| Research Article | ||
Open Vet. J.. 2026; 16(2): 1171-1177 Open Veterinary Journal, (2026), Vol. 16(2): 1171-1177 Research Article Antibodies in plasma and blood cells of horsesGeorgiy Demchenko1,2, Maxat Berdikulov1, Abylai Sansyzbay1, Serik Abdreshov1,2, Laura Koibasova2*, Damir Khusainov1 and Zhaniha Lessova11LLP, Scientific Production and Technical Center, Almaty, Republic of Kazakhstan 2Institute of Genetics and Physiology, Almaty, Republic of Kazakhstan *Corresponding Author: Laura Koibasova. Institute of Genetics and Physiology, Almaty, Republic of Kazakhstan. Email: laura.koibasova74 [at] mail.ru Submitted: 10/09/2025 Revised: 29/12/2025 Accepted: 16/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
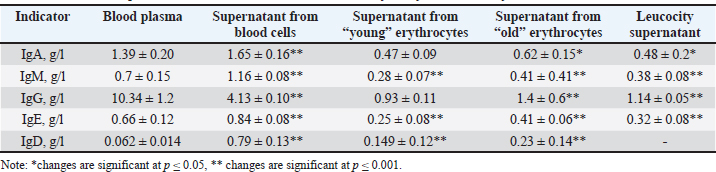
ABSTRACTBackground: Understanding the distribution of antibodies in the bloodstream is critical for advancing diagnostic and therapeutic approaches in veterinary medicine and comparative immunology. While most studies have focused on plasma antibodies, the presence and role of membrane-bound immunoglobulins remain underexplored, particularly in equine species such as the Jabe horse. Aim: This study aimed to analyze the amount of antibodies in the plasma and blood cells of Jabe horses for their potential future use. Methods: Blood samples were collected from Jabe horses and separated into plasma and cellular components. A specific technique was developed to isolate immunoglobulins from both plasma and blood cell membranes. The concentrations of different immunoglobulin classes [IgA , IgM, IgE, IgG, and Immunoglobulin D (IgD)] were measured using ELISA. Aged erythrocytes were given special attention to examine antibody accumulation over time. Results: The antibody concentration in plasma was generally higher than that on blood cell membranes. However, the membrane-bound fractions of IgA, IgM, and IgE ranged from 106% to 139.7%, indicating a significant membrane association. IgG levels in the supernatant were >60% lower than those in the plasma, suggesting selective membrane binding or sequestration. Notably, IgD was predominantly membrane-bound, with concentrations many times higher than those in the supernatant. A marked increase in membrane-bound immunoglobulins was observed in aged erythrocytes, highlighting a possible age-dependent accumulation. Furthermore, the content of other membrane-associated proteins correlated with their plasma concentrations. Conclusion: The study reveals that immunoglobulins are not only present in plasma but are also significantly associated with blood cell membranes in Jabe horses. The high levels of membrane-bound IgD and the accumulation of antibodies on aged erythrocytes suggest that functional or regulatory roles are yet to be fully understood. The newly proposed method for antibody isolation from both plasma and cellular fractions opens opportunities for further immunological research and potential clinical applications in veterinary medicine. Keywords: Blood components, Erythrocytes, Immunoassay, Immunoglobulins, Membrane transport. IntroductionAntibodies are glycoproteins produced by plasma cells. B cells differentiate into plasma cells upon stimulation by specific immunogens such as bacterial proteins (Jackson and Elsawa, 2015). These plasma cells are responsible for producing antibodies involved in humoral immune responses against bacteria, viruses, fungi, parasites, cellular antigens, chemicals, and synthetic substances (Elkuch et al., 2017; Jackson et al., 2017). Immunoglobulins constitute approximately 20% of total plasma proteins (Patel et al., 2023). The membranes of red and white blood cells, like those of other cells, serve as selective barriers and participate in both active and passive transport processes. They also play critical roles in biochemical interactions and immune responses. The red blood cell (RBC) membrane carries a surface electric charge, which is essential for sustaining cell viability in various physiological processes. In addition, the RBC membrane contains numerous signaling molecules, suggesting a role in regulatory mechanisms that contribute to the organism’s integrative functioning (Olumuyiwa-Akeredolu et al., 2017; Pretini et al, 2019). Advances in pharmacology and medicine have enabled the extraction and therapeutic use of valuable blood-derived products, such as albumin, creatinine, fibrinogen, thrombin, and gamma globulin. These components serve as immunological, hemostatic, nutritional, or blood-substitute agents. Blood-derived substances are also incorporated