| Research Article | ||
Open Vet. J.. 2026; 16(2): 934-943 Open Veterinary Journal, (2026), Vol. 16(2): 934-943 Research Article Effect of Moringa oleifera leaf extract on body length, malondialdehyde, and interleukin-6 levels in zebrafish embryo (Danio rerio)-induced stunting model with rotenoneMaslichah Mafruchati1*, Lailatul Fadliyah2, Akhmad Kusuma Wardhana3 and Intan Idiana Hassan41Department of Veterinary Anatomy, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 2Faculty of Vocational Studies, Universitas Airlangga, Surabaya, Indonesia 3Faculty of Environmental Management, Prince of Songkla University, Hatyai, Thailand 4School of Health Sciences, Universiti Sains Malaysia, George Town, Malaysia *Corresponding Author: Maslichah Mafruchati. Department of Veterinary Anatomy, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: maslichah-m [at] fkh.unair.ac.id Submitted: 26/09/2025 Revised: 11/01/2026 Accepted: 29/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
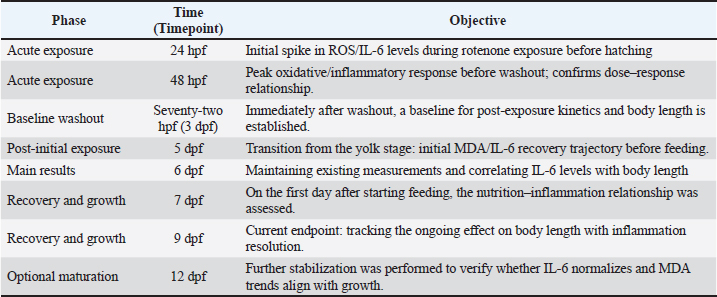
AbstractBackground: Stunting is the disruption of growth in both humans and animals brought on by ingesting inadequate nourishment over an extended period or receiving inadequately nutritious food. Aim: This study aimed to analyze the effect of the extracted leaf of Moringa oleifera on body length, malondialdehyde (MDA), and IL-6 levels in zebrafish larvae to mitigate stunting induced by rotenone. Methods: This study used an embryo of zebrafish at the age of 2–72 hours post-fertilization (hpf), which were exposed to rotenone with a concentration of 12.5 ppb to induce stunting, as well as exposed to the leaf extract of M. oleifera using various concentrations (0.56, 1.12, 2.24 µg/ml). On day 6 post-fertilization (dpf), zebrafish embryos were measured using the enzyme-linked immunosorbent assay method to determine MDA levels and IL-6 inside their body. Results: Moringa oleifera leaf extract reduced IL-6 levels in the water. MDA level was increased in the group given rotenone and moringa leaf extract (p < 0.05). However, the MDA rate had no significant relationship with zebrafish embryo body length (p > 0.05). Rotenone significantly reduced body length and increased IL-6 and MDA levels compared with controls. Moringa oleifera leaf extract improved body length in a dose-dependent manner, with the greatest effect at 2.24 µg/ml. IL-6 reduction was significant only at the highest dose, while lower doses improved growth without statistically lowering IL-6. MDA levels were paradoxically elevated in rotenone + moringa groups, likely reflecting assay interference, mineral-driven lipid peroxidation, or transient ROS burst during metabolic recovery. Conclusion: Moringa oleifera leaf extract effectively mitigates stunting in zebrafish embryos by reducing IL-6 levels and improving body length, despite elevated MDA levels. These findings support its potential as a safe, natural anti-inflammatory intervention for early-life growth disturbance. Keywords: Body length, Life expectancy, IL-6, Moringa oleifera, Zebrafish embryos. IntroductionStunting is the disruption of growth in both humans and animals brought on by ingesting inadequate nourishment over an extended period or receiving inadequately nutritious food (Lassi et al., 2013). Stunting, as shown by the limit (z-score) being <−2 SDs, occurs when body length or height is compared to age (Walker et al., 2005). Several factors, including oxidative stress and chronic inflammation, are considered the primary contributors to stunting (Gong et al., 2023). Growth abnormalities can be caused by insulin-like growth factor-1 (IGF-1) (Van Der Pol et al., 2019), which is less frequently produced in chronic inflammation. Interleukin-6 (IL-6) is primarily responsible for reducing IGF-1 synthesis (Ferraccioli et al., 2010). Increased proteolytic degradation of insulin-like growth factor binding protein-3 (IGFBP-3) and decreased IGF-1 expression can result from chronic inflammation and excessive IL-6 production (Hamid et al., 2022). IGF-1 does not bind to IGFBP-3; hence, IGF-1 has a shorter half-life by lowering IGFBP-3. In addition, IL-6 could inhibit osteoblast activity. Additionally, an increase in osteoclastogenesis might make IL-6 a proinflammatory cytokine with the potential to inhibit growth (Hamid et al., 2022). According to Wu et al. (2022), the pesticide rotenone is hazardous to both humans and animals. Stunting is a failure of linear growth during the first 2 years of life and is caused by rotenone toxicity. Because rotenone began with a barrier on the chain respiration complex I mitochondria, which increases reactive oxygen species (ROS). ROS are organic compounds with a functional group and an oxygen atom that has more electrons. ROS is formed naturally, especially in complex I of the mitochondrial respiratory chain, during normal cellular activity, or during pathological development (Higashi et al., 2014). If ROS was increased, the