| Research Article | ||
Open Vet. J.. 2026; 16(1): 510-514 Open Veterinary Journal, (2026), Vol. 16(1): 510-514 Research Article Repeatability and reproducibility of intraocular pressure measurements in horses using rebound and applanation tonometryTatiana Martinez Moreno, Bárbara Letícia Machado Fraga, Sofia Silva Petri, Amannda Moreira Tavares, Paula Mariah César Garcia, Maria Cristina Caldart de Andrade, Paula Stieven Hünning, Fabiana Quartiero Pereira, Eduardo Ghiggi and João Antonio Tadeu Pigatto*College of Veterinary Medicine, Federal University of Rio Grande do Sul, Porto Alegre, Brazil *Corresponding Author: João Antonio Tadeu Pigatto. College of Veterinary, Federal University of Rio Grande do Sul (UFRGS), Porto Alegre, RS, Brazil. Email: pigatto [at] ufrgs.br Submitted: 13/09/2025 Revised: 12/12/2025 Accepted: 31/12/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
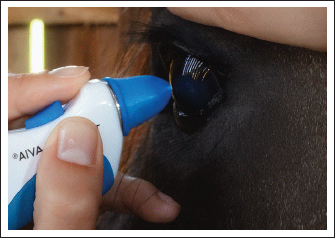
AbstractBackground: Intraocular pressure (IOP) assessment is a fundamental component of ophthalmic examinations in both veterinary and human medicine. Due to variations in IOP across species and potential differences among examiners, knowledge of normal IOP values obtained using different tonometers is essential. Aim: This study aimed to compare the repeatability and reproducibility of IOP values in horses obtained by three different examiners using rebound (Tonovet®) and applanation (Tonopen Avia®) tonometers. Methods: Thirty healthy criollo horses were examined. IOP was measured by three veterinarians with different levels of experience following routine ophthalmic evaluation. Statistical analyses included analysis of variance, Student’s t-test, and calculation of the intraclass correlation coefficient (ICC). Results: The mean IOP values were 26.16 ± 5.13 mm Hg with rebound tonometry and 22.55 ± 7.32 mm Hg with applanation tonometry, indicating a significant difference between the two devices. For the rebound measurements, the examiner-specific means were as follows: A=25.27 ± 4.9 mm Hg, B=25.80 ± 5.2 mm Hg, and C=27.40 ± 5.1 mm Hg. For applanation tonometry, the mean values were: A=20.03 ± 4.5 mm Hg, B=19.67 ± 6.0 mm Hg, and C=27.97 ± 10.3 mm Hg. Examiner C consistently obtained higher IOP values with the Tonopen Avia®. The ICC was 0.41 for rebound tonometry, indicating satisfactory reproducibility, and 0.18 for applanation tonometry, indicating poor reproducibility. Conclusion: Rebound tonometry (Tonovet®) yielded higher IOP values than applanation tonometry (Tonopen Avia®) but demonstrated superior reproducibility and was less affected by examiner variability. These findings demonstrate that Tonovet® is a more reliable tool for measuring IOP in horses under clinical and research conditions. Keywords: Intraocular pressure, Tonometry, Rebound, Applanation, Reproducibility. IntroductionAssessing intraocular pressure (IOP) is essential in ocular examinations of both humans and animals (Wada, 2006). Moreover, it is an important indicator of ocular health and a prognostic factor for ocular surgery outcomes (Whitacre and Stein, 1993). Portable instruments are used to measure IOP in animals. The two primary methods for IOP assessment are rebound tonometry and applanation tonometry (Kontiola et al., 2001; Leiva et al., 2006). In veterinary medicine, IOP values have been reported in various species, including birds of prey, cows, dogs, goats, rabbits, horses, alligators, owls, ferrets, cats, lions, mice, and sheep (Gelatt et al., 1981; Miller et al., 1991; Stiles et al., 1994; Pigatto et al., 2011). McDonald et al. (2017) compared Gelatt and Mackay (1998) rebound and applanation tonometry in alpacas, demonstrating species-specific