| Research Article | ||
Open Vet. J.. 2026; 16(1): 96-104 Open Veterinary Journal, (2026), Vol. 16(1): 96-104 Review Article First report on the prevalence and histopathological characterization of Histomonas meleagridis in backyard chickens from Batna Province, Eastern AlgeriaAbdelouahab Ouarest1*, Salah Meradi1, Ahmed Yassine Kalbaza1, Houari Hemida2, and Mustapha Adnane Smadi31Laboratory of Environment, Health and Animal Production (LESPA), Avian Pathology Service, Institute of Veterinary and Agronomic Sciences, University of Batna 1, Batna, Algeria 2Ibn Khaldoun University of Tiaret, Department of Biomedicine, Institute of Veterinary Sciences,University of Tiaret, Tiaret, Algeria 3Biotechnology Research Centre (C.R.Bt), Constantine, Algeria *Corresponding Author: Abdelouahab Ouarest. Laboratory of Environment, Health and Animal Production (LESPA), Avian Pathology Service, Institute of Veterinary and Agronomic Sciences, University of Batna1, Algeria. Email: a.ouarest [at] univ-batna.dz Submitted: 29/09/2025 Revised: 08/12/2025 Accepted: 23/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
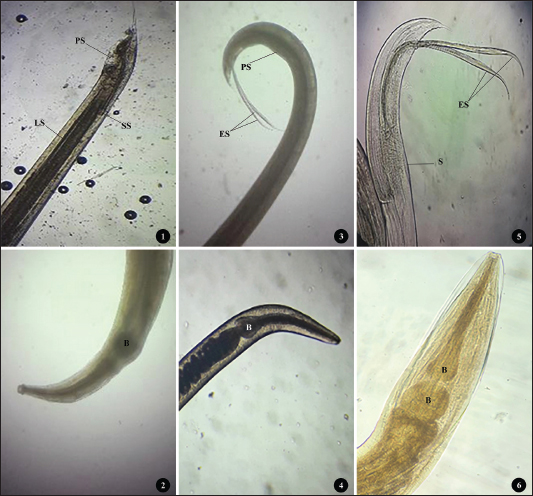
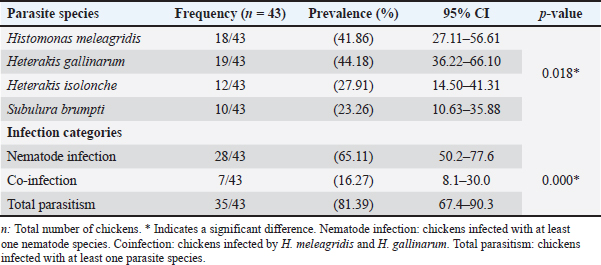
AbstractBackground: Histomonas meleagridis is a protozoan parasite responsible for histomoniasis, a severe disease in turkeys that causes characteristic cecal and hepatic lesions and potentially high mortality. Chickens are generally less affected but are recognized as important reservoirs and asymptomatic carriers. Despite its global distribution, the epidemiological situation of histomoniasis in Algerian backyard poultry remains poorly documented. This is of particular concern because backyard flocks, including turkeys, are often located near commercial poultry farms, and no effective treatment is currently available. Aim: This study aimed to investigate the prevalence and histopathological characterization of H. meleagridis in backyard chicken farms in the Batna region (Eastern Algeria). Methods: A total of 43 backyard chickens (Gallus gallus domesticus, Linnaeus, 1758), including 17 males and 26 females, were examined from September 2024 to May 2025. The backyard farms (n=20) within the study region were randomly selected. Chickens presenting clinical signs suggestive of histomoniasis were submitted for diagnostic investigation and screened for H. meleagridis infection using macroscopic and microscopic methods. Positive and negative H. meleagridis samples were subsequently evaluated using a lesion scoring system to assess the severity of cecal and hepatic lesions. Results: A total of 21 chickens (48.83%) from 12 farms showed macroscopic lesions typical of histomoniasis, whereas 18 chickens (41.86%) from 13 farms tested positive through histological analysis. Among the 43 chickens examined, 19 were infected with Heterakis gallinarum, corresponding to a prevalence of 44.18%. Co-infection with both parasites was detected in 7 birds (16.27%). Infected chickens exhibited significantly more severe lesions in the liver and ceca than uninfected birds (p < 0.05). No significant difference was found between lesions in H. meleagridis-infected birds and those co-infected with H. gallinarum. Conclusion: To the best of our knowledge, this is the first report on the prevalence and histopathological characterization of H. meleagridis in backyard chicken farms in Batna province, Eastern Algeria. Our findings suggest that H. meleagridis is the primary cause of cecal lesions, whereas H. gallinarum does not significantly aggravate the lesions. Given the proximity of these farms to commercial poultry operations, especially turkey farms, the study highlights the pathogenic importance of H. meleagridis, which could pose a serious threat to the commercial poultry industry, particularly due to the limited availability of effective treatments. Keywords: Histomonas meleagridis, Heterakis gallinarum, Backyard chickens, Parasitology, Algeria. IntroductionHistomoniasis, also known as enterohepatitis or enzootic typhlohepatitis, is a disease caused by the protozoan parasite Histomonas meleagridis. It primarily affects turkeys and other gallinaceous birds (Tyzzer, 1920). Transmission occurs mainly through the ingestion of embryonated eggs of the cecal nematode Heterakis gallinarum or via earthworms that have ingested these eggs (McDougald, 2005; Singh et al., 2022). While highly pathogenic in turkeys, causing severe cecal and hepatic lesions with mortality rates up to 90% (Nguyen et al., 2015), chickens are less susceptible and often serve as asymptomatic reservoirs (Hess et al., 2015; Eduardo et al., 2025). Until recently, antihistomonal drugs were used to prevent or treat histomoniasis. However, recent legislation in the European Union and the United