| Research Article | ||
Open Vet. J.. 2026; 16(1): 492-502 Open Veterinary Journal, (2026 ), Vol. 16(1): 492-502 Research Article Role of coenzyme Q10 on some physiological aspects of male rat exposure to silicon dioxide nanoparticlesMahmood S. Al-Maatheedi, Suha A. Rasheed* and Rana A. AsimDepartment of Physiology, Biochemistry and Pharmacology, College of Veterinary Medicine, University of Mosul, Mosul, Iraq *Corresponding Author: Suha A. Rasheed. Department of Physiology, Biochemistry and Pharmacology, College of Veterinary Medicine, University of Mosul, Mosul, Iraq. Email: suharasheed [at] uomosul.edu.iq Submitted: 06/09/2025 Revised: 01/12/2025 Accepted: 09/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
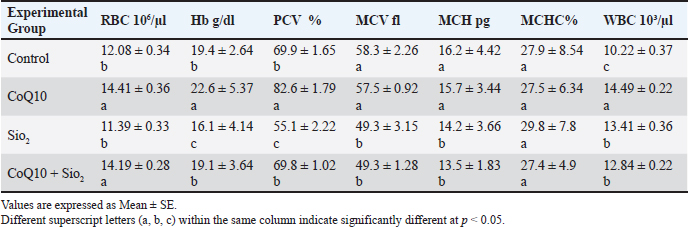
AbstractBackground: This study investigated how coenzyme Q10 (CoQ10) supplementation protected male rats against hematological abnormalities, oxidative stress, hormonal imbalance, and reproductive failure imposed by silicon oxide (SiO2) nanoparticles (SiO2) Aim: This study aimed to elucidate the mechanism of action of coenzyme Q10 supplement on some physiological disorders induced by SiO2-NPs in male Wistar rats. Methods: Four experimental groups were investigated: control group, CoQ10 group, SiO2 group, and (CoQ10 + SiO2) combination treatment groups. Results: There were significantly altered results of the blood profile. Also, Higher neutrophil to lymphocyte ratios show that SiO2 also significantly altered the leukocyte differential in favor of neutrophilia, indicating inflammatory stress. By lowering malondialdehyde levels and hence preserving erythrocytic parameters within reference limits, CoQ10 therapy alone considerably increased Red blood cell, Hemoglobin, Packed cell volume and white blood cell counts while greatly improving total antioxidant capacity. CoQ10 also reduced the hormonal alterations brought on by SiO2, therefore conserving luteinizing hormone and testosterone levels. While CoQ10 treatment considerably enhanced sperm shape, motility, and count, SiO2 drastically reduced assessments of sperm quality, including these components. CoQ10 was shown to reduce SiO2-induced testicular damage, hence preserving seminiferous tubule shape and spermatogenic integrity. Under SiO2 immunohistochemistry studies, CoQ10 supplementation reduced apoptosis-inducing factor and enhanced synptonemal complex protein 3. Conclusion: Our study concludes the mechanisms of meiosis and mitochondrial function retention. Since CoQ10 clearly shields against oxidative, hormonal, reproductive, and hematological damage, our data, combined, reveal the therapeutic potential in treating physiological abnormalities induced by SiO2 NPs. Keywords: Coenzyme Q10, Immunohistochemistry, physiological dysfunction, Silicon dioxide nanoparticles, Sperm quality in rats. IntroductionNanotechnology has revolutionized various fields, including medicine, cosmetics, agriculture, and food production (Malik et al., 2023; Sekhon, 2014). Among the most widely used nanomaterials are silicon dioxide nanoparticles (SiO2-NPs), which are commonly used due to their chemical stability, biocompatibility, and wide-ranging functional applications (Ahuja et al., 2024). However, the very properties that make these NPs versatile also raise concerns about their potential to penetrate biological systems and disrupt cellular homeostasis (Yusuf et al., 2023). Recent studies have shown that exposure to many NPs, including SiO2-NP, can cause a range of toxicological effects, largely mediated by oxidative stress, inflammation, and Deoxyribonucleic acid (DNA) damage (Cagalinec, 2017). These effects are particularly pronounced in tissues with high metabolic and proliferative activity, such as the hematopoietic system and reproductive organs. Infertility is typically a result of active sexual dysfunction, congenital or acquired abnormalities, cancers, elevated scrotal temperature, endocrine problems, and genetic, immunological, and idiopathic reasons (Guo et al., 2022). The male reproductive system, including the testes, is highly susceptible to oxidative damage due to the abundance of polyunsaturated fatty acids in sperm membranes and the reliance of spermatogenesis on tightly regulated redox balance (Ferramosca and Zara, 2022; Van Tran et al., 2016). Exposure to SiO2-NPs has been associated with decreased sperm quality, impaired steroidogenesis, histological abnormalities in the testicular architecture, and hormonal imbalances (Guo et al., 2022). Given the growing prevalence of environmental nanoparticle exposure, there is an urgent need to identify agents that can mitigate or prevent such damage. Reduced testicular blood flow, resulting in hypoxia and accumulation of harmful metabolites, causes male reproductive failure through energy depletion and oxidative damage (Obokhwo et al., 2024). Characterized by too high reactive oxygen species (ROS), oxidative stress negatively influences sperm DNA integrity, protein function, and lipid membranes, compromising sperm function (Chianese and Pierantoni, 2021) that rich in polyunsaturated fatty acids, mammal spermatozoa are especially susceptible to ROS-induced lipid peroxidation, reducing reproductive potential. Ubiquinone, the crucial antioxidant that makes up coenzyme Q10, is often present in higher organisms and is the only fat-soluble antioxidant that can be generated by the body (antioxidant that can be generated by the body that is fat-soluble (Podar et al., 2023; Feher et al., 2007). Ubiquinone is one of the molecules that transport electrons and protons in the oxidative phosphorylation forms found in cell mitochondria and plays a crucial role in the creation of vitality (Zong et al., 2024; Nolfi-Donegan et al., 2020). It is a naturally occurring lipophilic antioxidant located in the inner mitochondrial membrane and plays a dual role in cellular bioenergetics and protection against oxidative damage (Gasmi et al., 2024; Albadrany and Naser, 2020). coenzyme Q10 (CoQ10) participates in the electron transport chain, facilitating adenosine triphosphate production, while simultaneously scavenging ROS and regenerating other antioxidants, such as vitamin E (He et al., 2017). It enhances antioxidant defense, improves mitochondrial integrity, and supports reproductive function (Nie et al., 2023). It also modulates apoptosis and inflammatory pathways, making it a promising therapeutic candidate for reducing nanoparticle-induced toxicity (Qiu et al., 2024). In this study, we aimed to explain the mechanism of action of coenzyme Q10 supplements against some physiological disorders induced by SiO2-NPs in male Wistar rats, including comprehensive assessments of hematological parameters, oxidative stress biomarkers, sperm quality indices, and immunohistopathological alterations in testicular tissue. The outcomes of this research are expected to provide a scientific basis for assessing the danger of nano-SiO2-induced toxicity and the therapeutic potential of protective effects of CoQ10 as a