| Case Report | ||
Open Vet. J.. 2026; 16(1): 747-753 Open Veterinary Journal, (2026), Vol. 16(1): 747-753 Case Report Middle cranial fossa syndrome presumptively secondary to leishmaniasis in a dog: A case reportSergio Sánchez Briones*, Alba Farré Mariné and Alejandro Luján Feliu-PascualNeurology/Neurosurgery Service, Hospital Veterinario AUNA Especialidades Veterinarias IVC Evidensia, Valencia, Spain *Corresponding Author: Sergio Sánchez Briones. Neurology/Neurosurgery Service, Hospital Veterinario AUNA Especialidades Veterinarias IVC Evidensia, Valencia, Spain. Email: sergio.sanchez [at] ivcevidensia.es Submitted: 28/10/2025 Revised: 30/11/2025 Accepted: 05/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
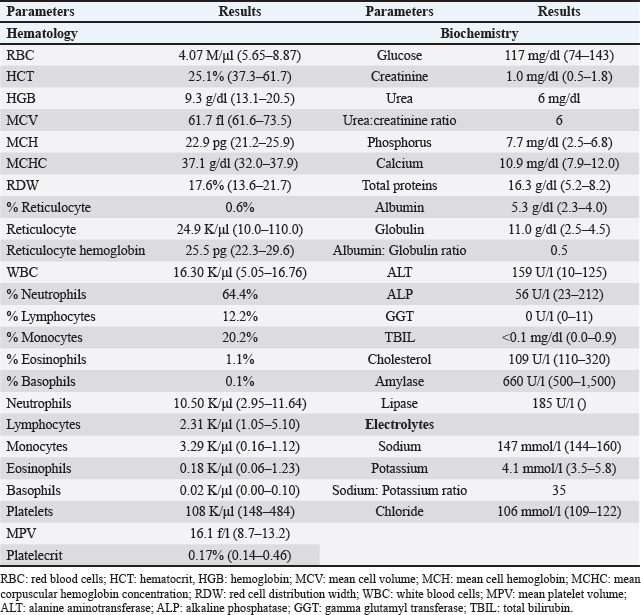
AbstractBackground: Middle cranial fossa syndrome refers to a group of neurological signs caused by damage to the oculomotor, trochlear, abducent, and trigeminal nerves and sympathetic innervation to the eye as they cross the middle cranial fossa due to different pathologies. Neurological deficits include external ophthalmoparesis or ophthalmoplegia, ptosis, reduced or absent direct and indirect pupillary light reflex, mydriasis, and reduced or absent corneal and nasal sensation. Neoplasia is the most common etiology in dogs and cats. Canine leishmaniosis is a vector-borne infectious disease caused by Leishmania spp., which has been shown to affect several organs. Although rare, cases of damage to the central and peripheral nervous systems have been reported; however, no cases with signs of middle cranial fossa syndrome have been described. Case Description: A 16-month-old male neutered 1.5 kg cross breed dog was presented for evaluation with neurological signs consistent with a multifocal neuroanatomical localization (middle cranial fossa, optic chiasm, and cervical and thoracolumbar spinal cord). Laboratory tests revealed hematological and biochemical abnormalities compatible with an infectious process. Computed tomography revealed osteolytic lesions of the basisphenoid bone at the middle cranial fossa and several vertebrae. Cytological analysis of the vertebral lesions identified Leishmania spp. amastigotes Treatment with meglumine antimoniate, allopurinol, and prednisone resulted in rapid clinical and radiological improvement. Conclusion: Leishmaniasis is a new potential etiology for middle cranial fossa syndrome in dogs. Keywords: Dog, Leishmania, Middle cranial fossa syndrome. IntroductionThe middle cranial fossa syndrome (MCFS), previously misnamed as cavernous sinus syndrome, is an infrequent but easily recognized group of neurological signs characterized by dysfunction of one or more of the following cranial nerves CN: III (oculomotor nerve), IV (trochlear nerve), VI (abducent nerve), and the maxillary and ophthalmic branches of V (trigeminal nerve) as they course rostrally through the middle cranial fossa in the base of the cranium (Rossmeisl et al., 2005; Jones et al., 2018). In addition, sympathetic innervation to the eye may be impaired. All these nerves exit the cranial vault through the same opening, the orbital fissure, located between the presphenoid and basisphenoid bones (Rossmeisl et al., 2005; Jones et al., 2018). To recognize this condition, it is imperative to understand the function of the previously mentioned nerves. The general somatic efferent neurons of the oculomotor nerve innervate the striated voluntary extraocular muscles (dorsal, medial, and ventral rectus muscles and the ventral oblique muscle of the eyeball) and the upper eyelid levator palpebrae muscle. Parasympathetic general visceral efferent neurons of CN III innervate the smooth muscles of the iris through the ciliary muscles. The trochlear nerve only carries general somatic efferent axons to the striated extraocular dorsal oblique muscle. The abducent nerve supplies general somatic efferent input to the striated extraocular lateral rectus and retractor bulbi muscles. Finally, the trigeminal nerve (maxillary, mandibular, and ophthalmic) carries general