| Research Article | ||
Open Vet. J.. 2026; 16(2): 809-816
Open Veterinary Journal, (2026), Vol. 16(2): 809-816 Research Article Histomorphological study of ovarian development in local Iraqi awassi sheep fetuses during the prenatal periodHiba Adeibel and Jafar Ghazi Abbas Al-Jebori*Department of Veterinary Anatomy, Veterinary College, Al-Qasim Green University, Babylon, Iraq *Corresponding Author: Jafar Ghazi Abbas Al-Jebori. Department of Veterinary Anatomy, Veterinary College, Al-Qasim Green University, Babylon, Iraq. Email: jafar [at] vet.uoqasim.edu.iq Submitted: 20/10/2025 Revised: 10/01/2026 Accepted: 18/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
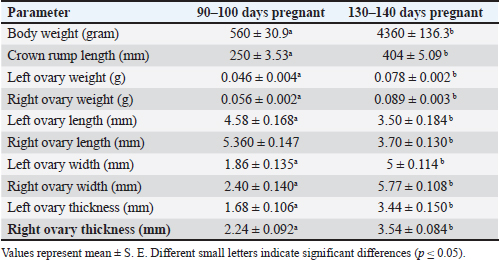
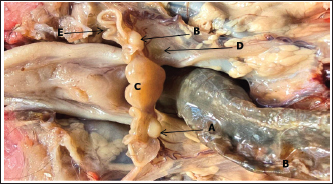
ABSTRACTBackground: Prenatal ovarian development is a critical process that determines future reproductive capacity, yet detailed morphological and histological data in sheep fetuses remain limited. Aim: This study aimed to investigate the prenatal morphological and histological development of sheep ovaries at different gestational stages. Methods: Ten fetuses were obtained from healthy pregnant ewes and classified into two gestational groups based on crown–rump length: Group I (90–100 days) and Group II (140–145 days). Gross anatomical observations were performed, followed by histological examination of ovarian tissues. Results: In Group I, the ovaries were small and elongated, with asymmetry in shape, as the left ovary appeared pear-shaped while the right was more uniformly elongated. Both ovaries were translucent, whitish, and located caudal to the metanephros within the abdominal cavity. Histologically, the ovaries consisted of distinguishable cortical and medullary regions based on the distribution of germ cells and undifferentiated mesenchymal cells. In Group II, the ovaries were larger, elongated, and more clearly defined, exhibiting a translucent whitish to creamy appearance. Microscopically, a well-developed cortex and medulla were evident, with a distinct tunica albuginea, clearly defined germinal epithelium, and a densely cellular cortex containing primordial, primary, and secondary follicles. Conclusion: Fetal sheep ovaries exhibit marked morphological and histological maturation from the second to the third trimester, with major ovarian differentiation and early folliculogenesis occurring predominantly during late gestation, thereby establishing the structural foundation for postnatal reproductive function. Keywords: Development, Fetuses, Ovary, Prenatal, Sheep. IntroductionAwassi sheep are the main local breed in Iraq. They are highly adapted to heat and drought; provide excellent milk, meat, and wool with strong disease resistance, and have low management costs. Approximately 65% of all livestock in Iraq are sheep (FAO, 2010), which are considered highly economical ruminants for various types of production. The female reproductive system includes two ovaries and a tract composed of two uterine tubes, uterus, cervix, vagina, and vulva (Samuelson, 2007). The ovaries perform dual function: the exocrine production of ova and the endocrine secretion of sex hormones, mainly progesterone and estrogen (Deokar et al., 2005). The ovary surface is covered by a simple squamous serosal epithelium, beneath which lies a thin layer of vascularized, dense, irregular connective tissue called the tunica albuginea. The ovary is organized into the cortex and medulla. The cortex contains clusters of fibroblasts (stromal cells), interstitial gland cells, and theca cells involved in follicular development (Liebich, 2019). The cortex contains follicles at various stages of development (primordial, primary, secondary, and antral), a small fraction of which will progress to become mature, ovulatory follicles, ready to rupture and release the oocyte, a process called ovulation. The medulla consists of large blood vessels surrounded by loose connective tissue containing a sufficient amount of elastic and reticular fibers (Samuelson, 2007; Devi, 2016). Few studies have examined ovarian development in ewe fetuses, particularly in the Awassi breed. Sheep represent a suitable model for this investigation due to their low cost, short gestation, and the fact that ovine prenatal ovarian development closely resembles