States has prohibited these compounds due to food safety concerns (Liebhart et al., 2017). Currently, no approved curative treatments or effective commercial vaccines are available, making histomoniasis a persistent threat to livestock, especially in free-range poultry (Abd El-Wahab et al., 2021; Palmieri et al., 2021). In Algeria, backyard poultry farming is widespread and frequently involves close contact between chickens and turkeys under unsanitary conditions. This practice may facilitate the spread of H. meleagridis and potentially impact nearby farms. Despite the global documentation of histomoniasis, its prevalence and pathological impact on Algerian backyard flocks remain poorly understood. This study aimed to investigate the prevalence of H. meleagridis and characterize associated histopathological lesions in backyard chickens in the Batna region (Eastern Algeria), providing new insights into the epidemiological situation of this infection and its implications for poultry health management. Materials and MethodsSample collectionThis study was conducted from September 2024 to May 2025 in Batna, Eastern Algeria. A total of 20 backyard farms (each housing approximately 30–100 birds) were randomly selected within the study area. From these farms, 43 domestic chickens (Gallus gallus domesticus, Linnaeus, 1758), including 17 males and 26 females, exhibiting clinical signs suggestive of histomoniasis, were submitted for diagnostic investigation to the Avian Pathology Service at the Veterinary Sciences Institute, Batna-1 University. Clinical signs recorded by field veterinarians included apathy, depression, and ruffled feathers with decreased feed and water uptake (Liebhart and Hess, 2019). Most cases involved dead chickens transported under refrigerated conditions, whereas morbid birds were euthanized by jugular section. All submitted birds were included in the study, regardless of the presence or absence of macroscopic lesions observed during necropsy. Because the exact chronological age of backyard chickens is often unknown or imprecisely reported by owners, age classification was performed at necropsy based on the development of the reproductive organs. Birds were classified as adults or subadults based on the development of their reproductive organs. The presence of developed ovaries and oviducts in females or well-developed testicles in males indicates adulthood, whereas their absence indicates subadult status (Makouloutou-Nzassi et al., 2024; Adcock et al., 2025). Postmortem necropsic examinations were conducted according to standard procedures (Zander et al., 1997). Gross lesionsFresh samples from the liver and caeca were examined for gross lesions of histomoniasis as described by Nguyen et al. (2015) and Liu et al. (2018). The observed lesions included pronounced caecal swelling and wall thickening, accompanied by hemorrhagic patches on the caecal mucosa and the presence of a caseous core within the lumen. Livers were also assessed for enlargement and the presence of ulcerative, circular depressions on the surface. These hepatic lesions were characterized by a light yellow to yellowish–green necrotic center surrounded by raised peripheral margins, giving them a distinctive crater-like appearance. Birds showing any of the above-mentioned lesions were initially considered positive for histomoniasis. The macroscopic lesion score (MLS) system proposed by Callait-Cardinal et al. (2006)was used to score cecal and liver lesions. They were classified as follows: (0) none; (1) moderate thickening of the cecal wall with normal cecal content; (2) moderate thickening of the cecal wall with a caseous core partially filling the lumen and slightly hemorrhagic mucosa; (3) severe thickening of the cecal wall with a caseous core partially filling the lumen and hemorrhagic mucosa; and (4) severe thickening of the cecal wall with a caseous core filling the lumen and epithelial necrosis of the mucosa. Similarly, we graded liver lesions based on the number of target-like yellowish-green circular depressed areas as follows: (0) none; (1) one to five small foci; (2) more than five small foci; (3) numerous small and large foci; and (4) numerous large foci with extended necrosis. Nematodes were collected from ceca, rinsed in 0.85% normal saline, and fixed in 70% ethanol (Zajac et al., 2021). They were later identified and counted using a stereoscopic microscope and a standard morphological key guide (Soulsby, 1982). HistopathologyRepresentative tissue samples from the ceca and liver were collected from areas with visible lesions, fixed in 10% neutral-buffered formalin (tissue: fixative ratio 1:10), and stored in labeled containers for 24–48 hours. Samples were processed at the Histopathology Laboratory, Institute