| Research Article | ||
Open Vet. J.. 2026; 16(2): 1193-1203 Open Veterinary Journal, (2026), Vol. 16(2): 1193-1203 Research Article Modified fibrin sealant-cellulose efficacy on wound healing: Histopathological studyHussein I. Kareem1*, Abbas Hadi Jasim Al-Mahmoudi1, Muslim F. Diwan2, Hala Abbas Naji1 and Amir I. Towfik21Department of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Al-Qadisiyah, Al Diwaniyah, Iraq 2Department of Veterinary Surgery and Obstetric, College of Veterinary Medicine, University of Al-Qadisiyah, Al Diwaniyah, Iraq *Corresponding Author: Hussein I. Kareem. Department of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Al-Qadisiyah, Al Diwaniyah, Iraq. Email: Hussein.Imran [at] qu.edu.iq Submitted: 13/09/2025 Revised: 15/01/2026 Accepted: 26/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
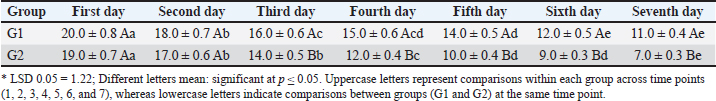
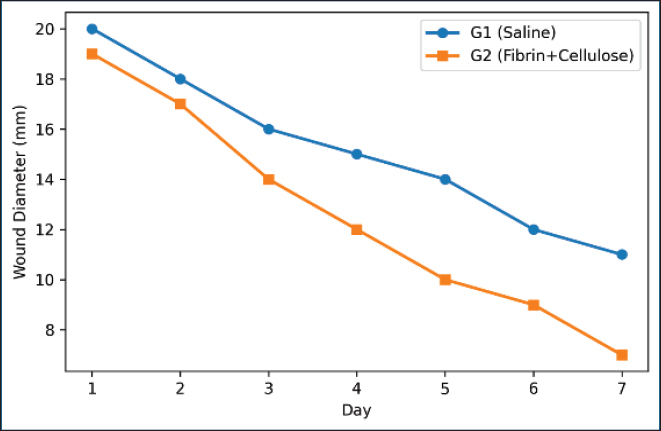
AbstractBackground: Wound healing takes a long time. To heal correctly, cells need specific conditions. Natural materials may assist cells in faster wound healing. Aim: This study aimed to test the healing qualities of a modified fibrin sealant containing cellulose isolated from date palm fronds (Phoenix dactylifera). Methods: Twelve local dogs were used, and they were divided into two groups: 1: control, administered normal saline; 2: test, treated with the fibrin–cellulose mixture. Each dog was given a circular wound of 20 mm. The assigned therapy was administered for 7 consecutive days. On days 7, 14, and 21, the wounds were measured, and tissue samples were collected for histological analysis. Results: As of day 7, the average size of the wounds in Group 2 decreased to 7 mm, whereas that in Group 1 remained at 11 mm (p ≤ 0.05). On day 14, the epithelial regeneration in the treatment group was at a more advanced stage than that in the control group. The treatment group had more developed collagen bundles and more mature vessels, suggesting that the group had advanced more than the others in the remodeling stage of the cycle. By day 21, wounds in Group 2 had fully healed, and their skin had completely regenerated. These wounds had a thick keratinized epidermis. On the other hand, Group 1 still had incomplete healing and noticeably thinner collagen networks. The treated group exhibited denser collagen bundles and more developed vascular growth, indicating a faster progression to the remodeling phase. Conclusion: The combination of fibrin and cellulose enhanced tissue restoration speed and quality. Cellulose served as a supportive lattice for the cells, enabling rapid proliferation and repair. This study demonstrates the ability of plant cellulose incorporated into fibrin sealant to promote rapid skin wound deterioration in dogs. Keywords: Angiogenesis, Collagen, Hemostasis, Palm,Regeneration. IntroductionHealing a wound is a slow, intricate process that involves many events. The tissue environment must be optimal to facilitate and enable healing. The wound bed environment must be kept warm, moist, clean, and protected. One of the modern wound healing strategies is the development of novel wound dressings that promote healing rather than merely protecting the wound. Hydrogels are one of the most innovative dressings developed. Chitosan and cellulose hydrogels are particularly effective as dressings. They have an excellent water retention capacity and therefore maintain a moist wound environment that is conducive to cellular proliferation (Al-Deresawi et al., 2022). Wound dressings also prevent the entry of bacteria (Al-Deresawi et al., 2022). This further reduces the chances of contracting an infection (Abd-Alhassen et al., 2021; Ghazi et al., 2024). Moisture also alleviates discomfort and promotes tissue proliferation more rapidly. Such dressings can absorb moisture while also preventing the entry of pathogenic microorganisms (Karim et al., 2018; Yaseen et al., 2020). Bacterial cellulose (BC) is amenable to various modifications to enhance its functional properties, for example, the inclusion of therapeutic agents that are typically beneficial for wound healing. The addition of anti-inflammatory or antibiotic agents transforms a dressing into a therapeutic device that is more than a passive protective layer. It is particularly suitable for dressing over contours of various configurations and irregular wound surfaces (Al-Deresawi et al., 2022). Both plant and bacterial cellulose can be used to create very thin yet powerful fibers or nanofibers (Al-Deresawi, 2024). Plant cellulose is more abundant and less expensive than other alternatives. Additionally, it is biodegradable without harming the body, which is an advantage. Although it is not the same as BC, it possesses many healing properties. Using both types of cellulose as scaffolds to support tissue regeneration is highly beneficial. Moreover, scientists can incorporate other materials with cellulose. For instance, the addition of nanoparticles results in a composite with superior properties. Such composites are able to combat bacterial infections and enhance healing, which is especially beneficial for infected or slow-healing wounds (Al-Deresawi et