that of humans. Additionally, we expanded the background information regarding the significance of studying ovarian development in sheep. The revised text now explains the benefits of understanding ovarian growth for improving reproductive efficiency, enhancing fertility management, and supporting advanced reproductive technologies, such as estrus synchronization and artificial insemination. This is the first detailed morphological and histological description of fetal ovarian development in the Awassi breed in Iraq at different fetal stages. Materials and MethodsThe current study was conducted at Al-Qasim Green University in College of Veterinary Medicine to describe the histomorphological developmental changes of the ovary in the prenatal periods of local Awassi sheep’s fetuses (Ovis Aries). Ovarian samples were collected from pregnant ewes that had been slaughtered from September to November for prenatal research. The fetuses were selected based on their normal morphological appearance, absence of congenital abnormalities, and availability at the desired gestational ages. Only healthy fetuses with intact membranes and no signs of autolysis were included in the study and distributed into two groups (five fetuses for each group): at end the of the first and second trimester of gestation according to the gestational age, which was determined based on (CRL) (the crown rump length a standard embryonic and fetal measurement that represents the distance from the top of the head (crown) to the base of the buttocks (rump). It is commonly used in developmental biology, veterinary science, and obstetrics by using the following formula (Y=2.74X + 30.15), where “Y” is the developmental age of the fetus in days and “X” is the crown-rump length in cm (Mohassen and Al-Jebori, 2020; Hamid and Al-Jebori, 2025), and morphological parameters are achieved by balance and vernier caliper. The sheep fetus's ovary was preserved in 10% neutral buffered formalin, dehydrated through a graded alcohol series, cleaned in xylene, and then embedded in paraffin wax. Using a rotary microtome, the blocks were sectioned at 5–6 μm thickness of slice, and histological sections were stained with Mason's trichrome stain (for the connective tissue), (PAS) periodic acid schiff (for basement membrane), Alcian blue (Alcian Blue, it helps to visualize glycosaminoglycans and extracellular matrix components in structures such as follicular basal lamina, stromal tissue, and developing follicles) and hematoxylin and eosin (show the generally compound of tissues) (Banchroft et al., 2018; Niyf and Al-Jebori, 2024; Ali and Al-Jebori, 2024). Data analysis of histological and morphological parameters was performed using the Statistical Package for the Social Sciences (SPSS) version 24. A one-way analysis of variance was performed, and differences were considered significant at p < 0.05. Ethical approvalThe experimental procedures were approved by the Institutional Animal Ethics Committee and conducted at the Veterinary Medicine College, Al-Qassim Green University, Iraq. Ethical regulation of this work was assigned by the university research committee and adhered to the guidelines of the American Veterinary Medical Association under approval number (35 in 20112025). ResultsEnd of the second trimester of gestation (90–100) daysMorphological resultsThe body weight of sheep fetuses was 560 ± 30.99 gram and the crown rump length was 250 ± 3.53 mm (Table 1). The ovaries are small and elongated in shape, with the left ovary being pear-shaped, while the right ovary is elongated, smooth surfaces, translucent, and whitish in color. They were located in the abdominal cavity caudally to the metanephros and highly attached to the uterine body and horn, covered by small and large intestinal loop (Fig. 1). The ovaries were laterally attached by the mesentery (mesovarium). The left ovary was adjacent to the left metanephros and cranially continuous to reach the left oviduct, where the right ovary was located caudally to the right metanephros. Overall, the left ovary had a more extensive relationship to the right metanephros than the right ovary (Fig. 2). Table 1. Quantitative assessment of fetal biometric traits and ovarian morphometry at two gestational stages in foetuses by balance and Vernia caliper.