death of the complex-1 mitochondria cells was activated, causing the pathophysiology of zebrafish to be stunted. The interference of rotenone with the transport of electrons to the chain respiration complex I in mitochondria increases the amount and quality of free electrons (Ye et al., 2021). This will build up on complex-1 mitochondria until a leak occurs, allowing electrons to escape (Chen et al., 2018). Zebrafish have genes that are 70% homologous to human genes. Zebrafish have high reproduction rates, long embryonic stages, and a unique level of sensitivity to chemical exposure. Moreover, it also has the characteristics of growing bone, which is fast, so it is easier to observe bone morphology in living embryos. This type of fish is suitable for use as a sample in a research model on the toxicity of chemicals or drugs (Etchin et al., 2011). As a result, this study was interested in using zebrafish as the sample of this study because of its use that could be implemented in pharmaceutical and medical purposes related to humans (Primihastuti et al., 2022). Moringa oleifera is a plant that grows in tropical and subtropical countries and acts as an antioxidant to hinder oxidative stress and an anti-inflammatory. M. oleifera contains flavonoid antioxidants, which could inhibit cascade reactions caused by rotenone as a source of oxidative stress induction. Moreover, M. oleifera has the potential to protect against the neurotoxic effects of rotenone. Based on that background, this study aimed to observe the effect of the extracted leaf of M. oleifera on body length, malondialdehyde (MDA), and IL-6 levels in zebrafish larvae to impede stunting, which is induced with rotenone. Moreover, M. oleifera was readily available in Indonesia due to its ease of cultivation in tropical countries, and the stock was abundant. Materials and methodsZebrafish broodstock maintenance and fertilization preparationA 4-month-old zebrafish brood, consisting of female and male brooders with a ratio of 2:1, was placed in an aquarium. The aquarium is cleaned at least once a week, and the water filter is replaced every 3 days. The water is treated with antichlorine, and it needs at least 30 minutes before the fish can be placed in the aquarium. They were fed with tetramine 3 times a day. A trap is installed at the bottom of the aquarium for the embryo trap, which is activated before the fertilization process begins. Then, the aquarium is closed (using cardboard and cloth), and the light is turned off to create a dark cycle (10 hours), during which time the researcher (20.30–06.30 WIB). A comparison of the dark-light cycle was conducted with a ratio of 10:14 hours (Raguso et al., 2024). The next day at 06.30, the lights were turned on for 30 minutes before the lid was lifted to give it a chance of conception. The trap was removed, and the eggs were cleaned with filtered water until they were clean and no more than 2 hours post-fertilization (hpf). The embryos were placed in a 6-well plate, with each well containing 30 embryos. Embryonic medium is administered according to the treatment group and replaced every day until 72 hpf according to the following: Group 1: Medium embryonic Group 2: Rotenone 12.5 pub + medium embryonic Group 3: Rotenone 12.5 pub + extract leaf moringa 0.56 µg/ml + embryonic medium Group 4: Rotenone 12.5 pub + extract leaf moringa 1.12 µg/ml + embryonic medium Group 5: Rotenone 12.5 pub + extract leaf moringa 2.24 µg/ml + embryonic medium Group 6: Extract moringa leaves 0.56 µg/ml) + embryonic medium Group 7: Extract leaf moringa 1.12 µg/ml + medium embryonic Group 8: Extract leaf moringa 2.24 µg/ml embryonic medium After that, zebrafish were placed in embryonic medium and incubated from 3 to 9 dpf. All groups used embryonic medium only and were stored in an incubator at 28°C ± 1°C (Raguso et al., 2024). Feeding 6 dpf zebrafish embryo with tetramine, taken to taste, crushed until smooth, adding water, and giving it to the zebrafish embryo. Making rotenone solution (Sigma R8875)A rotenone solution was made by dissolving rotenone powder in 10% dimethyl sulfoxide to reach a concentration of 2 × 103 µg/ml using the following formula: V1 × M1=V2 × M2 V1: volume of the diluent/filtration water V2: the volume to be administered according to the requested dose M1: rotenone concentration M2: requested dose The rotenone stock that has been made was put in the Eppendorf, labeled according to the date of manufacture in the laboratory, and stored in the freezer. Rotenone was thawed in the water where the zebrafish were treated and diluted with embryonic medium before use. Dosage of rotenone used:M: 2 × 103 µg/ml M2: 12.5 µg/m; V1: 15 ml (embryonic medium) (15 × 2.103=V2 × 12.5) V2: 93.75.10–3 ml=93.75 µl Making medium embryonicsAll ingredients (0.08 g of CaCl, 0.06 g of KCl, 2 g of NaCl, and 43.2 g of MgSO) were weighed using a digital scale and placed in a test tube containing 200 ml of filtered water. Stir