| Research Article | ||
Open Vet. J.. 2026; 16(1): 256-264 Open Veterinary Journal, (2026), Vol. 16(1): 256-264 Review Article Renal protective effect of coenzyme Q10 against methotrexate-induced nephrotoxicity in ratsHiewa Othman Dyary*Department of Basic Sciences, College of Veterinary Medicine, University of Sulaimani, Sulaymaniyah, Iraq *Corresponding Author: Hiewa Othman Dyary. Department of Basic Sciences, College of Veterinary Medicine, University of Sulaimani, Sulaymaniyah, Iraq. Email: dyary.othman [at] univsul.edu.iq Submitted: 16/09/2025 Revised: 07/12/2025 Accepted: 17/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
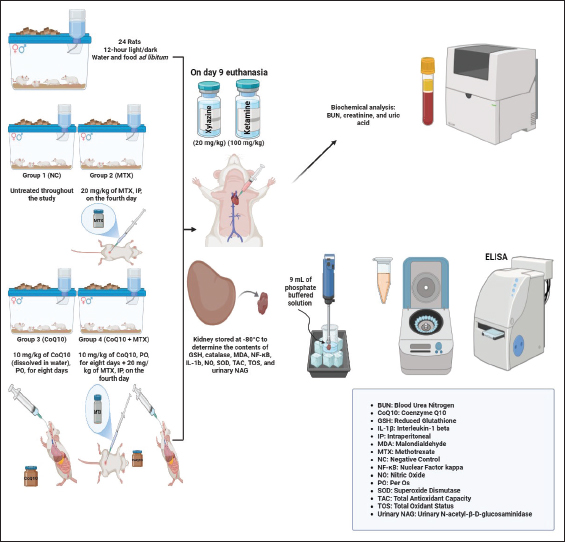
AbstractBackground: Methotrexate (MTX) is an effective chemotherapeutic and immunosuppressive agent; however, its clinical use is limited by nephrotoxicity, primarily due to oxidative stress (OS) and inflammation. Aim: Coenzyme Q10 (CoQ10) is a mitochondrial electron carrier with antioxidant and anti-inflammatory properties. It has been proposed as a potential agent against MTX-induced nephrotoxicity. This study assessed its renoprotective potential. Methods: Twenty-four Wistar rats were randomly divided into four equal groups: Group 1 (normal control) was left untreated, Group 2 (positive control) was intraperitoneally injected with MTX (20 mg/kg) on day 4 of the experiment, Group 3 (CoQ10) was orally administered CoQ10 (10 mg/kg) for 8 days, and Group 4 was administered CoQ10 + MTX. The following biomarkers were measured: OS biomarkers [glutathione (GSH), superoxide dismutase (SOD), malondialdehyde (MDA), total antioxidant capacity (TAC), total oxidant status (TOS), and nitric oxide (NO)], inflammatory markers (IL-1β, TNF-α, and NF-κB), and renal function markers [serum creatinine, uric acid, blood urea nitrogen (BUN), and urinary N-acetyl-β-D-glucosaminidase (NAG)]. Results: MTX induced nephrotoxicity, elevating BUN, creatinine, and uric acid levels, as well as urinary NAG, and increasing MDA, TOS, NO, IL-1β, TNF-α, and NF-κB, while reducing GSH, SOD, and TAC levels. In contrast, CoQ10 significantly reduced oxidative and inflammatory markers and increased the antioxidant capacity of renal tissue, even though it failed to restore values to baseline levels. CoQ10 showed significant renoprotective effects by suppressing OS and inflammatory pathways. Conclusion: These findings show that CoQ10 is a potential renoprotective antioxidant that could serve as an adjunct therapy to reduce MTX-associated renal injury. Keywords: Antioxidant, Coenzyme Q10, Drug-induced nephrotoxicity, Methotrexate, Oxidative damage. IntroductionMethotrexate (MTX) is an anticancer agent used to treat various malignancies, including acute lymphoblastic leukemia, osteosarcoma, and lymphoma (Hamed et al., 2022). MTX also has immunosuppressive effects for the treatment of autoimmune diseases, such as rheumatoid arthritis, inflammatory arthritis, juvenile idiopathic arthritis, and inflammatory bowel disease (van de Meeberg et al., 2023). Its clinical effectiveness is due to its dihydrofolate reductase inhibition, which impairs folic acid metabolism and disrupts DNA synthesis and cellular replication (Marin et al., 2022). Despite its many therapeutic applications, the use of MTX in therapy is associated with toxicities, including nephrotoxicity, which is crucial because the kidneys are the main organ for MTX excretion (Awad and Ali, 2021). MTX-induced nephrotoxicity is often dose-dependent and characterized by acute kidney injury (AKI) (Latcha et al., 2023), occurring in 2%–12% of patients (Howard et al., 2016). Nephrotoxicity occurs when MTX crystallizes in the renal tubules, causing injury to the epithelial cells. AKI causes several problems, such as treatment delays, dose reductions, or therapy discontinuation; hence, it adversely impacts patient outcomes (Liang et al., 2023). Nevertheless, high-dose MTX is life-saving and life-extending. Hence, reducing or preventing the adverse effects of MTX is a better approach than discontinuing it. MTX-induced nephrotoxicity is associated with oxidative stress (OS), inflammation, and mitochondrial dysfunction (Abdel-Wahab et al., 2023). Nephrotoxicity is manifested by decreased levels of superoxide dismutase (SOD), total antioxidant capacity (TAC), glutathione (GSH), and catalase (CAT), whereas the levels of TOS, malondialdehyde (MDA), and interleukin-6 are increased (Ozturk et al., 2022; Hassanein et al., 2023). Reactive oxygen species (ROS) are generated during MTX metabolism, leading to lipid peroxidation, DNA damage, and apoptotic pathway activation, thereby exacerbating tubular cell damage (Schmidt et al., 2022). Currently, several strategies are used to prevent and manage MTX-induced nephrotoxicity. These include high-volume intravenous hydration to maintain optimal renal perfusion and enhance drug elimination (Howard et al., 2016), urine