sensory afferent information from the face and head organs, parasympathetic visceral efferent innervation to the salivary glands, and general somatic efferent innervation to the masticatory muscles. Neurological deficits associated with MCFS include external ophthalmoparesis or ophthalmoplegia, reduced or absent direct and indirect pupillary light reflexes, mydriasis, and reduced or absent corneal and facial sensation (Rossmeisl et al., 2005; Jones et al., 2018). Neoplasia is the most common etiology of MCFS in dogs and cats, with infectious causes being the second most common (Jones et al., 2018). In cats, feline infectious peritonitis, toxoplasmosis, and cryptococcosis have been associated with MCFS (Theisen et al., 1996). Furthermore, an additional report described two dogs and one cat with suspected sphenoid bone osteomyelitis and neurological deficits compatible with MCFS (Busse et al., 2009). Leishmaniasis is a zoonotic systemic disease caused by protozoan parasites of the genus Leishmania spp. (Baneth and Solano-Gallego, 2022). Leishmania infantum is the most common species in dogs in Europe and is considered an endemic parasite in Spain. Because of its pathogenesis, clinical presentations can be very variable. The main clinical signs associated with leishmaniasis are weight loss, weakness, skin lesions, ocular abnormalities, epistaxis, and signs of renal disease such as polyuria and polydipsia. Moreover, dogs may develop gastrointestinal signs and lameness (Baneth and Solano-Gallego, 2022). Nervous system involvement is considered rare. However, some dogs diagnosed with Leishmania spp. and neurological signs of central and peripheral nervous system dysfunction have been reported in the literature (Giannuzzi et al., 2017). To date, no cases of dogs with MCFS secondary to Leishmania spp. have been described. The following report describes the clinical signs, imaging findings, treatment, and outcome of a dog with MCFS presumptively secondary to L. infantum. Case DetailsA 16-month-old male neutered 1.5 kg cross breed dog was evaluated for a 2-week history of progressive lethargy, disorientation, and vision loss. Symptomatic treatment with meloxicam (undisclosed dose) was administered before referral, with no improvement. Physical examination revealed moderate hyperthermia (rectal temperature 40ºC) and a low body score condition. Neurological examination showed that the dog was mildly obtunded during the consultation. Postural reactions were considered normal in all four limbs, as were myotatic spinal reflexes. Cranial nerve evaluation revealed bilateral absent menace responses, bilateral absent vestibulo-ocular reflexes, bilateral mydriasis, and bilateral absent direct and indirect pupillary light reflexes. The dog exhibited hyperesthesia during the palpation of the cervical and thoracolumbar regions. Based on the neurological examination, a multifocal lesion was suspected and localized to the middle cranial fossa, optic chiasm, and cervical and thoracolumbar vertebral column. The differential diagnoses included meningoencephalitis of unknown origin, infectious meningoencephalitis (Crytococcus spp., Neospora caninum, Toxoplasma gondii, and Leishmania spp.), or neoplasia (e.g., meningioma, mast cell tumor, or lymphoma). A complete blood cell count, serum biochemistry, and electrolytes (Table 1) showed normocytic normochromic non-regenerative anemia 25.1% (Reference range RR: 37.3%–61.7%), monocitosis 3.29 k/μl (RR: 0.16–1.12k/μl), thrombocytopenia 108 k/μl (RR: 148–484k/μl), low urea 6 mg/dl (RR: 7–27 mg/dl), hyperphosphatemia 7.7 mg/dl (RR: 2.5–6.8 mg/dl), elevated alanine aminotransferase 7.7 mg/dl (RR: 2.5–6.8 mg/dl), low cholesterol 109 mg/dl (RR: 110–320 mg/dl), low lipase 185 mg/dl (RR: 200–1,800 mg/dl), hypochloraemia 106 mmol/l (RR: 109–122 mmol/l), hyperalbuminemia 5.3 g/dl (RR: 2.3 g–4 g/dl), hyperglobulinemia 11 g/dl (RR: 2.5–4.5 g/dl), and hyperproteinaemia 16.3 g/dl (RR: 5.2 g–8.2 g/dl). Table 1. Hematology, biochemistry, and electrolytes are tested at the moment of diagnosis.