until the ingredients are well blended, put in a bottle, label (date of manufacture), and store in the refrigerator as stock. When it would be used, the stock is taken, and water is added, dilution as needed, with a medium stock ratio (1:9) for embryonic: filtered water. Making an extract from M. oleifera leavesAt least 100 g of Moringa simplicia was put in an Erlenmeyer glass. Simplicia is macerated with a 98% ethanol solvent of up to 900 ml while shaking until thoroughly mixed, and then allowed to stand for 24 hours to form a precipitate. The top layer is taken. The immersion process is carried out three times. Simplicia was submerged and filtered with filter paper using a Buchner funnel. The filtrate was placed in a 1 l evaporating flask. The evaporating flask is connected to the evaporator, the water bath is filled with water until it is full, and all the evaporation devices are assembled and connected to electricity. The water bath is heated to a temperature of 90°C and left for ±1.5–2 hours to separate the ethanol solution from the active substance in the holding flask. The extraction results are placed in plastic bottles, weighed, and stored in the freezer. The tubes are labeled with the date of manufacture and stored in the freezer. To make a stock solution of Moringa leaf extract, 1 mg/ml (1,000 µg/ml) of Moringa leaf extract was weighed as much as 10 mg and dissolved in 10 ml of sterile water until well mixed. Add it to the Valcon/Eppendorf, label it according to the manufacturer, and store it in the freezer. The dosage of Moringa leaf extract was obtained from Reyes Sánchez et al. (2006) regarding M. oleifera leaf extract in rats. Dose conversion was performed using the ratio of the body weight of the rats and zebrafish. Exposure to rotenone and moringa leaf extract, changing the medium, and feeding larvaeRotenone and Moringa leaf extract were administered to zebrafish embryos at 2–72 hpf. The zebrafish embryos were grouped into eight groups, each given exposure to rotenone concentrations of 12.5 ppb. Rotenone and moringa were administered in various doses of 0.56, 1.12, and 2.24 µg/ml, with the same interval, namely, 2, 24, and 48 hpf. At 72 hpf (3 dpf), the larvae hatched (out of the chorion) and were rinsed with embryonic medium solution three times to ensure that the exposure medium was removed from the larvae. All groups have replaced the embryonic medium daily from 9 dpf to prevent a contamination-free environment that can disturb the larvae. Zebrafish embryo are fed at 6 dpf because they still obtain nutrition from the yolk sac until 5 dpf. Zebrafish embryo are fed with tetramine at 6 dpf to meet nutritional needs and prevent death (Licitra et al., 2024). This study ground Tetramin and added it to filtered water, then dripped it into each well, up to 5 drops. Change of medium and feeding is done every day until the 9th day to fulfill their nutrition. Measurement of the long bodyBody length measurements on zebrafish embryo were carried out 3 times, namely at 72 hpf, 6 dpf, and 9 dpf. Zebrafish embryo were placed in a glass object and observed under a stereo microscope (Olympus SZ61) from PT Wadya Prima as a sole distributor for Olympus/Evident microscope in East Java, which is imported from Olympus Corporation, Tokyo, Japan. It was photographed in a straight position using Optilab software version 2.0 and not moving, and then measured using calibrated image raster software. Photographed and measured zebrafish embryo are included in a different plate containing embryonic medium. Based on the standard length (SL), the body length is measured from the tip of the nose (tip of the snout) to the base of the tail (caudal fin). Measurement rate of MDAs and IL-6Zebrafish embryo aged 6 dpf were euthanized in a deep immersion chamber filled with water and ice at a ratio of 5:1 for 10 min (Yin et al., 2022). This was for the anesthesia of the zebra fish. The zebrafish were then transferred to a Petri dish, lysed using 500 µL radioimmunoprecipitation assay (RIPA) buffer, homogenized using a plunger, and 250 µL RIPA buffer was added. The results of the suspension were put into a 2 mL microtube and cold centrifuged at 4°C at 3000 rpm for 20 min at 4°C. The supernatant was analyzed using the ELISA method to determine the levels of MDA (Bioassay Technology Laboratory; E0017FI) and IL-6 (Bioassay Technology Laboratory; E0026FI). Table 1 highlights the experimental timeline of zebrafish embryo exposed to rotenone and extract of Moringa Oleifera’s leaves. It shows that embryos grouped and treated at specific hours post-fertilization (2–72 hpf), with exposures repeated at 2, 24, and 48 hpf. The schedule ensured consistent induction of rotenone to check the stunting effect and systematic administration of Moringa extract doses for later measurement of body length, IL-6, and MDA. Table 1. Observation of the timeline of zebrafish embryo treated with rotenone and Moringa oleifera.