alkalinization to increase MTX solubility in renal tubular fluid and reduce the risk of intratubular crystal precipitation, and leucovorin rescue therapy to mitigate folate antagonism in healthy, non-malignant cells (Awad and Ali, 2021). Despite these interventions, the risk of renal dysfunction remains, particularly in the context of high-dose MTX therapy (Liang et al., 2023). The nephrotoxic potential of MTX has motivated increasing scientific research to develop adjunct therapies to provide renal cytoprotection. Agents that can modulate the oxidative and inflammatory pathways, which significantly contribute to MTX-induced renal injury, are receiving considerable attention (Dar et al., 2021; Arab et al., 2022). Coenzyme Q10 (CoQ10), or ubiquinol, is an endogenous, isoprenoid-substituted benzoquinone that is a lipophilic constituent of cellular membranes, mostly in the inner mitochondrial membrane (Aaseth et al., 2021). It plays a vital role as an essential electron carrier in the mitochondrial respiratory chain, mediating electron transfer between complexes I and II to complex III during oxidative phosphorylation, thereby enhancing adenosine triphosphate (ATP) synthesis (Banerjee et al., 2022). When reduced, CoQ10 is a potent antioxidant that directly neutralizes lipid peroxyl radicals and regenerates other antioxidants, such as vitamin E, contributing remarkably to the cellular redox defense system (Abdulrahman et al., 2024). CoQ10 is an excellent organoprotective compound against multiple organ injuries, including AKI (Zhao et al., 2022). It is a well-known dietary and nutritional supplement with various functions, including antioxidant and anti-inflammatory effects, ATP production, and gene expression modulation (Keshavarzi et al., 2025). The renoprotective effects of CoQ10 in AKI are likely due to its antioxidant, anti-apoptotic, and anti-inflammatory properties, which collectively prevent cellular damage and support renal function (Sifuentes-Franco et al., 2022). Therefore, CoQ10 is a promising candidate compound with the potential to reduce MTX-induced nephrotoxicity. Accordingly, this study investigated its effect against experimental MTX-induced nephrotoxicity in a rat model. Materials and MethodsAnimals and their housingAlbino Wistar rats of both sexes, weighing 170–190 g with an average weight of 179.7 g at arrival, were used in the study. The rats were housed in 40 × 30 × 20 cm3 polypropylene cages and provided with water and food ad libitum. Sterilized wood shavings were used as the bedding for the cages and were changed as needed. The room temperature was approximately 24°C, ventilation was controlled, and a 12-hour light/dark cycle was followed. The rats were acclimated for 1 week before the start of the experiment. Treatment proceduresTwenty-four rats were randomly divided into four groups of six each, with no statistically significant differences in weight between the groups. Group 1 (normal control; NC) was left untreated throughout the study. Group 2 (positive control; PC) rats were intraperitoneally injected with MTX at 20 mg/kg on the fourth day of the experiment (Ozturk et al., 2022). The rats in Group 3 (CoQ10) were orally administered 10 mg/kg of CoQ10 (dissolved in water) by gavage for 8 days (El-Sheikh et al., 2012). Group 4 (CoQ10 + MTX) rats were administered CoQ10 at a rate of 10 mg/kg for 8 days and injected with 20 mg/kg MTX intraperitoneally on the fourth day of the experiment (Fig. 1). MTX and CoQ10 were purchased from Sigma-Aldrich (Germany), while ketamine HCl and xylazine HCl were purchased from NexGen Pharmaceuticals (USA).
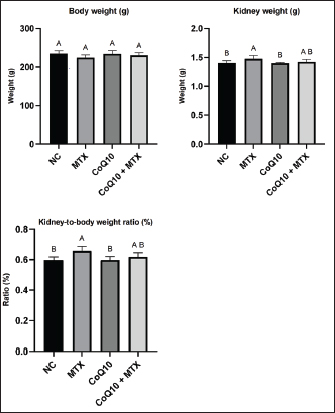
Fig. 1. The experimental design of the study. Animal euthanasia and blood and renal sample collectionOn day 9 of the experiment, each rat was weighed and euthanized by exsanguination after deep surgical anesthesia was induced by an intramuscular mixture of ketamine (100 mg/kg) and xylazine (20 mg/kg) (Mohammed-Rashid and Dyary, 2025). The chest cavity was opened, and blood was collected from the heart and placed into plain test tubes for serological analysis. The kidneys were soaked in normal saline, dried with sterile gauze, and weighed using a digital balance. The kidney-to-body weight (KBW) ratio was calculated as follows: KBW ratio=weight of both kidneys/Animal weight × 100. The kidney was homogenized in phosphate-buffered saline at a ratio of 1:9 and stored at −80°C for OS analysis, which involved measuring GSH, SOD, MDA, nuclear factor-kappa B (NF-κB), TAC, TOS, tumor necrosis factor-alpha (TNF-α), IL-1β, and nitric oxide (NO). Determination of biomarkers of renal functionSerum samples were centrifuged at 5,000 rpm for 15 minutes (Hettich Zentrifugen EBA20, Germany). The levels of blood urea nitrogen (BUN), uric acid (UA), and creatinine were measured using Cobas C 311 (Roche/Hitachi, Switzerland). Measurement of NAG