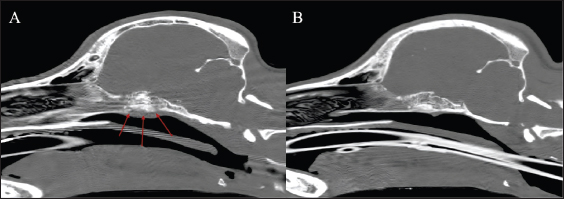
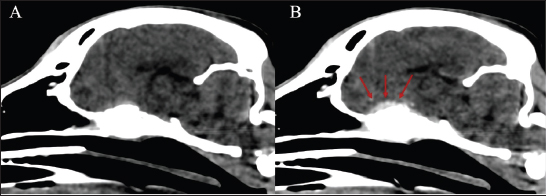
The dog received general anesthesia and underwent a computed tomography scan of the head and cervical vertebral column before and after iodinated intravenous contrast injection with iohexol at 400 mg/kg (Omnipaque, General Electrics Healthcare, Cork, Ireland). Anesthesia was induced with propofol (Propomitor, Orion Pharma, Madrid, Spain) at 4 mg/kg IV and was maintained with 1.5% isoflurane (Isofane, Piramal Critical Care B.V., Voorschoten, Netherlands) in oxygen under controlled ventilation. A 16-slice helical CT scanner (Brivo CT385, General Electrics Healthcare, Madrid, Spain) was used for imaging acquisition. The images revealed an aggressive polyostotic, osteolytic, and osteoproliferative lesion with periosteal reaction affecting the basisphenoid and presphenoid bones and, therefore, compromising the optic canal and orbital fissure (Fig. 1). The lesion extended intracranially at the level of the rostral and middle cranial fossa as an isoattenuating, ill-defined extra-axial space-occupying mass with marked and homogenous contrast enhancement (Fig. 2). No abnormalities were found in the cervical column images. A cisternal cerebrospinal fluid tap was performed, and the sample was analysed, the results of which were within the reference ranges. Polymerase chain reaction in the cerebrospinal fluid was not performed as the total nucleated cell count was within normal limits.
Fig. 1. Bone window (A) pre-treatment sagittal reconstruction and (B) post-treatment sagittal reconstruction of the head, revealing the osteolytic and osteoproliferative lesions (arrows) affecting the sphenoid bone and its resolution after treatment.
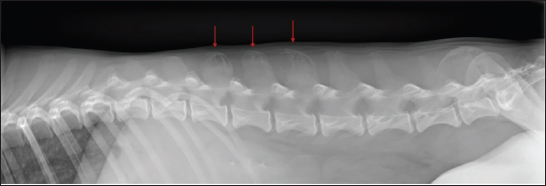
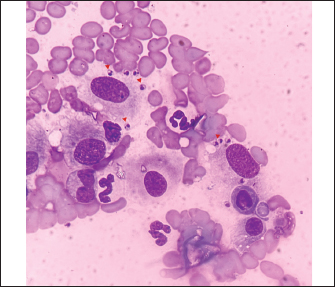
Fig. 2. Soft tissue window sagittal reconstruction of the head (A) pre-contrast and (B) post-contrast administration, showing the contrast-enhancing extra-axial lesion affecting the sphenoid bone (red arrows). Lateral thoracic and lumbar radiographs were performed to localize any additional osteolytic foci due to the multifocal hyperesthesia. Polyostotic radiolucent moth-eaten lesions were observed at the L1, L2, and L3 dorsal processes (Fig. 3). Subsequently, an abdominal ultrasound was performed, showing heterogeneous echogenicity of the spleen and liver parenchyma and enlarged iliac lymphatic nodules. Fine needle aspirates were obtained from the vertebral lesions, spleen, and liver. The cytological evaluation of the bone lytic lesions revealed a mixed inflammatory cellular population predominantly composed of macrophages, plasma cells, lymphocytes, and non-degenerated neutrophils. Intracytoplasmic structures compatible with parasitic forms of Leishmania spp. (amastigotes) were observed inside macrophages (Fig. 4). The same structures were observed in the spleen and liver, along with extramedular hematopoiesis. Based on the previous results, a proteinogram revealed monoclonal gammopathy. An Enzyme-Linked Immunosorbent Assay anti-body anti-leishmania serology resulted in a high seropositive result, 4.93 (R.R: 0–0.9).