variability and underscoring the importance of validating tonometers for each animal model. Studies comparing applanation and rebound tonometry have reported that TonoVet® tends to overestimate IOP values in cats (Rusanen et al., 2010), horses (Knollinger et al., 2005), and owls (Jeong et al., 2007). Applanation tonometers underestimate IOP values in dogs (Knollinger et al., 2005; Leiva et al., 2006; Park et al., 2011), rabbits (Pereira et al., 2011), mice (Goldblum et al., 2002), and owls (Jeong et al., 2007). Bacchin et al. (2021) demonstrated that both daily rhythm and age significantly affect IOP values, underscoring the importance of standardized measurement conditions. In contrast, reproducibility reflects the variation in measurements obtained under changing conditions, such as when different devices are used. Repeatability refers to the variation observed when repeated measurements are performed on the same subject under identical conditions. Bertens et al. (2021) reported differences in repeatability, reproducibility, and agreement among three tonometers in rabbits. In humans, studies have also investigated whether examiner-related factors influence IOP measurements (Gandhi et al., 2001). In horses, tonometry is essential for detecting glaucoma and uveitis that may progress silently and lead to irreversible vision loss. Accurate and reproducible IOP measurements in horses are critical for clinical diagnosis and therapeutic response monitoring. In addition, it is important to understand whether IOP values vary according to examiners because examiner-related variability could compromise diagnostic accuracy and clinical decision-making in equine ophthalmology. To the best of the authors’ knowledge, the influence of different examiners on IOP measurements in horses has not been previously investigated. This study aimed to evaluate the repeatability (intra-observer variability) and reproducibility (inter-observer variability) of IOP measurements in horses using rebound and applanation tonometers. Materials and MethodsSixty healthy eyes from thirty Criollo horses of different ages and both sexes, originating from Cabanha do Vigário (Viamão, RS, Brazil), were included in the study. All horses underwent a complete physical and ophthalmic examination before inclusion, and no abnormalities were detected. The ocular evaluation included slit-lamp biomicroscopy (Portable Slit Lamp SL 15, Kowa, Japan), Schirmer tear test (Ophthalmos, São Paulo, Brazil), fluorescein staining (Fluorescein Strips, Ophthalmos, São Paulo, Brazil), and IOP assessment using rebound tonometry (Tonovet®, Thiols, Helsinki, Finland) or applanation tonometry (Tonopen Avia®, Reichert, New York, USA). Measurements with each device were alternated and performed between 9:00 and 11:00 a.m. Three veterinarians measured IOP using both rebound (Tonovet®; Fig. 1) and applanation (Tonopen Avia®; Fig. 2) tonometers at 30-minute intervals. Examiner A had four years of experience with tonometry, examiner B had more than eight years of experience, and examiner C had no prior experience. The order of examinations was randomized for each horse to minimize stress and reduce examiner-related bias. Rebound tonometry was performed on the right eye, whereas applanation tonometry was performed on the left eye. The Tonovet® was calibrated in “h” mode (horse) for rebound tonometry. For applanation tonometry, three drops of 0.5% proxymetacaine hydrochloride (Anestalcon®, Allergan, Brazil) were instilled into the left eye, and IOP was measured 30 s later. Only readings with ≥95% confidence were included in the analysis.
Fig. 1. IOP measurement in a horse using rebound tonometry (TonoVet®).