countermeasure. Understanding these interactions is crucial not only for toxicological risk assessment but also for developing strategies to safeguard reproductive health in environments increasingly permeated by nanomaterials. Materials and MethodsExperimental animalsThis work has been done on 32 adults male Wistar albino rats. 3 months old, it weighed 320 ± 10 g. The College of Veterinary Medicine, University of Mosul, adopted them in their animal house. During the trial, the rats were housed in a well-ventilated room within plastic cages and fed a normal pellet diet along with drinking water ad libitum. During the trial period, the room temperature was 23ºC ± 2ºC for 12 hours, light/dark cycle (Jasim and Rasheed, 2021). Preparation of SiO2 NPs suspensionThe preparation of SiO2NPs used in this study was prepared by dissolving the powder of SiO2NPs in deionized water, and the suspension was dispersed by a vortex shaker for 10 minutes at 37°C immediately before administration to rats. A solution was prepared so that the dose could be administered at 1 ml via the intraperitoneal route (i.p) (Song et al., 2008). Experimental designThirty-two healthy male Wister albino rats were chosen at random and split into four equal groups (n=8 rats) and handled daily as follows: Group I (Control): Each rat received 1 ml of deionized water by oral gavage and intraperitoneal injection as a placebo once daily for 42 days. Group II: Each rat receives oral coenzyme Q10 (10 mg/kg body weight) once daily for 42 days. Group III: Each rat received SiO2 NPs (5 mg/kg body weight intraperitoneal injection, i.p.) once daily for 42 days. Group IV (Coenzyme Q10 + SiO2 NPs): Each rat received Coenzyme Q10 (10 mg/kg/day oral gavage) and SiO2 NPs (5 mg/kg body weight intraperitoneal injection, i.p.) once daily for 42 days. Each rat treated with Coenzyme Q10 received 1 ml of deionized water daily intraperitoneally, and the rat treated with SiO2 NPs received 1 ml of deionized water daily orally by gavage as a placebo. We decided on the route of administration and the concentration based on previous studies (Sharma et al., 2012; Almansour et al., 2018). Blood sampling and analysisAfter 42 days of treatment, rats were anesthetized using ether inhalation, and blood samples were drawn by retro-orbital Sinus technique into tubes containing Ethylene diamine tetra acetic acid as an anticoagulant. Blood samples were analyzed within 2 hours of collection (Al Nuaimmi and Rasheed, 2023). Hematological parameters were measured using an automated hematology analyzer (GP Animal, Getein Animal Medical BHA-5000 Vet). Serum was obtained by centrifugation of blood samples at 3,000 rpm for 10 minutes. Serum testosterone and luteinizing hormone (LH) levels were measured using enzyme-linked immunosorbent assay kits according to the manufacturer’s protocols (Rasheed and Al Nuaimi, 2022). Serum malondialdehyde (MDA) levels were measured using the BC0025 MDA Content Assay Kit-20210709 thiobarbituric acid reactive substance method. The total antioxidant capacity [BC1310 total antioxidant capacity (TAC) Assay Kit 20201020] was determined using an antioxidant assay kit following the manufacturer’s instructions (Al Ma’atheedi, 2014; He et al., 2017; Rasheed et al., 2022). Semen analysisThe epididymides were carefully excised and minced in physiological saline at 37°C to allow sperm release. Sperm count was measured using a hemocytometer under a light microscope. Motility was assessed immediately using a phase-contrast microscope, and the percentage of live and dead sperm was evaluated using eosin-nigrosine staining. The percentage of morphologically abnormal spermatozoa was determined by microscopic examination at × 400 magnification, with 200 sperm cells assessed per sample (Salvio et al., 2021; Ferramosca and Zara. 2022). Testicular preparedness for immunohistochemistry (IHC)Testicular tissues were extracted and stored in 10% neutral-buffered formalin for 24–48 hours just after euthanasia. After fixing, the tissues were dried in graded ethanol solutions, xylene-cleaned, paraffin embedded. Slides covered in cut sections (4–5 µm thickness) were coated with poly-L-lysine and then recovered antigenically in citrate buffer (pH 6.0). Three percent hydrogen peroxide inhibited the activity of endogenous peroxidase. Sections were next treated overnight at 4°C with primary antibodies and then with biotinylated secondary antibodies and streptavidin-horseradish peroxidase complex. Under a light microscope, Mayer’s hematoxylin counterstained slices; 3,3’-diaminobenzidine visualized immunoreactivity; dehydrated, cleaned, mounted, and examined under a light microscope (Shi et al., 2023; Qiu et al., 2024). Statistical analysisData are presented as means ± standard error of the mean. One-way ANOVA followed by Duncan’s multiple range test was considered statistically significant across groups at p < 0.05 (Salvio et al., 2021). Ethical approvalThis study was conducted in accordance with the ethical standards set by the Institutional Animal Care and Use Committee at the College of Veterinary Medicine, University of Mosul. All samples were collected with the owners’ consent under the approved ID: UM.VET.2023.150, Date: 1/12/2023. ResultsTable 1 shows that among the experimental group’s different hematological parameter values. Compared with the control group (p < 0.05), CoQ10 greatly increased Red blood cell (RBC), Hemoglobin (Hb), and Packed cell volume (PCV) Within the reference range, the mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), and mean corpuscular hemoglobin concentration (MCHC) stayed statistically equal between the CoQ10 and control groups. Exposure to SiO2 NPssignificantly reduced (p < 0.05) RBC (11.39 × 106/µl), Hb, and PCV relative to both the control and CoQ10 groups. Moreover, clearly, SiO2 therapy reduced MCV, MCH, and MCHC compared to the control and CoQ10 groups. Rats co-treated with CoQ10 and SiO2 showed partial restoration of RBC count (14.19 × 106/µl), Hb, and PCV, but not quite approaching those reported in the CoQ10-only group. Much above the SiO2 group. MCV, MCH, and MCHC values in the combined therapy group nonetheless improved over the SiO2 group alone, even if they stayed much lower than in the control and CoQ10-only groups. With the greatest levels seen in the CoQ10-treated rats, white blood cell (WBC) counts were considerably raised (p < 0.05) in both the CoQ10 (14.49 × 10³/µl) and SiO2 (13.41 × 10³/µl) groups relative to the control (10.22 × 10³/µl). Results fell between the CoQ10 and SiO2 groups; however, the CoQ10 + SiO2 group (12.84 × 10³/µl) also demonstrated a notable increase over the control. While SiO2 exposure caused notable hematological damage, generally CoQ10 treatment raised WBC count and improved erythrocytic indices. CoQ10 co-administration with SiO2 reduced but did not entirely counteract the negative hematological effects of SiO2. Table 1. Influence of Coenzyme Q10 and SI NPs on blood profile parameters across experimental groups.