Fig. 3. Laterolateral radiograph of the caudal thoracic and lumbar spine. Polyostotic radiolucent moth-eaten lesions were visible in the dorsal processes of L1, L2, and L3 (red arrows).
Fig. 4. Smear cytology of the vertebral lesion (hematoxylin and eosin staining). Note the presence of intracytoplasmic amastigotes (arrowheads) inside the macrophages. The dog was treated with a 4-week course of 100 mg/kg meglumine antimoniate (Antishmania, Fatro ibérica, Barcelona, Spain) subcutaneously once daily and 10 mg/kg alopurinol (Alopurinol Normon, Normon, Madrid, Spain) orally twice daily. In addition, prednisone (Prednisona Cinfa, Cinfa, Huarte, Spain) at 0.5 mg/kg twice daily orally was administered for 7 days to treat the possible vasogenic edema secondary to the inflammatory response produced by the intracranial lesion. No side effects were witnessed by the owner, and the dog tolerated the treatment well. Two and a half months after diagnosis, the dog was presented for a recheck. The physical and neurological examinations did not reveal any abnormalities. Additionally, the dog was reported to be clinically normal. A follow-up head CT scan with intravenous contrast was performed to evaluate the progression of the sphenoid bone lesions (Fig. 1). The previously described osteolytic and osteoproliferative lesions had healed, and the extra-axial mass lesion had resolved. Simultaneously, a follow-up proteinogram showed an improvement in the monoclonal gammopathy. During a telephone consultation 5 months after diagnosis, the owner reported that the dog was clinically normal and had an excellent quality of life. At that time, the dog was still receiving alopurinol, and the treatment was monitored by the referring veterinarian. Additional serological evaluations were declined due to financial constraints. DiscussionTo the best of the authors’ knowledge, this is the first case report that describes the CT findings and their resolution after 2 months of treatment in a dog with MCFS presumptively secondary to Leishmania spp. infection. Canine leishmaniasis has been reported to have the ability to infect multiple organs, including the nervous and musculoskeletal systems (Giannuzzi et al., 2017; Silva et al., 2022). Although nervous system involvement is considered rare in cases of canine leishmaniasis, the reported pathologies include vasculitis affecting the spinal cord and choroid plexi, myositis, intramedullary and extradural granulomas, granulomatous radiculoneuritis, meningomyelitis, meningoencephalitis, granulomatous meningitis, cerebral ischemic and hemorrhagic infarcts, ischemic myelopathy and compressive myelopathy (Pumarola et al., 1991; Vamvakidis et al., 2000; Viñuelas et al., 2001; Font et al., 2004; Ikeda et al., 2007; Paciello et al., 2009; Cauduro et al., 2011; José-López et al., 2012; José-López et al., 2014; Giannuzzi et al., 2017; Marquez et al., 2013; Farré Mariné et al., 2019). Regarding the musculoskeletal system, the ability of Leishmania spp. to produce myositis and osteolytic lesions, especially of joints, has been previously reported in the literature (Silva et al., 2021; Silva et al., 2022). Similar to the osseous changes seen in our case, the characteristic radiographic findings secondary to Leishmania spp. include a combination of periosteal reaction, intramedullary opacity alterations, and cortical or medullary structure destruction (Safrany et al., 2023). A previous case report published in 2023 described a dog presented with acute blindness presumptively secondary to bilateral optic neuritis and sphenoid bone osteomyelitis due to leishmaniosis diagnosed with serology. On neurological examination, the dog showed an absent menace response and bilaterally absent direct and indirect pupillary light reflexes. However, because the vestibulo-ocular reflexes were described as normal, a middle cranial fossa syndrome was not considered. The CT scan revealed similar findings to our case in the sphenoid bone, but a follow-up CT proving