| Review Article | ||
Open Vet. J.. 2026; 16(1): 41-55 Open Veterinary Journal, (2026), Vol. 16(1): 41-55 Review Article Preclinical studies of conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM for ischemic stroke: A systematic reviewRatih Rinendyaputri1,2, Uly Alfi Nikmah1, Novaria Sari Dewi Panjaitan1, Putri Reno Intan1, Jiro Hasegawa Situmorang1, Hasta Handayani Idrus1, Rilianawati Rilianawati3, Wireni Ayuningtyas4, Rachmawati Noverina4, Fathul Huda5 and Ahmad Faried6*1Center for Biomedical Research, Research Organization for Health, National Research and Innovation Agency, Bogor, Indonesia 2Doctoral Program, Faculty of Medicine, Universitas Padjadjaran, Bandung, Indonesia 3Research Center for Vaccine and Drugs, National Research and Innovation Agency (BRIN), PUSPIPTEK, Tangerang Selatan, Indonesia 4Bio Farma Stem Cell Research and Development, Bandung, Indonesia 5Department of Biomedical Sciences, Faculty of Medicine, Universitas Padjadjaran, Sumedang, Indonesia 6Neurosurgery Department, Faculty of Medicine, Universitas Padjadjaran, Bandung, Indonesia *Corresponding Author: Ahmad Faried. Neurosurgery Department, Faculty of Medicine, Universitas Padjadjaran, Bandung, Indonesia. Email: ahmad.faried [at] unpad.ac.id Submitted: 26/08/2025 Revised: 11/11/2025 Accepted: 30/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractThe current treatment for ischemic stroke, tissue plasminogen activator (tPA), is limited by a narrow therapeutic window, necessitating the development of adjuvant therapies. Hypoxic preconditioning is a strategy to improve the therapeutic potential of mesenchymal stem cells. The secretome of hypoxic mesenchymal stem cell-conditioned medium (hypMSC-CM) is an effective therapy for vascular lesions like stroke, as it promotes angiogenesis, neurogenesis, and neuroprotection. This systematic review aimed to assess the efficacy of hypMSC-CM in in vivo and in vitro stroke models to inform future clinical trials on stroke treatment. A systematic search was conducted for studies on hypMSC-CM in ischemic stroke models using the PubMed, Scopus, and Google Scholar databases up to December 2024. A total of 91 articles were identified, of which six met the eligibility criteria. Data were extracted from six studies (three in vivo and three in vitro), and their therapeutic outcomes are summarized in tables. All included studies consistently demonstrated beneficial effects of hypMSC-CM, with findings of reduced infarct volume, improved neurological function, and the promotion of neurogenesis and angiogenesis. This systematic review suggests that hypMSC-CM enhances neurogenesis and neuroprotection in the brain. However, high-quality preclinical studies and clinical trials are needed to support the therapeutic benefits of hypMSC-CM for ischemic stroke, especially as an adjuvant to standard tPA therapy. Keywords: Conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM, Stroke ischemic, Neuroprotection, Angiogenesis, Mesenchymal stem cell/MSC. IntroductionStroke remains one of the leading causes of mortality and a major contributor to long-term disability worldwide, posing a substantial burden on global public health systems (WHO, 2024). It is primarily categorized into ischemic stroke, which is caused by brain ischemia due to thrombosis, and hemorrhagic stroke. Ischemic stroke is more common and is responsible for a substantial proportion of stroke-related deaths and disabilities (Yuan et al., 2022; Fan et al., 2023). The current pharmacological treatment for ischemic stroke includes antiplatelet agents, anticoagulants, stent implantation to resolve arterial stenosis caused by atherosclerotic plaques, and recombinant tissue plasminogen activators (tPA) for reperfusion therapy (Kim, 2019). tPA dissolves thrombi and restores cerebral blood flow (Hasan et al., 2021). However, tPA clinical use is limited by a narrow therapeutic window and an increased risk of hemorrhagic complications (Quan et al., 2023; Jiang et al., 2024; Lin et al., 2024). These therapies are limited in preventing long-term neuronal death and are effective only within 4.5–6 hours after stroke onset. In patients over 80 years of age, the risks and benefits must be carefully considered (Peña I Dela et al., 2017; Bluhmki et al., 2020). In addition to pharmacological therapy, endovascular thrombectomy has become the standard of care for patients with large-vessel occlusion, demonstrating improved outcomes when performed within the appropriate time window (Goyal et al., 2016). Nevertheless, the efficacy of these treatments remains limited, particularly for patients who do not meet eligibility criteria (Nguyen et al., 2022). Therefore, alternative and adjuvant strategies are needed to promote neuroregeneration and improve functional recovery. To address these limitations, adjuvant therapies such as antioxidants, vitamins, and emerging biotechnology products, including stem cells and their secretomes, have been proposed as potential neuroprotective strategies (Qian et al., 2016; Yu et al., 2022). Among these, mesenchymal stem cells (MSCs) and their secretome have shown promise in promoting neuron regeneration and neuroprotection (Zacharek et al., 2007; Lapi et al., 2015; Ghazavi et al., 2017; Han et al., 2023). Therapy using MSCs, both autologous and allogenic, produced superior results compared to placebo, demonstrating safety as well as motor recovery and neuroprotective effects in ischemic stroke (Laskowitz et al., 2018; Jaillard et al., 2020; Yaqubi and Karimian, 2024). Furthermore, the clinical relevance of MSC-based therapy has been validated in various disease contexts. Clinical studies in idiopathic pulmonary fibrosis have demonstrated that MSC administration is safe and well-tolerated, with indications of potential therapeutic benefit, thereby endorsing the translational applicability of MSC-based interventions beyond neurological disorders (Chelluri et al., 2017). This is because the MSC secretome contains various growth factors, cytokines, and chemokines that contribute to neurogenesis, angiogenesis, and reduction of infarct size (Gutiérrez-Fernández et al., 2015; Abd El Motteleb et al., 2018). Hypoxic preconditioning is a strategy to enhance the bioactive secretions of MSCs, complementing other methods such as three-dimensional culture systems (Kim et al., 2015; Toghiani et al., 2024). Hypoxic preconditioning is a method for increasing bioactivity and has been reported for diabetes, as well as various organ damage (Kahrizi et al., 2023; Forkan et al., 2025). Optimization was carried out under hypoxic conditions of 1.5% oxygen. Success was indicated by measuring the level of extracellular vesicle (EV) concentration and protein concentration (Liu et al., 2021a,b; Song et al., 2025). Several studies have reported the efficacy of hypoxia preconditioning in the regeneration of lung, renal, and neuronal organs (Wang et al., 2015; Luo et al., 2019; Xu et al., 2022; Braga et al., 2023; Kuntjoro et al., 2024; Yuan et al., 2024). Importantly, secretomes derived from MSCs preconditioned under hypoxic conditions have been reported to exhibit enhanced therapeutic potential compared to normoxic MSC-derived secretomes, as demonstrated in several comparative preclinical studies, as reported in several comparative studies evaluating neuroprotective and angiogenic outcomes (Mazumder and Borah, 2015; Chrostek et al., 2019; Trzyna and Banaś-Ząbczyk, 2021; Agarwal et al., 2022). Citicoline may phosphorylate phosphoinositide 3-kinases (PI3K) and extracellular-signal-regulated kinase (ERK), activating the synthesis of vascular endothelial growth factor (VEGF) and fibroblast growth factor (FGF) proteins, which are involved in the angiogenesis process. Piroxicam also inhibits N-methyl-D-aspartate receptors, modulating calcium influx in neurons and thereby reducing excitotoxicity (Mazumder and Borah, 2014; Bermejo et al., 2023). Various investigations of MSC and neural stem cell (NSC) cells have demonstrated beneficial effects on neuronal regeneration (Xu et al., 2010; Guan et al., 2014; Kong et al., 2017). However, accumulating evidence suggests that the therapeutic benefits observed in MSC-based interventions are predominantly mediated by paracrine effects rather than direct cell replacement, thereby supporting the development of cell-free approaches such as MSC-derived conditioned medium (CM) (Cunningham et al., 2018; Li et al., 2021). Although MSCs demonstrate behavioral and neuroprotective benefits in stroke models, the optimal dosage, route, and timing of administration remain under active investigation (Zhang et al., 2021; Yang et al., 2022). Novel approaches, limits of