| Research Article | ||
Open Vet. J.. 2026; 16(1): 201-213 Open Veterinary Journal, (2026), Vol. 16(1): 201-213 Research Article Impact of cinnamon oil supplementation on broiler performance, intestinal health, gene expression, antioxidant capacity, and hematological parametersHagar Elashry1, Mahmoud Kamal2, Rania Mahmoud1, Abeer Aziza1* and Tarek Ibrahim11Department of Nutrition and Clinical Nutrition, Faculty of Veterinary Medicine, Mansoura University, Mansoura, Egypt 2Animal Production Research Institute, Agricultural Research Center, Giza, Egypt *Corresponding Author: Abeer Aziza. Department of Nutrition and Clinical Nutrition, Faculty of Veterinary Medicine, Mansoura University, Mansoura, Egypt. Email: abeeraziza [at] gmail.comArticles published in Open Veterinary Journal are licensed under a Creative Commons Attribution-NonCommercial 4.0 International License Submitted: 08/11/2025 Revised: 15/12/2025 Accepted: 20/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
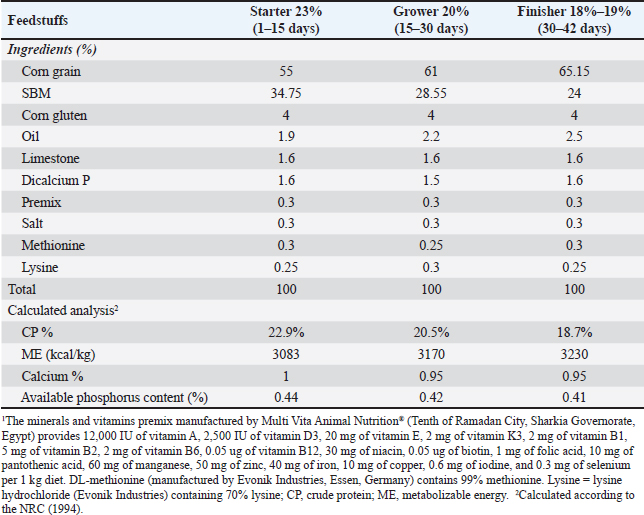
AbstractBackground: Cinnamon oil, used as a feed additive in poultry, enhances growth performance, improves antioxidant status, and augments immune response, resulting in superior meat quality and general productive efficiency. Aim: This study aimed to assess the effect of cinnamon oil (CO) in broiler diets on growth efficiency, hematological parameters, gene expression, and histological condition. Methods: Two hundred sixteen one-day-old broilers were assigned to three treatments in a fully randomized design, with each treatment subdivided into six replicates of 12 chicks. The first treatment is a control group, and the second and third are basic diets with 0.3% and 0.6% CO, respectively. Results: The 0.3% CO group had a higher final body weight, with no significant differences (p ≥ 0.05) in body weight and body weight gain. Birds in this group consumed less FI, leading to a notable improvement in feed conversion rate compared to the control. Additionally, 0.3% CO significantly (p ≤ 0.05) reduced total cholesterol levels without major impacts on most blood parameters, although mean corpuscular hemoglobin and mean corpuscular hemoglobin concentration increased in treated groups. Significant changes were observed in lipase and amylase levels, as well as higher catalase levels and lower malondialdehyde levels. Both 0.3% and 0.6% CO treatments (p ≤ 0.05) increased IGF-1, IL-10, and superoxide dismutase 1 levels, with the 0.6% concentration showing the highest levels. IL-1β levels remained low, indicating a specific effect of CO supplementation. Moreover, in the 0.3% CO group, CO supplementation enhanced intestinal absorption by increasing villus height and crypt depth. Conclusion: Adding 0.3% CO to the diet improves feed efficiency, lowers cholesterol, improves blood parameters, lowers inflammation, boosts antioxidant enzymes, and optimally improves intestinal structure, which is beneficial for poultry health and performance. Keywords: Cinnamon oil, Blood indicator, Broiler, Histology, Growth performance. IntroductionOver the past 50 years, the broiler industry has witnessed notable improvements in feed efficiency and a corresponding increase in broiler weight at the time of sale (Taleb et al., 2024; Ashour et al., 2025a). By 2050, the world’s population will need more animal protein. This situation implies that production methods must be more efficient and less environmentally harmful (Bist et al., 2024; Kamal et al., 2025). Poultry products, particularly those derived from broilers, constitute a significant source of animal protein (Abd El-Hack et al., 2024a,b; Mohamed et al., 2024). These birds are important in many fields because they are cost-effective, have a quick production cycle, and are very nutritious (Abo Egila et al., 2023; Abou-Kassem et al., 2024). Traditional poultry farming has problems with the environment and the economy because of higher production costs. To meet customer demand for healthier, organic products, broiler growth, health, and environmental sustainability must be improved (Alqahtani et al., 2024; El-Ratel et al., 2024; Kleyn and Ciacciariello, 2025). The incorporation of antibiotics in poultry feed has resulted in antibiotic-resistant bacteria, presenting considerable public health hazards and necessitating the pursuit of secure, natural substitutes (Kamal et al., 2023a; Yongjun and Lirong, 2024; Ashour et al., 2025b). Additionally, Hadad et al. (2024) discovered that incorporating Moringa oleifera leaves into rabbit diets improves health by modulating the immune system, enhancing the histological characteristics of the intestine, liver, and spleen, and optimizing physiological parameters due to antioxidative properties. Fathanah et al. (2024) indicated that adding probiotics to drinking water and 3% Lamtoro leaf meal as extra fiber positively enhanced Kampung chicken performance and small intestine absorption processes. Cinnamon, classified under “the genus Cinnamomum in the Lauraceae family,” comprises over 250–300 aromatic evergreen shrubs and trees. Nevertheless, only a limited number of these species possess notable commercial value as a globally used spice (Ali et al., 2021). Cinnamon contains a plethora of bioactive chemicals, including volatile oils, xanthones, alkaloids, coumarins, flavonoids, tannins, curcuminoids, terpenoids, and phenolics. These components enhance the inherent antioxidants, antibacterial, and anti-inflammatory properties of aqueous solutions (Liyanage et al., 2021). Cinnamon oil in broiler diets enhances BW, WG, and feed conversion rate (FCR) by boosting digestive enzymes and nutrient absorption without harming the carcass (Ali et al., 2021; Krauze et al., 2021). Furthermore, cinnamon oil has antimicrobial actions that change the gut microbiota by boosting beneficial bacteria, such as lactic acid bacteria, and lowering harmful bacteria. This process improves intestinal health and villi shape. Broiler-fed diets containing cinnamon oil supplements benefit from these effects, which enhance intestinal health and improve nutrient absorption (Saied et al., 2022). Furthermore, the addition of CO enhances the antioxidant capacity of broilers by increasing the activities of crucial antioxidant enzymes, “catalase (CAT), superoxide dismutase (SOD), and glutathione peroxidase,” while reducing indicators of lipid peroxidation, such as malondialdehyde (MDA) (Moustafa et al., 2020). This antioxidant function protects tissues from oxidative damage, which is important for maintaining the body’s balance under stress (Abd El-Hack et al., 2020). Cinnamon oil also alters the expression of genes associated with inflammatory responses. For example, it significantly lowers the levels of pro-inflammatory cytokines like TNF-α and NF-κB in broilers with infections, indicating that it has immunomodulatory and hepatoprotective effects (Tabatabaei et al., 2015). The incorporation of cinnamon oil into broiler meals enhances growth efficiency, bolsters antioxidant and immunological functions, and facilitates gene expression and tissue health, thereby improving overall production performance and meat quality. This study aimed to evaluate the impact of cinnamon oil on broiler growth efficiency, hematological parameters, gene expression, and histological conditions. Materials and MethodsBirds, diets, and experimental designTwo hundred sixteen one-day-old broiler chicks (n=216) were obtained from a single commercial hatchery to ensure genetic and health uniformity. All chicks were clinically healthy at the time of purchase. Individuals with similar initial body weight were randomly allocated into three experimental groups. This randomization protocol was rigorously applied throughout the study: for the initial allocation of birds to treatment groups, weekly selection of birds for growth performance monitoring, and final selection of birds for blood sampling, tissue collection, and slaughter. Each treatment comprised 6 repetitions, with each replicate containing 12 unsexed chicks. The experiment lasted for 6 weeks. The primary diet was developed according to the NRC (1994) criteria and was administered in pellet form during the study. The diet consisted of three phases: the first (1–15 days), second (15–30 days), and third (30–42 days), as shown in Table 1. The three dietary interventions were as follows: the control group received a basic diet, the second group received a basic diet supplemented with 0.3% CO, and the third group received a basic diet supplemented with 0.6% CO. The chicks were housed in standard cages (100 × 100 × 50 cm) with full access to food and fresh water. The study used a daily lighting schedule that included 23 hours of light followed by 1 hour of darkness. Birds that died during the study were necropsied for health monitoring, but their data were excluded from all final analyses. All reported results pertain only to healthy birds systematically sampled at the end of the trial. Table 1. Composition and calculation analysis of the basal diet.
