| Research Article | ||
Open Vet. J.. 2026; 16(2): 1090-1111
Open Veterinary Journal, (2026), Vol. 16(2): 1090-1111 Research Article Determination of the optimal cisplatin dose to induce anemia in streptozotocin-nicotinamide-induced type 2 diabetic rats: A preliminary studyAgus Jati Sunggoro1,2,3*, Paramasari Dirgahayu1,4, Bambang Purwanto1,Eti Poncorini Pamungkasari1,5, Brian Warsita1,2,6, Risya Cilmiaty1, Damiana Tribhuwaneswari Mahendra Wardhani7 and Yohanna Handika Putri Kusuma Dewi71Doctoral Program in Medical Science, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia 2Dr.Moewardi General Hospital, Surakarta, Indonesia 3Division of Hematology and Medical Oncology, Department of Internal Medicine, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia 4Department of Parasitology and Micology, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia 5Department of Public Health, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia 6Department of Anatomical Pathology, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia 7Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia *Corresponding Author: Agus Jati Sunggoro. Doctoral Program in Medical Science, Faculty of Medicine, Universitas Sebelas Maret, Surakarta, Indonesia. Email: agusjati [at] staff.uns.ac.id Submitted: 19/09/2025 Revised: 01/01/2026 Accepted: 15/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
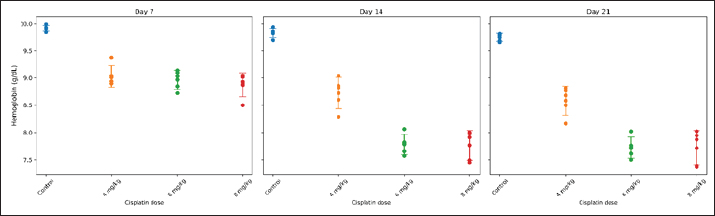
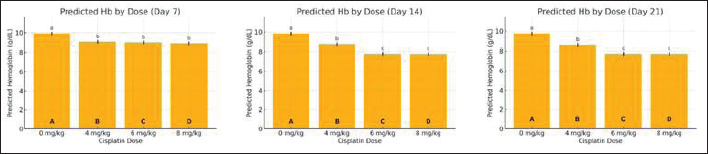
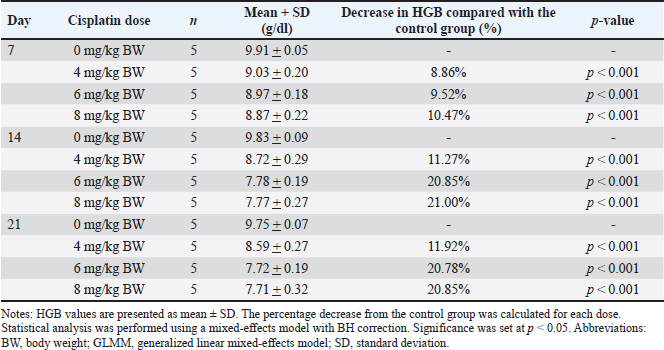
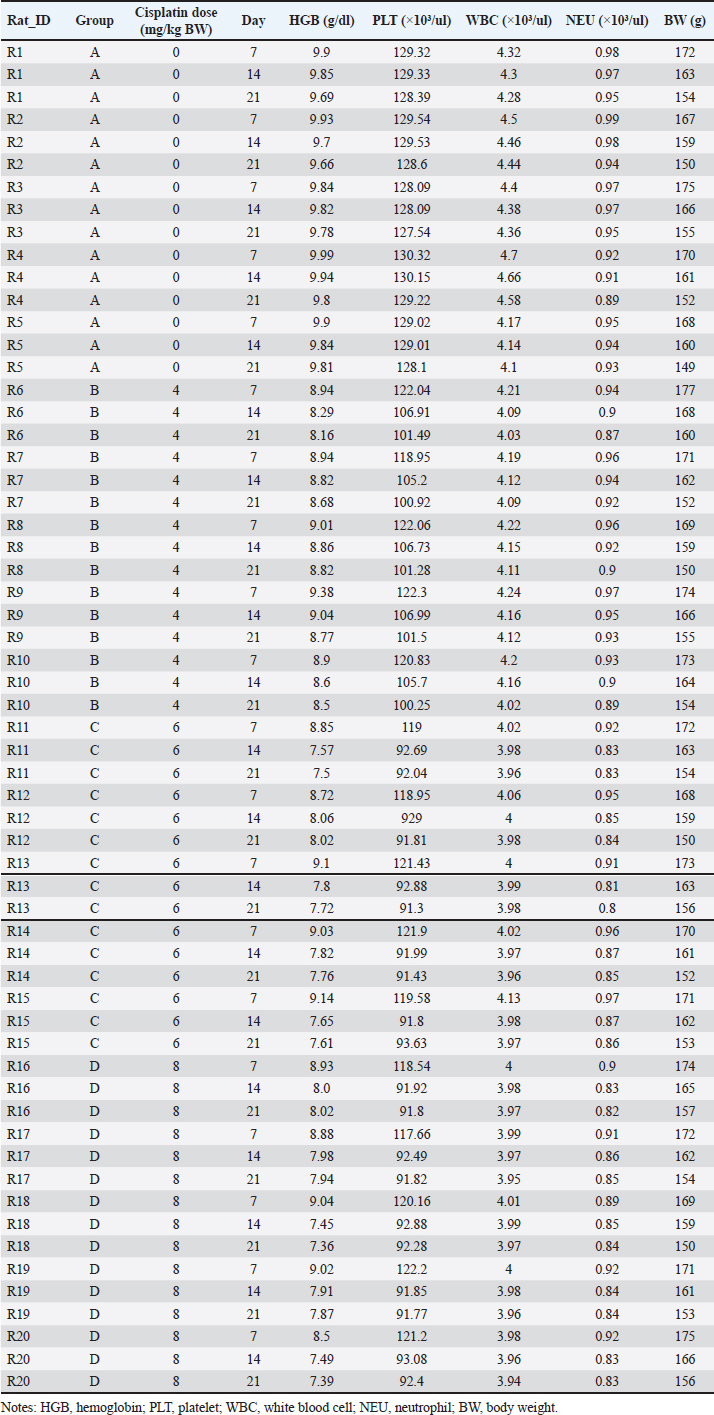
ABSTRACTBackground: Anemia is a major global health concern and a frequent complication of cancer chemotherapy. Patients with diabetes mellitus (DM) may experience chemotherapy-induced anemia (CIA), driven primarily by nephrotoxicity and impaired erythropoiesis. However, the interaction between pre-existing DM and CIA remains insufficiently understood, and preclinical models are limited. Aim: To determine the optimal single-dose, acute cisplatin regimen capable of inducing moderate anemia in a streptozotocin–nicotinamide type 2 diabetic Sprague Dawley rat model. Methods: Twenty male Sprague Dawley rats were induced to diabetes using nicotinamide [230 mg/kg body weight (BW), i.p.] followed by streptozotocin (65 mg/kg BW, i.v.). Diabetes was confirmed 72 hours later by fasting blood glucose ≥ 150 mg/dl, with repeat measurements performed to ensure stability before cisplatin administration. Diabetic rats were randomized into four groups (n=5 per group): control (saline) or a single IV injection of cisplatin at 4, 6, or 8 mg/kg BW. Hemoglobin (HGB), platelet (PLT), white blood cell (WBC), and neutrophil (NEU) counts were measured on days 7, 14, and 21 after injection. The optimal dose was defined as inducing >15% HGB reduction without mortality. Results: Cisplatin induced a significant dose-dependent reduction in HGB. The 4 mg/kg BW group developed mild anemia, whereas the 6 and 8 mg/kg BW doses produced moderate anemia (>15% HGB reduction) beginning on day 14 and maintained it through day 21 (≈20.8%–21.1%). All treated groups showed significant thrombocytopenia (decreased PLT count), leukocytosis (increased WBC count), and absolute neutrophilia (increased NEU count with increased WBC count). Weight loss (15.1%–16.2%) occurred across the groups, with the 6-mg/kg BW cohort exhibiting a less severe reduction than the 8-mg/kg BW group. No mortality occurred in any of the groups. Conclusion: A single dose of cisplatin (6 mg/kg BW) induces moderate, non-lethal CIA in diabetic Sprague Dawley rats while preserving tolerability. This preliminary study provides foundational dose-response data for developing diabetic-specific CIA models. However, additional validation incorporating comprehensive hematological and mechanistic assessments is required. Keywords: Erythropoietic suppression, Myelosuppression, Platinum compound toxicity, Preclinical anemia model, STZ-nicotinamide rat model. IntroductionAnemia is a condition characterized by an insufficient number of circulating erythrocytes to meet the body’s physiological oxygen demands. It is clinically diagnosed when hemoglobin (HGB) levels fall below established thresholds, which vary according to age, sex, and pregnancy status. As a significant global public health issue, anemia disproportionately affects adolescent girls, women of reproductive age (15–49 years), pregnant women, and children in low- and middle-income countries (World Health Organization [WHO], 2024). Historically, anemia severity was classified as mild, moderate, or severe based on HGB concentrations at >80%, 60%–80%, or <60% of the cutoff values, respectively (DeMaeyer et al., 1989). However, recent guidelines have revised these thresholds, indicating broader ranges—typically 85%–92% and 62%–70% of the cutoff for mild and severe anemia, respectively (WHO, 2024). Anemia is one of the most prevalent complications in patients with cancer, affecting 50%–65% of individuals at diagnosis and increasing to nearly 90% following treatment completion (Sharma et al., 2022). In clinical oncology, this condition is broadly categorized as either cancer-related anemia (a disease manifestation) or treatment-related anemia. Chemotherapy-associated anemia is a broad designation reflecting its multifactorial causes, which can include impaired erythropoiesis, nutritional deficits, hemolysis, or blood loss. However, this study focuses specifically on the direct impairment of erythropoiesis by chemotherapeutic agents, for which we employ the more precise term chemotherapy-induced anemia (CIA) (Madeddu et al., 2021). The abbreviation CIA is used throughout this manuscript to refer to this specific mechanism. Although most chemotherapeutics can cause CIA, platinum-based regimens such as cisplatin are particularly severe and prevalent (Madeddu et al., 2021). Studies indicate that 9%–40% of cancer patients treated with cisplatin develop anemia as a direct adverse effect (Sultana et al., 2022). Cisplatin-induced CIA is characterized by significant reductions in HGB levels, red blood cell counts, and other hematological parameters (Rajendrakumar et al., 2020). Its mechanism is hypoproliferative, resulting from the direct suppression of erythroid progenitor cells in the bone marrow and/or nephrotoxic effects that impair erythropoietin (EPO) production, ultimately decreasing HGB synthesis (Bryer and Henry, 2018; Madeddu et al., 2021). Anemia in patients with cancer has been linked to diminished treatment efficacy, elevated mortality risk, and an increased need for blood transfusions (Sharma et al., 2022). Understanding this specific mechanism is critical for developing targeted therapeutic strategies. In rat models, anemia is similarly defined by reduced HGB concentration and red blood cell mass, although reference values differ from those in humans. Normal HGB levels in male Sprague Dawley rats typically range from 14.7 to 16.6 g/dl (Frohlich, 2020). Experimental anemia in rats can be induced through various mechanisms, including phlebotomy, hemolytic agents (phenylhydrazine), iron deficiency, or myelosuppressive drugs such as cisplatin (Berger, 2007; Bunn, 2013). Cisplatin-induced anemia in rats mirrors the hypoproliferative anemia observed in human cancer patients, characterized by suppression of erythroid progenitors and impaired EPO production secondary to nephrotoxicity (Gao et al., 2006; Rajendrakumar et al., 2020). This pathophysiological similarity validates the use of rat models for investigating the mechanisms and potential therapeutic interventions of chemotherapy-induced anemia. The presence of comorbidities in patients with cancer undergoing chemotherapy may intensify adverse events and worsen clinical outcomes, including diabetes mellitus (DM). DM encompasses a spectrum of metabolic disorders characterized by chronic hyperglycemia, primarily caused by impaired insulin secretion (as in type 1 DM), insulin resistance [as in type 2 DM (T2DM)], or a combination of both (Schleicher et al., 2022). Mailliez et al. (2023)demonstrated that comorbid DM increases the risk of severe chemotherapy-related adverse events, with hematological toxicity (13%) being the second most common complication, of which anemia accounts for 10%. Notably, DM is the second most frequent comorbidity in patients with cancer before chemotherapy initiation, with reported prevalences of 