al., 2023). This study aims to expand the existing knowledge base by evaluating a novel cellulose and fibrin-based wound sealant. This combination may be synergistic in enhancing the functionality of PDC. The outcomes are being assessed in a preclinical model to validate the improvement in wound healing. Materials and MethodsPreparation of the cellulose fibrin sealantCellulose was extracted from the midrib of the fronds of date palms. The dried frond midribs were ground into a fine powder. Cellulose was then extracted using a Soxhlet extractor with 100% ethanol at 45°C for 6 hours. After extraction, the ethanol was evaporated by drying the cellulose in an oven at 40°C. Once dried, the cellulose was stored at 4°C for later use in the formulation. The study methods were applied for the first time. Fresh canine blood was taken from the jugular vein and immediately mixed with sodium citrate at a final concentration of 1% w/v to prepare the fibrin components to prevent clotting. The plasma was separated by centrifugation at 4,000 rpm for 10 minutes and stored at 20°C. Plasma was thawed at 4°C for 24 hours before use. Tranexamic acid (3,000 KIU/ml) was added to reduce clot degradation. Ethanol was added to the tranexamic acid to precipitate the fibrinogen. The resulting mixture was combined with the suspended cellulose in a ratio of 2.5:1. This mixture contained fibrinogen and cellulose components. Preparation of thrombin-cellulose solutionOne 100 ml of plasma was diluted to 1 l to prepare the thrombin solution. Subsequently, 3% acetic acid was added to adjust the pH to 5.3. The solution was centrifuged at 3,000 rpm for 5 minutes at 10°C. The resulting sediment was resuspended in 0.85% saline (25 ml). The pH was then neutralized to 7.0 by adding 2% sodium carbonate. Following the addition of sodium carbonate, cellulose was added to the solution in a ratio of 2.5:1 corresponding to the thrombin mix. This completed the thrombin and cellulose formulation. The fibrinogen, thrombin, and cellulose solutions were filled in a custom-built dual-chamber syringe. This design enabled the user to extrude the 2 solutions through a single outlet, thereby instantaneously mixing the solutions and applying the sealant to the wound. Experimental designTwelve healthy mixed-breed dogs aged 9 ± 2 months and weighing 20 ± 3 kg were randomly assigned to 2 groups at random. The first group (G1) was the control group that received only 0.9% sodium chloride (normal saline). Before being considered for inclusion in the study, all dogs underwent a thorough clinical examination. Physical health, a full blood count, a thorough examination of blood serum biochemistry, stool samples for fecal parasites, and confirmation of recent vaccinations were assessed. Only those pets that were clinically normal and did not have any infectious, metabolic, or dermatological diseases were included in the study. Group 2 (G2) was considered the treatment group and was treated with fibrin–cellulose sealant. A 20-mm circular incision was made on the back of each dog under aseptic surgical circumstances. Fibrin–cellulose mixture was thawed at 37°C and applied as a thin layer once a day for a week on G2’s wounds. G1 had normal saline as a control. The wound diameter was evaluated at predefined time points (days 0, 7, 14, and 21). The wounds were biopsied, and the edges of the wounds were taken on days 7, 14, and 21 of the experiment. The samples were taken, stained with hematoxylin and eosin (H and E), and observed under a light microscope to analyze the healing and repair of the tissue. Samples were taken from the wound border using a 6-mm stab biopsy sampler. Before tissue excision, each canine patient was sedated, and local anesthesia was administered. The biopsy was performed where normal surrounding skin and the edge of the wound met to retrieve a skin core sample in its entirety, comprising the epidermis, dermis layer, and upper portion of the subcutis. Gentle pressure was used to stop the bleeding immediately after the sample was taken, and the biopsy area was disinfected. On days 7, 14, and 21, all samples were taken from the same anatomical location in every animal to guarantee uniformity in the histological assessments. The wound diameter was recorded daily from day 0 to day 7. No further surface measurements were performed thereafter due to complete wound closure. Days 7, 14, and 21 were designated for histological analysis and hydroxyproline analysis. Anesthesia, analgesia, and perioperative careAnimals received preanesthetic medication xylazine at 1 mg/kg body weight (IM) before induction with ketamine at 5 mg/kg IM. To supplement preoperative analgesia, local infiltration of lidocaine 2% was used in the incision region. The surgical site was clipped, treated with 10% povidone–iodine solution, and left in contact for 3 minutes before incision. Throughout the procedure, the animals were monitored for heart and respiratory rates and the color of their mucous membranes. Meloxicam 0.2 mg/kg (IM) was administered every 24 hours for 3 consecutive days. If signs of discomfort were observed, rescue analgesia was achieved with the administration of 2 mg/kg IM