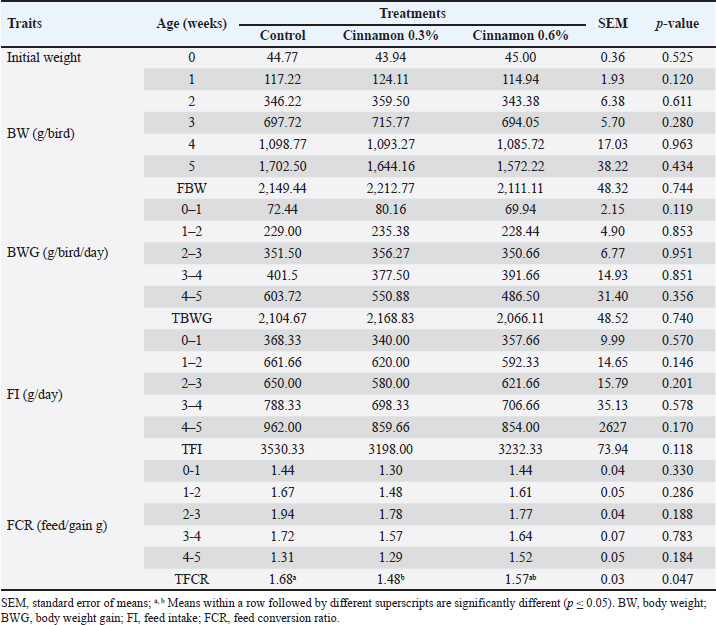
Growth performanceBird weights were weighed weekly. BWG was calculated weekly from 1 day of age to 42 days. The BWG was calculated by subtracting the final weight from the initial weight at the beginning of each period and weekly until the end of the experiment. FI was calculated by subtracting the remaining feed amount from the feed amount provided. The FCR was determined in grams of FI per unit of BWG. Blood parametersBlood specimens were taken from the wing veins of five broilers/treatment shortly before slaughter at the end of the trial. The blood was extracted into two separate tubes. White blood cell (WBC) counts were assessed in fresh blood samples using an NIHON KOHDEN automated hematology analyzer. The initial sample was obtained in a heparinized tube for hematological examination, including the evaluation of red blood cells, hemoglobin, and packed cell volume (PCV). “The mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), and mean corpuscular hemoglobin concentration (MCHC) were computed using the methods described by Harvey (2011).” A second specimen was obtained in a nonheparinized tube for serum separation and centrifuged at 3,000 rpm for 15 minutes. The serum was examined for total protein, albumin, total cholesterol, and creatinine, along with liver enzyme activity (ALT and AST levels), using the approach described by Young and Friedman (2001). Antioxidant and digestive enzymesAfter sacrifice, tissue specimens were taken from the liver and intestine of animals that were arbitrarily selected using phosphate-buffered saline (PBS) and then homogenized and centrifuged. The collected supernatants were used to estimate liver antioxidants (MDA and catalase) and intestinal digestive enzymes (lipase and amylase). A tissue sample (0.5 g) was rinsed in an ice-cold saline buffer (20 mM Tris-HCl, 0.14 M NaCl, pH 7.4) and subsequently homogenized in ice-cold PBS (pH 7.4). The homogenates were centrifuged at 3,000 rpm at 4°C for 15 minutes. The supernatants were meticulously collected and preserved at −20°C for the assessment of oxidative stress and digestive enzymes. Catalase, MDA as an oxidative biomarker, and lipase and amylase as indicators of digestion were evaluated using the enzymatic colorimetric method with commercial bio-diagnostic kits from Dokki, Giza, Egypt, in accordance with the manufacturer’s instructions (Fossati et al., 1980). Real-time polymerase chain reaction (RT-PCR)0.5 g of liver is taken to assess the expression of IGF-1 and SOD1. Another sample of 0.5 g of intestine was evaluated to evaluate some ILs, interleukin (IL-10), and IL-1β. The primers used are shown in Table 2. RNA was extracted according to the RNeasy Mini Kit instructions, and then RT-PCR results were analyzed. The RNA concentration and purity were checked using a Thermo Nanodrop 2,000 Spectrophotometer (Germany). Agilent MX3005p software was used to ascertain the amplification curves and Ct values. To assess the variance in gene expression of RNA across multiple samples, the CT values of each sample were compared with those of the control group using the “ΔΔCt” method as described by Yuan et al. (2006), applying the following ratio: (2–ΔΔCt). Table 2. Cinnamon primer sequence for RT-qPCR.
Histological statusAt the end of the trial, 5 birds were randomly chosen from each group for slaughter for histomorphological measurements. Tissue samples from the jejunum were fixed in 10% neutral buffered formalin for histopathological analysis. All fixed tissue specimens were embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin (Bancroft and Gamble, 2001). The jejunal slides were examined using an eyepiece (4×) in the Olympus BX41 microscope (Olympus, New York, NY, USA). Images were captured using a DVC 1300C color digital camera that was adjusted to the microscope. The intestinal villous height (VH) (μm) and crypt depth (CD) were measured according to the method of Bancroft and Gamble (2008). The VH/CD ratio was calculated. All morphometric parameters in jejunal sections with vertically oriented villi were determined by a pathologist using an image analysis (ImageJ=http://imagej.en.softonic.com). Statistical analysisData were examined statistically using one-way analysis of variance, employing the general linear model approach (SPSS, v.27). Sustainability tests for treatment differences were conducted using Duncan’s test. The following statistical model was employed to analyze variation and assess the influence of varying levels of CO: Yij=μ + Ti + eij, where Yij is the observation, μ, the observed mean, Ti is the effect of treatments, and eij is the experimental random error. Ethical approvalThis study was conducted on a poultry farm under the auspices of the Department of Nutrition and Clinical Nutrition, Faculty of Veterinary Medicine, Mansura University, Egypt. This study complied with the ethical standards of scientific research (No. MU-ACUC [VM.MS.23.10.92]) sanctioned by Mansura University, Egypt. ResultsGrowth efficiencyTable 3 shows how adding CO to broiler feed affects BW, BWG, FI, and FCR. The initial BW of the birds showed no significant changes (p=0.525), indicating the uniformity of the birds in the experimental treatments. The 0.3% CO group had a larger FBW than the other groups, but BW and BWG were similar in the trial; moreover, no substantial differences (p > 0.05) in BW and BWG were observed throughout the trial period. The results showed that the birds ate approximately the same quantity of feed, regardless of the treatment. On the other hand, the 0.3% CO group ate less feed than the other groups in the experiment. The difference led to significant changes (p < 0.05) in the FCR, with the 0.3% CO group having the best FCR and the 0.6% CO group having the second-best FCR compared to the control. Table 3. Effect of dietary cinnamon on broiler growth performance.