urinary excretionUrine samples were collected over 24 hours before scarification of the rats using metabolic cages, and N-acetyl-β-D-glucosaminidase (NAG) activity was measured using the QuantiChrom™ β-N-Acetylglucosaminidase Assay Kit (Catalog No. DNAG-100, Bioassay Systems, USA) according to the manufacturer’s protocol. Analysis of renal OS and antioxidant biomarkersAn MDA ELISA Kit (Code: SH0020, Bioassay Technology Laboratory/China) was used to measure MDA in kidney tissue, an indicator of lipid peroxidation that serves as a marker of OS and cell membrane damage. A TAC Assay Kit (Code: SH0148, Bioassay Technology Laboratory/China) was used to test for TAC in kidney tissue. This test helps evaluate the ability of the kidneys to counteract OS by neutralizing free radicals. The total oxidant status (TOS) colorimetric assay kit (E-BC-K802-M; manufactured by Elabscience®) was used to measure the levels of oxidant molecules and the degree of OS in liver tissues. A Rat Glutathione, GSH ELISA Kit (Code EA0113Ra, Bioassay Technology Laboratory/China) was used to measure GSH, which is critical for antioxidant defense and detoxification. GSH assists in ROS neutralization, detoxifies harmful compounds, and protects against oxidative damage. A Rat Catalase ELISA Kit (Code: E0869Ra Bioassay Technology Laboratory/China) was used to measure CAT levels, which are crucial in the antioxidant defense system by changing hydrogen peroxide (H2O2) into H2O and O2, protecting against OS and damage. Moreover, a QuantiChrom™ Nitric Oxide Assay Kit (Catalog No. 50-489-194, Fisher Scientific) was used to measure renal tissue NO levels. All procedures were conducted according to the manufacturer’s instructions. Determination of IL-1β, TNF-α, and NF-κB in renal tissueA rat IL-1β ELISA kit (Code: E0119Ra, Bioassay Technology Laboratory/China) was used to measure IL-1β. Additionally, a TNF-alpha ELISA kit (Code: E0299Po; Bioassay Technology Laboratory/China) was used to measure TNF-α, an indicator of inflammation, cell death, and the immunological response. Moreover, a Rat NF-κB ELISA kit (Cat. No. E0287Ra, Imugex/Germany) was used to measure NF-κB activation level, a crucial factor in inflammation, immune response, OS, and disease progression. Statistical analysisThe data are shown as the mean ± SD. A one-way analysis of variance was used to test for overall differences, and Tukey’s post hoc test was used to identify significant differences between groups. Differences were analyzed as follows: A p-value of 0.05 was considered significant. GraphPad Prism software, version 10.5, was used for all statistical analyses. Ethical approvalThe study protocol was approved by the scientific and ethical committees of the College of Veterinary Medicine, University of Sulaimani (approval number AUP-2025). ResultsBody weight (BW) and kidney-to-BW ratiosOn day 9 of the experiment, the mean body weight of the rats in the NC group was 234.7 g, which was not significantly different from that of the other groups (p > 0.05; Fig. 2). The mean combined kidney weight in the NC group was 1.40 g, whereas the kidneys in the PC group weighed 5.7% more than those in the NC group, representing a statistically significant increase. Renal hypertrophy in the PC is probably an indication of AKI induced by MTX treatment. Kidney weights in the CoQ10-only treated group averaged 1.40 g, indicating that CoQ10 alone did not cause significant changes in kidney weight compared to the NC. Remarkably, the combined kidney weight in the CoQ10 + MTX-treated group averaged 1.42 g, suggesting that CoQ10 coadministration mitigated MTX-induced renal hypertrophy.
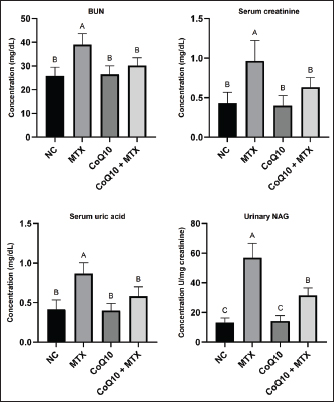
Fig. 2. The body weight, kidney weight, and KBW ratio in the various experimental groups. Values represent the mean (columns) ± SD (error bars) for six rats per group. Different uppercase letters indicate significant differences at p < 0.05. The KBW ratio provides a more concise indicator of changes in kidney size to account for body mass differences. The average KBW ratio of the NC was 0.598%, while that of the PC was 0.658%, reflecting a 10% increase compared with that of the NC (Fig. 2). Coadministration of CoQ10 resulted in a KBW ratio of 0.618%, which was not significantly different from that of the NC, supporting the notion that CoQ10 has a protective role against MTX-induced hypertrophy. BUN, creatinine, UA, and urinary NAGThe NC group exhibited a baseline BUN of 25.8 mg/dl, which was not significantly different from that of the CoQ10-only treated group (Fig. 3). In contrast, the PC group had a BUN level of 39.0 mg/dl, which was approximately 51.2% higher than that of the NC group, indicating renal stress or dysfunction. CoQ10 coadministration lowered the BUN level to 30.2 mg/dl, which was not significantly different from that of the NC group, highlighting the renoprotective effects of CoQ10.
