Open Veterinary Journal, (2026), Vol. 16(1): 265-274
Research Article
10.5455/OVJ.2026.v16.i1.24
Serological investigation of Toxoplasma gondii infection in urban goats from Makassar, Indonesia
Adriani Ris1, Raden Wisnu Nurcahyo2*, Dwi Priyowidodo2, Joko Prastowo2 and Rasdiyanah
Rasdiyanah3
1Doctoral Program of Veterinary Science, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Jakarta, Indonesia
2Department of Parasitology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Jakarta, Indonesia
3Veterinary Professional Education Study Program, Faculty of Medicine Hasanuddin University, Makassar, Indonesia
*Corresponding Author: Raden Wisnu Nurcahyo. Department of Parasitology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Jakarta, Indonesia. Email: wisnu-nc [at] ugm.ac.id
Submitted: 19/08/2025 Revised: 12/12/2025 Accepted: 27/12/2025 Published: 31/01/2026
© 2025 Open Veterinary Journal
This is an Open Access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited, and is not altered, transformed, or built upon in any way.
Abstract
Background: Makassar City, the largest metropolis in eastern Indonesia, represents a unique urban setting where the city core is becoming increasingly metropolitan, yet many peripheral districts continue to engage in small-scale mixed farming, including backyard goat keeping.
Aim: The aim of the study was to estimate the seroprevalence of Toxoplasma gondii in native goats (Capra hircus), also known locally as “Kambing Kacang,” kept in urban districts of Makassar, and to explore age- and sex-related risk patterns.
Methods: This cross-sectional study (November–December 2024) used stratified random sampling in three peri-urban sub-districts (Biringkanaya, Manggala, and Tamalate). Blood from 100 clinically healthy goats (≥ 6 months) was analyzed using a commercial indirect enzyme-linked immunosorbent assay (ID Screen® Toxoplasmosis Indirect Multi-species) to detect anti-T. gondii immunoglobulin G. The serostatus (positive ≥ 20 % S/P) was crosstabulated by age and sex; associations were tested with χ² at α=0.05.
Results: Twenty-five goats were seropositive, yielding an overall prevalence of 25% (95% confidence interval: 17%–34c%). Seroprevalence did not differ significantly by age (24%–26%; χ²=0.019; p=0.99) or sex (male 26.5 % vs. female 21.9 %; χ²=0.061; p=0.80). Spatially, prevalence ranged narrowly—21.1% in Manggala, 24.0% in Biringkanaya, and 25.4% in Tamalate (χ²=0.019; p=0.99).
Conclusion: 1 in four urban goats in Makassar carries T. gondii antibodies, with uniform exposure across demographic strata—implicating broad environmental contamination rather than focal risk factors. These findings highlight a tangible One-Health concern: backyard goat farming may sustain oocyst cycling close to human dwellings.
Keywords: Toxoplasma Gondii, Native goats, Urban livestock, Seroprevalence, ELISA.
Introduction
Toxoplasma gondii is a cosmopolitan protozoan parasite that infects virtually all warm-blooded animals and an estimated one-third of the world’s human population (Tenter et al., 2000; Dupont et al., 2023; Chen et al., 2024). Because infection is usually subclinical yet capable of causing severe disease in immunocompromised individuals and congenitally exposed infants, toxoplasmosis is recognized as a priority zoonosis by international public health bodies (De Barros et al., 2022; Kuruca et al., 2023). Transmission to humans occurs mainly through the ingestion of bradyzoite-containing tissue cysts in undercooked meat or oocysts shed by felids and disseminated in the environment; consequently, food-producing animals remain a cornerstone of preventive strategies (Pereira et al., 2010; Dzib Paredes et al., 2016; Shapiro et al., 2019). Transmission occurs when these species ingest sporulated oocysts through contaminated food, water, soil, or fomites (Dubey, 2022). For instance, small ruminants, such as goats, may become infected when grazing on pastures or consuming feed and water contaminated with felids, the definitive species. In humans, infection is often associated with the consumption of raw or undercooked meat containing tissue cysts or through the ingestion of oocyst-contaminated fresh produce and drinking water (Robert-Gangneux and Dardé, 2012). Thus, the critical factor in transmission is not the environmental presence of oocysts per se, but rather the exposure and ingestion pathways that facilitate parasite entry into the species. Among livestock species, goats are increasingly regarded as sensitive “sentinel” species (Stelzer et al., 2019; Dini et al., 2023). They browse close to the ground, drink from surface water sources, and share environments with free-roaming cats, all factors that increase their exposure to T. gondii oocysts (Shapiro et al., 2019).
However, most epidemiological evidence of T. gondii is derived from rural settings in temperate or arid regions (Abdul Hamid et al., 2020; Dahmane et al., 2024; Khan et al., 2024). Data from densely populated tropical cities in low- and middle-income countries remain scarce, despite rapid urban expansion that has blurred the boundary between peri-urban agriculture and residential neighborhoods (David et al., 2022; Qamar and Alsayeqh, 2023; Atif et al., 2024; Bokaba et al., 2024). Indonesia exemplifies this knowledge gap: only a few studies have addressed caprine toxoplasmosis, and virtually none have focused on urban livestock systems (Fatmawati et al., 2023; Sadiq et al., 2023; Mufa et al., 2025).