into culture media for microbiological studies (Bonnet et al., 2019; Bihari et al., 2020; Mathew et al., 2023). As part of the body’s physiological systems, blood indicators reflect the integrated functioning of the whole organism (Oore-ofe et al., 2017; Bonnet et al., 2019; Barajas-Martínez et al., 2021). Their roles are diverse and can be used to assess the adaptive responses of organisms to environmental conditions (Smagulova et al., 2007, 2010; Gareev, 2015). In earlier studies, we demonstrated that the transfer of substances to the surface of erythrocytes is an important component of blood–tissue–lymph exchange. The content of substances adsorbed on erythrocyte membranes changes most rapidly, followed by changes in plasma levels, with lymph levels changing most slowly (Mohandas and Gallagher 2008; Breslin et al., 2018; Zhang et al., 2024). However, many details of the substance transfer mechanisms involving erythrocytes remain poorly understood. The relationship between the adsorption–transport function of erythrocytes and the age of the erythrocytes has not been sufficiently explored. The primary role of this function is to adsorb essential substances onto erythrocyte surfaces and transfer them to the capillary exchange layer (Kuhn et al., 2017; Glassman et al., 2020). Regulatory molecules and other vital compounds adsorbed on erythrocytes are among the first to reach tissues, where they influence metabolism and physiological responses (Prechl et al., 2022). Our previous research revealed a significant dependence between the adsorption of proteins and other plasma constituents onto blood cell membranes and their plasma concentrations. This phenomenon was observed in samples with a normal age distribution of circulating erythrocytes. However, this relationship becomes less pronounced as the population of older erythrocytes increases. This observation is of great importance because it suggests age-related changes in plasma composition and its interaction with cell membranes (Florent et al., 2024). Therefore, studying the adsorption and transport functions of blood cells in farm animals is of both scientific and practical significance. This knowledge may be valuable in veterinary medicine, animal breeding, nutrition, and the development of new treatment strategies for diseases linked to blood composition alterations. In this context, our study aimed to investigate the presence and quantity of antibodies on the surface of erythrocytes and leukocytes. Special attention is given to their chemical nature and biological role. The results may contribute to the development of new biologically active compounds aimed at improving animal health and productivity, ultimately leading to better disease prevention and enhanced animal-derived product quality. Aim of the studyTo evaluate the quantity of antibodies in the plasma and blood cells of horses, with a view to their potential further application. Materials and MethodsThe study was conducted on 20 Jabe mares weighing 400 ± 20 kg and aged 2–5 years. Experiments were carried out on relatively healthy animals during the summer. The study material was the blood of horses. Blood for the study was taken from the jugular vein of an animal in an amount of 0.2–0.25 free-range liters in May–July. After collection, the blood was stabilized with heparin. After centrifugation (15 minutes at 1,500 rpm), the blood cells were separated from the plasma. The cells were separated by sequential selection of the upper and lower parts of the cell column. The upper layer of the column contains leukocytes and lymphocytes, and the erythrocytes were separated into fractions “young” (YE) and the lowest sediment layer “old” (SE) according to the method of Rockstroh et al. (2024). Antibodies from young and old erythrocytes and leukocytes were separately washed off once by adding and mixing the erythrocyte or leukocyte mass with a 3% sodium chloride solution and shaking for 30 minutes. The resulting suspensions were separately centrifuged again for 20 minutes at 3,000, and the supernatant (wash) was separated. The method for obtaining washes from erythrocytes was developed at our institute (methodological recommendations, patent of the Republic of Kazakhstan No. 16019, No. 16018) (Gareev, 2015). The antibody content was determined in the erythrocyte and leukocyte washes and in blood plasma. An enzyme immunoassay was performed. The concentration of antibodies (immunoglobulins A, M, G, E, and D) in plasma, blood, and washes from erythrocytes and leukocytes was