22.6% (Cannavale et al., 2019) and 18% (Xu et al., 2016), following only hypertension. Cancer patients with pre-existing DM exhibit higher perioperative and long-term mortality rates than non-diabetic cancer patients. This disparity may be partly attributed to a shift in clinical focus from DM management to cancer treatment, leading to reduced attention to glycemic control following a cancer diagnosis (Pinheiro et al., 2019). The streptozotocin-nicotinamide (STZ-NA)-induced diabetic rat model is widely used in diabetes research, generating a type 2 diabetes phenotype characterized by insulin deficiency without insulin resistance and stable, moderate hyperglycemia (Furman, 2021). Although studies have investigated various cisplatin-induced effects, including antitumor activity, nephrotoxicity, hepatotoxicity, and other metabolic toxicities, in diabetic rat models, research on hematological toxicity—particularly CIA—remains limited (da Silva Faria et al., 2015; Bulduk et al., 2022). Establishing an optimal dose of cisplatin to induce CIA in diabetic rats is essential for developing a reliable preclinical model to investigate underlying mechanisms and potential therapeutic interventions. Previous studies in non-diabetic rats have reported cisplatin-induced anemia at doses of 4–8 mg/kg body weight (BW) (Gao et al., 2006; Aston et al., 2017); however, the appropriate dose for inducing CIA in diabetic models has not been determined. This study aimed to determine the optimal single dose of cisplatin capable of inducing moderate, nonlethal CIA in STZ–NA-induced type 2 diabetic rats, thereby establishing a reliable preclinical model for future mechanistic and interventional studies. Materials and MethodsChemicalsCisplatin (generic OGBferron, 50 mg/50 ml injection) was obtained from PT (Perseroan Terbatas) Ferron Par Pharmaceuticals (Bekasi, Indonesia). STZ–NA were obtained from Sangon Biotech Company Ltd. (Shanghai, China). Sodium chloride (0.9% injection) was obtained from PT. B. Braun Pharmaceutical (Karawang, Indonesia). Trisodium citrate dihydrate buffer (Emsure®) was obtained from Merck KGaA (Darmstadt, Germany). Ketamine HCl (generic OGBdexa, 100 mg/ml injection) was purchased from PT. Dexa Medica (Tangerang, Indonesia). Xylazine [Rompun®, 2% (20 mg/ml) injection] was purchased from Bayer HealthCare LLC (Kansas, USA). All other chemicals and reagents were of analytical grade. Animals and group assignmentThis preliminary dose-finding study employed n=5 rats per group, consistent with established protocols for pilot investigations of chemotherapy-induced hematotoxicity (Gao et al., 2006; Aston et al., 2017). Sample size adequacy was confirmed through post hoc power analysis, which demonstrated >99% power for detecting the observed HGB differences between groups. A total of 20 male Sprague Dawley rats (age, 2–3 months; weight, 150–200 g) were obtained from the Food and Nutrition Study Center Laboratory, Pusat Antar-Universitas (PAU) Building, Universitas Gadjah Mada. Prior to experimentation, the rats were acclimated for 1 week under controlled conditions: temperature (24°C ± 1°C), humidity (55% ± 5%), 12-hour light-dark cycles, and ad libitum access to food and water (Furman, 2021). Subsequently, all rats were precisely weighed to the nearest 1 g and randomly allocated into the following four experimental groups: • Group A: T2DM rats without cisplatin administration. • Group B: T2DM rats administered a single intravenous (IV) dose of cisplatin (4 mg/kg BW). • Group C: T2DM rats administered a single IV dose of cisplatin (6 mg/kg BW). • Group D: T2DM rats administered a single IV dose of cisplatin (8 mg/kg BW). All 20 rats were randomly allocated into four experimental groups (n=5 per group) using a computer-generated random number sequence (Microsoft Excel RAND function). Each rat was assigned a unique identification number, and a random number was generated for each animal. Rats were then randomly sorted and sequentially allocated to Groups A–D. This simple randomization procedure ensured unbiased group assignment and minimized the selection bias. An investigator (initials) who was not involved in subsequent outcome assessments performed group allocation. Induction of T2DM ratsT2DM rat models were induced following Basic Protocol 3 described by Furman (2021) with slight modification. Briefly, rats were weighed and then fasted for 6–8 hours (7:00 AM to 1:00–3:00 PM) with free access to water, followed by an intraperitoneal (IP) injection of nicotinamide (230 mg/kg BW) dissolved in 0.9% sodium chloride, administered using a 1 ml syringe with a 23 G needle. STZ (65 mg/kg BW) dissolved in 50 mM citrate buffer (pH 4.5) was administered IV via a 1 ml syringe with a 25 G needle after 15 minutes. On days 1 and 2 post-induction, the rats were monitored for diabetes-related conditions, including signs of hypoglycemia or hyperglycemia. On day 3, the rats were fasted again for 6–8 hours, weighed, and fasting blood glucose (FBG) levels were measured from tail vein blood samples using the Accu-Chek® Active blood glucose monitoring system. The day for FBG examination was modified from day 10 in Basic Protocol 3 by Furman (2021)to day 3, referring to the study by Cen et al. (2022)to shorten the research duration. Rats with FBG levels of >150 mg/dl (8.3 mmol/l) were confirmed as diabetic (Furman, 2021). Induction of dose-dependent cisplatin anemia in ratsRats in the treatment group were induced with anemia on day 4 of the experiment via a single IV injection of cisplatin at doses of 4, 6, or 8 mg/kg BW, according to their assigned treatment group. Cisplatin was prepared in sterile saline and injected through the tail vein immediately before administration. Subsequently, the rats were monitored for acute side effects, including dehydration and reduced activity. Control animals (Group A) received saline only. Hematological examination and surgical terminationHematological parameters were evaluated on days 7, 14, and 21 after cisplatin administration (corresponding to experimental days 11, 18, and 25 from study initiation), together with concurrent BW measurements. For clarity, unless otherwise specified, all subsequent references to observation days (days 7, 14, and 21) refer to days post-cisplatin administration. The assessed hematological indices included HGB, platelet count (PLT), white blood cell count (WBC), and neutrophil count (NEU). HGB concentration was selected as the primary hematological endpoint, consistent with the WHO diagnostic criteria for anemia (WHO, 2024) and established methodology in cisplatin-induced anemia studies (Gao et al., 2006). The red blood cell counts and erythrocyte indices were not assessed in this preliminary investigation. Baseline HGB (day 0) was not assessed because the study focused on relative differences from control animals at each time point rather than absolute pretreatment values. Before treatment, all rats were randomized, and no reductions in HGB were expected within the brief interval following diabetes induction alone. Thus, day 7 functions as a stable reference point for evaluating cisplatin-induced changes. Blood samples were obtained from the tail vein and transferred into ethylenediaminetetraacetic acid-coated microtubes for analysis using a veterinary automated hematology analyzer (Urit 3000 Plus, China). The sampled volume ranged from 0.5 to 1.0 ml per collection, which remains within the recommended safe limits for 150–200 g rats (total blood volume 6–8 ml), representing 10%–15% removal over a 2-week period as permitted by FELASA/NIH guidelines. All procedures were performed by trained personnel, and the animals were monitored for signs of distress or anemia; no adverse effects were observed. The final assessment was conducted on day 21 post-cisplatin administration (experimental day 25 from study initiation), a time point selected based on previous reports (Gao et al., 2006) indicating maximal cisplatin-induced anemia severity. After the final blood sampling, all animals were humanely euthanized by IP injection of ketamine (200 mg/kg BW) and xylazine (20 mg/kg BW), in accordance with the American Veterinary Medical Association (AVMA) Guidelines for the Euthanasia of Animals (American Veterinary Medical Association [AVMA], 2020). Determination of the optimal dose of cisplatinThe optimal dose of cisplatin was determined based on an internal