tramadol. All animals were monitored for signs of delayed healing, infection, abnormal behavior, appetite around the surgical site, and surgical site appearance. Discomfort and pain were alleviated with the help of analgesia and daily postoperative follow-ups. Every effort was made to comply with the national animal welfare law and ARRIVE rules. The Institutional Animal Care and Use Committee provided the supervision necessary to guarantee the study’s ethical and welfare considerations. Histomorphometry analysisSlides containing the tissue samples were scanned, and the images were analyzed using ImageJ. The thickness of the epidermis, the amount of collagen at each level, and the number of blood vessels were recorded. Each slide was represented by the average of 5 fields. Immunohistochemistry sectionsThe sections were stained for CD68 to label the recruited macrophages and with Myeloperoxidase to label the neutrophils. Using a light microscope, they could view the stained samples. The number of activated macrophages and neutrophils in the random fields of view was documented. The average number of activated cells was calculated for each group. Hydroxyproline testAfter the skin samples had been dried, they were weighed, and the amount of hydroxyproline in each sample was determined by a colorimetric method. The absorbance at 560 nm was determined to quantify hydroxyproline. Results are presented in micrograms in relation to tissue weight in milligrams. The test was conducted on days 7, 14, and 21. The hydroxyproline concentration was estimated using a commercially available colorimetric determination of hydroxyproline assay kit (Cat. No. ab222941, Abcam, Cambridge, UK). Bacterial swab cultureDaily, sterile cotton swabs were used to collect the wound samples. Samples were inoculated in nutrient agar plates that were incubated at 37°C for 24 hours. Colony-forming units (CFU) were counted and recorded. No samples were collected after day 7 because all wounds were closed. Plates were incubated under aerobic conditions at 37°C for 24 hours. Profiles of the wound surface were collected using sterile cotton swabs moistened with sterile saline solution. Each swab was streaked on nutrition agar plates using a quadrant streaking technique. Plates were incubated aerobically at 37°C for 24 hours. Bacterial growth was evaluated on a semi-quantitative basis depending on the degree of adhesion of the colonies to the plates. Profiles were not taken after day 7 because the wounds had fully healed. Measurement of wound pHA mini pH probe was used to determine the wound pH. Measurements were recorded only once a day before cleaning or treating the wound. The pH was monitored throughout the week. No samples were collected after day 7 because all wounds were closed. Complete wound closure was defined as the formation of a full layer of epithelial tissue over the wound, with none of the following: drainage, dead tissue, or scabs. Statistical analysisTo evaluate the impact of the modified sealant, wound diameters were measured, and their change over time was analyzed using the Statistical Package for the Social Sciences (version 32). Differences over time and among the groups were analyzed using a 2-way Analysis of Variance (ANOVA). This test was used to assess differences in the healing rates of groups G1 and G2 over the entire duration of the study. The ANOVA was followed by the Hebrew test, which determines whether the differences between means were large enough for the researcher to conclude that the means were different. Values are considered statistically significant at p ≤ 0.05. This methodology shows that the observed treatment effects were significant and were not random occurrences. No statistical power analysis was performed before or after the study. Twelve dogs were included in the study due to their availability and inclusion in previous similar studies in veterinary research. Given the study’s primary objective of serving as an initial study to investigate the biological effect of the fibrin–cellulose sealant, the study design was not made to achieve a specific effect size. Ethical approvalThe Animal Care and Use Committee within the College of Veterinary Medicine at the University of Al-Qadisiyah reviewed and approved this study. Ethical approval was received per Proposal Reference Number 1877 dated September 11, 2022. ResultsMeasurements of wound contractionData on wound contraction for a single week revealed a marked difference between the two groups. On the first day, the wound diameter was almost the same in both groups (20 mm for G1 and 19 mm for G2). However, on the second day, G2 started to heal faster, with a reduction in wound diameter to 14 mm, whereas G1 decreased to 16 mm. However, this difference was determined to be statistically significant (p ≤ 0.05) starting on day 3. On day 7, the residual wound diameter in G2 was reduced to 7 mm, whereas that in G1 was 11 mm. The findings show that the group treated with the fibrin–cellulose mixture healed significantly faster (Table 1). Table 1. Daily wound contraction measurements (mm) in G1 (control) and G2 (treated with fibrin–cellulose sealant) over 7 days.