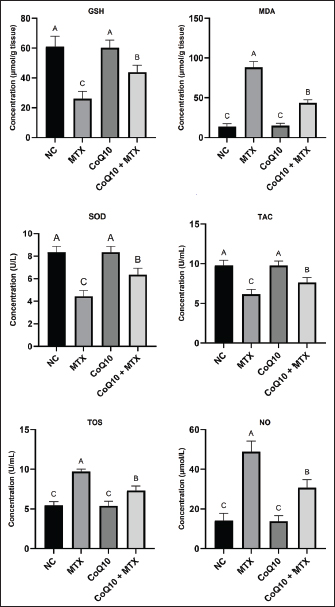
Fig. 3. BUN, creatinine, UA, and urinary NAG levels in the different experimental groups. Values are presented as mean (columns) ± SD (error bars) for six rats per group. Different uppercase letters indicate significant differences at p < 0.05. Serum creatinine levels in the NC group averaged 0.4 mg/dl, similar to those of the CoQ10-only group (0.4 mg/dl). In contrast, the PC group had a significantly higher concentration of 1.0 mg/dl than the other groups. This increase in serum creatinine levels in the PC indicates compromised renal function, even though the creatinine concentration did not reach toxic levels. Coadministration of CoQ10 lowered serum creatinine to 0.6 mg/dL, a value that was not significantly different from the NC (Fig. 3). These outcomes demonstrate that CoQ10 protects against MTX-induced renal impairment. Serum uric acid in the NC and CoQ10-only groups averaged 0.42 and 0.40 mg/dl, respectively, while the concentration significantly increased in the PC group to 0.87 mg/dl, probably reflecting reduced renal glomerular filtration rate. This finding further supports the notion that MTX induced nephrotoxicity in the PC group. Coadministration of CoQ10 resulted in a significantly lower uric acid concentration (0.58 mg/dl) compared to the PC group (Fig. 3), demonstrating CoQ10’s protective role against MTX-induced renal impairment. An elevated urinary NAG level, with a NAG concentration of 57.0 U/mg creatinine, is a marker of renal tubular injury, particularly proximal tubular injury, as observed in the PC group. This level was 4.35 times higher than the baseline NAG concentration of 13.1 and 14.2 U/mg creatinine in the NC and CoQ10-treated groups, respectively (Fig. 3). Co-treatment of MTX with CoQ10 in group 4 lowered the NAG concentration to 31.4 U/mg creatinine, which was significantly lower than that in the PC group, but it failed to return the concentration to baseline levels in the NC group. However, these findings highlighted the renoprotective impact of CoQ10 and demonstrated its mitigating effect on MTX-induced nephrotoxicity, probably through proximal tubular injury. Renal OS and antioxidant biomarkersThe average GSH level in the NC and CoQ10-treated groups was 60.9 and 60.2 µmol/g tissue, respectively, with no statistical differences between them. In contrast, the GSH concentration was reduced by 57.3% in the PC group, reaching 26.0 µmol/g (Fig. 4). This marked GSH depletion indicates severe OS induced by MTX exposure. Coadministration of CoQ10 with MTX in group 4 resulted in a GSH concentration of 43.8 µmol/g, which was significantly higher than that in the PC but lower than that in the NC. This result shows that CoQ10 partially reduced MTX-induced OS, although it was not sufficient to restore values to those of the NC.
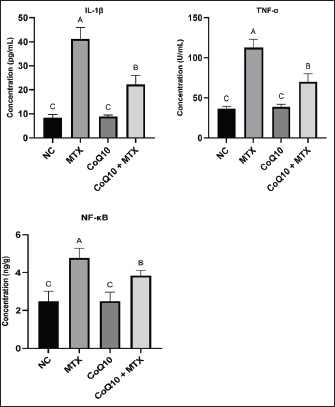
Fig. 4. GSH, MDA, SOD, TAC, TOS, and NO levels in the different experimental groups. Data are shown as mean (columns) ± SD (error bars) for six rats per group. Distinct uppercase letters indicate statistically significant differences at p < 0.05. Increased MDA levels in renal tissues are widely recognized as indicators of OS, lipid peroxidation, and potential nephrotoxicity. In this group, MDA concentrations were elevated 6.48-fold, rising from the baseline 13.6 µmol/g tissue in the NC to 88.1 µmol/g (Fig. 4). This remarkable increase in MDA in the PC group reflects potential oxidative damage to kidney tissue, indicating possible renal impairment. Coadministration of CoQ10 with MTX significantly reduced MDA concentrations to an average of 43.5 µmol/g, which was significantly lower than that of PC. However, the combination could not restore the MDA values to those observed in the NC group. Nevertheless, this finding reflects the potential of CoQ10 to reduce OS and lipid peroxidation and mitigate MTX-induced renal injury. SOD is an essential enzymatic antioxidant that catalyzes the dismutation of superoxide radicals into oxygen and H2O2. In this study, a lowered SOD concentration by 47.6% in the PC group from the baseline 8.4 U/l in the NC group to 4.4 U/l is an indication that MTX weakened the renal tissue’s capacity to neutralize ROS, leading to increased OS and potential renal injury. CoQ10 alone in group 3 did not alter SOD levels, whereas coadministration with MTX in group 4 increased the enzyme’s concentration to 6.4 U/l, significantly higher than that of the PC group but still lower than the baseline value in the NC group (Fig. 4). Altogether, these findings imply that CoQ10 mitigates MTX-induced OS, although it could not restore SOD enzymatic activity. The TAC of the renal tissue in the NC group averaged 9.8 U/ml, similar to that of the CoQ10-only group (Fig. 4). The treatment significantly reduced t by 36.7%, reaching 6.2 U/ml. Coadministration of CoQ10 with MTX elevated the TAC to 7.6 U/ml, which was significantly higher than that of the PC but still lower than the baseline value of the NC. These findings underscore the potential antioxidant effect of CoQ10 and its ability to counteract MTX-induced OS. The TOS in the PC group was higher than that in the NC group by 76.4%, indicating that the cumulative effect of ROS and other pro-oxidants was significantly higher in the MTX-only group than in the NC group (Fig. 4). In contrast, the TOS in the CoQ10 + MTX group averaged 7.3 U/ml, indicating that the combination lowered TOS levels by 24.7%, even though it failed to restore them to baseline levels observed in the NC group. This outcome further highlighted CoQ10’s potential antioxidant effect in mitigating MTX-induced OS. The NO levels were elevated about 3.4-fold from the baseline 14.2 µmol/l in the NC to 48.9 µmol/l in the PC group, indicating glomerulonephritis and drug-induced nephrotoxicity. Coadministration of CoQ10 remarkably reduced NO levels to 30.8 µmol/l (Fig. 4). This outcome further confirmed the antioxidant and renoprotective potential of CoQ10 in this study. IL-1β, TNF-α, and NF-κBThe levels of IL-1β, TNF-α, and NF-κB were significantly elevated in the PC group than in the other groups, suggesting increased inflammation and immune activation, which are typically associated with tissue injury and disease progression. Coadministration of CoQ10 with MTX in group 4 significantly reduced these values, although they did not return them to the baseline levels of the NC group (Fig. 5). These findings highlight the remarkable anti-inflammatory effect of CoQ10, although it failed to maintain the values within the baseline ranges.