operational criterion defined as producing >15% reduction in HGB concentration relative to the diabetic control group while maintaining mortality rates below 20%. This percentage-based threshold was selected to ensure clinically meaningful hematological impact while preserving animal welfare. This operational definition was developed specifically for dose selection purposes in this preliminary study and does not correspond to established clinical or preclinical anemia grading systems, which typically rely on absolute HGB values and erythrocyte indices. Data analysisStatistical analyses were performed using Python version 3.10. The normality of data distribution and homogeneity of variances were assessed using the Shapiro–Wilk and Levene’s tests, respectively (SciPy package). For the primary analysis of HGB levels across multiple groups and timepoints, a generalized linear mixed model (GLMM) was employed with cisplatin dose and observation day as fixed effects and rat ID as a random intercept to account for repeated measurements within subjects (StatsModels package). Post hoc pairwise comparisons between treatment groups at each timepoint were conducted using estimated marginal means (EMMs) derived from the GLMM. To control for multiple comparisons, the p-values were adjusted using the Benjamini–Hochberg (BH) procedure to limit the false discovery rate at 5%. Adjusted p-values <0.05 were considered statistically significant. For visualization, groups sharing the same lowercase letter within each timepoint indicate no statistically significant difference (BH-adjusted p ≥0.05), whereas groups sharing different lowercase letters indicate significant differences (BH-adjusted p < 0.05). This compact letter display method facilitates the interpretation of multiple pairwise comparisons (Piepho, 2004). Secondary outcomes (PLT count, WBC count, and NEU count) were analyzed using similar GLMM approaches with BH-adjusted post hoc comparisons. BW changes were analyzed using one-way analysis of variance (ANOVA) with Tukey’s honestly significant difference test for post hoc comparisons. Data are expressed as mean ± standard deviation (SD), and statistical significance was set at p < 0.05. Ethical approvalThis study was approved by the Research Ethics Committee of the Faculty of Medicine, Universitas Sebelas Maret (No: 129/UN27.06.11/KEP/EC/2025) and the Research Ethics Committee of the Faculty of Veterinary Medicine, Universitas Gadjah Mada (No: 68/EC-FKH/int./2025). Animal experiments were conducted at the Animal Research Facility Center for Food and Nutrition Studies PAU Building, Universitas Gadjah Mada. ResultsThis study used 20 Sprague–Dawley rats that were divided into four groups, each receiving a different treatment. Group A served as the control group and did not receive cisplatin during the study period. Cisplatin was administered at a dose of 4 mg/kg BW to group B, 6 mg/kg BW to group C, and 8 mg/kg BW to group D (Table S1). Characteristics of HGB in each groupIn this preliminary study, HGB levels were evaluated on days 7, 14, and 21 to assess the hematological effect of different single-dose cisplatin administrations. As shown in Figures 1 and 2, HGB concentrations tended to decrease in the cisplatin-treated groups compared with the control group. On day 7, the control group (0 mg/kg) showed the highest mean HGB level, whereas all cisplatin-treated groups (4, 6, and 8 mg/kg) exhibited significantly lower values (BH-adjusted p < 0.05). However, no significant differences were observed among the three cisplatin dose groups, as indicated by the shared letter “b.” By day 14, a clear separation was observed between the dose groups. The 6 and 8 mg/kg groups showed significantly lower HGB levels than both the control and the 4 mg/kg groups (different letters), whereas the 6 and 8 mg/kg groups did not significantly differ (same letter). On day 21, HGB levels remained lowest in the 6 and 8 mg/kg groups, which did not differ significantly from each other, but were significantly lower than both the control and 4 mg/kg groups. The control group consistently maintained the highest HGB concentration across all time points. Overall, the reduction in HGB levels differed significantly between groups beginning on Day 14 and remained significant on Day 21, particularly in the 6 and 8 mg/kg dose groups. HGB levels were measured on days 7, 14, and 21 after administration of a single dose of cisplatin across four dose groups (0, 4, 6, and 8 mg/kg). As shown in Table 1, a reduction in HGB was observed in all cisplatin-treated groups compared with the control group across all observation days. On day 7, the control group showed the highest mean HGB level (9.91 ± 0.05 g/dl), whereas the groups receiving 4, 6, and 8 mg/kg cisplatin demonstrated decreases of 8.86%, 9.52%, and 10.47%, respectively, compared with the control group. Although the reductions were modest at this early stage, a downward trend was evident with increasing cisplatin dose. By day 14, the decrease in HGB became more pronounced and dose-dependent. The mean HGB level in the control group was 9.83 ± 0.09 g/dl, whereas the 4 mg/kg group showed an 11.27% reduction, and the 6 and 8 mg/kg groups demonstrated larger reductions of 20.85% and 21.00%, respectively.
Fig. 1. Individual HGB values in STZ–NA-induced diabetic rats following single-dose cisplatin administration. Notes: Each dot represents an individual animal (n=5 per group). Horizontal markers indicate group means ± SD. HGB levels were measured on days 7, 14, and 21 after IV administration of cisplatin (0, 4, 6, or 8 mg/kg BW).
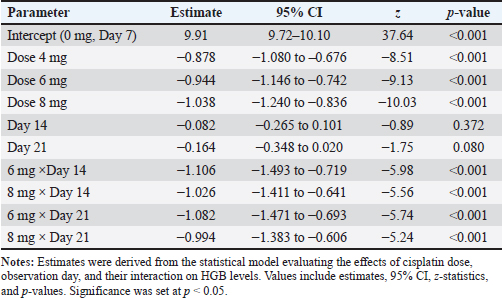
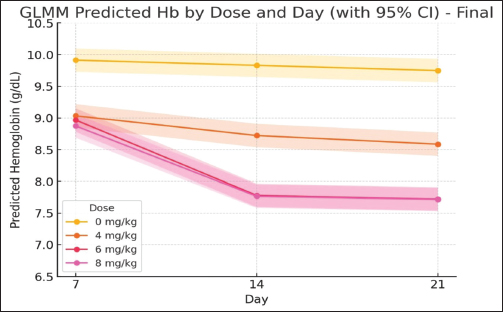
Fig. 2. EMMs of HGB levels across cisplatin dose groups on observation days 7, 14, and 21 in STZ–NA-induced diabetic rats. Notes: Bars represent the EMMs ± 95% confidence interval (CI) obtained from the statistical model. Bars sharing the same lowercase letter indicate no significant difference between the groups (BH-adjusted p ≥ 0.05). Different letters indicate statistically significant differences (p < 0.05). By day 21, the control group maintained HGB at 9.75 ± 0.07 g/dl, whereas the 6 and 8 mg/kg groups demonstrated HGB levels of 7.72 ± 0.19 and 7.71 ± 0.32 g/dl, respectively, representing reductions of approximately 21% from the diabetic controls. The control group consistently showed the highest HGB values across all time points, whereas the lowest levels were observed in the groups receiving 6 and 8 mg/kg cisplatin. Based on the predefined criteria for effective anemia induction—defined as a clinically relevant (>15%) reduction in HGB levels without exceeding acceptable mortality (<20%)—the 6 and 8 mg/kg groups met the criterion on both days 14 and 21. These findings indicate that 6 mg/kg cisplatin is an appropriate dose for reliably inducing moderate anemia in this experimental model. Table S2 presents the fixed-effects estimates of the GLMM. The dose of cisplatin showed a significant main effect on HGB levels, with reductions of −0.878, −0.944, and −1.038 g/dl for the 4, 6, and 8 mg/kg BW groups, respectively (all p < 0.001), compared with the reference group (0 mg/kg, day 7). These estimates indicate a clear dose-dependent decline in HGB concentration. In contrast, time alone (days 14 and 21) did not significantly affect HGB when the dose was held constant (p=0.372 and p=0.080, respectively), suggesting that CIA did not occur solely as a function of time. However, significant interaction effects were observed between dose and day. On day 14, the 6 and 8 mg/kg groups showed additional reductions of −1.106 and −1.026 g/dl, respectively (both p < 0.001), demonstrating that the decline in HGB was more pronounced at later time points in the higher-dose groups. A similar pattern was observed on day 21. These interaction terms confirm that the hematotoxic effect of cisplatin is dose-dependent and time-amplified, with the greatest reductions occurring in the 6 and 8 mg/kg BW groups at days 14 and 21. Notably, the similarity in the model estimates between the 6 mg/kg and 8 mg/kg BW groups at both days 14 and 21 supports the selection of 6 mg/kg BW as the optimal dose for inducing moderate anemia (>15% HGB reduction) while minimizing excessive toxicity. Table 1. HGB levels and percentage decrease across cisplatin dose groups and observation days in STZ–NA-induced diabetic rats.