determined using ELISA-BEST kits on a Roche Cobas e 411 Immunochemical Analyzer. The total protein and albumin contents in the blood plasma and supernatant were determined by conventional methods using a Cobas Integra 400 automatic biochemical analyzer (Roche Diagnostics). In some samples for subsequent use of plasma and supernatant for practical purposes, defibrination was performed. This is the process of removing fibrin from the blood resulting from blood clotting. Blood was defibrinated in a stainless-steel vessel in a mechanical paddle stirrer, the blood was stirred for 8–10 minutes, and then passed through a metal mesh filter. Statistical analysisThe results were statistically processed using Microsoft Excel, and the parameters were changed by considering the independent samples t-test (Student t-test) criterion. The results were considered reliable at p ≤ 0.05. Ethical approvalWe were guided by the decision of the Local Ethics Commission, an extract from the minutes of the Local Ethics Commission at the RSE “Institute of Genetics and Physiology” of the SC MSHE RK No. 2 dated 05. 23. 2024, outgoing No. 07-1.80. ResultsThis study examined the levels of total protein and its two main fractions—albumin and globulin (antibodies)—in blood plasma and in the supernatant obtained from erythrocytes and leukocytes, both “young” and “old”. The total protein concentration in plasma was 66.94 ± 1.23 g/l, whereas that in the supernatant was 31.12 ± 0.65 g/l. The albumin levels were 27.83 ± 1.8 g/l in the plasma and 16.4 ± 0.39 g/l in the supernatant. The total protein content in the supernatants from “young” and “old” erythrocytes was lower by 52% and 56%, respectively, compared to plasma. Albumin concentrations in washes from “young” and “old” erythrocytes were 16.2 and 17.4 g/l, respectively, representing a decrease of 41.34% and 41%, respectively, compared to plasma. Albumin levels in leukocyte supernatants were 40.7% lower than in plasma. Overall, both total protein and albumin concentrations in supernatants were higher in older blood cell fractions, indicating greater protein adsorption on aged erythrocyte and leukocyte membranes. Next, we analyzed the levels of immunoglobulins IgA, IgM, IgG, IgE, and Immunoglobulin D (IgD) in plasma and in the supernatants (washes) from erythrocyte and leukocyte membranes. Tables 1 and 2 present the data. Table 1 shows the statistically significant differences in immunoglobulin concentrations between the plasma and erythrocyte membranes. For example, the level of IgA in plasma was 19% lower than that on blood cell membranes, with 82% of the plasma level detected on erythrocyte membranes and 36% on leukocyte membranes. A similar trend was observed for IgM, where membrane-bound levels were 39.7% higher than in plasma (96.6% on erythrocyte membranes and 54.3% on leukocyte membranes) (Table 2). IgE was found in higher concentrations on the surface of blood cells than in plasma, i.e., 21.2% more in total. Specifically, erythrocyte-bound IgE matched plasma levels, whereas 48.5% of plasma IgE was present on leukocyte membranes. Our results indicate that most immunoglobulins, except IgG, are preferentially associated with blood cell membranes. Plasma IgG levels were 60% higher than those on cell membranes. On erythrocyte membranes, IgG was present at only 22.5% of its plasma concentration, and on leukocyte membranes, it was present at only 11%. This study examined the amount of total protein and its two fractions, albumin and globulin antibodies, in blood plasma and in the supernatant from erythrocytes and leukocytes from young and old cells. The study showed that the total protein content in plasma was 66.94 ± 1.23, in the supernatant 31.12 ± 0.65 g/l, albumin, and in plasma 27.83 ± 1.8, in the supernatant 16.4 ± 0.39 g /l. In the supernatant, the total protein from “young” and “old” erythrocytes compared to plasma was lower by 52% and 56%, respectively. The data reflecting the changes in the albumin concentration in the washes from the “young” 16.2 g/l and “old” 17.4 g/l erythrocytes were similarly lower by 41.34% and 41%, respectively, and those from leukocytes were lower by 40.7%. Total protein and albumin contents in the supernatant from the “young” and “old” erythrocytes and leukocytes were higher in the old blood cells. Table 1. Antibodies of plasma and contained on the membranes of erythrocytes and leukocytes in a horse of the Jabe breed.
Table 2. Immunoglobulin D levels in blood plasma and blood cell washes of animals with and without fibrinogen.