of Veterinary Sciences, Ibn Khaldoun University of Tiaret. Samples were embedded in paraffin, sectioned, and stained with hematoxylin and eosin for histological examination. Five-micrometer-thick sections were cut on a manual rotary microtome and then examined under a light microscope to detect H. meleagridis and evaluate microscopic lesions of the ceca and liver (Abdelhamid et al., 2020). The identification of protozoal trophozoites within the liver or cecal tissues was considered a confirmation of histomoniasis. Cecal and liver lesions were scored according to Callait-Cardinal et al. (2006) histological lesion score (HLS). Cecal lesions were graded as follows: (0) no lesion, (1) mild inflammatory infiltration, (2) extensive nodular infiltration by mononuclear cells and/or large mucosal erosions, (3) widespread epithelial necrosis, and (4) necrotic and fibrinous enteritis. Liver lesions were graded as follows: (0) no lesion, (1) mild mononuclear cell infiltrates near portal vessels, (2) few necrotic foci, (3) multiple necrotic foci, and (4) extensive parenchymal necrosis. Statistical analysisPearson’s chi-square test was used to compare prevalence across nematode species, infection categories, and host factors (sex and age). Lesion scores were analyzed using the Mann–Whitney U test because the data did not meet the normality assumption. Descriptive statistics were calculated, with p < 0.05 considered significant. All analyses were performed using SPSS version 23.0. Ethical approvalThe Scientific Council of the Institute of Veterinary Sciences and Agronomic Sciences at Batna 1 University, Algeria, issued and approved an ethical permit (Ethical approval No. 02/2024). ResultsPrevalence of parasitismOf the 43 examined chickens, 35 were infected with at least one parasite, corresponding to an overall prevalence of 81.39% (Table 1). Nematodes were detected in 28 birds (65.11%), with infection intensities ranging from 1 to 333 worms (mean=32). Heterakis gallinarum was the most prevalent species, identified in 19 chickens (44.18%) (Fig. 1:1–2). Heterakis isolonche was detected in 12 birds (27.91%) (Fig. 1:3–4), whereas Subulura brumpti was found in 10 birds (23.26%) (Fig. 1:5–6).
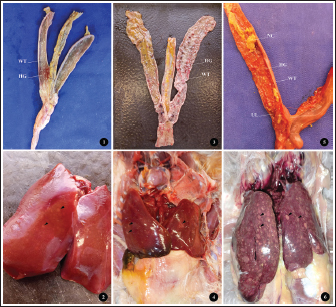
Fig. 1. (1-2) H. gallinarum: tail end of an adult male worm showing large unequal spicules; small (SS) and long spicule (LS), protruding circular precloacal sucker (PS), and anterior region with single esophageal bulb (B). (3-4) H. isolonche: tail end of male worm showing long and equal spicules (ES), preanal protruding sucker (PS), and anterior region with single esophageal bulb (B). (5-6) S. brumpti: tail end of male worm showing equal spicules (ES), preanal slit-like precloacal sucker with muscular striation (S), and anterior region with double esophageal bulb (B). Table 1. Prevalence of parasite species and infection categories in backyard chickens.
Histomoniasis was confirmed histologically in 18 chickens (41.86%) through microscopic visualization of H. meleagridis trophozoites in cecum and/or liver sections (Fig. 5; 1-2). Of these 18 infected birds, 17 showed macroscopic lesions, while one did not. Co-infection with H. meleagridis and H. gallinarum was observed in seven cases (16.27%). At the flock level, 13 of 20 backyard farms had at least one positive chicken, resulting in a prevalence of 65%.
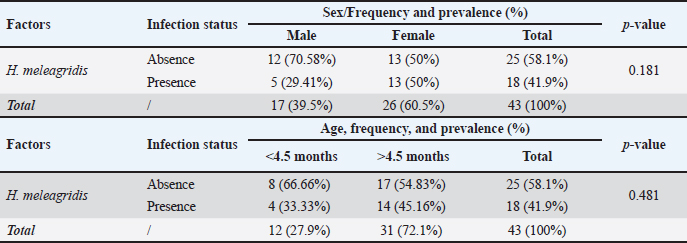
Fig. 5. Histopathological changes in livers and ceca of backyard chickens infected with histomoniasis. (1) Numerous histomonads (arrowhead) in the liver (HE, ×400). (2) Numerous histomonads (arrowhead) in ceca (HE, ×400). (3) Histological section of the liver showing periportal mononuclear infiltrate (black arrow), extensive parenchymal necrosis (red arrowhead), and steatosis (black arrowhead); (HE, ×400). (4) Histological section of ceca with large mucosal erosion (black arrow), extensive mononuclear infiltration (red arrowhead), and necrotic and fibrinous enteritis (black arrowhead); (HE, ×400). Pearson’s chi-square test showed a significant difference among the parasite species (p=0.018) and among the three infection categories: nematode infection, H. meleagridis coinfection with H. gallinarum, and overall parasitism prevalence (p=0.000) (Table 1). Table 2 shows the sex- and age-related distributions. The prevalence of H. meleagridis infection in male and female chickens was 5/17 (29.41%) and 13/26 (50%), respectively, with no significant difference between the two sexes (p=0.181). Additionally, there was no significant difference in prevalence between different age groups (p=0.481), with 45.16% (14/31) adult chickens and 33.33% (4/12) subadult chickens. Table 2. Influence of sex and age factors on the prevalence of H. meleagridis infection.