Fig. 5. Levels of IL-1β, TNF-α, NF-κB, and NO in the different experimental groups. Data are shown as mean (columns) ± SD (error bars) for six rats per group. Distinct uppercase letters indicate statistically significant differences at p < 0.05. DiscussionAlthough MTX is a commonly used anticancer and immunosuppressive compound, nephrotoxicity limits its clinical applications (Koźmiński et al., 2020), especially at higher doses, primarily due to OS-mediated inflammation and apoptosis (Alum et al., 2023). In this study, MTX administration caused renal injury, demonstrated by increased serum creatinine, BUN, uric acid, and urinary NAG levels, decreased antioxidants (GSH, SOD, and TAC), increased lipid peroxidation (increased MDA), and activation of inflammatory mediators (increased IL-1β, TNF-α, and NF-κB). These changes were responsible for MTX-induced nephrotoxicity. MTX-induced renal toxicity is primarily associated with OS, starting with ROS generation upon its metabolism, followed by mitochondrial dysfunction (Heidari et al., 2018). In this study, MTX induced OS by elevating MDA and TOS levels, while remarkably suppressing major renal antioxidants, including GSH, SOD, and TAC. This outcome aligns with previous studies reporting an association between ROS generation and the depletion of the body’s antioxidant systems with MTX-induced toxicity (Dar et al., 2021; Ezhilarasan, 2021). Oxidative damage dysregulates cellular macromolecules, amplifies inflammation, and triggers apoptosis (Roghani et al., 2020), leading to renal injury. Coadministration of CoQ10 demonstrated significant renoprotective effects by restoring antioxidant systems, as evidenced by higher GSH, SOD, and TAC concentrations in renal tissues compared with the MTX-only group. GSH is the most abundant non-enzymatic antioxidant, essential for ROS detoxification and redox homeostasis (Abdulrahman et al., 2024). The 57% reduction in GSH in the positive control group demonstrates severe depletion of the cellular antioxidant pool. Similarly, SOD depletion indicates an impaired enzymatic defense against superoxide radicals, thereby accelerating mitochondrial damage and lipid peroxidation (Anwar et al., 2025). Elevated MDA levels further confirm extensive membrane damage, as MDA is a definitive marker of lipid peroxidation (Yekti et al., 2018). Increased TOS in the positive control group indicates a cumulative increase in total oxidant load, reflecting intensified OS on renal tissues (Palm and Nordquist, 2011). Although complete restoration of the antioxidant systems did not occur, these findings demonstrated the antioxidant potential of CoQ10. This outcome is consistent with the well-documented free radical-scavenging ability of CoQ10 (Cirilli et al., 2021). The reduction in MDA concentrations due to CoQ10 pretreatment further supports its role in preventing lipid peroxidation and membrane damage. The alterations observed in BUN, creatinine, uric acid, and urinary NAG provide important insight into the extent and nature of renal involvement in this model. Elevated BUN and creatinine levels reflect reduced glomerular filtration and early functional impairment, which commonly accompany drug-induced nephrotoxicity (Wu and Huang, 2018; Sharma and Singh, 2023). Changes in UA levels indicate disturbances in tubular handling and oxidative metabolism (Gherghina et al., 2022); UA often rises secondary to OS, impaired tubular secretion, or increased cellular turnover (El Ridi and Tallima, 2017). Urinary NAG is the most sensitive marker of proximal tubular injury (Spasovski, 2013). This lysosomal enzyme is released into urine following subclinical or early tubular damage (Novak et al., 2023), which often precedes measurable changes in serum creatinine. Therefore, the concurrent rise in these biomarkers shows both functional (glomerular) and structural (tubular) injury. Their improvement with treatment indicates a genuine renoprotective effect, likely mediated through OS and inflammation attenuation. A deeper analysis of these biomarkers strengthens the mechanistic interpretation of the findings and aligns with previous studies demonstrating the utility of BUN, creatinine, UA, and NAG as early and reliable indicators of renal toxicity and therapeutic response. CoQ10 significantly attenuated MTX-induced biochemical and histopathological alterations. However, it did not fully restore all parameters to baseline levels due to the multifactorial nature of the induced injury, in which OS is only one component of the overall pathological process (Kala and Howard, 2025). CoQ10 is mainly an antioxidant and mitochondrial stabilizer. However, the underlying pathway of MTX-induced nephrotoxicity involves additional mechanisms, including inflammatory signaling, membrane lipid peroxidation, impaired protein synthesis, and