available conventional therapy for ischemic stroke, and cell-free therapeutic approaches based on the MSC secretome are gaining importance. In preclinical and clinical experiments, the paracrine effect of MSC secretome has been shown to have neuroprotective effects, promote neurogenesis, stimulate angiogenesis, and reduce the infarct area (Cunningham et al., 2018; Castelli et al., 2020; Gwam et al., 2021). The MSC secretome includes neurotrophic factors that promote neurogenesis, including brain-derived neurotrophic factor (BDNF), glial-derived neurotrophic factor (GDNF), and nerve growth factor/NGF, as well as chemokines like VEGF and stromal-derived factor-1 (SDF-1), which cause progenitor cells to migrate to the penumbra. This process enhances neurogenesis, neuroprotection, and angiogenesis, facilitating regeneration in the infarct region (Wang et al., 2007; Zhou et al., 2017; Noverina et al., 2020). Culture modification can be used to increase the release of growth factors, cytokines, chemokines, and EVs. The secretome has been shown to benefit from 3D culture modifications, cytokine induction, chemical addition, and hypoxic conditions (Azandeh et al., 2012; Almeria et al., 2019). These factors are critical for creating a secretome that can modulate inflammation, stimulate the migration of progenitor cells to the damaged brain region, and enhance recovery after ischemic stroke (Hofmann et al., 2012; Wei et al., 2012; Kim et al., 2015; Chen et al., 2017; Kong et al., 2017; Sidharta et al., 2018; Hu et al., 2019; Jiang et al., 2019; Zhang et al., 2019; Gregorius et al., 2021; Liu et al., 2021; Jumena et al., 2023; Tian et al., 2023). Hypoxic preconditioning can be used to boost the release of growth factors, chemokines, cytokines, exosomes, and EVs from MSCs (Ishiuchi et al., 2020; Jiang et al., 2022; Liu et al., 2022; Patil et al., 2022). Hypoxic preconditioning has been shown to boost the release of key molecules that promote neuroprotection, neurogenesis, and angiogenesis in preclinical studies (Hofmann et al., 2012; Wei et al., 2012; Kim et al., 2015; Chen et al., 2017; Kong et al., 2017; Sidharta et al., 2018; Hu et al., 2019; Jiang et al., 2019; Zhang et al., 2019; Gregorius et al., 2021; Liu et al., 2021; Jumena et al., 2023; Tian et al., 2023). Furthermore, hypoxic preconditioning activates hypoxia-inducible factor-1 alpha (HIF-1α), which regulates genes involved in inflammation, cell survival, and angiogenesis, thereby amplifying the regenerative properties of the conditioned medium of mesenchymal stem cells (MSC-CM) (Ye et al., 2021; Pulido-Escribano et al., 2022; Dong et al., 2023). It is considered to be a promising option for the treatment of ischemic stroke. There have been systematic review studies on the utilization of NSC, MSCs, and MSC-CM for ischemic stroke (Zhang et al., 2021; Yang et al., 2022; Behzadifard et al., 2023). However, no study on the effect of hypoxic mesenchymal stem cell-conditioned medium (hypMSC-CM) for ischemic stroke has been documented. Although MSCs and their secretomes have been studied extensively (Huang et al., 2015; Moisan et al., 2016; Ghazavi et al., 2017; Xia et al., 2020; Wang et al., 2022; Aderinto et al., 2024; Yaqubi and Karimian, 2024). The specific role of hypMSC-CM in ischemic stroke has not yet been systematically reviewed. This review focuses on preclinical studies exploring the use of hypMSC-CM for ischemic stroke treatment, with an emphasis on neurogenesis, neuroprotection, and angiogenesis. By comparing hypMSC-CM with NorMSC-CM, this review aimed to provide insights into the therapeutic potential of hypMSC-CM for ischemic stroke. This systematic review was created in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) standards (Moher et al., 2010). Although the study was not registered in the PROSPERO database or other equivalent databases, we confirmed that no similar reviews had been registered at the time of writing. Literature searchFor in vivo and in vitro stroke models through December 2024, publications for this review were selected from PubMed, Scopus, and Google Scholar. We conducted a PUBMED search using the following terms: (((((MSCs[MeSH Terms]) OR (MSC[Title/Abstract])) OR MSCs[Title/Abstract])) AND ((((Neuroprotection[MeSH Terms]) OR (neurons[Title/Abstract])) OR (neurogenesis[Title/Abstract])) OR (neuroprotective[Title/Abstract]))) AND ((((((Culture Media[MeSH Terms]) OR (CM [Title/Abstract])) OR (conditioned media[Title/Abstract])) OR (CM[Title/Abstract])) OR (secretome[Title/Abstract])) OR (preconditioning[Title/Abstract]))) AND (((((Hypoxia[MeSH Terms]) OR (Stroke[Title/Abstract])) OR (OGD[Title/Abstract])) OR (hypoxic[Title/Abstract])) OR (ischemic stroke[Title/Abstract])). Using Rayyan.ai, two researchers (UAN and HHI) performed the title selection step to identify relevant papers from the search results. Following the first screening, the same researcher applied the inclusion and exclusion criteria to each article. The final reference list was reviewed by a third researcher (RR), who was an expert in the field. Inclusion and exclusion criteriaArticles were selected using Rayyan.ai (Gafar et al., 2023). During the second screening phase, based on the following inclusion criteria: induced ischemic stroke, cerebral ischemia, or brain hypoxia; oxygen-glucose deprivation (OGD); therapy with hypMSC-CM; evaluation of changes in infarct size and neurological function/behavior; studies on neurogenesis and neuroprotection using sham or normoxia control groups; and original research publications. The exclusion criteria were as follows: human studies, absence of induced ischemic stroke, brain hemorrhagic models, no assessment of neurological or behavioral function, and studies presented as reviews, conference abstracts, letters, and non-English abstracts. The complete research approach is illustrated in the PRISMA flow diagram (Fig. 1).
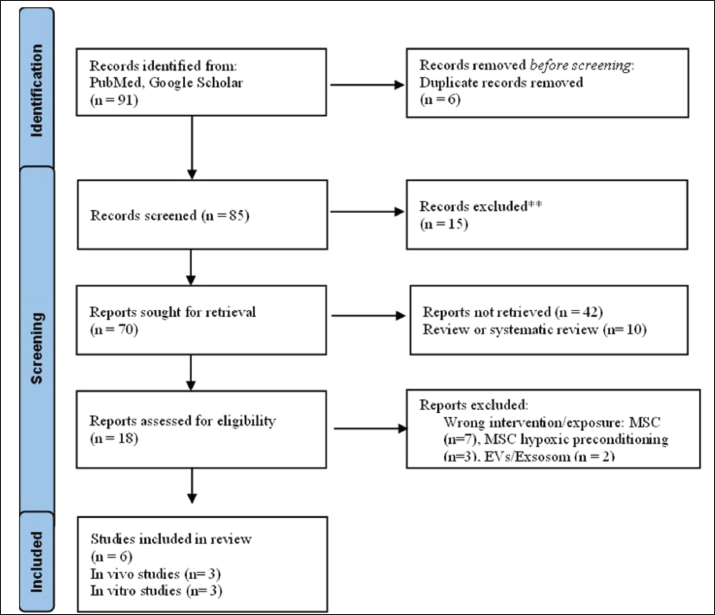
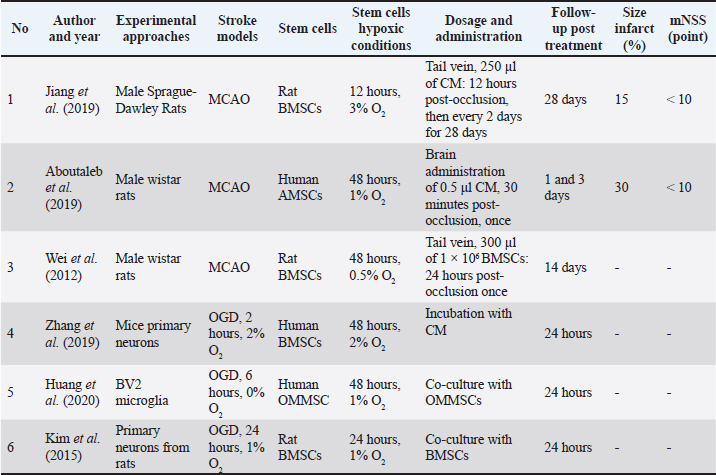
Fig. 1. Study selection flowchart. The PRISMA flow chart depicts the amount of data identified, included, and eliminated during various rounds of the systematic review. Data extractionWe collected the following information for each selected paper: author, journal, year of publication, country, ischemia model, animal type (including species, strain, sex, age, and weight), method of hypoxic preconditioning of the MSC, dosage, and outcomes. Findings were categorized based on lesion size, neurological function/behaviour, and indicators of neurogenesis, neuroprotection, and angiogenesis in the spinal cord. Quality assessmentWe assessed the methodological quality of the included studies using a checklist that evaluated criteria, such as growth factors, MSC hypoxia collection techniques, outcomes, controls, interventions, hypoxic conditions, duration, stem cell type, disease models, population, and study design. Each study was assigned a quality score on a 100-point scale. Two authors independently evaluated the methodological quality of the included studies. If there was a discrepancy in the quality scores between the reviewers, particularly when one rated a study below 50, discussions were held with a third reviewer (WA, JHS, or RN) to reach a consensus. Discrepancies were rare, occurring in less than 1% of the cases, as the two reviewers generally had similar assessments of the data. A total of 91 non-duplicate publications were identified. After screening the titles and abstracts, 15 were excluded for irrelevance or not meeting the criteria. Of the 72 full-text articles reviewed, 52 were excluded due to insufficient data on hypMSC-CM or study design limitations. Finally, six studies met the inclusion criteria and were selected for this systematic review (Fig. 1). The therapeutic effect of hypMSC-CM and its secretome in the treatment of ischemic stroke was the primary focus of these studies. Most of the included studies used the middle cerebral artery occlusion (MCAO) model in rodents, which is widely accepted as a representative preclinical model for ischemic stroke (Table 1). In vivo studies have consistently demonstrated that hypMSC-CM administration reduces infarct volume, improves neurological recovery, and promotes angiogenesis, neurogenesis, and neuroprotection through various signaling pathways (Table 2). Additionally, in vitro investigations using neuronal cultures and microglial cells under OGD conditions confirmed the anti-inflammatory and neuroprotective effects of hypMSC-CM. Table 1. Characterization of articles included in the study.
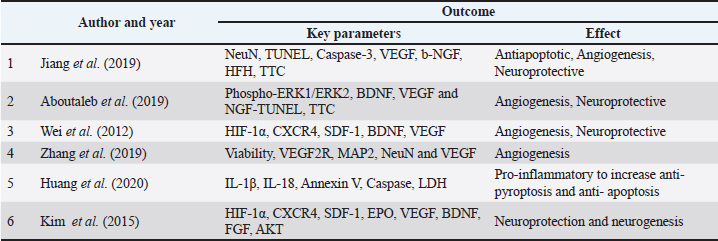
Table 2. Therapeutic effects or outcome using hypoxic preconditioning.