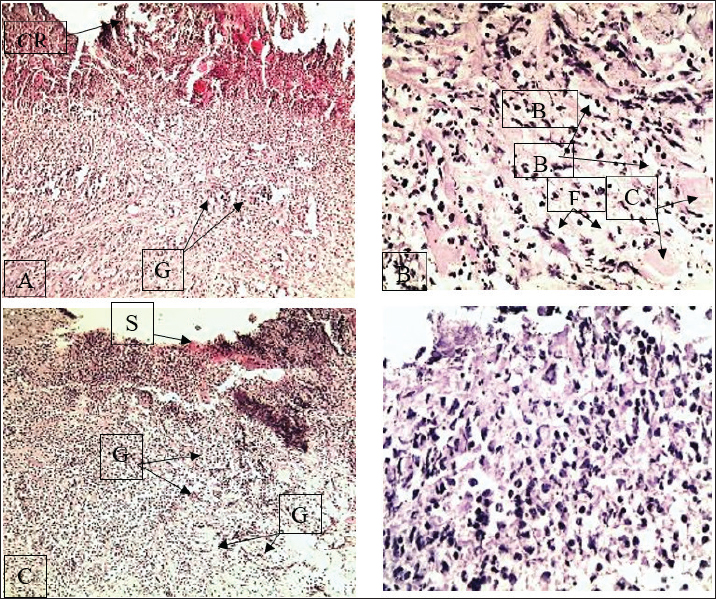
The LSD value of 1.22 also showed that changes in the wound size for G1 and G2 were significant as of day 3. This G2 had been smaller and smaller, a change of diameter that adjusted to the finding of the modified fibrin sealant that had been applied to the tissue to help it close faster. The findings show that cellulose-based enhancement is effective in topical wound care treatments. Histopathological findingsFirst weekHistological examination of the G1 samples revealed a thick crust on the wound surface, with a thick layer of granulation tissue underlying it. The findings demonstrate that G1 granulation was adequate, coupled with the crust, and formed new tissues during the granulation phase of healing. Although there were estimable wound diameters in the residual portions of the wounds, the complete closure of the new epithelium was in portions, mostly after day 7, and surface area estimations were not performed. Among the sample tissues, macrophage inflammatory cell infiltration was prominent. This was the formation of collagen. The collagen was woven loosely, indicating that the healing process had started in the early stages of the inflammatory and proliferative phases. At discharge, the tissue had not matured, whereas the epidermis had not matured and showed little healing. Therefore, the tissue structure was immature. In contrast, tissue samples in G2 showed far better and improved tissue cohesive organization. At this point, the crust had already started. This also resulted in the healing of the advanced and initial stages beyond. Granulation tissue with a more cohesive structure was abundant, where collagen fibers formed a thick, interwoven arrangement. There was a predominance of neutrophils as the inflammatory cells, and signs of new vascularization were evident. The collagen framework was well-formed, providing improved support to the tissue that was undergoing active growth. The treated group progressed through the stages of healing more rapidly than the control group (Fig. 1).
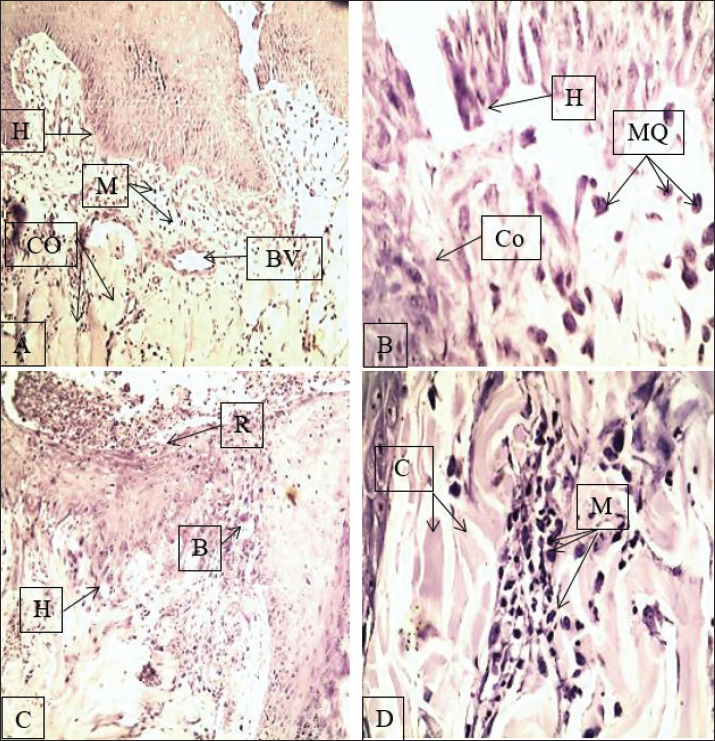
Fig. 1. Histology of the skin in the first week of the study showing the start of the sequence of events in wound repair. In G1 (A-B), the slices show a thick surface crust, granulation tissue (macrophages with collagen fibrosis), and collagen fibrosis, whereas G2 (C-D) shows crust sloughing, an organized pattern of collagen interlaced with dominant collagen and neutrophils, suggesting a more advanced stage of the stream of continuum. G=granulation tissue, B=blood vessel, F=fibrosis/collagen fibers, C=crust, S=epidermal surface, BV=blood vessel, CO=collagen, MQ=macrophages, H=hair follicle, M=muscle. A, C, 100 B, D 400. Second weekBy the second week, the histology of tissue samples from the G1 group showed moderate epidermal hyperplasia, particularly of the stratum basale. The collagen fiber bundles had an increased thickness, and new blood vessels continued to develop. The macrophages were still active in the interstitium. The wound was still not closing at the wound edges. Some signs of the inflammatory response continued. As healing progressed, more tissue regeneration appeared to be necessary for complete healing. On the other hand, the G2 samples displayed more accelerated healing. The wound surface had little remaining scab. The epidermis showed downgrowth, and the wound margins had already fused. The surface was underpinned by a dense collagen matrix, and the macrophages were still present to encourage remodeling. At higher magnification (X400), more compact and aligned collagen fibers were seen. In the second week, tissue samples demonstrated that the Fetal Calf Serum significantly advanced tissue regeneration and epidermal remodeling and enabled more rapid wound closure (Fig. 2).