resolution of bony lesions was not performed despite the dog responding well to treatment. Furthermore, no further clinicopathological and serological monitoring was performed after treatment. In contrast with our case, the presence of amastigotes was not observed (Safrany et al., 2023). Neoplasia is the most frequent condition in dogs with neurological deficits causing MCFS (Jones et al., 2018). Reported tumours affecting the sphenoid bone include meningioma, lymphoma, thyroid adenocarcinoma, melanoma, neuroendocrine carcinoma, primitive neuroectodermic tumor, chondrosarcoma, nerve sheath tumor, and mast cell tumor (Beltran et al., 2010; Rissi, 2015; Jones et al., 2018). Due to the aggressive characteristics of the skull base bone lesions, neoplasia was also considered in this case at the time of diagnosis. Biopsies are regularly taken from the skull base using neurointerventional techniques in human medicine (Wilseck et al., 2021). However, access to the ventral area of the cranial vault for biopsy sampling is considered a high-risk procedure in veterinary medicine (Owen et al., 2018; Chen et al., 2023). Therefore, in this case, samples were not obtained from the sphenoid bone lesions. Nevertheless, considering that the sphenoid bone and lumbar vertebrae lesions shared similar characteristics, the already known ability of Leishmania spp. to produce multifocal invasion of different organs, and the young age of the patient, it was reasonable to assume that the intracranial lesion was also caused by Leishmania spp. A neoplastic process or bacterial osteomyelitis was also considered and could not be completely ruled out. However, the presence of amastigotes in the lumbar vertebrae and the high Leishmania spp. anti-body titer result, in addition to the good clinical response to the treatment and the complete resolution of radiological findings in the control CT made a Leishmania spp. basisphenoid osteomyelitis is the most likely diagnosis. To the best of the authors´ knowledge, there is only one case reported of a dog with appendicular lytic bone lesions diagnosed with leishmaniasis that evaluated the response of CT findings after treatment. In that case, the initial CT scan revealed osteolysis involving the cortex and medulla of the left femoral trochlea, condyles, lateral epicondyles, intercondylar fossa, and the medial fabella. A control CT scan was performed 8.5 months after diagnosis, where the previous bone loss remained unchanged in size despite the dog’s favorable clinical response (König et al., 2019). This is in contrast with our case, where sphenoid bone changes were resolved in the control CT scan. The authors hypothesize that this difference could be related to the chronicity of the lesions before treatment initiation because the previously mentioned case presented with a 3-month history of lameness, while our patient´s clinical signs appeared 2 weeks before diagnosis. Although our case presented neurological deficits associated with anatomical structures not located in the middle cranial fossa, previous reports have described dogs with MCFS and neurological deficits related to involvement of adjacent structures due to expansion of diagnosed lesions in the skull base (Rossmeisl et al., 2005; Jones et al., 2018). Our patient’s neurological examination revealed vision loss with absent menace response and bilateral non-responsive mydriatic pupils. This latter finding can be explained by lesions affecting both retinae, both optic nerves, the optic chiasm, and the rostral portion of both optic tracts. An ophthalmological examination with fundic evaluation was not performed, which could have helped to rule out chorioretinitis. However, involvement of the optic canal or optic chiasm secondary to the osteoproliferative and osteolytic lesions affecting the sphenoid bone seems to be the most likely cause of the blindness in this case. In conclusion, this case provides a new differential diagnosis in dogs presented with neurological