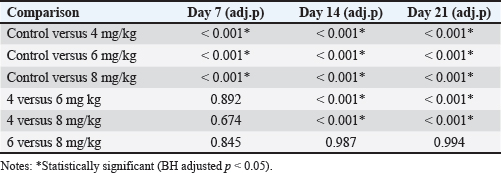
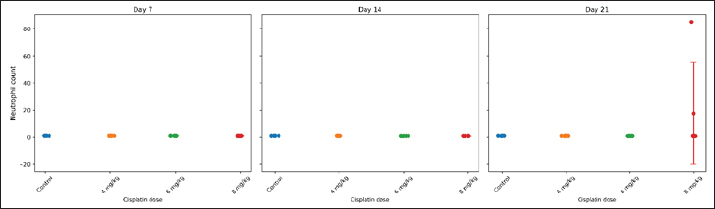
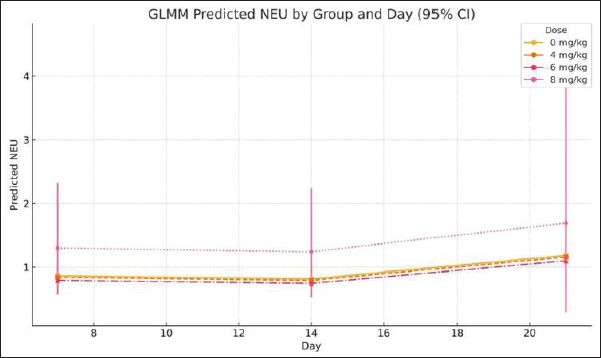
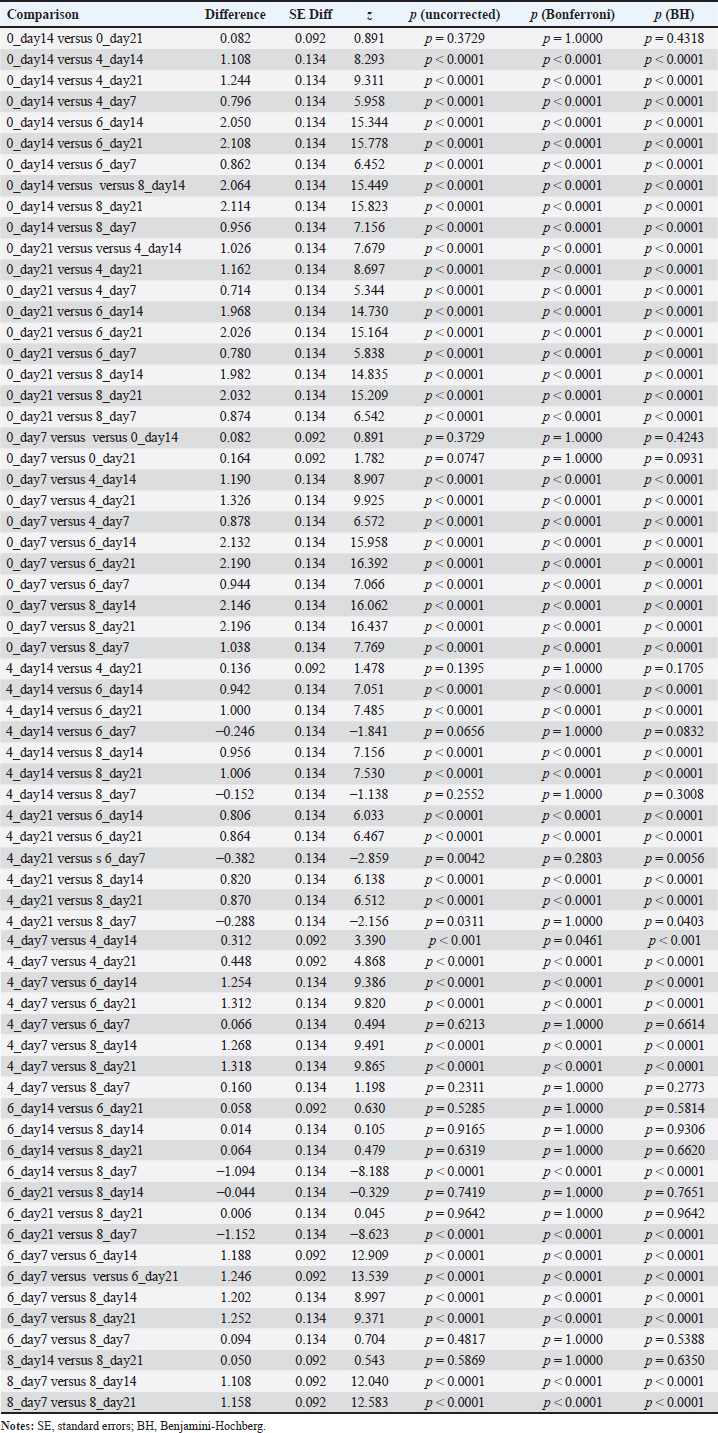
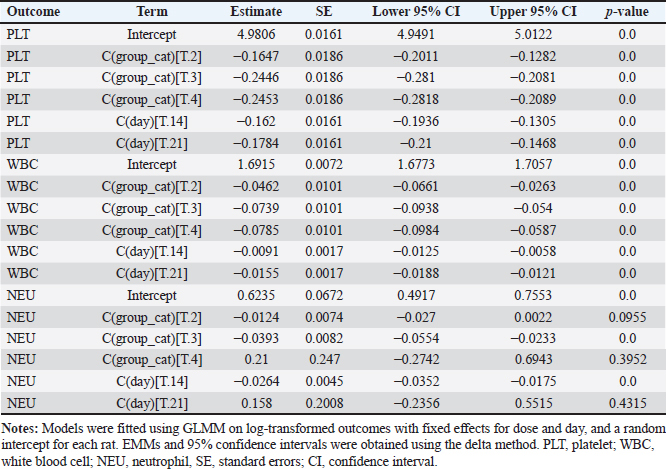
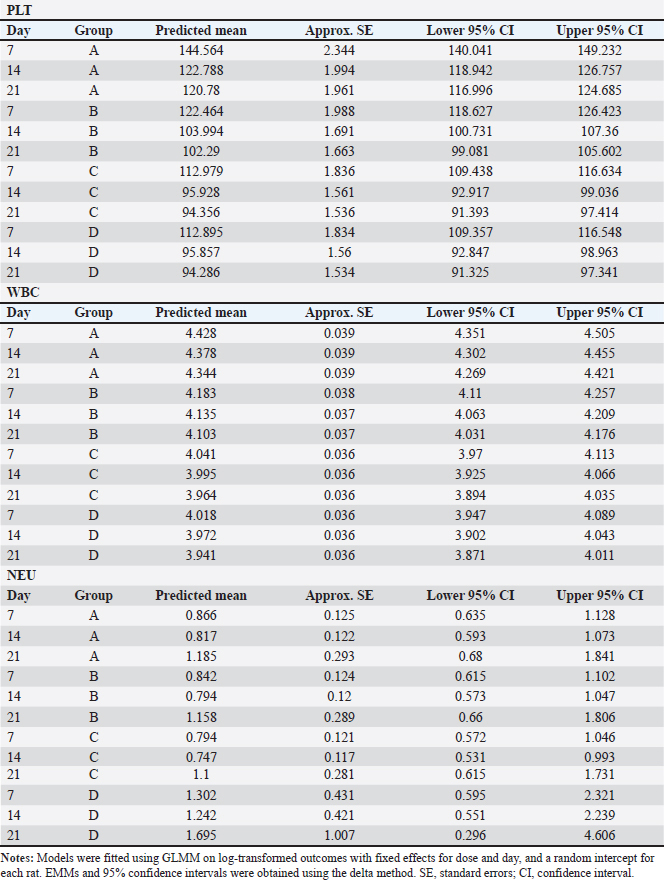
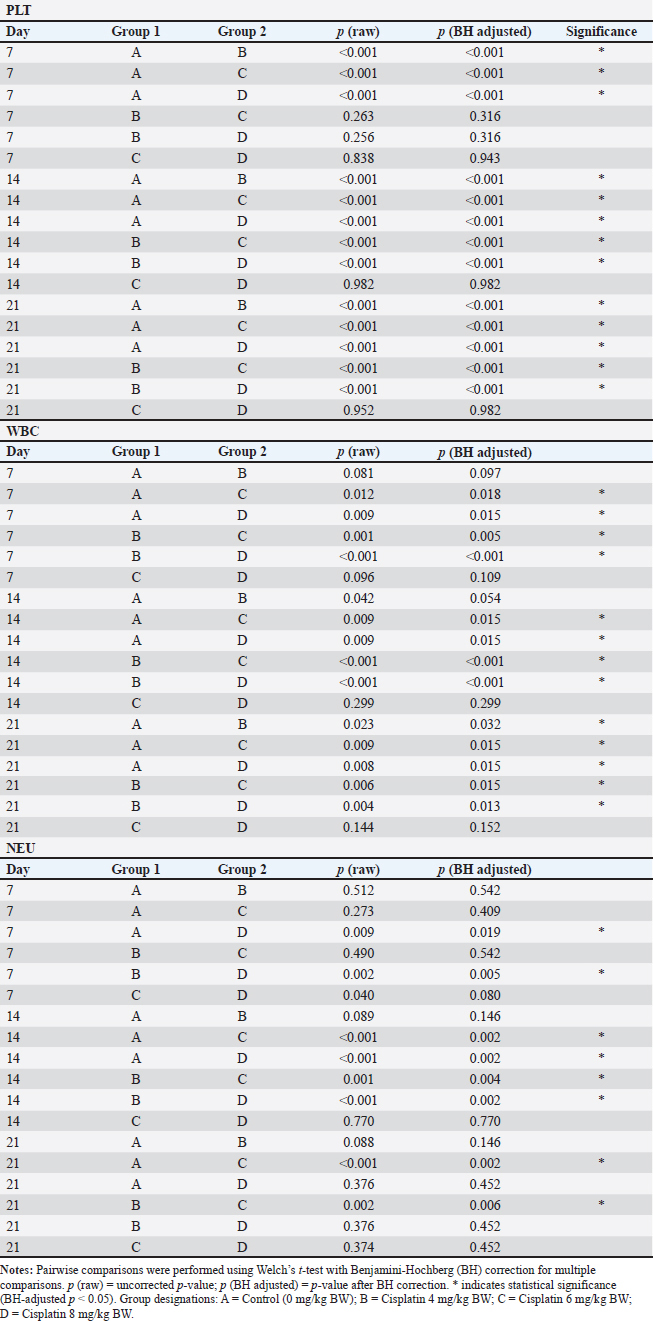
Comparison of HGB levels between the groupsHGB levels were compared across treatment groups at each observation timepoint using GLMM with BH adjusted post hoc pairwise comparisons. The overall trends are summarized below and presented in Figure 2, Table 1, and Figure S1, while the detailed pairwise comparisons and adjusted p-values for all dose groups and observation days are provided in Table S3. Day 7 post-cisplatin treatmentAll cisplatin-treated groups had significantly lower HGB levels than the diabetic control group (all adjusted p < 0.001). However, no significant differences were observed among the three cisplatin dose groups (4 vs. 6 mg/kg: adjusted p=0.892; 4 vs. 8 mg/kg: adjusted p=0.674; 6 vs. 8 mg/kg: adjusted p=0.845). Day 14 post-cisplatin treatmentA clear dose-dependent separation was observed. The control group (9.83 ± 0.09 g/dl) significantly differed from all treatment groups (all adjusted p < 0.001). The 4-mg/kg group (8.72 ± 0.29 g/dl) was significantly higher than both the 6-mg/kg (7.78 ± 0.19 g/dl; adjusted p < 0.001) and 8-mg/kg groups (7.77 ± 0.27 g/dl; adjusted p < 0.001). No significant difference was detected between the 6 and 8 mg/kg groups (adjusted p=0.987). Day 21 post-cisplatin treatmentThe pattern observed on day 14 was maintained. The control group (9.75 ± 0.07 g/dl) remained significantly higher than all cisplatin-treated groups (all adjusted p < 0.001). The 4 mg/kg group (8.59 ± 0.27 g/dl) showed significantly higher HGB than both the 6 mg/kg (7.72 ± 0.19 g/dl; adjusted p < 0.001) and 8 mg/kg groups (7.71 ± 0.32 g/dl; adjusted p < 0.001). The 6 and 8 mg/kg groups did not differ significantly (adjusted p=0.994). SummaryCisplatin induced a significant, dose-dependent reduction in HGB levels. The 6 and 8 mg/kg doses produced equivalent moderate anemia (approximately 21% reduction compared with the diabetic controls), meeting the predefined criterion of >15% HGB reduction. The similarity between these two doses supports the selection of the lower dose (6 mg/kg) as optimal to minimize potential toxicity while achieving the target hematological effect. Comparison of HGB levels between the groups is summarized in Table 2. Comparison of PLT, WBC, and NEU levels between groupsPLT, WBC, and NEU counts were analyzed using GLMMs, with cisplatin dose and day as fixed effects and rat ID as a random intercept to account for repeated measurements. Both dose and day exerted significant overall effects across all hematological parameters, indicating that the hematotoxic response to cisplatin was strongly dose-dependent and progressed over time. The fixed-effects estimates from these GLMMs, including the magnitude and significance of dose, day, and interaction terms for each parameter, are summarized in Table S4. Although interaction terms were explored, dose escalation and temporal changes consistently drove the primary effects. PlateletIndividual PLT counts following single-dose cisplatin administration are shown in Figure 3. GLMM demonstrated a pronounced and statistically significant main effect of cisplatin dose on PLT counts (p < 0.001), confirming a clear dose-dependent thrombocytopenia. Day was also significant (p < 0.001), reflecting progressive PLT suppression over the study period. Analysis of EMMs indicated uniformly lower PLT values in all treatment groups compared to controls; higher doses and later time points were associated with deeper nadirs (Fig. 4). BH–adjusted pairwise comparisons indicated that the control group differed significantly from all cisplatin-treated groups at nearly every day of assessment. The full EMM and pairwise comparison results are provided