The largest metropolis in eastern Indonesia, Makassar City, serves as a compelling case study. This study provides a compelling case study for understanding toxoplasmosis epidemiology in an urban livestock context. Unlike rural goat populations, urban goats in Makassar are reared in close proximity to dense human settlements, often with limited biosecurity measures and reliance on household food waste as feed. These unique husbandry practices increase the likelihood of contact with T. gondii oocysts, which are abundant in urban environments, shed by domestic and stray cats. In this setting, goats represent not only a sentinel species for environmental contamination but also a potential source of zoonotic transmission to humans. Thus, the urban goat population in Makassar is an important model for investigating the interface of animal husbandry, public health, and urban ecology within the One Health framework. While the urban core is becoming increasingly metropolitan, many peripheral districts continue to engage in small-scale mixed farming, including backyard goat keeping. This phenomenon is not unique to Makassar in fact, similar patterns are observed across other major Indonesian cities, where rapid urbanization coexists with remnants of traditional agriculture (Rustiadi et al., 2021; Sroka and Żmija, 2021; Yuan et al., 2022). Peri-urban communities continue practice subsistence farming and smallholder livestock rearing, especially in transitional zones adjacent to rural hinterlands (Agung et al., 2021). These transitional zones, where urban density meets informal animal husbandry and high cat populations, present ideal conditions for the silent circulation of T. gondii (Barros et al., 2018; Meireles et al., 2004; Zhu et al., 2023).
Although some Indonesian studies have documented the seroprevalence of T. gondii in a range of animal hosts (Fatmawati et al., 2023; Puspitasari et al., 2024), information on the seroprevalence of T. gondii in native. Goats kept in urban settings are still remarkably limited. Clarifying the serological status of these goats is essential for gauging the zoonotic risk to nearby human populations and designing targeted control measures. Enzyme-linked immunosorbent assay (ELISA) remains the serological method of choice because it offers high sensitivity and specificity for detecting anti-T. gondii immunoglobulin G (IgG), a marker of past or chronic exposure (Dong et al., 2024; Kim et al., 2024; Toaleb et al., 2025). Against this backdrop, this study investigates the serological evidence of T. gondii infection in native goats reared within Makassar’s urban and peri-urban zones. Using a cross-sectional design and enzyme-linked immunosorbent assay-based antibody detection, we estimated seroprevalence and evaluated two basic animal-level risk factors goat age and sex Greenfield and Arnold, (2008). The resulting data will fill a critical gap in the epidemiology of toxoplasmosis in urban livestock systems and provide evidence to guide one-health-oriented public-health interventions.
Materials and Methods
Study design
A cross-sectional study using a stratified random sampling method was conducted from November to December 2024 to assess the seroprevalence of T. gondii infection in selected urban districts of Makassar, South Sulawesi, Indonesia. All procedures involving animals were conducted in accordance with established animal welfare standards.
Study area and animal samples
The study was conducted in three urban subdistricts of Makassar City, South Sulawesi, Indonesia: Biringkanaya, Tamalate, and Manggala. These subdistricts are located along the city’s peri-urban boundary and represent transitional zones between urban development and the remaining agricultural land (Fig. 1). Although increasingly encroached upon by housing and infrastructure, these areas still retain narrow strips of farmland, including spaces used for small-scale livestock rearing. In these districts, goat husbandry is typically integrated into household or vacant-lot environments, characterized by semi-intensive, low-input systems.
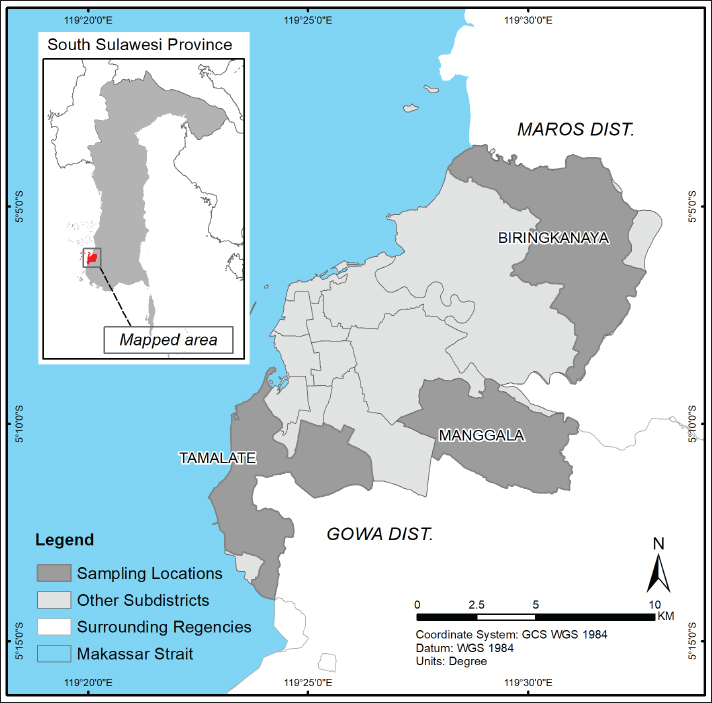
Fig. 1. Map showing the sampling locations in three urban subdistricts of Makassar City, South Sulawesi, Indonesia: Biringkanaya, Manggala, and Tamalate (Map vector Available via https://tanahair.indonesia.go.id).
The target population comprised clinically healthy goats. The goats used as samples in this study were first evaluated through a clinical examination to ensure good and representative health. The examination included general observations of consciousness, behavior, posture, and body condition score, all of which were within the normal range. Only clinically healthy goats were included, ensuring that the study results reflected the population’s health status without bias due to disease or other metabolic disorders.