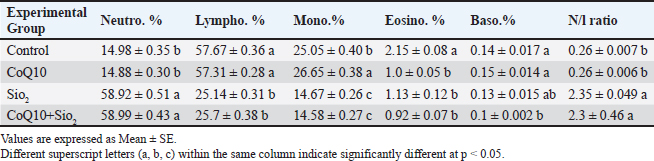
Table 2 shows the experimental group’s significant variations (p < 0.05) in the differential leukocyte counts and N/L ratio. While the lymphocyte proportions were highest (57.67% and 57.31%, respectively), accompanied by somewhat high monocyte counts (25.05% and 26.65%, respectively), the neutrophil percentages were low in the control and CoQ10-treated groups (14.98% and 14.89%). Between these groups, eosinophil and basophil percentages remained low and equal. I and II each had low N/L ratios, as well as 0.26 in everyone. A significantly higher N/L ratio (2.35) was obtained from a notable drop in lymphocyte percentage (25.14%) resulting from exposure to SiO2 and a considerable increase in neutrophil percentage (58.92). Although eosinophil and basophil values remained low, monocyte counts dropped dramatically (14.67%). The group co-treated with CoQ10 showed similar patterns to the SiO2 group with high neutrophil (58.99%), low lymphocyte (25.7%), reduced monocyte count (14.58%), and a rather high N/L ratio (2.3). Eosinophil and basophil percentages were low in all groups; basophils dropped somewhat but noticeably from the control in the CoQ10+SiO2 group. Reflecting a higher N/L ratio, SiO2 exposure clearly shifted the leukocyte differential profile relative to control toward neutrophilia and lymphopenia; CoQ10 supplementation by itself had no appreciable effect. Table 2. Influence of Coenzyme Q10 and Silicon Dioxide NPs on Differential Leukocyte Counts and Neutrophil-to-Lymphocyte Ratio Across Experimental Groups.
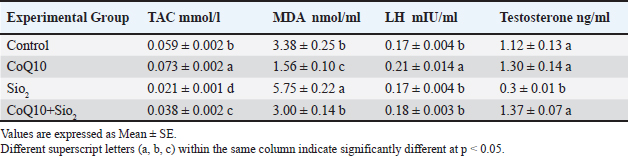
Comparing the exposition to SiO2 to the control group (p < 0.05, Table 3), oxidative stress markers and reproductive hormones were significantly changed. SiO2 clearly reduced total antioxidant capacity (TAC; 0.021 mmol/l) and dramatically increased the malondialdehyde (MDA; 5.75 nmol/ml), suggesting strong oxidative stress and lipid peroxidation. Moreover, luteinizing hormone stayed steady when compared to the control; testosterone levels in the SiO2 group were substantially lowered (0.3 ng/ml). Comparing therapy with CoQ10 alone to all other groups revealed a high antioxidant impact since it significantly raised TAC (0.073 mmol/l) and lowered MDA (1.56 nmol/ml). Furthermore, higher than the control, CoQ10 maintained 1.30 ng/ml of testosterone and greatly raised LH (0.21 mIU/ml). Together, CoQ10 and SiO2 in the group CoQ10+SiO2 assisted to slightly restored hormonal parameters and antioxidant state. Notwithstanding not returning to control or CoQ10 values, TAC (0.038 mmol/l) and MDA (3.00 nmol/ml) levels were far higher than those of the SiO2 group. In the combination group, LH and testosterone levels revealed that CoQ10 improved SiO2-induced hormonal disruption, similar to those of the control and CoQ10 groups (0.18 mIU/ml and 1.37 ng/ml, respectively). While overall CoQ10 supplements improved antioxidant capacity and maintained hormonal balance, SiO2 exposure induced significant oxidative stress and lowered testosterone levels. CoQ10 partially balanced its unfavorable effects to promote a preventive role against SiO2-induced oxidative and reproductive dysfunction caused by SiO2. Table 3. Influence of coenzyme Q10 and silicon dioxide NPs on total antioxidant capacity (TCA), malondialdehyde (MDA), luteinizing hormone (LH), and testosterone levels across experimental groups.
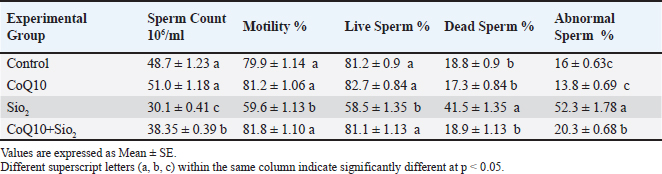
Table 4 summarizes that exposure to SiO2 significantly (p < 0.05) compromised several sperm quality measurements relative to the control and other experimental groups. Apart from a significant increase in the percentage of dead sperm (41.5%) and aberrant sperm (52.3%), the SiO2-treated group showed a clear drop in sperm count (30.1%), motility (59.6%), and live sperm percentage (58.5%). These findings unequivocally show that SiO2 has clearly negative effects on sperm health and spermatogenesis. Compared to the SiO2 group, rats receiving CoQ10 alone showed improved sperm parameters, with significantly greater sperm count (51.0 ×106/ml), motility (81.2%), and live sperm percentage (82.7%), as well as lower dead sperm (17.3%) and sperm abnormalities (13.8%). The group co-administered CoQ10 and SiO2 (CoQ10+SiO2), which showed a partial recovery of sperm quality. From the SiO2 group, the sperm count (38.35 × 106/ml) and live sperm percentage (81.1%) were much improved, even though they did not approach control or CoQ10-alone values. Particularly, motility (81.8%) and the proportion of dead sperm (18.9%) in the CoQ10+SiO2 group were restored to values comparable to those of the control and CoQ10 groups. Although they were rather lower than in the SiO2 group, sperm abnormalities (20.3%) stayed higher than those in the control and CoQ10 groups. Generally, CoQ10 treatment prevented SiO2-induced declines in sperm quality, hence supporting its function in lowering oxidative and reproductive damage. Table 4. Influence of Coenzyme Q10 and Silicon Dioxide NPs on Sperm Quality Parameters Across Experimental Groups.