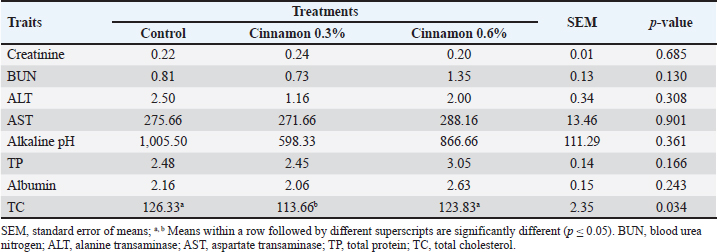
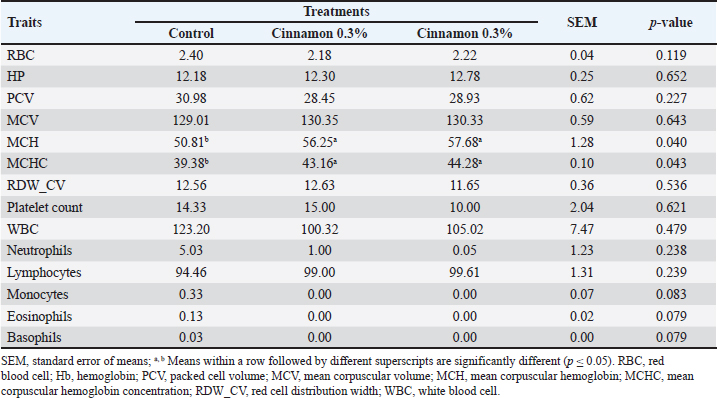
Blood indicatorsTable 4 shows the serum levels of creatinine, TP, cholesterol, ALT, AST, albumin, and alkaline pH. The total cholesterol levels were (p > 0.05) reduced in the 0.3% CO group compared with the other groups. Table 5 shows that adding CO to broiler feed did not affect most blood parameters, except that MCH and MCHC values were higher in the CO-treated groups than in the control group. Table 4. Impact of different cinnamon levels on blood constituents of broilers aged 6 weeks.
Table 5. Effect of different cinnamon levels on the hematology of 6-week-old broilers.
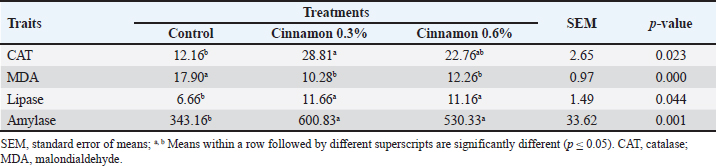
Antioxidants and digestive enzymesThe data in Table 6 indicate that cinnamon oil use mitigated CAT and MDA levels (p < 0.05). The birds given 0.3% CO exhibited the highest levels of CAT and the lowest MDA value. Significant changes (p < 0.05) in lipase and amylase levels were observed in the groups that received 0.3% and 0.6% CO compared with the other groups. Table 6. Effect of different levels of cinnamon on antioxidants and digestive enzymes in 6-week-old broilers.
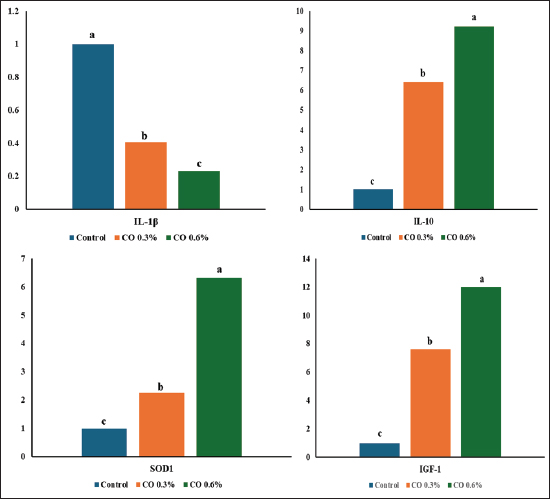
Real-time polymerase chain reactionFigure 1 illustrates that both 0.3% and 0.6% cinnamon oil treatments resulted in considerably increased gene expression levels of IGF-1, IL-10, and SOD1 compared with the control, with the 0.6% cinnamon oil group exhibiting the greatest expression for all evaluated genes. IGF-1 and IL-10 exhibited the most significant overexpression, particularly at 0.6% CO, but SOD1 expression was also substantially elevated compared with the control and increased with increasing CO dosage. On the other hand, IL-1β expression is consistently low in all treatments, which means that cinnamon oil does not have much of an effect. All genes exhibited normal levels in the control group, indicating that CO supplementation was the only way to observe the effect.
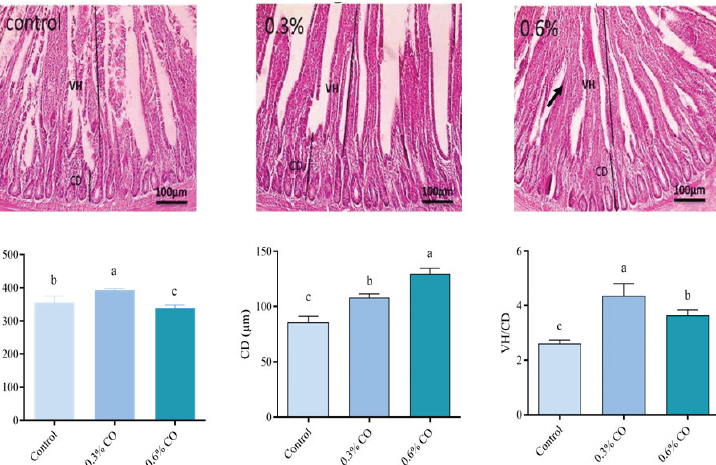
Fig. 1. Effect of adding cinnamon oil on broiler antioxidant and anti-inflammatory markers a, b, c Means within a row followed by different superscripts are significantly different (p ≤ 0.05). Histological traitsFigure 2 presents the effects of dietary supplementation with two additive amounts (0.3% and 0.6% CO) on intestinal morphology relative to the control. The villus height was markedly greater in the 0.3% CO group than in both the control and 0.6% CO groups. The control group exhibited intermediate values, whereas the 0.6% CO group displayed the lowest villus height among the treatments. The crypt depth was highest in the 0.6% CO group, moderate in the 0.3% CO group, and lowest in the control group.