Fig. 2. IOP measurement in a horse using applanation tonometry (Tonopen Avia®). Statistical analysisThe IOP values obtained from the three examiners were compared using repeated-measures analysis of variance (ANOVA) and between-group ANOVA. Additional comparisons were made using Student’s t-test, with statistical significance set at p < 0.05. The ICC was calculated to assess reproducibility. Ethical approvalThe Animal Use Ethics Committee of the Federal University of Rio Grande do Sul (CEUA-UFRGS) approved the study and conducted it in compliance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. ResultsBased on ophthalmic examinations, all horses were considered healthy, with findings within the reference ranges reported for the species. The IOP values could be successfully obtained using both tonometers. No significant differences were observed when comparing IOP values obtained from the right eye with those from the left eye, regardless of the device used or the examiner. The mean IOP values obtained with rebound tonometry (TonoVet®) were consistent across the three examiners, ranging from 25.27 ± 4.96 mmHg (examiner A) to 27.40 ± 5.14 mmHg (examiner C), with no significant statistical differences among them (p > 0.05). Applanation tonometry (Tonopen Avia®) demonstrated greater variability between examiners. Examiners A and B recorded similar values, 20.03 ± 4.52 mm Hg and 19.67 ± 6.00 mmHg, respectively, whereas examiner C obtained a substantially higher mean of 27.97 ± 10.36 mm Hg. These differences were statistically significant (p < 0.05). The results are presented in Table 1. The ICC was 0.41 for the rebound tonometer and 0.18 for the applanation tonometer. Table 1. Mean ± SD of intraocular pressure (IOP, mm Hg) in horses measured by three different examiners using rebound tonometry (TonoVet®) and applanation tonometry (Tonopen Avia®).
The intraclass correlation coefficient (ICC) for rebound tonometry was 0.41 [95% confidence interval (CI), p=0.003), while the ICC for applanation tonometry was 0.18 (95% CI, p=0.041). Overall, the results of rebound tonometry were more consistent among examiners, whereas those of applanation tonometry were more influenced by examiner variability, particularly for examiner C. DiscussionTonometry is essential for the accurate diagnosis of ocular diseases. A better understanding of each tonometer’s characteristics and limitations enhances diagnostic assessment reliability. The difference in IOP between the two eyes should not exceed 5 mmHg in healthy horses; greater asymmetry may indicate undiagnosed glaucoma or uveitis (Wada, 2006). Topical anesthetic drops may alter tear film viscosity, potentially interfering with rebound tonometry results (Chiu et al., 2008). In the current study, we performed rebound measurements (Tonovet®) on the right eye and applanation measurements (Tonopen Avia®) on the left eye. Therefore, applanation tonometry was always performed in the left eye to prevent possible contralateral interference. Although each device was consistently applied to the same eye in the present study, this design is unlikely to have influenced the findings. As reported in previous studies, clinically normal horses are not expected to exhibit significant differences in IOP between the right and left eyes (Bacchin et al., 2021). Multiple factors influence IOP, including corneal properties (Park et al., 2011), species-specific characteristics (Kontiola, 1997), head position (Komáromy et al., 2006), and the device employed (Moore et al., 1993). In this study, several measures were adopted to minimize these variables: horses were examined inside their stalls, without sedation or auriculopalpebral nerve block, and without any form of