Unvaccinated native goats (Capra hircus), also known locally as “Kambing Kacang” (Capra hircus, local “Kambing Kacang” breed), aged 6 months or older. All animals were reared under traditional smallholder management systems with limited or no confinement and were typically tethered near residential dwellings. Animals without visible signs of acute illness were included in the study. The minimum required sample size was calculated using a 95% confidence level, an expected prevalence of 30% based on prior regional data, and a desired precision of 10%. The target is only initial mapping/rough estimation to assess the level of “moderate” exposure in urban populations, and d=10% is rational (Arya et al., 2012), yielding a minimum of 81 animals. A total of 100 goats were selected using stratified random sampling to enhance statistical power and ensure proportional representation, with subdistricts serving as strata (Martin et al., 1987).
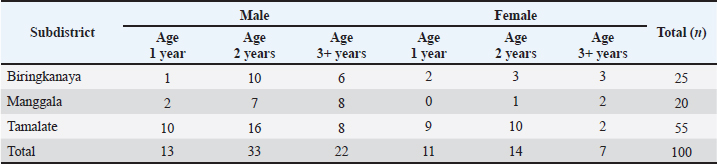
Table 1 summarizes the basic characteristics of the 100 goat populations sampled across the three selected subdistricts. All animals were reared under semi-intensive systems typical of a peri-urban environment. Age was determined by dental examination, and sex distribution reflected the natural structure of smallholder farmers’ backyard.
Table 1. Basic characteristics of the goat population sampled across the three selected subdistricts of Makassar, categorized by sex and age group.
Blood collection and serum preparation
Approximately 5 ml of blood was aseptically collected from the jugular vein of each goat using a vacutainer system with an 18-gauge needle, following standard veterinary protocols. The samples were transported in insulated cool boxes to the Serological Laboratory of the Maros Veterinary Center, where they were centrifuged at 3,000 rpm for 15 minutes. The resulting sera were transferred into labeled cryotubes and stored at −20°C until analysis.
ELISA testing procedure
Serological analysis was performed using a commercial indirect enzyme-linked immunosorbent assay kit (ID Screen® Toxoplasmosis Indirect Multi-species, ID.vet, France) to detect anti-T. gondii IgG antibodies. The assay was performed strictly following the manufacturer’s protocol. Optical density was measured at 450 nm using an enzyme-linked immunosorbent assay reader (Thermo Scientific Multiskan™, Thermo Fisher Scientific, Finland). The results were interpreted based on the sample-to-positive (S/P) ratio, with samples classified as seropositive when S/P% ≥ 20%, as recommended by the manufacturer.
Data analysis
All data were compiled and organized using Microsoft Excel, and further analysis was conducted using the R statistical software. The sample-to-positive percentage (S/P%) was the primary variable obtained from the ELISA test, as provided by the diagnostic laboratory. According to the manufacturer’s protocol (ID Screen® Toxoplasmosis Indirect Multi-species, ID.vet), the S/P% was calculated using the following formula:
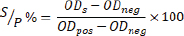
Where:
ODs=optical density of the sample, and ODpos=the optical density of the positive control, and ODneg=optical density of the negative control.
Samples with an S/P% ≥ 20% were classified as positive (seropositive), whereas those with an S/P% < 20% were considered negative (seronegative).
The distribution of seropositive and seronegative animals across subdistricts, age groups, and sex was summarized using descriptive statistics. For continuous data (S/P%), mean values and standard deviations were calculated within each subgroup to describe variability in antibody level. Associations between serostatus and animal-level risk factors (age and sex) were assessed using chi-square tests of independence, with statistical significance set at p < 0.05.
Ethical approval
Ethical approval for animal handling and blood collection was obtained from the Institutional Animal Care and Use Committee of Hasanuddin University under protocol number 010/UN4.1.RSHUH/BPP36/2025. Date:26/May/2025.
Results
A total of 25 of 100 goats (25.0%) tested seropositive for T. gondii, indicating a moderate level of exposure within the sampled urban population. Table 2 summarizes the distribution of serostatus across age groups, sex, and subdistricts, along with the results of the chi-square test to assess potential associations.
Table 2. Distribution of T. gondii serostatus by age, sex, and subdistrict with chi-squared results (n=100).
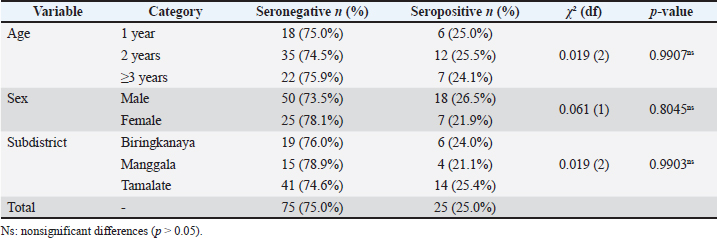
Overall, 25 of 100 goats (25.0%, 95% CI: 17%–34%) were seropositive for T. gondii. The median S/P% among positives was 128% (Q1=85%, Q3=180%), reflecting wide variability in antibody levels. No significant association was observed with age (p=0.99) or sex (p=0.80). Prevalence was consistent across subdistricts: 24.0%, 21.1%, and 25.4% in Biringkanaya, Manggala, and Tamalate, respectively (p=0.99). Collectively, these results indicate that exposure to T. gondii is evenly distributed across the sampled goat population, irrespective of age, sex, or subdistrict of origin.