metabolic enzyme disruption (Algendy et al., 2023). CoQ10 alone may not fully correct these mechanisms. In addition to OS, inflammation plays a crucial role in MTX-induced nephrotoxicity. Significant increases in IL-1β, TNF-α, and NF-κB levels, which are key inflammation mediators, were observed in the PC group. These results are consistent with previous reports of the inflammatory-activating and renal-damaging effects of MTX (Dar et al., 2021). CoQ10 coadministration markedly reduced these inflammatory mediators, demonstrating its ability to suppress NF-κB signaling and the inflammatory cascade. However, the effect of CoQ10 is more suppressive than curative because it failed to restore inflammatory cytokine levels to baseline normal levels, as in the NC group. Excessive NO generation contributes to OS by forming peroxynitrite, a highly reactive oxidant (Piacenza et al., 2022). In this study, NO levels were also elevated in the PC group, indicating potential endothelial dysfunction and glomerular injury. CoQ10 reduced renal NO concentrations remarkably, further exhibiting its potential to modulate redox-sensitive pathways and prevent nitrosative damage. The molecular and biochemical findings highlighted CoQ10’s clear renoprotective potential. Coadministering CoQ10 with MTX significantly mitigated kidney hypertrophy, restoring the KBW ratio and renal biomarkers, although complete restoration did not occur. These outcomes show that CoQ10 may not be able to provide complete nephrotoxicity prevention when used as monotherapy. Moreover, this study employed a short experimental duration, and longer-term studies may provide a more accurate assessment of whether CoQ10 offers sustained protection against MTX-induced nephrotoxicity. A structured dose-response study or a longer-term treatment protocol should be considered in future work to better characterize the therapeutic window and intervention durability. Extending the exposure duration would help determine whether the observed protective effects can be sustained over time, while a dose-response approach could clarify the minimal effective dose and identify any plateau or ceiling effect in efficacy. Nevertheless, the findings of this study are noteworthy. Given that MTX is an indispensable anticancer and immunomodulator, adjuvant treatments, such as CoQ10, could help mitigate renal complications without compromising the therapeutic objectives of MTX. ConclusionCoQ10 remarkably reduced MTX-induced nephrotoxicity by improving antioxidant systems, reducing lipid peroxidation, inhibiting proinflammatory biomarkers, and restoring renal function markers. Although CoQ10 failed to restore all values to baseline, it showed potential as a supportive renoprotective compound in MTX-induced nephrotoxicity. More studies are needed to confirm its long-term renoprotective effect. AcknowledgmentsNot applicable. Conflicts of interestThe author declares no conflicts of interest. FundingThis research received no funding. Authors’ contributionsHOD conceptualized and conducted the research, analyzed the data, and wrote the manuscript. Data availabilityAll data supporting this study’s findings are available within the manuscript, and no additional data sources are required. ReferencesAaseth, J., Alexander, J. and Alehagen, U. 2021. Coenzyme Q10 supplementation–in ageing and disease. Mech. Ageing Dev. 197, 111521; doi:10.1016/j.mad.2021.111521 Abdel-Wahab, W.M., Daifalla, N.S. and Essawy, A.E. 2023. L-methionine protects against nephrotoxicity induced by methotrexate through modulation of redox status and inflammation. Redox. Rep. 28, 2270886; doi:10.1080/13510002.2023.2270886 Abdulrahman, H., Dyary, H., Mohammed, R., Hamad, D., Star, F. and Saeed, N. 2024. Preventing free radical damage: the significance of including antioxidants in diet to strengthen immunity. Open. Vet. J. 14(7), 1526–1537; doi:10.5455/OVJ.2024.v14.i7.2 Algendy, A., Mantawy, E., Tadros, M. and Helal, G. 2023. Methotrexate induced nephrotoxicity: possible underlying mechanisms and promising natural protective agents in experimental models. Arch. Pharm. Sci. Ain. Shams. Univ. 7, 457–469; doi:10.21608/aps.2023.232811.1134 Alum, E.U., Famurewa, A.C., Orji, O.U., Aja, P.M., Nwite, F., Ohuche, S.E., Ukasoanya, S.C., Nnaji, L.O., Joshua, D., Igwe, K.U. and Chima, S.F. 2023. Nephroprotective effects of Datura stramonium leaves against methotrexate nephrotoxicity via attenuation of oxidative stress-mediated inflammation and apoptosis in rats. Avicenna. J. Phytomed. 13, 377–387; doi:10.22038/AJP.2023.21903 Anwar, S., Sarwar, T., Khan, A.A. and Rahmani, A.H. 2025. Therapeutic applications and mechanisms of superoxide dismutase (SOD) in different pathogenesis. Biomolecules 15, 1130; doi:10.3390/biom15081130 Arab, H.H., Abd El-aal, S.A., Eid, A.H., Arafa, E.S.A., Mahmoud, A.M. and Ashour, A.M. 2022. Targeting inflammation, autophagy, and apoptosis by troxerutin attenuates methotrexate-induced renal injury in rats. Int. Immunopharmacol. 