physical restraint, such as a lip twitch. No pressure was applied to the eyelids, head, or neck, and the animals’ heads were maintained above the heart level to avoid venous congestion. Previous studies in both humans and horses have demonstrated that IOP increases when the head is positioned below the heart level due to impaired venous drainage and increased extraocular muscle pressure (Linder et al., 1988; Komáromy et al., 2006; Broadwater et al., 2008). Sedatives such as xylazine, acepromazine, and ketamine were not used in our study because they are known to reduce IOP (Trim et al., 1985; Van der Woerdt et al., 1995). Similarly, auriculopalpebral nerve blocks do not affect IOP values (Van der Woerdt et al., 1995; Miller et al., 2000). Circadian rhythm is another factor that influences IOP fluctuations in horses, without necessarily indicating disease. Feeding habits and light regimens have been associated with IOP oscillations (Gelatt et al., 1981; Doyle et al., 2002; Bertolucci et al., 2009; Pereira et al., 2011). Therefore, all measurements were standardized to the morning period, when IOP values are typically lower (Wada, 2006; Bertolucci et al., 2009). The normal equine IOP values range between 15 and 30 mm Hg (Wilkie and Gilger, 2004; Wada, 2006). Although some values in this study exceeded 30 mm Hg, this was not interpreted as evidence of glaucoma. These elevations were likely related to examiner-dependent variability and transient increases in IOP caused by repeated handling (Gandhi et al., 2001). The applanation tonometer operates according to the Imbert–Fick law, meaning that the examiner can influence the force applied during measurement, potentially resulting in inaccurate IOP readings (Mcclure et al., 1976; Trim et al., 1985; Van der Woerdt et al., 1995). This may explain the discrepancies observed in the Tonopen Avia® measurements. Examiner-dependent variability has been previously described, as inadvertent pressure on the eyelids or globe during measurement can artificially elevate IOP values (Gandhi et al., 2001; Wilkie and Gilger, 2004). A study in humans demonstrated that conscious eyelid closure and examiner-applied pressure significantly increased IOP values (Gandhi et al., 2001). This effect was particularly evident with the less experienced examiner using the applanation device. Knollinger et al. (2005) compared rebound and applanation tonometry in dogs and horses and reported that Tonopen underestimated IOP in horses due to greater corneal thickness than in dogs. In this study, reproducibility was assessed using the ICC. According to the proposed scale, values between 0.75 and 1.0 are considered excellent, 0.40–0.75 satisfactory, and below 0.40 poor (Rau et al., 2012). According to the classification criteria proposed by Rau et al. (2012), the rebound tonometer (TonoVet®) demonstrated moderate reproducibility (ICC=0.41), indicating greater reproducibility than the applanation device. The ICC for the applanation tonometer (TonoPen Avia®) was 0.18, reflecting poor reproducibility. Examiner experience likely played a key role in the observed variability, particularly with applanation tonometry. Examiner C, who had no prior experience, consistently produced higher and more variable IOP values when using the Tonopen Avia®. This pattern indicates a learning curve effect, as applanation tonometry requires stable hand positioning, correct alignment, and minimal eyelid pressure. In contrast, RT is less dependent on examiner technique, which may explain the smaller inter-observer variation observed. These findings align with reports in human ophthalmology, where examiner pressure and eyelid manipulation significantly alter IOP readings, particularly among