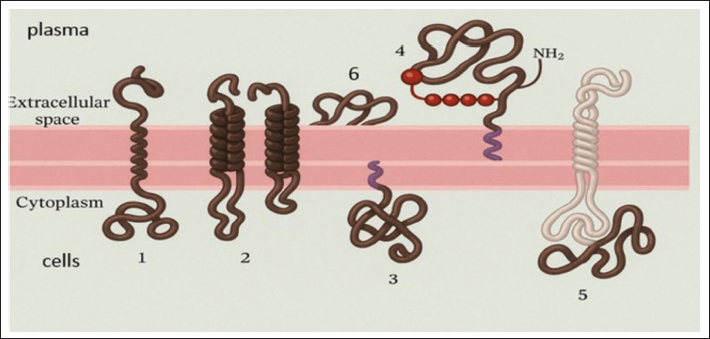
Furthermore, we determined the levels of immunoglobulins IgA, IgM, IgG, IgE, and IgD in blood plasma and supernatants from erythrocytes and leukocytes. The data are presented in Tables 1 and 2. Table 1 shows statistically significant differences between the immunoglobulin indicators in the plasma blood of animals and in the supernatant from erythrocyte membranes. Thus, the level of immunoglobulin A in plasma is 19% lower than that on blood cell membranes, and corresponds to 82 on the erythrocyte membrane and 36% on the leukocyte membrane. Immunoglobulin M showed a similar picture, with 39.7% higher on the blood cell membrane, 96.6% on the erythrocyte membrane, and 54.3% on the leukocyte membrane (Table 2). As can be seen, IgE in the blood is transferred to the surface of blood cells in an amount 21.2% higher than in plasma, and on erythrocytes in the same amount as plasma, and on leukocytes, 48.5% of plasma. We found that the majority (except for IgG) of immunoglobulins are transferred mainly to the surface of blood cells. There is 60% more immunoglobulin G in the plasma than in the cells. On the membrane of erythrocytes, it circulates in 22.5% of plasma and 11% of leukocytes. The level of IgG in washes from erythrocyte membranes was 45% lower than its concentration in plasma. Immunoglobulins of classes A, M, and E were predominantly found in membrane-bound forms, whereas IgG was mainly present in plasma in soluble forms. This study also examined IgD levels in horse blood plasma and in supernatants, both in the presence of fibrinogen and after its removal (defibrination). The concentration of IgD in plasma was very low, whereas its level in the supernatant was significantly elevated (Table 1). On blood cell membranes, IgD showed multiple-fold increases, with 91.1% of total membrane-bound IgD localized on erythrocytes. When fibrinogen was removed, an increase in IgD was detected. Statistically significant, but in our opinion, insignificant differences were revealed when comparing the washes in the presence of fibrinogen and with its removal, although the IgD concentration was higher without fibrinogen (Table 2). The mixing of supernatant substances from the blood cells of various animals did not result in the formation of protein-lipid aggregates. This creates the basis for creating therapeutic drugs from animal immunity substances, including horses. DiscussionThe function of erythrocytes extends beyond the classical role of oxygen and carbon dioxide transport. Erythrocytes and leukocytes are capable of attracting, adsorbing, and transporting a wide array of biologically active molecules through the bloodstream due to the electrochemical and physicochemical properties of their plasma membranes (Glassman et al., 2020; Prechl et al., 2022; Rockstroh et al., 2024). These properties suggest that these cells play a far more dynamic role in maintaining physiological homeostasis than previously thought. Our findings demonstrate that various proteins are carried on the membranes of horse erythrocytes and leukocytes; however, their plasma concentrations remain significantly higher, by a factor of 2.2 for total protein and 1.7 for albumin. These data indicate that the protein-transporting function of blood cells is primarily realized on a microcirculatory level, particularly within capillary networks, where localized regulation of substance delivery is critical for tissue function. Serum proteins play a key role in maintaining the homeostasis of the internal environment. Circulating (plasma) and membrane-bound proteins function in dynamic equilibrium with interstitial and tissue proteins and serve as important regulators of protective, immune, and transport mechanisms, thereby contributing to systemic metabolic stability. Consequently, quantitative and qualitative assessments of these proteins, particularly those associated with blood cells, may serve as valuable biomarkers for evaluating an animal’s physiological condition, immune status, and disease risk (Belinskaia et al., 2021). The functional integrity of erythrocytes depends heavily on the biochemical composition of blood plasma because erythrocytes are terminally differentiated cells and lack the organelles necessary for protein synthesis and self-renewal. Thus, they rely on extracellular sources for the maintenance of membrane structure and function (Chatzinikolaou