in Supplementary Tables S5 and S6. WBC countIndividual WBC counts following single-dose cisplatin administration are shown in Figure 5. WBC counts exhibited a strong main effect of dose (p < 0.001), characterized by a gradual, dose-dependent elevation following cisplatin administration. Day also contributed significantly (p < 0.05), indicating progressive leukocyte mobilization over time. EMM trajectories showed that the 6- and 8-mg groups consistently displayed higher WBC levels compared with controls, particularly on days 14 and 21 (Fig. 6). Post hoc tests confirmed significant increases in these higher-dose groups (-adjusted p < 0.05). Supplementary Tables S5 and S6 present detailed comparisons. NeutrophilIndividual NEU counts following single-dose cisplatin administration are shown in Figure 7. The NEU counts demonstrated significant effects of dose (p < 0.01) and time, along with a meaningful dose × day interaction (p < 0.01). Higher doses (6 and 8 mg) produced marked neutrophilia, which became particularly evident on days 14 and 21. On day 7, only the highest dose (8 mg) significantly differed from the controls; however, by days 14 and 21, both the 6 mg and 8 mg groups exhibited robust NEU elevations relative to the control animals (BH-adjusted p < 0.01) (Fig. 8). The full post hoc results are shown in Supplementary Tables S5 and S6. Table 2. Comparison of HGB levels between the groups.
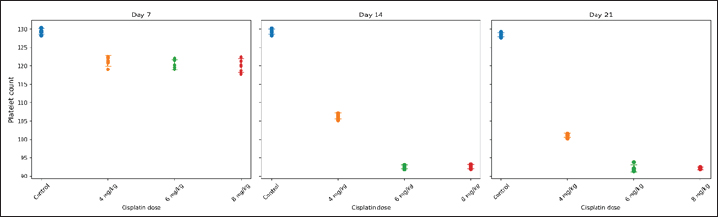
Fig. 3. Individual PLT count values in STZ–NA-induced diabetic rats following single-dose cisplatin administration. Notes: Each dot represents an individual animal (n=5 per group). Horizontal markers indicate group means ± SD. Measurements were obtained on days 7, 14, and 21 after cisplatin administration.
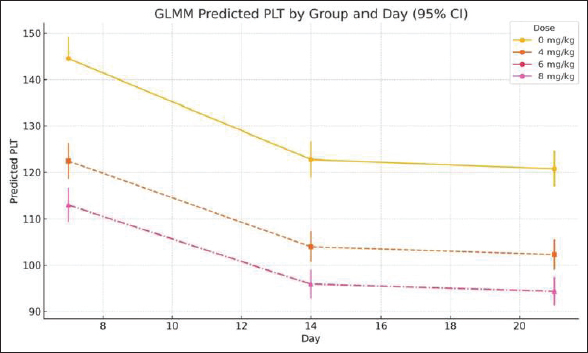
Fig. 4. EMMs of PLT counts (×10³/μl) across cisplatin dose groups and observation days (GLMM, mean ± 95% CI). Notes: GLMM, generalized linear mixed-effects model; PLT, platelet; CI, confidence interval.
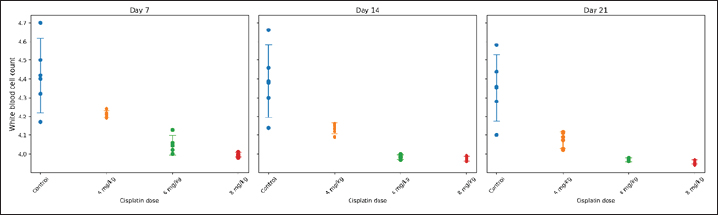
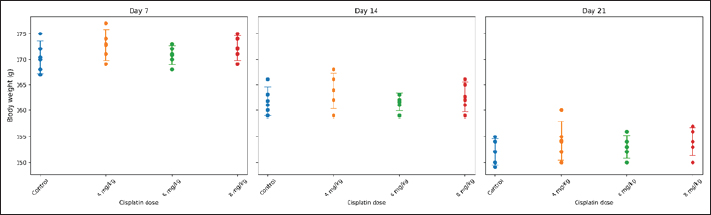
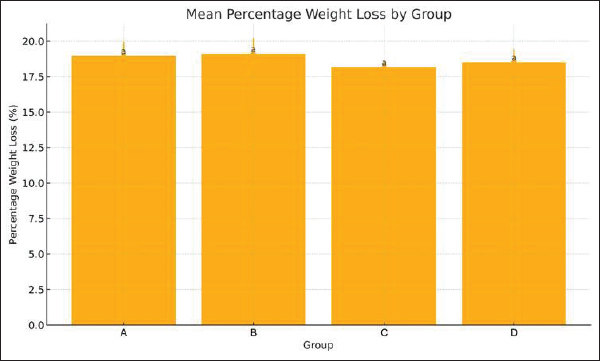
Fig. 5. Individual WBC counts in STZ–NA-induced diabetic rats following single-dose cisplatin administration. Notes: Each dot represents an individual animal (n=5 per group). Horizontal markers indicate group means ± SD. Measurements were obtained on days 7, 14, and 21. Percent weight loss of rats per groupIndividual BW values following single-dose cisplatin administration are shown in Figure 9. The mean BW loss ranged from 18.18% to 19.11% across all groups. Although Group B (DM + 4 mg/kg cisplatin) showed the greatest weight reduction (19.11% ± 0.87%), followed by Group A (DM control, 18.76% ± 0.62%), Group D (DM + 8 mg/kg, 18.42% ± 0.71%), and Group C (DM + 6 mg/kg, 18.18% ± 0.54%), one-way ANOVA revealed no statistically significant differences among groups (F=1.099, p=0.378) (Fig. 10). Post-hoc pairwise comparisons confirmed that the difference between the 4 and 6 mg/kg groups was not statistically significant (mean difference=0.93%, p=0.412). The narrow range of weight loss across all groups (< 1% difference between the highest and lowest) indicates comparable systemic tolerability regardless of cisplatin dose. DiscussionThis study successfully established a reliableSTZ–NA-induced type 2 diabetic rat model for investigating CIA. We demonstrated that a single 6-mg/kg BW dose of cisplatin produces moderate, non-lethal anemia with good systemic tolerability. This dose provides an optimal balance between hematotoxic efficacy, reproducibility, and animal welfare, making it a suitable model for future mechanistic and interventional studies.
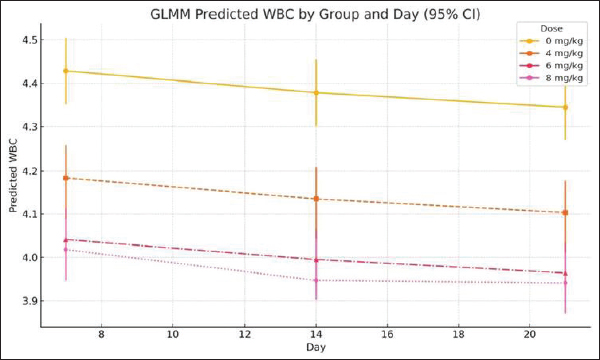
Fig. 6. EMMs of WBC counts (×10³/μl) across cisplatin dose groups and observation days (GLMM, mean ± 95% CI). Notes: GLMM, generalized linear mixed-effects model; WBC, white blood cell; CI, confidence interval.