103, 108284; doi:10.1016/j.intimp.2021.108284 Awad, H. and Ali, U.F. 2021. Management of methotrexate toxicity. J. Adv. Biomed. Pharm. Sci. 4, 32–36; doi:10.21608/jabps.2020.44872.1106 Banerjee, R., Purhonen, J. and Kallijärvi, J. 2022. The mitochondrial coenzyme Q junction and complex III: biochemistry and pathophysiology. FEBS. J. 289, 6936–6958; doi:10.1111/febs.16164 Cirilli, I., Damiani, E., Dludla, P.V., Hargreaves, I., Marcheggiani, F., Millichap, L.E., Orlando, P., Silvestri, S. and Tiano, L. 2021. Role of coenzyme Q10 in health and disease: an update on the last 10 years (2010–2020). Antioxidants 10, 1325; doi:10.3390/antiox10081325 Dar, A., Fehaid, A., Alkhatani, S., Alarifi, S., Alqahtani, W., Albasher, G., Almeer, R., Alfarraj, S. and Moneim, A.A. 2021. The protective role of luteolin against the methotrexate-induced hepato-renal toxicity via its antioxidative, anti-inflammatory, and anti-apoptotic effects in rats. Hum. Exp. Toxicol. 40, 1194–1207; doi:10.1177/0960327121991905 El Ridi, R. and Tallima, H. 2017. Physiological functions and pathogenic potential of uric acid: a review. J. Adv. Res. 8, 487–493; doi:10.1016/j.jare.2017.03.003 El-Sheikh, A.A., Morsy, M.A., Mahmoud, M.M., Rifaai, R.A. and Abdelrahman, A.M. 2012. Effect of coenzyme‐Q10 on doxorubicin‐induced nephrotoxicity in rats. Adv. Pharmacol. Pharm. Sci. 2012, 981461; doi:10.1155/2012/981461 Ezhilarasan, D. 2021. Hepatotoxic potentials of methotrexate: understanding the possible toxicological molecular mechanisms. Toxicology 458, 152840; doi:10.1016/j.tox.2021.152840 Gherghina, M.E., Peride, I., Tiglis, M., Neagu, T.P., Niculae, A. and Checherita, I.A. 2022. Uric acid and oxidative stress—relationship with cardiovascular, metabolic, and renal impairment. Int. J. Mol. Sci. 23, 3188; doi:10.3390/ijms23063188 Hamed, K.M., Dighriri, I.M., Baomar, A.F., Alharthy, B.T., Alenazi, F.E., Alali, G.H., Alenazy, R.H., Alhumaidi, N.T., Alhulayfi, D.H., Alotaibi, Y.B., Alhumaidan, S.S., Alhaddad, Z.A., Humadi, A.A., Alzahrani, S.A. and Alobaid, R.H. 2022. Overview of methotrexate toxicity: a comprehensive literature review. Cureus 14, e29518; doi:10.7759/cureus.29518 Hassanein, E.H.M., Sayed, A.M., El-Ghafar, O.A.M.A., Omar, Z.M.M., Rashwan, E.K., Mohammedsaleh, Z.M., Kyung, S.Y., Park, J.H., Kim, H.S. and Ali, F.E.M. 2023. Apocynin abrogates methotrexate-induced nephrotoxicity: role of TLR4/NF-κB-p65/p38-MAPK, IL-6/STAT-3, PPAR-γ, and SIRT1/FOXO3 signaling pathways. Arch. Pharm. Res. 46, 339–359; doi:10.1007/s12272-023-01436-3 Heidari, R., Ahmadi, A., Mohammadi, H., Ommati, M.M., Azarpira, N. and Niknahad, H. 2018. Mitochondrial dysfunction and oxidative stress are involved in the mechanism of methotrexate-induced renal injury and electrolytes imbalance. Biomed. Pharmacother. 107, 834–840; doi:10.1016/j.biopha.2018.08.050 Howard, S.C., McCormick, J., Pui, C.H., Buddington, R.K. and Harvey, R.D. 2016. Preventing and managing toxicities of high-dose methotrexate. Oncologist 21, 1471–1482; doi:10.1634/theoncologist.2015-0164 Kala, J. and Howard, S.C. 2025. High-dose methotrexate nephrotoxicity. Am. J. Nephrol. 2025, 41134727; doi: 10.1159/000549144 Keshavarzi, M., Naraki, K., Razavi, B.M. and Hosseinzadeh, H. 2025. Ameliorative and protective effects of coenzyme Q10 against natural and chemical toxicity: a narrative review. Naunyn-Schmiedeberg’s. Archiv. Für. Experimentelle. Pathologie. Und. Pharmakologie. 398, 9827–9850; doi:10.1007/s00210-025-03992-5 Koźmiński, P., Halik, P.K., Chesori, R. and Gniazdowska, E. 2020. Overview of dual-acting drug methotrexate in different neurological diseases, autoimmune pathologies and cancers. Int. J. Mol. Sci. 21, 3483; doi:10.3390/ijms21103483 Latcha, S., Gupta, M., Lin, I.H. and Jaimes, E.A. 2023. High dose methotrexate-induced acute kidney injury: incidence, risk factors, and recovery. Kidney Int. Rep. 8, 360–364; doi:10.1016/j.ekir.2022.10.029 Liang, C.A., Su, Y.C., Lin, S.J. and Tsai, T.H. 2023. Risk factors for acute kidney injury after high-dose methotrexate therapy: a single-center study and narrative review. Eur. J. Clin. Pharmacol. 79, 789–800; doi:10.1007/s00228-023-03491-7 Marin, G.E., Neag, M.A., Burlacu, C.C. and Buzoianu, A.D. 2022. The protective effects of nutraceutical components in methotrexate-induced toxicity models—an overview. Microorganisms 10, 2053; doi:10.3390/microorganisms10102053 Mohammed-Rashid, S.S. and Dyary, H.O. 2025. Effect of catechin on hepatotoxicity induced by combined doxorubicin and paclitaxel treatment. J. Toxicol. 