances hematological balance in CCoV-induced mice. The metabolic characteristics of these probiotics also contribute to their efficacy. Bifidobacterium species use the bifidum pathway to produce acetate and lactate, generating ATP while maintaining gut pH balance and limiting pathogen growth (Kaplan and Hutkins, 2000). Lactobacillus casei and L. plantarum thrive under acidic conditions and metabolize diverse carbohydrates, contributing to microbial stability and immunological resilience (Barrons and Tassone, 2008; Salas-Jara et al., 2016). These characteristics collectively reinforce the dual systemic and local immunoregulatory potential of the aforementioned cells. Limitations and prospectsThis study has several limitations. The sample size was relatively small, which may restrict the generalizability of the findings. Moreover, while the murine model is suitable for immunological assessment, it may not fully replicate the physiological and immunopathological responses of CCoV infection in its natural canine host. The study also focused on acute outcomes, and long-term probiotic effects were not evaluated. Future research should explore chronic or field-based CCoV infection models using the target species, including larger populations, and investigate molecular pathways such as NF-κB and TLR signaling to further clarify the mechanisms of probiotic action. Nonetheless, the current findings provide foundational evidence that probiotics can be used as adjunct therapeutic agents to improve CoV vaccine responses and immune regulation. ConclusionIn conclusion, probiotic supplementation with Bifidobacterium spp. and Lactobacillus spp. effectively modulated hematological and immunological responses in mice induced with the canine CoV vaccine. The significant improvement in leukocyte, hemoglobin, lymphocyte, and neutrophil profiles, along with the downregulation of Casp-3 and IFN-γ expression, indicates the dual hematoprotective and anti-inflammatory actions of these probiotics. Histological and immunohistochemical findings further support their role in maintaining tissue architecture and reducing apoptotic activity. Collectively, these results underscore the therapeutic potential of Bifidobacterium and Lactobacillus as immunomodulatory adjuncts for enhancing vaccine efficacy and mitigating inflammatory responses during viral challenge. Future investigations using target animal species and long-term evaluations are warranted to validate and extend these findings under practical veterinary conditions. AcknowledgmentsThe authors would like to thank the laboratory staff and technical assistants from the Department of Veterinary Medicine and Institute of Tropical Disesase, Universitas Airlangga Surabaya, and Laboratory of Pathology, Universitas Wijaya Kusuma Surabaya, for their valuable support in animal handling, histopathological preparations, and immunohistochemical analysis. Conflict of interestThe authors declare no competing financial or nonfinancial interests that could have influenced the work reported in this paper. FundingThis study was financially supported by the Hibah Riset Mandat Khusus COVID-19 Universitas Airlangga, SK Number: 1039/UN3.15/PT/2021. Authors’ contributionsISH and FF designed the study. RS, SK, MT, and SR conceived the methodology and supervised the laboratory procedures. Histopathological and immunohistochemical analyses were conducted by STM and MT. ISH, FF, SR, and MT performed data analysis and interpretation. ISH drafted the manuscript, and all authors, including SR, contributed to the manuscript revision and approved the final version for submission. Data availabilityAll data supporting this study’s findings are included within the article. Additional datasets generated or analyzed during the current study are available upon reasonable request from the corresponding author. ReferencesBarrons, R. and Tassone, D. 2008. Use of Lactobacillus probiotics for bacterial genitourinary infections in women: a review. Clin. Therapeutics. 30(3), 453–468; doi:10.1016/j.clinthera.2008.03.012 Buonavoglia, C. 2022. Canine coronavirus infections. Vet. Clinics. North. Amer. Small. Anim. Pract. 