Fig. 2. A microscopic examination of the intestine is illustrated. a, b, c Means within a row followed by different superscripts are significantly different (p ≤ 0.05). DiscussionEconomic dietary supplements provide feasible substitutes for poultry diets, potentially reducing feed formulation costs (Alyileili et al., 2020; Kamal et al., 2023b). The influence of these nutrients on chicken production may vary among different studies (Iqbal et al., 2022; El-Abbasy et al., 2025). Our results indicated that CO supplementation did not produce any significant changes in the BW of broiler chicks. Birds exposed to 0.3% CO demonstrated a negligible increase in both BW and BWG. Adding CO to their diets made them more efficient at eating because the birds that ate it had lower FI and a better FCR than the control. In this context, Daneshyar and Najafi (2016) and Fathi and El-Shahat (2017) showed that broiler hens fed diets with 3, 5, and 2 g/kg of cinnamon powder, respectively, exhibited markedly superior performance compared to alternative diets. Fathi et al. (2025) illustrated that the addition of cinnamon nanoparticles to feed reduced the population of pathogenic bacteria in the intestines of broiler chicks. Khanghahi et al. (2024) demonstrated that the inclusion of cinnamon and turmeric extract in oil-rich feeds with high peroxide values improves production efficiency and flock health. Yang et al. (2019) demonstrated that administering cinnamon extract to broiler chickens, particularly in terms of body weight, enhanced their growth. The increase in the BW of birds treated with cinnamon may be due to the antioxidant compounds found in these plants, such as eugenol, caryophyllene, cineole, and cinnamaldehyde (Saied et al., 2022), as well as curcumin (Naderi et al., 2014). Another study indicated that medicinal plants, such as cinnamon, turmeric, and thyme (both alone and in combination), affected the FCR of broilers (Mohamadamini et al., 2015). In contrast to the current outcomes, some studies have indicated that both a mixture of cinnamon and turmeric extract (Baghban et al., 2016) and a combination of turmeric and cinnamon powder (Khan and Ahmad, 2023) lower the FCR. Hematological indicators reflect the physiological condition of animals, illustrating their responses to diverse circumstances (Adebowale Adeyeye, 2020). They are often used to determine how environmental, dietary, and viral factors affect the health of animals (Kamal et al., 2023c). The findings demonstrated that the incorporation of CO into broiler feed markedly diminished TC levels while exerting negligible effects on most blood parameters, except for elevated MCH and MCHC values. These results align with studies on the effects of curcumin and turmeric supplementation in poultry nutrition (Aderemi and Alabi, 2023). Curcumin, the primary component of turmeric, has been extensively documented to exhibit hypocholesterolemic effects in broilers by modulating lipid metabolism (Yasmeen, 2024). Curcumin promotes the conversion of cholesterol into bile acids and inhibits enzymes, such as HMG-CoA reductase, involved in cholesterol synthesis. This results in reduced levels of TC, TG, and LDL cholesterol in broiler serum, without negatively impacting overall health indicators (Badran, 2020; Abdel-Moneim et al., 2025; Hernández-García et al., 2025). Curcumin administration typically preserves blood biochemical markers within normal physiological limits, indicating its safety and suitability as a dietary supplement (Ramadan et al., 2021). Slight increases in MCH and MCHC may indicate the antioxidant and anti-inflammatory properties of curcumin, potentially improving red blood cell function or hemoglobin concentrations (Abdelkader et al., 2024; Li et al., 2025). Improved nutrient absorption and oxidative stress regulation, which are known to affect erythrocyte integrity, may be linked to these effects. These studies indicated that curcumin does not have a significant negative effect on the number of WBC, hematocrit, or serum proteins. Curcumin not only lowers cholesterol but also protects broilers under stress in many ways. For example, it has antioxidant properties that reduce lipid peroxidation in muscles and hepatoprotective effects that improve liver enzyme profiles (Abd El-Hack et al., 2021). These bioactivities improve the health of birds, make feed more efficient, and improve meat quality. Changes in the properties of red blood cells, such as MCH and MCHC, may mean that they can carry more oxygen, which would be beneficial for the metabolic health of broilers (Salah et al., 2021). Thus, adding curcumin oil to broiler diets appears to be a beneficial way to improve lipid metabolism and maintain a healthy blood profile. It may also help with resistance to oxidative stress. The decrease in CAT and MDA levels in birds administered with CO, particularly at 0.3%, is consistent with the antioxidative properties of cinnamon. Catalase is an important enzyme that helps break down hydrogen peroxide, which protects cells from oxidative stress-induced damage. On the other hand, MDA is a sign of oxidative stress and lipid peroxidation (Krauze et al., 2021). The simultaneous increase in CAT activity and decrease in MDA levels indicate that cinnamon oil strengthens the avian antioxidant defense system and reduces cellular damage caused by oxidative stress (Wang et al., 2025). Studies on broiler chickens indicate that cinnamon oil supplementation significantly elevates CAT activity and reduces MDA levels, implying an enhanced oxidative state and diminished lipid peroxidation (Saied et al., 2022). This reaction makes chickens healthier overall and better able to handle oxidative stress. Significant changes in lipase and amylase levels in birds treated with 0.3% and 0.6% CO indicate an improvement in digestive function. Cinnamon oil enhances the secretion of digestive enzymes, thereby facilitating the digestion of lipids and carbohydrates, which improves nutrient absorption and avian performance (Ravardshiri et al., 2021). Studies have indicated that essential oils, including cinnamon, can augment the activities of pancreatic enzymes, such as lipase and amylase, consistent with the enzymatic changes observed in groups administered higher doses of cinnamon oil (Sotoudeh and Esmaeili, 2022; Wahyudi et al., 2023). This enzymatic enhancement improves feed efficiency and agrees with the bioactive compounds in cinnamon that promote gut health and digestive enzyme production. These results confirm that cinnamon oil has two benefits for poultry nutrition: it protects against oxidative damage and aids digestion. The observed enhancement in IGF-1, IL-10, and SOD1 gene expression with CO supplementation corresponds with the established bioactive characteristics of CO, notably its