Fig. 7. NEU counts in STZ–NA-induced diabetic rats following single-dose cisplatin administration. Notes: Each dot represents an individual animal (n=5 per group). Horizontal jitter was applied solely for visualization to prevent datapoint overlap. Horizontal markers indicate group means ± SD. Measurements were obtained on days 7, 14, and 21. Cisplatin-induced anemia and dose–time interactionHGB levels progressively declined in all cisplatin-treated groups, with significant and clinically relevant reductions (>15%) observed in the 6- and 8-mg/kg BW groups by day 14. Statistical analysis revealed that the cisplatin dose exerted a significant main effect on HGB reduction, whereas time alone was not associated with anemia progression. The significant interaction between dose and time indicates that hematological toxicity was not only dose-dependent but also amplified over time. These findings support a model in which anemia develops through cumulative hematotoxic effects rather than an immediate suppression of erythropoiesis after exposure. Cisplatin-induced anemia is primarily hypoproliferative, driven by direct suppression of erythroid precursors and impaired EPO synthesis due to nephrotoxicity (Bryer and Henry, 2018; Madeddu et al., 2021). This effect may be compounded in diabetic animals, considering that diabetic nephropathy also reduces the production of EPO (Arkew et al., 2023). The reduced baseline HGB in our diabetic control group (~9.75 g/dl), which was approximately 34% below the normal rat reference values (14.7–16.6 g/dl; Frohlich, 2020), is consistent with reports of hematological alterations in STZ-induced diabetic models, including reductions in packed cell volume, mean corpuscular HGB, and related erythrocyte indices (Fagbohun et al., 2020; Rehman et al., 2023). This diabetes-associated anemia may be attributed to chronic inflammation, impaired EPO production, increased oxidative stress, and non-enzymatic red blood cell glycosylation (Barbieri et al., 2015). However, previous studies have shown a paradoxical attenuation of cisplatin nephrotoxicity in diabetic models, possibly due to the downregulation of organic cation transporter 2, which limits the tubular uptake of cisplatin (Xu et al., 2017; Saito et al., 2022). This indicates that diabetic conditions may modulate cisplatin toxicity through competing renal mechanisms, underscoring the importance of developing diabetic-specific CIA models.
Fig. 8. EMMs of NEU counts (×10³/μl) across cisplatin dose groups and observation days (GLMM, mean ± 95% CI). Notes: GLMM, generalized linear mixed-effects model; NEU, neutrophil; CI, confidence interval.
Fig. 9. Individual BW values of STZ–NA-induced diabetic rats following single-dose cisplatin administration. Notes: Each dot represents an individual animal (n=5 per group). Horizontal markers indicate group means ± SD. BW was measured on days 7, 14, and 21 after IV cisplatin administration (0, 4, 6, or 8 mg/kg BW). Contextualizing the severity of anemia achieved in this model is important. Although our operational criterion defined “moderate anemia” as >15% HGB reduction from diabetic controls, the absolute HGB values observed in the 6 and 8 mg/kg groups by day 21 (approximately 7.7 g/dl) warrant careful interpretation. Reference HGB values for healthy adult male Sprague Dawley rats typically range from 14.7 to 16.6 g/dl (Frohlich, 2020). The reduced baseline HGB level in our diabetic control group (~9.75 g/dl) is consistent with the mild anemia commonly observed in the STZ-nicotinamide model (Arkew et al., 2023). The absolute HGB levels achieved in the cisplatin-treated groups correspond to moderate-to-severe anemia in rat physiology relative to normal rat reference ranges. Therefore, researchers utilizing this model should recognize that the term “moderate anemia,” as used in this study, refers strictly to our internal operational definition based on the percentage reduction from diabetic controls, rather than the absolute severity classifications used in standard hematological or toxicological frameworks. This distinction is critical for accurate interpretation and cross-study comparisons.
Fig. 10. Mean BW loss percentage in diabetic rats after 21 days of cisplatin administration. Notes: Values are expressed as mean ± SD, with n=5 rats per group. No statistically significant differences were found among dose groups (p=0.378). Groups sharing the same lowercase letter were not significantly different (BH adjusted p ≥ 0.05). Different letters indicate statistically significant differences (adjusted p < 0.05). Hematological alterations beyond HGB levelIn addition to anemia, cisplatin administration resulted in thrombocytopenia, leukocytosis, and neutrophilia, particularly in the 6 and 8 mg/kg BW groups. These findings are consistent with those of prior studies describing chemotherapy-induced myelosuppression, inflammatory activation, and bone marrow dysregulation (Finnigan et al., 2025; Zhang et al., 2025). The neutrophilic response observed in this acute, single-dose model likely represents an early inflammatory phase rather than the delayed neutropenia that is more typical in chronic or multi-cycle chemotherapy regimens (Yousif et al., 2021). The hematological profile obtained in this model closely reflects the clinical spectrum of CIA, which strengthens its translational relevance. Changes in BW and systemic tolerabilityA reduction in BW was observed across all groups, including the diabetic controls, which may primarily reflect the catabolic effects of diabetes, procedural stress associated with repeated blood sampling, or reduced food intake during the experimental period. The non-monotonic pattern of weight loss—wherein the 4 mg/kg group exhibited numerically greater weight reduction than the 6 and 8 mg/kg groups—was unexpected given the typical dose-dependent toxicity profile of cisplatin (Malik et al., 2006). However, this difference was not statistically significant (p=0.378), and the absolute differences between the groups were minimal (< 1%). Several factors, including variable metabolic responses to cisplatin, individual differences in diabetes severity affecting baseline metabolism, and the confounding catabolic effects of the diabetic state, may account for this observation in diabetic animals (Basu et al., 2025; López-Tofiño et al., 2025). Importantly, the comparison between the 6 and 8 mg/kg groups is most clinically relevant for optimal dose selection, as both doses achieved the target hematological effect (>15% HGB reduction). In this critical comparison, the 6 mg/kg dose demonstrated comparable weight loss to the 8 mg/kg dose while producing equivalent anemia induction with less variability in hematological parameters. This supports the selection of 6 mg/kg as the optimal biological dose to achieve the desired hematotoxic effect while maintaining acceptable systemic tolerability. The absence of significant weight loss differences across dose groups further suggests that cisplatin-induced systemic toxicity did not substantially differ within the tested dose range, and factors related to the diabetic condition may have been the predominant contributors to BW changes. Rationale for determining optimal doseAlthough both the 6- and 8-mg/kg BW doses induced moderate anemia, the 6-mg/kg BW dose was deemed optimal due to its hematological consistency, limited systemic stress (as indicated by BW maintenance), and narrower confidence intervals in the GLMM estimates. The dose of 8 mg/kg BW likely approaches the maximum tolerated dose (MTD), where excessive systemic toxicity and biological variability may obscure the drug-specific effects. In contrast, the 6 mg/kg BW dose serves as an optimal biological dose (OBD), producing a reproducible level of anemia suitable for mechanistic, pharmacological, or protective intervention studies. Translational considerations and generalizabilityThe generalizability of these findings to human diabetic populations and various cancer types warrants careful consideration. Several factors support this model’s potential translational relevance. First, the STZ-nicotinamide diabetic rat model recapitulates the key features of human type 2 diabetes, including moderate hyperglycemia, partial insulin deficiency, and preserved β-cell function (Furman, 2021). However, this model does not fully replicate the insulin resistance, obesity, and chronic inflammatory state characteristic of most human T2DM cases. Therefore, our findings may be most applicable to patients with lean diabetes or those with predominantly secretory defects. Second, the mechanisms of cisplatin-induced anemia—direct myelosuppression and nephrotoxicity-mediated EPO deficiency—are conserved across species (Madeddu et al., 2021). The hypoproliferative anemia observed in our model is pathophysiologically similar to human chemotherapy-induced anemia, supporting the mechanistic relevance of this model. Third, regarding cancer type generalizability, this model induces anemia through cisplatin administration without tumor implantation. Although this approach isolates drug-induced hematotoxicity from tumor-related effects, it does not account for the mechanisms of cancer-related anemia (inflammatory cytokines, tumor infiltration, nutritional depletion) that contribute to anemia in clinical settings. Future studies incorporating tumor-bearing diabetic animals would enhance applicability to specific cancer types. Finally, species-specific differences in cisplatin pharmacokinetics, renal physiology, and erythropoietic regulation must be acknowledged. The optimal dose identified in rats (6 mg/kg) cannot