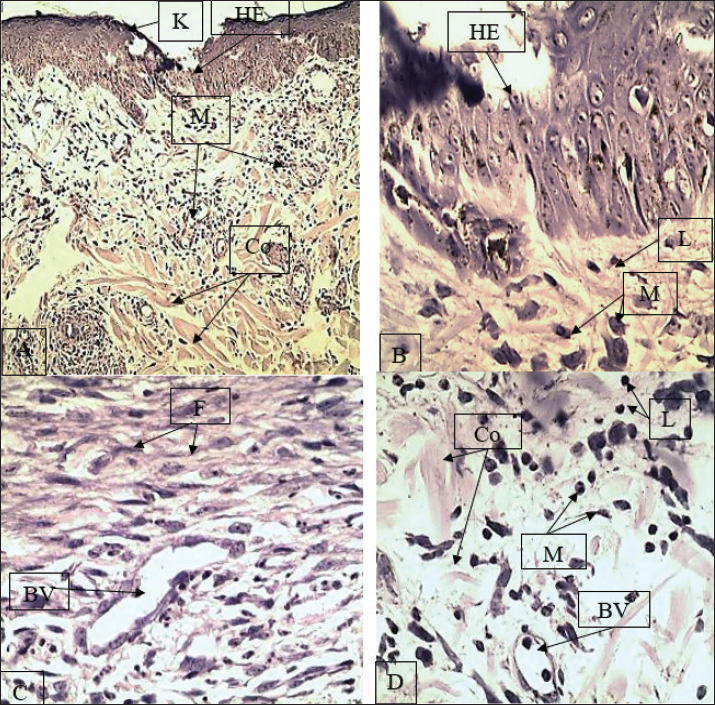
Fig. 2. Skin tissue repair at the second week. G1 (A–B) tissue sections show downward growth of the basal epidermis, collagen deposition, and the presence of new lood vessels (angiogenesis). G2 (C–D) tissue sections show epidermal bridging, slight crusted areas, collagen within the connective tissue, and macrophages in the tissue, which aids healing (remodeling). A=angiogenesis; K=keratin; HE=hyperplastic epidermis; M=macrophages; Co=collagen fibers; H=hair follicle; BV=blood vessel; L=lymphocytes; GA=granulation area. Magnification: A and C=X100; B and D=X400. Third weekIn the third week of the study, the G1 group showed a further improvement, although still lagging. Prominent horizontal crests of fibrosis were observed along the wound surface, along with the development of neovascularization. However, the collagen retrogression was still fragile and poor. Although lymphocytes and macrophages were still present, the tissue had not yet reached full structural maturity, suggesting ongoing inflammation. The G2 group underwent surface and microscopic examination, which showed complete wound healing. The epidermis’s basal layer was fully restored, and a newly formed thick keratinized stratum had formed. The dermis, the underlying layer of the skin, had collagen bundles with good structure and organization, interspersed with progressive fibrosis and a plentiful supply of new capillaries. Inflammatory cells were present, but in minor quantities, as they assisted in the remodeling step. Thus, the combination of cellulose and fibrin enhanced healing and tissue quality by the third week (Fig. 3).
Fig. 3. Wound healing after the third week with skin histological analysis. The segments in G1 (A-B) demonstrate horizontal fibrosis, loosely arranged and tenuous collagen fibers, and neovascularization. G2 (C-D) demonstrates complete epidermal renewal, a thick, heavily keratinized outer stratum, collagen fibrosis, and a remnant presence of fibrocytes. Late remodeling. R=regenerated epidermis; H=hair follicle; B=blood vessel; C=crust remnants; F=fibrosis/collagen fibers; M=muscle; Co=collagen; L=lymphocytes; BV=blood vessel. Magnification: A and C=×100 and B and D=×400. Throughout the 7-day measurement period, the treated group (G2) showed the fastest rate of wound closure. By the last measurement at 7 days, the wound measurement in the treated group (G2) decreased to 7 mm, compared with that in the control group (G1), which remained at 11 mm (p ≤ 0.05) (Fig. 4).
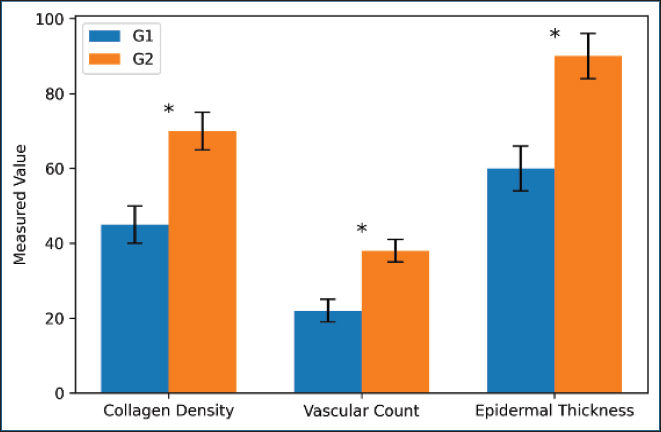
Fig. 4. Wound diameter over 7 days in the G1 and G2 groups. G2 wounds healed faster than G1 wounds. By day 7, G2 reached 7 mm, whereas G1 remained at 11 mm. On day 21, G2 exhibited increased collagen density, a higher quantity of blood vessels, and thicker epidermal layers when placed side by side with G1, and the differences were statistically significant (Fig. 5).