antioxidant and anti-inflammatory effects. The increase in IGF-1 aligns with cinnamon’s function in fostering growth and cellular vitality, as IGF-1 is essential for tissue development and repair (Jimoh et al., 2024). Cinnamon oil effectively enhances immunological modulation and reduces oxidative stress, as indicated by the substantial increase in IL-10, an anti-inflammatory cytokine, and the elevated expression of the antioxidant enzyme SOD1 (Davoudi and Ramazani, 2024; Jimoh et al., 2024). Previous research has shown that cinnamon and its constituents, such as cinnamaldehyde, inhibit pro-inflammatory cytokines (IL-1β, TNF-α) while enhancing IL-10 production, corroborating the minimal IL-1β response observed in the current study (Tabatabaei et al., 2015; Krauze et al., 2021). The immunomodulatory effects of cinnamon are linked to the alteration of inflammatory pathways, such as NF-κB and MAPK signaling, leading to diminished inflammation and improved antioxidant defense in diverse animal species, including chickens. The dose-dependent impact found, with 0.6% CO yielding the highest expression of beneficial genes, corresponds with existing literature indicating that elevated cinnamon oil dosages frequently amplify these molecular responses (Pagliari et al., 2023; Davoudi and Ramazani, 2024). This gene expression profile supports the hypothesis that CO acts as a natural modulator, improving antioxidant capacity, immunological balance, and possibly growth efficiency in poultry through growth factor and anti-inflammatory mediator production. These findings highlight the potential of cinnamon oil as a beneficial phytogenic feed additive for enhancing poultry health at appropriate dosages. The outcomes indicate that incorporating 0.3% CO into the diet significantly enhanced intestinal villus height and the VH/CD ratio, indicating increased intestinal health and possible nutrient absorption. Elevated supplementation (0.6% CO) augmented crypt depth, potentially signifying enhanced cell turnover; nevertheless, it was less successful in improving overall villus architecture and ratio compared with the 0.3% CO group. Increased VH typically correlates with improved nutrient absorption due to an expanded surface area, while a heightened VH/CD ratio indicates a more favorable balance between absorptive capacity and epithelial cell turnover, leading to improved digestive health and potentially enhanced growth efficiency (Solanki et al., 2022). The slight increase in CD, which is only 0.3%, could mean that cells are dividing more or that enterocytes are being replaced as part of normal gut maintenance or in response to dietary changes. Such changes could make the gut stronger and work better (Obianwuna et al., 2024). Our results are consistent with those of prior research, indicating that CO addition enhances gut morphology by elevating VH and preserving or marginally increasing CD, thereby promoting nutrient absorption and feed efficiency (Saied et al., 2022; Khan et al., 2025). A higher VH/CD ratio is often linked to a lower epithelial cell turnover rate. These findings can lower the amount of energy needed to keep the intestines healthy, which can then be used for growth and the immune system. The bioactive compounds of cinnamon oil, especially cinnamaldehyde, have antioxidant and anti-inflammatory properties that improve the gut barrier and affect the gut microbiota, which is advantageous for gut health (Kim et al., 2018; Pagliari et al., 2023). As a result, adding 0.6% cinnamon oil to the diet of chickens greatly improves the shape and function of their guts, thereby enhancing overall gastrointestinal health and nutrient absorption. ConclusionAdding 0.3% cinnamon oil to broiler diets makes feed more efficient by lowering FI and increasing FCR, while keeping BWG constant. It significantly lowers total cholesterol and has a beneficial effect on blood parameters, such as higher MCH and MCHC levels. Improving antioxidant status is linked to higher catalase activity, lower lipid peroxidation markers, and better changes in the digestive enzymes lipase and amylase. A gene expression study indicated that growth and anti-inflammatory markers (IGF-1, IL-10, and SOD1) increased with the dose, but proinflammatory IL-1β levels stayed low. This suggests that the treatment worked by changing the immune system in a specific manner. The morphology of the intestines improved, with taller villi and deeper crypts, especially at a dose of 0.3%. These changes make it easier for the body to absorb nutrients and stay healthy. Identifying the right dose that confers the most benefits to the structure of the intestines (as seen at 0.6%) without causing any problems or diminishing returns is still challenging. Future studies should examine the long-term effects, intestinal cellular turnover mechanisms, and comprehensive immunological responses in commercial settings. Using 0.3% cinnamon oil is a good way to make poultry healthier and better at what they do. Higher doses may be required depending on the production goals. AcknowledgmentsNone. Conflict of interestAll authors declare no conflicts of interest that could inappropriately influence this manuscript. FundingNone. Authors’ contributionsHE, RM, MK, AA, and TM drafted, revised, and edited the manuscript. All authors have read and approved the final version of the manuscript. Data availabilityThe data supporting the findings of this study are available upon reasonable request from the corresponding author. ReferencesAbd El-Hack, M.E., Alagawany, M., Abdel-Moneim, A.M.E., Mohammed, N.G., Khafaga, A.F., Bin-Jumah, M., Othman, S.I., Allam, A.A. and Elnesr, S.S. 2020. Cinnamon (Cinnamomum zeylanicum) oil as a potential alternative to antibiotics in poultry. Antibiotics 9, 210. Abd El-Hack, M.E., Alaidaroos, B.A., Farsi, R.M., Abou-Kassem, D.E., El-Saadony, M.T., Saad, A.M., Shafi, M.E., Albaqami, N.M., Taha, A.E. and Ashour, E.A. 2021. Impacts of supplementing broiler diets with biological curcumin, zinc nanoparticles and Bacillus licheniformis on growth, carcass traits, blood indices, meat quality and cecal microbial load. Animals 11(7), 1878. Abd El-Hack, M.E., Ashour, E.A., Aljahdali, N., Zabermawi, N.M., Baset, S.A., Kamal, M., Radhi, K.S., Moustafa, M., Algopishi, U., Alshaharni, M.O. and Bassiony, S.S. 2024a. Does the dietary supplementation of organic nano-zinc as a growth promoter impact broiler’s growth, carcass and meat quality traits, blood metabolites and cecal microbiota?. Poult. Sci. 103, 103550. Abd El-Hack, M.E., Ashour, E.A., Baset, S.A., Kamal M Swelum., Suliman, G.M. and Bassiony, S.S. 2024b. Effect of dietary supplementation of organic selenium nanoparticles on broiler chickens’ growth performance and carcass traits. Biol. Trace. Elem. Res. 202, 