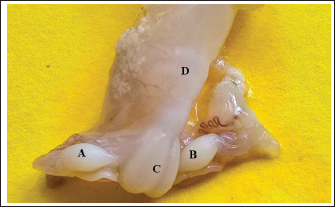
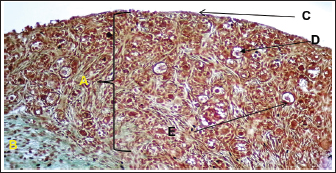
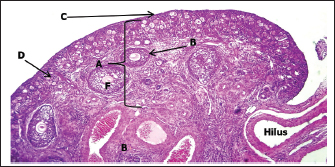
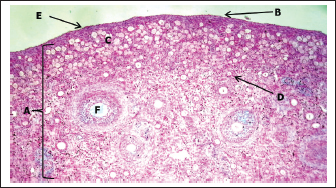
Histological examination showed that the ovary comprises two main regions: cortex and medulla, which were clearly distinguishable based on the distribution patterns of undifferentiated mesenchymal cells and germ cells. The tunica albuginea was clearly undifferentiated, and the right ovary was more developed and larger than the left ovary (Fig. 3). The cortical region consisted of densely packed cells, whereas the medulla appeared more diffuse and loosely organized (Fig. 4). The germinal epithelium gives rise to the surface layer; the “invagination” or “down-growth” of this epithelium is a normal developmental process that forms the cortical cords and rete ovarii, which were apparent. At this stage, germ cells at this stage were observed predominantly at the oogonial phase, appearing in clusters scattered along the ovarian periphery. They were closely associated with the surrounding mesenchymal tissue and arranged within ovarian sex cords, which were irregular in shape and extended from the germinal epithelium toward the developing cortex. These cords were surrounded by loose mesenchymal tissue, resulting in the formation of oogonia (primordial germ cells). The cortex contained numerous ovarian follicles at various stages of development, extending from the germinal epithelial zone to the medulla, and could be categorized into small, medium, and large sizes with many germ cells, particularly those in the center of the ovary (Figs. 4, 5). In contrast to the cortex, the medulla does not directly participate in follicle formation but plays a crucial role in the overall development and future physiological function of the ovary. The medullary region forms the central core of the fetal ovary and is surrounded by the developing cortex, which is the primary site of folliculogenesis. The medulla comprises loose connective tissue rich in undifferentiated mesenchymal cells and fibroblasts, along with well-developed blood vessels. The mesenchymal cells are spindle-shaped fibroblast-like cells embedded in a collagen fiber matrix, providing structural support and contributing to the ovarian stroma. Compared with the cortex, the medullary stroma is less cellular and more fibrous. Vascular elements in the medulla appear as small to medium-sized capillaries and venules, contributing to the early establishment of ovarian circulation. Scattered primitive interstitial cells are present among vascular structures. Notably, follicular structures are generally absent in the medulla (Fig. 6; Table 2).
Fig. 1. Photograph illustrates morphological structures of ovary at 90th day of gestation in sheep fetuses showed: A: Right ovary; B: Left ovary; C: Uterus; D: Metanephros; E: Oviduct.
Fig. 2. Photograph illustrates morphological structures of ovary at 90th day of gestation in sheep fetuses showed: A: Right ovary; B: Left ovary; C: Uterus; D: Vagina.
Fig. 3. Histological section showing the left ovary at 90-100 days of gestation. A: Cortex; B: Medulla; C: Germinal layer (H&E. 4X).
Fig. 4. Histological section showing the left ovary at 90–100 days of gestation. A: Cortex; B: Medulla; C: Germinal layer; D: Primordium follicle; E: Primary follicles (Masson's stain:10X).