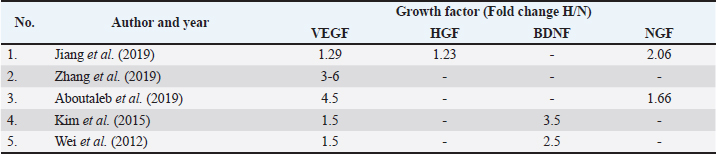
Reduction of infarct volume and improvement in motor functionAcross the reviewed studies, hypMSC-CM consistently reduced infarct size in rodent stroke models. Male Sprague-Dawley rats subjected to MCAO were treated with CM derived from bone marrow mesenchymal stem cells (BMMSC) 12 hours after reperfusion. CM was administered intravenously every 2 days for 28 days. The results showed a significant reduction in infarct volume compared to that in the normoxic MSC control group, as determined by triphenyl tetrazolium chloride (TTC) staining (p < 0.01). Furthermore, motor function, as measured by the modified neurological severity score (mNSS) and balance beam test, improved significantly in the hypMSC-CM treated group (p < 0.05).(Jiang et al., 2019) This suggests that hypMSC-CM enhances its therapeutic efficacy by increasing the secretion of neuroprotective factors that reduce brain damage and promote functional recovery. Hypoxic preconditioning increases the expression of various growth factors, including HIF-1α, BDNF, and erythropoietin (EPO), in BMMSC. When administered intravenously to MCAO rats, BMMSC significantly improved motor function, as assessed by rotarod performance, and led to higher levels of neuron-specific markers and glial fibrillary acidic protein (GFAP), indicating increased neuronal regeneration and decreased glial scarring (p < 0.05) (Wei et al., 2012). These results suggest that hypMSC provides comprehensive benefits in terms of both reducing damage and promoting recovery. Promotion of angiogenesis and neurogenesisThe reviewed studies have consistently reported that hypMSC-CM promotes angiogenesis and neurogenesis, which are critical processes in stroke recovery. These effects were primarily attributed to increased VEGF and other neurotrophic factor secretion under hypoxic conditions. VEGF levels in hypoxia-preconditioned BMSC-CM were significantly elevated compared to those in normoxic conditions (p < 0.001). The study demonstrated that these elevated VEGF levels were beneficial for enhancing neuron survival and supporting neuron regeneration in in vitro models using primary neurons from mouse brains subjected to OGD (Zhang et al., 2019a,b). Similarly, the CM of hypoxic preconditioned amniotic membrane mesenchymal stem cells (AMSC) in Wistar rats MCAO model significantly improved angiogenesis in the infarcted brain tissue, as evidenced by the upregulation of VEGF and neurogenesis-related factors such as BDNF (p < 0.05) (Aboutaleb et al., 2019). Additionally, therapy administration influences the success of therapy itself. The intranasal administration of hypoxia-preconditioned BMSC to the MCAO model in B6 mice resulted in increased levels of VEGF and its receptor (Tian et al., 2023). These findings support the idea that hypMSC-CM enhances neuroregeneration by stimulating both the formation of new blood vessels and the proliferation of neural cells in the ischemic brain. The increased expression of neurotrophic factors like BDNF and VEGF suggests that hypMSC-CM could have long-lasting benefits for stroke recovery by promoting tissue repair and functional improvement (Majumdar et al., 2013; Sidharta et al., 2018; Patil et al., 2022). The combination of stem cell therapy with tPA also provides a better level of effectiveness in inhibiting the occurrence of hemorrhage, inflammation, but inducing the angiogenesis process and the formation of neurite outgrowths in neurogenesis (Xin et al., 2013; Qian et al., 2016; Nakazaki et al., 2017; Fang et al., 2022). To enhance the therapeutic effects of MSC and its secretome, a combination is carried out with active herbal medicinal compounds such as sodium ferulate, salvianolic acid B, and extracts that can help increase post-stroke antioxidants (Kim et al., 2018; Guo et al., 2012; Zhang et al., 2016, 2017; Yan et al., 2023). The neurogenesis process is also expected to improve when MSCs become adjuvant therapy in the use of rehabilitation therapies such as acupuncture, physiotherapy, and administration of drugs such as stiripentol, mannitol, atorvastatin, and aspirin (Choi et al., 2018; Lee et al., 2020; Satani et al., 2021; Dhir et al., 2023; Aderinto et al., 2024). Anti-inflammatory and anti-apoptotic effectsInflammation is a hallmark of stroke, contributing to both immediate brain damage and delayed secondary injury. One study included in this review highlighted the anti-inflammatory effects of hypMSC-CM in both in vivo and in vitro stroke models. One key mechanism underlying these anti-inflammatory effects is the suppression of pro-inflammatory cytokines, such as IL-1 and IL-18. In a study using the BV2 microglia cell line as an in vitro model of ischemia, hypoxia-preconditioned olfactory mucosa mesenchymal stem cells (OMMSC) significantly reduced the production of IL-1β and IL-18 (p < 0.001 vs. normoxia) (Huang et al., 2020; Li et al., n.d.).This reduction in pro-inflammatory cytokine levels was associated with increased cell viability and reduced apoptosis, suggesting that hypMSC exert anti-inflammatory effects by modulating the microglial response to ischemic injury. The anti-apoptotic effects of hypMSC-CM were also observed, where hypMSC-CM significantly increased neuronal cell survival in the in vitro ischemia models through the upregulation of HIF-1α, AKT, and other pro-survival signaling pathways (p < 0.05 vs. normoxia) (Zhang et al., 2019a,b). These findings suggest that hypMSC-CM may protect against ischemic brain injury by both reducing the inflammatory response and promoting neuronal survival. Neuroprotection via increased growth factor secretionHypMSCs demonstrate significant neuroprotective effects by activating key signaling pathways involved in cell survival, neurogenesis, and angiogenesis, making them a promising therapy for ischemic stroke. HypMSC-CM activated PI3K/Akt signaling in ischemic brain tissue, resulting in enhanced neuroprotection and neurogenesis in a rodent model of MCAO. This activation led to improved functional outcomes and a reduction in brain damage (Jiang et al., 2019). Similarly, hypMSC-CM also upregulated the phosphorylation of ERK1/2 and increased BDNF expression in the infarcted region, further supporting its neuroprotective effects through these signaling mechanisms (Aboutaleb et al., 2019). HypMSC role in promoting the secretion of neurotrophic factors further amplifies its therapeutic potential. It significantly increased the production of BDNF, VEGF, and hepatocyte growth factor (HGF), which are critical for neuronal survival and protection against ischemic damage. HypMSC upregulated the expression of these neurotrophic factors, which in turn led to increased neuron regeneration and reduced infarct volume in rodent stroke models (Wei et al., 2012; Kim et al., 2015). These findings collectively point to hypMSC and hypMSC-CM ability to initiate a multifaceted neuroprotective response, targeting both neuron survival and tissue repair. The benefits of hypMSC-CM can also be attributed to its capacity to significantly enhance the secretion of various growth factors that play pivotal roles in post-stroke recovery. Studies have consistently reported that hypoxic preconditioning with MSC-CM leads to elevated levels of VEGF, BDNF, HGF, and β-nerve growth factor (β-NGF). Zhang et al. (2016, 2017, 2019a, b, 2021) noted a fourfold increase in VEGF levels in hypMSC-CM compared to normoxic MSC-CM (p < 0.001), while Jiang et al. (2019, 2022, 2024) found a twofold increase in β-NGF levels under hypoxic conditions (p < 0.01) (Zhang et al., 2019a,b; Jiang et al., 2019). The increased secretion of these growth factors directly contributed to improvements in angiogenesis, neuroprotection, and motor function observed in the stroke models. This review highlights the promising therapeutic potential of hypMSC and hypMSC-CM in treating ischemic stroke, with a particular focus on their ability to reduce infarct volume and enhance motor function. Most preclinical studies analyzed utilized rodent models, specifically the transient MCAO model, to assess the efficacy of hypMSC and hypMSC-CM. Both in vivo and in vitro studies have shown that hypMSC and hypMSC-CM not only reduce brain damage but also activate critical cellular signaling pathways involved in neuroprotection, angiogenesis, and neurogenesis. An in vivo study was performed and reported by Jiang et al. (2019, 2022, 2024). They induced stroke in 60 male Sprague-Dawley rats (240–270 g, 7–8 weeks old) using a 0.34-mm nylon monofilament coated with polylysine for the MCAO model. Secretome from hypoxic preconditioned conditioned medium rat BMMSC/rat BMMSCs-CM was administered through tail intravenous injection 12 hours after reperfusion and subsequently once every 2 days for 28 days during the stroke. The rat BMMSC-CM used for treatment was preconditioned for 12 hours under 3% O2 hypoxia and normoxia as a control. An examination of proteins in the form of cytokines and growth factors revealed a threefold increase, particularly in b-NGF. 2,3,5- TTC staining revealed (a) a decrease in infarct volume and an increase in neurological scores utilizing the (mNSS test and (b) balance beam test before and after therapy in hypMSC-CM compared to norMSC-CM (p < 