be directly extrapolated to humans, where cisplatin dosing is typically calculated based on body surface area (50–100 mg/m²) and adjusted for renal function. Despite these limitations, preclinical rat models remain essential tools for mechanistic investigations and initial screening of protective interventions before clinical translation. The model established in this study provides a platform for investigating diabetes-specific responses to platinum-based chemotherapy and evaluating candidate therapies aimed at mitigating hematotoxicity in this vulnerable patient population. Study limitations and future researchThis preliminary study has several limitations that warrant consideration. First, hematological characterization was incomplete. This study focused exclusively on HGB concentration without assessing erythrocyte indices (MCV, MCH, and MCHC), red blood cell count, hematocrit, reticulocyte count, or peripheral blood smear morphology. Additionally, EPO and renal function parameters (serum creatinine, BUN) were not measured. These omissions preclude the definitive classification of anemia morphology, differentiation between central (bone marrow) and peripheral (EPO-mediated) mechanisms, and direct evaluation of the renal-erythropoietic axis—a key pathway in cisplatin-induced anemia (Bryer and Henry, 2018; Madeddu et al., 2021). Future studies should incorporate comprehensive hematological and biochemical assessments to fully characterize induced anemia and its underlying mechanisms. Second, the experimental design included only diabetic animals without non-diabetic controls. This limitation limits our ability to isolate the specific contribution of diabetes to the observed hematological toxicity and weight changes and precludes the determination of whether the diabetic state intensified cisplatin-induced anemia. Future studies should employ a full factorial design including both diabetic and non-diabetic groups to comprehensively characterize the interaction between diabetes and cisplatin-induced hematotoxicity. Third, the sample size of five animals per group, while consistent with established protocols for preliminary dose-finding studies (Gao et al., 2006; Aston et al., 2017) and adequate for detecting the large observed effect sizes (post-hoc power >99%), represents a limitation for generalizability and detection of subtle differences in secondary endpoints. Future confirmatory studies should employ larger sample sizes (n ≥ 8 per group) as recommended in rodent toxicology methodology (Festing and Altman, 2002; Charan and Kantharia, 2013). Fourth, the acute, single-dose design with a 21-day observation period does not fully replicate clinical repeated-dosing regimens and may be insufficient to capture delayed toxicity or chronic hematological effects. Extended follow-up periods (≥28–42 days) with chronic or cyclical cisplatin administration would clarify the long-term tolerability profile and enable the assessment of hematological recovery kinetics. Despite these limitations, this study successfully established the foundational parameters necessary for developing a diabetic-specific CIA model and provided essential dose-response data for subsequent mechanistic and interventional investigations. Clinical and translational implicationsThis model addresses a clinically relevant gap because diabetes patients are predisposed to exacerbated chemotherapy-related hematotoxicity (Mailliez et al., 2023), with diabetes becoming increasingly prevalent among cancer patients. The model established in this study facilitates the investigation of diabetic-specific responses to chemotherapy and supports the early-stage testing of therapeutic interventions that aim to protect against anemia without compromising antitumor efficacy. Overall, these findings support the establishment of a pathophysiologically relevant CIA model in diabetic rats, which may serve as a basis for evaluating novel preventive or therapeutic strategies. ConclusionIn conclusion, a single dose of cisplatin (6 mg/kg BW) effectively induces moderate, non-lethal anemia in STZ–NA-induced diabetic Sprague Dawley rats while preserving acceptable systemic tolerability. This dose provides an appropriate starting point for modeling chemotherapy-induced anemia in the diabetic context. However, this preliminary study represents an initial foundation for model development rather than a fully validated preclinical tool. Comprehensive model validation will require: (a) larger sample sizes with extended observation periods, (b) complete hematological characterization including erythrocyte indices and peripheral blood smear analysis, (c) mechanistic assessments incorporating EPO and renal function parameters, and (d) comparison with non-diabetic controls to isolate diabetes-specific effects. Despite these limitations, this study provides essential dose-response data and demonstrates the feasibility of inducing anemia in diabetic rats using a single dose of cisplatin. These findings establish a promising direction for developing clinically relevant preclinical models that incorporate metabolic comorbidities, which may ultimately contribute to more personalized approaches in managing chemotherapy-induced hematotoxicity in diabetic cancer patients. AcknowledgmentsThe authors would like to thank all staff of the Animal Research Facility Center for Food and Nutrition Studies Universitas Gadjah Mada, for their support in this research. Conflict of interest All authors have no conflicts of interest to declare and are responsible for the content and writing of the article. Funding This study received no specific grant. Authors’ contributions A.J.S., P.D., B.P., E.P.P., and R.C. designed the protocol and interpreted the results of the experiments. A.J.S. and B.W. performed the experiments, analysed, and interpreted the data. A.J.S., D.T.M.W., and Y.H.P.K.D. drafted the initial manuscript. All authors have approved the final version of the manuscript. Data availability All data supporting this study are available upon request from the corresponding author. ReferencesAmerican Veterinary Medical Association (AVMA). 2020. AVMA guidelines for the euthanasia of animals: 2020 edition. Schaumburg, IL: American Veterinary Medical Association. Arkew, M., Asmerom, H., Gemechu, K. and Tesfa, T. 2023. Global prevalence of anemia among type 2 diabetic adult patients: a systematic review and meta-analysis. Diabetes. Metab. Syndrome Obes. 16, 2243–2254; doi:10.2147/DMSO.S421851 Aston, W.J., Hope, D.E., Nowak, A.K., Robinson, B.W., Lake, R.A. and Lesterhuis, W.J. 2017. A systematic investigation of the maximum tolerated dose of cytotoxic chemotherapy with and without supportive care in mice. BMC Cancer 17(1), 684; doi:10.1186/s12885-017-3677-7 Barbieri, J., Fontela, P.C., Winkelmann, E.R., Zimmermann, C.E.P., Sandri, Y.P., Mallet, E.K.V. and Frizzo, M.N. 2015. Anemia in patients with type 2 diabetes mellitus. Anemia 1(1), 354737; doi:10.1155/2015/354737 Basu, L., Grieco-St-Pierre, L., Ching, M.E.A., Stead, J.D.H., Hanson, A.A., Palaniyandi, J., Van Zyl, E., Hoyeck, M.P., Mckay, K.S., Van Allen, K.A., Lee, H., Dai, X.Q., Bautista, A., Fadzeyeva, E., Mulvihill, E.E., Yauk, C.L., Mennigen, J.A., Macdonald, P.E. and Bruin, J.E. 2025. Cisplatin exposure dysregulates insulin secretion in male and female mice. Diabetes 74(4), 528–543; doi:10.2337/db24-0419 Berger, J. 2007. Phenylhydrazine haematotoxicity. J. Appl. Biomed. 5(3), 125–130. Bryer, E. and Henry, D. 2018. Chemotherapy-induced anemia: etiology, pathophysiology, and implications for contemporary practice. Int. J. Clin. Transfusion. Med. 6, 21–31; doi:10.2147/IJCTM.S187569 Bulduk, B., Oto, G., Günbatar, N., Bulduk, M., Koçak, Y. and Elasan, S. 2022. The effect of resveratrol on toxicity caused by cisplatin in rats with experimentally created diabetes by streptozotocin. J. Health. Sci. Med. 5(1), 124–130. Bunn, H.F. 2013. Erythropoietin. Cold Spring Harbor Perspect. Med. 3(3), 11619; doi:10.1101/cshperspect.a011619 Cannavale, K., Xu, H., Xu, L., Sattayapiwat, O., Rodriguez, R., Bohac, C., Page, J. and Chao, C. 2019. Epidemiology of chemotherapy-induced anemia in patients with non-hodgkin lymphoma. Permanente. J. 23, 18–252. Cen, Y., Wang, P., Gao, F., Jing, M., Zhang, Z., Yi, P., Zhang, G., Sun, Y. and Wang, Y. 2022. Tetramethylpyrazine nitrone activates hypoxia-inducible factor and regulates iron homeostasis to improve renal anemia. Front. Pharmacol. 13, 964234. Charan, J. and Kantharia, N.D. 2013. How to calculate sample size in animal studies?. J. Pharmacol. Pharmacother. 4(4), 303–306; doi:10.4103/0976-500X.119726 Da Silva Faria, M.C., Santos, N.A.G.D., Carvalho Rodrigues, M.A., Rodrigues, J.L., Barbosa Junior, F. and Santos, A.C.D. 2015. Effect of diabetes on biodistribution, nephrotoxicity and antitumor activity of cisplatin in mice. Chem-Biol. Interact. 229, 119–131. DeMaeyer, E.M., Dallman, P., Gurney, J.M., Hallberg, L., Sood, S.K., Srikantia, S.G. and World Health Organization. 1989. Preventing and controlling iron deficiency anaemia through primary health care: a guide for health administrators and programme managers. Geneva, Switzerland: World Health Organization. Fagbohun, O.F., Awoniran, P.O., Babalola, O.O., Agboola, F.K. and Msagati, T.A.M. 2020. Changes in the biochemical, hematological and histopathological parameters in STZ-induced diabetic rats and the ameliorative effect of Kigelia africana fruit extract. Heliyon 6(5), e03989; doi:10.1016/j.heliyon.2020.e03989 Festing, M.F.W. and Altman, D.G. 2002. Guidelines for the design and statistical analysis of experiments using laboratory animals. ILAR. J. 43(4), 244–258; doi:10.1093/ilar.43.4.244 Finnigan, D., Hajjaj, O.I. and Othman, M. 2025. Red blood cell changes due to cancer and cancer treatments: a narrative review. Curr. Opinion. Hematol. 