52(2), 365–378; doi:10.1016/j.cvsm.2021.10.005 Costa, E.M., Silva, S. and Pintado, M.E. 2014. Coronaviridae in animals: a brief review. Ann. Anim. Sci. 14(3), 491–505; doi:10.2478/aoas-2014-0004 Davis, M.M., Tato, C.M. and Furman, D. 2017. Systems immunology: just getting started. Nature. Immunol. 18(7), 725–732; doi:10.1038/ni.3768 Decaro, N. and Buonavoglia, C. 2011. Canine coronavirus: not only an enteric pathogen. Vet. Clinics. North. Amer. Small. Anim. Pract. 41(6), 1121–1132; doi:10.1016/j.cvsm.2011.07.005 El-Shenawy, M., Fouad, M.T., Hassan, L.K., Seleet, F.L., El-Aziz, M.A. 2019. A probiotic beverage made from tiger-nut extract and milk permeate. Pak. J. Biol. Sci. 22(4), 180–187. Galluzzi, L., Vitale, I., Aaronson, S.A., Abrams, J.M., Adam, D., Agostinis, P., Alnemri, E.S., Altucci, L., Amelio, I., Andrews, D.W. and Annicchiarico-Petruzzelli, M. 2018. Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 25(3), 486–541. Gioti, V., Pavlidou, M. and Psalla, D. 2021. Canine coronavirus induces apoptosis in A-72 cells through caspase activation and PARP cleavage. J. Gen. Virol. 102(8), 1654; doi:10.1099/jgv.0.001654 Hamid, R., Rotshteyn, Y., Rabadi, L., Parikh, R. and Bullock, P. 2018. Evaluation of caspase-3 activation in tissue sections: a reliable marker of apoptosis. Apoptosis 23(5), 356–364; doi:10.1007/s10495-018-1455-9 Ibrahim, F., Halim, N.A. and Azhar, N. 2021. Role of probiotics in hematopoiesis and immune regulation: a review. Front. Nutr. 8, 678123; doi:10.3389/fnut.2021.678123 Jin, J., Hjerrild, K.A. and Feng, Y. 2020. The use of murine models in vaccine immunology research. Front. Immunol. 11, 599; doi:10.3389/fimmu.2020.00599 Kageyama, H., Tsuruhara, T. and Fujino, T. 2022. Oral administration of Lactobacillus plantarum enhances innate antiviral immunity and reduces influenza virus infection in mice. Microorganisms 10(4), 746; doi:10.3390/microorganisms10040746 Kaplan, H. and Hutkins, R.W. 2000. Metabolism of fructooligosaccharides by Lactobacillus paracasei 1195. Appl. Environ. Microbiol. 66(4), 1689–1695; doi:10.1128/AEM.66.4.1689-1695.2000 Kong, Q., Xiang, Z. and Zhang, J. 2024. Emerging animal coronaviruses: evolution, host adaptation, and zoonotic potential. Viruses 16(2), 320; doi:10.3390/v16020320 Kosmerl, E., Rocha-Mendoza, D., Ortega-Anaya, J., Jimenez-Flores, R. and Garcia-Cano, I. 2021. Bifidobacterium and Lactobacillus as potential probiotics for human health: a review of functional properties. Foods 10(6), 1306; doi:10.3390/foods10061306 Kim, S.K., Guevarra, R.B., Kim, Y.T., Kwon, J., Kim, H., Cho, J.H., Kim, H.B. and Lee, J.H. 2019. Role of probiotics in human gut microbiome-associated diseases. J. Microbiol. Biotechnol. 29, 1335–1340. Lehtoranta, L., Pitkäranta, A. and Korpela, R. 2020. Probiotics in respiratory virus infections. Eur. J. Clin. Microbiol. &. Infect. Dis. 39(4), 723–733; doi:10.1007/s10096-019-03703-4 Mathur, H., Beresford, T.P. and Cotter, P.D. 2020. Health benefits of lactic acid bacteria (LAB) fermentates. Nutrients 12(6), 1679; doi:10.3390/nu12061679 Purnama, M.T.E., Widyarini, S. and Handharyani, E. 2021. Hematological and biochemical parameters in mice treated with natural antioxidants. Vet. World 14(2), 476–483; doi:10.14202/vetworld.2021.476-483 Ranji, P., Haghshenas, B. and Khosroushahi, A.Y. 2019. Probiotic supplementation with Lactobacillus acidophilus and Bifidobacterium bifidum improves lipid profile and antioxidant status in colon cancer-induced mice. Nutr. Cancer. 71(6), 987–995; doi:10.1080/01635581.2019.1596824 Salas-Jara, M.J., Ilabaca, A., Vega, M. and García, A. 2016. Biofilm formation and virulence in Lactobacillus species. Microbial Pathogenesis 90, 1–9; doi:10.1016/j.micpath.2015.11.031 Salazar, N., Ruas-Madiedo, P. and Gueimonde, M. 2016. Bifidobacterium as a key member of the human gut microbiota: a review of its functional roles. Front. Microbiol. 7, 924; doi:10.3389/fmicb.2016.00924 Singh, V. and Rao, A. 2021. Probiotics in viral infections: mechanistic insights and potential clinical applications. Front. Microbiol. 12, 655064; doi:10.3389/fmicb.2021.655064 Villena, J. and Kitazawa, H. 2017. Probiotic microorganisms: a closer look at their beneficial effects on the host immune system. Front. Immunol. 