2025, 6775839; doi:10.1155/jt/6775839 Novak, R., Salai, G., Hrkac, S., Vojtusek, I.K. and Grgurevic, L. 2023. Revisiting the role of NAG across the continuum of kidney disease. Bioengineering 10, 444; doi:10.3390/bioengineering10040444 Ozturk, E., Karabulut, D., Akin, A.T., Kaymak, E., Kuloglu, N. and Yakan, B. 2022. Evaluation by different mechanisms of the protective effects of vitamin B12 on methotrexate nephrotoxicity. J. Mol. Histol. 53, 133–143; doi:10.1007/s10735-021-10027-9 Palm, F. and Nordquist, L. 2011. Renal oxidative stress, oxygenation, and hypertension. Am. J. Physiol. Regul. Integr. Comp. Physiol. 301, R1229–R1241; doi:10.1152/ajpregu.00720.2010 Piacenza, L., Zeida, A., Trujillo, M. and Radi, R. 2022. The superoxide radical switch in the biology of nitric oxide and peroxynitrite. Physiol. Rev. 102, 1881–1906. Roghani, M., Kalantari, H., Khodayar, M.J., Khorsandi, L., Kalantar, M., Goudarzi, M. and Kalantar, H. 2020. Alleviation of liver dysfunction, oxidative stress and inflammation underlies the protective effect of ferulic acid in methotrexate-induced hepatotoxicity. Drug. Des. Dev. Ther. 14, 1933–1941; doi:10.2147/DDDT.S237107 Schmidt, S., Messner, C.J., Gaiser, C., Hämmerli, C. and Suter-Dick, L. 2022. Methotrexate-induced liver injury is associated with oxidative stress, impaired mitochondrial respiration, and endoplasmic reticulum stress in vitro. Int. J. Mol. Sci. 23, 15116; doi:10.3390/ijms232315116 Sharma, V. and Singh, T.G. 2023. Drug induced nephrotoxicity-A mechanistic approach. Mol. Biol. Rep. 50, 6975–6986; doi:10.1007/s11033-023-08573-4 Sifuentes-Franco, S., Sánchez-Macías, D.C., Carrillo-Ibarra, S., Rivera-Valdés, J.J., Zuñiga, L.Y. and Sánchez-López, V.A. 2022. Antioxidant and anti-inflammatory effects of coenzyme Q10 supplementation on infectious diseases. Healthcare 10, 487; doi:10.3390/healthcare10030487 Spasovski, D. 2013. Renal markers for assessment of renal tubular and glomerular dysfunction. J. Nephropharmacol. 2, 23–25. Van De Meeberg, M.M., Hebing, R.C.F., Nurmohamed, M.T., Fidder, H.H., Heymans, M.W., Bouma, G., De Bruin‐weller, M.S., Tekstra, J., Van Den Bemt, B., De Jonge, R. and Bulatović Ćalasan, M. 2023. A meta‐analysis of methotrexate polyglutamates in relation to efficacy and toxicity of methotrexate in inflammatory arthritis, colitis and dermatitis. Br. J. Clin. Pharmacol. 89, 61–79; doi:10.1111/bcp.15579 Wu, H. and Huang, J. 2018. Drug-induced nephrotoxicity: pathogenic mechanisms, biomarkers and prevention strategies. Curr. Drug Metab. 19, 559–567; doi:10.2174/1389200218666171108154419 Yekti, R., Bukhari, A., Jafar, N. and Thaha, A.R. 2018. Measurement of malondialdehyde (MDA) as a good indicator of lipid peroxidation. IJAMSCR 6, 1–3. Zhao, S., Wu, W., Liao, J., Zhang, X., Shen, M., Li, X., Lin, Q. and Cao, C. 2022. Molecular mechanisms underlying the renal protective effects of coenzyme Q10 in acute kidney injury. Cell. Mol. Biol. Lett. 27, 57; doi:10.1186/s11658-022-00361-5 | ||
| How to Cite this Article |
| Pubmed Style Hiewa Othman Dyary. Renal protective effect of coenzyme Q10 against methotrexate-induced nephrotoxicity in rats. Open Vet. J.. 2026; 16(1): 256-264. doi:10.5455/OVJ.2026.v16.i1.23 Web Style Hiewa Othman Dyary. Renal protective effect of coenzyme Q10 against methotrexate-induced nephrotoxicity in rats. https://www.openveterinaryjournal.com/?mno=284325 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.23 AMA (American Medical Association) Style Hiewa Othman Dyary. Renal protective effect of coenzyme Q10 against methotrexate-induced nephrotoxicity in rats. Open Vet. J.. 2026; 16(1): 256-264. doi:10.5455/OVJ.2026.v16.i1.23 Vancouver/ICMJE Style Hiewa Othman Dyary. Renal protective effect of coenzyme Q10 against methotrexate-induced nephrotoxicity in rats. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 256-264. doi:10.5455/OVJ.2026.v16.i1.23 Harvard Style Hiewa Othman Dyary (2026) Renal protective effect of coenzyme Q10 against methotrexate-induced nephrotoxicity in rats. Open Vet. J., 16 (1), 256-264. doi:10.5455/OVJ.2026.v16.i1.23 Turabian Style Hiewa Othman Dyary. 2026. Renal protective effect of coenzyme Q10 against methotrexate-induced nephrotoxicity in rats. Open Veterinary Journal, 16 (1), 256-264. doi:10.5455/OVJ.2026.v16.i1.23 Chicago Style Hiewa Othman Dyary. "Renal protective effect of coenzyme Q10 against methotrexate-induced nephrotoxicity in rats." Open Veterinary Journal 16 (2026), 256-264. doi:10.5455/OVJ.2026.v16.i1.23 MLA (The Modern Language Association) Style Hiewa Othman Dyary. "Renal protective effect of coenzyme Q10 against methotrexate-induced nephrotoxicity in rats." Open Veterinary Journal 16.1 (2026), 256-264. Print. doi:10.5455/OVJ.2026.v16.i1.23 APA (American Psychological Association) Style Hiewa Othman Dyary (2026) Renal protective effect of coenzyme Q10 against methotrexate-induced nephrotoxicity in rats. Open Veterinary Journal, 16 (1), 256-264. doi:10.5455/OVJ.2026.v16.i1.23 |