inexperienced operators. Comparable results have been reported in other species. McDonald et al. (2017) found significant variability between Tonovet® and Tonopen-XL® in alpacas, reinforcing the need for caution when interpreting results across devices and observers. Similarly, Bertens et al. (2021) demonstrated that agreement between tonometers may be limited in rabbits, even under controlled conditions, highlighting the influence of corneal biomechanics and device technology. These findings support our conclusion that RT offers more consistent results than AP tonometry, which is more susceptible to examiner-related bias. Both tonometers allowed reliable measurement of IOP in horses. However, the Tonovet® values were consistently higher than the Tonopen Avia® values. Rebound tonometry demonstrated satisfactory reproducibility (ICC 0.41), whereas applanation tonometry showed poor reproducibility (ICC 0.18). Examiner experience influenced applanation tonometry results more strongly than did rebound tonometry. Tonovet® may be preferred in equine practice due to its greater consistency and reduced operator influence. AcknowledgmentsWe are grateful to Cabanha do Vigário Farm for providing the facilities and horses used in this study. We also thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for granting Tatiana Martinez Moreno a scholarship. Conflict of interestThe authors declare no conflicts of interest. FundingThis study received no specific grant. Authors’ contributionsTMM: conceptualization, methodology, investigation, writing, review, and editing; BLMF: methodology, investigation; SSP: methodology, investigation, and writing; AMT: methodology, investigation, review, and editing; PMCG: methodology, investigation; MCCA: methodology, investigation; PSH: methodology, investigation; FQP: methodology, investigation, and writing; EG: methodology, investigation; JATP: conceptualization, supervision, writing, and funding acquisition. Data availabilityThe data that support the findings of this study are available upon request from the corresponding author. ReferencesBacchin, A.B.O., Pereira, F.Q., Hünning, P.S., Albuquerque, L., Ghiggi, E. and Pigatto, J.A.T. 2021. Effects of daily curve and age on IOP in Criollo horses. Semina. Ciênc. Agrár. 42, 2049–2056. Bertens, C.J.F., Van Mechelen, R.J.S., Berendschot, T.T.J.M., Gijs, M., Wolters, J.E.J., Gorgels, T.G.M.F., Nuijts, R.M.M.A. and Beckers, H.J.M. 2021. Repeatability, reproducibility, and agreement of three tonometers for measuring intraocular pressure in rabbits. Sci. Rep. 11, 19217; doi:10.1016/j.scirep.19217 Bertolucci, C., Giudice, E., Fazio, F. and Piccione, G. 2009. Circadian IOP rhythms in athletic horses under different lighting regimes. Chronobiol. Int. 26, 348–358. Broadwater, J.J., Schorling, J.J., Herring, I.P. and Elvinger, F. 2008. Effect of body position on intraocular pressure in dogs without glaucoma. Am. J. Vet. Res. 69, 527–530. Chiu, W., Lam, A., Chen, R. and Chiu, R. 2008. Influence of corneal properties on RT. Ophthalmology 115, 80–84. Doyle, S.E., Grace, M.S., Mcivor, W. and Menaker, M. 2002. Circadian rhythms of dopamine in mouse retina: the role of melatonin. Vis. Neurosci. 19, 593–601. Gandhi, P.D., Gürses-Özden, R., Liebmann, J.M. and Ritch, R. 2001. Attempted eyelid closure affects intraocular pressure measurement. Am. J. Ophthalmol. 131, 417–420. Gelatt, K.N. and Mackay, E.O. 1998. Distribution of intraocular pressure in dogs. Vet. Ophthalmol. 1, 109–114. Gelatt, K.N., Gum, G.G., Barrie, K.P. and Williams, L. 1981. Diurnal variation in IOP in normotensive and glaucomatous beagles. Glaucoma 29, 21–24. Goldblum, D., Kontiola, A.I., Mittag, T., Chen, B. and Danias, J. 2002. Non-invasive determination of intraocular pressure in the rat eye: comparison of electronic tonometer (Tonopen®) and rebound (Impact probe) tonometer. Graefes. Arch. Clin. Exp. Ophthalmol. 