et al., 2024). This dependence makes erythrocytes sensitive indicators of systemic changes in the body’s internal environment. The immune system plays a pivotal role in preserving the antigenic and biochemical integrity of the internal milieu (Doltchinkova, et al., 2023; Bulatov et al., 2025). Our findings support the growing body of evidence suggesting that erythrocytes contribute to immune regulation indirectly and through direct interaction via surface-associated immunoglobulins. These immunoglobulins, anchored to erythrocyte membranes, are believed to participate in maintaining immune homeostasis, possibly by modulating local immune responses and contributing to immune surveillance. Membrane-associated proteins differ in their modes of attachment. Some peripheral membrane proteins are linked to the membrane via noncovalent interactions with other proteins and can be displaced by pH or ionic strength changes. Others, categorized as integral membrane proteins, penetrate the lipid bilayer or are anchored through lipid moieties such as phosphatidylinositol or fatty acid chains (Boes et al., 2021). These proteins require harsher conditions, such as detergent treatment, to be detached (Bonnet et al., 2019) (Fig. 1). This distinction is critical for understanding the biochemical behavior and functional specialization of MBIs in blood cells. Our results reveal that immunoglobulins IgA, IgM, IgE, and particularly IgD are found not only in plasma but also in significant concentrations on the membranes of erythrocytes and leukocytes. These findings support the hypothesis that certain immunoglobulins primarily exist in membrane-bound forms. The only exception is IgG, whose levels are considerably higher in plasma than on cell membranes, suggesting a different mode of immune function, likely associated with systemic rather than cell-surface immunity. This disparity highlights a potential specialization among equine immunoglobulin classes. Immunoglobulin D is a relatively recent discovery in comparative immunology. Our results show that IgD is present in minimal amounts in plasma but is markedly concentrated on the membranes of RBCs and White blood cells. This suggests that IgD functions predominantly in a membrane-bound state and may play a crucial role in both innate and adaptive immune responses (Pretini et al., 2019; Carpenter et al., 2024). Elevated IgD levels in erythrocyte and leukocyte washes reinforce the theory that IgD serves as a membrane-anchored immunoregulatory factor in horses. These findings open new perspectives in veterinary immunology. The data suggest that IgD may be a key component of the innate immune response in horses and that decreased surface expression or functional deficiency of this immunoglobulin could contribute to chronic inflammatory states or increased susceptibility to neoplastic processes. Further research into the structural and functional role of IgD could pave the way for the development of new diagnostic markers and therapeutic interventions in equine medicine (Chen et al., 2010). Considering the obtained results, we propose that the continuous mechanical and biochemical exposure experienced by circulating erythrocytes and leukocytes leads to progressive membrane remodeling. As demonstrated in previous studies and supported by our data, these changes result in dynamic shifts in their adsorptive and transport capabilities (Gutzeit et al., 2018; Demchenko et al., 2023, 2024; Thiagarajan et al., 2021). Such remodeling may have broader implications for the lifespan, immune function, and blood cell signaling capacity. Moreover, it has been demonstrated that under various physiological or pathological conditions—such as stress, inflammation, or aging—the ability of blood cells to adsorb and transport key substances changes significantly. This supports the idea that blood cell membranes serve not only as passive carriers but also as active modulators of systemic adaptation processes (Richardson et al., 2020; Thiagarajan et al., 2021). Therefore, a comparative analysis of the transport and binding capacities of erythrocytes and leukocytes under different conditions may provide crucial insights into the development of novel biologically active compounds, veterinary diagnostics, and therapeutics. Ultimately, these findings underscore the importance of studying blood cell membrane biochemistry not only for understanding physiological regulation but also for potential translational applications. We believe our results may serve as a scientific foundation for further investigations aimed at enhancing disease diagnosis, improving immune monitoring, and developing innovative strategies for animal health management and pharmaceutical production.