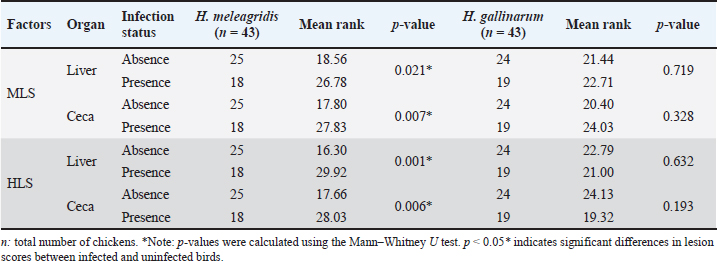
Pathological studyA total of 86 organ samples (43 liver and 43 ceca) from 43 chickens were examined to evaluate the association between H. meleagridis infection and lesion severity. Lesions were scored macroscopically (MLS) and histologically (HLS), and comparisons were made between infected and non-infected birds (Table 3). Table 3. Influence of H. meleagridis and H. gallinarum infection on macroscopic and HLS.
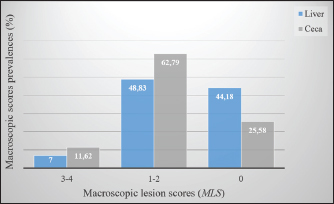
Macroscopic examination of the ceca and liver revealed variable severity among the examined birds (Fig. 2). Severe liver lesions (MLS=3-4) were recorded in approximately 7% (3/43) of chickens, characterized by pronounced hepatomegaly, congestion, ulcerative defects, and diffused greenish yellow necrotic foci (Fig. 3:6). Mild to moderate lesions (MLS=1-2), consisting of limited foci of degeneration, were observed in 48.83% (21/43) of the samples (Fig. 3:2-4), while the remaining 44.18% (19/43) of the samples showed no detectable macroscopic alterations (MLS=0). Similarly, the gross examination of the ceca showed that five birds (11.62%) exhibited moderate to severe lesions (MLS=3-4). Affected ceca appeared distended and contained necrotic debris. The mucosa frequently displayed hemorrhages, necrotic areas, and ulcerations after removal of the luminal content (Fig. 3: 5). Mild to moderate lesions (MLS=1-2) were observed in 27 birds (62.79%), characterized by slight thickening of the cecal wall and occasional petechial hemorrhages (Fig. 3: 1-3). No gross lesions (MLS=0) were detected in the remaining 11 (25.58%) birds.
Fig. 2. Prevalence of macroscopic lesions in the liver and ceca of the chickens studied.
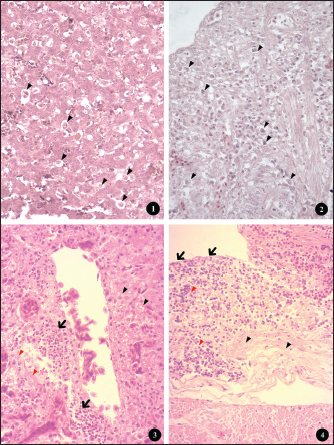
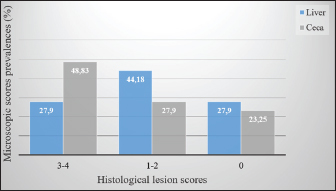
Fig. 3. (1, 3, and 5) Different degrees of MLS in affected ceca showing thickened intestinal walls (WT), hemorrhage (HG), ulceration (UL), and necrotic caseous material (NC). (2, 4, and 6) Different degrees of macroscopic lesions in the liver of affected chickens showing few to numerous small necrotic foci (arrowhead). Histopathological examination of the liver and ceca revealed a distribution consistent with the gross findings: 27.90% (12/43) of livers presented severe lesions (HLS=3-4), 44.18% (19/43) displayed mild to moderate lesions (HLS=1-2), and no histological changes were observed in 27.90% (12/43) of livers (HLS=0) (Fig. 4). Regardless of severity, H. meleagridis trophozoites, ranging from a few to numerous, were detected in hepatic tissue, mainly within fibrotic zones (Fig. 5: 1), exhibiting a dark central nucleus both extracellularly and within histiocytes. The histological sections of the liver show a periportal mononuclear infiltrate, extensive parenchymal necrosis, and steatosis (Fig. 5:3). Histological lesions of ceca were generally more prevalent and severe. Histomonas meleagridis trophozoites were frequently identified in association with damaged mucosa and submucosa (Fig. 5: 2). Severe lesions (HLS=3-4) were recorded in 21/43 (48.83%) samples, characterized by marked mucosal erosion, inflammatory cell infiltration, and abundant necrotic debris (Fig. 5:4). Mild-to-moderate lesions (HLS=1-2) were observed in 12/43 (27.90%) of cases, while 10/43 (23.25%) of ceca were histologically normal (HLS=0) (Fig. 4).