240, 942–946. Jeong, M., Kim, Y., Yi, N., Park, S., Kim, W., Chae, J., Kim, J., Lee, H. and Seo, K. 2007. Comparison of the rebound tonometer (Tonovet) with the applanation tonometer (Tonopen XL®) in normal Eurasian owls (Bubo bubo). Vet. Ophthalmol. 10, 376–379. Knollinger, A.M., La Croix, N.C., Barret, P.M. and Miller, P.E. 2005. Tonovet rebound tonometer for measuring intraocular pressure in dogs and horses. Invest. Ophthalmol. Vis. Sci. 46, 244–248. Komáromy, A.M., Garg, C.D., Ying, G.S. and Liu, C. 2006. Effect of head position on intraocular pressure in horses. Am. J. Vet. Res. 67, 1232–1235. Kontiola, A. 1997. A new electromechanical method for measuring intraocular pressure. Doc. Ophthalmol. 93, 265–276. Kontiola, A.I., Goldblum, D., Mittag, T. and Danias, J. 2001. The induction/impact tonometer: a new instrument to measure intraocular pressure in the rat. Exp. Eye. Res. 73, 781–785. Leiva, M., Naranjo, C. and Peña, M.T. 2006. Comparison of the rebound tonometer (Icare®) to the applanation tonometer (Tonopen XL®) in normotensive dogs. Vet. Ophthalmol. 9, 17–21. Linder, B.J., Trick, G.L. and Wolf, M.L. 1988. Altering body position affects intraocular pressure and visual function. Invest. Ophthalmol. Vis. Sci. 29, 1492–1497. Mcclure, J.R., Gelatt, K.N., Gum, G.G. and Manning, J.P. 1976. The effect of parenteral acepromazine and xylazine on intraocular pressure in the horse. Vet. Med. Small Anim. Clin. 32, 1727–1730. McDonald, J.E., Knollinger, A.M., Dustin-Dees, D., & MacLaren, N.E. 2017. Comparison of intraocular pressure measurements using rebound (TonoVet®) and applanation (TonoPen-XL®) tonometry in clinically normal alpacas (Vicugna pacos). Vet. Ophthalmol. 20, 155–159. Miller, P.E., Pickett, J.P. and Majors, L.J. 1991. Evaluation of two applanation tonometers. Am. J. Vet. Res. 52, 1917–1921. Miller, P.E., Pickett, J.P. and Majors, L.J. 2000. Evaluation of two applanation tonometers in horses. Am. J. Vet. Res. 51, 935–937. Moore, C. G., Milne, S. T. and Morrison, J. C. 1993. Noninvasive measurement of rat intraocular pressure with the Tono-pen. Invest. Ophthalmol. Vis. Sci. 34, 363–369. Park, Y.W., Jeong, M.B., Kim, T.H., Ahn, J.S. and Ahn, J.T. 2011. Effect of central cornea thickness on intraocular pressure using the rebound tonometer and the applanation tonometer in normal dogs. Vet. Ophthalmol. 14, 169–173. Pereira, F.Q., Bercht, B.S., Soares, M.G., Mota, M.G.B. and Pigatto, J.A.T. 2011. Comparison of rebound and applanation tonometer for measuring intraocular pressure in normal rabbits. Vet. Ophthalmol. 1, 321–326. Pigatto, J.A.T., Pereira, F.Q., Albuquerque, L., Dutra Corrêa, L.F., Bercht, B.S. and Hünning, P.S. 2011. Intraocular pressure measurement in sheep using an applanation tonometer. Ceres 58, 685–689. Rau, R., Silva, A.L.F., Salame, A.L.A., Junior, L.A.S.M. and Soares, L.A. 2012. Evaluation of the reproducibility of retinal nerve fiber layer thickness and optic nerve head parameters with optical coherence tomography. Oftalmol 71, 380–384. Rusanen, E., Florin, M., Hässig, M. and Spiess, B.M. 2013. Evaluation of a rebound tonometer (Tonovet®) in clinically normal cat eyes. Vet. Ophthalmol. 13, 31–36. Stiles, J., Buyukmih, N.C. and Farver, T.B. 1994. Tonometry of normal eye in raptors. Am. J. Vet. Res. 55, 477–479. Trim, C., Colbern, G. and Martin, C. 1985. Effect of xylazine and ketamine on intraocular pressure in horses. Vet. Rec. 117, 442–443. Van Der Woerdt, A., Gilbert, B.C., Wilkie, D.A. and Strauch, S.M. 1995. The effect of auriculopalpebral nerve block and intravenous administration of xylazine on intraocular pressure and corneal thickness in horses. Am. J. Vet. Res. 56, 155–158. Wada. 2006. Changes of intraocular pressure in uveitic horses. Equine Sci. 17, 67–73. Wilkie, D.A. and Gilger, B.C. 2004. Equine glaucoma. Vet. Clin. North. Am. Equine. Pract. 20, 381–391. Whitacre, M.M. and Stein, R. 1993. Sources of error with Goldmann-type tonometers. Surv. Ophthalmol. 38, 1–27. | ||