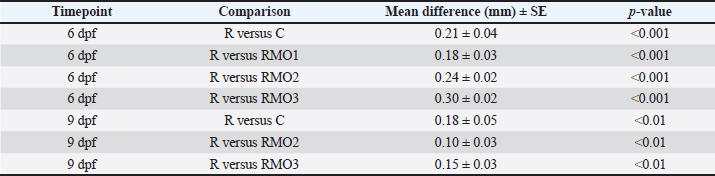
ELISA procedure(1) Put 50 µl standard into the standard well (2) Add 40 µl of the sample into the well. Then, add 10 µl of anti-MDA antibody zebrafish/zebrafish anti-IL-6 antibodies into the sample wells (3) Add 50 µl of streptavidin-HRP into the standard and sample wells (do not put into the control well), mix for 1 minute, close with adhesive, and incubate for 60 minutes at 37°C. (4) The adhesive is opened. Each well was then aspirated and washed three times with the wash buffer. At the end of the washing process, the remaining wash buffer was removed and dried with a paper dryer. (5) Add 50 µl of substrate solution A and 50 µl of substrate solution B in each well and incubate for 10 minutes at room temperature and 37°C, covered with adhesive to protect from light. (6) Add 50 µl stop solution into each well (7) In 30 minutes, data were analyzed using an ELISA reader at a wavelength of 450 nm to determine the optical density (value). Data analysisThe collected data were presented in the form of mean score ± SD. Then, all data were analyzed using SPSS version 21 with the method of parametric statistics, one-way analysis of variance after fulfilling the test of normality data and the test of homogeneity of variance. If the condition from the one-way ANOVA test is not fulfilled, then the chosen test is the non-parametric Kruskal-Wallis. A significance test score of 0.05 was used. Detailed pairwise comparisons are provided in the Tables 2–4. Table 2. Differences in body length (mean ± SE, p values).
Table 3. Differences in IL-6 levels (mean ± SE, p-values).
Table 4. Differences in MDA (mean ± SE, p-values).
Ethical approvalThis study used the sample from the Nidhom Foundation of Airlangga University, which has been accredited in the following ethical principles of using animal cells as a sample for research. The research and laboratory process samples were tested with an ethical test. The ethical clearance certificate number was 456/HRECC.FODM/V/2023, and it was tested by the Ethical Department of the Faculty of Veterinary Medicine of Airlangga University. ResultsTable 2 shows that at 6 dpf, rotenone significantly reduced body length compared with the control (p < 0.001). Moringa extract improved body length in a dose-response pattern, with the greatest effect in RMO3 (p < 0.001. The corrective effect persisted at 9 dpf, and the Moringa-alone group (MO1–MO3) did not differ from the control group at 3–9 dpf, confirming the safety profile. Table 3 shows that rotenone exposure significantly increased IL-6 levels compared with the control group (p < 0.001). Among the M. Oleifera extract-treated groups, only RMO3 showed a significant reduction in IL-6 compared with the rotenone group (p < 0.05). Lower doses (RMO1 and RMO2) showed a non-significant decrease, indicating that IL-6 suppression was dose-dependent. Table 4 shows that rotenone significantly increased MDA levels compared with the control group. The combination of rotenone + M. oleifera extract (RMO1, RMO2, and RMO3) produced a larger MDA level than the rotenone group alone. As discussed in the following section, this requires careful interpretation. DiscussionBody lengthAccording to the results above, there was a significant difference in body length between the control group (C) and the rotenone group (R) at 6 and 9 dpf. According to Seyoum et al. (2020), a toxin is the reason for the linear disturbance of growth in the early life of an animal. A previous study on the model of the stunting zebrafish (Sun et al., 2021) showed that exposure to rotenone concentration of 10 ppb in zebrafish aged 2-72 hours post fertilization (hpf). It caused disturbance in linear growth at age 6 dpf. Besides that, exposure to rotenone at a dose of 12.5 ppb in zebrafish (2-72 hpf) can cause stunting through decreased expression of insulin-like growth factor-1 (IGF-1) (Yan et al., 2023), extracellular signal-regulated kinase 1/2 (ERK 1/2), protein Ki-67 (Quadri et al., 2025), glucose transporter-1 (GLUT-1) (Wang et al., 2024), glucose transporter-4 (GLUT-4), vascular endothelial growth factor (VEGF), and vascular endothelial growth factor receptor-2 (VEGFR-2). Body length is the main indicator for determining stunting conditions. The results mentioned earlier showed that there was an increase in the body length of zebrafish larvae in the group given rotenone and various concentrations of M. oleifera leaf extract (RMO1, RMO2, and RMO3) compared to the rotenone (R) group. The average body length of the zebrafish larvae in the RMO1, RMO2, and RMO3 groups seemed to approach that of the control group (C) at 6 and 9 dpf. The results of the Pearson correlation test showed a strong positive connection between the extract concentration of the leaf of M. oleifera and the long bodies of the stunted zebrafish larvae. There was an increase in the concentration of Moringa leaf extract, followed by an increase in body length in the stunting model zebrafish larvae. Moringa oleifera leaf extract interferes with rotenone to induce stunting in zebrafish larvae. Moreover, there were no differences in the body length of the MO1, MO2, and MO3 groups with the control group (C) at the ages of 3, 6, and 9 dpf. This means that the leaf extract of