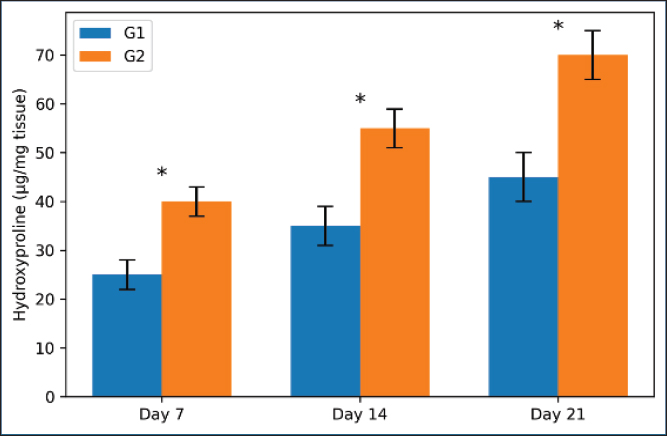
Fig. 5. Comparison of the collagen density (% area), vascular count (vessels/mm²), and epidermal thickness (µm) on day 21. G2 showed higher collagen content, more vessels, and thicker epidermis. Statistically significant differences (*p ≤ 0.05). G2 exhibited a greater hydroxyproline level in the tissue on days 7, 14, and 21. By day 21, the difference reached a statistically significant level (Fig. 6).
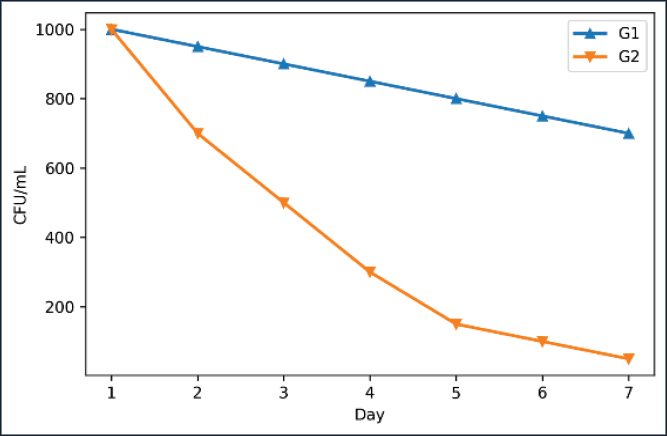
Fig. 6. Hydroxyproline levels on days 7, 14, and 21 in both groups. G2 had more collagen at all time points. The difference increased with time and was significant. Bacterial findingsG2 had a steep decline in bacterial counts starting on day 2 and showed clinically uninfected wounds by day 7, unlike the G1 group (Fig. 7).
Fig. 7. Bacterial load (CFU/ml) over 7 days in both groups. G2 showed a rapid decrease in bacterial counts. By day 7, G2 was nearly bacteria-free. Duration until complete closureThroughout the study period, both groups experienced no adverse incidents. Mild crusting and erythema, which occurred during the first few days, resolved on their own. None of the participants showed signs of wound infection, excessive discharge, dehiscence, necrosis, or other abnormalities. All dogs exhibited normal eating and behavioral patterns during the study period. DiscussionThis paper proved that using a cellulose-containing fibrin sealant in the treatment of wound healing in dogs is effective, as cellulosic materials, including fibrin, bacterial, and plant cellulosic materials, are significant and recent additions in the field of wound healing. Cellulosic biopolymers, because of their high-water retention, mechanical properties, and biocompatibility, are ideal in wound healing (Portela et al., 2019). In the histological study performed in the third week, epithelial encroachment was advanced, and the collagen in the tissue was more densely organized. Moreover, tissue neovascularization was more abundant in the dogs treated with fibrin–cellulose and the control dogs. Our fibrin cellulose mixture probably did the same as Datta et al. (2025) and provided a neovascularization and cell regeneration scaffolding that fibroblasts require to remodel and proliferate tissues. The observations and results of the study agree with and expand on the works of Paleczny et al. (2021) on biofunctional celluloses. In our study, the palm frond cellulose provided a nanofibrillar scaffold that was instrumental in wound hemostasis and stabilized the wound bed during the early stages of healing, similar to the findings of Cidreira et al. (2021), who showed that the cellulose nanocrystals rapidly promoted hemostasis and reinforced early wound bed architecture. Integrating cellulose with fibrin resulted in a sealant with multiple benefits: structural support for the sealant itself, extended local retention, diminished fluid loss, and inflammation control, all of which are paramount for wound closure. Moreover, the enhanced epithelial regeneration in G2 animals corroborates recently proposed approaches that employ bio-adhesive polysaccharide hydrogels to replicate the dermal layer, thus directing and enhancing cell migration (Yang and Wang, 2023). In comparison to the control group, the treated animals exhibited significantly greater reepithelialization and keratin layer formation. This was likely due to the combined effect of fibrin and cellulose as a matrix and growth stimulant. 3D biofabricated cellulose hydrogels have also been shown to accelerate healing by promoting hydration and stabilizing important growth factors (Tabatabaei Hosseini et al., 2024). Although our technique was simpler and computerized molding was not employed in our design, the end biological effect was still effective. This suggests the broad versatility of cellulose in scaffold-based healing and tissue engineering. Bacterial cellulose materials have preferential use in infected and chronic wounds for their antibacterial and anti-inflammatory properties (Zheng et al., 2020; Meng et al., 2023). Our study demonstrates that the presence of natural cellulose in surgically clean wounds provides an opportunity for a flexible environment that allows for unprecedented tissue remodeling without excessive inflammation. This agrees with Carvalho et al. (2019), who stated that BC is not only a drug carrier but also a passive catalyzer of the healing process. On the third week of the study, samples in the G2 group demonstrated the inactivity of primary polymorphonuclear cells, the presence of intensely streaked collagen aligned in the same direction, and complete restoration of skin and tissue in the deep dermis—these observations