0.01). Cellular studies on the effects of ipsilateral therapy, including enhanced angiogenesis, such as VEGF production, and suppression of tunneling as a signal of apoptosis, revealed a substantial, favorable therapeutic impact (p < 0.05). An increase in PI3K/Akt, which enhances neuroprotection and neurogenesis, signals the effect of secretome treatment (Jiang et al., 2019). AMSC-CM were produced via hypoxia preconditioning with 1% O2 for 48 hours, according to Aboutaleb et al. (2019). In Wistar rats, therapy was administered at a dosage of 0.5 µl 30 minutes after MCAO treatment via stereotactic intracerebral infusion. To measure the extent of the infarct region, the mNSS test, rotarod test, and TTC were used. The use of hypoxic hAMMSC-CM showed significant improvement compared to the MCAO and Sham groups (p < 0.05 and p < 0.01, respectively). Nissl staining, immunohistochemical evaluation, and TUNEL tests were performed at the cellular level to identify DNA fragmentation, suggesting apoptosis in the core region and penumbra areas. This study found that the intravenous administration of AMSC-CM protected against localized cerebral ischemia-reperfusion damage following MCAO. Human AMSC-CM hypoxic preconditioning promoted the BDNF/ERK signaling pathway, thereby suppressing apoptosis and exerting a neuroprotective effect (p < 0.05). In the infarct region, this process is regulated by ERK1/2 and the mitogen-activated protein kinase family, which controls neuronal survival and cell death. Inhibition of apoptosis, increased BDNF protein expression, and significantly enhanced VEGF and NGF (p < 0.001 and p < 0.05 vs. MCAO) expression in MCAO animals treated with AMSC-CM hypoxic preconditioning, as evidenced by subsequent upregulation of Bcl-2. This event was found to have a favorable influence on the neurogenesis and neuroprotection processes following MCAO (Aboutaleb et al., 2019). Wei et al. (2012) developed the idea of allogenic treatment for hypBMMSC-CM in Wistar MCAO rats by administering 300 μl intravenously. Motor function tests revealed that hypoxic preconditioning with BMMSC-CM significantly improved motor function. HIF-1α, BDNF, GDNF, SDF-1, and EPO were all expressed at higher levels in hypBMMSC-CM (p < 0.05 in norBMMSC-CM). Similarly, the glial factor GFAP/neuronN ratio increased in hypBMMSC-CM (p0.05 vs. norBMMSC-CM) (p < 0.05 vs. norBMMSC-CM). Erythropoietin receptor (EPOR) and VEGF levels are greater in regions where these proteins are involved in neuron regeneration, anti-inflammation, and angiogenesis (Wei et al., 2012). In an in vitro study by Zhang et al. (2016, 2017, 2019a, b, 2021) hypBMMSCs-CM in serum-free media for 48 hours under 2% O2 hypoxia. Primary mouse neurons were treated with OGD as an ischemia model for 2 hours in 2% O2, followed by hypoxic preconditioning with hBMMSC-CM for 24 hours, and the effect on neuronal cell survival was investigated. VEGF levels in hypoxia-preconditioned hBMMSC-CM increased fourfold compared with those in normoxic conditions. This level of VEGF is believed to protect neurons and facilitate neuronal regeneration. According to this study, there was a substantial increase in viability in primary cultures of mouse neurons, as well as an increase in VEGF production in hyp-hBMMSC-CM (p < 0.001 vs. OGD and p < 0.001 vs. normoxia) (Zhang et al., 2019a,b). In another in vitro investigation, the microglial BV2 cell line was used to construct a stroke model using OGD for 4 hours at 5% O2. Hypoxic preconditioning of OM-MSC with 1% O2 for 48 hours using co-culture was carried out for 12 hours, and then various analyses were performed. Microglia are crucial in neuroinflammation because they produce pro- and anti-inflammatory mediators. Hypoxic preconditioned OMMSC-CM promoted HIF-1α expression (p < 0.05 vs. normoxia), protecting microglial cells from pyroptotic cell death or apoptosis. This treatment lowers the levels of pro-inflammatory mediators (e.g., IL-1β and IL-18). This suggests that hypoxic preconditioning may be an effective method for increasing the anti-pyroptosis and anti-apoptotic effects of hypoxia-preconditioned hypMSC-CM (p < 0.001 vs. normoxia) (Huang et al., 2020). Another study used primary cultures of cortical neurons for the OGD model with 1% O2 for 24 hours, cultured with hyphBMMSC-CM for 24 hours, and analyzed the therapeutic effect. Post-culture levels of H/R-conditioned BM-MSCs improved the survival of ischemic rat cortical neurons. HyphBM-MSCs-CM increased pro-survival signals such as HIF-1alpha AKT, EPOR, trophic factor release, and cell migration marker SDF-1/CXCR7 appears to be enhancing its ability to rescue ischemic cortical neurons (p < 0.05 vs. normoxia) (Kim et al., 2015). DiscussionThis review aimed to assess the therapeutic efficacy of hypMSC-CM in the treatment of ischemic stroke. This study revealed that most preclinical research on hypMSC-CM as a treatment for stroke recovery is based on a rodent model of tMCAO. Furthermore, the present review established that hypMSC-CM has the capacity to diminish the TTC test results and enhance motor function in in vivo assessments of infarct volume and motor function. In addition to in vivo and in vitro studies, other cellular signaling pathways, such as neuroprotection induction, angiogenesis, and neurogenesis, have been investigated. Previous comprehensive evaluations have found that hypoxic preconditioning of MSCs enhances their therapeutic impact in stroke and increases the levels of several proteins that facilitate neuroregeneration (Behzadifard et al., 2023). Further research showed evidence from preclinical studies, both in vivo and in vitro, that treatment of ischemic stroke with hypMSC-CM had a better therapeutic effect on ischemic stroke than norMSC-CM (Table 3). The findings support the idea that hypoxia stimulation of MSCs induces PI3K/Akt phosphorylation, which in turn activates HIF-1α, resulting in increased production of growth factors, cytokines, chemokines, and EVs (exosomes and non-coding RNA/ncRNA) (Ye et al., 2021; Patil et al., 2022; Pulido-Escribano et al., 2022). Table 3. Bioactive enhancement in hypoxic preconditioning.
According to the findings of various research, this hypoxic preconditioning secretome may be a solution for ischemic stroke treatment, where paracrine actions aid in several neurogenesis processes (Wei et al., 2012; Leschik et al., 2022). Various growth factors, cytokines, chemokines, and EVs have a part in the penumbra area's angiogenic, anti-apoptotic, and neuroregenerative activities (Uzdensky, 2019; Li et al., 2020). Following an ischemic stroke, an infarction arises in the core region, which is not supplied with blood, resulting in total neuronal cell death, and in the penumbra, which is provided with blood but with extremely low blood flow (Bandera et al., 2006). It is believed that administering various antioxidants and vitamins, followed by enhancing blood flow, can help preserve the penumbra region (Mazumder and Borah, 2015; Agarwal et al., 2022). Similarly, hypMSC-CM is likely to aid in penumbra regeneration and rescue neuronal cells from death caused by ischemic damage. By cutting off the blood supply to the intracerebral artery region, the penumbral area can easily be acquired as an in vivo model. Meanwhile, primary cultures, cell lines, including both normal neuronal cells and neuroblastoma cells from humans or animals, may be used to simulate the penumbra region as an in vitro model (Li et al., 2020; Trotman-Lucas and Gibson, 2021). This systematic review of preclinical studies revealed that the paracrine effect of hypMSC-CM improved motor behavior and limb strength, lowered infarct size, and reduced inflammation, thereby exerting a neuroprotective effect. In general, paracrine effects play an important role in stroke treatment, particularly in; first, the effect of neurogenesis, neurotropic factors secreted by MSCs such as NGF, GDNF and BDNF play a role in axon growth, formation of new neurons and strengthening of existing neural connections (Sidharta et al., 2018; Rinendyaputri et al., 2023). Progenitor neurons will migrate to the infarct site, where they will undergo modifications in shape and function (Rahman et al., 2021). Apart from neurotrophic factors, the role of cytokines as an anti-inflammatory after ischemic stroke, which causes reperfusion injury, is also very important. MSCs will stimulate M2 macrophages to generate anti-inflammatory cytokines, allowing them to suppress pro-inflammatory cytokines such as IL-18 and IL-1β (Huang et al., 2014). HypMSC-CM contains anti-inflammatory compounds, such as IL-10 and TGF-β, which enhance the effects of MSCs following hypoxic preconditioning. Second, when VEGF and BDNF bind to their receptors, they phosphorylate PI3K, which subsequently activates ERK/Akt1 and suppresses the apoptotic cascade (Zhou et al., 2017; Aboutaleb et al., 2019; Zhang et al., 2019a,b). VEGF also aids angiogenesis in the infarction region. Repairing the blood vessels delivers oxygen and nutrients to the penumbra region, allowing neurogenesis to proceed and reducing the infarcted area. Behavioral and cognitive gains may indicate a decrease in the size of the infarct area. The positive paracrine effect of hypMSC-CM on neurogenesis and