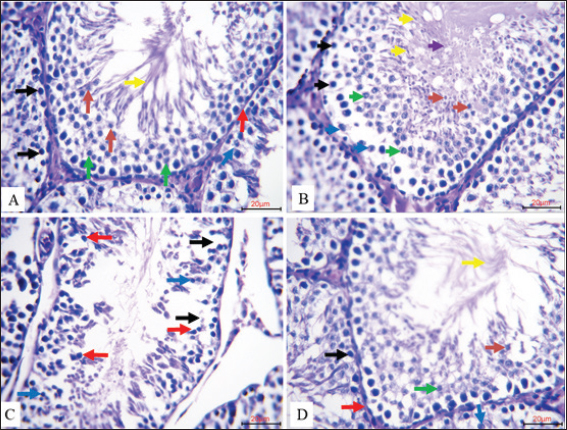
Four Experimental Groups’ valuable histological sections of rat testes are stained with hematoxylin and eosin (H&E, 20µm) from Figure 1. The seminiferous tubules in the control group (A) showed typical architecture with an ordered distribution of spermatogonia and Sertoli cells along an intact basement membrane. Clearly visible, indicating a good spermatogenic development, were primary spermatocytes, round and elongated spermatids, and mature spermatozoa, all phases of spermatogenesis. The Coenzyme Q10-treated group (B) exhibited enhanced spermatogenic activity, implying a stimulatory effect of CoQ10 on germ cell proliferation and maturation. Clearly distinct spermatogonia, Sertoli cells, primary spermatocytes, spermatids, and abundant mature spermatozoa defined this group. In contrast, the group treated with Nano-SiO2 (C) showed marked disturbance of seminiferous tubule organization, hallmarks of SiO2-induced testicular toxicity, including widening intercellular spaces and marked histopathological changes, including degeneration and necrosis of spermatogenic cells, prominent germ cell death, and detachment of spermatogonia from the basement membrane. Rats co-treated with CoQ10 and Nano-SiO2 (D) showed clearly marked preservation of testicular structure: the seminiferous tubules maintained their normal arrangement, with intact basement membranes, viable Sertoli cells, preserved spermatogonia, and the presence of all spermatogenic stages, closely resembling the control. The findings show overall how well CoQ10 protects against testicular harm triggered by SiO2.
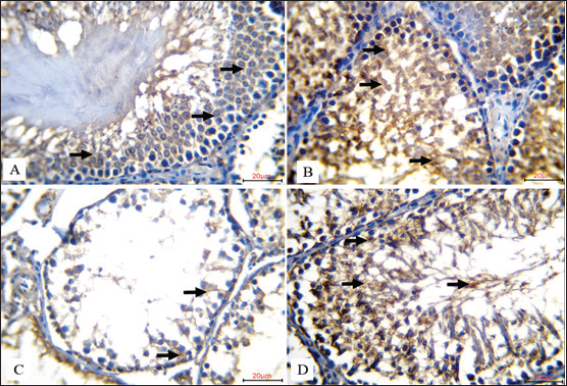
Fig. 1. Comparison of histological sections of rat testis in the four experimental groups are compared (20 µm, H&E Stain). A. Spermatogonia (black arrows) and Sertoli cells (blue arrows) in the seminiferous tubules of the control group were neatly arranged along an intact basement membrane (red arrow). The lumen displayed the typical stages of spermatogenesis, such as mature spermatozoa (yellow arrow), spherical spermatids (orange arrow), and primary spermatocytes (green arrow). B. The seminiferous tubules in the Coenzyme Q10-treated group exhibited increased spermatogenic activity with distinctive identification of spermatogonia (black arrows), Sertoli cells (blue arrows), primary spermatocytes (green arrow), round spermatids (orange arrow), elongated spermatids (yellow arrow), and mature sperm cells (purple arrow). C. spp. treated with Nano-SiO2 showed marked pathological alterations. These included spermatogenic cell degeneration and necrosis (black arrows), evident germ cell apoptosis (blue arrows), and spermatogonia detachment from the basement membrane, leaving wide intercellular spaces (red arrows), signifying toxic damage to the testicular structure D. The seminiferous tubules in the group that received both Coenzyme Q10 and Nano-SiO2 displayed with Sertoli cells (blue arrows), preserved spermatogonia (black arrows), an intact basement membrane (red arrow), and all stages of spermatogenesis present, including primary spermatocytes (green arrow), spermatids (orange arrow), and spermatozoa (yellow arrow)). Figure 2 shows, for each of the four experimental groups (20 µµm, IHC), the IHC localization of apoptosis-inducing factor (AIF) in rat testicular tissue. Strong cytoplasmic brown staining of spermatogenic cells in the seminiferous tubules of the control group (A) indicated strong physiological expression of AIF under normal conditions. Highly dense and extensive brown cytoplasmic staining revealed that the Coenzyme Q10-treated group (B) displayed a further increase in AIF expression in the spermatogenic cells. This implies that CoQ10 might help limit death by increasing mitochondrial activity. Rats treated with Nano-SiO2 (C) showed a significant reduction in AIF expression by merely faint, light brown staining in the cytoplasm of germ cells. This implies that testicular cell homeostasis is most certainly changed in sharp contrast and that the mitochondrial death signal is compromised. Strong cytoplasmic brown staining in spermatogenic cells suggests levels either equal to or greater than those observed in the control and CoQ10-only groups. Co-administration of Coenzyme Q10 with Nano-SiO2 (D) produced a notable amplification and restoration of AIF expression. These findings confirm that CoQ10 can effectively offset Nano-SiO2’s inhibitory effects on AIF expression, supporting its function in preserving mitochondrial integrity and apoptotic balance inside the testis.