| How to Cite this Article |
| Pubmed Style Moreno TM, Fraga BLM, Petri SS, Tavares AM, Garcia PMC, Andrade MCCD, Hunning PS, Pereira FQ, Ghiggi E, Pigatto JAT. Repeatability and reproducibility of intraocular pressure measurements in horses using rebound and applanation tonometry. doi:10.5455/OVJ.2026.v16.i1.48 Web Style Moreno TM, Fraga BLM, Petri SS, Tavares AM, Garcia PMC, Andrade MCCD, Hunning PS, Pereira FQ, Ghiggi E, Pigatto JAT. Repeatability and reproducibility of intraocular pressure measurements in horses using rebound and applanation tonometry. https://www.openveterinaryjournal.com/?mno=283678 [Access: February 01, 2026]. doi:10.5455/OVJ.2026.v16.i1.48 AMA (American Medical Association) Style Moreno TM, Fraga BLM, Petri SS, Tavares AM, Garcia PMC, Andrade MCCD, Hunning PS, Pereira FQ, Ghiggi E, Pigatto JAT. Repeatability and reproducibility of intraocular pressure measurements in horses using rebound and applanation tonometry. doi:10.5455/OVJ.2026.v16.i1.48 Vancouver/ICMJE Style Moreno TM, Fraga BLM, Petri SS, Tavares AM, Garcia PMC, Andrade MCCD, Hunning PS, Pereira FQ, Ghiggi E, Pigatto JAT. Repeatability and reproducibility of intraocular pressure measurements in horses using rebound and applanation tonometry. doi:10.5455/OVJ.2026.v16.i1.48 Harvard Style Moreno, T. M., Fraga, . B. L. M., Petri, . S. S., Tavares, . A. M., Garcia, . P. M. C., Andrade, . M. C. C. D., Hunning, . P. S., Pereira, . F. Q., Ghiggi, . E. & Pigatto, . J. A. T. (2026) Repeatability and reproducibility of intraocular pressure measurements in horses using rebound and applanation tonometry. doi:10.5455/OVJ.2026.v16.i1.48 Turabian Style Moreno, Tatiana Martinez, Bárbara Letícia Machado Fraga, Sofia Silva Petri, Amannda Moreira Tavares, Paula Mariah César Garcia, Maria Cristina Caldart De Andrade, Paula Stieven Hunning, Fabiana Quartiero Pereira, Eduardo Ghiggi, and João Antonio Tadeu Pigatto. 2026. Repeatability and reproducibility of intraocular pressure measurements in horses using rebound and applanation tonometry. doi:10.5455/OVJ.2026.v16.i1.48 Chicago Style Moreno, Tatiana Martinez, Bárbara Letícia Machado Fraga, Sofia Silva Petri, Amannda Moreira Tavares, Paula Mariah César Garcia, Maria Cristina Caldart De Andrade, Paula Stieven Hunning, Fabiana Quartiero Pereira, Eduardo Ghiggi, and João Antonio Tadeu Pigatto. "Repeatability and reproducibility of intraocular pressure measurements in horses using rebound and applanation tonometry." doi:10.5455/OVJ.2026.v16.i1.48 MLA (The Modern Language Association) Style Moreno, Tatiana Martinez, Bárbara Letícia Machado Fraga, Sofia Silva Petri, Amannda Moreira Tavares, Paula Mariah César Garcia, Maria Cristina Caldart De Andrade, Paula Stieven Hunning, Fabiana Quartiero Pereira, Eduardo Ghiggi, and João Antonio Tadeu Pigatto. "Repeatability and reproducibility of intraocular pressure measurements in horses using rebound and applanation tonometry." doi:10.5455/OVJ.2026.v16.i1.48 APA (American Psychological Association) Style Moreno, T. M., Fraga, . B. L. M., Petri, . S. S., Tavares, . A. M., Garcia, . P. M. C., Andrade, . M. C. C. D., Hunning, . P. S., Pereira, . F. Q., Ghiggi, . E. & Pigatto, . J. A. T. (2026) Repeatability and reproducibility of intraocular pressure measurements in horses using rebound and applanation tonometry. doi:10.5455/OVJ.2026.v16.i1.48 |