Figure 2 summarizes the central tendency and spread of S/P% values for each demographic stratum. The titers were uniformly low across all seronegative goats (upper panels), with an overall mean of 3.9% ± 4.5 SD (range 0.02–19.9 p%). The three age classes differed only marginally: 1-year-old goats averaged 3.6 i%, 2-year-old goats 4.3 a%, and ≥ 3-year goats 3.6 a%, all with overlapping dispersions. In sharp contrast, seropositive animals (lower panels) displayed markedly higher readings, yielding an overall mean of 137% ± 69 SD (range, 20.4%–235.9a%). Mean titers rose from 98% ± 80 SD in the 1-year group to 151% ± 68 SD in 2-year goats, then stabilized at 146% ± 58 SD in theoldest group. However, the width of the boxes and whiskers shows that individual variation far outweighs these age-level differences. Dispersion was comparable between sexes: females averaged 134% ± 87 SD and males 138% ± 64 SD among seropositive, whereas both sexes remained below 5% among seronegative.
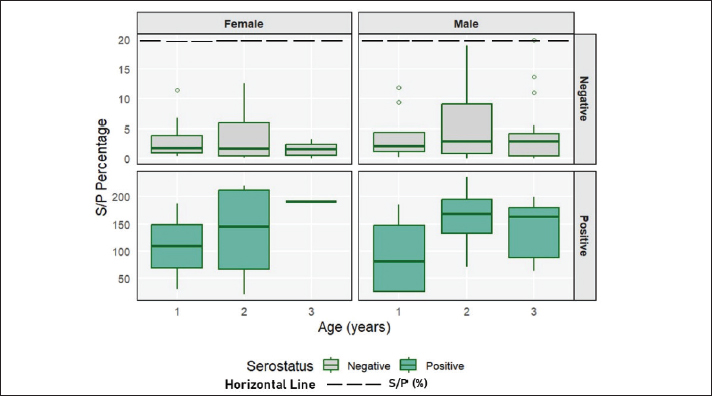
Fig. 2. Boxplot of the percentage distribution of S/P in goats categorized by age group, sex, and serostatus.
Figure 2. Boxplots of S/P percentage values of goats categorized by age group, sex, and serostatus. The boxes represent the interquartile range, the horizontal line within each box indicates the median, the whiskers show the minimum and maximum values, and the dots denote outliers. The cut-off threshold for seropositivity is indicated by a horizontal dashed line at 20%. Negative and positive samples are displayed together for clarity.
Interestingly, the variance of antibody titers appeared to be lower in older female goats than in their younger counterparts. This pattern suggests that once infected, older animals maintain more stable antibody levels, possibly due to immunological maturity or repeated low-level re-exposure. Conversely, the 2-year age group showed greater variability in titers, which could reflect heterogeneous exposure during this transitional stage—some individuals may have experienced primary infection with high antibody responses, while others remained naïve or had waning titers. Although not statistically significant, these patterns highlight the dynamic nature of host–parasite interactions across age and sex strata. Notably, formal statistical tests of variance were not performed; therefore, the observations are descriptive and intended to provide contextual interpretation rather than inferential conclusions.
Discussion
This cross-sectional survey provides the first serological snapshot of T. gondii exposure in native goats reared at the urban–peri-urban interface of Makassar. An overall IgG seroprevalence of 25% places this population close to the most recent global pooled estimate for goats (31.8%) generated from 225 studies across 70 countries (Ahaduzzaman and Hasan, 2022) and aligns with other moderate-prevalence reports such as in Somalia with 29.6% (Kakimori et al., 2024) and in South Punjab, Pakistan, which found seropositivity among goats of 31.5% (Khan et al., 2024). A comparable moderate prevalence with lower seropositivity was also observed in goats from Hunan Province, China, where a seropositivity rate of 13.3% was reported among 1,147 animals sampled from multiple farm types (Yi et al., 2024). In contrast, the levels reported for intensively managed herds elsewhere in Southeast Asia and North Africa are substantially below. For instance, lactating dairy goats in East Java exhibited extraordinarily high prevalences, 100% in Malang and 90.7% in Lumajang (Fatmawati et al., 2023); semi-intensive meat goats in Selangor, Malaysia, showed 69.8% seropositivity (Abdul Hamid et al., 2020); and smallholders in northeastern Algeria recorded 53.3% seropositivity (Dahmane et al., 2024). Likewise, in Espírito Santo, Brazil, Vicentini et al. (2024) reported a strikingly higher prevalence of 46.6% among goats sampled from peri-urban and rural municipalities, a figure nearly double that observed in Makassar. The differences observed across these contexts may reflect heterogeneity in herd management systems, environmental sanitation, feline population density, and public health infrastructure, underscoring the importance of localized epidemiological baselines for zoonotic risk assessment.
Contrary to initial expectations that cumulative environmental exposure would result in an age-dependent increase in seroprevalence, the data revealed a remarkably consistent pattern across age categories—25.0% in 1-year-olds, 25.5% in 2-year-olds, and 24.1% in goats aged ≥3 years. Seropositivity was also similar between the sexes (Fig. 2). These findings mirror observations from both high-prevalence settings, such as intensively reared dairy goats in East Java (Fatmawati et al., 2023), and moderate-prevalence populations, including smallholder flocks in Selangor (Sadiq et al., 2023) and southern China (Yi et al., 2024), where age and sex were not significant predictors of infection. Collectively, such uniformity supports the hypothesis that in endemic environments, most goats acquire T. gondii infection early in life, likely during their first grazing period when exposure to oocyst-contaminated substrates is highest (Dubey, 2021). The risk of new infection appears to plateau following this initial exposure, consistent with the development of lasting humoral immunity. Nevertheless, the wide dispersion of S/P % values among seropositive animals (ranging from just above the threshold to over 230%) suggests considerable heterogeneity in parasite burden or immunological responses. The slightly elevated median titers observed in 2-year-old animals may reflect the booster effects of intermittent re-exposure to T. gondii oocysts, a pattern also suggested in longitudinal studies from temperate and tropical settings (Stelzer et al., 2019; Dhuha et al., 2021).