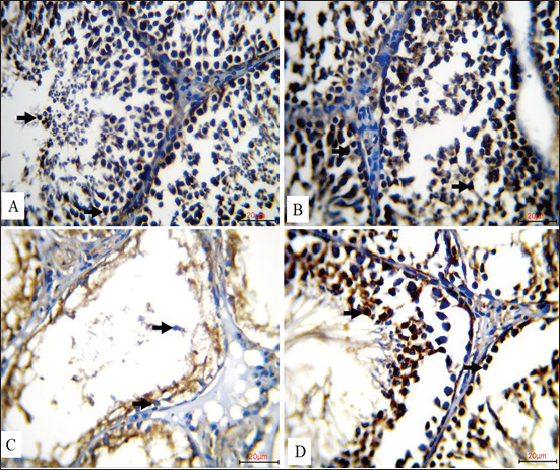
Fig. 2. Immunohistochemical expression of AIF in rat testes across experimental groups (20 µm, IHC). A. AIF was highly expressed in the control group, as evidenced by intense cytoplasmic brown staining in the spermatogenic cells of the seminiferous tubules (black arrows), reflecting normal physiological expression of the protein. B. AIF expression was significantly increased in the CoQ10-treated group, revealing a very high density of brown cytoplasmic staining in spermatogenic cells (black arrows), implying a possible function of CoQ10 in improving mitochondrial activity and apoptotic control. The cytoplasm of germ cells of C. rats exposed to Nano-SiO2 showed modest brown staining (black arrows), suggesting a decrease in AIF expression that may have been caused by harmful interference with mitochondrial apoptotic signaling. D. A prominent cytoplasmic brown signal in spermatogenic cells (black arrows) indicated that Coenzyme Q10 and Nano-SiO2 co administration greatly increased AIF expression. Figure 3 shows the immunohistochemical staining patterns for synaptonemal complex protein 3 (SYCP3) in rat testicular tissue slices from many experimental groups (20 µm, IHC). Strong nuclear brown staining in the spermatogenic cells in the control group (A) amply illustrates a high degree of SYCP3 expression, therefore showing strong and healthy meiotic development. Likewise, the group treated with Coenzyme Q10 (B) showed resilient nuclear brown staining, thereby confirming ongoing and high meiotic activity following CoQ10 injection. In contrast, the group treated with Nano-SiO2 (C) showed a 100% decrease in SYCP3 expression; spermatogenic cells showed no obvious nuclear brown staining. The evidence either amazing testicular toxicity or germ cell loss, most likely leading to decreased meiosis. Notably, nuclear brown staining in the spermatogenic cells, co-administration of Coenzyme Q10 with Nano-SiO2 (D) restored strong SYCP3 expression, in order indicating that CoQ10 supplementation was effective in counteracting nano-SiO2-induced suppression of meiotic protein expression and in supporting the recovery of normal spermatogenic processes.
Fig. 3. Illustrates the immunohistochemical expression of synaptonemal complex protein 3 (SYCP3) in rat testes across experimental groups (20 µm, IHC).A. a high degree of SYCP3 expression was noted in the control group; this was shown by strong nuclear brown staining in spermatogenic cells (black arrows), suggesting vigorous and healthy meiotic activity. Similarly, B. the group treated with Coenzyme Q10 showed a strong SYCP3 expression together with notable nuclear brown staining (black arrows). As no discernible nuclear brown staining was seen in the spermatogenic cells (black arrows), C. rats treated to Nano-SiO2 showed a total reduction of SYCP3 expression, presumably through testicular toxicity or germ cell depletion. Strong nuclear brown staining in spermatogenic cells indicates that D. co-treatment with Coenzyme Q10 and Nano-SiO2 restored high SYCP3 expression (Black arrows). DiscussionThe present study was designed to elucidate whether SiO2-NPs affect the physiological status of the male reproductive system and explore whether CoQ10 could reverse such effects. SiO2-NP led to severe oxidative stress, hematological disorder, hormonal imbalance, and reproductive toxicity in male rats. Nanoparticle-induced oxidative stress may result in hemolysis and perturb erythropoiesis. This is consistent with the elevated levels of ROS that damage cells, kill cells, and suppress the formation of erythrocytes from bone marrow (Madkour et al., 2023). In addition, the presence of NPs increases inflammation by having nanomaterials interact directly with cellular components, enhancing inflammatory reactions and giving rise to further hematological disturbances. These results are supported by other authors, who indicated that the cytotoxic and pro-oxidant, and genotoxic actions of NPs tend to be higher in more metabolically active and reproductive organs (Cagalinec, 2017; Huang et al., 2022). One of the most salient findings is that RBC, Hb, PCV, and other erythrocytic indices significantly decreased in the SiO2 group, suggesting that this anemia may be induced by oxidative injury of RBCs. Oxidative damage to erythrocyte membranes or decreased bone marrow activity is most likely the cause of this anemia. All these show an inflammatory response and immune modulation, and are consistent with the earlier reports on hematotoxic effects of metal oxide NPs (Sekhon, 2014). On the other hand, the hematological parameters showed improvement with the supplementation of CoQ10, which could be ascribed to its stabilization effect on mitochondrial membranes and reduction of lipid peroxidation in addition to increasing erythropoiesis (Albadrany and Naser, 2020; Salvio et al., 2021). All of which are conducive to the recovery of hematologic parameters. This protective effect is in line with previous studies that have reported on the potential of CoQ10 to improve general hematological status against OS and to ameliorate erythrocyte antioxidant defense systems (Paunovic et al., 2017). Analysis of leukocytes showed that the ratio of neutrophil to lymphocyte was significantly increased by SiO2 exposure (a marker of systemic inflammation). This is in line with the fact that nanoparticle accumulation leads to the activation of the innate immune responses and immunotoxicity (Sergent et al., 2012; Zaiter, 2022). Leukocyte distribution was interestingly not-significantly disturbed by CoQ10 alone (with partial inhibition of neutrophilia when co-administered with SiO2), indicating a regulating role in immunological homeostasis. In agreement with previous studies, the findings showed that CoQ10 alone reduced explosively WBC counts and raised RBC indices significantly, hence supporting erythropoiesis and reduction of inflammation (Obokohwo et al., 2024). The oxidative stress parameters supported strong SiO2-pro-oxidant activity, as indicated by increased MDA and decreased TAC. These changes are ascribable to the peroxidation of lipids and consumption of antioxidant systems. These results are in line with recent studies that exposure to SiO2 caused ROS-induced damage to the ovary and testis (Liu et al., 2024). Nevertheless, TAC