neuroprotection is expected to be an alternative therapy for ischemic stroke. HypMSC-CM, in conjunction with conventional treatments such as vitamins and antioxidants, may provide an alternative adjuvant therapy for ischemic stroke. These studies have reported significant effects on neurogenesis; however, there is no proof of concept for allogeneic therapy. Research is still being conducted on cells and mice; therefore, many studies are needed before clinical trials can be initiated. Research on the effects of hypMSC-CM in stroke has not been widely conducted. To determine the impact of adjuvant therapy, it can be combined with standard treatments, such as tPA. In various studies, the use of herbal medicines has been reported to increase therapeutic effects. The year of publication was not limited in this study; however, only English-language articles were used for analysis. Hence, papers in languages other than English were omitted from the search. Our search encompassed three databases; however, we may have overlooked the relevant material, and the sample sizes in several of the included studies were small. The number of publications obtained was minimal because our inclusion criteria focused on the hypoxia-conditioned MSC secretome for ischemic stroke. It is anticipated that this research will lead to preclinical and clinical trials, overcoming these difficulties and generating substantial data to inform the treatment of ischemic stroke using the MSC secretome, particularly in conjunction with hypoxic preconditioning. The main strength of our study lies in the combination of in vitro and in vivo studies; however, this systematic review has several limitations. 1) Owing to the limited original data, we used the overall data from the table in a percentage form. 2) Variations in hypMSC-CM and norMSC-CM doses, routes, and transplantation times have not been reported to yield optimal results. We did not consider the influence of transplantation time and route on the results when analyzing the optimal dose, which may have changed the conclusion. 3) The rationale for this practice is worth exploring because not all articles have reported the bioactive composition of hypMSC-CM and norhypMSC-CM. This demonstrates the sound therapeutic effects of the information obtained from the included studies. Limitations and Translational Challenges: Although this study provides significant insights into the therapeutic potential of hypMSC-CM for ischemic stroke, several limitations must be acknowledged. A meta-analysis was not conducted because of the significant heterogeneity of the available data, which encompassed variations in study design (in vivo versus in vitro), MSC sources, routes of administration (intravenous versus intracerebral), duration and intensity of hypoxic preconditioning, and outcome measurements. To date, there has not been a thorough systematic evaluation of the use of hypoxia-preconditioned MSCs in stroke models. This makes it difficult to reproduce and standardize findings across different research studies. Despite promising preclinical outcomes, several translational challenges must be addressed before hypMSC-CM can be used in clinical settings. The optimal dosage, delivery route, and therapeutic timing remain undefined, impeding clinical translation. The bioactive composition of hypMSC-CM varies with culture conditions, complicating regulatory approval and large-scale production. Additionally, preclinical animal models do not fully replicate human stroke pathophysiology, and immune responses to allogeneic products require further investigation. Resolving these challenges is critical for translating preclinical research outcomes into safe and effective clinical practice. ConclusionIn conclusion, the reviewed studies provide strong evidence that hypMSC offers neuroprotection and enhances neurogenesis following ischemic stroke through the secretion of growth factors, cytokines, and EVs. AcknowledgmentsWe thank the National Research and Innovation Agency and the Graduate School of Biomedical Sciences, Doctoral Program, Faculty of Medicine, Padjadjaran University, Indonesia, for supporting the Degree by Research program and providing APC payments. Conflict of interestThe authors declare no conflicts of interest in this study. Authors' contributionRR, RN, and AF conceived and designed the study. FH and WA contributed to data analysis and interpretation. UAH, PRI, and R drafted the manuscript. NSDP critically revised the manuscript. AF and FH supervised the study. NHS and HHI contributed to the final approval of the manuscript. Data availabilityThe manuscript contains all the necessary information to corroborate the study results. ReferencesAbd El Motteleb, D.M., Hussein, S., Hasan, M.M. and Mosaad, H. 2018. Comparison between the effect of human Wharton’s jelly-derived mesenchymal stem cells and levetiracetam on brain infarcts in rats. J. Cell. Biochem. 119(12), 9790–9800. Aboutaleb, N., Faezi, M., Nasseri Maleki, S., Nazarinia, D., Razavi Tousi, S.M.T. and Hashemirad, N. 2019. Conditioned medium obtained from mesenchymal stem cells attenuates focal cerebral ischemia reperfusion injury through activation of ERK1/ERK2-BDNF signaling pathway. J. Chem. Neuroanat. 97, 87–98. Aderinto, N., Olatunji, G., Kokori, E., Babalola, A.E., Yusuf, I.A., Apampa, O.O., Ukoaka, B.M., Aboje, J.E., Adefusi, T., Moradeyo, A., Obasanjo, O.M., Ogieuhi, I.J., Omoworare, O., and Olatunji, D. 2024. Stem cell therapies in stroke rehabilitation: a narrative review of current strategies and future prospects. Egypt. J. Neurol. Psychiatry Neurosurg. 60(1), 79. Agarwal, A., Vishnu, V.Y., Sharma, J., Bhatia, R., Garg, A., Dwivedi, S., Upadhyay, A., Goyal, V., Singh, M.B., Gupta, A., Rajan, R. and Srivastava, M.V.P. 2022. Citicoline in acute ischemic stroke: a randomized controlled trial. PLos One 17, e0267870. Almeria, C., Weiss, R., Roy, M., Tripisciano, C. and Kasper, C. 2019. Hypoxia conditioned mesenchymal stem cell-derived extracellular vesicles induce increased vascular tube formation in vitro. Front. Bioeng. Biotechnol. 7(292), 1–13. Azandeh, S., Orazizadeh, M., Hashemitabar, M., Khodadadi, A., Shayesteh, A.A., Nejad, D.B., Gharravi, A.M. and Allahbakhshi, E. 2012. Mixed enzymatic-explant protocol for isolation of mesenchymal stem cells from Wharton’s jelly and encapsulation in 3D culture system. J. Biomed. Sci. Eng. 05(10), 580–586. Bandera, E., Botteri, M., Minelli, C., Sutton, A., Abrams, K.R. and Latronico, N. 2006. Cerebral blood flow threshold of ischemic penumbra and infarct core in acute ischemic stroke: a systematic review. Stroke 1334, 1334–1339. Behzadifard, M., Aboutaleb, N., Dolatshahi, M., Khorramizadeh, M., Mirshekari Jahangiri, H., Kord, Z. and Nazarinia, D. 2023. Neuroprotective effects of conditioned medium of Mesenchymal stem cells (MSC-CM) as a therapy for ischemic stroke recovery: a systematic review. Neurochem. Res. 48(5), 1280–1292. Bermejo, P.E., Dorado, R. and Zea-Sevilla, M.A. 2023. Role of Citicoline in patients with mild cognitive impairment. Neurosci. Insights 18. Bluhmki, E., Danays, T., Biegert, G., Hacke, W. and Lees, K.R. 2020. Alteplase for acute ischemic stroke in patients aged u003e80 years: pooled analyses of individual patient data. Stroke 51(8), 2322–2331. Braga, C.L., Santos, R.T., da Silva, C.M., de Novaes Rocha, N., Felix, N.S., Medeiros, M., Melo, M.M., Silva, J.D., Teixeira, D.E., Neves, C.C., Rocco, P.R.M., Cruz, F.F. and Silva, P.L. 2023. Therapeutic effects of hypoxia-preconditioned bone marrow-derived mesenchymal stromal cells and their extracellular vesicles in experimental pulmonary arterial hypertension. Life Sci. 329, 121988. Castelli, V., Antonucci, I., D'Angelo, M., Tessitore, A., Zelli, V., Benedetti, E., Ferri, C., Desideri, G., Borlongan, C., Stuppia, L. and Cimini, A. 2020. Neuroprotective effects of human amniotic fluid stem cells-derived secretome in an ischemia / reperfusion model. Stem Cells. Transl. Med. 10, 251–266. Chelluri, L.K., Upadhyay, U., Nallagonda, R., Prasad, S., Samiuddin, M., Mohanty, R., Mallarpu, C., Ponnana, M., Rawul, S. and Chelluri, E.P. 2017. Safety study of autologous adult bone marrow derived mesenchymal stromal cells in idiopathic pulmonary fibrosis - pilot data. New. Horiz. Transl. Med. 4(1–4), 15–22. Chen, J., Yang, Y., Shen, L., Ding, W., Chen, X., Wu, E., Cai, K. and Wang, G. 2017. Hypoxic preconditioning augments the therapeutic efficacy of bone marrow stromal cells in a rat ischemic stroke model. Cell. Mol. Neurobiol. 37(6), 1115–1129. Choi, C., Kim, H.M., Shon, J., Park, J., Kim, H.T., Kang, S.H., Oh, S.H., Kim, N.K. and Kim, O.J. 2018. The combination of mannitol and temozolomide increases the effectiveness of stem cell treatment in a chronic stroke model. Cytotherapy 20(6), 820–829. Chrostek, M.R., Fellows, E.G., Crane, A.T., Grande, A.W. and Low, W.C. 2019. Efficacy of stem cell-based therapies for stroke. Brain Res. 1722, 146362. Cunningham, C.J., Redondo-Castro, E. and Allan, S.M. 2018. The therapeutic potential of the mesenchymal stem cell secretome in ischaemic stroke. J. Cerebral Blood Flow Metab. 