32(2), 93–103. Fröhlich, J. 2020. Rats and mice. In Ferrets, Rabbits, and Rodents. Eds., Quesenberry, K., Mans, C. and Orcutt, C. St. Louis, MO: Elsevier Health Sciences, pp: 345 -367; doi: 10.1016/B978-0-323-48435-0.00025-3 Furman, B.L. 2021. Streptozotocin-induced diabetic models in mice and rats. Curr Protocols. 1(4), e78; doi: 10.1002/cpz1.78 Gao, L., Ma, R., Zhou, J. and Cheng, S. 2006. Changes of serum erythropoietin during cisplatin-or 5-fluorouracil-induced anemia in rats. Toxicol. Mech. Methods. 16(9), 501–506. López-Tofiño, Y., López-Gómez, L., Martín-Ruíz, M., Uranga, J.A., Nurgali, K., Vera, G. and Abalo, R. 2025. Effects of repeated cisplatin and monosodium glutamate on visceral sensitivity in rats. Cells 14(1), 26; doi:10.3390/cells14010026 Madeddu, C., Neri, M., Sanna, E., Oppi, S. and Macciò, A. 2021. Experimental drugs for chemotherapy-and cancer-related anemia. J. Exp. Pharmacol. 13, 593–611; doi:10.2147/JEP.S262349 Mailliez, A., Ternynck, C., Duhamel, A., Mailliez, A., Ploquin, A., Desauw, C., Lemaitre, M., Bertrand, N., Vambergue, A. and Turpin, A. 2023. Diabetes is associated with high risk of severe adverse events during chemotherapy for cancer patients: a single-center study. Int. J. Cancer 152(3), 408–416. Malik, N.M., Moore, G.B.T., Smith, G., Liu, Y.L., Sanger, G.J. and Andrews, P.L.R. 2006. Behavioural and hypothalamic molecular effects of the anti-cancer agent cisplatin in the rat: a model of chemotherapy-related malaise?. Pharmacol. Biochem. Behav. 83(1), 9–20. Piepho, H.P. 2004. An algorithm for a letter-based representation of all-pairwise comparisons. J. Comput. Graph. Statist. 13(2), 456–466. Pinheiro, L.C., Kaur, H., Nilo, D., Safford, M.M., Derosa, A.P. and Kern, L.M. 2019. Determining the impact of a cancer diagnosis on diabetes management: a systematic literature review. Am. J. Clin. Oncol. 42(11), 870–883. Rajendrakumar, T., Rao, S., Satyanarayana, M.L., Narayanaswamy, H.D., Byregowda, S.M. and Shridhar, N.B. 2020. Hematological alterations in cisplatin induced toxicity in rats and its amelioration by Andrographis paniculata. Int. J. Curr. Microbiol. Appl. Sci. 9(4), 525–532. Rehman, H.U., Ullah, K., Rasool, A., Manzoor, R., Yuan, Y., Tareen, A.M., Kaleem, I., Riaz, N., Hameed, S. and Bashir, S. 2023. Comparative impact of streptozotocin on altering normal glucose homeostasis in diabetic rats compared to normoglycemic rats. Scientific. Rep. 13(1), 7921; doi:10.1038/s41598-023-29445-8 Saito, Y., Sakamoto, T., Takekuma, Y., Kobayashi, M., Okamoto, K., Shinagawa, N., Shimizu, Y., Kinoshita, I. and Sugawara, M. 2022. Diabetes mellitus degenerates cisplatin-induced nephrotoxicity in short hydration method: a propensity score-matching analysis. Scientific. Rep. 12(1), 21819. Schleicher, E., Gerdes, C., Petersmann, A., Müller-Wieland, D., Müller, U.A., Freckmann, G., Heinemann, L., Nauck, M. and Landgraf, R. 2022. Definition, classification and diagnosis of diabetes mellitus. Exp. Clin. Endocrinol. Diabetes. 130(S 01), S1–S8; doi: 10.1055/a-1624-2897 Sharma, P., Georgy, J.T., Andrews, A.G., John, A.O., Joel, A., Chacko, R.T., Premkumar, P.S. and Singh, A. 2022. Anemia requiring transfusion in breast cancer patients on dose-dense chemotherapy: prevalence, risk factors, cost and effect on disease outcome. Support. Care. Cancer. 30(6), 5519–5526. Sultana, F., Islam, M.F., Amin, M.N., Jahan, N., Kabir, A., Emran, T.B., Sutradhar, B.C. and Banik, S. 2022. Evaluation of biochemical and pathological parameters at different doses of cisplatin in experimental animal model: toxicological study on an antineoplastic drug. Makara J. Health Res. 26(1), 67–73. World Health Organization. 2024. Guideline on haemoglobin cutoffs to define anaemia in individuals and populations. Geneva, Switzerland: World Health Organization. Xu, H., Xu, L., Page, J.H., Cannavale, K., Sattayapiwat, O., Rodriguez, R. and Chao, C. 2016. Incidence of anemia in patients diagnosed with solid tumors receiving chemotherapy, 2010–2013. Clin. Epidemiol. 8, 61–71; doi:10.2147/CLEP.S89480 Xu, Y.J., Wang, Y., Lu, Y.F., Xu, S.F., Wu, Q. and Liu, J. 2017. Age-associated differences in transporter gene expression in kidneys of male rats. Mol. Med. Rep. 15(1), 474–482. Yousif, A.A.M., Merghani, M.M. and Babiker, N.E. 2021. The effect of cisplatin on complete blood count among chemotherapy sudanese patients at Taiba Cancers Center in Khartoum state. J. Drug Del. Ther. 11(6-S), 104–113. Zhang, L., Xu, P. and Yan, X. 2025. Mechanism-based pharmacokinetic/pharmacodynamic modeling of erythroferrone in anemic rats with chronic kidney disease and chemotherapy-induced anemia: an early biomarker for hemoglobin response and rHuEPO hyporesponsiveness. ACS. Pharmacol. Translational. Sci. 8(1), 189–202; doi:10.1021/acsptsci.4c00575 Supplementary TablesTable S1. Individual animal data for all hematological parameters and BW measurements. Data are presented for each rat (identified by unique ID) across all treatment groups and observation timepoints.
Table S2. Fixed-effects estimates from GLMM assessing cisplatin dose, observation day, and their interaction on HGB levels.
Table S3. Pairwise comparisons of HGB levels across cisplatin dose groups and observation days (GLMM EMMs with Bonferroni and BH-adjusted p-values).
Table S4. Fixed-effects estimates from GLMM for PLT, WBC, and NEU counts across cisplatin dose groups and observation days.
Table S5. EMMs for PLT, WBC, and NEU counts by cisplatin dose group and observation day (GLMM analysis).
Table S6. Pairwise comparisons of PLT, WBC, and NEU counts between treatment groups for each observation day (Welch’s t-test with BH-adjusted p-values).
Supplementary Figure Appendix
Figure S1. EMMs of HGB levels across cisplatin dose groups and observation days (GLMM, mean ± 95% CI). Notes: GLMM, generalized linear mixed-effects models; Hb, hemoglobin; CI, confidence interval. | ||
| How to Cite this Article |
| Pubmed Style Sunggoro AJ, Dirgahayu P, Purwanto B, Pamungkasari EP, Warsita B, Cilmiaty R, Wardhani DTM, Dewi YHPK. Determination of the optimal cisplatin dose to induce anemia in streptozotocin-nicotinamide-induced type 2 diabetic rats: A preliminary study. Open Vet. J.. 2026; 16(2): 1090-1111. doi:10.5455/OVJ.2026.v16.i2.29 Web Style Sunggoro AJ, Dirgahayu P, Purwanto B, Pamungkasari EP, Warsita B, Cilmiaty R, Wardhani DTM, Dewi YHPK. Determination of the optimal cisplatin dose to induce anemia in streptozotocin-nicotinamide-induced type 2 diabetic rats: A preliminary study. https://www.openveterinaryjournal.com/?mno=285025 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.29 AMA (American Medical Association) Style Sunggoro AJ, Dirgahayu P, Purwanto B, Pamungkasari EP, Warsita B, Cilmiaty R, Wardhani DTM, Dewi YHPK. Determination of the optimal cisplatin dose to induce anemia in streptozotocin-nicotinamide-induced type 2 diabetic rats: A preliminary study. Open Vet. J.. 2026; 16(2): 1090-1111. doi:10.5455/OVJ.2026.v16.i2.29 Vancouver/ICMJE Style Sunggoro AJ, Dirgahayu P, Purwanto B, Pamungkasari EP, Warsita B, Cilmiaty R, Wardhani DTM, Dewi YHPK. Determination of the optimal cisplatin dose to induce anemia in streptozotocin-nicotinamide-induced type 2 diabetic rats: A preliminary study. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1090-1111. doi:10.5455/OVJ.2026.v16.i2.29 Harvard Style Sunggoro, A. J., Dirgahayu, . P., Purwanto, . B., Pamungkasari, . E. P., Warsita, . B., Cilmiaty, . R., Wardhani, . D. T. M. & Dewi, . Y. H. P. K. (2026) Determination of the optimal cisplatin dose to induce anemia in streptozotocin-nicotinamide-induced type 2 diabetic rats: A preliminary study. Open Vet. J., 16 (2), 1090-1111. doi:10.5455/OVJ.2026.v16.i2.29 Turabian Style Sunggoro, Agus Jati, Paramasari Dirgahayu, Bambang Purwanto, Eti Poncorini Pamungkasari, Brian Warsita, Risya Cilmiaty, Damiana Tribhuwaneswari Mahendra Wardhani, and Yohanna Handika Putri Kusuma Dewi. 2026. Determination of the optimal cisplatin dose to induce anemia in streptozotocin-nicotinamide-induced type 2 diabetic rats: A preliminary study. Open Veterinary Journal, 16 (2), 1090-1111. doi:10.5455/OVJ.2026.v16.i2.29 Chicago Style Sunggoro, Agus Jati, Paramasari Dirgahayu, Bambang Purwanto, Eti Poncorini Pamungkasari, Brian Warsita, Risya Cilmiaty, Damiana Tribhuwaneswari Mahendra Wardhani, and Yohanna Handika Putri Kusuma Dewi. "Determination of the optimal cisplatin dose to induce anemia in streptozotocin-nicotinamide-induced type 2 diabetic rats: A preliminary study." Open Veterinary Journal 16 (2026), 1090-1111. doi:10.5455/OVJ.2026.v16.i2.29 MLA (The Modern Language Association) Style Sunggoro, Agus Jati, Paramasari Dirgahayu, Bambang Purwanto, Eti Poncorini Pamungkasari, Brian Warsita, Risya Cilmiaty, Damiana Tribhuwaneswari Mahendra Wardhani, and Yohanna Handika Putri Kusuma Dewi. "Determination of the optimal cisplatin dose to induce anemia in streptozotocin-nicotinamide-induced type 2 diabetic rats: A preliminary study." Open Veterinary Journal 16.2 (2026), 1090-1111. Print. doi:10.5455/OVJ.2026.v16.i2.29 APA (American Psychological Association) Style Sunggoro, A. J., Dirgahayu, . P., Purwanto, . B., Pamungkasari, . E. P., Warsita, . B., Cilmiaty, . R., Wardhani, . D. T. M. & Dewi, . Y. H. P. K. (2026) Determination of the optimal cisplatin dose to induce anemia in streptozotocin-nicotinamide-induced type 2 diabetic rats: A preliminary study. Open Veterinary Journal, 16 (2), 1090-1111. doi:10.5455/OVJ.2026.v16.i2.29 |