prolong the passive role of healing. Future work, as stated by Torkaman et al. (2021), is expected to combine amino-acid-modified chitosan with cellulose to offer more opportunities to augment the healing process of the tissue, and similarly, the work of D A Ghazi et al. (2024) to clinical practices will be offered to patients of 3D prepared BC dressings. This provides an opportunity to offer our patients a more stable healing environment. Moreover, with the results we obtained, it is possible to conclude that a simple fibrin–cellulose mixture is a combination that offers a lot of healing opportunities, and that multifunctional and designed fabrics will be able to offer their healing opportunities in chronic infected extensive wounds. This study has at least 1 limitation. A comparison group where only fibrin was used was lacking. Because this study only compares the fibrin–cellulose sealant to saline, one cannot disentangle the effects of the fibrin itself from the effects of cellulose. Given the material constraints and the primary aim of this preliminary study to assess the performance of the composite formulation, a fibrin-only arm was omitted. Future studies should consider a three-arm design of saline control, fibrin alone, and combined cellulose and fibrin to adequately understand the specific contributions of each constituent to the healing response. Another limitation is that the experiment was performed in healthy animals with clean, organized wounds. The challenge for the sealant was to evaluate its effectiveness on chronic or infected wounds. Clinical challenges remain to be determined, and infected or chronic wounds and healthy wounds will be used to evaluate the clinical effect of the sealant. ConclusionCompared with saline treatment, the sealant made from palm fronds, fibrin, and cellulose showed more rapid wound contraction and more organized granulation tissue in clean surgical wounds. Of all the studies conducted to date, this study was the most convincingly conducted under controlled conditions, providing evidence to support the first-order biological activity of the composite formulation. In consultation with the study team, we recommend that future studies include a fibrin-only and cellulose-only group and that the sealant be evaluated in chronic and infected wounds to fully delineate its therapeutic potential. AcknowledgmentThe authors thank the College of Veterinary Medicine, University of Al-Qadisiyah, for their support in this study. Conflict of interestThe authors declare no conflicts of interest. FundingThe authors self-funded the study. No external funding source is available. Authors’ contributionsHussein I. Kareem: Conceptualization, Methodology, Experimental design, Data interpretation, Writing—Original draft preparation. Abbas Hadi Jasim Al-Mahmoudi: Histopathological analysis, Microscopy, Data curation, Writing—Review and Editing. Muslim F. Diwan: Surgical procedures, Animal handling, Sample collection, Investigation. Hala Abbas Naji: Laboratory analysis, Hydroxyproline assay, Microbiological evaluation, Validation. Amir I. Towfik: Supervision, Project administration, Final manuscript revision, Writing – Reviewing and Editing. Data availabilityData are available when requested via the corresponding author. ReferencesAbd-Alhassen, J.K., Janabi, A.H.D. and Aboktifa, M.A. 2021. Antioxidant and antimicrobial evaluation of lycopene isolated from watermelon. Biochem. Cellular. Arch. 21, 2905–2910. Al-Deresawi, T.S. 2024. Isolation and characterization of fermenting bacterial isolates from vinegar industry waste in local markets of Wasit Province. Karbala Int. J. Modern Sci. 10(3), 442–448. Al-Deresawi, T.S., Mohammed, M.K. and Khudhair, S.H. 2022. The use of bacterial cellulose produced from the local isolate Komagataeibacter xylinus TELE as an antibacterial for wounds. Int. J. Drug. Del. Technol. 12(4), 1825–1830; doi:10.25258/ijddt.12.4.56 Al-Deresawi, T.S., Mohammed, M.K. and Khudhair, S.H. 2023. Isolation, screening, and identification of local bacterial isolates producing bio-cellulose. J. Med. Chem. Sci. 6(3), 622–633. Carvalho, T., Guedes, G., Sousa, F.L., Freire, C.S.R. and Santos, H.A. 2019. Latest advances on bacterial cellulose-based materials for wound healing, delivery systems, and tissue engineering. Biotechnol. J. 14(12), e1900059; doi:10.1002/biot.201900059 Cidreira, A.C.M., De Castro, K.C., Hatami, T., Linan, L.Z. and Mei, L.H.I. 2021. Cellulose nanocrystals-based materials as hemostatic agents for wound dressings: a review. Biomed. Microdevices. 