Spatially, the seroprevalence of T. gondii varied only modestly across the three subdistricts studied (ranging from 21.1% to 25.4%), suggesting that the exposure pressure is not localized but rather reflects city-wide environmental contamination. This pattern is consistent with the high density and free-roaming nature of domestic cats observed throughout the study areas, reinforcing their role as primary oocyst environmental disseminators (Robert-Gangneux and Dardé, 2012; Shapiro et al., 2019; Vilela et al., 2024). The relative uniformity in seroprevalence across subdistricts is consistent with findings from other urban and peri-urban settings, suggesting that spatial gradients may be obscured by widespread baseline environmental contamination in certain ecological and management contexts (Chen et al., 2024).
This phenomenon may be further explained by the high degree of interconnectedness within urban goat and livestock husbandry systems, including frequent interactions with humans, animal keepers, and other potential host species. The frequent movement of animals across urban neighborhoods (for grazing, trade, or slaughter) can facilitate homogenized exposure patterns, effectively diluting localized differences in transmission pressure (Stelzer et al., 2019; Abdul Hamid et al., 2020; González-Barrio et al., 2024). Toxoplasma gondii seroprevalence in pigs and goats has been strongly associated with close human-animal interactions, unrestricted animal movement, and shared domestic spaces in smallholder and tribal livestock settings, such as those reported in northeastern India and northern Argentina. Exposure to cats and farm functions (e.g., breeding operations) significantly increased the risk of toxoplasmosis in pigs (Milton et al., 2024), whereas goats raised under family farming conditions with free-ranging dogs and communal land use had high rates of T. gondii and N. caninum coinfection (Basset et al., 2024).
From the perspective of One Health, even this moderate seroprevalence implies a substantial zoonotic reservoir. Approximately one in four slaughtered goats may carry viable tissue cysts that pose infection risks to consumers if undercooked. In Makassar, goat meat can be consumed on religious holidays such as Eid al-Adha, which sometimes uses undercooked meat. Furthermore, goats in yards are usually tethered <50 m from human habitation, thus increasing the possibility of oocyte exposure in the shared environment. As stated by Hadi (2022), several risk factors associated with the high seroprevalence of goat farming in South Sulawesi include natural mating, open pens that allow cats to enter freely, and grazing in grassy areas around residential areas, which allows goats to consume feed contaminated with cat feces. Toxoplasmosis is increasingly recognized as a leading foodborne parasitic disease in low- and middle-income countries, particularly due to the consumption of small-ruminant meat (Barros et al., 2018; Kuruca et al., 2023). Meat from goats has been identified as a significant transmission vehicle, especially during cultural or religious festivities, where undercooked preparations are more common (Dzib Paredes et al., 2016; Dubey, 2021). Moreover, recent assessments underscore the emerging threat posed by the consumption of raw or unpasteurized goat milk—a practice that is gaining popularity through informal markets and social media promotion (Pereira et al., 2010; Fatmawati et al., 2023). Although the primary transmission route remains ingestion of tissue cysts in meat, T. gondii has been detected in milk, and vertical transmission in humans and animals remains a concern in endemic regions (Tenter et al., 2000; Dupont et al., 2023).
Strength and limitations
This study provides valuable baseline information on T. gondii exposure among goats raised in urban and peri-urban environments of Makassar, Indonesia. The use of a validated multispecies ELISA ensured the reliable detection of anti-T. gondii IgG antibodies, allowing for a robust estimation of seroprevalence across demographic strata.
However, several limitations should be acknowledged. This study was conducted only during the rainy season (November–December), when higher humidity may prolong oocyst survival. Thus, the prevalence reported here may not fully represent seasonal variation in exposure risk. Toxoplasma gondii oocysts are known to remain viable longer under moist and cool conditions, which could potentially lead to an overestimation of prevalence during this period. Therefore, the failure to consider the impact of seasonality on exposure risk represents an important limitation that should be addressed in future studies through multi-seasonal sampling.
In addition, the absence of molecular confirmation (e.g., PCR or genotyping) limits the ability to distinguish between active and past infections or to determine circulating genotypes. Despite these constraints, this study provides the first critical insight into the epidemiology of caprine toxoplasmosis in urban livestock systems in Indonesia and establishes a foundation for broader One Health-oriented surveillance and control strategies. Future research should integrate both serological and molecular approaches to enhance epidemiological precision and comprehensively elucidate transmission dynamics.
Conclusion
This study demonstrates, for the first time, that T. gondii circulates at a moderate level among native Kacang goats reared in Makassar’s urban and peri-urban areas, with an overall IgG seroprevalence of 25%. The uniform distribution of seropositivity across age groups, sexes, and sub-districts indicates widespread environmental contamination. These findings underscore a significant One Health concern, as the close interaction between smallholder goats, stray cats, and dense human settlements may create overlooked pathways for zoonotic transmission. Despite limitations in sample size and diagnostic scope, the study’s rigorous design and focus on indigenous livestock strengthen confidence in the results. Overall, this study provides essential baseline evidence that highlights the urgent need for integrated control strategies to mitigate T. gondii transmission risks at the human–animal–environment interface.