Fig. 1. Association of membrane proteins with the lipid bilayer and membrane proteins on erythrocytes. Transmembrane proteins penetrate the bilayer in the form of a single β-helix (1) or several β-helices (2). Proteins (1 and 2) have a covalently attached fatty acid chain immersed in the cytoplasmic monolayer (1). Membrane proteins associate with the bilayer only due to a covalently attached lipid or a fatty acid chain immersed in the cytoplasmic monolayer (3), or, much less often, through the phospholipid phosphatidylinositol immersed in the outer monolayer and connected to the protein through an oligosaccharide (4). Proteins associated with the membrane only due to non-covalent interactions with other membrane proteins (5). Membrane proteins held by charge difference (6). ConclusionThis study identified the quantitative of total protein and albumin circulation on the surfaces of erythrocytes and leukocytes in Jabe breed horses. The concentrations of these proteins in the blood plasma significantly exceeded their levels on the cellular membranes. In contrast, IgA, IgM, IgE, and IgD were predominantly associated with blood cell membranes. Among them, IgD exhibited the greatest disparity between plasma and membrane-bound forms, suggesting a primarily membrane-bound role. IgG was the only immunoglobulin to demonstrate an inverse distribution, with significantly higher concentrations in plasma than on membranes. Additionally, the study revealed an increascharacteristicse in membrane-associated total protein, albumin, and immunoglobulin content on aged erythrocytes, indicating a possible age-related enhancement of their adsorptive properties. A novel method for the isolation of immunoglobulins from the blood of farm animals is currently under development. This technology holds promise for advancing biomedical drug production, potentially leading to innovative applications in veterinary and human medicine. AcknowledgmentsNone. Conflicts of interest The authors of this study have no conflicts of interest to declare. Funding This research was funded by the Committee of Science of the Ministry of Science and Higher Education of the Republic of Kazakhstan (Grant No. BR24993032). Authors’ contributions Conceptualization: G.D and M.B.; methodology: A.S, S.A., J.G. P.; validation: L.K., D.Kh.; formal analysis: D.Kh. and Zh. L.; investigation: S.A., D.Kh, and Zh. L.; resources: G.D., M.B.; data curation: L.K. and Zh. L.; writing—original draft preparation: G.D., M.B.; writing—review and editing: G.D., M.B., A.S., S.A.; visualization: S.A., D.Kh. All authors have read and agreed to the published version of the manuscript. Data availability All relevant data are included in the manuscript and its supplementary materials. Additional information is available from the authors upon request, if needed. ReferencesDemchenko, G A., Abdreshov S.N., Koibasova, L U., Yessenova, M A. and Yeshmukhanbet, A N. 2024. The impact of hypoxic training at different altitudes on human physiology, biochemistry, and cytogenetics. Edelweiss Appl. Sci. Technol. 8(4), 1119–1130; doi: 10.55214/25768484.v8i4.1486<AQ5> Barajas-Martínez, A., Ibarra-Coronado, E., Sierra-Vargas, M.P., Cruz-Bautista, I., Almeda-Valdes, P., Aguilar-Salinas, C.A., Fossion, R., Stephens, C.R., Vargas-Domínguez, C., Atzatzi-Aguilar, O.G., Debray-García, Y., García-Torrentera, R., Bobadilla, K., Naranjo Meneses, M.A., Mena Orozco, D.A., Lam-Chung, C.E., Martínez Garcés, V., Lecona, O.A., Marín-García, A.O., Frank, A. and Rivera, A.L. 2021. Physiological network from anthropometric and blood test biomarkers. Front. Physiol. 11, 612598; doi.org/10.3389/fphys.2020.612598 Belinskaia., DA., Voronina., PA., Goncharov. and NV. 2021. Integrative role of albumin: evolutionary, biochemical and pathophysiological aspects. J. Evol. Biochem. Physiol. 57(6), 1419–1448. Bihari, S., Bannard-Smith, J. and Bellomo, R. 2020. Albumin as a drug: its biological effects beyond volume expansion. Crit. Care Resusc. 22(3), 257–265. Boes, D.M., Godoy-Hernandez, A. and McMillan, D.G.G. 2021. Peripheral Membrane Proteins: promising Therapeutic Targets across Domains of Life. Membranes (Basel). 