Fig. 4. Prevalence of histological lesions in the liver and ceca of the studied chickens. Regarding lesion severity, the samples without H. meleagridis consistently showed lower mean ranks in both organs (Table 3). For MLS, the mean rank in the liver was 18.56 in samples without H. meleagridis compared to 26.78 in infected samples (p=0.021). A similar trend was observed in the ceca, with the mean ranks increasing from 17.80 (absence) to 27.83 (presence) (p=0.007). A comparable pattern was observed for HLS. In the liver, the mean ranks were 16.30 in the absence of H. meleagridis and 29.92 when the parasite was present, indicating a highly significant difference (p=0.001). In the ceca, the mean rank increased from 17.66 (absence) to 28.03 (presence) (p=0.006). In contrast, H. gallinarum infection did not significantly affect either the macroscopic or HLS in the examined organs (p > 0.05). The mean ranks of the infected and non-infected groups were similar, indicating that H. gallinarum had no significant pathological impact (Table 3). DiscussionIn this study, we report the first histological detection of H. meleagridis in backyard chickens in the Batna province. The findings demonstrate that histomoniasis, which has been widely reported in several countries, also exists in backyard poultry farms in this region of Eastern Algeria and could pose an emerging risk to the national poultry sector. A high prevalence of intestinal parasitism was also observed in the study population, highlighting the importance of enteric infections in traditional poultry systems. This result is comparable to those reported in Morocco (Hassouni and Belghyti, 2006), Colombia (Vergara et al., 2021), and Gabon (Makouloutou-Nzassi et al., 2024). In contrast, lower rates were reported in the Kashmir Valley (Salam et al., 2018) and in the district of Odisha (Hembram et al., 2015). These inter-country variations may reflect differences in climate, management practices, flock health, and preventive measures. In our setting, the high prevalence can be explained by the lack of prophylaxis, close contact with other animal species, and poor hygiene conditions typical of backyard systems. Although free-range poultry farming supports animal welfare, it also increases exposure to diverse pathogens (Jeni et al., 2021). This is consistent with our finding that 65.11% of chickens carried at least one nematode species, highlighting gastrointestinal helminths as a major health issue in backyard flocks. Among the identified parasites, H. gallinarum showed the highest prevalence, followed by H. meleagridis, H. isolonche, and S. brumpti. Although less frequent, the presence of H. isolonche and S. brumpti demonstrates notable parasite diversity and reflects the considerable environmental contamination. However, the high occurrence of H. gallinarum is of particular concern because it is a biological vector of H. meleagridis, the causative agent of histomoniasis, a serious and often fatal disease in turkeys (de Jesus Ramires et al., 2025). The concomitant detection of H. gallinarum and H. meleagridis occurred in only 16.27% of cases, which contrasts with the widely reported synergy between the protozoan and the nematode (Eduardo et al., 2025). Comparable discrepancies have been documented internationally. In Nigeria, H. gallinarum and H. meleagridis were detected in only 14.7% and 10.3% of chickens, respectively, with coinfections accounting for 3.1% of cases (Oriaku et al., 2024). In contrast, Xu et al. (2018) reported a significantly higher prevalence of histomoniasis (94.6%) and a co-infection rate of 50.8% in China. Therefore, the relatively low co-infection rate observed in our study indicates that H. meleagridis could circulate independently of H. gallinarum under local conditions. Alternative transmission routes include direct bird-to-bird transmission via cloacal drinking and reverse peristalsis, insect ingestion, or human-mediated spread (Clarke et al., 2017). Several experimental and clinical studies have demonstrated that cecal infestation by H. meleagridis can reduce the establishment and development of H. gallinarum. Daş et al. (2021) suggested that the absence of a linear relationship between the number of histomonads and H. gallinarum might be explained by the pathological changes induced by histomonads in the cecal environment, which is the habitat of H. gallinarum. Similarly, Schwarz et al. (2011) reported that the inflammatory and destructive lesions of the cecal mucosa observed during H. meleagridis infections can alter the tissue and immune environment, thereby limiting the normal development of H. gallinarum. These mechanisms could explain the low rate of coinfection between the two parasites in our study. Considering the sex and age parameters, although females and adult chickens were more parasitized than males and subadult chickens, no statistically significant difference was found in