38(8), 1276–1292. Dhir, N., Jain, A., Sharma, A.R., Sharma, S., Mahendru, D., Patial, A., Malik, D., Prakash, A., Attri, S.V., Bhattacharyya, S., Das Radotra, B. and Medhi, B. 2023. Rat BM-MSCs secretome alone and in combination with stiripentol and ISRIB, ameliorated microglial activation and apoptosis in experimental stroke. Behav. Brain. Res. 449, 114471. Dong, J., Wu, B. and Tian, W. 2023. Exosomes derived from hypoxia-preconditioned mesenchymal stem cells (hypoMSCs-Exo): advantages in disease treatment. Cell Tissue Res. 392(3), 621–629. Fan, J., Li, X., Yu, X., Liu, Z., Jiang, Y., Fang, Y., Zong, M., Suo, C., Man, Q. and Xiong, L. 2023. Global burden, risk factor analysis, and prediction study of ischemic stroke, 1990–2030. Neurology 101(2), 85–94. Fang, C.N., Song, T., Tao, X., Liu, S.F. and Feng, Z. 2022. Bone marrow stromal cells attenuate oxygen and glucose deprivation followed by re-oxygenation-induced brain microvascular endothelial cell injury by mediating the plasminogen activator/plasminogen activator receptor/stromal cell-derived factor-1α/C-X-C chemokine receptor type 4 pathway. Acta. Neurobiol. Exp. (Wars). 82(3), 398–407. Forkan, C.P., Shrestha, A., Yu, A., Chuang, C., Pociot, F. and Yarani, R. 2025. Could hypoxic conditioning augment the potential of mesenchymal stromal cell-derived extracellular vesicles as a treatment for type 1 diabetes? Stem Cell Res. Ther. 16(1), 37. Gafar, F., Wasmann, R.E., McIlleron, H.M., Aarnoutse, R.E., Schaaf, H.S., Marais, B.J., Agarwal, D., Antwi, S., Bang, N.D., Bekker, A., Bell, D.J., Chabala, C., Choo, L., Davies, G.R., Day, J.N., Dayal, R., Denti, P., Donald, P.R., Engidawork, E., Garcia-Prats, A.J., Gibb, D., Graham, S.M., Hesseling, A.C., Heysell, S.K., Idris, M.I., Kabra, S.K., Kinikar, A., Kumar, A.K.H., Kwara, A., Lodha, R., Magis-Escurra, C., Martinez, N., Mathew, B.S., Mave, V., Mduma, E., Mlotha-Mitole, R., Mpagama, S.G., Mukherjee, A., Nataprawira, H.M., Peloquin, C.A., Pouplin, T., Ramachandran, G., Ranjalkar, J., Roy, V., Ruslami, R., Shah, I., Singh, Y., Sturkenboom, M.G.G., Svensson, E.M., Swaminathan, S., Thatte, U., Thee, S., Thomas, T.A., Tikiso, T., Touw, D.J., Turkova, A., Velpandian, T., Verhagen, L.M., Winckler, J.L., Yang, H., Yunivita, V., Taxis, K., Stevens, J. and Alffenaar, J.W.C. 2023. Global estimates and determinants of antituberculosis drug pharmacokinetics in children and adolescents: a systematic review and individual patient data meta-analysis. Eur. Respir. J. 61(3), 2201596. Ghazavi, H., Hoseini, S.J., Ebrahimzadeh-Bideskan, A., Mashkani, B., Mehri, S., Ghorbani, A., Sadri, K., Mahdipour, E., Ghasemi, F., Forouzanfar, F., Hoseini, A., Pasdar, A.R., Sadeghnia, H.R. and Ghayour-Mobarhan, M. 2017. Fibroblast growth factor type 1 (FGF1)-overexpressed adipose-derived mesenchaymal stem cells (AD-MSCFGF1) induce neuroprotection and functional recovery in a rat stroke model. Stem Cell Rev. 13(5), 670–685. Goyal, M., Menon, B.K., Van Zwam, W.H., Dippel, D.W.J., Mitchell, P.J., Demchuk, A.M., Dávalos, A., Majoie, C.B.L.M., Van Der Lugt, A., De Miquel, M.A., Donnan, G.A., Roos, Y.B.W.E.M., Bonafe, A., Jahan, R., Diener, H.C., Van Den Berg, L.A., Levy, E.I., Berkhemer, O.A., Pereira, V.M., Rempel, J., Millán, M., Davis, S.M., Roy, D., Thornton, J., Román, L.S., Ribó, M., Beumer, D., Stouch, B., Brown, S., Campbell, B.C.V., Van Oostenbrugge, R.J., Saver, J.L., Hill, M.D. and Jovin, T.G. 2016. Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials. Lancet 387(10029), 1723–1731. Gregorius, J., Wang, C., Stambouli, O., Hussner, T., Qi, Y., Tertel, T., Börger, V., Mohamud Yusuf, A., Hagemann, N., Yin, D., Dittrich, R., Mouloud, Y., Mairinger, F.D., Magraoui, F.E., Popa-Wagner, A., Kleinschnitz, C., Doeppner, T.R., Gunzer, M., Meyer, H.E., Giebel, B. and Hermann, D.M. 2021. Small extracellular vesicles obtained from hypoxic mesenchymal stromal cells have unique characteristics that promote cerebral angiogenesis, brain remodeling and neurological recovery after focal cerebral ischemia in mice. Basic. Res. Cardiol. 116(1), 40. Guan, Y.M., Zhu, Y., Liu, X.C., Huang, H.L., Wang, Z.W., Liu, B., Zhu, Y.Z. and Wang, Q.S. 2014. Effect of human umbilical cord blood mesenchymal stem cell transplantation on neuronal metabolites in ischemic rabbits. BMC. Neurosci. 15(1), 41. Guo, J.W., Chen, C., Huang, Y. and Li, B. 2012. Combinatorial effects of Naomai Yihao Capsules and vascular endothelial growth factor gene-transfected bone marrow mesenchymal stem cells on angiogenesis in cerebral ischemic tissues in rats. J. Traditional. Chin. Med. 32(1), 87–92. Gutiérrez-Fernández, M., Rodríguez-Frutos, B., Ramos-Cejudo, J., Otero-Ortega, L., Fuentes, B., Vallejo-Cremades, M., Sanz-Cuesta, B. and Díez-Tejedor, E. 2015. Comparison between xenogeneic and allogeneic adipose mesenchymal stem cells in the treatment of acute cerebral infarct: proof of concept in rats. J. Transl. Med. 13(1), 46. Gwam, C., Mohammed, N. and Ma, X. 2021. Stem cell secretome, regeneration, and clinical translation: a narrative review. Ann. Transl. Med. 9(1), 70. Han, M., Zhang, Z., Liu, Z., Liu, Y., Zhao, H., Wang, B., Zhang, C., Shang, H., Li, Y., Wang, S., and Xin, T. 2023. Three-dimensional-cultured MSC-derived exosome with hydrogel for cerebral ischemia repair. Biomater. Adv. 149, 213396. Hasan, T.F., Hasan, H. and Kelley, R.E. 2021. Overview of acute ischemic stroke evaluation and management. Biomedicines 9(10), 1486. Hofmann, N.A., Ortner, A., Jacamo, R., Reinisch, A., Schallmoser, K., Rohban, R., Frühwirth, M., Liechtenstein, N., Beham-Schmid, C., Linkesch, W., Andreeff, M. and Strunk, D. 2012. A novel role for mesenchymal stem/progenitor cells as hypoxia sensors during initiation of neo-vasculogenesis in vivo. Blood 120(21), 613. Hu, Y., Chen, W., Wu, L., Jiang, L., Qin, H. and Tang, N. 2019. Hypoxic preconditioning improves the survival and neural effects of transplanted mesenchymal stem cells via CXCL12/CXCR4 signalling in a rat model of cerebral infarction. Cell Biochem. Funct. 37(7), 504–515. Huang, P., Gebhart, N., Richelson, E., Brott, T.G., Meschia, J.F. and Zubair, A.C. 2014. Mechanism of mesenchymal stem cell-induced neuron recovery and anti-inflammation. Cytotherapy 16(10), 1336–1344. Huang, W., Lv, B., Zeng, H., Shi, D., Liu, Y., Chen, F., Li, F., Liu, X., Zhu, R., Yu, L. and Jiang, X. 2015. Paracrine factors secreted by MSCs promote astrocyte survival associated with GFAP downregulation after ischemic stroke via p38 MAPK and JNK. J. Cell. Physiol. 230(10), 2461–2475. Huang, Y., Tan, F., Zhuo, Y., Liu, J., He, J., Duan, D., Lu, M. and Hu, Z. 2020. Hypoxia-preconditioned olfactory mucosa mesenchymal stem cells abolish cerebral ischemia/reperfusion-induced pyroptosis and apoptotic death of microglial cells by activating HIF-1α. AGING 12(11), 10932–11050. Ishiuchi, N., Nakashima, A., Doi, S., Yoshida, K., Maeda, S., Kanai, R., Yamada, Y., Ike, T., Doi, T., Kato, Y. and Masaki, T. 2020. Hypoxia-preconditioned mesenchymal stem cells prevent renal fibrosis and inflammation in ischemia-reperfusion rats. Stem. Cell. Res. Therapy. 11(30), 1–15. Jaillard, A., Hommel, M., Moisan, A., Zeffiro, T.A., Favre-Wiki, I.M., Barbieux-Guillot, M., Vadot, W., Marcel, S., Lamalle, L., Grand, S. and Detante, O. 2020. Autologous mesenchymal stem cells improve motor recovery in subacute ischemic stroke: a randomized clinical trial. Transl. Stroke Res. 11(5), 910. Jiang, H., Zhao, H., Zhang, M., He, Y., Li, X., Xu, Y. and Liu, X. 2022. Hypoxia induced changes of exosome cargo and subsequent biological effects. Front. Immunol. 13, 1–26. Jiang, R.H., Wu, C.J., Xu, X.Q., Lu, S.S., Zu, Q.Q., Zhao, L.B., Wang, J., Liu, S. and Shi, H.B. 2019. Hypoxic conditioned medium derived from bone marrow mesenchymal stromal cells protects against ischemic stroke in rats. J. Cell. Physiol. 234(2), 1354–1368. Jiang, W., Zhao, Y., Liu, R., Zhang, B., Xie, Y., Gao, B., Shi, K., Zou, M., Jia, D., Ding, J., Hu, X., Duan, Y., Han, R., Huang, D., Van Kaer, L. and Shi, F.D. 2024. Histidine-rich glycoprotein modulates neutrophils and thrombolysis-associated hemorrhagic transformation. EMBO. Mol. Med. 16(9), 2146–2169. Jumena, F., Amalina, N.D. and Dewi Antari, A. 2023. The effect of hypoxic mesenchymal cell secretome administration on VEGF levels in type 1 diabetes rats model. Int. J. Cell Biomed. Sci. 1(3), 86–92. Kahrizi, M.S., Mousavi, E., Khosravi, A., Rahnama, S., Salehi, A., Nasrabadi, N., Ebrahimzadeh, F. and Jamali, S. 2023. Recent advances in pre-conditioned mesenchymal stem/stromal cell (MSCs) therapy in organ failure; a comprehensive review of preclinical studies. Stem Cell Res. Ther. 14(1), 155. Kim, J.S. 2019. TPA helpers in the treatment of acute ischemic stroke: are they ready for clinical use?. J. Stroke 160–174. Kim, R., Kim, P., Lee, C.Y., Lee, S., Yun, H., Lee, M.Y., Kim, J., Baek, K. and Chang, W. 2018. Multiple combination of Angelica gigas extract and mesenchymal stem cells enhances therapeutic effect. Biol. Pharm. Bull. 41(12), 1748–1756. Kim, Y.S., Noh, M.Y., Cho, K.A., Kim, H., Kwon, M.S., Kim, K.S., Kim, J., Koh, S.H. and Kim, S.H. 2015. Hypoxia/Reoxygenation-preconditioned human bone marrow-derived mesenchymal stromal cells rescue ischemic rat cortical neurons by enhancing trophic factor release. Mol. Neurobiol. 52(1), 792–803. Kong, D., Zhu, J., Liu, Q., Jiang, Y., Xu, L., Luo, N., Zhao, Z., Zhai, Q., Zhang, H., Zhu, M. and Liu, X. 2017. Mesenchymal stem cells protect neurons against hypoxic-ischemic injury via inhibiting parthanatos, necroptosis, and apoptosis, but not autophagy. Cell. Mol. Neurobiol. 