M. oleifera with concentrations of 0.56, 1.12, and 2.24 µg/ml had no significant effect on stunting in zebrafish larvae. Various animal studies have assessed the safety of the leaf extract of M. oleifera and have shown a high level of safety for consumption. Moreover, the reports stated that no side effects were reported in animal and human studies. The enhancement rate of MDAs in groups RMO1, RMO2, and RMO3 caused by the combination of rotenone and M. oleifera administered to the zebrafish larvae. Hamed et al. (2022) stated that M. oleifera treatment can increase the metabolism of mitochondria and increase the O2 capacity of the samples. However, rotenone can inhibit mitochondrial complex 1, resulting in a declining ability of oxidative phosphorylation and hindering electron transport from mitochondrial complex I (Lee et al., 2020). As a result, the combination of rotenone and M. oleifera exposure can increase the metabolism of mitochondria that have impaired function, as well as increase the formation of free radicals (ROS), leading to oxidative stress (Nakai et al., 2003). IL-6Results of the study above showed an enhancement rate of IL-6 in the rotenone group (R) compared with the control group (C). The group given rotenone and extract of the leaf of M. oleifera using various concentrations (RMO1, RMO2, and RMO3) showed a declining rate of IL-6 compared with the R group. However, according to the statistical tests of one-way ANOVA, the extract of leaf moringa (M. oleifera) showed a significant effect in group RMO3 (concentration 2.24 µg/ml). This result agrees with the results of a study conducted by Hayer et al. (2010), which stated that rotenone can increase the activity of nuclear factor-κB (NF-κB). Moreover, those studies stated that IL-6 expression was correlated with inflammation. Exposure to rotenone significantly increased IL-6 levels, confirming an inflammatory response. Among the groups treated with M. Oleifera leaf extract, only the highest dose (RMO3, 2.24 µg/ml) produced a statistically significant reduction in IL-6 compared with the group administered with rotenone alone. Moreover, the lower-dose groups (RMO1 and RMO2) improved body length despite not significantly lowering IL-6 levels. This suggests that growth recovery at submaximal doses may occur through non-inflammatory pathways, such as partial stabilization of IGF-1 signaling, improved nutrient utilization, or enhanced cellular resilience independent of cytokine suppression (Xia et al., 2024). In other words, while IL-6 reduction appeared to be dose-dependent, the effect of M. Oleifera leaf extract on promoting the growth of zebrafish embryos could also involve mechanisms beyond inflammation control. This dual action explains why the body length of the zebrafish embryo increased in all treatment groups with M. Oleifera extract, but the reduction in IL-6 was only significant at the highest concentration (Gaber et al., 2025). MDAIn the above-mentioned results, rotenone exposure significantly increased the MDA level, consistent with lipid peroxidation and oxidative stress. However, MDA was further increased in the group administered a combination of rotenone and M. oleifera. This paradoxical finding can be explained by three mechanisms. The first was a technical artifact (assay interference), where polyphenolic compounds inside M. oleifera leaf, such as kaempferol and quercetin, could interfere with ELISA or TBARS-based assays, leading to artificially elevated MDA readings. Assay sensitivity to secondary oxidation products and polyphenol protein interactions has been documented in lipid peroxidation measurements (Martínez-González et al., 2017). The second reaction was metal-catalyzed peroxidation (Fenton reaction). Moringa oleifera sp. leaves contain minerals such as magnesium (Mg), iron (Fe), and cobalt (Co). These substances can catalyze hydroxyl radical formation through Fenton-like reactions. Therefore, lipid peroxidation is accelerated even in the presence of antioxidants (Ebeid et al., 2020). Third, recovery-related transient increase in ROS. Moringa oleifera stimulates mitochondrial activity and improves metabolic recovery. During this process, transient bursts of ROS are produced as part of normal mitochondrial adaptation (Jaja-Chimedza et al., 2017). Therefore, elevated MDA may reflect a temporary increase in ROS associated with accelerated metabolism, rather than pathological damage (Kim et al., 2018). Taken together, the concurrent increase in MDA along with improved body length and reduced IL-6 levels are attributed to M. Oleifera’s effect on anti-inflammatory and growth boost remains biologically consistent. On the other hand, the increase in MDA may represent either methodological interference, mineral-driven peroxidation, or recovery-related oxidative signaling (Anggraini et al., 2025). There was no significant relationship between body length and MDA levels in the zebrafish larval stunting model. This was because in this study, only MDA levels were measured at 6 dpf. The results showed that there was a negative correlation between body length and IL-6 levels in the stunting model of zebrafish larvae with moderate strength. This indicates a declining rate of IL-6, followed by an increase in body length in