Acknowledgments
The author expresses his gratitude and appreciation to the Indonesia Endowment Funds for Education and the Center for Higher Education Funding for Publication.
Conflict of interest
The authors declare no conflict of interest.
Funding
Scholarships were provided by the Indonesia Endowment Funds for Education and the Center for Higher Education Funding.
Authors’ contributions
AR, RWN, DP, and JP designed this study. AR and R field research and laboratory sample analysis. All authors wrote, edited, read, and approved the final version of the manuscript.
Data availability
All data supporting the findings of this study are available within the manuscript, and no additional data sources are required.
References
Abdul Hamid, N., Sadiq SZ Ramanoon., Mansor M Watanabe., Md Isa, J. Kamaludeen. and Syed-Hussain. 2020. Seroprevalence and risk factors of T. gondii in ruminant meats from wet markets in Klang Valley and abattoirs in Selangor, Malaysia. Animals 10(7), 1139; doi:10.3390/ani10071139
Agung, H., Benny, R. and Arif, S. 2021. A review of a food buffer system for urban areas in Jakarta: a case study. Afr. J. Agricult. Res. 17(1), 118–127; doi:10.5897/AJAR2020.15210
Ahaduzzaman, M. and Hasan, T. 2022. Seroprevalence of T. gondii infection in sheep and goats from different geographical regions of the world: systematic review and meta-analysis. Transboundary. Emerg. Dis. 69(6), 3790–3822; doi: 10.1111/tbed.14753
Arya, R., Antonisamy. and Kumar. 2012. Estimation of sample size in prevalence studies. Indian J. Pediatrics 79(11), 1482–1488; doi:10.1007/s12098-012-0763-3
Atif, I., Touloun. and Boussaa, S. 2024. Toxoplasma gondii in humans, animals and in the environment in Morocco: a literature review. Gut. Pathogens 16(1), 53; doi:10.1186/s13099-024-00645-5
Barros, M., Cabezón, O., Dubey, J.P., Almería, S., Ribas, M.P., Escobar, L.E., Ramos, B. and Medina-Vogel, G. 2018. T. gondii infection in wild mustelids and cats across an urban-rural gradient. PLos One. 13(6), e0199085; doi:10.1371/journal.pone.0199085
Basset, C., Gos, M.L., Steffen, K.D., Helman, E., Fitte, B., Olaizola, P.L. and Unzaga, J.M. 2024. Seroprevalence of T. gondii and Neospora caninum in goats from the Luracatao Valley, Salta, Argentina. Vet. Parasitology. Regional. Stud. Rep. 49, 100992; doi:10.1016/j.vprsr.2024.100992
Bokaba, R.P., Dermauw, V., Morar-Leather, D., Dorny, P., Van Schalkwyk, L. and Neves, L. 2024. Investigation of Toxoplasma gondii at the human-livestockwildlife interface, South Africa. Onderstepoort. J. Vet. Res. 91(1), 1–8; doi: 10.4102/ojvr.v91i1.2130
Chen, C., Qin, S., -Y.., Yang, X., Li, X., -M.., Cai, Y., Lei, C., -C.., Zhao, Q., Elsheikha, H.M. and Cao, H. 2024. Global prevalence and risk factors associated with T. gondii infection in wild birds: a systematic review and meta-analysis. Prev. Vet. Med. 226, 106187; doi:10.1016/j.prevetmed.2024.106187
Dahmane, A., Almeida, D., Reghaissia, N., Baroudi, D., Samari, H., Abdelli, A., Laatamna, A. and Mesquita, J.R. 2024. Seroprevalence Assessment and Risk Factor Analysis of T. gondii Infection in Goats from Northeastern Algeria. Animals 14(6), 883; doi:10.3390/ani14060883
David, S., Kela, S., Nkup, J. and Cirfat, N. 2022. Seroprevalence and risk factors associated with toxoplasmosis among women of child-bearing age in the Gombe metropolis, Gombe state, Nigeria. Microbes Infect. Dis. 0(0), 0; doi:10.21608/mid.2022.147764.1342
De Barros, R.A.M., Torrecilhas, A.C., Marciano, M.A.M., Mazuz, M.L., Pereira-Chioccola, V.L. and Fux, B. 2022. Toxoplasmosis in Humans and Animals. Diagnosis and Perspectives in the One-Healthy Approach. Acta Tropica 231, 1–106432; doi:10.1016/j.actatropica.2022.106432
Dhuha, J.M., Muayad, M.T.A., Saeed, O.A., Al-Bayar, M.A., Saeid, Z.J.M., Al-Bakri, S.A., Che Musa, H., Safaa, A., Abed, Q.N., Kaabi, S.A.G., Haniza, H.M.Z. and Shaari, A. 2021. Impact of tropical seasonal changes on hematological parameters of goats. J. Indonesian. Trop. Anim. Agriculture. 46(3), 219–226; doi:10.14710/jitaa.46.3.219-226
Dini, F.M., Morselli, S., Marangoni, A., Taddei, R., Maioli, G., Roncarati, G., Balboni, A., Dondi, F., Lunetta, F. and Galuppi, R. 2023. T. gondii spread among animals and humans in Northern Italy: a retrospective analysis in a One-Health framework. Food. Waterborne. Parasitology. 32, 197; doi:10.1016/j.fawpar.2023.e00197
Dong, H., Zhang, J., Wang, Q., Shen, Y., Zhou, B., Dai, L., Zhu, W., Sun, H., Xie, X., Xie, H., Xu, C., Zhao, G. and Yin, K. 2024. Development of an indirect ELISA for detecting T. gondii IgG antibodies based on a recombinant TgIMP1 protein. PLos Negl. Trop. Dis. 18(8), 12421; doi:10.1371/journal.pntd.0012421
Dubey, J.P. 2022. Sarcocystis neurona, Neospora spp. and Toxoplasma gondii infections in horses and equine protozoal myeloencephalitis (EPM): five decades of personal experience, perspectives and update. Parasitology 149(6), 717–728.