37(2), 303–313. Kuntjoro, M., Hendrijantini, N., Prasetyo, E.P., Agustono, B. and Hong, G. 2025. Hypoxia-preconditioned human umbilical cord mesenchymal stem cells transplantation Ameliorates Inflammation and Bone Regeneration in Peri-Implantitis Rat Model. Eur. J. Dent. 19(02), 420–427. Lapi, D., Vagnani, S., Sapio, D., Mastantuono, T., Boscia, F., Pignataro, G., Penna, C., Pagliaro, P. and Colantuoni, A. 2015. Effects of bone marrow mesenchymal stem cells (BM-MSCs) on rat pial microvascular remodeling after transient middle cerebral artery occlusion. Front. Cell. Neurosci. 9, 329. Laskowitz, D.T., Bennett, E.R., Durham, R.J., Volpi, J.J., Wiese, J.R., Frankel, M., Shpall, E., Wilson, J.M., Troy, J. and Kurtzberg, J. 2018. Allogeneic umbilical cord blood infusion for adults with ischemic stroke: clinical outcomes from a phase I safety study. Stem. Cells. Transl. Med. 7(7), 521–529. Lee, J.Y., Kim, H.S., Kim, S.H., Kim, H.S. and Cho, B.P. 2020. Combination of human mesenchymal stem cells and repetitive transcranial magnetic stimulation enhances neurological recovery of 6-hydroxydopamine model of parkinsonian’s disease. Tissue Eng. Regen. Med. 17(1), 67–80. Leschik, J., Gentile, A., Cicek, C., Péron, S., Tevosian, M., Beer, A., Radyushkin, K., Bludau, A., Ebner, K., Neumann, I., Singewald, N., Berninger, B., Lessmann, V. and Lutz, B. 2022. Brain-derived neurotrophic factor expression in serotonergic neurons improves stress resilience and promotes adult hippocampal neurogenesis. Prog. Neurobiol. 217, 102333. Li, H., Kittur, F.S., Hung, C.Y., Li, P.A., Ge, X., Sane, D.C. and Xie, J. 2020. Quantitative proteomics reveals the beneficial effects of low glucose on neuronal cell survival in an in vitro Ischemic penumbral model. Front. Cell. Neurosci. 14. Li, W., Shi, L., Hu, B., Hong, Y., Zhang, H., Li, X. and Zhang, Y. 2021. Mesenchymal stem cell-based therapy for stroke: current understanding and challenges. Front. Cell. Neurosci. 15 . Li, X., Elsherbiny, N.M., Ezquer, F., Del Desarrollo, U., Mudasir, C., Gugjoo, B., Bai, X.-Y. and Zhou, C. n.d. Strategies for the induction of anti-inflammatory mesenchymal stem cells and their application in the treatment of immune-related nephropathy. Lin, L., Ba, Z., Tian, H., Qin, H., Chen, X., Zhou, X., Zhao, S., Li, L., Xue, F., Li, H., He, L., Li, X., Du, J., Zhou, Z. and Zeng, W. 2024. Ultrasound-responsive theranostic platform for the timely monitoring and efficient thrombolysis in thrombi of tPA resistance. Nature Commun. 15(1), 6610. Liu, J., He, J., Ge, L., Xiao, H., Huang, Y., Zeng, L., Jiang, Z., Lu, M. and Hu, Z. 2021b. Hypoxic preconditioning rejuvenates mesenchymal stem cells and enhances neuroprotection following intracerebral hemorrhage via the miR-326-mediated autophagy. Stem Cell Res. Therapy 12(1), 1. Liu, J., He, J., Huang, Y., Ge, L., Xiao, H., Zeng, L., Jiang, Z., Lu, M. and Hu, Z. 2021a. Hypoxia-preconditioned mesenchymal stem cells attenuate microglial pyroptosis after intracerebral hemorrhage. Ann. Transl. Med. 9(17), 1362. Liu, X., Wang, J., Wang, P., Zhong, L., Wang, S., Feng, Q., Wei, X. and Zhou, L. 2022. Hypoxia-pretreated mesenchymal stem cell-derived exosomes-loaded low-temperature extrusion 3D-printed implants for neural regeneration after traumatic brain injury in canines. Front. Bioeng. Biotechnol. 10(September), 1–18. Luo, Z., Wu, F., Xue, E., Huang, L., Yan, P., Pan, X. and Zhou, Y. 2019. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival by inducing HIF-1α in injured neuronal cells derived exosomes culture system. Cell Death Discovery 10(2), 1–11. Majumdar, D., Bhonde, R. and Datta, I. 2013. Influence of ischemic microenvironment on human Wharton’s Jelly mesenchymal stromal cells. Placenta 34(8), 642–649. Mazumder, M. and Borah, A. 2015. Piroxicam confer neuroprotection in cerebral ischemia by inhibiting cyclooxygenases, acid-sensing ion channel-1a and aquaporin-4: an in silico comparison with Aspirin and Nimesulide. Bioinformation 11(4), 217–222. Mazumder, M.K. and Borah, A. 2014. Piroxicam inhibits NMDA receptor-mediated excitotoxicity through allosteric inhibition of the GluN2B subunit: an Moher, D., Liberati, A., Tetzlaff, J. and Altman, D.G. 2010. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int. J. Surg. 8(5), 336–341. Moisan, A., Favre, I., Rome, C., De Fraipont, F., Grillon, E., Coquery, N., Mathieu, H., Mayan, V., Naegele, B., Hommel, M., Richard, M.J., Barbier, E.L., Remy, C. and Detante, O. 2016. Intravenous injection of clinical grade human MSCs after experimental stroke: functional benefit and microvascular effect. Cell Transplant. 25(12), 2157–2171. Nakazaki, M., Sasaki, M., Kataoka-Sasaki, Y., Oka, S., Namioka, T., Namioka, A., Onodera, R., Suzuki, J., Sasaki, Y., Nagahama, H., Mikami, T., Wanibuchi, M., Kocsis, J.D. and Honmou, O. 2017. Intravenous infusion of mesenchymal stem cells inhibits intracranial hemorrhage after recombinant tissue plasminogen activator therapy for transient middle cerebral artery occlusion in rats. J. Neurosurg. 127(4), 917–926. Nguyen, T.N., Raymond, J., Nogueira, R.G., Fischer, U. and Siegler, J.E. 2022. the problem of restrictive thrombectomy trial eligibility criteria. Stroke 53(9), 2988–2990. Noverina, R., Faried, A., Ayuningtyas, W., Kurniawan, D., Bolly, H., Bachtiar, I. and Wirakusumah, F.F. 2019. Allogeneic amniotic membrane-derived 3-dimensional culture of mesenchymal stem cells promotes rapid burn wound closure in a non-human primate burn wound injury Model. SSRN Electron. J. Patil, S., Fageeh, H.N., Fageeh, H.I., Ibraheem, W., Alshehri, A.S., Al-Brakati, A., Almoammar, S., Almagbol, M., Dewan, H., Khan, S.S., Baeshen, H.A., Patil, V.R., Raj, A.T. and Bhandi, S. 2022. Hypoxia, a dynamic tool to amplify the gingival mesenchymal stem cells potential for neurotrophic factor secretion. Saudi J. Biol. Sci. 29(5), 3568–3576. Peña, I. dela, Borlongan, C., Shen, G. and Davis, W. 2017. Strategies to extend thrombolytic time window for Ischemic stroke treatment: an unmet clinical need. J. Stroke 19(1), 50–60. Pulido-Escribano, V., Torrecillas-Baena, B., Camacho-Cardenosa, M., Dorado, G., Gálvez-Moreno, M.A. and Casado-Díaz, A. 2022. Role of hypoxia preconditioning in therapeutic potential of mesenchymal stem-cell-derived extracellular vesicles. World J. Stem Cells 14(7), 435–576. Qian, J.Y., Chopp, M. and Liu, Z. 2016. Mesenchymal stromal cells promote axonal outgrowth alone and synergistically with astrocytes via tPA. PLos One 11(12), 168345. Quan, X., Liang, X., Ding, Y., Han, Y., Li, J., Yuan, M., Li, Y., Yuan, Z., Wang, R. and Zhao, Y. 2023. Cryo-shocked platelet coupled with ros-responsive nanomedicine for targeted treatment of thromboembolic disease. ACS. Nano. 17(7), 6519–6533. Rahman, A.A., Amruta, N., Pinteaux, E. and Bix, G.J. 2021. Neurogenesis after stroke: a therapeutic perspective. Transl. Stroke Res. 12, 1–14. Rinendyaputri, R., Noviantari, A., Zainuri, M., Nikmah, U.A., Dany, F. and Intan, P.R. 2023. Secretion of growth factor in conditioned medium rat bone marrow-derived mesenchymal stem cells (CM-rBMMSC). American Institute of Physics Inc, p 040003. Satani, N., Zhang, X., Giridhar, K., Wewior, N., Cai, C., Aronowski, J. and Savitz, S.I. 2021. A combination of atorvastatin and aspirin enhances the pro-regenerative interactions of marrow stromal cells and stroke-derived monocytes in vitro. Front. Pharmacol. 12, 589418. Sidharta, V.M., Herningtyas, E.H., Lagonda, C.A., Fauza, D., Kusnadi, Y., Susilowati, R. and Partadiredja, G. 2018. High VEGF level is produced by human umbilical cord- mesenchymal stem cells (hUC-MSCs) in amino acid-rich medium and under hypoxia condition. Indonesian Biomed. J. 10(3), 222–230. Song, Y., Liang, F., Tian, W., Rayhill, E., Ye, L. and Tian, X. 2025. Optimizing therapeutic outcomes: preconditioning strategies for MSC-derived extracellular vesicles. Front. Pharmacol. 16. Tian, H., Yang, X., Zhao, J., Liu, X., Liu, X., Cai, Y., Wehbe, A., Ding, Y., Yu, S., Wei, L. and Liu, L. 2023. Hypoxia-preconditioned bone marrow mesenchymal stem cells improved cerebral collateral circulation and stroke outcome in mice. Arterioscler. Thromb. Vasc. Biol. 43(7), 1281–1294. Toghiani, R., Azimian Zavareh, V., Najafi, H., Mirian, M., Azarpira, N., Abolmaali, S.S., Varshosaz, J. and Tamaddon, A.M. 2024. Hypoxia-preconditioned WJ-MSC spheroid-derived exosomes delivering miR-210 for renal cell restoration in hypoxia-reoxygenation injury. Stem Cell Res. Ther. 15(1), 240. Trotman-Lucas, M. and Gibson, C.L. 2021. A review of experimental models of focal cerebral ischemia focusing on the middle cerebral artery occlusion model. F1000Research 10, 242. Trzyna, A. and Banaś-Ząbczyk, A. 2021. Adipose-derived stem cells secretome and its potential application in ‘stem cell-free therapy’. Biomolecules 11(878), 1–24. Uzdensky, A.B. 2019. Apoptosis regulation in the penumbra after ischemic stroke: expression of pro- and antiapoptotic proteins. Apoptosis 24(9–10), 687–702. Wang, Y., Deng, Y. and Zhou, G. 2007. SDF-1 α / CXCR4-mediated migration of systemically transplanted bone marrow stromal cells towards ischemic brain lesion in a rat model. 