was remarkably improved, and MDA was also decreased after the administration of CoQ10, which could be indicative of its role as a mitochondria antioxidant and free radical scavenger (He et al., 2017). The best thing is that CoQ10 supplements correct hormonal imbalances and improve antioxidant protection, nullifying these harmful effects. CoQ10 has the potential to be a therapeutic agent that protects against oxidative and reproductive damage induced by nanoparticle exposure; however, only partial recovery is achieved when such therapy is combined with SiO2 NPs. Despite remarkable elevations in the LH and testosterone levels of their hormone profiles, the antioxidant status in CoQ10+SiO2 group was partly recovered. Endocrine disruption induced by SiO2 was attenuated by CoQ10. The gonadotoxic effects of nanoparticle exposure were reflected by the significant decrease in testosterone and LH serum levels due to SiO2 exposure. This may be due to: testicular Leydig cell malfunction or impaired steroidogenesis. In contrast, CoQ10 not only maintained testosterone levels but also increased LH, thereby showing a dual effect on the activation of hypothalamic–pituitary–gonadal axis and protection against oxidative damage to Leydig cells (Li et al., 2019). Although significantly reducing sperm count, motility, and viability, SiO2 increased the number of dead and morphologically abnormal sperm. The result is consistent with previous reports, where oxidative stress and inflammation’s impact on spermatogenesis and sperm function has been well documented (Özgür et al., 2019). CoQ10 supplementation had a protective effect on testicular function by significantly elevating live sperm and reducing abnormal ones. While the total sperm count remained below that of control, increases in both motility and viability showed that CoQ10 functioned to protect reproductive ability by enhancing mitochondrial performance during toxic insult. The current immunohistochemical findings identify patterns of AIF expression in the rat testis with disparate experimental manipulations. The mitochondrial flavoprotein AIF, which is required for caspase-independent pathways, is a kind of death controller (Novo et al., 2021) that significantly affects not only mitochondrial quality but also the survival of spermatogenic cells. The high level of AIF expression within the testes of control rats treated with saline indicates healthy mitochondria and cellular viability in seminiferous tubules. Interestingly, the very high AIF induction observed in the CoQ10-treated group likely reflects a protective influence of CoQ10’s powerful antioxidant action at stabilizing mitochondrial membranes and preventing oxidative stress-induced cell death (Gutierrez et al., 2020). On the other hand, slightly decreased expression of AIF in rats treated with Nano-SiO2 demonstrated that silica NPs were harmful for the testes, which were usually appearing to exert via oxidative stress, mitochondrial dysfunction, and lower spermatogenesis (Rashidian et al., 2023). Notably, AIF expression was rescued when CoQ10 and Nano-SiO2 were simultaneously administered, which further illustrated a powerful prevention effect against nanoparticle-induced oxidative damage of CoQ10. Taken together, these findings lend support to the importance of mitochondrial proteins, such as AIF, in reproductive health and also importantly indicate potential avenues for therapy using antioxidants, e.g., CoQ10 to mitigate against the deleterious effects of NPs on male fertility. The results of the immunohistochemical study revealing of SYCP3 expression in rat testis are interesting new findings about reproductive biology and toxicology. Meiosis relies on SYCP3, especially in promoting recombination and synapsis of homologous chromosomes during spermatogenesis for the maintenance of sperm quality and genetic stability (De La Fuente et al., 2007; Adams and Davies, 2023). The strongly marked nuclear expression of SYCP3 detected in the control and coenzyme Q10-treated groups underscores its critical role in normal spermatogenesis. Presumably, the effectiveness of CoQ10 is derived from its ability to protect spermatogenic cells against oxidative stress and enhance meiotic efficiency. Conversely, the absence of SYCP3 in Nano-SiO2 group indicated that meiotic events might be primarily altered, probably due to oxidative stress, inflammation, or direct cytotoxic effect of such nanoparticles towards spermatogenic cells. It is NPs that the group exposed to Nano-SiO2 + CoQ10 displays recovered SYCP3 expression, suggesting a protective or regenerative effect of CoQ10 on the reproduction-damaging function of NPs. CoQ10 reduces oxidative stress or inflammatory responses induced by NPs, which can prevent meiotic activity and testicular health. The biochemical findings were confirmed by the histological and immunohistochemical investigations. The depletion of apoptotic (AIF) and meiotic (SYCP3) proteins resulted in severe testicular degeneration, failed spermatogenic layers, and a typical decreased sex ratio in SiO2 c-exposed group, indicating mitochondrial injury with defected meiosis. The CoQ10 significantly reversed these deviations, increasing the expression of AIF and SYCP3, and also ensuring the integrity of seminiferous tubules. These results demonstrate that CoQ10 can also appear support by maintaining mitochondrial function, inhibit apoptosis, and preserve germ cell development (Shi et al., 2023). Together, these findings confirm the pleiotropic action of CoQ10 as an antioxidant, endocrine modulator, and reproductive protector. Although it did not completely restore all parameters back to control levels, its capability of decreasing SiO2-mediated physiological damage significantly indicates therapeutic potential in circumstances of unavoidable NP exposure. ConclusionThe present findings provide robust and broad-spectrum evidence regarding the severity of toxic effects induced by SiO2-NPs) in male rats, including oxidative stress, blood dyscrasia, hormonal imbalance, and impairment of reproductive performance. In addition, supplementation with CoQ10 protected against testicular damage and maintained normal hematological and hormonal status. Overall, these results highlight CoQ10 as a pleiotropic preventive mediator against nanoparticle-evoked