the infection rate between the two sexes and age categories. This finding aligns with several experimental studies that also reported no statistical difference in infection levels between the age and sex of turkeys inoculated with H. meleagridis (Liebhart et al., 2008). The relatively small size of our sample, limited to 43 chickens (26 females and 17 males, including 12 young and 31 adults), constitutes a limitation to the interpretation of our results. Future work, including a larger sample that considers these variables, would be necessary to confirm or refute the actual impact of these factors on infection. The lesion patterns were consistent with previous histomoniasis descriptions in gallinaceous birds (De Araújo et al., 2015; Hess et al., 2015). Cecal enlargement with wall thickening, severe typhlitis with fibrinous content, and foci of necrosis in the liver were the main macroscopic changes. Simultaneously, histological examination of tissues from both organs confirmed the presence of H. meleagridis trophozoites. Lesion evaluation showed that histomoniasis-positive birds had more severe cecal and liver alterations than negative birds, regardless of the presence of H. gallinarum. These results confirm that H. meleagridis mainly causes tissue lesions, whereas H. gallinarum plays a limited direct pathogenic role and mainly acts as an epidemiological vector. These remarks are in perfect agreement with Salam et al. (2018). Interestingly, in histomoniasis-positive birds, cecal lesions were more severe than the hepatic lesions. This observation agrees with previous reports indicating that liver lesions are often mild or absent in chickens infected with H. meleagridis, whereas they are prominent in turkeys (Liebhart and Hess, 2019). Clarke et al. (2017) also reported that H. meleagridis genotype 2 can induce pronounced cecal damage without marked hepatic involvement. These findings indicate that lesion distribution and severity may be influenced by strain-specific virulence factors, underscoring the importance of parasite genetic variability for disease expression. ConclusionThe relatively high prevalence of H. meleagridis observed in the study population constitutes a novel and concerning finding for the region. These findings indicate active circulation of histomoniasis within backyard chicken flocks, representing a potential threat to commercial poultry farms, especially turkeys, which are highly susceptible to histomoniasis. In the absence of effective treatments and adequate biosecurity measures, the emergence and spread of the disease on a larger scale could be favored. This study represents the first step in understanding histomoniasis in backyard poultry in Algeria, paving the way for future research. The sample size and lack of molecular analyses create opportunities for additional studies to identify and characterize local H. meleagridis strains. Although focusing on the Batna region was a relevant starting point, it also emphasizes the need to expand research to other areas to better understand the distribution of parasites. AcknowledgmentsThe authors would like to express their gratitude to the veterinarians in charge for arranging the farm visits and sampling on the farms included in the study, as well as to the farmers for their consent and assistance during the farm visits. Conflict of interestThe authors declare no conflicts of interest. FundingThis research received no specific grant. Authors’ contributionsAbdelouahab Ouarest: conducted the experiment, collected and analyzed the data, and drafted the manuscript. Houari Hemida: performed the histopathological analysis and data interpretation. Ahmed Yassine Kalbaza and Mustapha Adnane Smadi: performed the statistical analysis and data interpretation. Salah Meradi: supervised the entire study, critically reviewed the manuscript for intellectual content, and approved the final version for publication. All authors have read and approved the final version of the manuscript. Data availabilityThe data that support the findings of this study are not openly available due to sensitivity reasons and are available from the corresponding author upon reasonable request. ReferencesAbd El-wahab, A., Visscher, C., Haider, W. and Dimitri, R. 2021. A case study of histomoniasis in fattening turkeys identified in histopathological investigations. German. J. Vet. Res. 1(3), 13–18; doi:10.51585/gjvr.2021.3.0015 Abdelhamid, M.K., Quijada, N.M., Dzieciol, M., Hatfaludi, T., Bilic, I., Selberherr, E., Liebhart, D., Hess, C., Hess, M. and Paudel, S. 2020. Co-infection of chicken layers with Histomonas meleagridis and avian pathogenic Escherichia coli is associated with dysbiosis, cecal colonization and translocation of the bacteria from the gut lumen. Front. Microbiol. 