8;11(5), 346. Bonnet, M., Lagier, J.C., Raoult, D. and Khelaifia, S. 2019. Bacterial culture through selective and non-selective conditions: the evolution of culture media in clinical microbiology. New Microbes New Infect. 34, 100622; doi:10.1016/j.nmni.2019.100622 Breslin, J.W., Yang, Y., Scallan, J.P., Sweat, R.S., Adderley, S.P. and Murfee, W.L. 2018. Lymphatic Vessel Network Structure and Physiology. Compr. Physiol. 9(1), 7–299. Bulatov, Y., Kurmasheva, A., Amanova, Z., Abitaev, R., Sametova, Z., Kyrgyzbayeva, A., Kondybaeva, Z., Turyskeldi, S., Ussembay, A., Toktyrova, D., Mazbayeva, D., Shayakhmetov, Y., Kerimbayev, A., Khussainov, D., Wentao, M., Rsaliyev, A. and Abduraimov, Y. 2025. The Influence of the Associated Inactivated Vaccine Against Infectious Rhinotracheitis and Bovine Viral Diarrhea on the Formation and Duration of Colostral Immunity in Kazakh Whiteheaded Calves. Vaccines 13(4), 408. Carpenter, S. and O’Neill, L.A.J. 2024. From periphery to centerstage: 50 years of advancements in innate immunity. Cell 187(9), 2030–2051. Chatzinikolaou, P.N., Margaritelis, N.V. and Paschalis, V. 2024. Erythrocyte metabolism. Acta Physiol. 240(3), 1–49; doi:10.1111/apha.14081 Chen, K. and Cerutti, A. 2010. New insights into the enigma of immunoglobulin D. Immunol. Rev. 237(1), 160–179; doi:10.1111/j.1600-065X.2010.00929.x Demchenko, G.A., Nurmakhanova, B.A., Abdreshov, S.N. and Koibasova, L.U. 2023. Phytocorrection of Age-Related Changes in the Composition of Blood Plasma, Lymph, and Interstitial Fluid. Bull. Exp. Biol. Med. 175, 785–790; doi:10.1007/s10517-023-05947-3 Doltchinkova, V., Kitanova, M., Nikolov, R., Stoyanova-Ivanova, A., Petkov, O., Dikova, Y. and Vitkova, V. 2023. Erythrocyte Membrane Biophysical Changes Mediated by Pooled Immunoglobulin G and Hematin: electrokinetic and Lipid Peroxidation Studies. Membranes 13(3), 281; doi:10.3390/membranes13030281 Elkuch, M., Greiff, V., Berger, C.T., Bouchenaki, M., Daikeler, T., Bircher, A., Navarini, A.A., Heijnen, I. and Recher, M. 2017. Low immunoglobulin E flags two distinct types of immune dysregulation. Clin. Exp. Immunol. 187(3), 345–352. Florent S., Schvartz, M., Renault, J., Vieira, J., Devineau, S., Leroy, J., Taché, O., Boulard, Y. and Pin, S., 2024. The impact of virgin and aged microstructured plastics on proteins: the case of hemoglobin adsorption and oxygenation. Int. J. Mol. Sci. 25(13), 7047; doi.org/10.3390/ijms25137047 Gareev, R.A., 2015. Method for determining endogenous and exogenous blood markers Patent number 30294 IPC: G01N 33/50, G01N 33/48. Available at: https://kz.patents.su/4-30294-sposob-opredeleniya endogennyh-i-ekzogennyh-markerov-krovi.html. Glassman, P.M., Villa, C.H., Ukidve, A., Zhao, Z., Smith, P., Mitragotri, S., Russell, A.J., Brenner, J.S. and Muzykantov, V.R. 2020. Vascular Drug Delivery Using Carrier Red Blood Cells: focus on RBC Surface Loading and Pharmacokinetics. Pharmaceutics 12, 440; doi:10.3390/pharmaceutics12050440 Gutzeit, C., Chen, K. and Cerutti, A. 2018. The enigmatic function of IgD: some answers at last. Eur. J. Immunol. 248(7), 1101–1113. Jackson, D. and Elsawa, S. 2015. Factors Regulating Immunoglobulin Production by Normal and Disease-Associated Plasma Cells. Biomolecules 5(1), 20–40; doi:10.3390/biom5010020 Kuhn, V., Diederich, L., Keller, T.C.S., Kramer, C.M., Lückstädt, W., Panknin, C., Suvorava, T., Isakson, B.E., Kelm, M. and Cortese-Krott, M.M. 2017. Red Blood Cell Function and Dysfunction: redox Regulation, Nitric Oxide Metabolism, Anemia. Antioxid. Redox. Signal. 26(13), 718–742. Mathew, J., Sankar, P., Varacallo, M, A., 2023. Physiology, Blood Plasma. [Updated 2023 Apr 24]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK531504 Mohandas, N. and Gallagher, P.G. 2008. Red cell membrane: past, present, and future. BLOOD 112(10), 3939–3948; doi:10.1182/blood-2008-07-161166 Olumuyiwa-Akeredolu, O.O.O., Soma, P., Buys, A.V., Debusho, L.K. and Pretorius, E. 2017. Characterizing pathology in erythrocytes using morphological and biophysical membrane properties: relation to impaired hemorheology and cardiovascular function in rheumatoid arthritis. Biochimica Et Biophysica Acta (BBA). –. Biomembranes 1859(12), 2381–2391; doi:10.1016/j.bbamem.2017.09.014 Olumuyiwa-Akeredolu, O.O., Soma, P., Buys, A.V., Debusho, L.K. and Pretorius, E. 2017. Characterizing pathology in erythrocytes using morphological and biophysical membrane properties: relation to impaired hemorheology and cardiovascular function in rheumatoid arthritis, Biochim. Biophys. Acta Biomembr. 