systemic dysfunctions. Therefore, CoQ10 may serve as a promising therapeutic or preventive candidate for mitigating the adverse physiological impacts associated with exposure to SiO2 NPs. AcknowledgmentWe appreciate the Deanship of the College of Veterinary Medicine for providing support in completing this study. Conflict of interestThe authors declare that no conflicts of interest arose during the writing or data analysis phases. FundingNone. Authors contributionsStudy design: M.S. Al-Maatheedi Suha A. Rasheed Collecting and analyzing data: M.S. Al-Maatheedi, Suha A. Rasheed, Rana A. Asim Examination of samples: M.S. Al-Maatheedi, Suha A. Rasheed, Rana A. Asim Statistical analysis: M.S. Al-Maatheedi Suha A. Rasheed Rana A. Asim Writing: M.S. Al-Maatheedi, Suha A. Rasheed, Rana A. Asim. Data availabilityAll data supporting the findings of this study are available within the manuscript. ReferencesAdams, I.R. and Davies, O.R. 2023. Meiotic chromosome structure, the synaptonemal complex, and infertility. Annu. Rev. Genom. Hum. Genet. 24(1), 35–61; doi:10.1146/annurev-genom-101122-103453 Ahuja, D., Akhila, M.R., Singh, A.K. and Batra, P. 2024. Impact of nanoparticles on dental composites: a systematic review and meta-analysis. J. Int. Oral Health 16(6), 439–448. Albadrany, Y. and Naser, A. 2020. Coenzyme Q10 coadministration with diclofenac augmented impaired renal function in broiler chickens (Gallus gallus domesticus). Vet. World 13(4), 642–647; doi: 10.14202/vetworld.2020.642-647 Al-Maatheedi, M.S. 2014. Effect of partial and complete extirpation of uropygial gland on some physiological traits for males quail (Coturnix coturnix). Iraqi J. Vet. Sci. 28(2), 115–125. Almansour, M., Alarifi, S. and Jarrar, B. 2018. In vivo investigation on the chronic hepatotoxicity induced by intraperitoneal administration of 10-nm silicon dioxide nanoparticles. Int. J. Nanomed. 7(13), 2685–2696; doi:10.2147/IJN.S162847 Cagalinec, M. 2017. Nanoparticles targeting mitochondria neurodegenerative diseases: challenge for nanotherapeutics. Nanomed. Neurosci. 2, 61–100; doi:10.1007/978-3-319-50661-6_4 Chianese, R. and Pierantoni, R. 2021. Mitochondrial reactive oxygen species (ROS) production alters sperm quality. Antioxidants 10(1), 92; doi:10.3390/antiox10010092 De La Fuente, R., Parra, M.T., Viera, A., Calvente, A., Gomez, R., Suja, J.A., Rufas, J.S. and Page, J. 2007. Meiotic pairing and segregation of achiasmate sex chromosomes in eutherian mammals: the role of SYCP3 protein. PLos Genet. 3(11), e198; doi:10.1371/journal.pgen.0030198 Feher, J., Nemeth, E., Nagy, V. and Lengyel, G. 2007. The preventive role of coenzyme Q10 and other antioxidants in injuries caused by oxidative stress. Arch. Med. Sci. 3(4), 305–314; doi:10.5114/aoms.2007.44838 Ferramosca, A. and Zara, V. 2022. Diet and male fertility: the impact of nutrients and antioxidants on sperm energetic metabolism. Int. J. Mol. Sci. 23(5), 2542; doi:10.3390/ijms23052542 Gasmi, A., Bjørklund, G., Mujawdiya, P.K., Semenova, Y., Piscopo, S. and Peana, M. 2024. Coenzyme Q10 in aging and disease. Crit. Rev. Food Sci. Nutr. 64(12), 3907–3919; doi:10.1080/10408398.2022.2137776 Guo, Z., Wang, X., Zhang, P., Sun, F., Chen, Z., Ma, W., Meng, F., Hao, H. and Shang, X. 2022. Silica nanoparticles cause spermatogenesis dysfunction in mice via inducing cell cycle arrest and apoptosis. Ecotoxicol. Environ. Saf. 231, 113210; doi: 10.1016/j.ecoenv.2022.113210 Gutierrez-Mariscal, F.M., Arenas-de Larriva, A.P., Limia-Perez, L., Romero-Cabrera, J.L., Yubero-Serrano, E.M. and López-Miranda, J. 2020. Coenzyme Q10 supplementation for the reduction of oxidative stress: clinical implications in the treatment of chronic diseases. Int. J. Mol. Sci. 21(21), 7870; doi:10.3390/ijms21217870 He, L., He, T., Farrar, S., Ji, L., Liu, T. and Ma, X. 2017. Antioxidants maintain cellular redox homeostasis by elimination of reactive oxygen species. Cell Physiol. Biochem. 44(2), 532–553; doi:10.1159/000485089 Huang, Y., Li, P., Zhao, R., Zhao, L., Liu, J., Peng, S., Fu, X., Wang, X., Luo, R. and Wang, R. 2022. Silica nanoparticles: biomedical applications and toxicity. Biomed. Pharmacother. 151, 113053; doi:10.1016/j.biopha.2022.113053 Jasim, A.Y. and Rasheed, S.A. 2021. Effect of vitamin C treatment on some central nervous system functions in young rats whose mothers treated with hydrogen peroxide during the lactation period. Iraqi. J. Vet. Sci. 35(4), 713–717; doi:10.33899/ijvs.2021.127894.1544 Li, X., Zhan, J., Hou, Y., Chen, S., Hou, Y., Xiao, Z., Luo, D. and Lin, D. 2019. Coenzyme Q10 suppresses oxidative stress and apoptosis via activating the Nrf-2/NQO-1 and NF-κB signaling pathway after spinal cord injury in rats. Am. J. Transl. Res. 11(10), 6544–6552. Liu, W., Liu, H., Zhang, S., Hao, H., Meng, F., Ma, W., Guo, Z., Jiang, S. and Shang, X. 2024. Silica nanoparticles cause ovarian dysfunction and fertility decrease in mice via oxidative stress-activated autophagy and apoptosis. Ecotoxicol. Environ. Saf. 285, 117049; doi:10.1016/j.ecoenv.2024.117049 Madkour, L.H. 2023. Reactive oxygen species (ROS), nanoparticles, and endoplasmic reticulum (ER) stress-induced cell death mechanisms. Cambridge, UK: Academic Press; doi: 10.1016/C2019-0-04102-7. Malik, S., Muhammad, K. and Waheed, Y. 2023. Nanotechnology: a revolution in modern industry. Molecules 28(2), 661; doi:10.3390/molecules28020661 Nie, X., Dong, X., Hu, Y., Xu, F., Hu, C. and Shu, C. 2023. Coenzyme Q10 stimulate reproductive vitality. Drug Des. Devel. Ther. 17, 2623–2637; doi:10.2147/DDDT.S414731 Nolfi-Donegan, D., Braganza, A. and Shiva, S. 2020. Mitochondrial electron transport chain: oxidative phosphorylation, oxidant production, and methods of measurement. Redox Biol. 37, 101674; doi:10.1016/j.redox.2020.101674 Novo, N., Ferreira, P. and Medina, M. 2021. The apoptosis-inducing factor family: moonlighting proteins in the crosstalk between mitochondria and nuclei. IUBMB. Life 73(3), 568–581; doi:10.1002/iub.2452 Al Nuaimmi, S.M. and Rasheed, S.A. 2023. The effect of dry yeast and folic acid treatment on the reproductive and physiological aspects of Quail stressed with Hydrogen Peroxide. Veterinarija Ir Zootechnika 81(2), i1–i7; Obokhwo, O.M., Ben-Azu, B., Nwangwa, E.K., Ohwin, E.P., Igweh, J.C. and Adeogun Adetomiwa, E. 2024. Adverse hematological profiles associated with chlorpromazine antipsychotic treatment in male rats: preventive and reversal mechanisms of taurine and coenzyme-Q10. Toxicol. Rep. 12, 448–462; doi:10.1016/j.toxrep.2024.03.008 Özgür, M.E., Ulu, A., Özcan, I., Balcioglu, S., Ateş, B. and Köytepe, S. 2019. Investigation of toxic effects of amorphous SiO2 nanoparticles on motility and oxidative stress markers in rainbow trout sperm cells. Environ. Sci. Pollut. Res. 26, 15641–15652; doi:10.1007/s11356-019-04980-y Paunović, M.G., Matić, M.M., Ognjanović, B.I. and Saičić, Z.S. 2017. Antioxidative and haematoprotective activity of coenzyme Q10 and vitamin E against cadmium-induced oxidative stress in Wistar rats. Toxicol. Ind. Health. 