Dupont D Robert., Brenier, M.G., -Pinchart MP Lefevre., Wallon, A. and Pelloux, H. 2023. T. gondii, a plea for a thorough investigation of its oncogenic potential. Helicon 9(11), e22147; doi:10.1016/j.heliyon.2023.e22147
Dubey, J.P. Murata, F.H.A., Cerqueira-Cézar, C.K., Kwok, O.C.H. and Su, C. 2021. Epidemiological significance of Toxoplasma gondii infections in wild rodents: 2009-2020. J. Parasitol. 107, 182–204.
Dzib Paredes, G.F., Ortega-Pacheco, A., Rosado-Aguilar, J.A., Acosta Viana, K.Y., Guzmán-Marín, E. and Jiménez-Coello, M. 2016. T. gondii in meat for human consumption – A brief review of the most described strategies for its detection and quantification.In Significance, Prevention and Control of Food Related Diseases. UK: Jakun InTech, 39, pp: 69–79. https://doi.org/ 10.5772/63224
Fatmawati, M., Suwanti, L.T., Mufasirin, M., Fong, S., Valinata, S., Subekti, D.T., Ekawasti, F. and Awad, H.A.A. 2023. Serological survey of immunoglobulin G from T. gondii infection in East Java dairy goats. Vet. World. 1926, 1926–1932; doi:10.14202/vetworld.2023.1926-1932
González-Barrio, D., Carpio, A.J., Preite, L., Miguel-Vicedo, M., Estévez-Reboredo, R.M., González-Viadero, M., Barba-Sánchez, R., Calero-Bernal, R., Carmena, D. and Fuentes, I. 2024. T. gondii exposure in wildlife in Spain: is there any predictable threat for humans and domestic animals?. Sci. Total Environ. 935, 173290; doi:10.1016/j.scitotenv.2024.173290
Greenfield, H.J. and Arnold, E.R. 2008. Absolute age and tooth eruption and wear sequences in sheep and goat: determining age-at-death in zooarchaeology using a modern control sample. J. Archeological. Sci. 35(4), 836–849; doi:10.1016/j.jas.2007.06.003
Hadi, S. 2022. Uji Serologis Toxoplasmosis pada Kambing Lokal di Sulawesi Selatan dengan Metode ELISA. Jurnal Triton 13(1), 22–29; doi:10.47687/jt.v13i1.223
Kakimori, M.T.A., Osman, A.M., Silva, A.C.S., Ibrahim, A.M., Shair, M.A., Cavallieri, A.C., Barros, L.D., Garcia, J.L., Vieira, T.S.W.J., Hassan-Kadle, A.A. and Vieira, R.F.C. 2024. Serological and molecular detection of T. gondii and Neospora caninum in Somali ruminants. Parasitology Res. 123(11), 376; doi:10.1007/s00436-024-08397-6
Khan, M.Y., Barlaam, A., Gazzonis, A.L., Ferrari, N. and Giangaspero, A. 2024. Seroprevalence and risk factors of T. gondii infection in goats from the South Punjab Province, Pakistan. Vet. Parasitology. Regional Stud. Rep. 50, 101018; doi:10.1016/j.vprsr.2024.101018
Kim, M., Park, S.J. and Park, H. 2024. Trends in serological and molecular diagnostic methods for T. gondii infection. Eur. J. Med. Res. 29(1), 520; doi:10.1186/s40001-024-02055-4
Kuruca L Belluco., Vieira, S., -Pinto M Antic. and Blagojevic, B. 2023. Current control options and a method for risk-based control of T. gondii in the meat chain. Food Control 146, 109556; doi:10.1016/j.foodcont.2022.109556
Martin, S.W., Meek, A.H. and Willeberg, P. 1987. Veterinary epidemiology: principles and methods. Iowa State University Press. Sudan: Iowa State University Press.