1859 (12), 2381–2391; doi: 10.1016/j.bbamem.2017.09.014 Patel, P, Jamal, Z, Ramphul, K. Immunoglobulin., 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK513460/ Prechl, J., Papp, K., Kovács. and Pfeil, T. 2022. The Binding Landscape of Serum Antibodies: how Physical and Mathematical Concepts Can Advance Immunology Systems. Antibodies (Basel). 11(3), 43; doi:10.3390/antib11030043 Pretini, V., Koenen, M.H., Kaestner, L., Fens, A.M., Schiffelers, R.M., Bartels, M. and Van Wijk, R. 2019. Red Blood Cells: chasing Interactions. Front. Physiol. 10, 945. Richardson, K.J., Kuck, L. and Simmonds, M.J. 2020. Beyond oxygen transport: active role of erythrocytes in the regulation of blood flow. Am. J. Physiol. Heart. Circ. Physiol. 319(4), 866–872. Rockstroh, C., Hintz, K., Kannenberg, J. and Jassoy, C. 2024. Measurement of the Memory B Cell Response via Antibodies from Activated Cells. Antibodies 13, 81; doi:10.3390/antib13040081 Smagulova, Z.Sh., Makarushko, S.G., Kim, T.D., Ostapchuk, E.O. and Tashenov, K.T. 2010. Indicators of the adsorption-transport function of erythrocytes under the action of acetylsalicylic acid. Rep. Natl. Acad. Sci. Republic of Kazakhstan 6, 75–78. Smagulova, Z.Sh., Makarushko, S.G., Sadykova, H.M., Gareev, R.A., 2007. Transfer of organic substances on the surface of erythrocytes during acute intoxication with CCl4. // Medical and physiological problems of human ecology: materials of the All-Russian conference with international participation. – Ulyanovsk. Russia. 56 p. Thiagarajan, P., Parker, C.J. and Prchal, J.T. 2021. How Do Red Blood Cells Die?. FrontPhysiol 12, 655393; doi:10.3389/fphys.2021.655393 Zhang, X., Lin, Y., Xin, J., Zhang, Y., Yang, K., Luo, Y. and Wang, B. 2024. Red blood cells in biology and translational medicine: natural vehicles inspire new biomedical applications. Theranostics 14(1), 220–248. | ||
| How to Cite this Article |
| Pubmed Style Demchenko G, Berdikulov M, Sansyzbay A, Abdreshov S, Koibasova L, Khusainov D, Lessova Z. Antibodies in plasma and blood cells of horses. Open Vet. J.. 2026; 16(2): 1171-1177. doi:10.5455/OVJ.2026.v16.i2.35 Web Style Demchenko G, Berdikulov M, Sansyzbay A, Abdreshov S, Koibasova L, Khusainov D, Lessova Z. Antibodies in plasma and blood cells of horses. https://www.openveterinaryjournal.com/?mno=283010 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.35 AMA (American Medical Association) Style Demchenko G, Berdikulov M, Sansyzbay A, Abdreshov S, Koibasova L, Khusainov D, Lessova Z. Antibodies in plasma and blood cells of horses. Open Vet. J.. 2026; 16(2): 1171-1177. doi:10.5455/OVJ.2026.v16.i2.35 Vancouver/ICMJE Style Demchenko G, Berdikulov M, Sansyzbay A, Abdreshov S, Koibasova L, Khusainov D, Lessova Z. Antibodies in plasma and blood cells of horses. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1171-1177. doi:10.5455/OVJ.2026.v16.i2.35 Harvard Style Demchenko, G., Berdikulov, . M., Sansyzbay, . A., Abdreshov, . S., Koibasova, . L., Khusainov, . D. & Lessova, . Z. (2026) Antibodies in plasma and blood cells of horses. Open Vet. J., 16 (2), 1171-1177. doi:10.5455/OVJ.2026.v16.i2.35 Turabian Style Demchenko, Georgiy, Maxat Berdikulov, Abylai Sansyzbay, Serik Abdreshov, Laura Koibasova, Damir Khusainov, and Zhaniha Lessova. 2026. Antibodies in plasma and blood cells of horses. Open Veterinary Journal, 16 (2), 1171-1177. doi:10.5455/OVJ.2026.v16.i2.35 Chicago Style Demchenko, Georgiy, Maxat Berdikulov, Abylai Sansyzbay, Serik Abdreshov, Laura Koibasova, Damir Khusainov, and Zhaniha Lessova. "Antibodies in plasma and blood cells of horses." Open Veterinary Journal 16 (2026), 1171-1177. doi:10.5455/OVJ.2026.v16.i2.35 MLA (The Modern Language Association) Style Demchenko, Georgiy, Maxat Berdikulov, Abylai Sansyzbay, Serik Abdreshov, Laura Koibasova, Damir Khusainov, and Zhaniha Lessova. "Antibodies in plasma and blood cells of horses." Open Veterinary Journal 16.2 (2026), 1171-1177. Print. doi:10.5455/OVJ.2026.v16.i2.35 APA (American Psychological Association) Style Demchenko, G., Berdikulov, . M., Sansyzbay, . A., Abdreshov, . S., Koibasova, . L., Khusainov, . D. & Lessova, . Z. (2026) Antibodies in plasma and blood cells of horses. Open Veterinary Journal, 16 (2), 1171-1177. doi:10.5455/OVJ.2026.v16.i2.35 |