33(10), 746–756; doi:10.1177/0748233717696935 Podar, A.S., Semeniuc, C.A., Ionescu, S.R., Socaciu, M.I., Fogarasi, M., Fărcaș, A.C., Vodnar, D.C. and Socaci, S.A. 2023. An overview of analytical methods for quantitative determination of coenzyme Q10 in foods. Metabolites 13(2), 272; doi:10.3390/metabo13020272 Qiu, S., Zhu, F. and Tong, L. 2024. Application of targeted drug delivery by cell membrane-based biomimetic nanoparticles for inflammatory diseases and cancers. Eur. J. Med. Res. 29(1), 523; doi:10.1186/s40001-024-02020-7 Rasheed, S.A. and Al Nuaimmi, S.M.A. 2022. Effect of adding dry yeast and folic acid on improving the physiological and productive performance of Quail. Iraqi J. Agricult. Sci. 53(4), 789–797; doi:10.36103/ijas.v53i4.1590 Rasheed, S.A., Asim, R.A. and Jasim, H.M. 2022. The moderating effect of Panax ginseng roots on the male reproductive system and heat shock protein 70 in heat-stressed Japanese quails. Iraqi J. Vet. Sci. 36(4), 853–859; doi:10.33899/IJVS.2022.132356.2084 Rashidian, G., Mohammadi-Aloucheh, R., Hosseinzadeh-Otaghvari, F., Chupani, L., Stejskal, V., Samadikhah, H., Zamanlui, S., Multisanti, C.R. and Faggio, C. 2023. Long-term exposure to small-sized silica nanoparticles (SiO2-NPs) induces oxidative stress and impairs reproductive performance in adult zebrafish (Danio rerio). Comp. Biochem. Physiol. C. Toxicol. Pharmacol. 273, 109715; doi:10.1016/j.cbpc.2023.109715 Salvio, G., Cutini, M., Ciarloni, A., Giovannini, L., Perrone, M. and Balercia, G. 2021. Coenzyme Q10 and male infertility: a systematic review. Antioxidants 10(6), 874; doi:10.3390/antiox10060874 Sekhon, B. 2014. Nanotechnology in agri-food production: an overview. Nanotechnol. Sci. Appl. 7, 31–53; doi:10.2147/NSA.S39406 Sergent, J.A., Paget, V. and Chevillard, S. 2012. Toxicity and genotoxicity of nano-SiO2 on human epithelial intestinal HT-29 cell line. Ann. Occup. Hyg. 56(5), 622–630; doi:10.1093/annhyg/mes005 Sharma, V., Singh, P., Pandey, A.K. and Dhawan, A. 2012. Induction of oxidative stress, DNA damage and apoptosis in mouse liver after sub-acute oral exposure to zinc oxide nanoparticles. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 745(1-2), 84–91; doi:10.1016/j.mrgentox.2011.12.008 Shi, Y.Q., Zhu, X.T., Zhang, S.N., Ma, Y.F., Han, Y.H., Jiang, Y. and Zhang, Y.H. 2023. Premature ovarian insufficiency: a review on the role of oxidative stress and the application of antioxidants. Front. Endocrinol. 14, 1172481; doi:10.3389/fendo.2023.1172481 Song, X., Jiang, N., Li, Y., Xu, D. and Qiu, G. 2008. Synthesis of CeO2-coated SiO2 nanoparticle and dispersion stability of its suspension. Mater. Chem. Phys. 110(1), 128–135; doi:10.1016/j.matchemphys.2008.01.042 Van Tran, L., Malla, B.A., Kumar, S. and Tyagi, A.K. 2016. Polyunsaturated fatty acids in male ruminant reproduction—a review. Asian-Australasian J. Anim. Sci. 30(5), 622; doi:10.5713/ajas.15.1034 Yusuf, A., Almotairy, A.R.Z., Henidi, H., Alshehri, O.Y. and Aldughaim, M.S. 2023. Nanoparticles as drug delivery systems: a review of the implication of nanoparticles’ physicochemical properties on responses in biological systems. Polymers 15(7), 1596; doi:10.3390/polym15071596 Zaiter, T. 2022. In vitro evaluation of the toxicological profile of silica nanoparticles and nanoemulsions.. Université Bourgogne Franche-Comté. Available via https://theses.hal.science/tel-04112071/ Zong, Y., Li, H., Liao, P., Chen, L., Pan, Y., Zheng, Y., Zhang, C., Liu, D., Zheng, M. and Gao, J. 2024. Mitochondrial dysfunction: mechanisms and advances in therapy. Signal Transduct. Target. Ther. 9(1), 124; doi: 10.1038/s41392-024-01839-8. | ||
| How to Cite this Article |
| Pubmed Style Al-maatheedi MS, Rasheed SA, Asim RA. Role of coenzyme Q10 on some physiological aspects of male rat exposure to silicon dioxide nanoparticles. doi:10.5455/OVJ.2026.v16.i1.46 Web Style Al-maatheedi MS, Rasheed SA, Asim RA. Role of coenzyme Q10 on some physiological aspects of male rat exposure to silicon dioxide nanoparticles. https://www.openveterinaryjournal.com/?mno=282327 [Access: February 01, 2026]. doi:10.5455/OVJ.2026.v16.i1.46 AMA (American Medical Association) Style Al-maatheedi MS, Rasheed SA, Asim RA. Role of coenzyme Q10 on some physiological aspects of male rat exposure to silicon dioxide nanoparticles. doi:10.5455/OVJ.2026.v16.i1.46 Vancouver/ICMJE Style Al-maatheedi MS, Rasheed SA, Asim RA. Role of coenzyme Q10 on some physiological aspects of male rat exposure to silicon dioxide nanoparticles. doi:10.5455/OVJ.2026.v16.i1.46 Harvard Style Al-maatheedi, M. S., Rasheed, . S. A. & Asim, . R. A. (2026) Role of coenzyme Q10 on some physiological aspects of male rat exposure to silicon dioxide nanoparticles. doi:10.5455/OVJ.2026.v16.i1.46 Turabian Style Al-maatheedi, Mahmood S., Suha A. Rasheed, and Rana A. Asim. 2026. Role of coenzyme Q10 on some physiological aspects of male rat exposure to silicon dioxide nanoparticles. doi:10.5455/OVJ.2026.v16.i1.46 Chicago Style Al-maatheedi, Mahmood S., Suha A. Rasheed, and Rana A. Asim. "Role of coenzyme Q10 on some physiological aspects of male rat exposure to silicon dioxide nanoparticles." doi:10.5455/OVJ.2026.v16.i1.46 MLA (The Modern Language Association) Style Al-maatheedi, Mahmood S., Suha A. Rasheed, and Rana A. Asim. "Role of coenzyme Q10 on some physiological aspects of male rat exposure to silicon dioxide nanoparticles." doi:10.5455/OVJ.2026.v16.i1.46 APA (American Psychological Association) Style Al-maatheedi, M. S., Rasheed, . S. A. & Asim, . R. A. (2026) Role of coenzyme Q10 on some physiological aspects of male rat exposure to silicon dioxide nanoparticles. doi:10.5455/OVJ.2026.v16.i1.46 |