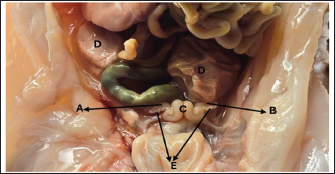
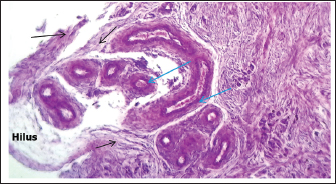
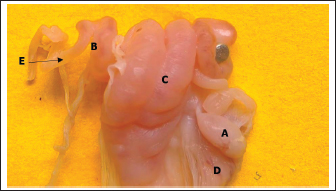
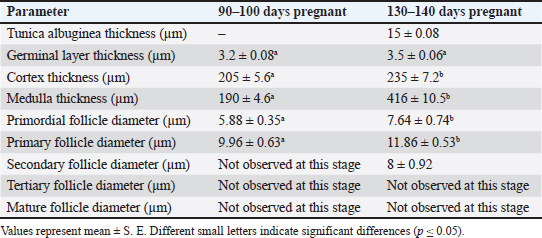
Fig. 5. Histological section showing the left ovary cortex and medulla at 90–100 days of gestation. A: Cortex; B: Medulla; C: Germinal layer; D: Primordium follicles; E: Primary follicles; F: Sex cords (periodic acid schiff stain:20X). End of the third trimester of gestation (130–140) daysMorphological results: The body weight of fetuses' 4360 ± 136.3 gram and the crown rump length 404 ± 5.09 mm (Table 1). Both ovaries more clearly and still growth with advancement of age to appear enlarged in size comparative with second trimester with elongated translucent whitish to creamy in color situated in the abdominal cavity, had two surfaces (medial and lateral) and two borders (free border, attachment border) and two pole (cranial and caudal) hanging by mesovarium to lateral abdominal wall caudally to metanephros on each side of uterus in abdominal cavity to fixative by obvious and well development broad ligament (Figs. 7,8).
Fig. 6. Histological section showing the right ovary medulla at 90–100 days of gestation at ovarian stroma showed distribution of the connective tissue stroma in the medulla (black arrows) and between ovarian blood vessels (blue arrows) (H&E:10X).
Fig. 7. Photograph illustrates morphological structures of ovary at 140–145 day of gestation in sheep fetuses. A: Right ovary; B: Left ovary; C; Uterus; D: Broad ligament; E: Oviduct.
Fig. 8. Photograph illustrates morphological structures of ovary at 140–145 day of gestation in sheep fetuses. A: Right ovary; B: Left ovary; C: Uterus; D: Broad ligament; E: Oviduct. Table 2. Quantitative assessment of ovarian tissue layers and follicular diameters at two gestational stages in foetuses.
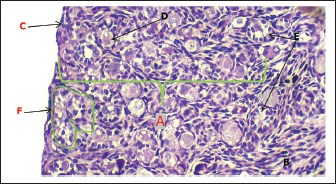
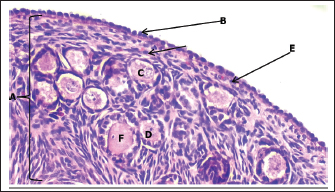
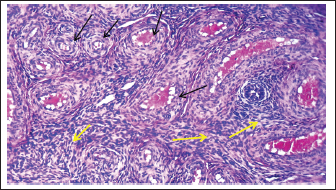
Histological examination revealed that the fetal ovary undergoes significant structural maturation with age. It is composed of a well-developed cortex and medulla with a clear tunica albuginea with well-organized architecture and clearly defined germinal epithelium, and a densely cellular cortex containing primordial (single layer of flattened granulosa cells), primary (single layer of cuboidal granulosa cells), and secondary follicles (multiple layers of granulosa cells) (Fig. 9). The ovaries contain major components of the mature ovary, except for the tertiary and Graafian follicles. The germinal epithelium appears as a strip of cells surrounding the surface of the fetal ovary, consisting of cuboidal cells with rounded nuclei, and positioned on the visible basement membrane, and some flat cells with elongated nuclei (Fig. 10). There is a dense connective tissue layer in the tunica albuginea beneath the germinal epithelium. It is progressively more defined during the third trimester of gestation as the ovary undergoes differentiation and compartmentalization to become more prominent and well organized, helping to demarcate the cortical region of the ovary from the germinal epithelium with the disappearance of the sex cords from the sub-germinal region, compared to the second trimester of gestation, in which the sex cords were clearly visible.