23(4), 43; doi:10.1007/s10544-021-00581-0 Datta, D., Bandi, S.P., Colaco, V., Dhas, N., Saha, S.S., Hussain, S.Z. and Singh, S. 2025. Cellulose-based nanofibers infused with biotherapeutics for enhanced wound-healing applications. ACS Polym. Au 5(2), 80–104; doi:10.1021/acspolymersau.4c00092 Ghazi, A.M., Ali Al-bayati, M.A. and Janabi, A.H. 2024. Metabolomics-detected alterations generated by phytosomal propolis and phytosomal lycopene in male rats with induced benign prostatic hyperplasia. Iraqi J. Vet. Sci. 38(Suppl I–IV), 7–15. Karim, S., Mansour, K., Janabi, A. and Al-Nakeeb, N. 2018. First phylogenetic characterization of pseudocowpox virus from cattle in Al-Qadisiyah province, Iraq. Iraqi J. Vet. Sci. 33(1), 123–126. Meng, S., Wu, H., Xiao, D., Lan, S. and Dong, A. 2023. Recent advances in bacterial cellulose-based antibacterial composites for infected wound therapy. Carbohydrate Polym. 316, 121082; doi:10.1016/j.carbpol.2023.121082 Paleczny, J., Brożyna, M., Junka, A., Bartoszewicz, M. and Dudek-Wicher, R. 2021. Modifications of bacterial cellulose in wound care. Polimery. W. Medycynie. 51(2), 77–84; doi:10.17219/pim/143330 Portela, R., Leal, C.R., Almeida, P.L. and Sobral, R.G. 2019. Bacterial cellulose: a versatile biopolymer for wound dressing applications. Microbial Biotechnol. 12(4), 586–610; doi:10.1111/1751-7915.13392 Tabatabaei Hosseini, B.S., Meadows, K., Gabriel, V., Hu, J. and Kim, K. 2024. Biofabrication of cellulose-based hydrogels for advanced wound healing: a special emphasis on 3D bioprinting. Macromolecular Bioscience 24(5), e2300376; doi:10.1002/mabi.202300376 Torkaman, S., Rahmani, H., Ashori, A. and Najafi, S.H.M. 2021. Modification of chitosan using amino acids for wound healing purposes: a review. Carbohydrate. Polym. 258, 117675; doi:10.1016/j.carbpol.2021.117675 Yang, J. and Wang, S. 2023. Polysaccharide-based multifunctional hydrogel bio-adhesives for wound healing: a review. Gels 9(2), 138; doi:10.3390/gels9020138 Yaseen, M.M., Karawan, A.C., Alfatlawi, M.A.A. and Janabi, A.H.D. 2020. The role of gut bacterial cytochrome-P450 of mosquito larvae in degradation of temephos insecticide. Ann. Trop. Med. Public Health 23(1), S412. Zheng, L., Li, S., Luo, J. and Wang, X. 2020. Latest advances on bacterial cellulose-based antibacterial materials as wound dressings. Front. Bioengineering Biotechnol. 8, 593768; doi:10.3389/fbioe.2020.593768 | ||
| How to Cite this Article |
| Pubmed Style Kareem HI, Al-mahmoudi AHJ, Diwan MF, Naji HA, Towfik AI. Modified fibrin sealant-cellulose efficacy on wound healing: Histopathological study. Open Vet. J.. 2026; 16(2): 1193-1203. doi:10.5455/OVJ.2026.v16.i2.37 Web Style Kareem HI, Al-mahmoudi AHJ, Diwan MF, Naji HA, Towfik AI. Modified fibrin sealant-cellulose efficacy on wound healing: Histopathological study. https://www.openveterinaryjournal.com/?mno=283667 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.37 AMA (American Medical Association) Style Kareem HI, Al-mahmoudi AHJ, Diwan MF, Naji HA, Towfik AI. Modified fibrin sealant-cellulose efficacy on wound healing: Histopathological study. Open Vet. J.. 2026; 16(2): 1193-1203. doi:10.5455/OVJ.2026.v16.i2.37 Vancouver/ICMJE Style Kareem HI, Al-mahmoudi AHJ, Diwan MF, Naji HA, Towfik AI. Modified fibrin sealant-cellulose efficacy on wound healing: Histopathological study. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1193-1203. doi:10.5455/OVJ.2026.v16.i2.37 Harvard Style Kareem, H. I., Al-mahmoudi, . A. H. J., Diwan, . M. F., Naji, . H. A. & Towfik, . A. I. (2026) Modified fibrin sealant-cellulose efficacy on wound healing: Histopathological study. Open Vet. J., 16 (2), 1193-1203. doi:10.5455/OVJ.2026.v16.i2.37 Turabian Style Kareem, Hussein I., Abbas Hadi Jasim Al-mahmoudi, Muslim F. Diwan, Hala Abbas Naji, and Amir I. Towfik. 2026. Modified fibrin sealant-cellulose efficacy on wound healing: Histopathological study. Open Veterinary Journal, 16 (2), 1193-1203. doi:10.5455/OVJ.2026.v16.i2.37 Chicago Style Kareem, Hussein I., Abbas Hadi Jasim Al-mahmoudi, Muslim F. Diwan, Hala Abbas Naji, and Amir I. Towfik. "Modified fibrin sealant-cellulose efficacy on wound healing: Histopathological study." Open Veterinary Journal 16 (2026), 1193-1203. doi:10.5455/OVJ.2026.v16.i2.37 MLA (The Modern Language Association) Style Kareem, Hussein I., Abbas Hadi Jasim Al-mahmoudi, Muslim F. Diwan, Hala Abbas Naji, and Amir I. Towfik. "Modified fibrin sealant-cellulose efficacy on wound healing: Histopathological study." Open Veterinary Journal 16.2 (2026), 1193-1203. Print. doi:10.5455/OVJ.2026.v16.i2.37 APA (American Psychological Association) Style Kareem, H. I., Al-mahmoudi, . A. H. J., Diwan, . M. F., Naji, . H. A. & Towfik, . A. I. (2026) Modified fibrin sealant-cellulose efficacy on wound healing: Histopathological study. Open Veterinary Journal, 16 (2), 1193-1203. doi:10.5455/OVJ.2026.v16.i2.37 |