zebrafish larvae of the stunting model. These results are consistent with the theory that IL-6 can affect growth hormone and bone growth (McGonagle et al., 2020). The results showed no significant relationship between MDA levels and IL-6 levels in zebrafish larvae of the stunting model. This result differed from the result of a previous study (Primihastuti et al., 2022), which stated that damage to the oxidative system due to lipid peroxidation produces lipid aldehydes, such as 4-hydroxy-trans-2-nonenal (HNE), acrolein, and MDA. These lipid aldehydes could affect the activation of transcription factor NF-κB, which will enhance the production of proinflammatory cytokines. Moreover, a study conducted by Katmawanti et al. (2021) reported a decrease in MDA levels caused by the exposure of zebrafish to rotenone and Centella asiatica extract at 4, 5, and 6 dpf, followed by a decrease in IL-6 levels in zebrafish larvae. No significant relationship was found between MDA and IL-6 levels in larval zebrafish. It was because the measurement was done in one, that is, 6 dpf. As a result, it is unknown how the chart rate changes MDAs to rate IL-6 larvae zebrafish at 3, 6, and 9 dpf (Wang et al., 2017). ConclusionMoringa oleifera leaf extract effectively mitigated rotenone-induced stunting in zebrafish larvae by improving body length and reducing IL-6 levels. The growth-promoting effect was dose-dependent, with the highest concentration producing significant anti-inflammatory activity, whereas lower doses supported growth through possible non-inflammatory pathways. Although MDA levels were elevated in the rotenone + M. oleifera groups, this increase could reflect methodological interference, mineral-catalyzed lipid peroxidation, or transient ROS generation during metabolic recovery. More importantly, the increase in MDA did not correlate with impaired growth. There were beneficial effects of M. oleifera leaf extract on development and inflammation, which outweigh these oxidative markers. These findings support M. oleifera leaf extract as a safe, natural intervention for early-life growth disturbance. Future studies are needed to clarify the mechanistic basis of MDA changes. AcknowledgmentWe thank the current rector of Universitas Airlangga for the support in providing us with the incentive for publication upon publication of papers. We are also thankful to the vice rector of Universitas Airlangga for reminding the corresponding author to finish the research to be used to propose for a professorship. FundingNo funding was provided for the research process of this paper. However, the Article Processing Charge of the journal for this paper would be funded by Airlangga University upon acceptance by the editor of the intended journal. Authors' contributionMM performed data collection, data curation, funding acquisition, methodology, project administration, data analysis, and wrote the draft paper. LF performed conceptualization, investigation, supervision, validation, and data visualization. AKW performed data curation, translation, review, and editing of the paper, and data analysis. IIH proofread the paper’s grammar and wrote a draft of the paper. Conflict of interestThis study has no conflicts of interest to declare. Data availabilityThe study data would be provided if needed for the review process ReferencesAnggraini, N.W.P., Sulistyowati, S., Soetrisno, S., Purwanto, B. and Dirgahayu, P. 2025. Effects of Moringa oleifera leaves on CRP, MDA, and A levels in a mouse model of preeclampsia. Open Vet. J. 15(4), 1784. Chen, H., Chen, F., Hu, W. and Gou, S. 2018. Effective platinum (IV) prodrugs conjugated with lonidamine as a functional group working on the mitochondria. J. Inorganic Biochem. 180, 119–128. Ebeid, H.M., Kholif, A.E., Chrenkova, M. and Anele, U.Y. 2020. Ruminal fermentation kinetics of Moringa oleifera leaf and seed as protein feeds in dairy cow diets: in sacco degradability and protein and fiber fractions assessed by the CNCPS method. Agroforestry. Syst. 94(3), 905–915. Etchin, J., Kanki, J.P. and Look, A.T. 2011. Zebrafish as a model for the study of human cancer. Methods Cell Biol. 105, 309–337. Ferraccioli, G., Bracci-Laudiero, L., Alivernini, S., Gremese, E., Tolusso, B. and De Benedetti, F. 2010. Interleukin-1β and interleukin-6 in arthritis animal models: roles in the early phase of transition from acute to chronic inflammation and relevance for human rheumatoid arthritis. Mol. Med. 16(11), 552–557. Gong, P., Li, J., Wang, G., Guan, C., Meng, Z. and Jia, Y. 2023. Influence of reef structure and its flow field effect on the spatial behavior of Sebastes schlegelii adults. Front. Mar. Sci. 10, 1–10; doi:10.3389/fmars.2023.1185898 Hamid, O.I.A., Domouky, A.M. and El-Fakharany, Y.M. 2022. Molecular evidence of the amelioration of toluene induced encephalopathy by human breast milk mesenchymal stem cells. Sci. Rep. 12(1), 1–13; doi:10.1038/s41598-022-13173-6 Hayer, S., Niederreiter, B., Nagelreiter, I., Smolen, J. and Redlich, K. 2010. Interleukin 6 is not a crucial regulator in an animal model of tumour necrosis factor-mediated bilateral sacroiliitis. Ann. Rheumatic Dis. 69(7), 1403–1406. Higashi, Y., Maruhashi, T., Noma, K. and Kihara, Y. 2014. Oxidative stress and endothelial dysfunction: clinical