8, 468; doi:10.3389/fimmu.2017.00468 Yan, S., Tian, Z., Li, M., Li, B. and Cui, W. 2019. Effects of probiotic supplementation on the regulation of blood lipid levels in overweight or obese subjects: a meta-analysis. Food Funct. 10(3), 1747–1759. Zhou, P., Yang, X.L., Wang, X.G., Hu, B., Zhang, L., Zhang, W., Si, H.R., Zhu, Y., Li, B., Huang, C.L., Chen, H.D., Chen, J., Luo, Y., Guo, H., Jiang, R.D., Liu, M.Q., Chen, Y., Shen, X.R., Wang, X., Zheng, X.S., Zhao, K., Chen, Q.J., Deng, F., Liu, L.L., Yan, B., Zhan, F.X., Wang, Y.Y., Xiao, G.F. and Shi, Z.L. 2021. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579(7798), 270–273; doi:10.1038/s41586-020-2012-7 Zommiti, M., Chikindas, M.L. and Ferchichi, M. 2020. Probiotics–lactic acid bacteria: classification, characteristics, and applications. Microorganisms 8(12), 1896; doi:10.3390/microorganisms8121896 | ||
| How to Cite this Article |
| Pubmed Style Hamid IS, Fikri F, Kuncorojakti S, Purnama MTE, Maslamama ST, Rehman S, Solfaine R. Beneficial immunomodulatory and hematological effects of bifidobacterium and lactobacillus probiotics in canine coronavirus vaccine induction in a murine model. Open Vet. J.. 2026; 16(1): 337-345. doi:10.5455/OVJ.2026.v16.i1.30 Web Style Hamid IS, Fikri F, Kuncorojakti S, Purnama MTE, Maslamama ST, Rehman S, Solfaine R. Beneficial immunomodulatory and hematological effects of bifidobacterium and lactobacillus probiotics in canine coronavirus vaccine induction in a murine model. https://www.openveterinaryjournal.com/?mno=277480 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.30 AMA (American Medical Association) Style Hamid IS, Fikri F, Kuncorojakti S, Purnama MTE, Maslamama ST, Rehman S, Solfaine R. Beneficial immunomodulatory and hematological effects of bifidobacterium and lactobacillus probiotics in canine coronavirus vaccine induction in a murine model. Open Vet. J.. 2026; 16(1): 337-345. doi:10.5455/OVJ.2026.v16.i1.30 Vancouver/ICMJE Style Hamid IS, Fikri F, Kuncorojakti S, Purnama MTE, Maslamama ST, Rehman S, Solfaine R. Beneficial immunomodulatory and hematological effects of bifidobacterium and lactobacillus probiotics in canine coronavirus vaccine induction in a murine model. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 337-345. doi:10.5455/OVJ.2026.v16.i1.30 Harvard Style Hamid, I. S., Fikri, . F., Kuncorojakti, . S., Purnama, . M. T. E., Maslamama, . S. T., Rehman, . S. & Solfaine, . R. (2026) Beneficial immunomodulatory and hematological effects of bifidobacterium and lactobacillus probiotics in canine coronavirus vaccine induction in a murine model. Open Vet. J., 16 (1), 337-345. doi:10.5455/OVJ.2026.v16.i1.30 Turabian Style Hamid, Iwan Sahrial, Faisal Fikri, Suryo Kuncorojakti, Muhammad Thohawi Elziyad Purnama, Salipudin Tasil Maslamama, Saifur Rehman, and Rondius Solfaine. 2026. Beneficial immunomodulatory and hematological effects of bifidobacterium and lactobacillus probiotics in canine coronavirus vaccine induction in a murine model. Open Veterinary Journal, 16 (1), 337-345. doi:10.5455/OVJ.2026.v16.i1.30 Chicago Style Hamid, Iwan Sahrial, Faisal Fikri, Suryo Kuncorojakti, Muhammad Thohawi Elziyad Purnama, Salipudin Tasil Maslamama, Saifur Rehman, and Rondius Solfaine. "Beneficial immunomodulatory and hematological effects of bifidobacterium and lactobacillus probiotics in canine coronavirus vaccine induction in a murine model." Open Veterinary Journal 16 (2026), 337-345. doi:10.5455/OVJ.2026.v16.i1.30 MLA (The Modern Language Association) Style Hamid, Iwan Sahrial, Faisal Fikri, Suryo Kuncorojakti, Muhammad Thohawi Elziyad Purnama, Salipudin Tasil Maslamama, Saifur Rehman, and Rondius Solfaine. "Beneficial immunomodulatory and hematological effects of bifidobacterium and lactobacillus probiotics in canine coronavirus vaccine induction in a murine model." Open Veterinary Journal 16.1 (2026), 337-345. Print. doi:10.5455/OVJ.2026.v16.i1.30 APA (American Psychological Association) Style Hamid, I. S., Fikri, . F., Kuncorojakti, . S., Purnama, . M. T. E., Maslamama, . S. T., Rehman, . S. & Solfaine, . R. (2026) Beneficial immunomodulatory and hematological effects of bifidobacterium and lactobacillus probiotics in canine coronavirus vaccine induction in a murine model. Open Veterinary Journal, 16 (1), 337-345. doi:10.5455/OVJ.2026.v16.i1.30 |