Fig. 9. Histological section showing the ovary at 140–145 days of gestation in sheep fetuses. A: Cortex; B: Medulla; C: Germinal layer; D: Primordium follicles; E: Primary follicles; F: Secondary follicles (H&E. 4X).
Fig. 10. Histological section showing the ovary cortex at 140–145 days of gestation in sheep fetuses. A: Cortex; B: Germinal layer; C: Primordium follicles; D: Primary follicles; E: Tunica albuginea; F: Secondary follicles (H&E. 20X).
Fig. 11. Histological section showing the ovary cortex at 140–145 days of gestation in sheep fetuses. A: Cortex; B; Germinal layer; C: Primordium follicles; D: Primary follicles; E: Tunica albuginea; F: Secondary follicles (Alcian blue stain. 10X).
Fig. 12. Histological section showing the right ovary medulla at 140–145 days of gestation at ovarian stroma showed distribution of the connective tissue stroma in the medulla (yellow arrows) and between ovarian blood vessels (black arrows) (H&E:10X). The cortex exhibited a high density of follicles at various developmental stages. Numerous primordial follicles were observed in the outer cortical zone, each comprising an oocyte surrounded by a single layer of flattened pre-granulosa cells. Early primary follicles, identified by a single layer of cuboidal granulosa cells, indicate the initiation of follicular activation. Fewer secondary follicles deeper in the cortex were detected, characterized by multiple layers of granulosa cells and early differentiation of the theca layer beyond the basement membrane, although no antral formation was noted (Fig. 11). The ovarian medulla appears to be loose connective tissue rich in collagen fibers, fibroblasts, and an increasing number of blood vessels, supporting ovarian vascularization with active fibroblasts and scattered primitive mesenchymal cells. No developing follicles are present in the medulla at this point. The emerging medullary architecture reflects a dynamic and preparatory phase of ovarian development, ensuring proper reproductive and endocrine function in the future (Fig. 12, Table 2). DiscussionThe morphological observations indicate that sheep fetuses exhibited normally developed ovaries that were small, elongated, and asymmetrically shaped, with the left ovary showing a closer anatomical association with the metanephros than the right. The caudal abdominal position, mesovarian attachment, and close relationship with the uterine structures reflect the typical developmental organization of the fetal female reproductive system, suggesting normal spatial differentiation during this stage of gestation. The results of the current study were parallel with (Deoker et al., 2005 and Kumari et al., 2017), who noted that the ovary in the second trimester in sheep and goat is oval in shape and attached to the mesonephros by mesovarium. In contrast, in the dromedary camel (Abdel-Elrazik et al., 2013) described fetal ovaries were described during the 4th and 5th months of gestation as circular or bean-shaped. Similarly, El-Ghannam and El-Nagger (1974) reported that buffalo fetal ovaries are oval or spindle-shaped with symmetrical thickening during the third month of gestation. During the second trimester, the fetal ovary exhibits clear cortical–medullary differentiation, with oogonia arranged in cortical sex cords and early follicles, indicating active germ cell proliferation and early folliculogenic commitment. In contrast, the follicle-free, vascularized medulla plays a primarily stromal and supportive role, while the relatively advanced development of the right ovary suggests physiological asymmetry during fetal ovarian morphogenesis. The current findings were in agreement with those of Deoker et al. (2005), who reported that the ovary lacked a clearly defined tunica albuginea in sheep fetuses at 46–60 days of gestation. The cortex and medulla can be distinguished in buffalo. During the second trimester, the fetal ovary undergoes significant morphological with cellular changes, and the gonads are at an advanced