evidence and therapeutic implications. Trends. Cardiovasc. Med. 24(4), 165–169; doi:10.1016/j.tcm.2013.12.001 Jaja-Chimedza, A., Graf, B.L., Simmler, C., Kim, Y., Kuhn, P., Pauli, G.F. and Raskin, I. 2017. Biochemical characterization and anti-inflammatory properties of an isothiocyanate-enriched moringa (Moringa oleifera) seed extract. PLos One 12(8), 182658. Katmawanti, S., Supriyadi. and Mariroh, F. 2021. Is instant porridge with a high calcium content based on Moringa oleifera as an alternative baby food to prevent stunting in Indonesia?. J. Public Health Res. 10(2), 2021. Kim, N.K., Zealous Gietbong, F., Andriyono, S., Kim, A.R. and Kim, H.W. 2018. The complete mitogenome of Bagrid catfish Chrysichthys nigrodigitatus (Siluriformes: claroteidae). Mitochondrial DNA Part B 3(2), 1239–1240. Lassi, Z.S., Das, J.K., Zahid, G., Imdad, A. and Bhutta, Z.A. 2013. Impact of education and provision of complementary feeding on growth and morbidity in children less than 2 years of age in developing countries: a systematic review. BMC. Public. Health. 13(SUPPL.3), 1–10; doi:10.1186/1471-2458-13-S3-S13 Lee, S.B., Park, Y.H., Chungu, K., Woo, S.J., Han, S.T., Choi, H.J., Rengaraj, D. and Han, J.Y. 2020. Targeted knockout of MDA5 and TLR3 in the DF-1 chicken fibroblast cell line impairs innate immune response against RNA ligands. Front. Immunol. 11, 678. Martínez-González, C.L., Martínez, L., Martínez-Ortiz, E.J., González-Trujano, M.E., Déciga-Campos, M., Ventura-Martínez, R. and Díaz-Reval, I. 2017. Moringa oleifera, a species with potential analgesic and anti-inflammatory activities. Biomed. Pharmacother. 87, 482–488. McGonagle, D., Sharif, K., O’Regan, A. and Bridgewood, C. 2020. Interleukin-6 use in COVID-19 pneumonia related macrophage activation syndrome. Autoimmun. Rev. 19, 102537. Nakai, N., Murata, M., Nagahama, M., Hirase, T., Tanaka, M., Fujikawa, T., Nakao, N., Nakashima, K. and Kawanishi, S. 2003. Oxidative DNA damage induced by toluene is involved in its male reproductive toxicity. Free Radical Res. 37(1), 69–76; doi:10.1080/1071576021000033103 Primihastuti, D., Wardani, D.W.K.K., Primaditya, V., Cory’ah, F.A.N., Ariati, L.I.P., Kalsum, U., Khotimah, H., Ali, M.M. and Riawan, W. 2022. The effect of ethanol extract of pegagan (Centella asiatica) on bone ossification and osteoclastogenesis on the stunting model of Zebrafish (Danio rerio) larvae induced by rotenone. GSC Biol. Pharm. Sci. 19(1), 91–99. Reyes Sánchez, N., Spörndly, E. and Ledin, I. 2006. Effect of feeding different levels of foliage of Moringa oleifera to creole dairy cows on intake, digestibility, milk production and composition. Livestock Sci. 101(1–3), 24–31. Sun, B., Liu, M., Tang, L., Hu, C., Huang, Z. and Chen, L. 2021. Probiotics inhibit the stunted growth defect of perfluorobutanesulfonate via stress and thyroid axes in zebrafish embryo. Environ. Pollut. 290, 118013. Seyoum, A., Pradhan, A., Jass, J. and Olsson, P.E. 2020. Perfluorinated alkyl substances impede growth, reproduction, lipid metabolism and lifespan in Daphnia magna. Sci. Total Environ. 737, 139682. Van Der Pol, C.W., Van Roovert-reijrink, I.A.M., Maatjens, C.M., Gussekloo, S.W.S., Kranenbarg, S., Wijnen, J., Pieters, R.P.M., Schipper, H., Kemp, B. and Van Den Brand, H. 2019. Light-dark rhythms during incubation of broiler chicken embryos and their effects on embryonic and post hatch leg bone development. PLos One 14(1), e0210886; doi:10.1371/JOURNAL.PONE.0210886 Walker, S.P., Chang, S.M., Powell, C.A. and Grantham-McGregor, S.M. 2005. Effects of early childhood psychosocial stimulation and nutritional supplementation on cognition and education in growth-stunted Jamaican children: prospective cohort study. Lancet 366(9499), 1804–1807; doi:10.1016/S0140-6736(05)67574-5 Wang, C.Y., Liang, C.Y., Feng, S.C., Lin, K.H., Lee, H.N., Shen, Y.C., Wei, L.C., Chang, C.J., Hsu, M.Y., Yang, Y.Y., Chiu, C.H. and Wang, C.Y. 2017. Analysis of the interleukin-6 (-174) locus polymorphism and serum IL-6 levels with the severity of normal tension glaucoma. Ophthalmic. Res. 57(4), 224–229. Wijayanti, A.R., Ridlayanti, A. and Ali, H.K.M. 2021. Protection of pegagan (Centella asiatica) extract through Hsp60 and bax expression on stunting model zebrafish larvae (Danio rerio) by rotenone inducted. J. Phys.: Conf. Ser. 2021 1764(1), p 12019. Wu, L., Zhong, L., Ru, H., Yao, F., Ni, Z. and Li, Y. 2022. Thyroid disruption and growth inhibition of zebrafish embryos/larvae by phenanthrene treatment at environmentally relevant concentrations. Aquatic. Toxicol. 243, 106053. Xia, R., Li, S., Yang, H., Zhao, Y., Teame, T., Hao, Q., Xie, M., Li, M., Chen, Q., Song, C., Ran, C., Yang, Y., Zhang, Z. and Zhou, Z. 2024. Effects of dietary bacterial culture of fengycin-producing Bacillus subtilis on growth performance, intestinal and hepatopancreas health, intestinal microbiota and disease resistance of common carp (Cyprinus carpio). Aquaculture 585, 740725. Ye, C., Zheng, S., Jiang, D., Lu, J., Huang, Z., Liu, Z., Zhou, H., Zhuang, C. and Li, J. 2021. Initiation and execution of programmed cell death and regulation of reactive oxygen species in plants. Int. J. Mol. Sci. 22(23), 12942. Supplementary data
The process of weighing and drying the simplicia of Moringa leaves (M. oleifera) in an oven at 40°C.