3760–3766. Abdelkader, A.M., El-Sheikh, A.M.H., El-Tahawy, W.S. and Hassan, S.S. 2024. Effect of curcumin and allicin on productive and physiological traits of mandra chickens. J. Agric. Environ. Sci. 23, 98–119. Abdel-Moneim, A.M.E., Mesalam, N.M., Yang, B. and Elsadek, M.F. 2025. Dietary incorporation of biological curcumin nanoparticles improved growth performance, ileal architecture, antioxidative status, serum lipid profile, and humoral immune response of heat-stressed broiler chickens. Poult. Sci. 104, 104740. Abo Egila, N.S.H., Dosoky, W.M., Khisheerah, N.S.M., Ahmed, M.H., Zahran, S.M., Almohmadi, N.H., Abusudah, W.F., Kamal, M., Moustafa, M., Tellez-Isaias, G., Al-Shehri, M. and Abd El-hack, M.E. 2023. Does dietary linseed or canola oil affect lipid metabolism, immunity, and n-3 polyunsaturated fatty acids content in quail eggs?. Poult. Sci. 102, 103116. Abou-Kassem, D.E., El-Sayiad, G.A., El-Samahy, R.A., Abd El-hack, M.E., Taha, A.E., Kamal, M., Alfassam, H.E., Rudayni, H.A., Allam, A.A., Moustafa, M., Algopishi, U. and Ashour, E.A. 2024. Impacts of storage period and egg weight on hatching and growth performance of growing Japanese quails. Poult. Sci. 103, 103772. Adebowale Adeyeye, S. 2020. Wild sunflower and goat weed leaf meals composite-mix supplementation in broiler chickens: effects on performance, health status and meat. Acta Fytotech. Zootech. 23(4), 205–212. Aderemi, F.A. and Alabi, O.M. 2023. Turmeric (Curcuma longa): an alternative to antibiotics in poultry nutrition. Translat. Anim. Sci. 7, 133. Akbarian, A., Michiels, J., Golian, A., Buyse, J., Wang, Y. and De Smet, S. 2014. Gene expression of heat shock protein 70 and antioxidant enzymes, oxidative status, and meat oxidative stability of cyclically heat-challenged finishing broilers fed Origanum compactum and Curcuma xanthorrhiza essential oils. Poult. Sci. 93, 1930–1941. Ali, A., Ponnampalam, E.N., Pushpakumara, G., Cottrell, J.J., Suleria, H.A.R. and Dunshea, F.R. 2021. Cinnamon: a natural feed additive for poultry health and production—A review. Animals 11, 2026. Alqahtani, F.S., Bahshwan, S., AL-Qurashi, M.M., Allohibi, A., Beyari, E.A., Alghamdi, M.A. and Mostafa, N.G. 2024. Impact of dietary Bacillus toyonensis M44 as an antibiotic alternative on growth, blood biochemical properties, immunity, gut microbiota, and meat quality of IR broilers. Pak. Vet. J. 44, 637–646. Alyileili, S.R., Belal, I.E.H., Hussein, A.S. and El-Tarabily, K.A. 2020. Effect of inclusion of degraded and non-degraded date pits in broilers’ diet on their intestinal microbiota and growth performance. Animals 10, 2041. Amills, M., Jimenez, N., Villalba, D., Tor, M., Molina, E., Cubilo, D., Marcos, C., Francesch, A., Sanchez, A. and Estany, J. 2003. Identification of three single-nucleotide polymorphisms in the chicken insulin-like growth factor 1 and 2 genes and their associations with growth and feeding traits. Poult. Sci. 82, 1485–1493. Ashour, E.A., Aldhalmi, A.K., Ismail, I.E., Kamal, M., Elolimy, A.A., Swelum, A.A. and Abd El-hack, M.E. 2025a. The effect of using Echinacea extract as an immune system stimulant and antioxidant on blood indicators, growth efficiency, and carcass characteristics in broiler chickens to produce a healthy product. Poult. Sci. 104, 104392. Ashour, E.A., Aldhalmi, A.K., Kamal, M., Salem, S.S., Mahgoub, S.A., Alqhtani, A.H., Madkour, M., Elolimy, A.A., Abd El-hack, M.E. and Swelum, A.A. 2025b. The efficacy of Artichoke leaf extract conjugated with organic zinc nanoparticles on growth, carcass traits and blood biochemical parameters of broilers. Poult. Sci. 104(1), 104521. Badran, A. 2020. Effect of dietary curcumin and curcumin nanoparticles supplementation on growth performance, immune response and antioxidant of broilers chickens. Egypt. Poult. Sci. J. 40, 325–343. Baghban, K.P., Daneshyar, M. and Najafi, R. 2016. Effects of cinnamon and turmeric powders supplementation on performance, carcass characteristics and some serum parameters of broiler chickens under heat stress conditions. Anim. Sci. Res. 26, 63–75. Bancroft, J.D. and Gamble, M. 2001. Theory and practice of histological techniques. 5th ed. New York (London, San Francisco, Tokyo): churchill Livingston. 5th ed., New York (London, San Francisco, Tokyo): Churchill Livingstone, New York, NY, 63, pp: 63–82. Bancroft, J.D. and Gamble, M. 2008. Theory and practice of histological techniques. Elsevier Health Sci. Bist, R.B., Bist, K., Poudel, S., Subedi, D., Yang, X., Paneru, B. and Chai, L. 2024. Sustainable poultry farming practices: A critical review of current strategies and future prospects. Poult. Sci. 104295. Daneshyar, M. and Najafi, R. 2016. Effects of Cinnamon (Cinnamomum Zeylanicum) and Turmeric (Curcuma Longa) Powder on Performance, Enzyme Activity, and Blood Parameters of Broiler Chickens Under Heat Stress. Poult. Sci. J. 4, 47–53. Davoudi, F. and Ramazani, E. 2024. Antioxidant and anti-inflammatory effects of Cinnamomum species and their bioactive compounds: an updated review of the molecular mechanisms. Physiol. Pharmacol. 28, 99–116. El-Abbasy, M.M., Aldhalmi, A.K., Ashour, E.A., Bassiony, S.S., Kamal, M., Alqhtani, A.H., Abou-Kassem, D.E., Elolimy, A.A., Abd El-hack, M.E. and Swelum, A.A. 2025. Enhancing broiler growth and carcass quality: impact of diets enriched with Moringa oleifera leaf powder conjugated with zinc nanoparticles. Poult. Sci. 104(1), 104519. El-Ratel, I.T., Amara, M.M., Beshara, M.M., El Basuini, M.F., Fouda, S.F., El-Kholy, K.H. and Mekawy, A. 2024. Effects of supplemental vitamin A on reproduction and antioxidative status of aged laying hens, and their offspring’s growth, blood indices and immunity. Poult. Sci. 103, 103453. Fathanah, S.Y.E., Rahardja, D.P. and Purwanti, S. 2024. Impacts of probiotic and dietary Lamtoro leaf meal on the growth performance, digestibility and small intestinal morphometry of Kampung chicken. Int. J. Agric. Biosci. 