Meireles, L.R., Galisteo, A.J., Pompeu, E. and Andrade, H.F. 2004. T. gondii spreading in an urban area evaluated by seroprevalence in free-living cats and dogs. J. Vet. Pathol. Trop. Med. Int. Health 9(8), 876–881; doi:10.1111/j.1365-3156.2004.01280.x
Milton, A.A.P., Das, S., Ghatak, S., Srinivas, K., Priya, G.B. and Angappan M. 2024. Seroepidemological investigation of Toxoplasma gondii and Trichinella spp. in pigs reared by tribal communities and small-holder livestock farmers in Northeastern India. PLoS One 19(2), e0298357; doi: 10.1371/journal.pone.0298357
Mufa, R.I.M., Eka Pramyrtha Hestianah, Lucia., Hani, T.S., Widya, P., Tony, P.L., Rina, H., Retno, V., Heni, Y., Puspitasari. and Mufasirin. 2025. Galaxy Guardian. Toxoplasma oocyst contamination in water sources in goat sheep farming in Sidoarjo District. J. Parasite Sci. 9(1), 20–24; doi:10.20473/jops.v9i1.67669
Pereira, K.S., Franco, R.M.B. and Leal, D.A.G. 2010. Transmission of Toxoplasmosis by Foods.In Advances in Food and Nutrition Research. Ed., Taylor, S.L. Rio de Janeiro, Brazil: Academic Press, 60, pp: 1–9. https://doi.org/ 10.1016/S1043-4526(10)60001-0
Puspitasari, H., Suwanti, L.T., Mufasirin, M., Kusnoto, K., Yudaniayanti, I.S., Setiawan, B., Suprihati, E., Aksono, E.B., Widodo, D.P., Wardhana, A.H., Matsubayashi, M. and Indasari, E.N. 2024. Molecular detection of toxoplasmosis in wild rats using a loop-mediated isothermal amplification assay. Vet. World. 1575, 1575–1580; doi:10.14202/vetworld.2024.1575-1580
Qamar, W. and Alsayeqh, A.F. 2023. Foodborne T. gondii with a special focus on its prevalence in Pakistan from 2000 to 2022: a review. Front. Vet. Sci. 9, 1080139; doi:10.3389/fvets.2022.1080139
Robert-Gangneux, F. and Dardé, M.L. 2012. Epidemiology and Diagnostic Strategies for Toxoplasmosis. Clin. Microbiol. Rev. 25(2), 264–296; doi:10.1128/CMR.05013-11
Rustiadi, E., Pravitasari, A.E., Setiawan, Y., Mulya, S.P., Pribadi, D.O. and Tsutsumida, N. 2021. Impact of continuous Jakarta megacity urban expansion on the formation of the Jakarta-Bandung conurbation over the rice farm regions. Cities 111, 103000; doi:10.1016/j.cities.2020.103000
Sadiq, M.B., Muhamad, A.S., Hamdan, S.A., Ramanoon, S.Z., Zakaria, Z., Aziz, N.A.A., Mansor, R., Arshad, S.S., Khalid, N., Abdul Hamid, N., Kamaludeen, J. and Syed-Hussain, S.S. 2023. Seroprevalence and Factors Associated with T. gondii, Neospora caninum, and Besnoitia besnoiti Infections in Cattle and Goats in Selangor, Malaysia. Animals 13(5), 948; doi:10.3390/ani13050948
Shapiro, K., Bahia-Oliveira, L., Dixon, B., Dumètre, A., De Wit, L.A., VanWormer, E. and Villena, I. 2019. Environmental transmission of T. gondii: oocysts in water, soil, and food. Food Waterborne Parasitology 15, 49; doi:10.1016/j.fawpar.2019.e00049
Sroka, W. and Żmija, D. 2021. Changes in Urban Farming Systems: a Mixed Approach Based on Statistical Analysis and Expert Surveys. Agriculture 11(5), 455; doi:10.3390/agriculture11050455
Stelzer, S., Basso, W., Benavides Silván, J., Ortega-Mora, L.M., Maksimov, P., Gethmann, J., Conraths, F.J. and Schares, G. 2019. T. gondii infection and toxoplasmosis in farm animals: risk factors and economic impact. J. Vet. Med. Food Waterborne Parasitology, 15; doi: 10.1016/j.fawpar.2019.e00037
Tenter, A.M., Heckeroth, A.R. and Weiss, L.M. 2000. T. gondii: from animals to humans. Int. J. For Parasitology 30(12–13), 1217–1258; doi:10.1016/S0020-7519(00)00124-7
Toaleb, N.I., Shaapan, R.M. and Aboelsoued, D. 2025. Comparison of commercial ELISA, in-house indirect ELISA, and dot-ELISA developed for the detection of T. gondii antibodies in farm animals. J. Vet. Diagnostic Invest. 10406387251331637, 10406387251331637; doi:10.1177/10406387251331637
Vicentini, L.P.P., Damiani, L.S.A., Beltrame, M.A.V., Santos, G.L., Mazuz, M.L., Müller, R.A., Lopes, R.E.N., Vitor, R.W.A. and Fux, B. 2024. Seroprevalence and risk factors of T. gondii infection in goats (Capra hircus) in the state of Espírito Santo, Southeastern Brazil. Vet. Parasitology. Regional. Stud. Rep. 51, 101032; doi:10.1016/j.vprsr.2024.101032
Vilela, V.L.R., Feitosa, T.F., Simões, S.V.D., Mota, R.A., Katzer, F. and Bartley, P.M. 2024. An abortion storm in a goat farm in Northeast Brazil was caused by the atypical T. gondii genotype #13. Curr. Res. Parasitology Vector-Borne Dis. 5, 100157; doi:10.1016/j.crpvbd.2023.100157
Yi, X., -L.., Yang, W., -H.., Zheng, H., -L.., Cao, M., -L.., Xiong, J., Chen, W., -C.., Zhou, Y., -J.., Li, F., Zhu, X., -Q.., Liu, G. and -H. 2024. Seroprevalence and molecular detection of T. gondii and N. caninum in beef cattle and goats in Hunan province, China. Parasites. Vectors. 17(1), 195; doi:10.1186/s13071-024-06283-9
Yuan, G.N., Marquez, G.P.B., Deng, H., Iu, A., Fabella, M., Salonga, R.B., Ashardiono, F. and Cartagena, J.A. 2022. Urban agriculture: technology, socio-economy, and policy. Helicon 8(11), e11583; doi:10.1016/j.heliyon.2022.e11583
Zhu, S., VanWormer, E. and Shapiro, K. 2023. More people, more cats, more parasites: human population density and temperature variation predict prevalence of T. gondii oocyst shedding in free-ranging domestic and wild felids. PLos One. 18(6), 286808; doi:10.1371/journal.pone.0286808