stage of differentiation with tunica albuginea remains poorly developed (Sharma et al., 2014) and conformable (Sharum, 2023) in ovine fetuses at 95 days age, the fetal ovary displays distinct histological features indicative of early folliculogenesis and the cortical region is densely populated with primordial follicles, predominantly situated just beneath the ovarian surface epithelium. A smaller proportion of primary follicles was observed more centrally within the cortex without secondary or antral follicles. In contrast, the findings of the current study disagree with those of Aruna et al. (2017), who reported that the ovary does not exhibit a clear distinction between cortex and medulla in goat fetuses at 60 days of gestation. Germ cells are organized into ovarian cords, which consist of both somatic or pre-granulosa cells and oogonia. The observed interspecies differences in the histological organization of the ovary can be attributed to inherent variations in developmental timing, genetic programming, and fetal endocrine regulation. Each species follows a distinct ovarian differentiation trajectory; therefore, structures such as the tunica albuginea, cortical and medullary regions, and ovarian cords may appear clearly defined in some species at a given gestational age while remaining less developed in others. Variation in gestation length and fetal hormonal profiles—particularly estrogen, follicles stimulating hormone (FSH), and growth factors—further modulates germ cell proliferation, stromal maturation, and the folliculogenesis onset. In addition, maternal physiological conditions contribute to differences in stromal organization and vascularization. Consequently, the discrepancies reported among studies reflect species-specific developmental dynamics and differences in the embryonic ages examined, rather than true contradictions. The increased fetal body weight and crown–rump length in the third trimester of gestation were accompanied by clear ovarian enlargement, reflecting advanced ovarian growth with gestational progression. The elongated, translucent ovaries and their well-defined anatomical features, including distinct surfaces, borders, and poles, indicate structural maturation compared with earlier stages of development. Their stable positioning and firm suspension by the mesovarium and well-developed broad ligament suggest functional stabilization of the ovaries within the abdominal cavity during late fetal development. The present study in third trimester, showed that both ovaries more clearly and still growth with advancement of age to appear enlarged in size comparative with second trimester with elongated translucent whitish to creamy in color situated in the abdominal cavity and inconsistent with in the goat and camel fetuses, the two ovaries were pin head, oval in shape with smooth surface, cream in color to descended from below the kidneys to the pelvic brim adjacent to the uterine tubes in third trimester of gestation (Pratap et al., 2012) and with (Abdel-Elrazik et al., 2013) in dromedary Camel who mentioned that the final pelvic position of the fetal ovaries was demonstrated in fetuses during second trimester of gestation. The observed decrease in ovarian length at 130–140 days of gestation does not indicate arrested development but rather reflects gestation-dependent fetal ovary morphological reorganization. During late prenatal life, volumetric expansion and cortical thickening driven by follicular assembly, stromal maturation, and increased vascularization, ovarian growth. These processes promote a shift from a relatively elongated configuration to a shorter, wider, and thicker organ. Histological analysis in the third stage of development demonstrated advanced age-dependent maturation of the fetal ovary, characterized by clear cortical–medullary differentiation, a well-defined germinal epithelium, and a prominent tunica albuginea. The cortex showed a high density of follicles at early developmental stages (primordial, primary, and limited secondary), indicating active follicular organization without antral or Graafian formation. In contrast, the medulla consisted of loosely arranged connective tissue rich in fibroblasts and blood vessels, with no follicular structures, supporting its primarily stromal and vascular role during late fetal ovarian development. In sheep, this finding coincides with Douker et al. (2005) that the germinal epithelium becomes more prominent and appears flattened. Beneath it, the cortex appears darker in staining with numerous follicles at various stages of development, whereas the medulla is lighter and contains sparse blood vessels. In goats, the contemporary study agrees with the findings of Aruna et al. (2017), during the third trimester of gestation, the ovary can be distinctly divided into two main regions: the cortex and the medulla, while in bovine (El-Ghannam and El-Naggar, 1974) at (7–8) months, the tunica albuginea reaches its maximum thickness and the germinal epithelium is flattened, with the cortex consisted of germinal elements, while medulla consisted of vascularized connective tissue lacking germ cells. ConclusionsThis study showed that fetal sheep ovaries undergo notable morphological and histological development during the second and third trimesters. By 90–100 days, the ovaries were small, elongated, and translucent with indistinct cortex and medulla, clusters of oocytes, and developing ovarian cords, while the tunica albuginea was poorly defined. By 130–140 days, the ovaries were enlarged, with a well-developed tunica albuginea, organized cortical and medullary regions, and follicles progressing to the secondary stage, although tertiary follicles were absent. These findings indicate that major ovarian differentiation and folliculogenesis occur mainly during the third trimester, preparing the ovary for postnatal reproductive function and providing a crucial baseline for understanding normal ovine fetal development. AcknowledgementThe authors gratefully acknowledge the support of Al-Qasim Green University and thank all colleagues and staff who contributed to this study's success. FundingNo funding was received. Author’s contributionHiba Adeibel and Jafar Ghazi Abbas Al-Jebori confirm that they contributed equally to the paper. Conflict of interestThe authors have no conflicts to declare. Data availabilityThe datasets used for analysis during are available from the corresponding author on reasonable request. ReferencesAbdel-Elrazik, A.M.A., Nawito, M.F., El-Sayed, T. and Mahmoud, K.G.M. 2013. Prenatal morphological development of the ovary in dromedary camel (Camelus dromedaries). Glob. Vet. 11(4), 372–376;doi:10.5829/idosi.gv.2013.11.4.7683 Ali, R.T. and Al-Jebori, J.G.A. 2024. Prenatal and histomorphological development of fungiform and circumvallate papillae in local Awassi sheep (Ovis arise). J. Anim. Health. Prod. 12(S1), 187–195;doi:10.17582/journal.jahp/2024/12.s1.187.195 Bancroft, J. D., Suvarna, K. S. and Layton, C. 2018. Bancroft’s theory and practice of histological techniques. 8th ed. London: Elsevier Health Sciences. https://doi.org/10.1016/C2015-0-00143-5 Deokar, M. S., Dhande, P. L., Patil, A. D. and Gaikwad, S. A. 2005. Histomorphological and histochemical changes in the ovary and oviduct in prenatal sheep (Ovis aries).https://www.researchgate.net/publication /257138806_Histomorphological_and_Histochemical_changes_in_the_Ovary_and_Oviduct_in_Prenatal_Sheep_Ovis_aries?utm_source=chatgpt.com Devi, S. V. 2016. Basic histology: A color atlas and text. JP Medical Ltd. doi: 10.5005/jp/books/12791 El-Ghannam, F. and El-Naggar, M.A. 1974. The prenatal development of the buffalo ovary. J. Reprod. Fertil. 36(3), 479–483; doi:10.1530/jrf.0.0410479 Food and Agriculture Organization of the United Nations (FAO).