deficits compatible with MCFS and reports a novel clinical presentation for leishmaniasis. AcknowledgmentsWe wish to thank IVC Evidensia, via the IVC Evidensia Research Fund, for providing funding for this study. Conflict of interestThe authors declare no conflicts of interest. FundingThe authors received financial support for publication of this article by IVC Evidensia. Authors’ contributionsS.S.B. managed the case, prepared the figures, and wrote the manuscript. A.F.M and A.L.F supervised the case and reviewed the manuscript. Data availabilityThe data supporting the findings of this study are available from the corresponding author upon request. ReferencesBaneth, G. and Solano-Gallego, L. 2022. Leishmaniasis. Vet. Clinics North Amer. Small Anim. Pract. 52, 1359–1375. Beltran, E., De Stefani, A., Stewart, J., De Risio, L. and Johnson, V. 2010. Disseminated mast cell tumor infiltrating the sphenoid bone and causing blindness in a dog. Vet. Ophthalmology 13, 184–189. Busse, C., Dennis, R. and Platt, S.R. 2009. Suspected sphenoid bone osteomyelitis causing visual impairment in two dogs and one cat. Vet. Ophthalmology 12, 71–77. Cauduro, A., Favole, P., Lorenzo, V., Simonetto, L., Barda, B., Cantile, C. and Asperio, R.M. 2011. Paraparesis caused by vertebral canal leishmaniotic granuloma in a dog. J. Vet. Internal. Med. 25, 398–399. Chen, S., Young, M.G., Bush, W.W., Shores, A. and Levine, C. 2023. Detailed anatomic description of the lateral, transzygomatic approach to the middle fossa and rostral brainstem and its use in three dogs. Vet. Surg. 52, 1180–1190. Farré Mariné, A., Borrego, J.F., Pumarola, M. and Luján Feliu-pascual, A. 2019. What Is Your Neurologic Diagnosis? Myelopathy attributable to Leishmania-induced occipito-atlanto-axial joint arthritis and capsular hypertrophy. J. Am. Vet. Med. Assoc. 255, 789–792. Font, A., Mascort, J., Altimira, J., Closa, J.M. and Vilafranca, M. 2004. Acute paraplegia associated with vasculitis in a dog with leishmaniasis. J. Small Anim. Pract. 45, 199–201. Giannuzzi, A.P., Ricciardi, M., De Simone, A. and Gernone, F. 2017. Neurological manifestations in dogs naturally infected by Leishmania infantum: descriptions of 10 cases and a review of the literature. J. Small Anim. Pract. 58, 125–138. Ikeda, F.A., Laurenti, M.D., Corbett, C.E., Feitosa, M.M., Machado, G.F. and Perri, S.H. 2007. Histological and immunohistochemical study of the central nervous system of dogs naturally infected by Leishmania (Leishmania) chagasi. Brazilizan. J. Vet. Res. Anim. Sci. 44, 5–11. Jones, A.M., Bentley, E. and Rylander, H. 2018. Cavernous sinus syndrome in dogs and cats: case series (2002–2015). Open Vet. J. 8, 86–192. José-López, R., De La Fuente, C., Pumarola, M. and Añor, S. 2014. Intramedullary spinal cord mass presumptively associated with leishmaniasis in a dog. J. Am. Vet. Med. Assoc. 244, 200–204. José-López, R., La Fuente, C.D. and Añor, S. 2012. Presumed brain infarctions in two dogs with systemic leishmaniasis. J. Small Anim. Pract. 53, 554–557. König, M.L., Howard, J., Schmidhalter, M., Hentrich, B. and Hettlich, B. 2019. Leishmaniosis manifesting as osteomyelitis and monoarthritis in a dog and outcome following treatment with miltefosine and allopurinol. Vet. Rec. Case. Rep. 7, 793. Márquez, M., Pedregosa, J.R., López, J., Marco-Salazar, P., Fondevila, D. and Pumarola, M. 2013. Leishmania amastigotes in the central nervous system of a naturally infected dog. J. Vet. Diagnostic. Invest. 25, 142–146. Owen, T.J., Martin, L.G. and Chen, A.V. 2018. Transsphenoidal Surgery for Pituitary Tumors and Other Sellar Masses. Vet. Clinics North Amer. Small Anim. Pract. 48, 129–151. Paciello, O., Oliva, G., Gradoni, L., Manna, L., Manzillo, V.F., Wojcik, S., Trapani, F. and Papparella, S. 2009. Canine inflammatory myopathy associated with Leishmania infantum infection. Neuromuscular Disord. 19, 124–130. Pumarola, M., Brevik, L., Badiola, J., Vargas, A., Domingo, M. and Ferrer, L. 1991. Canine leishmaniasis associated with systemic vasculitis in two dogs. J. Comparative. Pathol. 