Process for preparing embryonic medium: all ingredients (CaCl 0.08 g, KCl 0.06 g, NaCl 2 g, MgSO4 3.2 g) were weighed using a digital balance and put in a test tube containing 200 ml filtered water, then stirred until the material was mixed thoroughly, bottled, labeled (date of manufacture), and stored in the fridge as stock.
Plates containing zebrafish and mosquito larvae are in an incubator at 37°C.
Body length observations of zebrafish and mosquito larvae were used with a stereo microscope (Olympus SZ61) linked to Optilab software and then measured using a calibrated image raster software. | ||
| How to Cite this Article |
| Pubmed Style Mafruchati M, Fadliyah L, Wardhana AK, Hassan II. Effect of Moringa oleifera leaf extract on body length, malondialdehyde, and interleukin-6 levels in zebrafish embryo (Danio rerio)-induced stunting model with rotenone. Open Vet. J.. 2026; 16(2): 934-943. doi:10.5455/OVJ.2026.v16.i2.15 Web Style Mafruchati M, Fadliyah L, Wardhana AK, Hassan II. Effect of Moringa oleifera leaf extract on body length, malondialdehyde, and interleukin-6 levels in zebrafish embryo (Danio rerio)-induced stunting model with rotenone. https://www.openveterinaryjournal.com/?mno=286613 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.15 AMA (American Medical Association) Style Mafruchati M, Fadliyah L, Wardhana AK, Hassan II. Effect of Moringa oleifera leaf extract on body length, malondialdehyde, and interleukin-6 levels in zebrafish embryo (Danio rerio)-induced stunting model with rotenone. Open Vet. J.. 2026; 16(2): 934-943. doi:10.5455/OVJ.2026.v16.i2.15 Vancouver/ICMJE Style Mafruchati M, Fadliyah L, Wardhana AK, Hassan II. Effect of Moringa oleifera leaf extract on body length, malondialdehyde, and interleukin-6 levels in zebrafish embryo (Danio rerio)-induced stunting model with rotenone. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 934-943. doi:10.5455/OVJ.2026.v16.i2.15 Harvard Style Mafruchati, M., Fadliyah, . L., Wardhana, . A. K. & Hassan, . I. I. (2026) Effect of Moringa oleifera leaf extract on body length, malondialdehyde, and interleukin-6 levels in zebrafish embryo (Danio rerio)-induced stunting model with rotenone. Open Vet. J., 16 (2), 934-943. doi:10.5455/OVJ.2026.v16.i2.15 Turabian Style Mafruchati, Maslichah, Lailatul Fadliyah, Akhmad Kusuma Wardhana, and Intan Idiana Hassan. 2026. Effect of Moringa oleifera leaf extract on body length, malondialdehyde, and interleukin-6 levels in zebrafish embryo (Danio rerio)-induced stunting model with rotenone. Open Veterinary Journal, 16 (2), 934-943. doi:10.5455/OVJ.2026.v16.i2.15 Chicago Style Mafruchati, Maslichah, Lailatul Fadliyah, Akhmad Kusuma Wardhana, and Intan Idiana Hassan. "Effect of Moringa oleifera leaf extract on body length, malondialdehyde, and interleukin-6 levels in zebrafish embryo (Danio rerio)-induced stunting model with rotenone." Open Veterinary Journal 16 (2026), 934-943. doi:10.5455/OVJ.2026.v16.i2.15 MLA (The Modern Language Association) Style Mafruchati, Maslichah, Lailatul Fadliyah, Akhmad Kusuma Wardhana, and Intan Idiana Hassan. "Effect of Moringa oleifera leaf extract on body length, malondialdehyde, and interleukin-6 levels in zebrafish embryo (Danio rerio)-induced stunting model with rotenone." Open Veterinary Journal 16.2 (2026), 934-943. Print. doi:10.5455/OVJ.2026.v16.i2.15 APA (American Psychological Association) Style Mafruchati, M., Fadliyah, . L., Wardhana, . A. K. & Hassan, . I. I. (2026) Effect of Moringa oleifera leaf extract on body length, malondialdehyde, and interleukin-6 levels in zebrafish embryo (Danio rerio)-induced stunting model with rotenone. Open Veterinary Journal, 16 (2), 934-943. doi:10.5455/OVJ.2026.v16.i2.15 |