13, 333–339. Fathi, M. and El-Shahat, K.H. 2017. L-carnitine enhances oocyte maturation and improves in vitro development of embryos in dromedary camels (Camelus dromedaries). Theriogenology 104, 18–22. Fathi, M., Mohammad, A., Zarrinkavani, K. and Biranvand, Z. 2025. The effect of cinnamon nanoparticles produced with ultrasonic waves on growth performance, digestive enzyme activity, thyroid hormone levels, and ileal bacterial population in broiler chickens. Iranian. J. Anim. Sci. Res. 17, 193–203. Fossati, P., Prencipe, L. and Berti, G. 1980. Use of 3, 5-dichloro-2-hydroxybenzenesulfonic acid/4-aminophenazone chromogenic system in direct enzymic assay of uric acid in serum and urine. Clin. Chem. 26, 227–231. Hadad, S.S., Aziz, E.K., Saad, A.H., El-Gendy, H., Abdel-Megeid, N., Masoud, S.R., Shukry, M., Swelum, A.A., Al-Ghadi, M.Q., Alhimaidi, A.R., Tellez-Isaias, G., Abd El-hack, M.E. and Abumandour, M.M.A. 2024. Impact of dietary Moringa oleifera leaf supplementation on gut morphometry, behaviour, and physiological parameters in growing male rabbits. J. Anim. Physiol. Anim. Nutr. 108, 1214–1230. Harvey, J.W. 2011. Veterinary hematology: a diagnostic guide and color atlas. Elsevier Health Sci. 31. Hernández-García, P.A., Granados-Rivera, L.D., Orzuna-Orzuna, J.F., Vázquez-Silva, G., Díaz-Galván, C. and Razo-Ortíz, P.B. 2025. Meta-Analysis of dietary curcumin supplementation in broiler chickens: growth performance, antioxidant status, intestinal morphology, and meat quality. Antioxidants 14, 460. Iqbal, J., Sharif, M., Suleman, M.N., Saeed, M., Ahamd, F., Kamboh, A.A., Ayaşan, T. and Arslan, M. 2022. Effect of dietary supplementation of a non-antibiotic growth promoter on growth performance and intestinal histomorphology in broilers. Pak. J. Zool. 54, 1629–1636. Jimoh, O.A., Ayodele, A.D., Ojo, O.A., Okin-Aminu, H.O. and Olarotimi, O.J. 2024. Effects of turmeric, ginger, cinnamon, and garlic essential oils on HSP70, NFκB, oxidative DNA damage, inflammatory cytokines, and oxidative markers in broiler chickens. Translat. Anim. Sci. 8, txae127. Kamal, M., Aljahdali, N., Jaber, F.A., Majrashi, K.A., Kishk, W.H., Khalil, H.A. and Abd El-hack, M.E. 2023c. Influence of dietary chitosan supplementation on ovarian development and reproductive performance of New Zealand White rabbit does. Ann. Anim. Sci. 23, 757–764. Kamal, M., Kishk, W.H., Khalil, H.A., Abdel-Khalek, A.M., Ayoub, M.A., Swelum, A.A., Alqhtani, A.H., Ba-Awadh, H.A. and Abd El-hack, M.E. 2023b. Effect of dietary chitosan supplementation on productive and physiological performance parameters of growing New Zealand white rabbits. Int. J. Biol. Macromol. 230, 123166. Kamal, M., Sindi, R.A., El-Azzazi, F.E., Kishk, W.H., Khalil, H.A., Abdel-Khalek, A.M., Ayoub, M.A., Tufarelli, V. and Abd El-hack, M.E. 2023a. Sexual behaviour response, testicular development and semen quality of New Zealand white rabbit bucks as influenced by dietary chitosan. Reproducr. Dom. Anim. 58, 405–413. Kamal, M., Zhu, L., Abd El-hack, M.E., Arif, M., Li, F. and Cheng, Y. 2025. Functional Roles of Mannan and Chitosan Oligosaccharides on Animal Health and Nutrition: a Review. Carbohydr. Polym. Tech. 10, 100764. Khan, K. and Ahmad, N. 2023. Using cinnamon (Cinnamomum zeylanicum) and turmeric (Curcuma longa L) powders as an antibiotic growth promoter substitutions in broiler chicken’s diets. Anim. Biotechnol. 34, 4466–4473. Khan, K., Al-Khalaifah, H., Ahmad, M.T., Khan, M.T., Alonaizan, R., Khan, and R.U. Alhidary, I.A. 2025. Dietary supplementation of cinnamon and turmeric powder enhances growth performance, nutrient digestibility, immune response, and renal function in broiler chickens. Poult. Sci. 105556. Khanghahi, M., Jafari, M., Khorshidi, K. and Gharahveysi, S. 2024. Effect of Feed Containing Cinnamon and Turmeric Extract with Peroxide Oils on the Growth Performance, Antibody Titers, Cecal Microbial Population, and Jejunum Morphology of Broiler Chickens. Braz. J. Poult. Sci. 26, 2024. Kim, J.I., Lee, J.H., Song, Y., Kim, Y.T., Lee, Y.H. and Kang, H. 2018. Oral consumption of cinnamon enhances the expression of immunity and lipid absorption genes in the small intestinal epithelium and alters the gut microbiota in normal mice. J. Func. Foods. 49, 96–104. Kleyn, R. and Ciacciariello, M. 2025. Putting sustainable poultry production into perspective. W. Poult. Sci. J. 81(2), 343–357. Krauze, M., Cendrowska-Pinkosz, M., Matuseviĉius, P., Stępniowska, A., Jurczak, P. and Ognik, K. 2021. The effect of administration of a phytobiotic containing cinnamon oil and citric acid on the metabolism, immunity, and growth performance of broiler chickens. Animals 11, 399. Li, Z., Qumar, M., Irfan, M., Khan, M.T., Faran, G., Abbas, G., Prince, K., Dilbar, G.A., Gondal, M.A., Rauf, M., Ahmed, I., Ilyas, M., Hussain, A., Yuan, H. and Fouad, D. 2025. Impact of dietary turmeric and ginger powder on performance, serum chemistry and gut microbiota in laying hens. J. Appl. Poult. Res. 34(4), 100605. Liyanage, N.M.N., Bandusekara, B.S., Kanchanamala, R.W.M.K., Hathurusinghe, H.A.B.M., Rathnayaka, A.M.R.W.S.D., Pushpakumara, D.K.N.G., Samita, S., Wijesinghe, K.G.G., Jayasinghe, G.G., Liyanage, W.K. and Bandaranayake, P.C.G. 2021. Identification of superior Cinnamomum zeylanicum Blume germplasm for future true cinnamon breeding in the world. J. Food Compos. Anal. 96, 103747. Mohamadamini, M., Shariatmadari, F. and Hosseini, S.A. 2015. The effects of turmeric, thyme and cinnamon on parameters related to ascites syndrome in Arian broilers. Iranian J. Med. Arom. Plants 31, e436–e444. Mohamed, L.A., Dosoky, W.M., Kamal, M., Alshehry, G., Algarni, E.H., Aldekhail, N.M., Mohamed, H.S., Abd El-hack, M.E. and Farag, S.A. 2024. Growth performance, carcass traits, and meat physical characteristics of growing Japanese quail fed ginger powder and frankincense oil as feed additives. Poult. Sci. 103, 103771. Moustafa, N., Aziza, A., Orma, O. and Ibrahim, T. 2020. Effect of supplementation of broiler diets with essential oils on growth performance, antioxidant status, and general health. Mansoura Vet. Med. J. 21, 14–20. Naderi, M., Akbari, M.R., Asadi-Khoshoei, E., Khaksar, K. and Khajali, F. 2014. Effects of dietary inclusion of turmeric (Curcuma longa) and cinnamon (Cinnamomum verum) powders on performance, organs relative weight and some immune system parameters in broiler chickens. J. Poult. Sci. 2, 153–163. NRC. 1994. National Research Council. Nutrient Requirements of Poultry, 9th ed.; National Academy Press: Washington, DC, USA. Obianwuna, U.E., Chang, X., Oleforuh-Okoleh, V.U., Onu, P.N., Zhang, H., Qiu, K. and Wu, S. 2024. Phytobiotics in poultry: revolutionizing broiler chicken nutrition with plant-derived gut health enhancers. J. Anim. Sci. Biotechnol. 15, 169. Pagliari, S., Forcella, M., Lonati, E., Sacco, G., Romaniello, F., Rovellini, P., Fusi, P., Palestini, P., Campone, L., Labra, M., Bulbarelli, A. and Bruni, I. 2023. Antioxidant and anti-inflammatory effect of cinnamon (Cinnamomum verum J. Presl) bark extract after in vitro digestion simulation. Foods 12, 452. Ramadan, O., Hagag, S.E.S., El-Gharib, M.S. and Zahran, F. 2021. The protective role of turmeric (Curcuma longa) on broilers exposed to chronic heat stress. Alfarama J. Basic Appl. Sci. 2, 194–204. Ravardshiri, M., Bahram, S., Javadian, S.R. and Bahrekazemi, M. 2021. Cinnamon promotes growth performance, digestive enzyme, blood parameters, and antioxidant activity of rainbow trout (Oncorhynchus mykiss) in low-carbohydrate diets. Turkish J. Fish. Aquatic Sci. 21, 309–322. Saied, A.M., Attia, A.I., El-Kholy, M.S., Reda, F.M. and El Nagar, A.G. 2022. Effect of cinnamon oil supplementation into broiler chicken diets on growth, carcass traits, haemato-biochemical parameters, immune function, antioxidant status and caecal microbial count. J. Anim. Feed. Sci. 31, 21–33. Salah, A.S., Ahmed-Farid, O.A., Nassan, M.A. and El-Tarabany, M.S. 2021. Dietary curcumin improves energy metabolism, brain monoamines, carcass traits, muscle oxidative stability and fatty acid profile in heat-stressed broiler chickens. Antioxidants 1265, 1265. Samy, A.A., El-Enbaawy, M.I., El-Sanousi, A.A., Abd El-wanes, S.A., Ammar, A.M., Hikono, H. and Saito, T. 2015. In-vitro assessment of differential cytokine gene expression in response to infections with Egyptian classic and variant strains of highly pathogenic H5N1 avian influenza virus. Int. J. Vet. Sci. Med. 3, N1–N8. Solanki, D.M., Dave, C.J., Bhanderi, B.B., Sheth, A.L. and Ghodasara, D.J. 2022. Effect of cinnamon (Cinnamomum zeylanicum) essential oil as an alternative to antibiotic growth promoter in broilers. Indian. J. Vet. Sci. Biotechnol. 