105, 279–286. Rissi, D.R. 2015. A retrospective study of skull base neoplasia in 42 dogs. J. Vet. Diagnostic Invest. 27, 743–748. Rossmeisl, J.H., Higgins, M.A., Inzana, K.D., Herring, I.P. and Grant, D.C. 2005. Bilateral cavernous sinus syndrome in dogs: 6 cases (1999–2004). J. Am. Vet. Med. Assoc. 226, 1105–1111. Safrany, B., Rushton, J., Barthez, P.Y. and Holmes, A. 2023. Blindness associated with presumed bilateral optic neuritis and sphenoid bone osteomyelitis in a dog with leishmaniosis. Vet. Rec. Case Rep. 11, 559. Silva, A.R.S., Gomes, A.A.D., Aquino, M.C.C., Almeida, B.F.M., Lima, V.M.F., Ciarlini, P.C., Pinoti, L.D.R., Marcondes, M. and Vieira, R.F.C. 2022. Synovial fluid and radiographic evaluation of joints from dogs with visceral leishmaniasis. Parasites & Vectors 15, 320. Silva, A.R.S., Oliveira, H.S., Gomes, A.A.D., Beserra, H.E.O., Silva, J.P., Santos-Doni, T.R., Tsunemi, M.H., Marcondes, M., Rahal, S.C. and Mamprim, M.J. 2021. Joint involvement in canine visceral leishmaniasis: orthopedic physical examination, radiographic and computed tomographic findings. Vet. Parasitology 299, 109569. Theisen, S.K., Podell, M., Schneider, T., Wilkie, D.A. and Fenner, W.R. 1996. A retrospective study of cavernous sinus syndrome in 4 dogs and 8 cats. J. Vet. Internal Med. 10, 65–71. Vamvakidis, C.D., Koutinas, A.F., Kanakoudis, G., Georgiadis, G. and Saridomichelakis, M. 2000. Masticatory and skeletal muscle myositis in canine leishmaniasis (Leishmania infantum). Vet. Rec. 146, 698–703. Viñuelas, J., Garcı́a-Alonso, M., Ferrando, L., Navarrete, I., Molano, I., Mirón, C., Carcelén, J., Alonso, C. and Nieto, C.G. 2001. Meningeal leishmaniosis induced by Leishmania infantum in naturally infected dogs. Vet. Parasitology. 101, 23–27. Wilseck, Z.M., Lin, L., Gemmete, J.J., Pandey, A.S., Srinivasan, A. and Chaudhary, N. 2021. Skull Base Neurointerventional Techniques. Neuroimaging Clinics North Amer. 3, 649–664. | ||
| How to Cite this Article |
| Pubmed Style Briones SS, Mariné AF, Feliu-pascual AL. Middle cranial fossa syndrome presumptively secondary to leishmaniasis in a dog: A case report. doi:10.5455/OVJ.2026.v16.i1.71 Web Style Briones SS, Mariné AF, Feliu-pascual AL. Middle cranial fossa syndrome presumptively secondary to leishmaniasis in a dog: A case report. https://www.openveterinaryjournal.com/?mno=293189 [Access: February 04, 2026]. doi:10.5455/OVJ.2026.v16.i1.71 AMA (American Medical Association) Style Briones SS, Mariné AF, Feliu-pascual AL. Middle cranial fossa syndrome presumptively secondary to leishmaniasis in a dog: A case report. doi:10.5455/OVJ.2026.v16.i1.71 Vancouver/ICMJE Style Briones SS, Mariné AF, Feliu-pascual AL. Middle cranial fossa syndrome presumptively secondary to leishmaniasis in a dog: A case report. doi:10.5455/OVJ.2026.v16.i1.71 Harvard Style Briones, S. S., Mariné, . A. F. & Feliu-pascual, . A. L. (2026) Middle cranial fossa syndrome presumptively secondary to leishmaniasis in a dog: A case report. doi:10.5455/OVJ.2026.v16.i1.71 Turabian Style Briones, Sergio Sánchez, Alba Farré Mariné, and Alejandro Luján Feliu-pascual. 2026. Middle cranial fossa syndrome presumptively secondary to leishmaniasis in a dog: A case report. doi:10.5455/OVJ.2026.v16.i1.71 Chicago Style Briones, Sergio Sánchez, Alba Farré Mariné, and Alejandro Luján Feliu-pascual. "Middle cranial fossa syndrome presumptively secondary to leishmaniasis in a dog: A case report." doi:10.5455/OVJ.2026.v16.i1.71 MLA (The Modern Language Association) Style Briones, Sergio Sánchez, Alba Farré Mariné, and Alejandro Luján Feliu-pascual. "Middle cranial fossa syndrome presumptively secondary to leishmaniasis in a dog: A case report." doi:10.5455/OVJ.2026.v16.i1.71 APA (American Psychological Association) Style Briones, S. S., Mariné, . A. F. & Feliu-pascual, . A. L. (2026) Middle cranial fossa syndrome presumptively secondary to leishmaniasis in a dog: A case report. doi:10.5455/OVJ.2026.v16.i1.71 |