(2010). Production Yearbook. Vol. 196. Rome, Italy: FAO; 2010. p. 20. Available from https://www.fao.org/3/i2100e/i2100e. Hamid, M.H. and Al-Jebori, J.G.A. 2025. Histomorphological developmental study of the uterus in local Awassi sheep fetuses during the postnatal period. J. Anim. Health Prod. 13(s1), 181–186. Kumari, A.G., Amin, R.U. and Rao, K.S. 2017. Morphometric and histological characterization of fetal goat ovaries. J. Dairy Vet. Sci. 3, 1–7; doi: 10.19080/JDVS.2017.03.555605 Liebich, H.G. 2019. Veterinary histology of domestic mammals and birds 5th edition: textbook and colour atlas. 5th ed., Sheffield, UK: 5m Books Ltd. Available via https://5mbookscom/product/veterinary-histology-of-domestic-mammals-and-birds-5theditiion-textbook-and-colouratlas?utm_source=chatgpt.com Mohassen, F.W. and Al-Jebori, J.G.A. 2020. Ontogenesis of thyroid gland in awassi sheep foetuses: prenatal study. Plant Arch. 20(1), 1096–1100;https://www.plantarchives.org/special%20issue%2020-1/1096 Niyf, A.M. and Al-Jebori, J.G.A. 2024. Ontogenesis of rectum in local Awassi sheep foetuses (Ovis aries) during prenatal periods. J. Anim. Health. Prod. 12(s1), 45–54; doi:10.17582/journal.jahp/2024/12.s1.45.54 Pratap, N., Manjunatha, B.M. and Al Bulushi, S. Incidence of Early Pregnancy Loss in Dromedary Camels (Camelus dromedarius). In Proc of the third conference of international society of camelid research and development, 2012 ,poloniae, italya pp 109–110. Samuelson. 2007. Textbook of veterinary histology. St.Louis, USA: Saunders Elsevier, pp: 442–486. Sharma, R.K., Jerome, A. and Purohit, G.N. 2014. Reproductive physiology of the male and female buffalo. Theriogenology 5702, 0614; doi:10.1016/j.theriogenology.2014.04.014 Sharum, I.B. 2023. Determination of the ovine ovarian reserve during the prenatal and neonatal periods. Asian. Pacific. J. Reprod. 12(4), 194–200; doi:10.4103/2305-0500.380986 | ||
| How to Cite this Article |
| Pubmed Style Adeibel H, Al-jebori JGA. Histomorphological study of ovarian development in local Iraqi awassi sheep fetuses during the prenatal period. Open Vet. J.. 2026; 16(2): 809-816. doi:10.5455/OVJ.2026.v16.i2.4 Web Style Adeibel H, Al-jebori JGA. Histomorphological study of ovarian development in local Iraqi awassi sheep fetuses during the prenatal period. https://www.openveterinaryjournal.com/?mno=291654 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.4 AMA (American Medical Association) Style Adeibel H, Al-jebori JGA. Histomorphological study of ovarian development in local Iraqi awassi sheep fetuses during the prenatal period. Open Vet. J.. 2026; 16(2): 809-816. doi:10.5455/OVJ.2026.v16.i2.4 Vancouver/ICMJE Style Adeibel H, Al-jebori JGA. Histomorphological study of ovarian development in local Iraqi awassi sheep fetuses during the prenatal period. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 809-816. doi:10.5455/OVJ.2026.v16.i2.4 Harvard Style Adeibel, H. & Al-jebori, . J. G. A. (2026) Histomorphological study of ovarian development in local Iraqi awassi sheep fetuses during the prenatal period. Open Vet. J., 16 (2), 809-816. doi:10.5455/OVJ.2026.v16.i2.4 Turabian Style Adeibel, Hiba, and Jafar Ghazi Abbas Al-jebori. 2026. Histomorphological study of ovarian development in local Iraqi awassi sheep fetuses during the prenatal period. Open Veterinary Journal, 16 (2), 809-816. doi:10.5455/OVJ.2026.v16.i2.4 Chicago Style Adeibel, Hiba, and Jafar Ghazi Abbas Al-jebori. "Histomorphological study of ovarian development in local Iraqi awassi sheep fetuses during the prenatal period." Open Veterinary Journal 16 (2026), 809-816. doi:10.5455/OVJ.2026.v16.i2.4 MLA (The Modern Language Association) Style Adeibel, Hiba, and Jafar Ghazi Abbas Al-jebori. "Histomorphological study of ovarian development in local Iraqi awassi sheep fetuses during the prenatal period." Open Veterinary Journal 16.2 (2026), 809-816. Print. doi:10.5455/OVJ.2026.v16.i2.4 APA (American Psychological Association) Style Adeibel, H. & Al-jebori, . J. G. A. (2026) Histomorphological study of ovarian development in local Iraqi awassi sheep fetuses during the prenatal period. Open Veterinary Journal, 16 (2), 809-816. doi:10.5455/OVJ.2026.v16.i2.4 |