18, 73–80. Sotoudeh, E. and Esmaeili, N. 2022. Effects of Biotronic® Top3, a feed additive containing organic acids, cinnamaldehyde and a permeabilizing complex on growth, digestive enzyme activities, immunity, antioxidant system and gene expression of barramundi (Lates calcarifer). Aquac. Rep. 24, 101152. Suzuki, K., Okada, H., Itoh, T., Tada, T., Mase, M., Nakamura, K. and Tsukamoto, K. 2009. Association of increased pathogenicity of Asian H5N1 highly pathogenic avian influenza viruses in chickens with highly efficient viral replication accompanied by early destruction of innate immune responses. J. Virol. 83, 7475–7486. Tabatabaei, S.M., Badalzadeh, R., Mohammadnezhad, G.R. and Balaei, R. 2015. Effects of Cinnamon extract on biochemical enzymes, TNF-α and NF-κB gene expression levels in liver of broiler chickens inoculated with Escherichia coli. Pesq. Vet. Bras. 35, 781–787. Taleb, H.M., Abdel-Halim, A.A., Ramadan, G.S., Shazly, S.A., Kamal, M., Alwaili, M.A., Rudayni, H.A., Allam, A.A., Taha, A.E., Al-Sayed, H.M.A., Moustafa, M., Alshaharni, M.O., Mahrose, K. and Fouad, A.M. 2024. Does the marketing age impact growth performance, carcass traits, economic feasibility and hemato-biochemical properties of genetically-modified quails?. Poult. Sci. 103, 103793. Wahyudi, I.T., Jusadi, D., Setiawati, M. and Ekasari, J. 2023. Effects of dietary supplementation with cinnamon powder and lysine on blood chemistry, liver histology, growth performance, and fillet quality of striped catfish Pangasianodon hypophthalmus. Aquac. Int. 31, 3513–3529. Wang, D., Sayed, M.A.M., Galal, A.E., Attaai, A.H., Makled, M.N., Ali, A.H.H., Wei, C., Habib, M.A., Abdelfattah, M.G. and Abouelezz, K. 2025. The antioxidative properties of thyme, cinnamon, and pomegranate oils in heat-stressed broilers. Poultry Sci. 104(7), 105228. Yang Yun Feng, Y.Y., Zhao Lu Lu, Z.L., Shao Yu Xin, S.Y., Liao Xiu Dong, L.X., Zhang LiYang, Z.L., Lu Lin, L.L. Luo Xu Gang, L.X. 2019. Effects of dietary graded levels of cinnamon essential oil and its combination with bamboo leaf flavonoid on immune function, antioxidative ability and intestinal microbiota of broilers. J. Integr. Agric. 18, 2123–2132 Yasmeen. and R. 2024. Effect of Curcuma Longa on Growth and Lipid Profile of Broiler Chickens across Seasons. J. Bioresource Manage. 11, 21. Yongjun, D.O.N.G. and Lirong, W.A.N.G. 2024. Effect of Dietary Inclusion of Alfalfa Polysaccharide on Performance and Immune System Efficacy of Broiler Chickens. Kafkas. Üniversitesi. Veteriner. Fakültesi. Dergisi. 30(3), 335–340. Young, D.S. and Friedman, R.B. 2001. Effects of disease on clinical laboratory tests. 4th ed. AACC Press. 4th ed., Washington, D.C.: AACC Press. Yuan, J.M., Guo, Y.M., Yang, Y. and Wang, Z. 2007. Characterization of fatty acid digestion of Beijing fatty and arbor acres chickens. Asian-Austral. J. Anim. Sci. 20, 1222–1228. Yuan, J.S., Reed, A., Chen, F. and Stewart, C.N. 2006. Statistical analysis of real-time PCR data. BMC. Bioinf. 7, 85. | ||
| How to Cite this Article |
| Pubmed Style Elashry H, Kamal M, Mahmoud R, Aziza A, Ibrahim T. Impact of cinnamon oil supplementation on broiler performance, intestinal health, gene expression, antioxidant capacity, and hematological parameters. Open Vet. J.. 2026; 16(1): 201-213. doi:10.5455/OVJ.2026.v16.i1.19 Web Style Elashry H, Kamal M, Mahmoud R, Aziza A, Ibrahim T. Impact of cinnamon oil supplementation on broiler performance, intestinal health, gene expression, antioxidant capacity, and hematological parameters. https://www.openveterinaryjournal.com/?mno=295333 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.19 AMA (American Medical Association) Style Elashry H, Kamal M, Mahmoud R, Aziza A, Ibrahim T. Impact of cinnamon oil supplementation on broiler performance, intestinal health, gene expression, antioxidant capacity, and hematological parameters. Open Vet. J.. 2026; 16(1): 201-213. doi:10.5455/OVJ.2026.v16.i1.19 Vancouver/ICMJE Style Elashry H, Kamal M, Mahmoud R, Aziza A, Ibrahim T. Impact of cinnamon oil supplementation on broiler performance, intestinal health, gene expression, antioxidant capacity, and hematological parameters. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 201-213. doi:10.5455/OVJ.2026.v16.i1.19 Harvard Style Elashry, H., Kamal, . M., Mahmoud, . R., Aziza, . A. & Ibrahim, . T. (2026) Impact of cinnamon oil supplementation on broiler performance, intestinal health, gene expression, antioxidant capacity, and hematological parameters. Open Vet. J., 16 (1), 201-213. doi:10.5455/OVJ.2026.v16.i1.19 Turabian Style Elashry, Hagar, Mahmoud Kamal, Rania Mahmoud, Abeer Aziza, and Tarek Ibrahim. 2026. Impact of cinnamon oil supplementation on broiler performance, intestinal health, gene expression, antioxidant capacity, and hematological parameters. Open Veterinary Journal, 16 (1), 201-213. doi:10.5455/OVJ.2026.v16.i1.19 Chicago Style Elashry, Hagar, Mahmoud Kamal, Rania Mahmoud, Abeer Aziza, and Tarek Ibrahim. "Impact of cinnamon oil supplementation on broiler performance, intestinal health, gene expression, antioxidant capacity, and hematological parameters." Open Veterinary Journal 16 (2026), 201-213. doi:10.5455/OVJ.2026.v16.i1.19 MLA (The Modern Language Association) Style Elashry, Hagar, Mahmoud Kamal, Rania Mahmoud, Abeer Aziza, and Tarek Ibrahim. "Impact of cinnamon oil supplementation on broiler performance, intestinal health, gene expression, antioxidant capacity, and hematological parameters." Open Veterinary Journal 16.1 (2026), 201-213. Print. doi:10.5455/OVJ.2026.v16.i1.19 APA (American Psychological Association) Style Elashry, H., Kamal, . M., Mahmoud, . R., Aziza, . A. & Ibrahim, . T. (2026) Impact of cinnamon oil supplementation on broiler performance, intestinal health, gene expression, antioxidant capacity, and hematological parameters. Open Veterinary Journal, 16 (1), 201-213. doi:10.5455/OVJ.2026.v16.i1.19 |