11, 1–17; doi:10.3389/fmicb.2020.586437 Adcock, K.G., Weyna, A.A.W., Yabsley, M.J., Bäck, R.E., Garrett, K.B., Niedringhaus, K.D., Kunkel, M.R., Fenton, H.M.A., Keel, M.K., Bahnson, C.S., Elsmo, E. and Nemeth, N.M. 2025. Trichomonad disease in wild Turkeys (Meleagris gallopavo): pathology and molecular characterization of Histomonas, Tetratrichomonas, Tritrichomonas, and Simplicimonas spp. J. Wildlife Dis. 61(1), 131–147; doi:10.7589/JWD-D-24-00050 Callait-Cardinal, M.P., Chauve, C., Reynaud, M.C., Alogninouwa, T. and Zenner, L. 2006. Infectivity of Histomonas meleagridis in ducks. Avian. Pathol. 35(2), 109–116; doi:10.1080/03079450600597626 Clarke, L.L., Beckstead, R.B., Hayes, J.R. and Rissi, D.R. 2017. Pathologic and molecular characterization of histomoniasis in peafowl (Pavo cristatus). J. Vet. Diagnostic Invest. 29(2), 237–241; doi:10.1177/1040638716687002 Daş, G., Wachter, L., Stehr, M., Bilic, I., Grafl, B., Wernsdorf, P., Metges, C.C., Hess, M. and Liebhart, D. 2021. Excretion of Histomonas meleagridis following experimental co-infection of distinct chicken lines with Heterakis gallinarum and Ascaridia galli. Parasites. Vectors. 14(1), 1–15; doi:10.1186/s13071-021-04823-1 De Araújo, J.L., Olinda, R.G., Frade, M.T.S., Maia, L. and Dantas, A.F.M. 2015. Histomoniasis outbreak in free-range chickens in semiarid Paraíba, Brazil. SeminaCiencias. Agrarias. 36(1), 307–312; doi:10.5433/1679-0359.2015v36n1p307 De Jesus Ramires, M., Hummel, K., Hatfaludi, T., Hess, M. and Bilic, I. 2025. Host-specific targets of Histomonas meleagridis antigens revealed by immunoprecipitation. Sci. Rep. 15(1), 5800; doi:10.1038/s41598-025-88855-y Eduardo, M.B.J., Alexis, R.G., Magnolia, F.C.B., Tellez-Isaias, G. and Shehata, A.A. 2025. Heterakis gallinarum and Histomonas meleagridis in laying hens reared in cage system: a case report. German. J. Vet. Res. 5(1), 1–7; doi:10.51585/gjvr.2025.1.0114 Hassouni, T. and Belghyti, D. 2006. Distribution of gastrointestinal helminths in chicken farms in the Gharb region - Morocco. Parasitol. Res. 99(2), 181–183; doi:10.1007/s00436-006-0145-8 Hembram, A., Panda, M.R., Mohanty, B.N., Pradhan, C.R., Dehuri, M., Sahu, A. and Behera, M. 2015. Prevalence of gastrointestinal helminths in Banaraja fowls reared in semi-intensive system of management in Mayurbhanj district of Odisha. Vet. World 8(6), 723–726; doi:10.14202/vetworld.2015.723-726 Hess, M., Liebhart, D., Bilic, I. and Ganas, P. 2015. Histomonas meleagridis-new insights into an old pathogen. Vet. Parasitol. 208(1–2), 67–76; doi:10.1016/j.vetpar.2014.12.018 Jeni, R.E., Dittoe, D.K., Olson, E.G., Lourenco, J., Seidel, D.S., Ricke, S.C. and Callaway, T.R. 2021. An overview of health challenges in alternative poultry production systems. Poultry Sci. 100(7), 101173; doi:10.1016/j.psj.2021.101173 Oriaku, L.O., Silas, I.I., Nnanna-Chigozie, E.P., Inyang, S.X., Gbadebo, A.O. and Jesse, S.B. 2024. Prevalence of gastrointestinal parasites of poultry chicken slaughtered and sold in Uyo Metropolis, Akwa Ibom State, Nigeria. Int. J. Pathogen Res. 13(5), 23–31; doi:10.9734/ijpr/2024/v13i5306 Liebhart, D. and Hess, M. 2019. Spotlight on Histomonosis (blackhead disease): a re-emerging disease in turkeys and chickens. Avian Pathol. 0(0), 1–4; doi:10.1080/03079457.2019.1654087 Liebhart, D., Ganas, P., Sulejmanovic, T. and Hess, M. 2017. Histomonosis in poultry: previous and current strategies for prevention and therapy*. Avian Pathol. 46(1), 1–18; doi:10.1080/03079457.2016.1229458 Liebhart, D., Grabensteiner, E. and Hess, M. 2008. A virulent mono-eukaryotic culture of Histomonas meleagridis is capable of inducing fatal histomonosis in different aged turkeys of both sexes, regardless of the infective dose. Avian. Dis. 52(1), 168–172; doi:10.1637/8107-090707-ResNote Liu, D., Kong, L., Tao, J. and Xu, J. 2018. An outbreak of histomoniasis in backyard sanhuang chickens. Korean. J. Parasitol. 56(6), 597–602; doi:10.3347/kjp.2018.56.6.597 Makouloutou-Nzassi, P., Longo-Pendy, N.M., Nguema, L.K.A., Lendzele, S.S., Bangueboussa, F., Bouchedi, B., Maganga, G.D. and Boundenga, L. 2024. Prevalence of gastrointestinal parasites in chickens (Gallus gallus domesticus) and associated risk factors in M’passa department, Southeast Gabon. Open Vet. J. 14(12), 3232–3240; doi:10.5455/OVJ.2024.v14.i12.8 Mcdougald, L.R. 2005. Blackhead disease (Histomoniasis) in poultry: a critical review. Avian. Dis. 49(4), 462–476; doi:10.1637/7420-081005R.1 Nguyen, D.T., Bilic, I., Jaskulska, B., Hess, M., Le, D.Q., Le Hua, L.N., Huynh, V.V., Nguyen, S.T. and Vu-Khac, H. 2015. Prevalence and genetic characterization of Histomonas meleagridis in Chickens in Vietnam. Avian Dis. 59(2), 309–314; doi:10.1637/10964-102414-reg Palmieri, N., De Jesus Ramires, M., Hess, M. and Bilic, I. 2021. Complete genomes of the eukaryotic poultry parasite Histomonas meleagridis: linking sequence analysis with virulence / attenuation. BMC. Genomics. 22(1), 1–18; doi:10.1186/s12864-021-08059-2 Salam, S.T., Wani, S.A., Dar, P.A. and Ahangar, F.A. 2018. Prevalence and pathology of Heterakis gallinarum mono-infection and co-infection with Histomonas meleagridis in free roaming domestic chicken of Kashmir Valley. Int. J. Academic. Res. Develop. 