5. Wang, C., Tian, C., Cai, D., Jiang, H., Zhang, W., Liu, S., Peng, L. and Hu, X. 2022. BDNF-overexpressing MSCs delivered by hydrogel in acute ischemic stroke treatment. Ann. Transl. Med. 10(24), 1393. Wang, X., Liu, C., Li, S., Xu, Y., Chen, P., Liu, Y., Ding, Q., Wahafu, W., Hong, B. and Yang, M. 2015. Hypoxia precondition promotes adipose-derived mesenchymal stem cells based repair of diabetic erectile dysfunction via augmenting angiogenesis and neuroprotection. PLos One 10(3), 3. Wei, L., Fraser, J.L., Lu, Z.Y., Hu, X. and Yu, S.P. 2012. Transplantation of hypoxia preconditioned bone marrow mesenchymal stem cells enhances angiogenesis and neurogenesis after cerebral ischemia in rats. Neurobiol. Dis. 46(3), 635–645. WHO. 2024. The top 10 causes of death. Available via https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (Accessed 15 December 2025) Xia, Y., Ling, X., Hu, G., Zhu, Q., Zhang, J., Li, Q., Zhao, B., Wang, Y. and Deng, Z. 2020. Small extracellular vesicles secreted by human iPSC-derived MSC enhance angiogenesis through inhibiting STAT3-dependent autophagy in ischemic stroke. Stem Cell Res. Therapy 11(1), 1. Xin, H., Chopp, M., Shen, L.H., Zhang, R.L., Zhang, L., Zhang, Z.G. and Li, Y. 2013. Multipotent mesenchymal stromal cells decrease transforming growth factor β1 expression in microglia/macrophages and down-regulate plasminogen activator inhibitor 1 expression in astrocytes after stroke. Neurosci. Lett. 542, 81–86. Xu, H., Miki, K., Ishibashi, S., Inoue, J., Sun, L., Endo, S., Sekiya, I., Muneta, T., Inazawa, J., Dezawa, M. and Mizusawa, H. 2010. Transplantation of neuronal cells induced from human mesenchymal stem cells improves neurological functions after stroke without cell fusion. J. Neurosci. Res. 88(16), 3598–3609. Xu, Z., Lin, L., Fan, Y., Huselstein, C., De Isla, N., He, X., Chen, Y. and Li, Y. 2022. Secretome of mesenchymal stem cells from consecutive hypoxic cultures promotes resolution of lung inflammation by reprogramming anti-inflammatory macrophages. Int. J. Mol. Sci. 23(8), 4333. Yan, M., Li, Z., Dai, S., Li, S. and Yu, P. 2023. The potential effect of salvianolic acid B against rat ischemic brain injury in combination with mesenchymal stem cells. J. Chem. Neuroanat. 133(102338), 102338. Yang, Y., Hu, X., Qin, Q., Kong, F., Peng, X., Zhao, J., Si, J., Yang, Z. and Xie, S. 2022. Optimal therapeutic conditions for the neural stem cell-based management of ischemic stroke: a systematic review and network meta-analysis based on animal studies. BMC Neurol. 22(1), 1. Yaqubi, S. and Karimian, M. 2024. Stem cell therapy as a promising approach for ischemic stroke treatment. Curr. Res. Pharmacol. Drug Discov. 6, 100183. Ye, Y., Zhao, X., Xu, Y. and Yu, J. 2021. Hypoxia-inducible non-coding RNAs in mesenchymal stem cell fate and regeneration. Front. Dent. Med. 2. Yu, X., Zheng, X., and Cheng, D. 2022. Curative effect of interventional therapy and neurological changes in Ischemic stroke of posterior circulation evaluated by magnetic resonance imaging under genetic Algorithm. Sci. Program. 2022, 1–11. Yuan, F., Liu, J., Zhong, L., Liu, P., Li, T., Yang, K., Gao, W., Zhang, G., Sun, J. and Zou, X. 2024. Enhanced therapeutic effects of hypoxia-preconditioned mesenchymal stromal cell-derived extracellular vesicles in renal ischemic injury. Yuan, H., Liu, J., Gu, Y., Ji, X. and Nan, G. 2022. Intermittent hypoxia conditioning as a potential prevention and treatment strategy for ischemic stroke: current evidence and future directions. Front. Neurosci. 16, 1. Zacharek, A., Chen, J., Li, A., Cui, X., Li, Y., Roberts, C., Feng, Y., Gao, Q. and Chopp, M. 2007. Angiopoietin1/Tie2 and VEGF/Flk1 induced by MSC treatment amplifies angiogenesis and vascular stabilization after stroke. J. Cerebral Blood Flow Metab. 27(10), 1684–1691. Zhang, Q., Chen, Z.W., Zhao, Y.H., Liu, B.W., Liu, N.W., Ke, C.C. and Tan, H.M. 2017. Bone marrow stromal cells combined with sodium ferulate and n-butylidenephthalide promote the effect of therapeutic angiogenesis via advancing astrocyte-derived trophic factors after ischemic stroke. Cell Transplant. 26(2), 229–242. Zhang, Q., Zhao, Y., Xu, Y., Chen, Z., Liu, N., Ke, C., Liu, B. and Wu, W. 2016. Sodium ferulate and n-butylidenephthalate combined with bone marrow stromal cells (BMSCs) improve the therapeutic effects of angiogenesis and neurogenesis after rat focal cerebral ischemia. J. Transl. Med. 14(1), 223. Zhang, X.L., Zhang, X.G., Huang, Y.R., Zheng, Y.Y., Ying, P.J., Zhang, X.J., Lu, X., Wang, Y.J. and Zheng, G.Q. 2021. Stem cell-based therapy for experimental ischemic stroke: a preclinical systematic review. Front. Cell. Neurosci. 15. Zhang, Y., Ma, L., Su, Y., Su, L., Lan, X., Wu, D., Han, S., Li, J., Kvederis, L., Corey, S., Borlongan, C.V. and Ji, X. 2019a. Hypoxia conditioning enhances neuroprotective effects of aged human bone marrow mesenchymal stem cell-derived conditioned medium against cerebral ischemia in vitro. Brain Res. 1725(146432):146432. Zhang, Y., Ma, L., Su, Y., Su, L., Lan, X., Wu, D., Han, S., Li, J., Kvederis, L., Corey, S., Borlongan, C.V. and Ji, X. 2019b. Hypoxia conditioning enhances neuroprotective effects of aged human bone marrow mesenchymal stem cell-derived conditioned medium against cerebral ischemia in vitro. Brain Res. 1725, 146432. Zhou, L., Lin, Q., Wang, P., Yao, L., Leong, K., Tan, Z. and Huang, Z. 2017. Enhanced neuroprotective efficacy of bone marrow mesenchymal stem cells co-overexpressing BDNF and VEGF in a rat model of cardiac arrest-induced global cerebral ischemia. Cell Death Dis. 8(5), e2774–e2710. | ||
| How to Cite this Article |
| Pubmed Style Rinendyaputri R, Nikmah UA, Panjaitan NSD, Intan PR, Situmorang JH, Idrus HH, Rilianawati R, Ayuningtyas W, Noverina R, Huda F, Faried A. Preclinical studies of conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM for ischemic stroke: A systematic review. Open Vet. J.. 2026; 16(1): 41-55. doi:10.5455/OVJ.2026.v16.i1.4 Web Style Rinendyaputri R, Nikmah UA, Panjaitan NSD, Intan PR, Situmorang JH, Idrus HH, Rilianawati R, Ayuningtyas W, Noverina R, Huda F, Faried A. Preclinical studies of conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM for ischemic stroke: A systematic review. https://www.openveterinaryjournal.com/?mno=279773 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.4 AMA (American Medical Association) Style Rinendyaputri R, Nikmah UA, Panjaitan NSD, Intan PR, Situmorang JH, Idrus HH, Rilianawati R, Ayuningtyas W, Noverina R, Huda F, Faried A. Preclinical studies of conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM for ischemic stroke: A systematic review. Open Vet. J.. 2026; 16(1): 41-55. doi:10.5455/OVJ.2026.v16.i1.4 Vancouver/ICMJE Style Rinendyaputri R, Nikmah UA, Panjaitan NSD, Intan PR, Situmorang JH, Idrus HH, Rilianawati R, Ayuningtyas W, Noverina R, Huda F, Faried A. Preclinical studies of conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM for ischemic stroke: A systematic review. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 41-55. doi:10.5455/OVJ.2026.v16.i1.4 Harvard Style Rinendyaputri, R., Nikmah, . U. A., Panjaitan, . N. S. D., Intan, . P. R., Situmorang, . J. H., Idrus, . H. H., Rilianawati, . R., Ayuningtyas, . W., Noverina, . R., Huda, . F. & Faried, . A. (2026) Preclinical studies of conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM for ischemic stroke: A systematic review. Open Vet. J., 16 (1), 41-55. doi:10.5455/OVJ.2026.v16.i1.4 Turabian Style Rinendyaputri, Ratih, Uly Alfi Nikmah, Novaria Sari Dewi Panjaitan, Putri Reno Intan, Jiro Hasegawa Situmorang, Hasta Handayani Idrus, Rilianawati Rilianawati, Wireni Ayuningtyas, Rachmawati Noverina, Fathul Huda, and Ahmad Faried. 2026. Preclinical studies of conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM for ischemic stroke: A systematic review. Open Veterinary Journal, 16 (1), 41-55. doi:10.5455/OVJ.2026.v16.i1.4 Chicago Style Rinendyaputri, Ratih, Uly Alfi Nikmah, Novaria Sari Dewi Panjaitan, Putri Reno Intan, Jiro Hasegawa Situmorang, Hasta Handayani Idrus, Rilianawati Rilianawati, Wireni Ayuningtyas, Rachmawati Noverina, Fathul Huda, and Ahmad Faried. "Preclinical studies of conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM for ischemic stroke: A systematic review." Open Veterinary Journal 16 (2026), 41-55. doi:10.5455/OVJ.2026.v16.i1.4 MLA (The Modern Language Association) Style Rinendyaputri, Ratih, Uly Alfi Nikmah, Novaria Sari Dewi Panjaitan, Putri Reno Intan, Jiro Hasegawa Situmorang, Hasta Handayani Idrus, Rilianawati Rilianawati, Wireni Ayuningtyas, Rachmawati Noverina, Fathul Huda, and Ahmad Faried. "Preclinical studies of conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM for ischemic stroke: A systematic review." Open Veterinary Journal 16.1 (2026), 41-55. Print. doi:10.5455/OVJ.2026.v16.i1.4 APA (American Psychological Association) Style Rinendyaputri, R., Nikmah, . U. A., Panjaitan, . N. S. D., Intan, . P. R., Situmorang, . J. H., Idrus, . H. H., Rilianawati, . R., Ayuningtyas, . W., Noverina, . R., Huda, . F. & Faried, . A. (2026) Preclinical studies of conditioned medium of hypoxic-preconditioned mesenchymal stem cell/hypMSC-CM for ischemic stroke: A systematic review. Open Veterinary Journal, 16 (1), 41-55. doi:10.5455/OVJ.2026.v16.i1.4 |