3(1995), 505–508. Schwarz, A., Gauly, M., Abel, H., Daş, G., Humburg, J., Weiss, A.T.A., Breves, G. and Rautenschlein, S. 2011. Pathobiology of Heterakis gallinarum mono-infection and co-infection with Histomonas meleagridis in layer chickens. Avian Pathol. 40(3), 277–287; doi:10.1080/03079457.2011.561280 Singh, M.N., Kesdangsakonwut, S., Jitsamai, W., Sonia, C., Phupolpan, C. and Taweethavonsawat, P. 2022. Molecular detection and characterization of Histomonas meleagridis in fighting cocks, Thailand. SSRN. Electron. J. 09(5), 7352–7363; doi:10.2139/ssrn.4195270 Soulsby, E.J.L. 1982. Helminths, arthropods and protozoars of domesticated animals, 7ème edition. London, UK: Bailliere Tindall, p 808. Tyzzer, E.E. 1920. The flagellate character and reclassification of the parasite producing “Blackhead” in Turkeys: Histomonas meleagridis (Gen. nov.) (Smith). J. Parasitol. 6(3), 124; doi:10.2307/3271065 Vergara, D., Alvarez, J. and Cordero, A. 2021. Prevalence of gastrointestinal parasites in three groups of domestic poultry managed under backyard system in the Savanna subregion, Department of Sucre, Colombia. J. Adv. Vet. Anim. Res. 8(4), 606–611; doi:10.5455/javar.2021.h551 Xu, J., Qu, C., Guo, P., Zhuo, Z., Liu, D. and Tao, J. 2018. Epidemic characteristics of clinical histomoniasis in chicken flocks in eastern China. Avian. Dis. 62(2), 189–194. Zajac, A.M., Conoboy, G.A., Little S.E. and Reichard, M.V. 2021. Veterinary clinical parasitology, 9th ed. Hoboken, NJ: Jhon Wiley & Sons, Inc. p 39. Zander, D.V., Bermudez, A.J. and Mallinson, E.T. 1997. Principles of disease prevention: diagnosis and control. In Diseases of poultry, 10th ed. Eds., Calnek, B.W., Barnes, H.J., Beard, C.W., McDougald, L.R. and Saif, Y.M. Ames, IA: Iowa State University Press, pp: 3–45. | ||
| How to Cite this Article |
| Pubmed Style Ouarest A, Meradi S, Kalbaza AY, Hemida H, Smadi MA. First report on the prevalence and histopathological characterization of Histomonas meleagridis in backyard chickens from Batna Province, Eastern Algeria. Open Vet. J.. 2026; 16(1): 96-104. doi:10.5455/OVJ.2026.v16.i1.9 Web Style Ouarest A, Meradi S, Kalbaza AY, Hemida H, Smadi MA. First report on the prevalence and histopathological characterization of Histomonas meleagridis in backyard chickens from Batna Province, Eastern Algeria. https://www.openveterinaryjournal.com/?mno=287224 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.9 AMA (American Medical Association) Style Ouarest A, Meradi S, Kalbaza AY, Hemida H, Smadi MA. First report on the prevalence and histopathological characterization of Histomonas meleagridis in backyard chickens from Batna Province, Eastern Algeria. Open Vet. J.. 2026; 16(1): 96-104. doi:10.5455/OVJ.2026.v16.i1.9 Vancouver/ICMJE Style Ouarest A, Meradi S, Kalbaza AY, Hemida H, Smadi MA. First report on the prevalence and histopathological characterization of Histomonas meleagridis in backyard chickens from Batna Province, Eastern Algeria. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 96-104. doi:10.5455/OVJ.2026.v16.i1.9 Harvard Style Ouarest, A., Meradi, . S., Kalbaza, . A. Y., Hemida, . H. & Smadi, . M. A. (2026) First report on the prevalence and histopathological characterization of Histomonas meleagridis in backyard chickens from Batna Province, Eastern Algeria. Open Vet. J., 16 (1), 96-104. doi:10.5455/OVJ.2026.v16.i1.9 Turabian Style Ouarest, Abdelouahab, Salah Meradi, Ahmed Yassine Kalbaza, Houari Hemida, and Mustapha Adnane Smadi. 2026. First report on the prevalence and histopathological characterization of Histomonas meleagridis in backyard chickens from Batna Province, Eastern Algeria. Open Veterinary Journal, 16 (1), 96-104. doi:10.5455/OVJ.2026.v16.i1.9 Chicago Style Ouarest, Abdelouahab, Salah Meradi, Ahmed Yassine Kalbaza, Houari Hemida, and Mustapha Adnane Smadi. "First report on the prevalence and histopathological characterization of Histomonas meleagridis in backyard chickens from Batna Province, Eastern Algeria." Open Veterinary Journal 16 (2026), 96-104. doi:10.5455/OVJ.2026.v16.i1.9 MLA (The Modern Language Association) Style Ouarest, Abdelouahab, Salah Meradi, Ahmed Yassine Kalbaza, Houari Hemida, and Mustapha Adnane Smadi. "First report on the prevalence and histopathological characterization of Histomonas meleagridis in backyard chickens from Batna Province, Eastern Algeria." Open Veterinary Journal 16.1 (2026), 96-104. Print. doi:10.5455/OVJ.2026.v16.i1.9 APA (American Psychological Association) Style Ouarest, A., Meradi, . S., Kalbaza, . A. Y., Hemida, . H. & Smadi, . M. A. (2026) First report on the prevalence and histopathological characterization of Histomonas meleagridis in backyard chickens from Batna Province, Eastern Algeria. Open Veterinary Journal, 16 (1), 96-104. doi:10.5455/OVJ.2026.v16.i1.9 |